Incorporating Monoclonal Antibodies into the First-Line Treatment of Classical Hodgkin Lymphoma
Abstract
1. Introduction
1.1. The Journey from Pathobiology to Novel Therapeutic Approaches
1.2. Treatment of Hodgkin Lymphoma in the Past Millennium: Lights and Shades
2. Randomized Trials Incorporating Novel Agents in the First-Line Therapy of Advanced-Stage Classical Hodgkin Lymphoma
2.1. The BV-AVD Combination: Incorporating Brentuximab Vedotin to ABVD
2.2. Novel Combinations with Brentuximab Vedotin and Checkpoint Inhibitors: Moving beyond BV-AVD
3. Brentuximab Vedotin in the First-Line Therapy of Early-Stage Classical Hodgkin Lymphoma
4. Checkpoint Inhibitors in the First-Line Therapy of Classical Hodgkin Lymphoma
5. Brentuximab Vedotin and Checkpoint Inhibitor Combinations in the First-Line Treatment of Classical Hodgkin Lymphoma
6. Novel Agents in the First-Line Treatment of Classical Hodgkin Lymphoma of the Elderly
6.1. BV Combined with AVD
6.2. BV Monotherapy
6.3. Anthracycline-Free BV-Chemo Combinations
6.4. Checkpoint Inhibitor Monotherapy with or without BV for Frail or CT-Ineligible Patients
7. Ongoing and Forthcoming Randomized Trials
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Aoki, T.; Steidl, C. Novel insights into Hodgkin lymphoma biology by single-cell analysis. Blood 2023, 141, 1791–1801. [Google Scholar] [CrossRef] [PubMed]
- Kuppers, R.; Rajewsky, K.; Zhao, M.; Simons, G.; Laumann, R.; Fischer, R.; Hansmann, M.L. Hodgkin disease: Hodgkin and Reed-Sternberg cells picked from histological sections show clonal immunoglobulin gene rearrangements and appear to be derived from B cells at various stages of development. Proc. Natl. Acad. Sci. USA 1994, 91, 10962–10966. [Google Scholar] [CrossRef] [PubMed]
- Kanzler, H.; Kuppers, R.; Hansmann, M.L.; Rajewsky, K. Hodgkin and Reed-Sternberg cells in Hodgkin’s disease represent the outgrowth of a dominant tumor clone derived from (crippled) germinal center B cells. J. Exp. Med. 1996, 184, 1495–1505. [Google Scholar] [CrossRef] [PubMed]
- Schmitz, R.; Hansmann, M.L.; Bohle, V.; Martin-Subero, J.I.; Hartmann, S.; Mechtersheimer, G.; Klapper, W.; Vater, I.; Giefing, M.; Gesk, S.; et al. TNFAIP3 (A20) is a tumor suppressor gene in Hodgkin lymphoma and primary mediastinal B cell lymphoma. J. Exp. Med. 2009, 206, 981–989. [Google Scholar] [CrossRef]
- Stein, H.; Mason, D.Y.; Gerdes, J.; O’Connor, N.; Wainscoat, J.; Pallesen, G.; Gatter, K.; Falini, B.; Delsol, G.; Lemke, H.; et al. The expression of the Hodgkin’s disease associated antigen Ki-1 in reactive and neoplastic lymphoid tissue: Evidence that Reed-Sternberg cells and histiocytic malignancies are derived from activated lymphoid cells. Blood 1985, 66, 848–858. [Google Scholar] [CrossRef]
- Younes, A.; Bartlett, N.L.; Leonard, J.P.; Kennedy, D.A.; Lynch, C.M.; Sievers, E.L.; Forero-Torres, A. Brentuximab vedotin (SGN-35) for relapsed CD30-positive lymphomas. N. Engl. J. Med. 2010, 363, 1812–1821. [Google Scholar] [CrossRef]
- Younes, A.; Gopal, A.K.; Smith, S.E.; Ansell, S.M.; Rosenblatt, J.D.; Savage, K.J.; Ramchandren, R.; Bartlett, N.L.; Cheson, B.D.; de Vos, S.; et al. Results of a pivotal phase II study of brentuximab vedotin for patients with relapsed or refractory Hodgkin’s lymphoma. J. Clin. Oncol. 2012, 30, 2183–2189. [Google Scholar] [CrossRef]
- Carey, C.D.; Gusenleitner, D.; Lipschitz, M.; Roemer, M.G.M.; Stack, E.C.; Gjini, E.; Hu, X.; Redd, R.; Freeman, G.J.; Neuberg, D.; et al. Topological analysis reveals a PD-L1-associated microenvironmental niche for Reed-Sternberg cells in Hodgkin lymphoma. Blood 2017, 130, 2420–2430. [Google Scholar] [CrossRef]
- Patel, S.S.; Weirather, J.L.; Lipschitz, M.; Lako, A.; Chen, P.H.; Griffin, G.K.; Armand, P.; Shipp, M.A.; Rodig, S.J. The microenvironmental niche in classic Hodgkin lymphoma is enriched for CTLA-4-positive T cells that are PD-1-negative. Blood 2019, 134, 2059–2069. [Google Scholar] [CrossRef]
- Canellos, G.P.; Rosenberg, S.A.; Friedberg, J.W.; Lister, T.A.; Devita, V.T. Treatment of Hodgkin lymphoma: A 50-year perspective. J. Clin. Oncol. 2014, 32, 163–168. [Google Scholar] [CrossRef]
- Vassilakopoulos, T.P.; Angelopoulou, M.K. Advanced and relapsed/refractory Hodgkin lymphoma: What has been achieved during the last 50 years. Semin. Hematol. 2013, 50, 4–14. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Engert, A.; Franklin, J.; Eich, H.T.; Brillant, C.; Sehlen, S.; Cartoni, C.; Herrmann, R.; Pfreundschuh, M.; Sieber, M.; Tesch, H.; et al. Two cycles of doxorubicin, bleomycin, vinblastine, and dacarbazine plus extended-field radiotherapy is superior to radiotherapy alone in early favorable Hodgkin’s lymphoma: Final results of the GHSG HD7 trial. J. Clin. Oncol. 2007, 25, 3495–3502. [Google Scholar] [CrossRef] [PubMed]
- Carde, P.; Burgers, J.M.; Henry-Amar, M.; Hayat, M.; Sizoo, W.; Van der Schueren, E.; Monconduit, M.; Noordijk, E.M.; Lustman-Marechal, J.; Tanguy, A.; et al. Clinical stages I and II Hodgkin’s disease: A specifically tailored therapy according to prognostic factors. J. Clin. Oncol. 1988, 6, 239–252. [Google Scholar] [CrossRef] [PubMed]
- Carde, P.; Hagenbeek, A.; Hayat, M.; Monconduit, M.; Thomas, J.; Burgers, M.J.; Noordijk, E.M.; Tanguy, A.; Meerwaldt, J.H.; Le Fur, R.; et al. Clinical staging versus laparotomy and combined modality with MOPP versus ABVD in early-stage Hodgkin’s disease: The H6 twin randomized trials from the European Organization for Research and Treatment of Cancer Lymphoma Cooperative Group. J. Clin. Oncol. 1993, 11, 2258–2272. [Google Scholar] [CrossRef]
- Noordijk, E.M.; Carde, P.; Dupouy, N.; Hagenbeek, A.; Krol, A.D.; Kluin-Nelemans, J.C.; Tirelli, U.; Monconduit, M.; Thomas, J.; Eghbali, H.; et al. Combined-modality therapy for clinical stage I or II Hodgkin’s lymphoma: Long-term results of the European Organisation for Research and Treatment of Cancer H7 randomized controlled trials. J. Clin. Oncol. 2006, 24, 3128–3135. [Google Scholar] [CrossRef]
- Meyer, R.M.; Gospodarowicz, M.K.; Connors, J.M.; Pearcey, R.G.; Wells, W.A.; Winter, J.N.; Horning, S.J.; Dar, A.R.; Shustik, C.; Stewart, D.A.; et al. ABVD alone versus radiation-based therapy in limited-stage Hodgkin’s lymphoma. N. Engl. J. Med. 2012, 366, 399–408. [Google Scholar] [CrossRef]
- Straus, D.J.; Portlock, C.S.; Qin, J.; Myers, J.; Zelenetz, A.D.; Moskowitz, C.; Noy, A.; Goy, A.; Yahalom, J. Results of a prospective randomized clinical trial of doxorubicin, bleomycin, vinblastine, and dacarbazine (ABVD) followed by radiation therapy (RT) versus ABVD alone for stages I, II, and IIIA nonbulky Hodgkin disease. Blood 2004, 104, 3483–3489. [Google Scholar] [CrossRef]
- Andre, M.P.E.; Girinsky, T.; Federico, M.; Reman, O.; Fortpied, C.; Gotti, M.; Casasnovas, O.; Brice, P.; van der Maazen, R.; Re, A.; et al. Early Positron Emission Tomography Response-Adapted Treatment in Stage I and II Hodgkin Lymphoma: Final Results of the Randomized EORTC/LYSA/FIL H10 Trial. J. Clin. Oncol. 2017, 35, 1786–1794. [Google Scholar] [CrossRef]
- Radford, J.; Illidge, T.; Counsell, N.; Hancock, B.; Pettengell, R.; Johnson, P.; Wimperis, J.; Culligan, D.; Popova, B.; Smith, P.; et al. Results of a trial of PET-directed therapy for early-stage Hodgkin’s lymphoma. N. Engl. J. Med. 2015, 372, 1598–1607. [Google Scholar] [CrossRef]
- Fuchs, M.; Goergen, H.; Kobe, C.; Kuhnert, G.; Lohri, A.; Greil, R.; Sasse, S.; Topp, M.S.; Schafer, E.; Hertenstein, B.; et al. Positron Emission Tomography-Guided Treatment in Early-Stage Favorable Hodgkin Lymphoma: Final Results of the International, Randomized Phase III HD16 Trial by the German Hodgkin Study Group. J. Clin. Oncol. 2019, 37, 2835–2845. [Google Scholar] [CrossRef]
- Borchmann, P.; Plutschow, A.; Kobe, C.; Greil, R.; Meissner, J.; Topp, M.S.; Ostermann, H.; Dierlamm, J.; Mohm, J.; Thiemer, J.; et al. PET-guided omission of radiotherapy in early-stage unfavourable Hodgkin lymphoma (GHSG HD17): A multicentre, open-label, randomised, phase 3 trial. Lancet Oncol. 2021, 22, 223–234. [Google Scholar] [CrossRef] [PubMed]
- Devita, V.T., Jr.; Serpick, A.A.; Carbone, P.P. Combination chemotherapy in the treatment of advanced Hodgkin’s disease. Ann. Intern. Med. 1970, 73, 881–895. [Google Scholar] [CrossRef] [PubMed]
- Bonadonna, G.; Zucali, R.; Monfardini, S.; De Lena, M.; Uslenghi, C. Combination chemotherapy of Hodgkin’s disease with adriamycin, bleomycin, vinblastine, and imidazole carboxamide versus MOPP. Cancer 1975, 36, 252–259. [Google Scholar] [CrossRef] [PubMed]
- Canellos, G.P.; Anderson, J.R.; Propert, K.J.; Nissen, N.; Cooper, M.R.; Henderson, E.S.; Green, M.R.; Gottlieb, A.; Peterson, B.A. Chemotherapy of advanced Hodgkin’s disease with MOPP, ABVD, or MOPP alternating with ABVD. N. Engl. J. Med. 1992, 327, 1478–1484. [Google Scholar] [CrossRef]
- Glick, J.H.; Young, M.L.; Harrington, D.; Schilsky, R.L.; Beck, T.; Neiman, R.; Fisher, R.I.; Peterson, B.A.; Oken, M.M. MOPP/ABV hybrid chemotherapy for advanced Hodgkin’s disease significantly improves failure-free and overall survival: The 8-year results of the intergroup trial. J. Clin. Oncol. 1998, 16, 19–26. [Google Scholar] [CrossRef]
- Chisesi, T.; Bellei, M.; Luminari, S.; Montanini, A.; Marcheselli, L.; Levis, A.; Gobbi, P.; Vitolo, U.; Stelitano, C.; Pavone, V.; et al. Long-term follow-up analysis of HD9601 trial comparing ABVD versus Stanford V versus MOPP/EBV/CAD in patients with newly diagnosed advanced-stage Hodgkin’s lymphoma: A study from the Intergruppo Italiano Linfomi. J. Clin. Oncol. 2011, 29, 4227–4233. [Google Scholar] [CrossRef]
- Gordon, L.I.; Hong, F.; Fisher, R.I.; Bartlett, N.L.; Connors, J.M.; Gascoyne, R.D.; Wagner, H.; Stiff, P.J.; Cheson, B.D.; Gospodarowicz, M.; et al. Randomized phase III trial of ABVD versus Stanford V with or without radiation therapy in locally extensive and advanced-stage Hodgkin lymphoma: An intergroup study coordinated by the Eastern Cooperative Oncology Group (E2496). J. Clin. Oncol. 2013, 31, 684–691. [Google Scholar] [CrossRef]
- Diehl, V.; Franklin, J.; Pfreundschuh, M.; Lathan, B.; Paulus, U.; Hasenclever, D.; Tesch, H.; Herrmann, R.; Dorken, B.; Muller-Hermelink, H.K.; et al. Standard and increased-dose BEACOPP chemotherapy compared with COPP-ABVD for advanced Hodgkin’s disease. N. Engl. J. Med. 2003, 348, 2386–2395. [Google Scholar] [CrossRef]
- Skoetz, N.; Trelle, S.; Rancea, M.; Haverkamp, H.; Diehl, V.; Engert, A.; Borchmann, P. Effect of initial treatment strategy on survival of patients with advanced-stage Hodgkin’s lymphoma: A systematic review and network meta-analysis. Lancet Oncol. 2013, 14, 943–952. [Google Scholar] [CrossRef]
- Vassilakopoulos, T.P.; Johnson, P.W. Treatment of advanced-stage Hodgkin lymphoma. Semin. Hematol. 2016, 53, 171–179. [Google Scholar] [CrossRef]
- Gallamini, A.; Hutchings, M.; Rigacci, L.; Specht, L.; Merli, F.; Hansen, M.; Patti, C.; Loft, A.; Di Raimondo, F.; D’Amore, F.; et al. Early interim 2-[18F]fluoro-2-deoxy-D-glucose positron emission tomography is prognostically superior to international prognostic score in advanced-stage Hodgkin’s lymphoma: A report from a joint Italian-Danish study. J. Clin. Oncol. 2007, 25, 3746–3752. [Google Scholar] [CrossRef] [PubMed]
- Gallamini, A.; Patti, C.; Viviani, S.; Rossi, A.; Fiore, F.; Di Raimondo, F.; Cantonetti, M.; Stelitano, C.; Feldman, T.; Gavarotti, P.; et al. Early chemotherapy intensification with BEACOPP in advanced-stage Hodgkin lymphoma patients with a interim-PET positive after two ABVD courses. Br. J. Haematol. 2011, 152, 551–560. [Google Scholar] [CrossRef] [PubMed]
- Johnson, P.; Federico, M.; Kirkwood, A.; Fossa, A.; Berkahn, L.; Carella, A.; d’Amore, F.; Enblad, G.; Franceschetto, A.; Fulham, M.; et al. Adapted Treatment Guided by Interim PET-CT Scan in Advanced Hodgkin’s Lymphoma. N. Engl. J. Med. 2016, 374, 2419–2429. [Google Scholar] [CrossRef] [PubMed]
- Gallamini, A.; Tarella, C.; Viviani, S.; Rossi, A.; Patti, C.; Mule, A.; Picardi, M.; Romano, A.; Cantonetti, M.; La Nasa, G.; et al. Early Chemotherapy Intensification with Escalated BEACOPP in Patients with Advanced-Stage Hodgkin Lymphoma With a Positive Interim Positron Emission Tomography/Computed Tomography Scan After Two ABVD Cycles: Long-Term Results of the GITIL/FIL HD 0607 Trial. J. Clin. Oncol. 2018, 36, 454–462. [Google Scholar] [CrossRef] [PubMed]
- Stephens, D.M.; Li, H.; Schoder, H.; Straus, D.J.; Moskowitz, C.H.; LeBlanc, M.; Rimsza, L.M.; Bartlett, N.L.; Evens, A.M.; LaCasce, A.S.; et al. Five-year follow-up of SWOG S0816: Limitations and values of a PET-adapted approach with stage III/IV Hodgkin lymphoma. Blood 2019, 134, 1238–1246. [Google Scholar] [CrossRef]
- Gallamini, A.; Rossi, A.; Patti, C.; Picardi, M.; Romano, A.; Cantonetti, M.; Oppi, S.; Viviani, S.; Bolis, S.; Trentin, L.; et al. Consolidation Radiotherapy Could Be Safely Omitted in Advanced Hodgkin Lymphoma with Large Nodal Mass in Complete Metabolic Response After ABVD: Final Analysis of the Randomized GITIL/FIL HD0607 Trial. J. Clin. Oncol. 2020, 38, 3905–3913. [Google Scholar] [CrossRef]
- Borchmann, P.; Goergen, H.; Kobe, C.; Lohri, A.; Greil, R.; Eichenauer, D.A.; Zijlstra, J.M.; Markova, J.; Meissner, J.; Feuring-Buske, M.; et al. PET-guided treatment in patients with advanced-stage Hodgkin’s lymphoma (HD18): Final results of an open-label, international, randomised phase 3 trial by the German Hodgkin Study Group. Lancet 2017, 390, 2790–2802. [Google Scholar] [CrossRef]
- Casasnovas, R.O.; Bouabdallah, R.; Brice, P.; Lazarovici, J.; Ghesquieres, H.; Stamatoullas, A.; Dupuis, J.; Gac, A.C.; Gastinne, T.; Joly, B.; et al. PET-adapted treatment for newly diagnosed advanced Hodgkin lymphoma (AHL2011): A randomised, multicentre, non-inferiority, phase 3 study. Lancet Oncol. 2019, 20, 202–215. [Google Scholar] [CrossRef]
- Casasnovas, R.O.; Bouabdallah, R.; Brice, P.; Lazarovici, J.; Ghesquieres, H.; Stamatoullas, A.; Dupuis, J.; Gac, A.C.; Gastinne, T.; Joly, B.; et al. Positron Emission Tomography-Driven Strategy in Advanced Hodgkin Lymphoma: Prolonged Follow-Up of the AHL2011 Phase III Lymphoma Study Association Study. J. Clin. Oncol. 2022, 40, 1091–1101. [Google Scholar] [CrossRef]
- Engert, A.; Haverkamp, H.; Kobe, C.; Markova, J.; Renner, C.; Ho, A.; Zijlstra, J.; Kral, Z.; Fuchs, M.; Hallek, M.; et al. Reduced-intensity chemotherapy and PET-guided radiotherapy in patients with advanced stage Hodgkin’s lymphoma (HD15 trial): A randomised, open-label, phase 3 non-inferiority trial. Lancet 2012, 379, 1791–1799. [Google Scholar] [CrossRef]
- Connors, J.M.; Ansell, S.M.; Fanale, M.; Park, S.I.; Younes, A. Five-year follow-up of brentuximab vedotin combined with ABVD or AVD for advanced-stage classical Hodgkin lymphoma. Blood 2017, 130, 1375–1377. [Google Scholar] [CrossRef] [PubMed]
- Connors, J.M.; Jurczak, W.; Straus, D.J.; Ansell, S.M.; Kim, W.S.; Gallamini, A.; Younes, A.; Alekseev, S.; Illes, A.; Picardi, M.; et al. Brentuximab Vedotin with Chemotherapy for Stage III or IV Hodgkin’s Lymphoma. N. Engl. J. Med. 2018, 378, 331–344. [Google Scholar] [CrossRef] [PubMed]
- Ansell, S.M.; Radford, J.; Connors, J.M.; Dlugosz-Danecka, M.; Kim, W.S.; Gallamini, A.; Ramchandren, R.; Friedberg, J.W.; Advani, R.; Hutchings, M.; et al. Overall Survival with Brentuximab Vedotin in Stage III or IV Hodgkin’s Lymphoma. N. Engl. J. Med. 2022, 387, 310–320. [Google Scholar] [CrossRef]
- Straus, D.J.; Dlugosz-Danecka, M.; Connors, J.M.; Alekseev, S.; Illes, A.; Picardi, M.; Lech-Maranda, E.; Feldman, T.; Smolewski, P.; Savage, K.J.; et al. Brentuximab vedotin with chemotherapy for stage III or IV classical Hodgkin lymphoma (ECHELON-1): 5-year update of an international, open-label, randomised, phase 3 trial. Lancet Haematol. 2021, 8, e410–e421. [Google Scholar] [CrossRef] [PubMed]
- Herrera, A.F.; LeBlanc, M.; Castellino, S.M.; Li, H.; Rutherford, S.C.; Evens, A.M.; Davison, K.; Punnett, A.; Hodgson, D.; Parsons, S.K.; et al. Nivolumab (N)-AVD improves progression-free survival compared to Brentuximab Vedotin (BV)-AVD in advanced stage (AS) classic Hodgkin Lymphoma (HL): Results of SWOG S1826. Hematol. Oncol. 2023, 41, 33–35. [Google Scholar] [CrossRef]
- Press, O.W.; Li, H.; Schoder, H.; Straus, D.J.; Moskowitz, C.H.; LeBlanc, M.; Rimsza, L.M.; Bartlett, N.L.; Evens, A.M.; Mittra, E.S.; et al. US Intergroup Trial of Response-Adapted Therapy for Stage III to IV Hodgkin Lymphoma Using Early Interim Fluorodeoxyglucose-Positron Emission Tomography Imaging: Southwest Oncology Group S0816. J. Clin. Oncol. 2016, 34, 2020–2027. [Google Scholar] [CrossRef]
- Zinzani, P.L.; Broccoli, A.; Gioia, D.M.; Castagnoli, A.; Ciccone, G.; Evangelista, A.; Santoro, A.; Ricardi, U.; Bonfichi, M.; Brusamolino, E.; et al. Interim Positron Emission Tomography Response-Adapted Therapy in Advanced-Stage Hodgkin Lymphoma: Final Results of the Phase II Part of the HD0801 Study. J. Clin. Oncol. 2016, 34, 1376–1385. [Google Scholar] [CrossRef]
- Diepstra, A.; Visser, L.; Fortpied, C.; Noordzij, W.; Loft, A.; Arens, A.; Sureda-Balari, A.; Carvalho, S.; Vranovský, A.; Sents, W.; et al. T001: FDG-PET and serum TARC levels after one cycle of BV-AVD in advanced stage Hodgkin lymphoma patients: Results from the very early PET-response adapted EORTC-COBRA trial. HemaSphere 2022, 6, 1. [Google Scholar] [CrossRef]
- Borchmann, P.; Haverkamp, H.; Lohri, A.; Mey, U.; Kreissl, S.; Greil, R.; Markova, J.; Feuring-Buske, M.; Meissner, J.; Duhrsen, U.; et al. Progression-free survival of early interim PET-positive patients with advanced stage Hodgkin’s lymphoma treated with BEACOPP(escalated) alone or in combination with rituximab (HD18): An open-label, international, randomised phase 3 study by the German Hodgkin Study Group. Lancet Oncol. 2017, 18, 454–463. [Google Scholar]
- Eichenauer, D.A.; Plutschow, A.; Kreissl, S.; Sokler, M.; Hellmuth, J.C.; Meissner, J.; Mathas, S.; Topp, M.S.; Behringer, K.; Klapper, W.; et al. Incorporation of brentuximab vedotin into first-line treatment of advanced classical Hodgkin’s lymphoma: Final analysis of a phase 2 randomised trial by the German Hodgkin Study Group. Lancet Oncol. 2017, 18, 1680–1687. [Google Scholar] [CrossRef]
- Damaschin, C.; Goergen, H.; Kreissl, S.; Plutschow, A.; Breywisch, F.; Mathas, S.; Meissner, J.; Sokler, M.; Topp, M.S.; Vucinic, V.; et al. Brentuximab vedotin-containing escalated BEACOPP variants for newly diagnosed advanced-stage classical Hodgkin lymphoma: Follow-up analysis of a randomized phase II study from the German Hodgkin Study Group. Leukemia 2022, 36, 580–582. [Google Scholar] [CrossRef] [PubMed]
- Borchmann, P.; Moccia, A.; Greil, R.; Hertzberg, M.; Schaub, V.; Hüttmann, A.; Keil, F.; Dierlamm, J.; Haenel, M.; Novak, U.; et al. Treatment Related Morbidity in Patients with Classical Hodgkin Lymphoma: Results of the Ongoing, Randomized Phase III HD21 Trial by the German Hodgkin Study Group. Blood 2022, 140, 771–773. [Google Scholar] [CrossRef]
- Borchmann, P.; Moccia, A.A.; Greil, R.; Schneider, G.; Hertzberg, M.; Schaub, V.; Hüttmann, A.; Keil, F.; Dierlamm, J.; Hänel, M.; et al. BRECADD is non-inferior to eBEACOPP in patients with advanced stage classical Hodgkin Lymphoma: Efficacy results of the GHSG phase III HD21 trial. Hematol. Oncol. 2023, 41, 881–882. [Google Scholar] [CrossRef]
- Abramson, J.S.; Arnason, J.E.; LaCasce, A.S.; Redd, R.; Barnes, J.A.; Sokol, L.; Joyce, R.; Avigan, D.; Neuberg, D.; Takvorian, R.W.; et al. Brentuximab vedotin, doxorubicin, vinblastine, and dacarbazine for nonbulky limited-stage classical Hodgkin lymphoma. Blood 2019, 134, 606–613. [Google Scholar] [CrossRef]
- Abramson, J.S.; Bengston, E.; Redd, R.; Barnes, J.A.; Takvorian, T.; Sokol, L.; Lansigan, F.; Armand, P.; Shah, B.; Jacobsen, E.; et al. Brentuximab vedotin plus doxorubicin and dacarbazine in nonbulky limited-stage classical Hodgkin lymphoma. Blood Adv. 2023, 7, 1130–1136. [Google Scholar] [CrossRef]
- Park, S.I.; Shea, T.C.; Olajide, O.; Reddy, N.M.; Budde, L.E.; Ghosh, N.; Deal, A.M.; Noe, J.F.; Ansell, S.M. ABVD followed by BV consolidation in risk-stratified patients with limited-stage Hodgkin lymphoma. Blood Adv. 2020, 4, 2548–2555. [Google Scholar] [CrossRef]
- Kumar, A.; Casulo, C.; Advani, R.H.; Budde, E.; Barr, P.M.; Batlevi, C.L.; Caron, P.; Constine, L.S.; Dandapani, S.V.; Drill, E.; et al. Brentuximab Vedotin Combined With Chemotherapy in Patients With Newly Diagnosed Early-Stage, Unfavorable-Risk Hodgkin Lymphoma. J. Clin. Oncol. 2021, 39, 2257–2265. [Google Scholar] [CrossRef]
- Fornecker, L.M.; Lazarovici, J.; Aurer, I.; Casasnovas, R.O.; Gac, A.C.; Bonnet, C.; Bouabdallah, K.; Feugier, P.; Specht, L.; Molina, L.; et al. Brentuximab Vedotin Plus AVD for First-Line Treatment of Early-Stage Unfavorable Hodgkin Lymphoma (BREACH): A Multicenter, Open-Label, Randomized, Phase II Trial. J. Clin. Oncol. 2023, 41, 327–335. [Google Scholar] [CrossRef]
- Behringer, K.; Goergen, H.; Hitz, F.; Zijlstra, J.M.; Greil, R.; Markova, J.; Sasse, S.; Fuchs, M.; Topp, M.S.; Soekler, M.; et al. Omission of dacarbazine or bleomycin, or both, from the ABVD regimen in treatment of early-stage favourable Hodgkin’s lymphoma (GHSG HD13): An open-label, randomised, non-inferiority trial. Lancet 2015, 385, 1418–1427. [Google Scholar] [CrossRef]
- Barrington, S.F.; Phillips, E.H.; Counsell, N.; Hancock, B.; Pettengell, R.; Johnson, P.; Townsend, W.; Culligan, D.; Popova, B.; Clifton-Hadley, L.; et al. Positron Emission Tomography Score Has Greater Prognostic Significance Than Pretreatment Risk Stratification in Early-Stage Hodgkin Lymphoma in the UK RAPID Study. J. Clin. Oncol. 2019, 37, 1732–1741. [Google Scholar] [CrossRef]
- Stuver, R.; Michaud, L.; Casulo, C.; Advani, R.H.; Budde, E.L.; Barr, P.M.; Batlevi, C.L.; Caron, P.C.; Constine, L.S.; Dandapani, S.; et al. Brentuximab Vedotin Combined with Chemotherapy in Newly Diagnosed, Early-Stage, Unfavorable-Risk Hodgkin Lymphoma: Extended Follow-up with Evaluation of Baseline Metabolic Tumor Volume and PET2. Blood 2022, 140 (Suppl. S1), 1756–1758. [Google Scholar] [CrossRef]
- Armand, P.; Engert, A.; Younes, A.; Fanale, M.; Santoro, A.; Zinzani, P.L.; Timmerman, J.M.; Collins, G.P.; Ramchandren, R.; Cohen, J.B.; et al. Nivolumab for Relapsed/Refractory Classic Hodgkin Lymphoma After Failure of Autologous Hematopoietic Cell Transplantation: Extended Follow-Up of the Multicohort Single-Arm Phase II CheckMate 205 Trial. J. Clin. Oncol. 2018, 36, 1428–1439. [Google Scholar] [CrossRef] [PubMed]
- Younes, A.; Santoro, A.; Shipp, M.; Zinzani, P.L.; Timmerman, J.M.; Ansell, S.; Armand, P.; Fanale, M.; Ratanatharathorn, V.; Kuruvilla, J.; et al. Nivolumab for classical Hodgkin’s lymphoma after failure of both autologous stem-cell transplantation and brentuximab vedotin: A multicentre, multicohort, single-arm phase 2 trial. Lancet Oncol. 2016, 17, 1283–1294. [Google Scholar] [CrossRef]
- Chen, R.; Zinzani, P.L.; Fanale, M.A.; Armand, P.; Johnson, N.A.; Brice, P.; Radford, J.; Ribrag, V.; Molin, D.; Vassilakopoulos, T.P.; et al. Phase II Study of the Efficacy and Safety of Pembrolizumab for Relapsed/Refractory Classic Hodgkin Lymphoma. J. Clin. Oncol. 2017, 35, 2125–2132. [Google Scholar] [CrossRef]
- Chen, R.; Zinzani, P.L.; Lee, H.J.; Armand, P.; Johnson, N.A.; Brice, P.; Radford, J.; Ribrag, V.; Molin, D.; Vassilakopoulos, T.P.; et al. Pembrolizumab in relapsed or refractory Hodgkin lymphoma: 2-year follow-up of KEYNOTE-087. Blood 2019, 134, 1144–1153. [Google Scholar] [CrossRef] [PubMed]
- Zinzani, P.L.; Chen, R.; Armand, P.; Johnson, N.A.; Brice, P.; Radford, J.; Ribrag, V.; Molin, D.; Vassilakopoulos, T.P.; Tomita, A.; et al. Pembrolizumab monotherapy in patients with primary refractory classical hodgkin lymphoma who relapsed after salvage autologous stem cell transplantation and/or brentuximab vedotin therapy: KEYNOTE-087 subgroup analysis. Leuk. Lymphoma 2020, 61, 950–954. [Google Scholar] [CrossRef]
- Armand, P.; Zinzani, P.L.; Lee, H.J.; Johnson, N.A.; Brice, P.; Radford, J.; Ribrag, V.; Molin, D.; Vassilakopoulos, T.P.; Tomita, A.; et al. Five-year follow-up of KEYNOTE-087: Pembrolizumab monotherapy in relapsed/refractory classical Hodgkin lymphoma. Blood 2023. [Google Scholar] [CrossRef]
- Kuruvilla, J.; Ramchandren, R.; Santoro, A.; Paszkiewicz-Kozik, E.; Gasiorowski, R.; Johnson, N.A.; Fogliatto, L.M.; Goncalves, I.; de Oliveira, J.S.R.; Buccheri, V.; et al. Pembrolizumab versus brentuximab vedotin in relapsed or refractory classical Hodgkin lymphoma (KEYNOTE-204): An interim analysis of a multicentre, randomised, open-label, phase 3 study. Lancet Oncol. 2021, 22, 512–524. [Google Scholar] [CrossRef]
- Vassilakopoulos, T.P. Relapsed or refractory classical Hodgkin lymphoma: Which immunotherapy, and when? Lancet Oncol. 2021, 22, 417–419. [Google Scholar] [CrossRef]
- Ansell, S.M.; Bröckelmann, P.J.; Keudell, G.; Lee, H.J.; Santoro, A.; Zinzani, P.L.; Collins, G.P.; Cohen, J.B.; Boer, J.P.; Kuruvilla, J.; et al. Nivolumab for Relapsed or Refractory (R/R) Classical Hodgkin Lymphoma (Chl) after Autologous Transplantation: 5-Year Overall Survival from the Phase 2 Checkmate 205 Study. Hematol. Oncol. 2021, 39, 18–22. [Google Scholar] [CrossRef]
- Ramchandren, R.; Domingo-Domenech, E.; Rueda, A.; Trneny, M.; Feldman, T.A.; Lee, H.J.; Provencio, M.; Sillaber, C.; Cohen, J.B.; Savage, K.J.; et al. Nivolumab for Newly Diagnosed Advanced-Stage Classic Hodgkin Lymphoma: Safety and Efficacy in the Phase II CheckMate 205 Study. J. Clin. Oncol. 2019, 37, 1997–2007. [Google Scholar] [CrossRef] [PubMed]
- Brockelmann, P.J.; Buhnen, I.; Meissner, J.; Trautmann-Grill, K.; Herhaus, P.; Halbsguth, T.V.; Schaub, V.; Kerkhoff, A.; Mathas, S.; Bormann, M.; et al. Nivolumab and Doxorubicin, Vinblastine, and Dacarbazine in Early-Stage Unfavorable Hodgkin Lymphoma: Final Analysis of the Randomized German Hodgkin Study Group Phase II NIVAHL Trial. J. Clin. Oncol. 2023, 41, 1193–1199. [Google Scholar] [CrossRef] [PubMed]
- Allen, P.B.; Lu, X.; Chen, Q.; O’Shea, K.; Chmiel, J.S.; Slonim, L.B.; Sukhanova, M.; Savas, H.; Evens, A.M.; Advani, R.; et al. Sequential pembrolizumab and AVD are highly effective at any PD-L1 expression level in untreated Hodgkin lymphoma. Blood Adv. 2023, 7, 2670–2676. [Google Scholar] [CrossRef] [PubMed]
- Lynch, R.C.; Ujjani, C.S.; Poh, C.; Warren, E.H.; Smith, S.D.; Shadman, M.; Till, B.; Raghunathan, V.M.; Alig, S.; Alizadeh, A.A.; et al. Concurrent pembrolizumab with AVD for untreated classic Hodgkin lymphoma. Blood 2023, 141, 2576–2586. [Google Scholar] [CrossRef]
- Cheson, B.D.; Pfistner, B.; Juweid, M.E.; Gascoyne, R.D.; Specht, L.; Horning, S.J.; Coiffier, B.; Fisher, R.I.; Hagenbeek, A.; Zucca, E.; et al. Revised response criteria for malignant lymphoma. J. Clin. Oncol. 2007, 25, 579–586. [Google Scholar] [CrossRef] [PubMed]
- Cheson, B.D.; Fisher, R.I.; Barrington, S.F.; Cavalli, F.; Schwartz, L.H.; Zucca, E.; Lister, T.A.; Alliance, A.L.; Lymphoma, G.; Eastern Cooperative Oncology, G.; et al. Recommendations for initial evaluation, staging, and response assessment of Hodgkin and non-Hodgkin lymphoma: The Lugano classification. J. Clin. Oncol. 2014, 32, 3059–3068. [Google Scholar] [CrossRef]
- Cheson, B.D.; Ansell, S.; Schwartz, L.; Gordon, L.I.; Advani, R.; Jacene, H.A.; Hoos, A.; Barrington, S.F.; Armand, P. Refinement of the Lugano Classification lymphoma response criteria in the era of immunomodulatory therapy. Blood 2016, 128, 2489–2496. [Google Scholar] [CrossRef]
- Brockelmann, P.J.; Goergen, H.; Keller, U.; Meissner, J.; Ordemann, R.; Halbsguth, T.V.; Sasse, S.; Sokler, M.; Kerkhoff, A.; Mathas, S.; et al. Efficacy of Nivolumab and AVD in Early-Stage Unfavorable Classic Hodgkin Lymphoma: The Randomized Phase 2 German Hodgkin Study Group NIVAHL Trial. JAMA Oncol. 2020, 6, 872–880. [Google Scholar] [CrossRef]
- Allen, P.B.; Savas, H.; Evens, A.M.; Advani, R.H.; Palmer, B.; Pro, B.; Karmali, R.; Mou, E.; Bearden, J.; Dillehay, G.; et al. Pembrolizumab followed by AVD in untreated early unfavorable and advanced-stage classical Hodgkin lymphoma. Blood 2021, 137, 1318–1326. [Google Scholar] [CrossRef]
- Lynch, R.C.; Uijani, C.S.; Poh, C.; Warren, E.H.; Smith, S.D.; Shadman, M.; Shustov, A.R.; Till, B.G.; Tseng, Y.D.; Coye, H.; et al. Concurrent Pembrolizumab with AVD for Untreated Classical Hodgkin Lymphoma. Blood 2021, 138 (Suppl. S1), 233. [Google Scholar] [CrossRef]
- Ricardi, U.; Levis, M.; Evangelista, A.; Gioia, D.M.; Sacchetti, G.M.; Gotti, M.; Re, A.; Buglione, M.; Pavone, V.; Nardella, A.; et al. Role of radiotherapy to bulky sites of advanced Hodgkin lymphoma treated with ABVD: Final results of FIL HD0801 trial. Blood Adv. 2021, 5, 4504–4514. [Google Scholar] [CrossRef]
- Advani, R.H.; Avigdor, A.; Sureda, A.; Lavie, D.; Hohaus, S.; Zaucha, J.M.; Hua, V.M.; Zilioli, V.R.; Gazitua, R.; Özcan, M.; et al. Pembrolizumab and Chemotherapy in Newly-Diagnosed, Early Unfavorable or Advanced Stage Classic Hodgkin Lymphoma: The Phase 2 Keynote-C11 Study. Blood 2022, 140 (Suppl. S1), 1759–1760. [Google Scholar] [CrossRef]
- Herrera, A.F.; Burton, C.; Radford, J.; Miall, F.; Townsend, W.; Santoro, A.; Zinzani, P.L.; Lewis, D.; Fowst, C.; Brar, S.; et al. Avelumab in relapsed/refractory classical Hodgkin lymphoma: Phase 1b results from the JAVELIN Hodgkins trial. Blood Adv. 2021, 5, 3387–3396. [Google Scholar] [CrossRef] [PubMed]
- Collins, G.P.; Hawkes, E.A.; Kirkwood, A.A.; Clifton-Hadley, L.; Tyson, C.; Barsoum, E.; Pettengell, R.; Medd, P.G.; McKay, P.; Miall, F.; et al. Avelumab Monotherapy Followed by a Pet Adapted Chemotherapy Approach in the First Line Treatment of Classical Hodgkin Lymphoma: Initial Results from the Avenue Window Study. Hematol. Oncol. 2023, 41, 162–163. [Google Scholar] [CrossRef]
- Diehl, V.; Sextro, M.; Franklin, J.; Hansmann, M.L.; Harris, N.; Jaffe, E.; Poppema, S.; Harris, M.; Franssila, K.; van Krieken, J.; et al. Clinical presentation, course, and prognostic factors in lymphocyte-predominant Hodgkin’s disease and lymphocyte-rich classical Hodgkin’s disease: Report from the European Task Force on Lymphoma Project on Lymphocyte-Predominant Hodgkin’s Disease. J. Clin. Oncol. 1999, 17, 776–783. [Google Scholar] [CrossRef] [PubMed]
- Venkataraman, G.; Mirza, M.K.; Eichenauer, D.A.; Diehl, V. Current status of prognostication in classical Hodgkin lymphoma. Br. J. Haematol. 2014, 165, 287–299. [Google Scholar] [CrossRef] [PubMed]
- Brockelmann, P.J.; Angelopoulou, M.K.; Vassilakopoulos, T.P. Prognostic factors in Hodgkin lymphoma. Semin. Hematol. 2016, 53, 155–164. [Google Scholar] [CrossRef]
- Lee, H.; Flinn, I.W.; Melear, J.; Ramchandren, R.; Friedman, J.; Burke, J.M.; Linhares, Y.; Gonzales, P.A.; Raval, M.; Chintapatla, R.; et al. Brentuximab Vedotin, Nivolumab, Doxorubicin, and Dacarbazine (AN+AD) for Advanced Stage Classic Hodgkin Lymphoma: Updated Efficacy and Safety Results from the Single-Arm Phase 2 Study (SGN35-027 Part B). Blood 2022, 140, 763–765. [Google Scholar] [CrossRef]
- Lee, H.J.; Abramson, J.S.; Bartlett, N.L.; Burke, J.M.; Lynch, R.C.; Eva, D.D.; Hess, B.T.; Schuster, S.R.; Linhares, Y.; Ramchandren, R.; et al. Brentuximab Vedotin, Nivolumab, Doxorubicin, and Dacarbazine (AN+AD) for Early Stage Classic Hodgkin Lymphoma: Interim Efficacy and Safety Results from the Single-Arm Phase 2 Study (SGN35-027 Part C). Blood 2022, 140, 9399–9401. [Google Scholar] [CrossRef]
- Park, S.I.; Ansell, S.M.; Giri, S.; Svoboda, J.; Smith, S.D.; Feldman, T.; Budde, E.L.; Ness, A.J.; Choi, Y.; Bierman, P.J.; et al. Frontline PET-Directed Therapy with Brentuximab Vedotin Plus AVD Followed By Nivolumab Consolidation in Patients with Limited Stage Hodgkin Lymphoma. Blood 2022, 140, 1751–1752. [Google Scholar] [CrossRef]
- Goh, Z.; Latimer, M.; Lewis, K.L.; Cheah, C.Y.; Ciaccio, P.D.; Cushion, T.; Hawkes, E.A.; Harrop, S.; Ku, M.; Campbell, A.; et al. Characteristics and Outcomes of Older Patients with Classical Hodgkin Lymphoma: An Australasian Lymphoma Alliance, and Lymphoma and Related Diseases Registry Study. Clin. Lymphoma Myeloma Leuk. 2023, 23, 370–378. [Google Scholar] [CrossRef]
- Vassilakopoulos, T.P.; Pangalis, G.A.; Boutsikas, G.; Kokoris, S.; Galani, Z.; Siakantaris, M.; Dimitriadou, E.; Georgiou, G.; Dimou, M.; Kalpadaki, C.; et al. Hodgkin Lymphoma (HL) in patients ≥60 years old: Clinical and laboratory features, outcome after anthracycline-based treatment and comparison with younger patients: A single center experience. Haematol. Hematol. J. 2011, 96 (Suppl. S2), 87. [Google Scholar]
- Evens, A.M.; Connors, J.M.; Younes, A.; Ansell, S.M.; Kim, W.S.; Radford, J.; Feldman, T.; Tuscano, J.; Savage, K.J.; Oki, Y.; et al. Older patients (aged ≥ 60 years) with previously untreated advanced-stage classical Hodgkin lymphoma: A detailed analysis from the phase III ECHELON-1 study. Haematologica 2022, 107, 1086–1094. [Google Scholar] [CrossRef] [PubMed]
- Evens, A.M.; Advani, R.H.; Helenowski, I.B.; Fanale, M.; Smith, S.M.; Jovanovic, B.D.; Bociek, G.R.; Klein, A.K.; Winter, J.N.; Gordon, L.I.; et al. Multicenter Phase II Study of Sequential Brentuximab Vedotin and Doxorubicin, Vinblastine, and Dacarbazine Chemotherapy for Older Patients with Untreated Classical Hodgkin Lymphoma. J. Clin. Oncol. 2018, 36, 3015–3022. [Google Scholar] [CrossRef] [PubMed]
- Gibb, A.; Pirrie, S.J.; Linton, K.; Warbey, V.; Paterson, K.; Davies, A.J.; Collins, G.P.; Menne, T.; McKay, P.; Fields, P.A.; et al. Results of a UK National Cancer Research Institute Phase II study of brentuximab vedotin using a response-adapted design in the first-line treatment of patients with classical Hodgkin lymphoma unsuitable for chemotherapy due to age, frailty or comorbidity (BREVITY). Br. J. Haematol. 2021, 193, 63–71. [Google Scholar] [PubMed]
- Forero-Torres, A.; Holkova, B.; Goldschmidt, J.; Chen, R.; Olsen, G.; Boccia, R.V.; Bordoni, R.E.; Friedberg, J.W.; Sharman, J.P.; Palanca-Wessels, M.C.; et al. Phase 2 study of frontline brentuximab vedotin monotherapy in Hodgkin lymphoma patients aged 60 years and older. Blood 2015, 126, 2798–2804. [Google Scholar] [CrossRef] [PubMed]
- Friedberg, J.W.; Forero-Torres, A.; Bordoni, R.E.; Cline, V.J.M.; Patel Donnelly, D.; Flynn, P.J.; Olsen, G.; Chen, R.; Fong, A.; Wang, Y.; et al. Frontline brentuximab vedotin in combination with dacarbazine or bendamustine in patients aged ≥ 60 years with HL. Blood 2017, 130, 2829–2837. [Google Scholar] [CrossRef] [PubMed]
- Schiano de Colella, J.M.; Viviani, S.; Rapezzi, D.; Patti, C.; Clement Filliatre, L.; Rossi, A.; Bijou, F.; Cantonetti, M.; Borel, C.; Thamphya, B.; et al. Brentuximab vedotin and bendamustine as first-line treatment of Hodgkin lymphoma in the elderly (HALO Trial). J. Clin. Oncol. 2020, 38, 8029. [Google Scholar] [CrossRef]
- Yasenchak, C.A.; Bordoni, R.; Patel-Donnelly, D.; Larson, T.; Goldschmidt, J.; Boccia, R.V.; Cline, V.J.M.; Sacchi, M.; Forero-Torres, A.; Sims, R.B.; et al. Frontline Brentuximab Vedotin as Monotherapy or in Combination for Older Hodgkin Lymphoma Patients. Blood 2020, 136, 18–19. [Google Scholar] [CrossRef]
- LaCasce, A.S.; Bociek, R.G.; Sawas, A.; Caimi, P.; Agura, E.; Matous, J.; Ansell, S.M.; Crosswell, H.E.; Islas-Ohlmayer, M.; Behler, C.; et al. Brentuximab vedotin plus bendamustine: A highly active first salvage regimen for relapsed or refractory Hodgkin lymphoma. Blood 2018, 132, 40–48. [Google Scholar] [CrossRef]
- Advani, R.H.; Moskowitz, A.J.; Bartlett, N.L.; Vose, J.M.; Ramchandren, R.; Feldman, T.A.; LaCasce, A.S.; Christian, B.A.; Ansell, S.M.; Moskowitz, C.H.; et al. Brentuximab vedotin in combination with nivolumab in relapsed or refractory Hodgkin lymphoma: 3-year study results. Blood 2021, 138, 427–438. [Google Scholar] [CrossRef] [PubMed]
- Cheson, B.D.; Bartlett, N.L.; LaPlant, B.; Lee, H.J.; Advani, R.J.; Christian, B.; Diefenbach, C.S.; Feldman, T.A.; Ansell, S.M. Brentuximab vedotin plus nivolumab as first-line therapy in older or chemotherapy-ineligible patients with Hodgkin lymphoma (ACCRU): A multicentre, single-arm, phase 2 trial. Lancet Haematol. 2020, 7, e808–e815. [Google Scholar] [CrossRef] [PubMed]
- Lazarovici, J.; Amorim, S.; Bouabdallah, K.; Guidez, S.; Molina, L.; Morschhauser, F.; Gac, A.-C.; Gastinne, T.; Laribi, K.; Launay, V.; et al. Nivolumab First-Line Therapy for Elderly, Frail Hodgkin Lymphoma Patients: Niviniho, a Lysa Phase II Study. Blood 2021, 138, 232. [Google Scholar] [CrossRef]
- Dickinson, M.J.; Trotman, J.; Berkahn, L.; Butler, J.; Bressel, M.; Neeson, P.; Minson, A. Pembrolizumab as First Therapy for Hodgkin Lymphoma Is Deliverable in Older or Abvd-Ineligible Patients, Allows Subsequent Therapy, and Gives Adequate Survival. Hematol. Oncol. 2023, 41, 160–161. [Google Scholar] [CrossRef]
- Torka, P.; Feldman, T.; Savage, K.; Ganesan, N.; Hancock, H.; Davey, T.; Perez, L.; Santarosa, A.; Subzwari, S.; Capadona, C.; et al. Phase 2 Trial of Nivolumab Plus Adriamycin, Vinblastine, Dacarbazine (N-Avd) as Frontline Therapy in Older Adults with Hodgkin Lymphoma. Hematol. Oncol. 2023, 41, 161–162. [Google Scholar] [CrossRef]
- Liaskas, A.; Panitsas, F.; Kravvariti, E.; Angelopoulou, M.K.; Pangalis, G.A.; Vassilakopoulos, T.P. Very Late Relapses in Hodgkin Lymphoma: A Critical Comparison of Major Published Studies. Haema 2022, 13, 14–19. [Google Scholar]
- Song, Y.; Gao, Q.; Zhang, H.; Fan, L.; Zhou, J.; Zou, D.; Li, W.; Yang, H.; Liu, T.; Wang, Q.; et al. Treatment of relapsed or refractory classical Hodgkin lymphoma with the anti-PD-1, tislelizumab: Results of a phase 2, single-arm, multicenter study. Leukemia 2020, 34, 533–542. [Google Scholar] [CrossRef]
- Bröckelmann, P.J.; Kaul, H.; Fuchs, M.; Kobe, C.; Baues, C.; Borchmann, P.; Engert, A. P063: Trial in Progress: Individualized Immunotherapy in Early-Stage Unfavorable Hodgkin Lymphoma—The Investigator-Initiated Phase II GHSG INDIE Trial. HemaSphere 2022, 6, 29. [Google Scholar] [CrossRef]
- Radford, J.; Adedayo, T.; Ardavan, A.; Barrington, S.F.; Berkahn, L.; Chauvie, S.; Clifton-Hadley, L.; Collins, G.P.; Crump, M.; Cutter, D.; et al. P025: RADAR: An international phase III, PET response-adapted, randomised trial in progress, comparing ABVD±ISRT with brentuximab vedotin+AVD±ISRT in patients with previously untreated limited-stage classical Hodgkin lymphoma. HemaSphere 2022, 6, 12–13. [Google Scholar] [CrossRef]
- Spina, V.; Bruscaggin, A.; Cuccaro, A.; Martini, M.; Di Trani, M.; Forestieri, G.; Manzoni, M.; Condoluci, A.; Arribas, A.; Terzi-Di-Bergamo, L.; et al. Circulating tumor DNA reveals genetics, clonal evolution, and residual disease in classical Hodgkin lymphoma. Blood 2018, 131, 2413–2425. [Google Scholar] [CrossRef]
- Gallamini, A.; Picardi, M.; Filonenko, K.; Gotti, M.; Céspedes, J.N.; Rossi, A.; Domingo-Domènech, E.; Giza, A.; Sorasio, R.; Trentin, L.; et al. T021: Radiation-Free Therapy as the INitial treatment of Good-prognosis early non-bulky Hodgkin lymphoma, defined by a low Metabolic Tumor Volume and a negative PET-2—RAFTING Trial. HemaSphere 2022, 6, 10–11. [Google Scholar] [CrossRef]
- Vassilakopoulos, T.P.; Kravvariti, E.; Panitsas, F.; Angelopoulou, M.K.; Liaskas, A.; Kontopidou, F.N.; Yiakoumis, X.; Variami, E.; Dimopoulou, M.N.; Siakantaris, M.P.; et al. Very late relapses in Hodgkin lymphoma treated with chemotherapy with or without radiotherapy: Linear pattern and distinct prognostic factors. Blood Cancer J. 2022, 12, 102. [Google Scholar] [CrossRef] [PubMed]
- Andersen, M.D.; Hamilton-Dutoit, S.; Modvig, L.; Vase, M.; Christiansen, I.; Christensen, J.H.; Dahl-Sorensen, R.B.; Stoltenberg, D.; Kamper, P.; d’Amore, F. Late recurrence of lymphoid malignancies after initial treatment for Hodgkin lymphoma—A study from the Danish Lymphoma Registry. Br. J. Haematol. 2022, 198, 50–61. [Google Scholar] [CrossRef] [PubMed]
- Brockelmann, P.J.; Goergen, H.; Kohnhorst, C.; von Tresckow, B.; Moccia, A.; Markova, J.; Meissner, J.; Kerkhoff, A.; Ludwig, W.D.; Fuchs, M.; et al. Late Relapse of Classical Hodgkin Lymphoma: An Analysis of the German Hodgkin Study Group HD7 to HD12 Trials. J. Clin. Oncol. 2017, 35, 1444–1450. [Google Scholar] [CrossRef]



| Patients’ Characteristics and Key Outcome and Toxicity Measures | ECHELON-1 | SWOG S1826 | ||
|---|---|---|---|---|
| ABVD | BV-AVD | BV-AVD | N-AVD | |
| Patients (Ν) and patient characteristics [42,44,45] | 670 | 664 | 487 | 489 |
| Age (median (range)) | 37 (18–83) | 35 (18–82) | 26 (12–81) | 27 (12–83) |
| Stage IV (%) | 63 | 64 | 65 | 62 |
| IPS 4–7 (%) | 26 | 25 | 32 | 32 |
| Outcome measures [42,43,44,45] | ||||
| All patients | ||||
| 1-year PFS (%) * | 81.4 | 88.6 | 86 m | 94 m |
| 2-year mPFS per IRC (%) (primary endpoint) [42] | 77.2 a | 82.1 a | ND | ND |
| 6-year PFS per INV (%) [43] | 74.5 b | 82.3 b | NYA | NYA |
| 6-year OS (%) [43] | 89.4 c | 93.9 c | NYA | NYA |
| Stage IV or III subgroup | ||||
| 2-year mPFS per IRC, Stage IV (%) [42] | 74.9 d | 82.0 d | ND | ND |
| 6-year PFS per INV, stage III (%) [43] | NR e | NR e | NYA | NYA |
| 6-year PFS per INV, stage IV (%) [43] | NR f | NR f | NYA | NYA |
| 6-year OS, stage III (%) [43] | NR g | NR g | NYA | NYA |
| 6-year OS, stage IV (%) [43] | NR h | NR h | NYA | NYA |
| Interim PET negative status [44] | n = 578 | n = 588 | ||
| 5-year PFS per INV, all patients (%) | 78.9 i | 84.9 i | NYA | NYA |
| 5-year PFS per INV, <60 years old (%) | 81.5 j | 86.6 j | NYA | NYA |
| Interim PET positive status [44] | n = 58 | n = 47 | ||
| 5-year PFS per INV, all patients (%) | 45.9 k | 60.6 k | NYA | NYA |
| 5-year PFS per INV, <60 years old (%) | 49.3 l | 63.1 l | NYA | NYA |
| Toxicity [42,45] | ||||
| Toxic deaths, all patients [N (%)] | 13 (1.9) | 9 (1.4) | NR | 1 (<%) |
| Hospitalization, all patients (%) | 28 | 37 | NR | NR |
| Peripheral sensory neuropathy, all patients, all grades (%) | 17 | 29 | 55 | 29 |
| Peripheral sensory neuropathy, all patients, grade ≥ 3 (%) | <1 | 5 | 8 | 1 |
| Peripheral motor neuropathy, all patients, all grades (%) | 4 | 11 | 7 | 4 |
| Febrile neutropenia, all patients (%) | 8 | 19 | 7 | 5 |
| Febrile neutropenia, elderly (%) | 17 | 37 | - | - |
| Abramson J.S. et al., 2019 [54] | Abramson J.S. et al., 2023 [55] | Park S.I. et al., 2020 [56] | Kumar A. et al., 2021 [57] | Fornecker L.M. et al., 2022 [58] | |
|---|---|---|---|---|---|
| Patients (N) | 36 | 34 | 39 | 116 | 170 |
| Eligible stages | I/II non-bulky ◊ | I/II non-bulky ◊ | I/II non-bulky ± | Stage I, II unfavorable ¶ | Stage I/II unfavorable & |
| Initial treatment | BV-AVD × 2 | BV-AD × 2 | ABVD × 2 | BV-AVD × 2 | BV-AVD × 2 vs. ABVD × 2 (113 vs. 57 patients) |
| Further treatment | BV-AVD × 4 (#4) (not PET2 directed) | BV-AVD × 2 (#4) vs. ABVD × 2 (#4) (not PET2 directed) | |||
| PET2− | BV-AVD × 2 (#4) | BV-AD × 2 (#4) | F: BV × 6 U: ABVD × 2 (#4) + BV × 6 | ||
| PET2+ | BV-AVD × 4 (#6) | BV-AD × 4 (#6) | F: ABVD × 2 (#4) + BV × 6 U: ABVD × 4 (#6) + BV × 6 | ||
| PET2+ definition | DS 4–5 | DS 4–5 | DS 3–5 | DS 4–5 | DS 4–5 |
| PET2+ patients (N) | 0 | 6% | 28% | 13% | 17.7% vs. 24.6% |
| RT (% of patients) | No | No | Yes, for EoT PET+ (5%) | Yes, for cohorts 1–3 | Yes, for all patients after treatment (30 Gy) |
| PFS | 3-year: 94% | 5-year: 91% | 3-year: 92% (100% for patients with EoT PET−) | 2-year PFS: 93.1%, 96.6%, 89.7%, 96.6% for cohorts 1–4 | 2-year: 97.3% vs. 92.6% For PET2−: 97.8% vs. 97.7% For PET2+: 93.8 vs. 71.6% |
| OS | 3-year: 97% | 5-year: 96% | 3-year: 97% (100% for patients with EoT PET−) | 2-year: 99% in the whole population | NR |
| Toxicity (% of patients) | |||||
| PN grade ≥ 3 | 24% | 0% * | 2.5% | 3% | 3 vs. 2% |
| FN | 29% | 0% ** | NR | 8% | 8% vs. 6% |
| EoT PET-negative (% of patients) | 91.2% | 97% | 95% | 93%, 100%, 93%, and 97% for cohorts 1–4 | 88% vs. 77% |
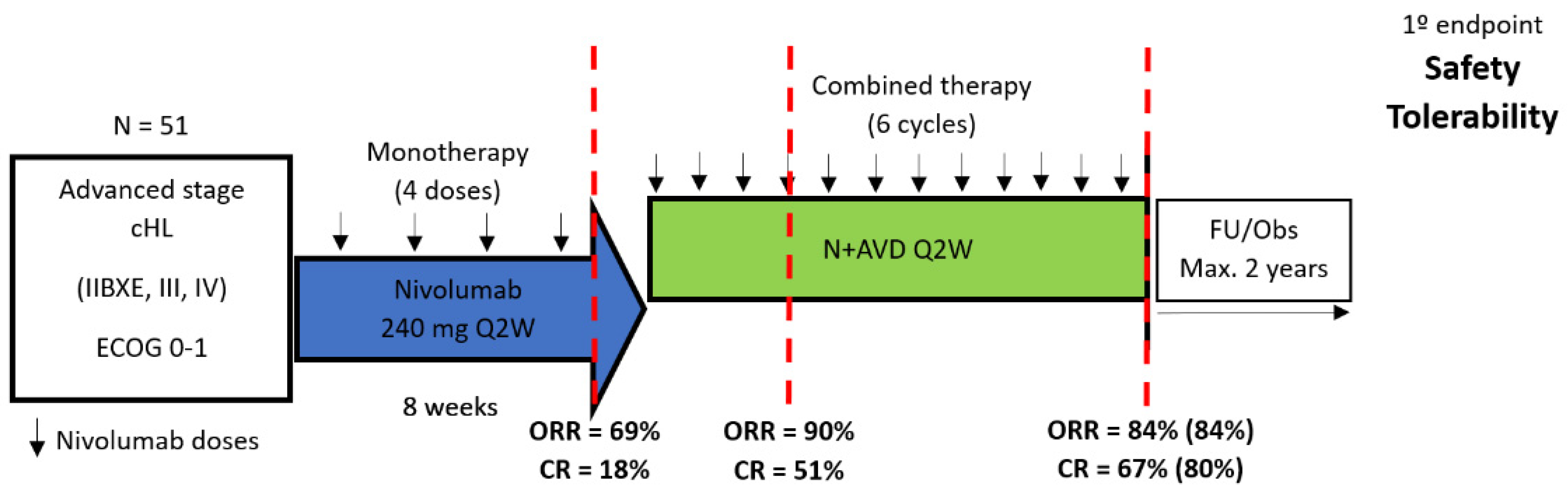
| Ramchandren R. et al., CHECKMATE-205, Arm D, 2019 [71] | Bröckelmann P.J. et al., NIVAHL, 2023 [72] | Allen P.B. et al., 2022 [73] | Lynch E.C. et al., 2023 [74] | |
|---|---|---|---|---|
| Patients (N) | 51 | 109 | 30 | 30 |
| Eligible stages | Advanced * (III/IV, IIBX/E) | I/II unfavorable * | III/IV, I/II unfavorable ¶¶ | All |
| CPI lead-in phase | Nivo × 4 | Nivo × 4 (Only Arm B) | Pembro × 3 | no |
| Response to CPI monotherapy [ORR (CR)] | 69% (18%) | NR (51%) | NR (37% + 23%) *** | NA |
| Overall Treatment Schedule | Nivo × 4 followed by AVD × 6 | Arm A: Nivo-AVD × 4 + 30 Gy ISRT Arm B: Nivo × 4 → Nivo-AVD × 2 → AVD × 2 + 30 Gy ISRT | Pembro × 3 followed by AVD × 4–6 ** | Pembro-AVD × 2–6 ** |
| CR rate at EoT | 80% | Arm A: 83% ¶ Arm B: 84% ¶ | 100% (at EoT) | 78% (at EoT) |
| PFS | 92% (9-month) | Arm A: 100% (3-year) Arm B: 98% (3-year) | 100% (2-year) | 97% (2-year) |
| OS | 98% (9-month) | 3-year OS 100% in both arms | 100% (2-year) | 100% (2-year) |
| Patients’ Characteristics and Outcomes | BV-AVD (Sequential Regimen) [94] | BV-AVD (Concomitant Regimen) [93] | ABVD Comparator [93] |
|---|---|---|---|
| Patients (N) | 48 | 84 | 102 |
| Age [years, median (range)] | 69 (60–88) | 68 (60–82) | 66 (60–83) |
| ECOG PS ≥ 2 | 19% | 12% | 10% |
| Ann Arbor Stage | |||
| II | 19% | 0% | 0% |
| III | 37% | 37% | 34% |
| IV | 44% | 61% | 66% |
| Efficacy | |||
| Response to BV × 2 [ORR (CR)] | 82% (36%) | NA | NA |
| Interim PET positive | 24% (10/42 pts) | 20% | 18% |
| EoT PET-negative | 90% (38/42 pts) | 71% | 74% |
| 2-year PFS | 84% | 70.3% * | 71.4% * |
| 5-year PFS | NA | 67.1% | 61.6% |
| 2-year OS | 93% | NA | NA |
| Toxicity | |||
| TRM | 2% | 3.6% | 5.1% |
| Neutropenia, grade ≥ 3 | 44% | 70% | 59% |
| Febrile neutropenia, grade ≥ 3 | 8% | 37% | 17% |
| Peripheral neuropathy | |||
| any grade | NA | 65% | 43% |
| grade ≥ 3 | 4% | 18% | 3% |
| grade 2 | 27% | 37% | 16% |
| resolution | 69% | 80% | 83% |
| Pulmonary toxicity, any grade | NA | 2% | 13% |
| Patients’ Characteristics, Outcome, and Toxicity | BV Monotherapy (BREVITY) [95] | BV Monotherapy [96] | BV-Dacarbazine [97] | BV-Bendamustine [97] | BV-Bendamustine (HALO) [98] | BV-Nivolumab [99] |
|---|---|---|---|---|---|---|
| Patients (total, N) | 35 | 27 | 22 | 20 | 60 | 20 |
| Patients (evaluable, N) | 31 | 26 | 19 | 17 | 59 | 19 |
| Eligibility criteria | Stage IIBX/III/IV unfit for standard CT * | ≥60 years old | ≥60 years old | ≥60 years old | Stage IIB/III/IV ≥60 years old | ≥60 years old |
| Follow-up time (median) | 3-years | 59.4 months | 58.6 months | 51.3 months | 20.6 months | 19.4 months |
| Treatment cycles | up to 16 | up to 16 | up to 12 plus BV ≥ 4 | up to 6 plus BV ≥ 10 | up to 6 | NA |
| Age [median (IQR or range)] | 77 (72–82) | 78 (64–92) | 69 (62–88) | 75 (63–86) | 70.32 (62–79) | 72 (NR–NR) |
| Ann Arbor Stage III, IV | 80% | 63% | 68% | 75% | 80% | 80% |
| ECOG PS ≥ 2 | 48% | 22% | 32% | 20% | 10% | 5% |
| B-symptoms | 71% | 22% | 29% | 41% | 68% | NA |
| CIRS [median (IQR or range)] | 5 (4, 7) | NA | NA | NA | NA | NA |
| TRM | 0.35% | 0% | 0% | 0% | NA | 0% |
| ORR | 84% ** | 92% *** | 100% *** | 100% *** | 63% | 95% |
| CMR | 26% ** | 73% *** | 62% *** | 88% *** | 80.36% | NA |
| PFS | ||||||
| Median | 7.3 months | 10.5 months | 46.8 months | 40.3 months | NR | NR |
| 1-year | 14% | ~35% | NA | NA | 84% | NA |
| 2-year | 7% | ~30% + | NA | NA | 54% | NA |
| OS | ||||||
| Median | 19.5 months | 77.5 months | 64 months | 46.9 months | 83% | NR |
| 1-year | 73% | NA | NA | NA | 97% | NA |
| 2-year | 42% | NA | NA | NA | 83% | NA |
| Peripheral neuropathy grade ≥ 3 | NR ++ | 30% & | 26% | 20% | 0% | 35% |
| Discontinuation rate | 29% | 42% | 40% | 40% | 30.5% | 30% |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vassilakopoulos, T.P.; Liaskas, A.; Pereyra, P.; Panayiotidis, P.; Angelopoulou, M.K.; Gallamini, A. Incorporating Monoclonal Antibodies into the First-Line Treatment of Classical Hodgkin Lymphoma. Int. J. Mol. Sci. 2023, 24, 13187. https://doi.org/10.3390/ijms241713187
Vassilakopoulos TP, Liaskas A, Pereyra P, Panayiotidis P, Angelopoulou MK, Gallamini A. Incorporating Monoclonal Antibodies into the First-Line Treatment of Classical Hodgkin Lymphoma. International Journal of Molecular Sciences. 2023; 24(17):13187. https://doi.org/10.3390/ijms241713187
Chicago/Turabian StyleVassilakopoulos, Theodoros P., Athanasios Liaskas, Patricio Pereyra, Panayiotis Panayiotidis, Maria K. Angelopoulou, and Andrea Gallamini. 2023. "Incorporating Monoclonal Antibodies into the First-Line Treatment of Classical Hodgkin Lymphoma" International Journal of Molecular Sciences 24, no. 17: 13187. https://doi.org/10.3390/ijms241713187
APA StyleVassilakopoulos, T. P., Liaskas, A., Pereyra, P., Panayiotidis, P., Angelopoulou, M. K., & Gallamini, A. (2023). Incorporating Monoclonal Antibodies into the First-Line Treatment of Classical Hodgkin Lymphoma. International Journal of Molecular Sciences, 24(17), 13187. https://doi.org/10.3390/ijms241713187







