Abstract
With the wide usage of organic compounds, the assessment of their acute toxicity has drawn great attention to reduce animal testing and human labor. The development of graph models provides new opportunities for acute toxicity prediction. In this study, five graph models (message-passing neural network, graph convolution network, graph attention network, path-augmented graph transformer network, and Attentive FP) were applied on four toxicity tasks (fish, Daphnia magna, Tetrahymena pyriformis, and Vibrio fischeri). With the lowest prediction error, Attentive FP was reported to have the best performance in all four tasks. Moreover, the attention weights of the Attentive FP model helped to construct atomic heatmaps and provide good explainability.
1. Introduction
The growing utilization of various chemicals, including medicines, pesticides, and chemical fertilizers, has sparked increasing interest due to their considerable toxicity [1]. Apart from criticism for animal testing, assessing the toxicity and risk of chemicals is a complex task that often requires significant time and effort [2]. Therefore, the research for chemical toxicity assessment is crucial in the advancement of alternative methods to animal testing and will support the development of predictive modeling [3].
Many drugs also fail clinical trials due to their unmanageable toxicity, which is a significant cause of the high failure rate in clinical drug development. To alleviate this problem in an already long and costly process, it would be ideal to gauge toxicity directly from the molecular structure [4]. However, this is a challenging task for humans because chemical systems are complex and such properties are not always immediately evident from molecular substructures. In fact, determining effective methods to represent molecules for toxicity prediction remains challenging, though many exist, including theoretical, empirical, and semi-empirical representations. Descriptors representing a molecular structure may be divided into graph-based and geometry-based types. Graph-based representations primarily describe the connectivity between atoms, while geometry-based representations include information about the molecule’s physical geometry, including bond lengths and angles [5].
Traditionally, many quantitative structure–toxicity relationship (QSTR) models have been introduced as a means to evaluate and predict chemical risk, relying solely on molecular structure information [6,7,8]. The molecular structure is featurized by physical and chemical properties such as size, covalent bonds, and polarity, and it can be effectively characterized through quantitative molecular descriptors. Among the most common descriptors for toxicity prediction are Molecular Access System keys (MACCS Keys), PubChem Substructure Fingerprints, Klekotha–Roth, and Extended-Connectivity Fingerprints (ECFPs) [9]. However, these models require predefined features and molecular descriptors, and a lack of exhaustiveness would lead to poor performance. With more than 5000 molecular descriptors available for characterizing molecules, it is not always clear how to select the most appropriate representation while avoiding selection bias. In place of such general descriptors, there has been growing interest in alternative data-driven representations via deep learning [5,10,11].
With the development of molecular graph representations and deep learning, graph neural networks (GNNs) are one of the leading research areas for molecular property prediction. In a molecular graph, atoms are treated as vertices and bonds are treated as edges. By functioning directly on molecular graphs, GNNs provide a more accurate description of the chemical structure with connectivity information and can allow for more relevant representations of molecules without having to consider many-body interactions or complex electronic structures [5,12,13]. Starting from message-passing neural networks (MPNNs) [14], many variations have been developed with higher complexity or additional attention mechanisms, such as the graph convolution network (GCN), graph attention network (GAT), path-augmented graph transformer network (PAGTN) [15], and Attentive FP model [5]. These architectures employ various techniques to overcome the complexity and irregularity of molecular graphs.
In recent years, deep learning has grown popular for toxicity prediction due to its successful performance and because it avoids a challenging feature selection process. For example, Mayr et al.’s DeepTox pipeline proved effective on the Tox21 data challenge and outperformed shallower machine learning methods [16]. The choice of molecular representation has also shown to be important in this domain, and graphs have been shown to be useful in encoding a molecular structure, as vertices can represent atoms and edges can represent connections between atoms [17]. In 2017, Xu et al., introduced molecular graph-encoding convolutional neural networks for acute oral toxicity (AOT) prediction via their deepAOT model [10]. This model uses graph convolutions to represent molecules, removing the need to calculate fingerprints and allowing the algorithm to learn more directly from the data. Wu and Wei presented another topology-based molecular representation in 2018 that paired element-specific persistent homology with multitask deep learning and proved successful on four benchmark toxicity datasets [18].
In this study, the performance of five graph models (MPNN, GCN, GAT, PAGTN, and Attentive FP) was evaluated on four acute toxicity tasks (fish, Daphnia magna, Tetrahymena pyriformis, and Vibrio fischeri). With rigorous training and testing, Attentive FP was identified as the best-performing model with low prediction error. Furthermore, the attention weights from the trained Attentive FP model can help visualize atom importance with a molecular heatmap. This interpretability is especially important in molecular property prediction for understanding the model’s internal workings and gaining new scientific insights [19].
2. Results
2.1. Graph Models Training
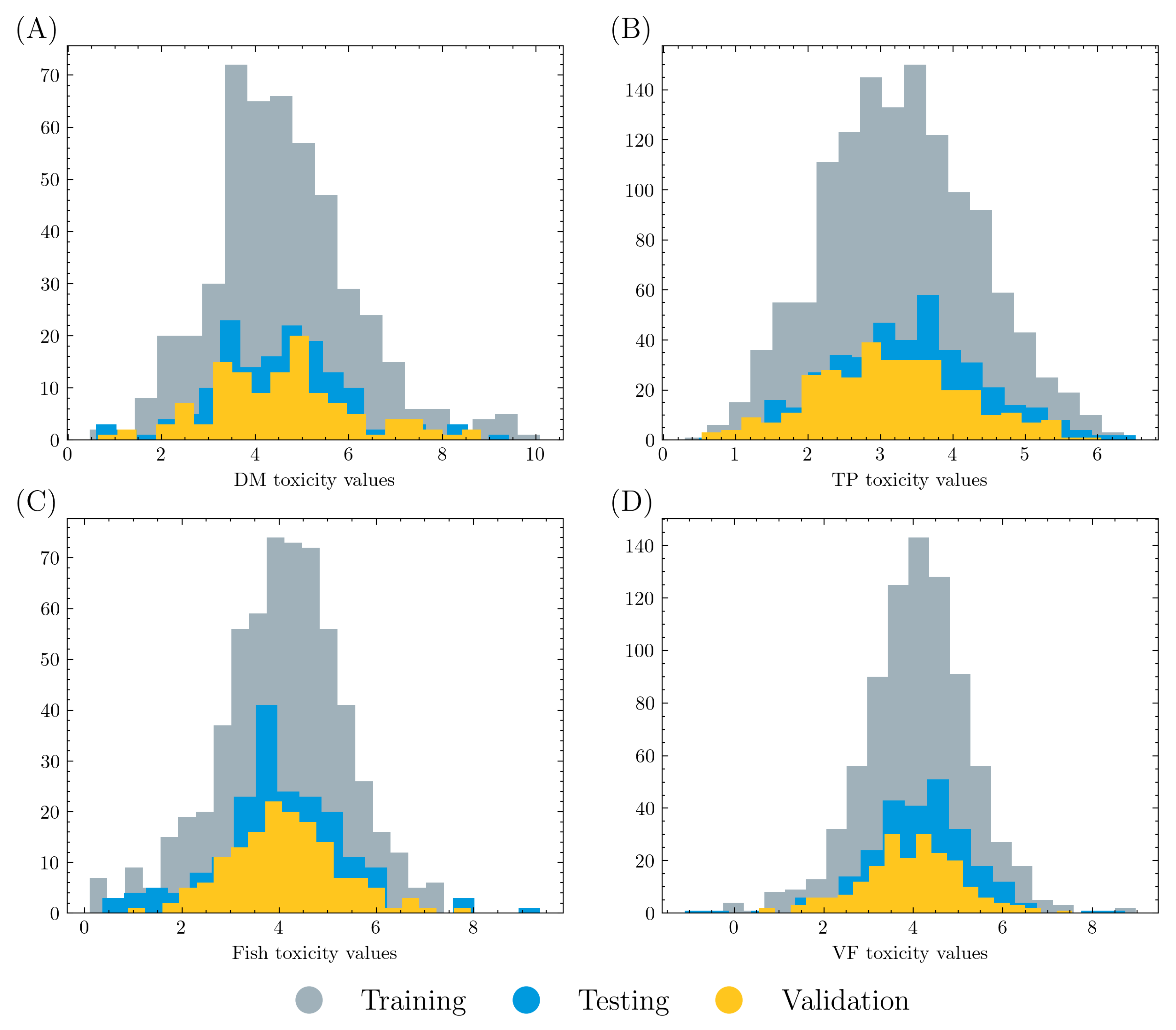
The overall dataset was randomly split into a training set (80%) and a test set (20%). Five-fold cross validation was applied on the training set, where 80% of the training data were used to train a graph model and 20% of the data were used for validation in each training process. Therefore, training, validation, and testing data account for 64%, 16%, and 20% of the overall dataset, respectively. The data distributions are displayed in Figure 1. It is shown that all three datasets have similar distributions in each acute toxicity task.
Figure 1.
Count and value distribution in (A) DM, (B) TP, (C) fish, and (D) VF acute toxicity tasks. Training, validation, and testing data account for 64%, 16%, and 20% of the overall dataset, respectively. All three datasets in each task have similar distribution.
The parameters being fine-tuned in the graph models are listed in Table 1. The learning rate was fixed as 0.001 in all graph models. The batch size and dropout rate parameters were tuned in all models. The number of GNN layers was tuned for the PAGTN and Attentive FP models with potential values in 1, 2, and 4. The number of graph convolution layers was tuned for the GCN and GAT models from two layers of 64, 128, and 256 units. The MPNN model tuned a similar parameter of the number of units in the hidden layer. It also tuned the number of atom output features with 15, 30 and 60. Similarly, the PAGTN model tuned the size for the hidden node features with 64, 128, and 256. The selected parameters can be classified into sample size (batch size), regularization (dropout), and complexity (number of GNN layers, number of graph convolution layers, number of units, number of atom output features, and size for the hidden node features). The full combination of all the parameters was tested.

Table 1.
Parameters being fine-tuned in graph models.
2.2. Superior Performance of Attentive FP
The mean MSE, MAPE, and R2 metrics were calculated under each parameter setting. The metric calculations are detailed in Equations (4)–(6). The model with the lower MSE and MAPE and higher R2 was considered to have better performance. The best model setting on the training set was taken and applied in the test set. The model was trained in five independent runs, and the mean and standard deviation are calculated and summarized in Table 2. In all four tasks, Attentive FP performed the best among the five graph models. Specifically, in the VF and TP tasks, Attentive FP was significantly better than the other models with a 12.3% and 13.3% decrease in the MSE as compared to the second-best GCN model, respectively. The GCN model was the second-best model with 3rd place in the DM task and 2nd place in the other three tasks. The other three models have varying performance in different tasks. For example, the GAT model was ranked 2nd in the DM task but last in the VF task. At the task level, the graph models had the best performance on the TP task and similar performance on the VF and fish tasks, with DM being the worst.

Table 2.
Model performance evaluated on test set.
To further validate that graph models are truly reflective of a structure–activity relationship, a y-randomization test was performed for the Attentive FP and GCN models. The toxicity values on the training set were randomly shuffled and used for model training under the same best-performing hyperparameter sets. For the Attentive FP model, the R2 values were 0.0142, 0.0048, 0.0780, and 0.0126 for the VF, DM, TP, and fish tasks. For the GCN models, the corresponding values were 0.0591, 0.0568, 0.0624, and 0.0007.
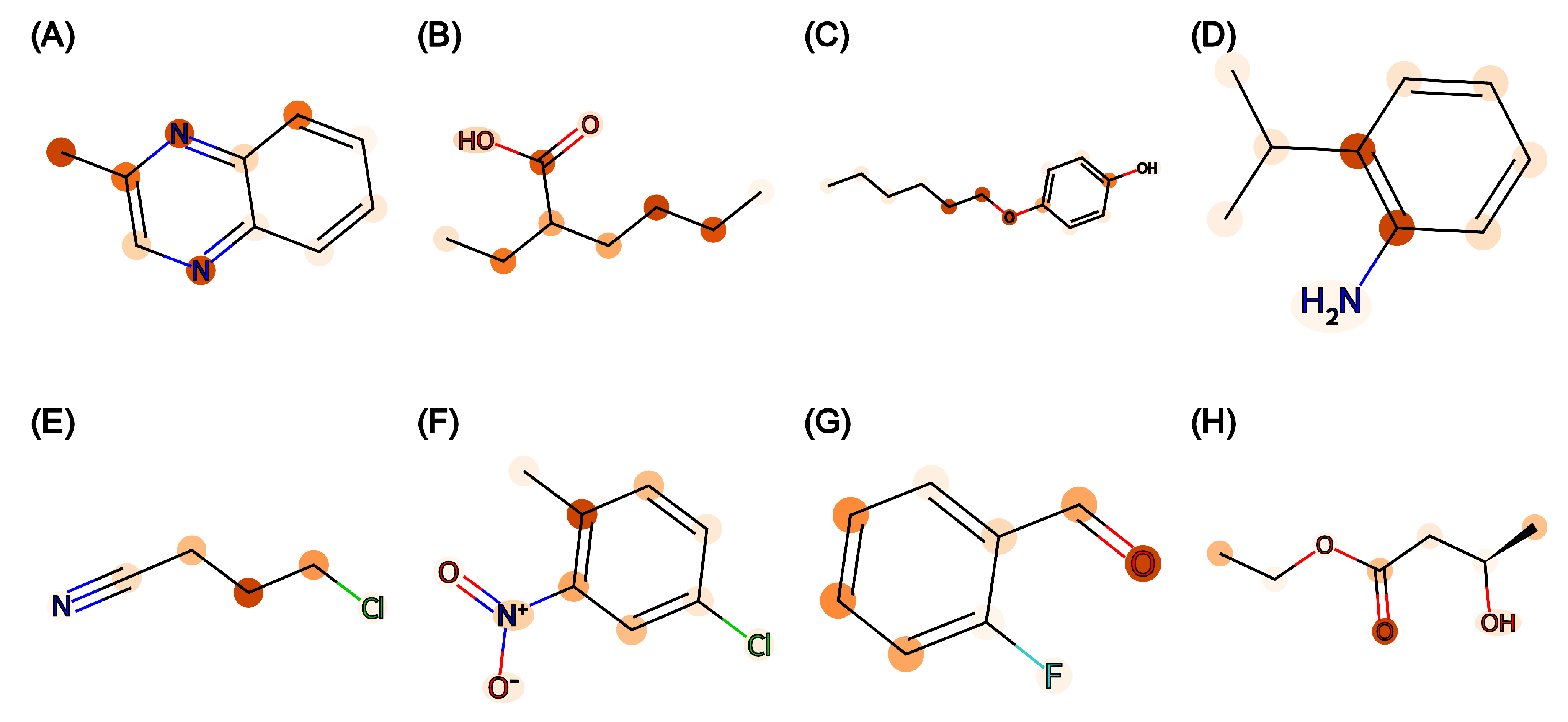
The attention weights on the attention layer of Attentive FP were extracted to produce molecular heatmaps. Figure 2 displays the heatmap of eight molecules with a low prediction error (<0.01) from the trained Attentive FP model on the TP task. It is shown that many carbon and nitrogen atoms on aromatic rings were assigned a high weight. While other graph models do not have weights at the atomic level, the attention weights and heatmap from the Attentive FP model can be used to explain model prediction against single prediction and can be extended for rational design.
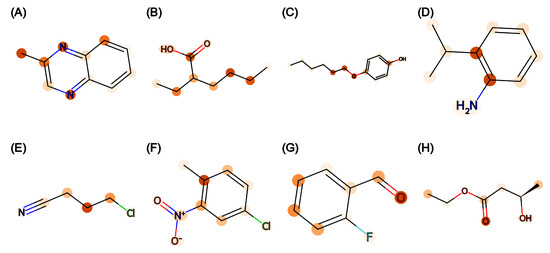
Figure 2.
Molecular heatmap with attention weights of Attentive FP trained for the TP task. (A–H) are eight randomly selected molecules in the test set of TP task. In each molecule, atomic bonds are represented in black, blue, red, or green lines according to atom types. The atom with higher weight has darker color.
3. Discussion
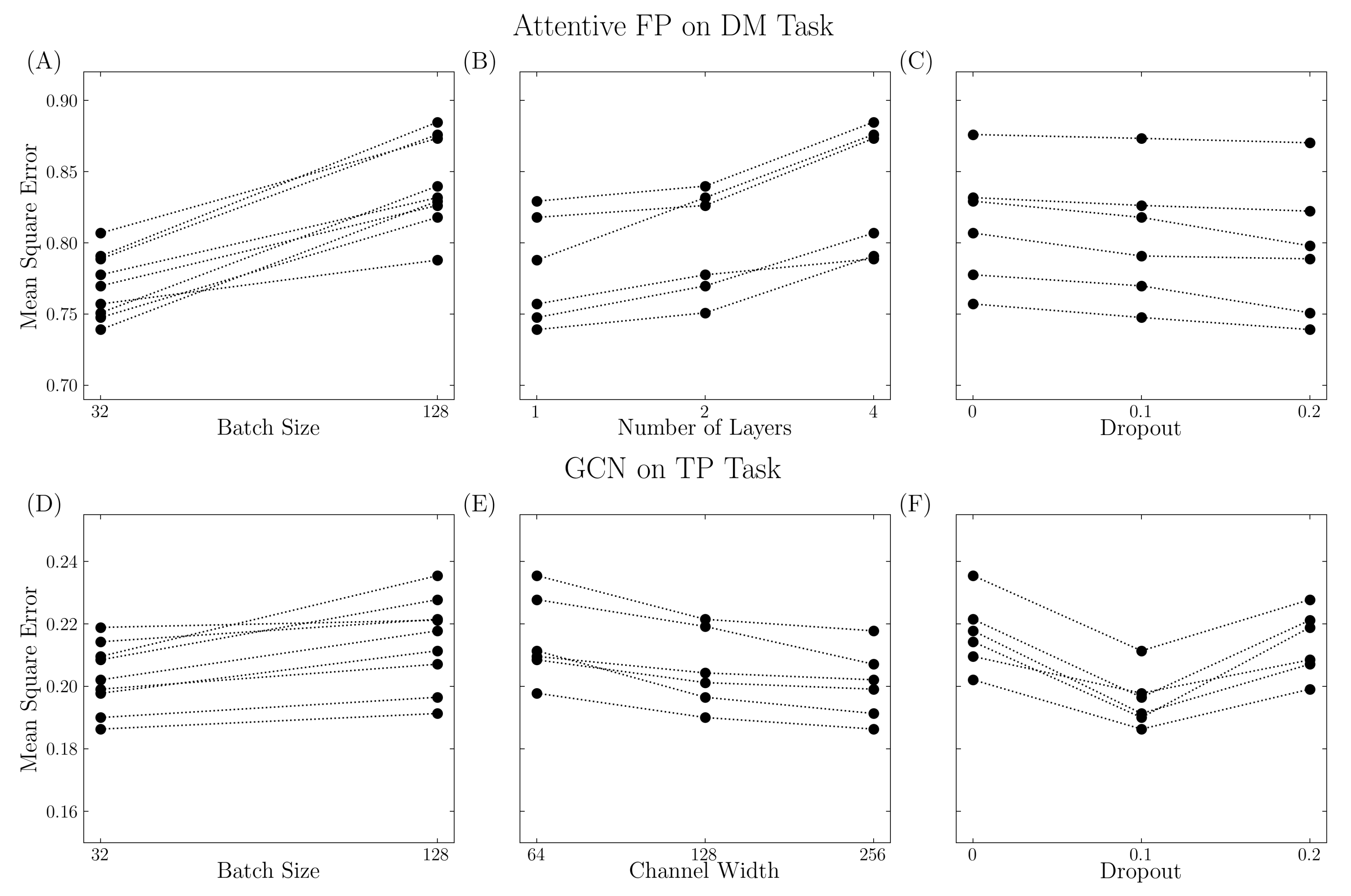
Appropriate parameters are needed to reach the best performance. As shown in Figure 3, model performance varies with different parameter settings. For example, in the Attentive FP model on the DM task, the model performed poorly with a large batch size, large number of layers, and low dropout rate. These indicate that increased model complexity might not always lead to better performance, and each model must be tuned appropriately. For the GCN model on the TP task, the model performed better with a smaller batch size, large channel width, and suitable dropout rate. The V shape in the dropout (Figure 3F) implies that a reasonable dropout rate is required to overcome overfitting, while a large dropout rate might lead to insufficient training. In this work, a grid search cross-validation method was used to explore the hyperparameter space. However, it is not possible to explore all possible configurations, and the grid search method does not necessarily lead to the optimum set. A Bayesian optimization approach, as implemented in the hyperopt package [20], may prove useful for future work.
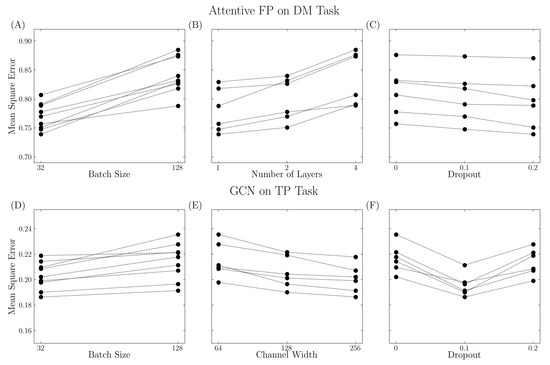
Figure 3.
Various model performance with different parameter settings. The performance was quantified using mean square error. (A–C) Attentive FP model trained on DM task. (D–F) GCN model trained on TP task. Three factors were considered such as sample size (batch size parameter), model complexity (number of layers for Attentive FP model and channel width for GCN model), and regularization (dropout parameter).
Sample size is an important factor for machine learning models. As reported in this study, graph models reached the best and worst performance in the TP and DM tasks, respectively. This aligns with the comparison of the training size, where the TP task includes 2035 molecules while the DM task has only 753 molecules. The VF and fish tasks have similar sample sizes of 1271 and 956, and thus have comparable performance. Therefore, it is reasonable to conclude that a larger dataset would lead to better performance. In addition, the training datasets in this work lacked a significant number of nontoxic molecules. Real-world applications may not reflect this data distribution, so these models may be limited in such environments and should be used with caution where a large number of molecules have low or zero toxicity. Future research may investigate graph model performance on a dataset curated with regard to real-life scenarios.
In the current study, each graph model was trained independently on four tasks, which does not take advantage of the fact that some tasks share common molecules. Transfer learning should be considered in future applications where a low-level embedding layer can be trained to learn common molecular structures with high-level task-specific layers.
It should be noted that the primary purpose of this study is to evaluate and compare the five selected graph models quantitatively instead of using model interpretation and molecular structural explanation. Attentive FP is highlighted to be unique in producing atomic weights from which a heatmap may be generated for further analysis, but the study is limited in analyzing the structural basis of toxicity.
4. Materials and Methods
4.1. Data Collection
The dataset was collected from Li et al. [21]. As reported, several publications and databases were utilized to compose a collection of 5048 toxicity data points for chemicals from fish, Daphnia magna (DM), Tetrahymena pyriformis (TP), and Vibrio fischeri (VF). Li et al. [22] was the primary source of data points, with additional toxicity values for TP taken from Ruusmann and Maran [23]. The dataset is freely available at https://github.com/hhaootian/toxicity/blob/main/data/toxicity.xlsx (accessed on 20 May 2023).
The fish toxicity values express median lethal concentration () after 96 h and were taken from data for the fathead minnow (Pimephales promelas), guppy (Poecilia reticulata), medaka (Oryzias latipes), and rainbow trout (Oncorhynchus mykiss). Prior work by Li et al., from which this dataset was partially curated, showed strong correlation of toxicity between the four species [22]. The DM data use or half maximal effective concentration () after 48 h. The TP toxicity data express the concentration required for 50% growth inhibition () after 40 or 48 h. Highly similar data were observed between the two timeframes. The VF values indicate 50% bioluminescence inhibition concentration () after either 15 or 30 min, with higher preference given to 30-minute data points. The average toxicity value was used for compounds with more than one value for a certain species. For all acute toxicity values, the negative logarithm of the molar concentration (mol/L) was taken.
For each entry, featurization techniques corresponding to each graph model were applied. A molecule is dropped if it fails to produce a proper featurization result. In total, 14 out of 2049 molecules were dropped in the TP task, 4 out of 757 molecules in the DM task, 6 out of 1277 molecules in the VF task, and 9 out of 965 molecules in the fish task. The featurization script to produce cleaned datasets is available at https://github.com/hhaootian/toxicity/blob/main/src/clean.py (accessed on 20 May 2023).
The GAT, GCN, MPNN, and Attentive FP models use MolGraphConvFeaturizer, v2.7.1 which creates graph convolutional representations of molecules. This method encodes the molecular structure, including atoms, bonds, and bond types, directly in the featurization result. This allows for more data-driven machine learning than with the use of traditional molecular fingerprints [24]. The PAGTN model uses PagtnMolGraphFeaturizer, v2.7.1. This also exploits molecular graph convolutions but specifically connects all atoms within the molecule while accounting for interactions between them relative to their distance [15]. These featurizers were implemented in DeepChem [25].
4.2. Graph Models
4.2.1. Message-Passing Neural Network
A message-passing neural network (Algorithm 1) is a type of graph neural network that operates on graph-structured data and incorporates message passing, which allows information to be exchanged between nodes in a graph. These nodes represent atoms, while edges between them represent bonds. MPNNs exploit this graph structure to learn interactions between the nodes. The neural network iteratively updates the hidden state of each node by first passing messages and then aggregating information from its neighboring nodes. The MPNN architecture has found success in a variety of applications, including molecular property prediction, due to its ability to process complex molecular graphs [13].
The MPNN architecture comprises three main steps: message passing, feature update among vertices, and readout. First, features are assigned to each atom () and bond (). Then, nodes send messages to their neighbors, and features are updated accordingly. Finally, a readout function creates graph-level features to be used in machine learning tasks.
The message-passing protocol consists of two main components: message passing along edges (Equation (1)) and vertex update (Equation (2)). The message function aggregates edge features with their incident nodes, while the update function updates each node’s hidden state based on the message received at iteration . Here, denotes the neighbors of v. In the final readout step, the function R combines the atom features to represent the whole graph in a vector (Equation (3)) [14].
| Algorithm 1 Message-Passing Neural Network. |
|
4.2.2. Graph Convolution Networks and Graph Attention Networks
Graph convolution networks (GCNs, Algorithm 2) are a form of neural network architecture designed to operate on graph structures using convolutions. They utilize a fixed convolutional operation based on traditional convolutional neural networks (CNNs), either in the spectral or spatial domain of the graph [26]. GCNs typically aggregate feature data from a node’s immediate neighbors to determine its hidden state, which is often used as its final representation. These neural networks can operate directly on molecular graphs, rather than using fingerprint vectors, in order to improve accuracy, efficiency, and interpretability [27].
Graph attention networks (GATs) combine a neural network with an attention mechanism that determines the relative importance of neighboring nodes. Unlike GCNs, which apply fixed convolutional operations, GATs compute attention coefficients for each neighbor and weigh their contributions when updating the hidden state. This hidden state is an aggregation of the weighted contributions of neighboring nodes, providing each node with a tailored representation based on its neighbors. This allows GATs to selectively emphasize different neighboring nodes based on their importance and focus on the most salient aspects of an input, which can also improve the architecture’s interpretability [28].
| Algorithm 2 Graph Convolution/Attention Networks. |
|
4.2.3. Path-Augmented Graph Transformer Network
PAGTNs (Algorithm 3) were introduced by Chen et al., in 2019 to solve the problem of GCNs failing to recognize long-range structural relationships in molecules [15]. This type of model utilizes the transformer architecture on graph-structured data. In comparison to other models like GCNs, which aggregate primarily local information on nodes, PAGTNs utilize a globally connected attention framework. This means that each node attends to all other nodes while still focusing on the most important ones. PAGTNs exploit path features to learn long-range relationships between nodes without needing several layers. These paths may provide valuable information on molecular substructures, which could explain the success of PAGTNs in molecular property prediction.
| Algorithm 3 Path-Augmented Graph Transformer Network. |
|
4.2.4. Attentive FP
The Attentive FP model (Algorithm 4) is a GNN architecture that represents molecules using molecular fingerprints and a graph attention mechanism. Rather than calculating molecular conformation or connecting all atoms with virtual edges, as in the MPNN model, Attentive FP prioritizes interactions between nearby atoms while still considering the effects of distant ones, beyond a node’s immediate neighbors. This is made possible by the attention mechanism, which assigns greater weights to more important substructures in order to focus on the most relevant parts of a molecule. This property of Attentive FP can be leveraged to interpret the model, which is key for comparing it to existing domain knowledge and potentially gaining insight into novel chemical patterns [5].
| Algorithm 4 Attentive FP. |
|
Graph models were implemented in Python (v3.8.11) DeepChem [25] (v2.7.1), PyTorch (v2.0.0), and DGL (v1.1.0) packages.
4.3. Performance Metrics
Three metrics were calculated to quantify model performance on each regression task. With n pairs of true value and predicted value , mean square error (MSE), mean absolute percent error (MAPE), and R-squared (R2) are calculated as follows:
5. Conclusions
The development of graph models provides an alternative method to QSTR models for acute toxicity prediction. In this study, a pipeline was provided to compare different graph models for this purpose. Performance metrics were evaluated and key differences were reported among five popular graph models, namely, MPNNs, GCNs, GATs, PAGTNs, and Attentive FP. As reported, the Attentive FP model performed the best with a low prediction error, a high coefficient of determination R2, and good interpretability. The importance of parameter fine-tuning and sample size was discussed, along with the potential application of transfer learning.
Author Contributions
Conceptualization, R.K. and H.T.; methodology, R.K. and H.T.; software, H.T. and Y.L.; validation, H.T. and Y.L.; formal analysis, H.T.; data curation, H.T.; writing—original draft preparation, R.K., H.T., H.W. and Y.L.; writing—review and editing, R.K., H.T., H.W. and Y.L.; visualization, H.T.; supervision, H.T.; project administration, H.T. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The data and codes to reproduce the results are available at https://github.com/hhaootian/toxicity (accessed on 20 May 2023).
Acknowledgments
The authors thank Xi Jiang from the Biostatistics Ph.D. program in the Statistics department of Southern Methodist University for fruitful discussion.
Conflicts of Interest
The authors declare no conflict of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| AOT | Acute oral toxicity |
| ADMET | Absorption, Distribution, Metabolism, Excretion, and Toxicity |
| CNN | Convolutional neural network |
| DM | Daphnia magna |
| Half maximal effective concentration | |
| GAT | Graph attention network |
| GCN | Graph convolution network |
| GRU | Gated recurrent unit |
| 50% bioluminescence inhibition concentration | |
| 50% growth inhibition concentration | |
| Median lethal concentration | |
| MAPE | Mean absolute percent error |
| MLP | Multilayer perceptron |
| MPNN | Message-passing neural network |
| MSE | Mean squared error |
| PAGTN | Path-augmented graph transformer network |
| QSTR | Quantitative structure–toxicity relationship |
| TP | Tetrahymena pyriformis |
| VF | Vibrio fischeri |
References
- Mo, L.; Yuan, B.; Zhu, J.; Qin, L.; Dai, J. QSAR models for predicting additive and synergistic toxicities of binary pesticide mixtures on Scenedesmus obliquus. Chin. J. Struct. Chem. 2022, 41, 2203166–2203177. [Google Scholar]
- Zhang, S.; Wang, N.; Su, L.; Xu, X.; Li, C.; Qin, W.; Zhao, Y. MOA-based linear and nonlinear QSAR models for predicting the toxicity of organic chemicals to Vibrio fischeri. Environ. Sci. Pollut. Res. 2020, 27, 9114–9125. [Google Scholar] [CrossRef] [PubMed]
- Barron, M.; Lilavois, C.; Martin, T. MOAtox: A comprehensive mode of action and acute aquatic toxicity database for predictive model development. Aquat. Toxicol. 2015, 161, 102–107. [Google Scholar] [CrossRef] [PubMed]
- Sun, D.; Gao, W.; Hu, H.; Zhou, S. Why 90% of clinical drug development fails and how to improve it? Acta Pharm. Sin. 2022, 12, 3049–3062. [Google Scholar] [CrossRef] [PubMed]
- Xiong, Z.; Wang, D.; Liu, X.; Zhong, F.; Wan, X.; Li, X.; Li, Z.; Luo, X.; Chen, K.; Jiang, H. Others Pushing the boundaries of molecular representation for drug discovery with the graph attention mechanism. J. Med. Chem. 2019, 63, 8749–8760. [Google Scholar] [CrossRef] [PubMed]
- Tian, H.; Ketkar, R.; Tao, P. ADMETboost: A web server for accurate ADMET prediction. J. Mol. Model. 2022, 28, 408. [Google Scholar] [CrossRef]
- Lagunin, A.; Zakharov, A.; Filimonov, D.; Poroikov, V. QSAR modelling of rat acute toxicity on the basis of PASS prediction. Mol. Inform. 2011, 30, 241–250. [Google Scholar] [CrossRef]
- Abramenko, N.; Kustov, L.; Metelytsia, L.; Kovalishyn, V.; Tetko, I.; Peijnenburg, W. A review of recent advances towards the development of QSAR models for toxicity assessment of ionic liquids. J. Hazard. Mater. 2020, 384, 121429. [Google Scholar] [CrossRef]
- Cavasotto, C.; Scardino, V. Machine learning toxicity prediction: Latest advances by toxicity end point. ACS Omega 2022, 7, 47536–47546. [Google Scholar] [CrossRef]
- Xu, Y.; Pei, J.; Lai, L. Deep learning based regression and multiclass models for acute oral toxicity prediction with automatic chemical feature extraction. J. Chem. Inf. Model. 2017, 57, 2672–2685. [Google Scholar] [CrossRef]
- Rücker, C.; Rücker, G.; Meringer, M. Y-randomization–a useful tool in QSAR validation, or folklore. J. Chem. Inf. Model 2007, 47, 2345–2357. [Google Scholar] [CrossRef]
- Tian, H.; Xiao, S.; Jiang, X.; Tao, P. PASSer: Fast and accurate prediction of protein allosteric sites. Nucleic Acids Res. 2023, 541, gkad303. [Google Scholar] [CrossRef] [PubMed]
- Tang, M.; Li, B.; Chen, H. Application of message passing neural networks for molecular property prediction. Curr. Opin. Struct. Biol. 2023, 81, 102616. [Google Scholar] [CrossRef]
- Gilmer, J.; Schoenholz, S.; Riley, P.; Vinyals, O.; Dahl, G. Neural message passing for quantum chemistry. Int. Conf. Mach. Learn. 2017, 70, 1263–1272. [Google Scholar]
- Chen, B.; Barzilay, R.; Jaakkola, T. Path-augmented graph transformer network. arXiv 2019, arXiv:1905.12712. [Google Scholar]
- Mayr, A.; Klambauer, G.; Unterthiner, T.; Hochreiter, S. DeepTox: Toxicity prediction using deep learning. Front. Environ. Sci. 2016, 3, 80. [Google Scholar] [CrossRef]
- Jiang, J.; Wang, R.; Wei, G. GGL-Tox: Geometric graph learning for toxicity prediction. J. Chem. Inf. Model. 2021, 61, 1691–1700. [Google Scholar] [CrossRef]
- Wu, K.; Wei, G. Quantitative toxicity prediction using topology based multitask deep neural networks. J. Chem. Inf. Model. 2018, 58, 520–531. [Google Scholar] [CrossRef]
- Polishchuk, P. Interpretation of Quantitative Structure—Activity Relationship Models: Past, Present, and Future. J. Chem. Inf. Model. 2017, 57, 2618–2639. [Google Scholar] [CrossRef]
- Bergstra, J.; Yamins, D.; Cox, D. Making a Science of Model Search: Hyperparameter Optimization in Hundreds of Dimensions for Vision Architectures. In Proceedings of the 30th International Conference on Machine Learning, Atlanta, GA, USA, 17–19 June 2013; pp. 115–123. [Google Scholar]
- Li, J.; Zhang, X.; Yang, Y.; Huang, T.; Li, C.; Su, L.; Zhao, Y.; Cronin, M. Development of thresholds of excess toxicity for environmental species and their application to identification of modes of acute toxic action. Sci. Total. Environ. 2018, 616, 491–499. [Google Scholar] [CrossRef]
- Li, J.; Wang, X.; Wang, Y.; Wen, Y.; Qin, W.; Su, L.; Zhao, Y. Discrimination of excess toxicity from narcotic effect: Influence of species sensitivity and bioconcentration on the classification of modes of action. Chemosphere 2015, 120, 660–673. [Google Scholar] [CrossRef] [PubMed]
- Ruusmann, V.; Maran, U. From data point timelines to a well curated data set, data mining of experimental data and chemical structure data from scientific articles, problems and possible solutions. J. Comput. Aided Mol. Des. 2013, 27, 583–603. [Google Scholar] [CrossRef]
- Kearnes, S.; McCloskey, K.; Berndl, M.; Pande, V.; Riley, P. Molecular graph convolutions: Moving beyond fingerprints. J. Comput. Aided Mol. Des. 2016, 30, 595–608. [Google Scholar] [CrossRef] [PubMed]
- Ramsundar, B.; Eastman, P.; Walters, P.; Pande, V.; Leswing, K.; Wu, Z. Deep Learning for the Life Sciences; O’Reilly Media: Sebastopol, CA, USA, 2019. [Google Scholar]
- Zhang, S.; Tong, H.; Xu, J.; Maciejewski, R. Graph Convolutional Networks: A comprehensive review. Comput. Soc. Netw. 2019, 6, 11. [Google Scholar] [CrossRef]
- Duvenaud, D.; Maclaurin, D.; Iparraguirre, J.; Bombarell, R.; Hirzel, T.; Aspuru-Guzik, A.; Adams, R. Convolutional networks on graphs for learning molecular fingerprints. Adv. Neural Inf. Process. Syst. 2015, 28, 1–9. [Google Scholar]
- Veličković, P.; Cucurull, G.; Casanova, A.; Romero, A.; Liò, P.; Bengio, Y. Graph Attention Networks. arXiv 2018, arXiv:1710.10903. [Google Scholar]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).