MIC19 Exerts Neuroprotective Role via Maintaining the Mitochondrial Structure in a Rat Model of Intracerebral Hemorrhage
Abstract
1. Introduction
2. Results
2.1. General Observations
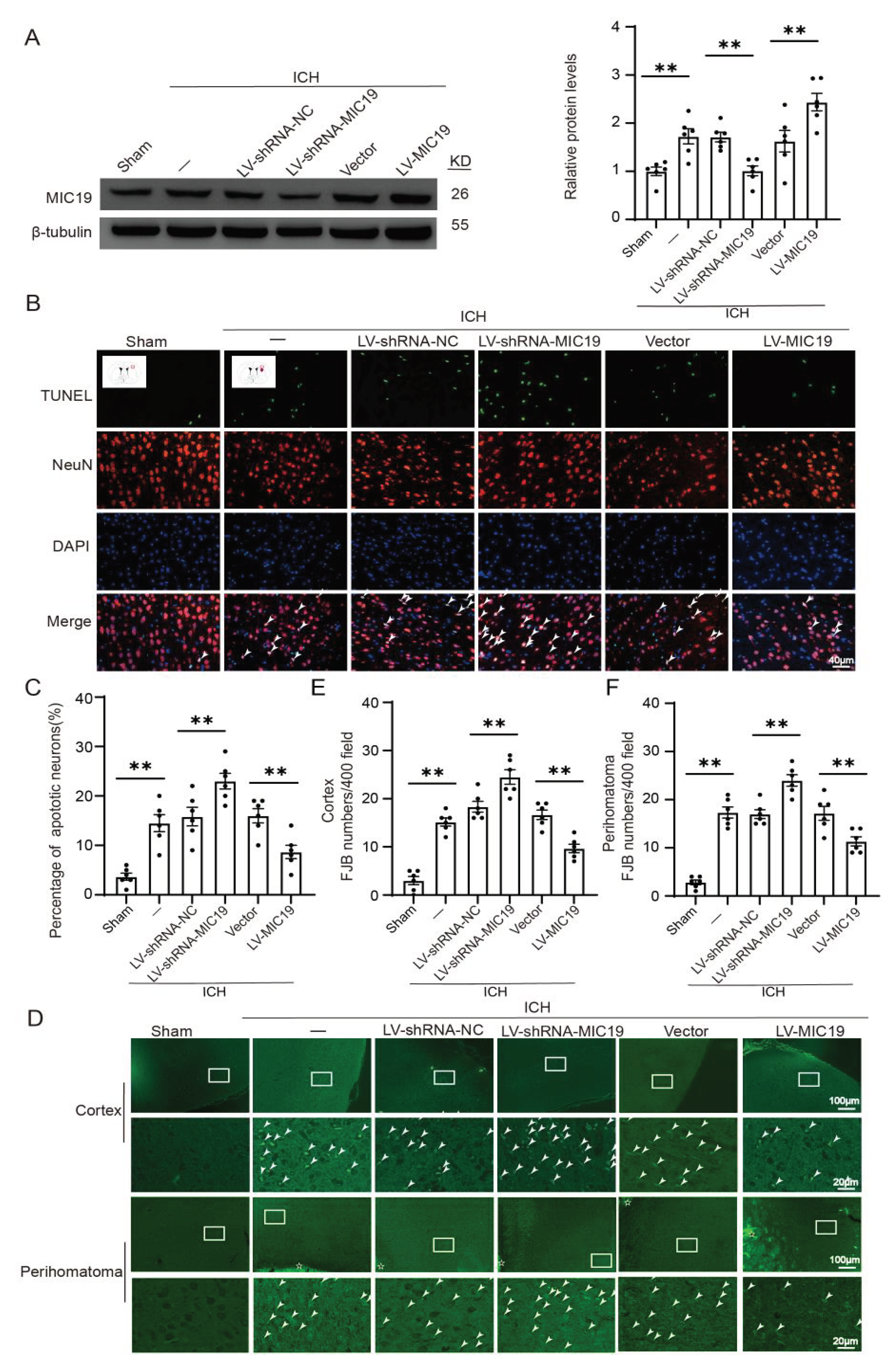
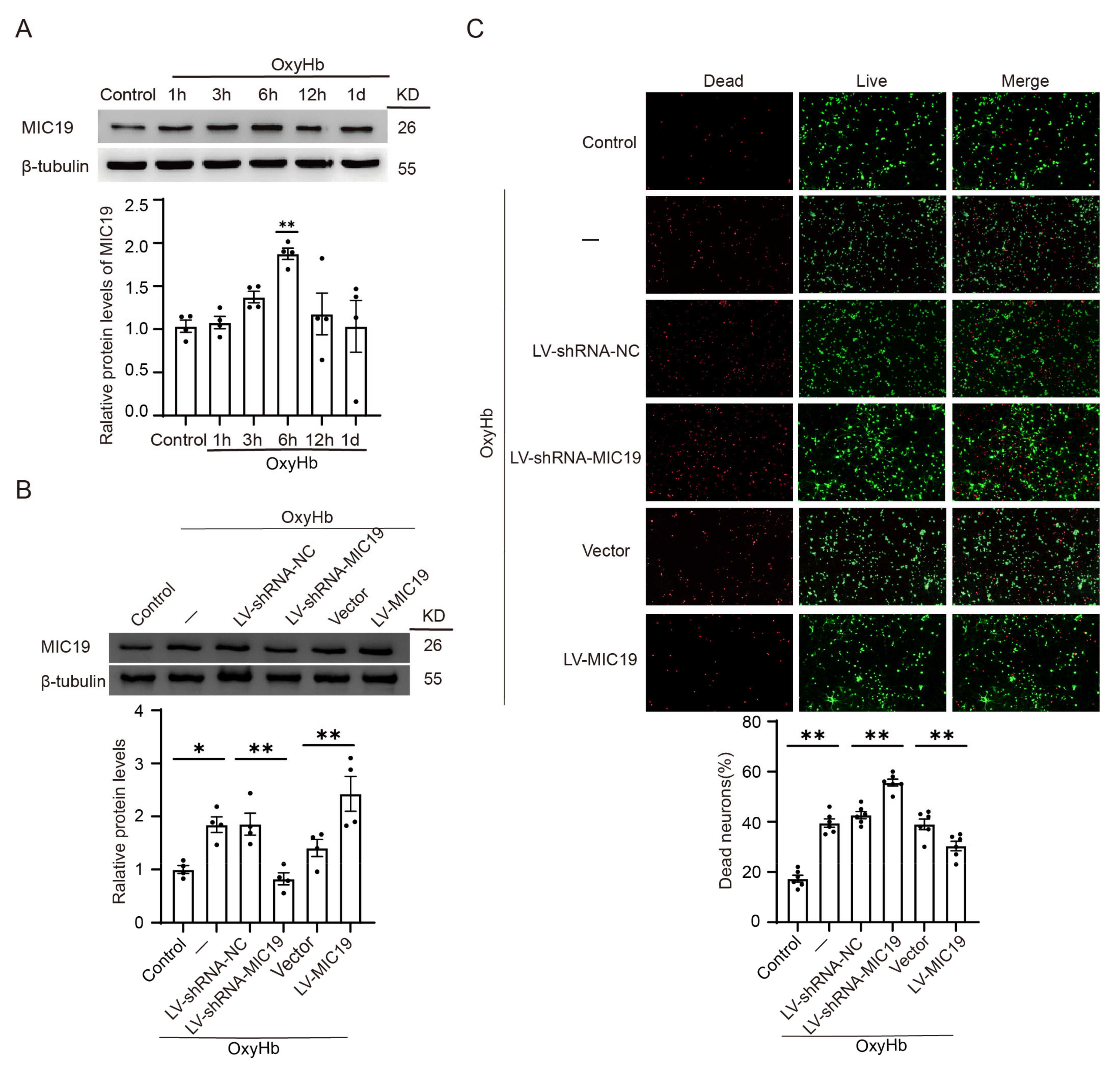
2.2. MIC19 Expression Increases in Brain Tissues and Primary-Cultured Cortical Neurons after ICH Induction
2.3. MIC19 Overexpression Alleviates ICH-Induced Neurodegeneration and Neuronal Apoptosis in Rats
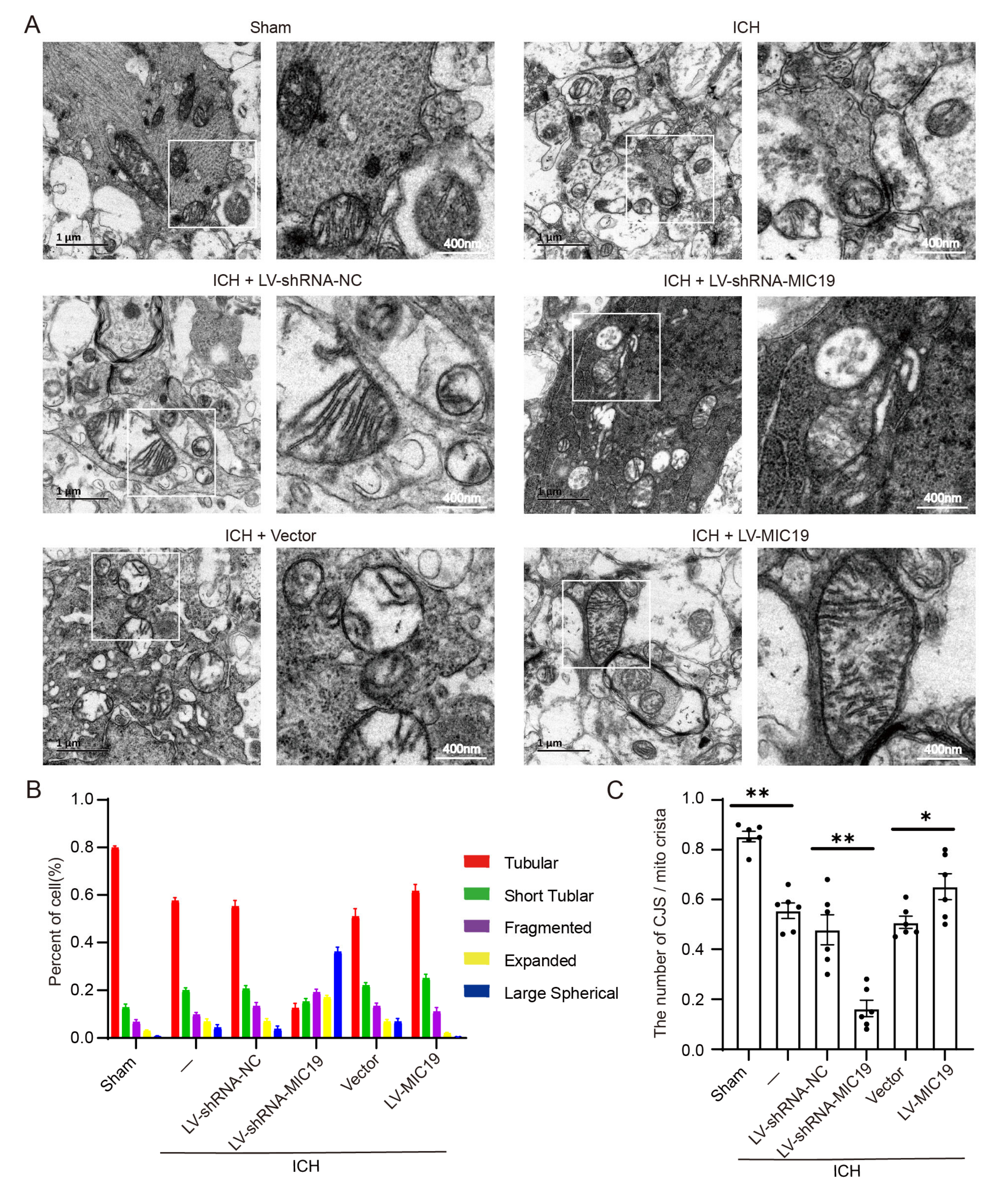
2.4. Overexpression of MIC19 Reverses ICH-Induced Mitochondrial Damage in Rats
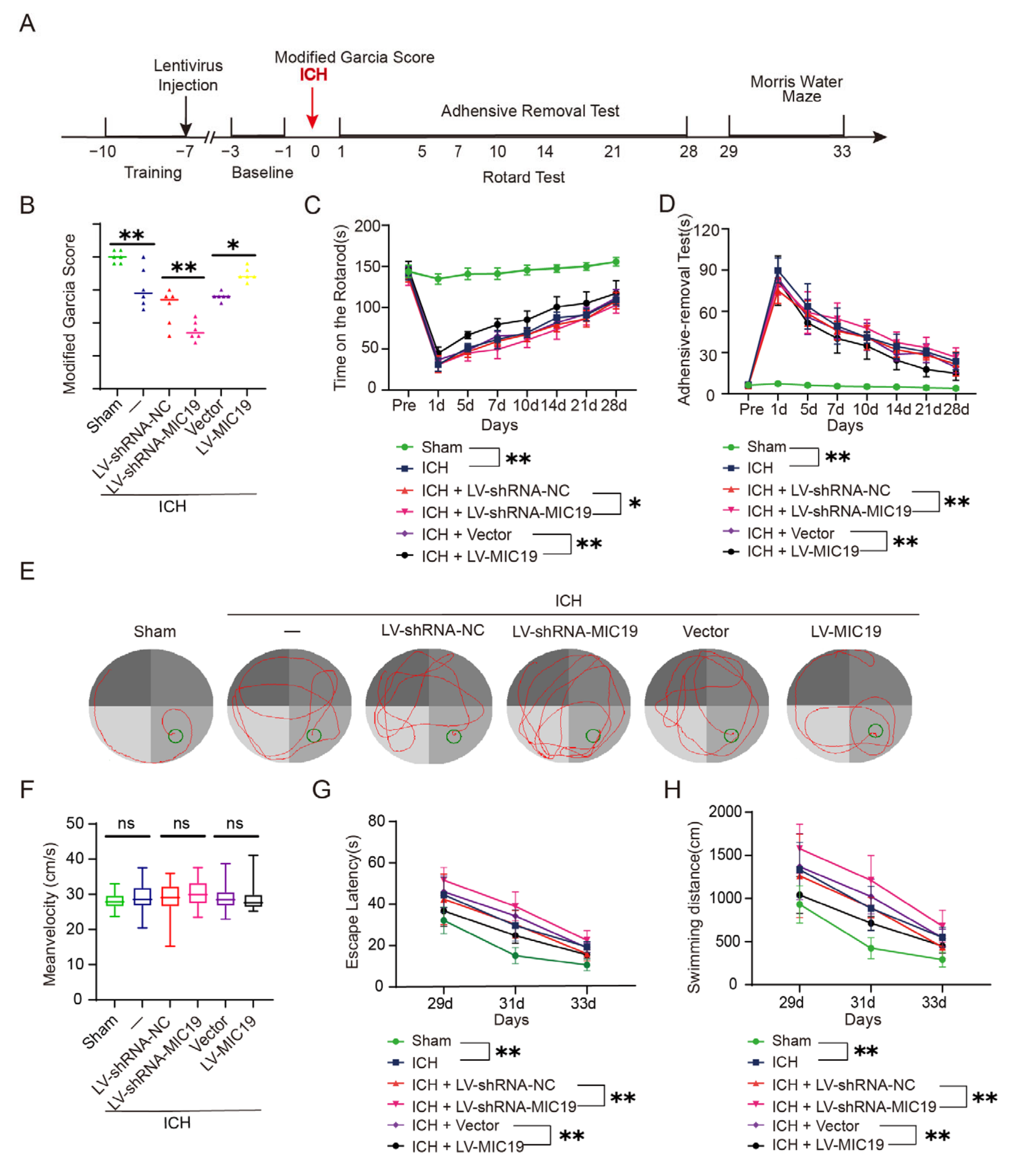
2.5. Overexpression of MIC19 Improves Behavioral Deficits in Rats after ICH Induction
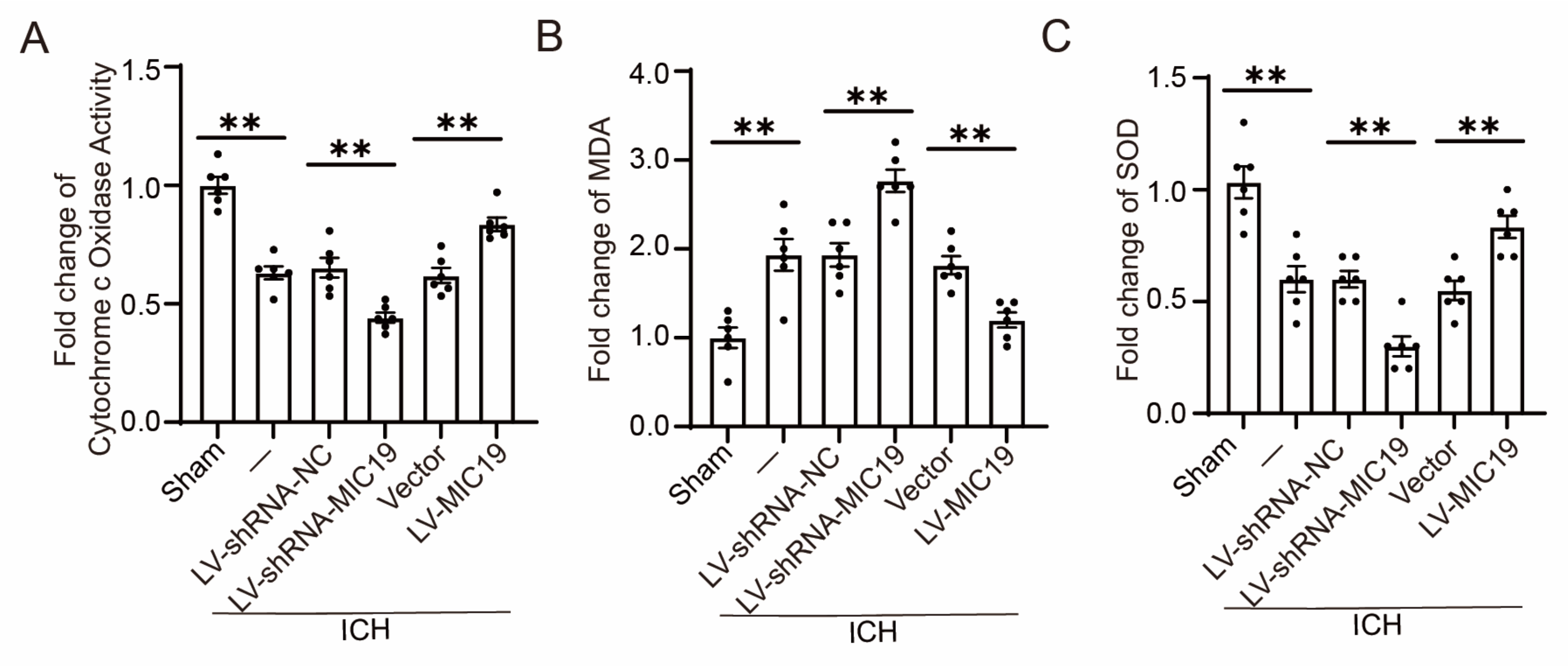
2.6. Overexpression of MIC19 Inhabits Oxidative Stress Induced by ICH and Enhances Cytochrome c Oxidase Activity in Rats
2.7. Overexpression of MIC19 Exerts Neuroprotective Effects in Neurons
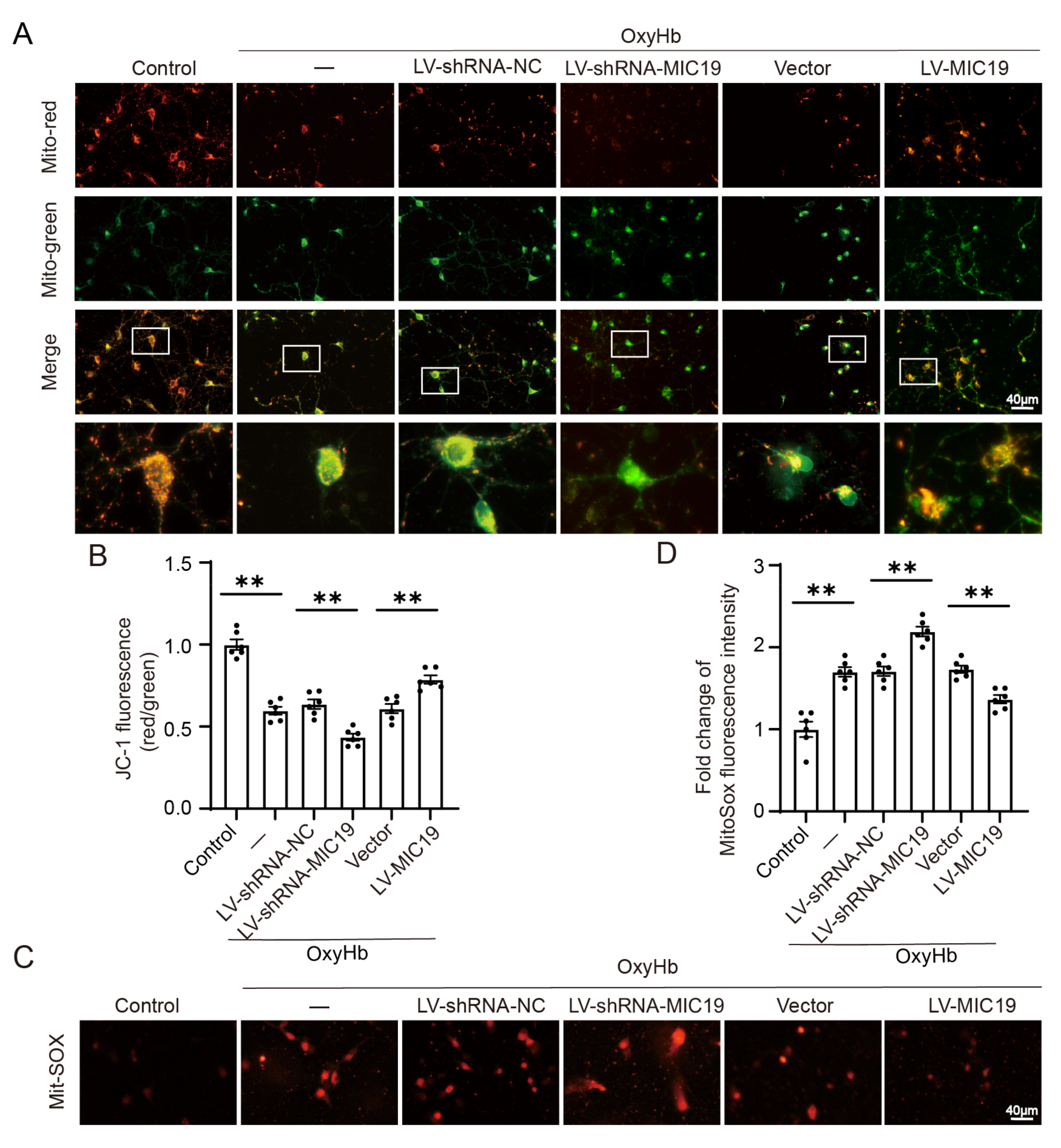
2.8. Overexpression of MIC19 Inhabits Collapsed Mitochondrial Membrane Potential and Oxidative Stress Induced by OxyHb In Vitro
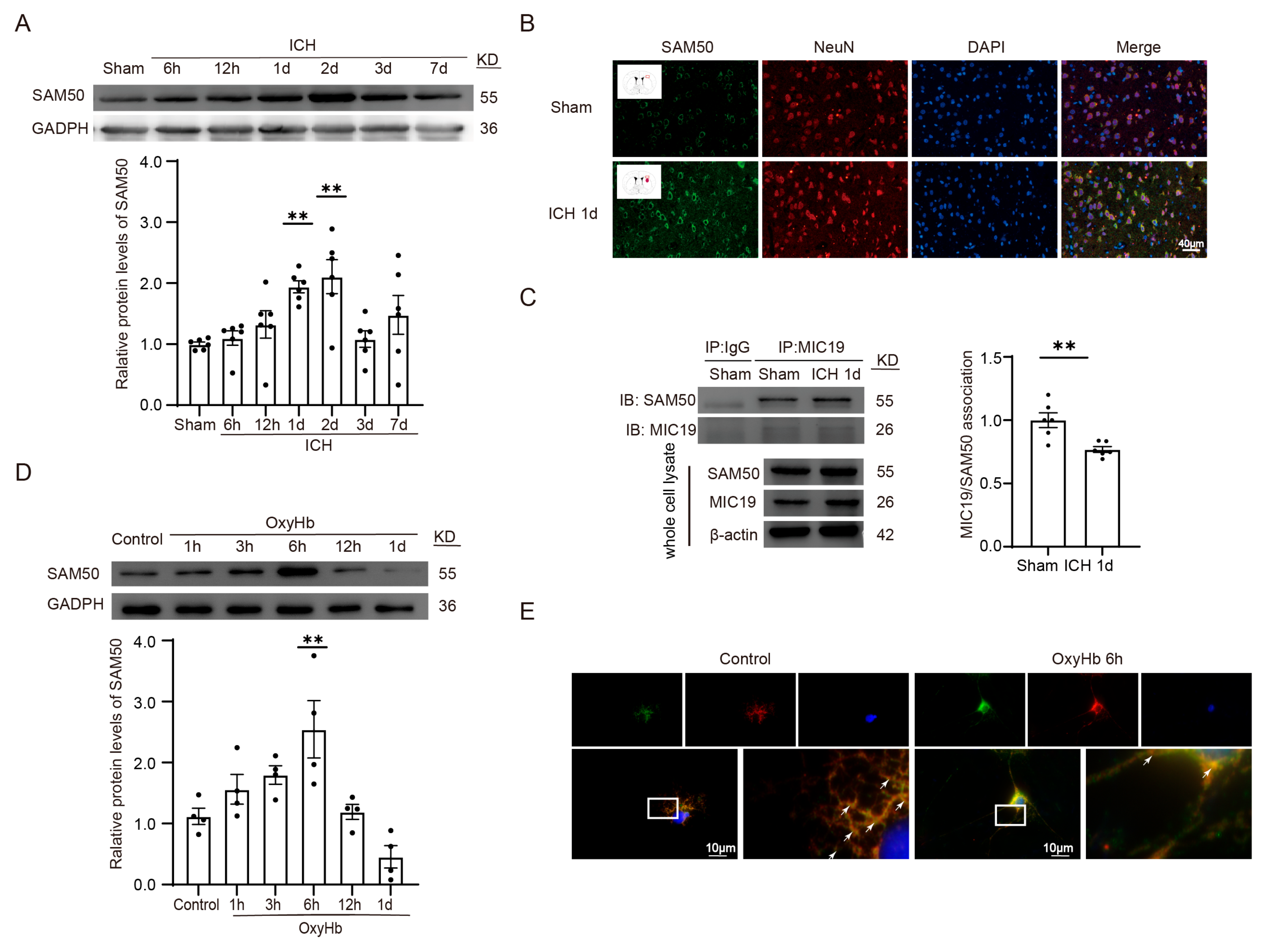
2.9. MIC19 Exerts Its Protective Role via Maintaining the Stability of MIC19–SAM50 Axis
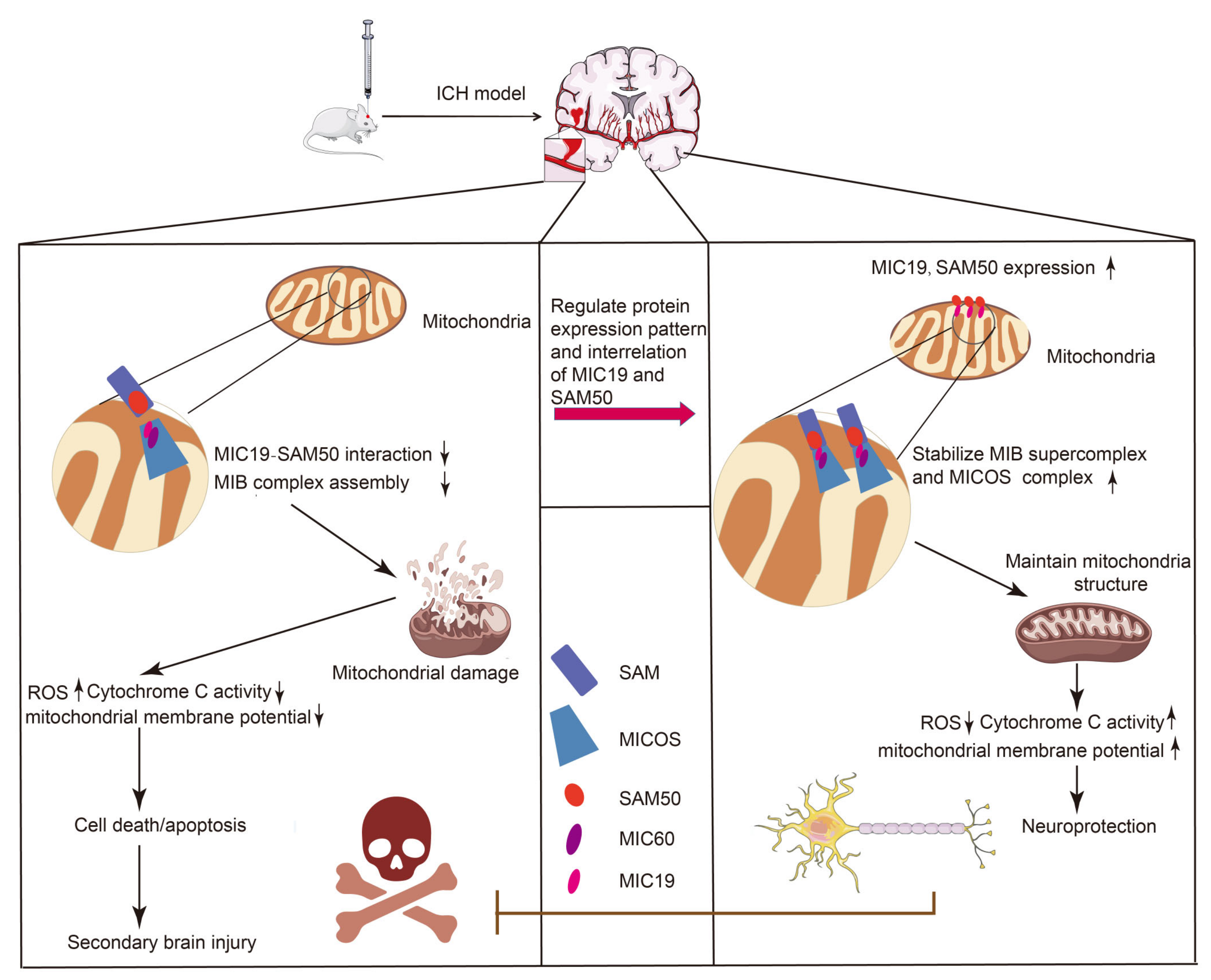
3. Discussion
4. Methods and Materials
4.1. Animals
4.2. Experimental Design
4.3. Rat Model of ICH
4.4. Primary-Cultured Cortical Neurons and ICH Induction
4.5. Intracerebral Lentivirus Injections
- Sense: 5′-GTCCTCTCCATCTGGCTCTAA-3′. titer: 4.5 × 108 TU/mL;
- Sense: 5′-AACCGCACAAAGGTGAAGCAT-3′. titer: 4.5 × 108 TU/mL;
- Sense: 5′-GAGGAGCTGGCATTGGAACAA-3′. titer: 3 × 108 TU/mL (last choice).
4.6. Western Blot
4.7. Immunofluorescent Microscopy
4.8. TUNEL and FJB Staining
4.9. Electron Microscope
4.10. Mitochondrial Morphology Analysis
4.11. Neurological Scoring
4.12. Adhesive Removal Test
4.13. Rotarod Test
4.14. Morris Water Maze
4.15. Live–Dead Cellular Staining
4.16. MitoSOX Staining
4.17. Malondialdehyde (MDA) and Superoxide Dismutase (SOD) Detection
4.18. Mitochondrial Membrane Potential Detection
4.19. Cytochrome c Oxidase Activity Colorimetric Assay
4.20. Co-Immunoprecipitation Analysis
4.21. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Xue, M.; Yong, V.W. Neuroinflammation in intracerebral haemorrhage: Immunotherapies with potential for translation. Lancet Neurol. 2020, 19, 1023–1032. [Google Scholar] [CrossRef] [PubMed]
- Rajashekar, D.; Liang, J.W. Intracerebral Hemorrhage. In StatPearls; StatPearls Publishing LLC.: Treasure Island, FL, USA, 2022. [Google Scholar]
- Cordonnier, C.; Demchuk, A.; Ziai, W.; Anderson, C.S. Intracerebral haemorrhage: Current approaches to acute management. Lancet 2018, 392, 1257–1268. [Google Scholar] [CrossRef] [PubMed]
- Kamal, H.; Ahmed, M.K.; Zha, A.; Lail, N.S.; Shirani, P.; Sawyer, R.N.; Mowla, A. Strokes occurring in the hospital: Symptom recognition and eligibility for treatment in the intensive care units versus hospital wards. Brain Circ. 2020, 6, 196–199. [Google Scholar] [PubMed]
- Masomi-Bornwasser, J.; Kurz, E.; Frenz, C.; Schmitt, J.; Wesp, D.M.A.; König, J.; Lotz, J.; Ringel, F.; Kerz, T.; Krenzlin, H.; et al. The Influence of Oxidative Stress on Neurological Outcomes in Spontaneous Intracerebral Hemorrhage. Biomolecules 2021, 11, 1615. [Google Scholar] [CrossRef] [PubMed]
- Bautista, W.; Adelson, P.D.; Bicher, N.; Themistocleous, M.; Tsivgoulis, G.; Chang, J.J. Secondary mechanisms of injury and viable pathophysiological targets in intracerebral hemorrhage. Ther. Adv. Neurol. Disord. 2021, 14, 17562864211049208. [Google Scholar] [CrossRef]
- Alkhalil, M. A promising tool to tackle the risk of cerebral vascular disease, the emergence of novel carotid wall imaging. Brain Circ. 2020, 6, 81–86. [Google Scholar] [CrossRef]
- Ren, H.; Han, R.; Chen, X.; Liu, X.; Wan, J.; Wang, L.; Yang, X.; Wang, J. Potential therapeutic targets for intracerebral hemorrhage-associated inflammation: An update. J. Cereb. Blood Flow Metab. 2020, 40, 1752–1768. [Google Scholar] [CrossRef]
- Lule, S.; Wu, L.; Sarro-Schwartz, A.; Edmiston, W.J., 3rd; Izzy, S.; Songtachalert, T.; Ahn, S.H.; Fernandes, N.D.; Jin, G.; Chung, J.Y.; et al. Cell-specific activation of RIPK1 and MLKL after intracerebral hemorrhage in mice. J. Cereb. Blood Flow Metab. 2021, 41, 1623–1633. [Google Scholar] [CrossRef]
- Mayer, S.A.; Rincon, F. Treatment of intracerebral haemorrhage. Lancet Neurol. 2005, 4, 662–672. [Google Scholar] [CrossRef]
- Zheng, J.; Shi, L.; Liang, F.; Xu, W.; Li, T.; Gao, L.; Sun, Z.; Yu, J.; Zhang, J. Sirt3 Ameliorates Oxidative Stress and Mitochondrial Dysfunction After Intracerebral Hemorrhage in Diabetic Rats. Front. Neurosci. 2018, 12, 414. [Google Scholar] [CrossRef]
- van Asch, C.J.; Luitse, M.J.; Rinkel, G.J.; van der Tweel, I.; Algra, A.; Klijn, C.J. Incidence, case fatality, and functional outcome of intracerebral haemorrhage over time, according to age, sex, and ethnic origin: A systematic review and meta-analysis. Lancet Neurol. 2010, 9, 167–176. [Google Scholar] [CrossRef] [PubMed]
- Annesley, S.J.; Fisher, P.R. Mitochondria in Health and Disease. Cells 2019, 8, 680. [Google Scholar] [CrossRef] [PubMed]
- Kramer, P.; Bressan, P. Our (Mother’s) Mitochondria and Our Mind. Perspect. Psychol. Sci. 2018, 13, 88–100. [Google Scholar] [CrossRef] [PubMed]
- Carota, G.; Distefano, A.; Spampinato, M.; Giallongo, C.; Broggi, G.; Longhitano, L.; Palumbo, G.A.; Parenti, R.; Caltabiano, R.; Giallongo, S.; et al. Neuroprotective Role of α-Lipoic Acid in Iron-Overload-Mediated Toxicity and Inflammation in In Vitro and In Vivo Models. Antioxidants 2022, 11, 1596. [Google Scholar] [CrossRef] [PubMed]
- Kim-Han, J.S.; Kopp, S.J.; Dugan, L.L.; Diringer, M.N. Perihematomal mitochondrial dysfunction after intracerebral hemorrhage. Stroke 2006, 37, 2457–2462. [Google Scholar] [CrossRef]
- Sakowska, P.; Jans, D.C.; Mohanraj, K.; Riedel, D.; Jakobs, S.; Chacinska, A. The Oxidation Status of Mic19 Regulates MICOS Assembly. Mol. Cell. Biol. 2015, 35, 4222–4237. [Google Scholar] [CrossRef]
- Li, H.; Ruan, Y.; Zhang, K.; Jian, F.; Hu, C.; Miao, L.; Gong, L.; Sun, L.; Zhang, X.; Chen, S.; et al. Mic60/Mitofilin determines MICOS assembly essential for mitochondrial dynamics and mtDNA nucleoid organization. Cell Death Differ. 2016, 23, 380–392. [Google Scholar] [CrossRef]
- Tang, J.; Zhang, K.; Dong, J.; Yan, C.; Hu, C.; Ji, H.; Chen, L.; Chen, S.; Zhao, H.; Song, Z. Sam50-Mic19-Mic60 axis determines mitochondrial cristae architecture by mediating mitochondrial outer and inner membrane contact. Cell Death Differ. 2020, 27, 146–160. [Google Scholar] [CrossRef]
- Sastri, M.; Darshi, M.; Mackey, M.; Ramachandra, R.; Ju, S.; Phan, S.; Adams, S.; Stein, K.; Douglas, C.R.; Kim, J.J.; et al. Sub-mitochondrial localization of the genetic-tagged mitochondrial intermembrane space-bridging components Mic19, Mic60 and Sam50. J. Cell Sci. 2017, 130, 3248–3260. [Google Scholar]
- Deinsberger, W.; Vogel, J.; Kuschinsky, W.; Auer, L.M.; Böker, D.K. Experimental intracerebral hemorrhage: Description of a double injection model in rats. Neurol. Res. 1996, 18, 475–477. [Google Scholar] [CrossRef]
- Deng, R.; Wang, W.; Xu, X.; Ding, J.; Wang, J.; Yang, S.; Li, H.; Shen, H.; Li, X.; Chen, G. Loss of MIC60 Aggravates Neuronal Death by Inducing Mitochondrial Dysfunction in a Rat Model of Intracerebral Hemorrhage. Mol. Neurobiol. 2021, 58, 4999–5013. [Google Scholar] [CrossRef] [PubMed]
- Ren, R.; Fang, Y.; Sherchan, P.; Lu, Q.; Lenahan, C.; Zhang, J.H.; Zhang, J.; Tang, J. Kynurenine/Aryl Hydrocarbon Receptor Modulates Mitochondria-Mediated Oxidative Stress and Neuronal Apoptosis in Experimental Intracerebral Hemorrhage. Antioxid. Redox Signal. 2022, 37, 1111–1129. [Google Scholar] [CrossRef]
- Nunnari, J.; Suomalainen, A. Mitochondria: In sickness and in health. Cell 2012, 148, 1145–1159. [Google Scholar] [CrossRef]
- Cogliati, S.; Enriquez, J.A.; Scorrano, L. Mitochondrial Cristae: Where Beauty Meets Functionality. Trends Biochem. Sci. 2016, 41, 261–273. [Google Scholar] [CrossRef] [PubMed]
- Anand, R.; Strecker, V.; Urbach, J.; Wittig, I.; Reichert, A.S. Mic13 Is Essential for Formation of Crista Junctions in Mammalian Cells. PLoS ONE 2016, 11, e0160258. [Google Scholar] [CrossRef]
- Friedman, J.R.; Mourier, A.; Yamada, J.; McCaffery, J.M.; Nunnari, J. MICOS coordinates with respiratory complexes and lipids to establish mitochondrial inner membrane architecture. eLife 2015, 4, e07739. [Google Scholar] [CrossRef]
- Bock-Bierbaum, T.; Funck, K.; Wollweber, F.; Lisicki, E.; von der Malsburg, K.; von der Malsburg, A.; Laborenz, J.; Noel, J.K.; Hessenberger, M.; Jungbluth, S.; et al. Structural insights into crista junction formation by the Mic60-Mic19 complex. Sci. Adv. 2022, 8, eabo4946. [Google Scholar] [CrossRef]
- Huynen, M.A.; Mühlmeister, M.; Gotthardt, K.; Guerrero-Castillo, S.; Brandt, U. Evolution and structural organization of the mitochondrial contact site (MICOS) complex and the mitochondrial intermembrane space bridging (MIB) complex. Biochim. Biophys. Acta 2016, 1863, 91–101. [Google Scholar] [CrossRef] [PubMed]
- Darshi, M.; Trinh, K.N.; Murphy, A.N.; Taylor, S.S. Targeting and import mechanism of coiled-coil helix coiled-coil helix domain-containing protein 3 (ChChd3) into the mitochondrial intermembrane space. J. Biol. Chem. 2012, 287, 39480–39491. [Google Scholar] [CrossRef]
- Xue, X.; Ma, L.; Zhang, X.; Xu, X.; Guo, S.; Wang, Y.; Qiu, S.; Cui, J.; Guo, W.; Yu, Y.; et al. Tumour cells are sensitised to ferroptosis via RB1CC1-mediated transcriptional reprogramming. Clin. Transl. Med. 2022, 12, e747. [Google Scholar] [CrossRef]
- Meng, C.; Zhang, J.; Dang, B.; Li, H.; Shen, H.; Li, X.; Wang, Z. PERK Pathway Activation Promotes Intracerebral Hemorrhage Induced Secondary Brain Injury by Inducing Neuronal Apoptosis Both in Vivo and in Vitro. Front. Neurosci. 2018, 12, 111. [Google Scholar] [CrossRef] [PubMed]
- Latorre-Muro, P.; O’Malley, K.E.; Bennett, C.F.; Perry, E.A.; Balsa, E.; Tavares, C.D.J.; Jedrychowski, M.; Gygi, S.P.; Puigserver, P. A cold-stress-inducible PERK/OGT axis controls TOM70-assisted mitochondrial protein import and cristae formation. Cell Metab. 2021, 33, 598–614.e7. [Google Scholar] [CrossRef] [PubMed]
- Percie du Sert, N.; Hurst, V.; Ahluwalia, A.; Alam, S.; Avey, M.T.; Baker, M.; Browne, W.J.; Clark, A.; Cuthill, I.C.; Dirnagl, U.; et al. The ARRIVE guidelines 2.0: Updated guidelines for reporting animal research. Br. J. Pharmacol. 2020, 177, 3617–3624. [Google Scholar] [CrossRef]
- Yuan, G.; Cao, C.; Cao, D.; Li, B.; Li, X.; Li, H.; Shen, H.; Wang, Z.; Chen, G. Receptor-interacting protein 3-phosphorylated Ca(2+)/calmodulin-dependent protein kinase II and mixed lineage kinase domain-like protein mediate intracerebral hemorrhage-induced neuronal necroptosis. J. Neurochem. 2022, 164, 94–114. [Google Scholar] [CrossRef]
- Xu, H.; Cao, J.; Xu, J.; Li, H.; Shen, H.; Li, X.; Wang, Z.; Wu, J.; Chen, G. GATA-4 regulates neuronal apoptosis after intracerebral hemorrhage via the NF-κB/Bax/Caspase-3 pathway both in vivo and in vitro. Exp. Neurol. 2019, 315, 21–31. [Google Scholar] [CrossRef]
- Zhuang, Y.; Xu, H.; Richard, S.A.; Cao, J.; Li, H.; Shen, H.; Yu, Z.; Zhang, J.; Wang, Z.; Li, X.; et al. Inhibition of EPAC2 Attenuates Intracerebral Hemorrhage-Induced Secondary Brain Injury via the p38/BIM/Caspase-3 Pathway. J. Mol. Neurosci. 2019, 67, 353–363. [Google Scholar] [CrossRef]
- Fan, W.; Li, X.; Zhang, D.; Li, H.; Shen, H.; Liu, Y.; Chen, G. Detrimental Role of miRNA-144-3p in Intracerebral Hemorrhage Induced Secondary Brain Injury is Mediated by Formyl Peptide Receptor 2 Downregulation Both In Vivo and In Vitro. Cell Transplant. 2019, 28, 723–738. [Google Scholar] [CrossRef]
- Chen, C.; Lu, L.; Zhu, J.; Gu, X.; Liu, B.; Li, D.; Chen, G. Miro1 provides neuroprotection via the mitochondrial trafficking pathway in a rat model of traumatic brain injury. Brain Res. 2021, 1773, 147685. [Google Scholar] [CrossRef]
- Shi, X.; Bai, H.; Wang, J.; Wang, J.; Huang, L.; He, M.; Zheng, X.; Duan, Z.; Chen, D.; Zhang, J.; et al. Behavioral Assessment of Sensory, Motor, Emotion, and Cognition in Rodent Models of Intracerebral Hemorrhage. Front. Neurol. 2021, 12, 667511. [Google Scholar] [CrossRef]
- Xiao, J.; Cai, T.; Fang, Y.; Liu, R.; Flores, J.J.; Wang, W.; Gao, L.; Liu, Y.; Lu, Q.; Tang, L.; et al. Activation of GPR40 attenuates neuroinflammation and improves neurological function via PAK4/CREB/KDM6B pathway in an experimental GMH rat model. J. Neuroinflammation 2021, 18, 160. [Google Scholar] [CrossRef] [PubMed]
- Lei, B.; James, M.L.; Liu, J.; Zhou, G.; Venkatraman, T.N.; Lascola, C.D.; Acheson, S.K.; Dubois, L.G.; Laskowitz, D.T.; Wang, H. Neuroprotective pentapeptide CN-105 improves functional and histological outcomes in a murine model of intracerebral hemorrhage. Sci. Rep. 2016, 6, 34834. [Google Scholar] [CrossRef] [PubMed]
- Lei, B.; Wang, H.; Jeong, S.; Hsieh, J.T.; Majeed, M.; Dawson, H.; Sheng, H.; Warner, D.S.; James, M.L. Progesterone Improves Neurobehavioral Outcome in Models of Intracerebral Hemorrhage. Neuroendocrinology 2016, 103, 665–677. [Google Scholar] [CrossRef] [PubMed]
- Tan, X.; Yang, Y.; Xu, J.; Zhang, P.; Deng, R.; Mao, Y.; He, J.; Chen, Y.; Zhang, Y.; Ding, J.; et al. Luteolin Exerts Neuroprotection via Modulation of the p62/Keap1/Nrf2 Pathway in Intracerebral Hemorrhage. Front. Pharmacol. 2019, 10, 1551. [Google Scholar] [CrossRef] [PubMed]
- Liang, T.; Ma, C.; Wang, T.; Deng, R.; Ding, J.; Wang, W.; Xu, Z.; Li, X.; Li, H.; Sun, Q.; et al. Galectin-9 Promotes Neuronal Restoration via Binding TLR-4 in a Rat Intracerebral Hemorrhage Model. Neuromolecular Med. 2021, 23, 267–284. [Google Scholar] [CrossRef]

| Sub-Test | 0 | 1 | 2 | 3 |
|---|---|---|---|---|
| Spontaneous activity (SA) | Animal was akinetic | Animal moved slowly or minimally | Animal approached 1–2 walls | Animal approached at least 3 walls of the cage or raised on hindlimbs to explore the top of the cage |
| Body proprioception (BP) | Animal had a unilateral response | Animal had either a weak bilateral response or weak left response and brisk right response | Animal had a brisk bilateral response | |
| Vibrissae touch (VT) | Animal had a unilateral response | Animal had either a weak bilateral response or weak left response and brisk right response | Animal had a brisk bilateral response | |
| Limb symmetry (LS) | Hemiparesis | Left forelimb or left hindlimb fixed | Asymmetric extension | All limbs were extended symmetrically |
| Lateral turning (LT) | Animal had no turning at all on one side | Animal had unequal turning | Animal turned bilaterally less than 45° on both sides | Animal turned bilaterally at least 45° on both sides |
| Forelimb outstretching (FO) | Animal had a paretic forelimb | Animal walked in circles | Animal walked asymmetrically or to one side | Animal briskly walked symmetrically on forepaws |
| Climbing (CL) | Animal failed to climb or circled instead of climbing | Animal climbed to the top and had a weak grip or animal climbed but had a strong grip | Animal climbed to the top and had a strong grip |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, S.; Yin, X.; Wang, J.; Li, H.; Shen, H.; Sun, Q.; Li, X. MIC19 Exerts Neuroprotective Role via Maintaining the Mitochondrial Structure in a Rat Model of Intracerebral Hemorrhage. Int. J. Mol. Sci. 2023, 24, 11553. https://doi.org/10.3390/ijms241411553
Yang S, Yin X, Wang J, Li H, Shen H, Sun Q, Li X. MIC19 Exerts Neuroprotective Role via Maintaining the Mitochondrial Structure in a Rat Model of Intracerebral Hemorrhage. International Journal of Molecular Sciences. 2023; 24(14):11553. https://doi.org/10.3390/ijms241411553
Chicago/Turabian StyleYang, Siyuan, Xulong Yin, Jiahe Wang, Haiying Li, Haitao Shen, Qing Sun, and Xiang Li. 2023. "MIC19 Exerts Neuroprotective Role via Maintaining the Mitochondrial Structure in a Rat Model of Intracerebral Hemorrhage" International Journal of Molecular Sciences 24, no. 14: 11553. https://doi.org/10.3390/ijms241411553
APA StyleYang, S., Yin, X., Wang, J., Li, H., Shen, H., Sun, Q., & Li, X. (2023). MIC19 Exerts Neuroprotective Role via Maintaining the Mitochondrial Structure in a Rat Model of Intracerebral Hemorrhage. International Journal of Molecular Sciences, 24(14), 11553. https://doi.org/10.3390/ijms241411553






