1. Introduction
Cancer is undoubtedly a potentially life-threatening disease for humanity that can greatly affect the quality of human life [
1,
2,
3,
4,
5]. The numerous side effects of cisplatin such as nausea, kidney and liver failure (typical of heavy metal toxicity) have induced the search for better alternative candidates to non-platinum metal complexes such as copper, cobalt, nickel, zinc, ruthenium, and iron [
6,
7,
8,
9,
10]. Copper is the third most abundant metal, behind iron and zinc, and is found in the body in trace amounts. The total amount of copper in the human body is only 75–100 mg, but is present in every tissue of the body. It is primarily stored in the liver, with smaller amounts found in the brain, heart, kidneys, and muscles [
7]. As an essential trace element, copper plays a very important role in many physiological cellular processes [
8,
9]. Metal complexes of the Schiff base ligands (coordinated via ONS and NO donor atoms) with copper(II) ions are interesting due to their structural, spectral, and redox properties [
10]. Complexes containing a sulfur atom can be expected to possess great potential for medical applications because such molecules may mimic the functional properties of sulfur-containing proteins. Sulfur is well-known to be an important component of biomolecules and it plays a key role in biological systems [
11]. Cytotoxic activity is primarily mediated by targeting DNA and proteins. Interactions with small molecules leads to DNA damage in cancer cells by blocking uncontrolled division, resulting in cell death [
12,
13]. Many inorganic compounds have been successfully used as drugs in the treatment of different types of cancers. Copper(II) complexes have been considered and suggested to be better candidates for cancer treatment due to their significant role in biological systems, having negligible or no side effects [
14,
15]. Mononuclear copper(II) complexes with the phenanthroline type of ligand show lesser affinities to DNA binding compared to binuclear copper(II) complexes [
16,
17]. In angiogenesis, copper appears to be involved in stimulating proliferation and endothelial cell migration, and acts as a cofactor for angiogenic factors (VEGF, bFGF, TNF-α, and IL-1) [
18,
19,
20,
21]. However, in embryogenic cells, the human copper transporter (hCTR1) inhibits the activation of cellular signaling pathways, resulting in the development and progression of cancer [
22]. The difference in the tumor cells’ response to copper compared to normal cells probably laid the basis for the development of anticancer drugs [
15,
23].
The aim of the present study was to research the interactions of copper(II)complexes with S-isoalkyl derivatives of thiosalicylic acid (isoalkyl = isopropyl-, isobutenyl-, and isopentyl) marked as
C1–
C3 with guanosine-5′-monophosphate (5′-GMP) and calf thymus DNA (CT-DNA) as well as the evaluation of their in vitro and in vivo antitumor activity in a mouse colon cancer model. In this article, we present a docking study of binuclear copper(II) complexes with S-isoalkyl derivatives of thiosalicylic acid. Previously, we showed the synthesis, characterization, and cytotoxic effect of newly synthetized copper(II) complexes (
C1–
C3) with isoalkyl derivatives of thiosalicylic acid as ligands (
L1–
L3). Their chemical structures are presented in
Scheme 1. These complexes showed a lower cytotoxic effect than cisplatin toward normal fibroblasts as evaluated by the MTT assay as well as significant apoptotic effects against HCT116 cells. We also found antiproliferative effects of
C1, a copper(II)complex with an isopropyl derivative of thiosalicylic acid toward HCT116 cells [
24]. All complexes induced the arrest of HCT116 cells in the G2 phase of the cell cycle [
24]. Taking into account the well-known anti-inflammatory activity of derivatives of salicylates [
25] and the involvement of inflammatory pathways in cancerogenesis and the progression of cancer [
26], the anticancer properties of transition metal complexes with different derivatives of salicylates could be considered to have higher in vivo effects in comparison with their effects in cell culture systems. By reducing inflammation, salicylates are known to reduce the risk of large bowel cancer development [
27]. Hence, in this study, we analyzed the antitumor effects of copper(II) complexes with isoalkyl derivatives of thiosalicylic acid against murine colon carcinoma cells, CT26, and we also explored the in vivo antitumor effect of the most active complex in a murine heterotopic model of colon carcinoma.
3. Discussion
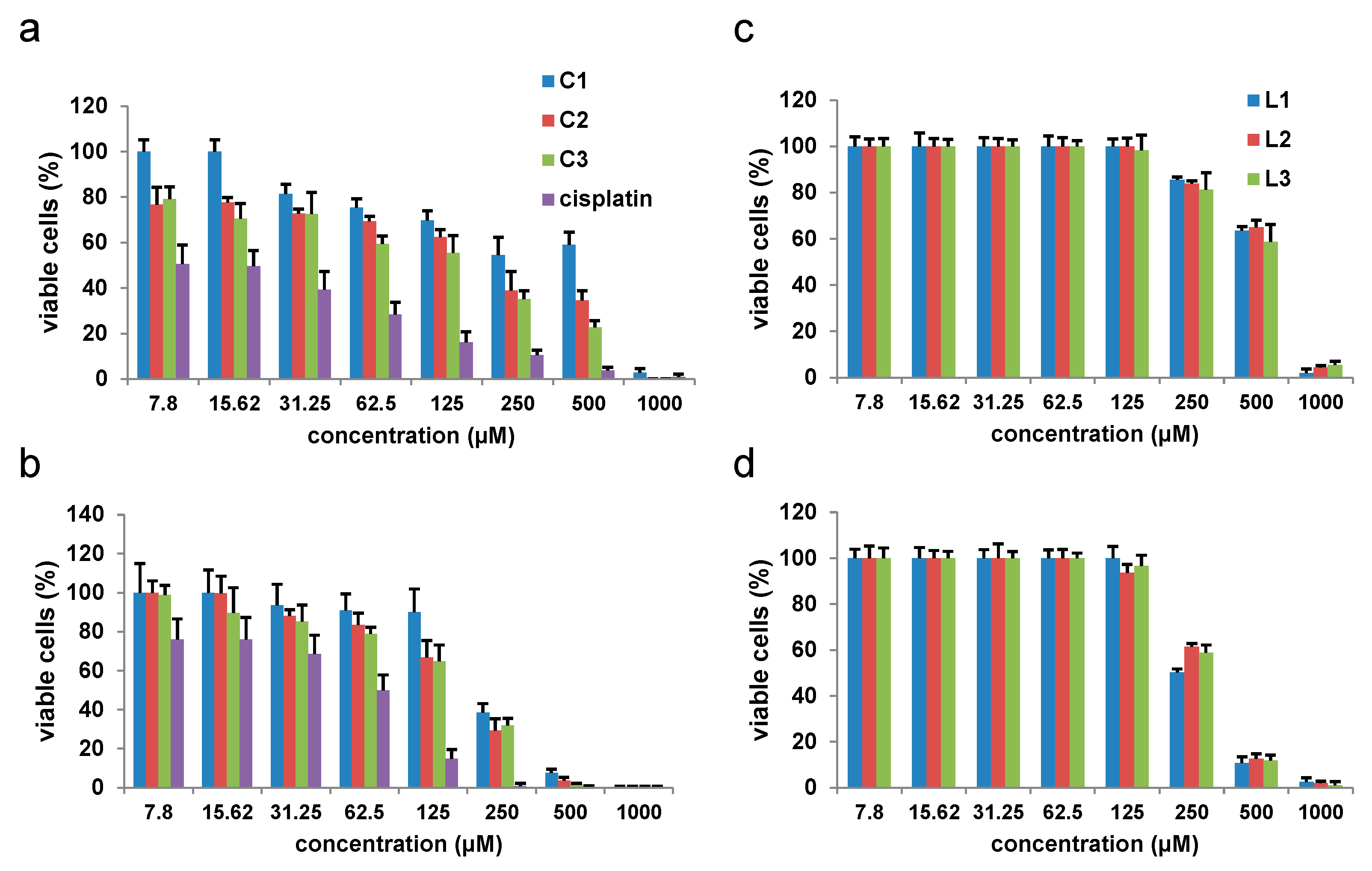
Significant progress is being made in the development of drugs to combat previously incurable forms of cancer. One approach involves targeting and disrupting the structure of DNA to inhibit replication and transcription. In this study, we focused on Cu(II) complexes containing S-isoalkyl derivatives of salicylic acid (referred to as C1, C2, and C3) for this purpose. Through a combination of experimental and theoretical methods, we investigated how these complexes interacted with DNA.
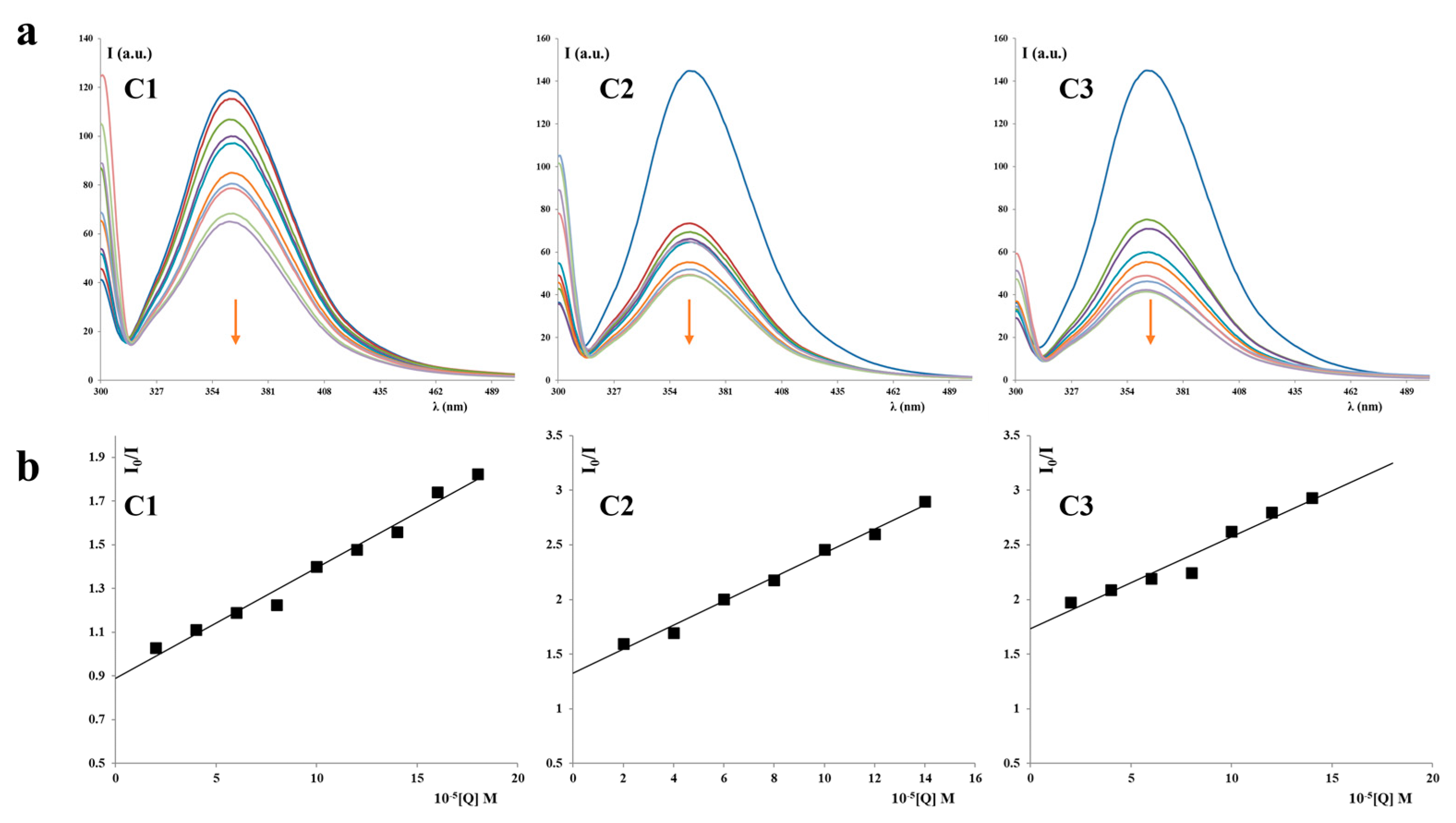
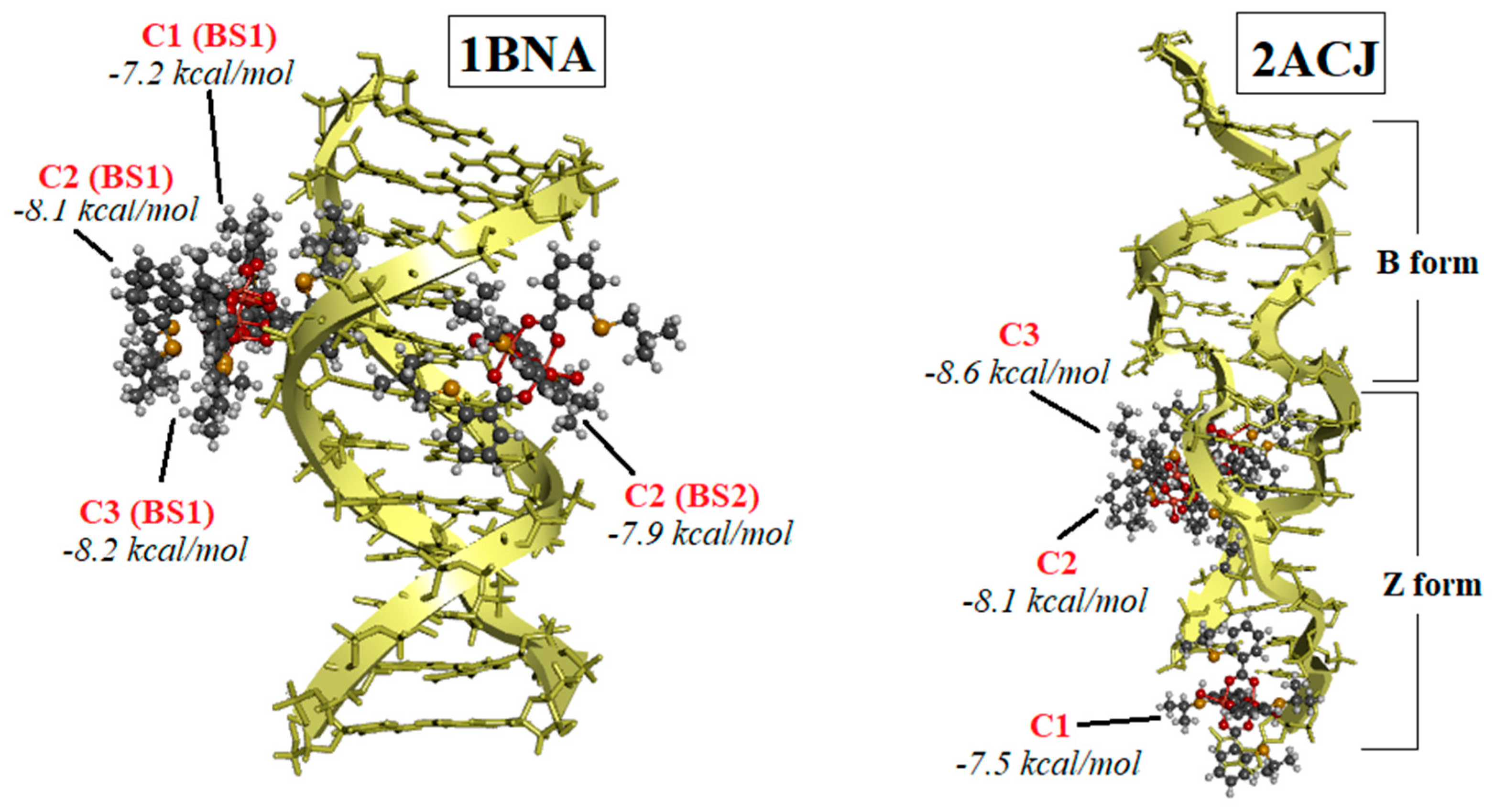
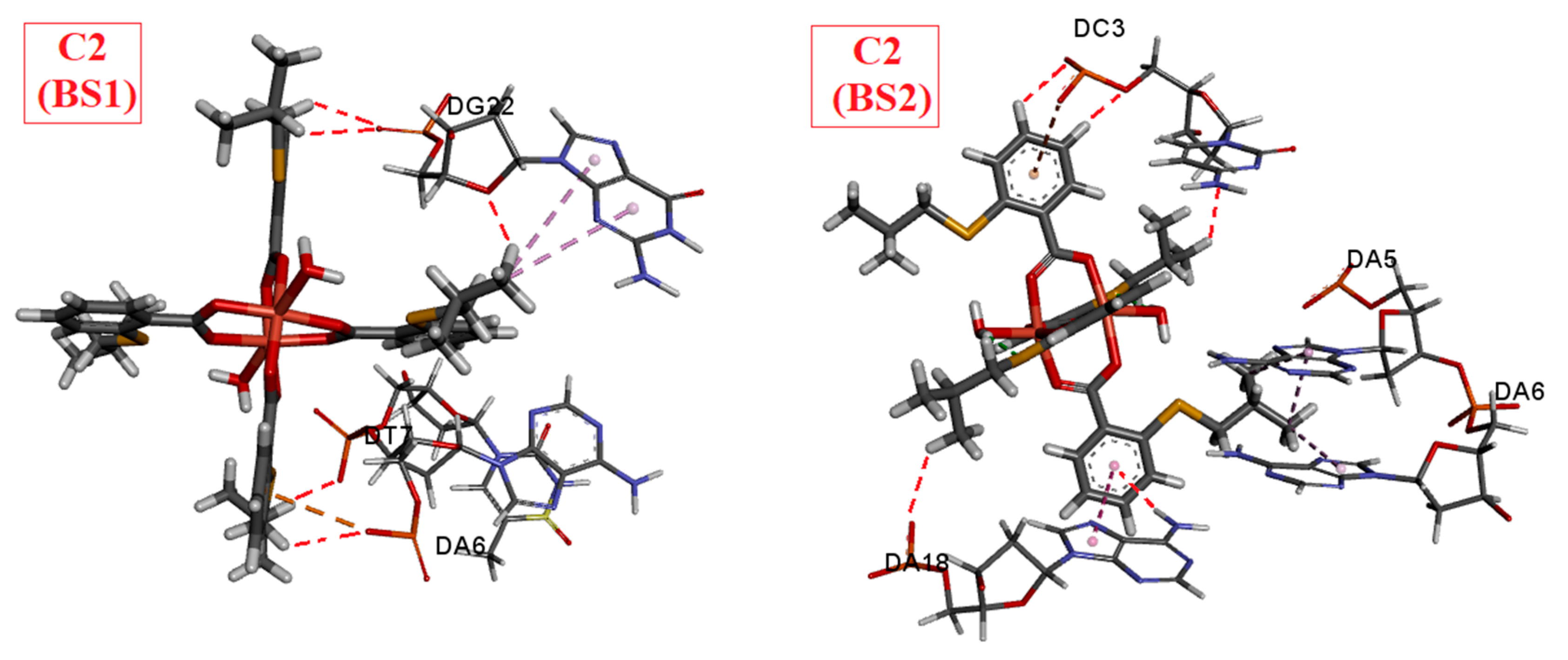
Our experiments using ethidium bromide (EB) revealed that the complexes did not displace EB, indicating a lack of intercalation. Molecular docking studies provided insights into the binding preferences of the complexes, suggesting that they may bind in the major groove to the sugar–phosphate backbone, or, most favorably, in the minor groove. Viscosity experiments showed a modest increase in the viscosity of DNA solutions upon the addition of the C1 or C3 complexes, indicating some interaction. Each complex exhibited a single binding site in the minor groove. However, the viscosity strikingly increased when DNA was mixed with the C2 complex, which was bound at two distinct binding sites—first in the minor groove, and the second position in the major groove. The presence of two bulky C2 complexes of Cu(II) induced the transformation of B-DNA into Z-DNA. The binding energies calculated for all three Cu(II) complexes to Z-DNA were notably higher compared to B-DNA. Z-DNA had an elongated structure compared to B-DNA, which explains the significant increase in viscosity observed when DNA interacted with two C2 complexes simultaneously.
In this study, it was shown that the binuclear complex of copper(II) with the S-isoamyl derivative of thiosalicylic acid, C3, arrests cancer cells in the G2/M phase, achieves a cytotoxic effect in murine colon cancer cells, and significantly reduces the expression of inflammatory cytokines in the tissue of primary CT26 mouse colon cancer, resulting in a significant reduction in primary tumor growth and metastasis.
Copper(II) complexes with alkyl derivatives of thiosalicylic acid were previously synthesized and characterized [
53] and their antitumor activity was analyzed [
24]. These copper complexes were shown to have a weak cytotoxic effect on the murine colon cancer cells CT26 and CT26. CL25, and human colorectal cancer cells HCT116 [
24]. Palladium complexes with the same ligands showed a better antitumor effect on human colorectal cancer cells, HCT116 and CaCo-2, and human lung adenocarcinoma, A549, compared with the same copper complexes [
53,
54,
55]. Zn(II) complexes with the same ligands showed relatively moderate cytotoxicity on murine, 4T1, and human, MDA-MB-468, breast carcinoma cells [
56]. Platinum(IV) complexes with the same ligands showed significant cytotoxicity on murine leukemia cell, BCL1, and a moderate to weak effect on human B-prolymphocytic leukemia cell, JVM-13 [
57]. The dinuclear complex 3 investigated in this study showed a better cytotoxic effect on murine colon carcinoma cells compared to similar mononuclear complexes [
24]. In this study, the
C3 complex also showed a better cytotoxic effect toward the murine colon cancer line CT26 than on the human colorectal cancer cell line SW480, in contrast to the study showing that the Cu(II) complex with goserelin acetate exhibited better cytotoxic effect on A549 human lung adenocarcinoma cells than on the CT26 murine colon carcinoma cell line [
58]. The different effect of dinuclear copper complexes with S-isoalkyl derivatives of thiosalicylic acid on different tumor cells is consistent with the results of a recent study claiming that Cu(II) complexes with derivatives of Schiff’s bases exert different effects on colorectal cancer cells DLD-1 and human breast cancer cells MDA-MB-231 [
59]. The in vitro cytotoxicity of the copper complexes examined in this study was weaker compared to the similarly examined cytotoxicity of the Cu(II) complexes with pyrrolysines [
60]. Similar to the results of this study, a moderate cytotoxic effect of Cu(II) complexes with tridentate Schiff bases with different halogens was demonstrated on HCT116 human colorectal cancer cells, while the same complexes showed a very pronounced effect on human ovarian cancer cells [
61]. Very good cytotoxic effects of Cu(II) complexes with salicylic acid derivatives on HT29 human colorectal cancer cells were described [
62].
Compounds exhibiting antitumor activity often achieve this activity by interacting with different molecules such as DNA, proteins, and parts of the cell membrane [
63]. The dinuclear copper complexes examined in this study had relatively weak interactions with human serum albumin and the DNA molecule, and the strongest interactions with these two molecules were achieved by complex
C2 (
Table 1 and
Table 2). As the best cytotoxicity in vitro was achieved by complex
C3, it is most likely that interaction with DNA contributes little to the antitumor activity of dinuclear Cu(II) complexes with S-isoalkyl derivatives of thiosalicylic acid.
Copper(II) complexes with alkyl derivatives of thiosalicylic acid exert a moderate apoptotic effect on murine and human colorectal cancer cells [
24]. In accordance with that finding, there was a relatively weak apoptotic effect of dinuclear complexes of copper(II) with S-isoalkyl derivatives of thiosalicylic acid on CT26 cells (
Figure 9). Good apoptotic activity of various Cu(II) on human colorectal cancer cell lines has been previously described [
64,
65,
66,
67]. Similar to the results of this study, Cu(I) thiocyanate complexes with phosphine derivatives of sparfloxacin exert a mild to moderate apoptotic effect on the CT26 cells [
68]. Chromosome instability, which precedes in 60–80% of colorectal cancers, very often affects genes whose products participate in the control of apoptosis. The p53 gene is mutated in about 70% of colorectal cancers [
69]. The p53 protein participates in the regulation of the cell cycle, DNA repair mechanisms, and apoptosis; which of these processes is the result of p53 activation, depends on the cell type and microenvironment [
70]. The p53 mutation in HCT116 human colorectal cancer cells makes these cells more sensitive to radiotherapy and adriamycin, and less sensitive to 5-fluorouracil [
71]. It is possible that the mutations in CT26 cells make these cells relatively resistant to apoptosis induced by dinuclear complexes of copper(II) with alkyl derivatives of thiosalicylic acid, which does not exclude other mechanisms of antitumor activity.
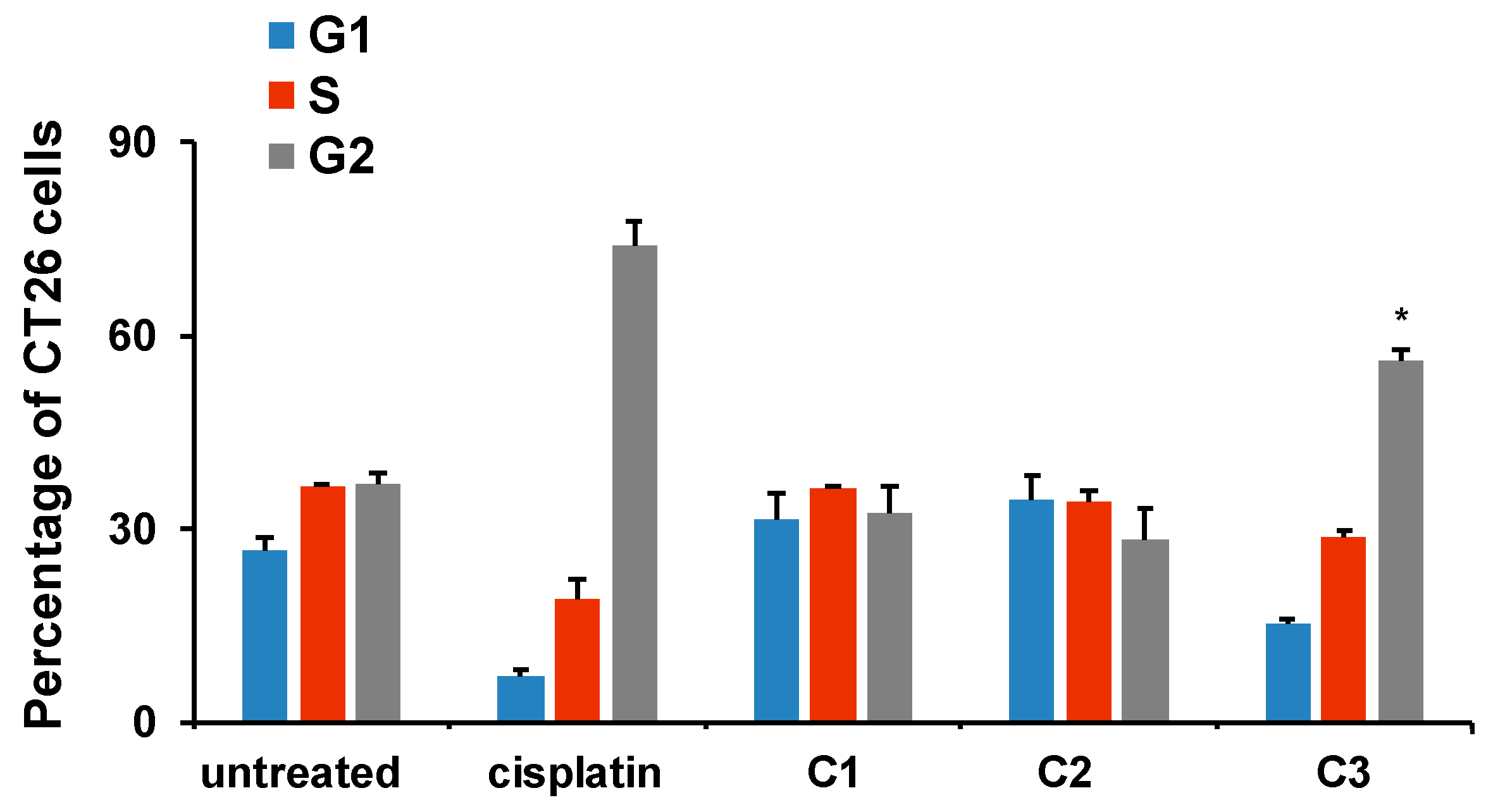
Cells undergoing mitotic catastrophe are in the G2/M phase of the cell cycle [
72]. Mitotic catastrophe is a disrupted mitosis resulting in cell death, which differs in mechanism from apoptotic or necrotic death. The
C3 complex, which exhibited the strongest cytotoxic activity on all examined tumor cell lines (
Table 3), also caused the accumulation of CT26 cells in the G2/M phase of the cell cycle (
Figure 10). Thus, although
C3 induced no significant apoptosis of CT26 cells, its greatest potential to reduce CT26 viability, assessed by the MTT assay, could be explained by the arrest of CT26 cells in the G2/M phase of the cell cycle under the action of
C3 and the reduction in CT26 viability by other mechanisms (such as mitotic catastrophe) [
73]. A similar effect of the induction of mitotic catastrophe and subsequent cell death of human non-small cell lung carcinoma, H-460, was described for the ternary complex of Cu(II) with L-tyrosine and diimines [
74].
Numerous studies have shown that aspirin (acetylsalicylic acid) reduced the rate of colorectal adenoma recurrence, advanced adenoma, the number of recurrent adenomas, and delayed the time to adenoma recurrence [
75,
76,
77,
78]. It also reduced the risk of developing colorectal cancer [
79,
80]. Aspirin acts as an inhibitor of cyclooxygenase-2. It reduces the production of PGE2 and reduces inflammation, therefore reducing the risk of the development and progression of colorectal cancer [
81]. Thiosalicylic acid, like aspirin, belongs to the group of non-steroidal anti-inflammatory drugs, so it is possible that metal complexes with derivatives of this acid such as the dinuclear Cu(II) complex with the S-isoamyl derivative of thiosalicylic acid achieve an enhanced antitumor effect in vivo in colon cancer by acting directly on tumor cells, but also by acting indirectly by inhibiting inflammation.
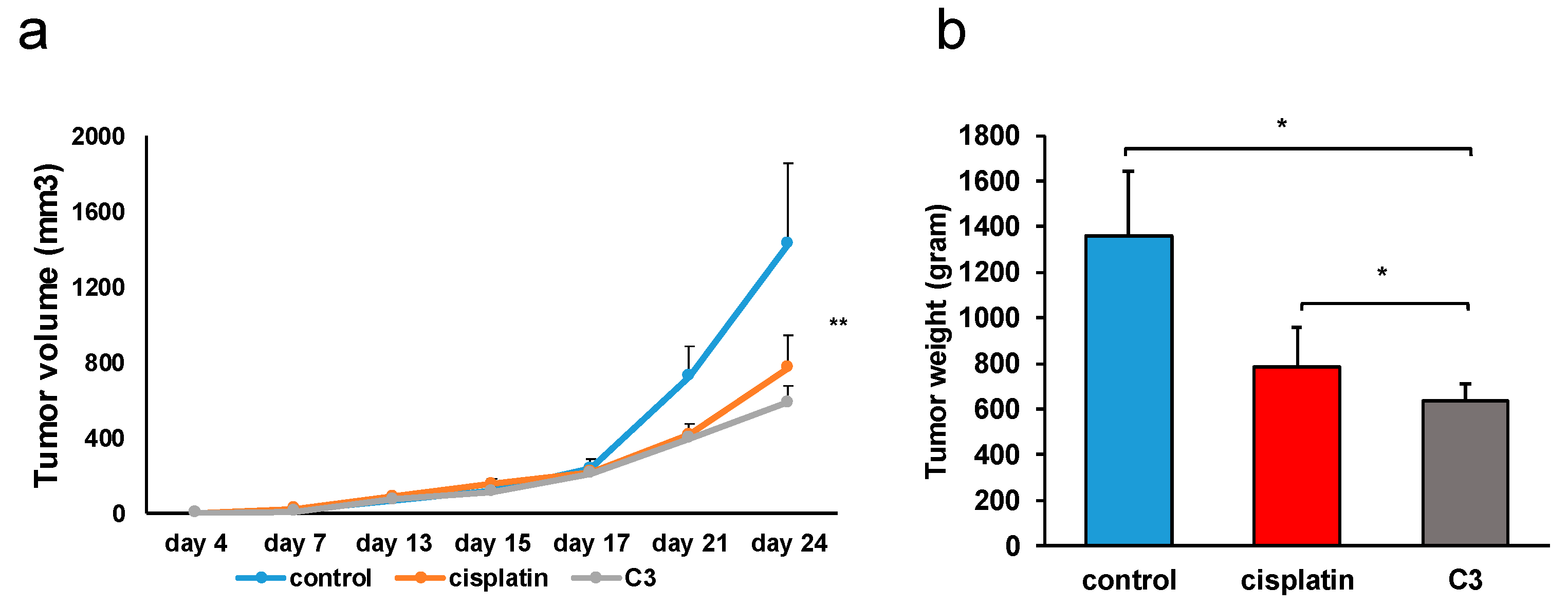
C3 significantly reduced the growth of the primary heterotopic CT26 mouse carcinoma compared to untreated mice (
Figure 11). An even more significant result is the significantly lower weight of the excised primary tumor in the group of mice treated with complex
C3 compared to the group of mice treated with cisplatin (
Figure 11). The treatment of mice with the
C3 complex was accompanied by a lower incidence of metastases (
Table 5) in the lungs and liver and a smaller size of metastatic changes (
Figure 13 and
Figure 14), which is in accordance with the strongest cytotoxic activity of
C3 on CT26 cells, as evaluated by MTT. In the tissue of the primary tumor of mice treated with the
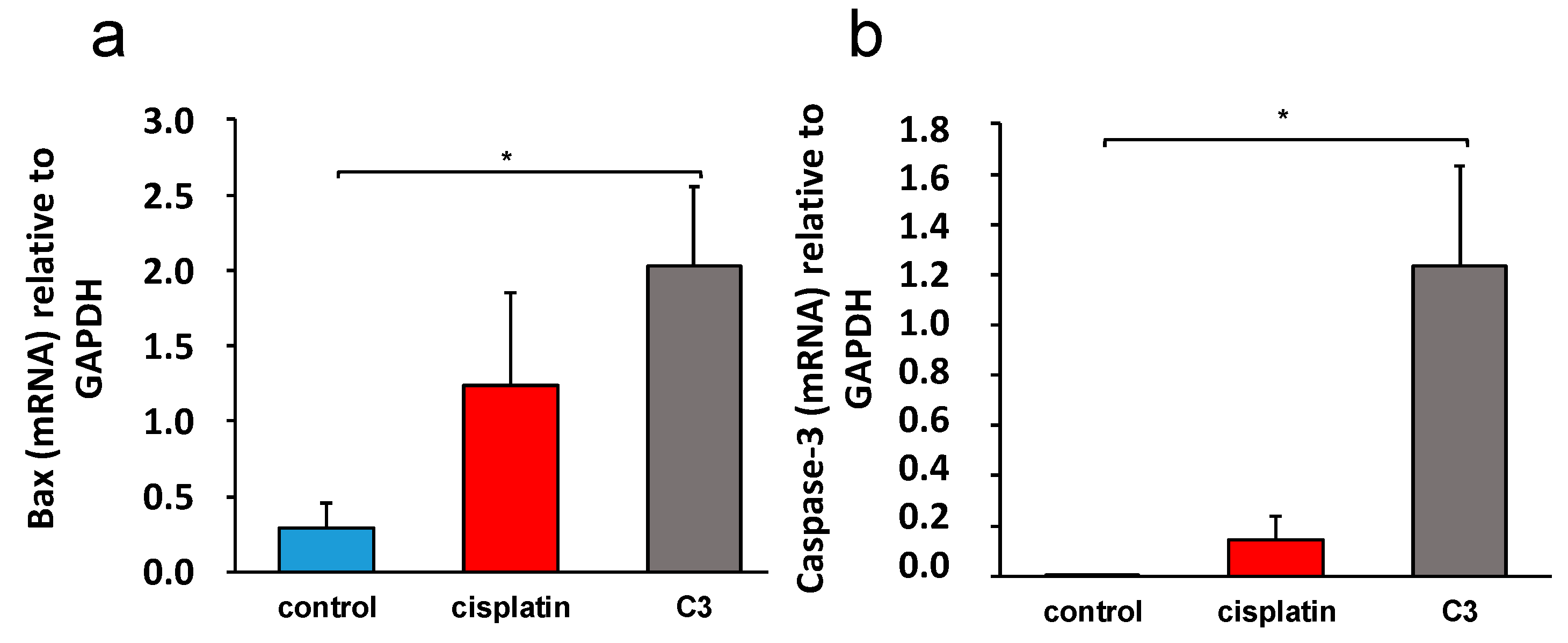
C3 complex, a significantly higher expression of mRNA molecules that are crucial for the process of apoptosis, Bax and caspase-3, was recorded (
Figure 12), and a significantly lower expression of the proinflammatory molecules TNF-α, pro-IL-β, and ICAM-1 and VCAM-1 (
Figure 15) were detected compared to the tissue from untreated mice.
Blocking IL-1β in a murine model of colitis and associated colon cancer significantly reduces the size and invasiveness of tumors [
82,
83], which indicates a significant role of this cytokine in the development and progression of colorectal cancer. TNF-α promotes the migration and invasiveness of colorectal cancer [
84] and participates in colorectal carcinogenesis associated with ulcerative colitis [
85]. VCAM-1 is a molecule that is expressed in colorectal cancer; it is associated with the presence of CD3+ lymphocytes [
86] and participates in tumor infiltration by immune cells. ICAM-1 is a molecule whose expression is controlled by the cytokines TNF-α and IL-β, which plays an important role in colorectal pathogenesis, participating in mutual interactions between the tumor and extracellular matrix in signal transduction and numerous immune processes [
87]. Colorectal cancer cells, in contrast to normal intestinal epithelial cells, express ICAM-1, which, through interaction with ligands, promotes tumor metastasis [
88,
89]. The expression of ICAM-1 determines the malignant potential of colorectal cancer; it is correlated with a worse prognosis [
90,
91], although there are also studies indicating that the membrane expression of this molecule correlates with better prognosis of colorectal cancer [
92], which is explained by the possibility that ICAM-1 expression on the membrane of tumor cells stimulates the killing of tumor cells by cytotoxic lymphocytes, if they are present in the tumor microenvironment. Soluble ICAM-1 promotes angiogenesis and the growth of colorectal cancer [
93]. Greater expression of ICAM-1 molecules in tumor tissue can also result in enhanced tumor cell apoptosis [
94].
The binuclear copper(II) complex with the S-isoamyl derivative of thiosalicylic acid achieved a very convincing antitumor effect in vivo in a mouse colon cancer model, significantly reducing the growth of the primary tumor and the incidence and size of metastases in the lungs and liver. As the in vitro antitumor effect of this complex is moderate, it is possible that the reduced growth and metastasis of murine colon cancer is a consequence of the modulation of the tumor microenvironment by the tested complex. Bearing in mind the importance of inflammation for the formation and progression of colorectal cancer [
81,
82,
83,
84,
85], it is possible that
C3, by influencing the expression of inflammatory molecules TNF-α, pro-IL-β, ICAM-1, and VCAM-1 in the tissue of the primary tumor, reduces tumor growth and progression, and also stimulates the apoptosis of tumor cells in vivo.