TDP-43 Epigenetic Facets and Their Neurodegenerative Implications
Abstract
:1. Introduction
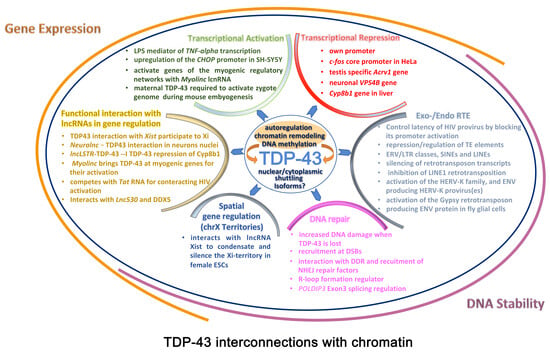
2. TDP-43 Role in Chromatin Remodeling and Transcription
2.1. TDP-43 Is a Global Chromatin Modifier
2.2. TDP-43 and Local/Specific Gene Transcriptional Regulation
2.3. TDP-43 and Genome-Wide Transcriptional Regulation
2.4. TDP-43 and DNA Repair
2.5. TDP-43 and Regulation of the Genome Dark Matter
2.5.1. The Singular Case of Human HERV-K Env Protein Activated by TDP-43
2.5.2. TDP-43 Control of LINE1 Retrotransposition
2.5.3. Conservation of TDP-43/TDPH Regulation of TE in Drosophila

2.6. ALS and Epigenetic Functionality of TDP-43 Short Splicing Isoforms
2.7. Epigenetic Role of TDP-43 Alternative Forms
2.8. Epigenetic Landscape Modifications Associated with TDP-43 Mutants
3. TARDBP/TDP-43 Autonomous/Non-autonomous Regulation
3.1. TDP-43 Autoregulation
3.2. Production of Alternatively Spliced Protein-Coding Isoforms
3.3. Promoter Control of Pan TARDBP Expression
4. Potential Druggable Targets
5. Concluding Remarks
6. Resources for the Review
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Berson, A.; Nativio, R.; Berger, S.L.; Bonini, N.M. Epigenetic Regulation in Neurodegenerative Diseases. Trends Neurosci. 2018, 41, 587–598. [Google Scholar] [CrossRef] [PubMed]
- Bennett, S.A.; Tanaz, R.; Cobos, S.N.; Torrente, M.P. Epigenetics in amyotrophic lateral sclerosis: A role for histone post-translational modifications in neurodegenerative disease. Transl. Res. 2019, 204, 19–30. [Google Scholar] [CrossRef]
- Basavarajappa, B.S.; Subbanna, S. Histone Methylation Regulation in Neurodegenerative Disorders. Int. J. Mol. Sci. 2021, 22, 4654. [Google Scholar] [CrossRef] [PubMed]
- Bodai, L. Epigenetic Regulation in Neurodegeneration Disease. Int. J. Mol. Sci. 2022, 23, 6185. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Ren, J.; Zhang, M.; Wang, H.; Yi, F.; Wu, J.; Tang, Y. The heterogeneity of microglial activation and its epigenetic and non-coding RNA regulations in the immunopathogenesis of neurodegenerative diseases. Cell. Mol. Life Sci. 2022, 79, 511. [Google Scholar] [CrossRef]
- Sharma, R.; Sharma, S.; Thakur, A.; Singh, A.; Singh, J.; Nepali, K.; Liou, J.P. The Role of Epigenetic Mechanisms in Autoimmune, Neurodegenerative, Cardiovascular, and Imprinting Disorders. Mini Rev. Med. Chem. 2022, 22, 1977–2011. [Google Scholar] [CrossRef]
- Neumann, M.; Sampathu, D.M.; Kwong, L.K.; Truax, A.C.; Micsenyi, M.C.; Chou, T.T.; Bruce, J.; Schuck, T.; Grossman, M.; Clark, C.M.; et al. Ubiquitinated TDP-43 in Frontotemporal Lobar Degeneration and Amyotrophic Lateral Sclerosis. Science 2006, 314, 130–133. [Google Scholar] [CrossRef]
- Arai, T.; Hasegawa, M.; Akiyama, H.; Ikeda, K.; Nonaka, T.; Mori, H.; Mann, D.; Tsuchiya, K.; Yoshida, M.; Hashizume, Y.; et al. TDP-43 is a component of ubiquitin-positive tau-negative inclusions in frontotemporal lobar degeneration and amyotrophic lateral sclerosis. Biochem. Biophys. Res. Commun. 2006, 351, 602–611. [Google Scholar] [CrossRef]
- Prasad, A.; Bharathi, V.; Sivalingam, V.; Girdhar, A.; Patel, B.K. Molecular Mechanisms of TDP-43 Misfolding and Pathology in Amyotrophic Lateral Sclerosis. Front. Mol. Neurosci. 2019, 12, 25. [Google Scholar] [CrossRef]
- Schwab, C.; Arai, T.; Hasegawa, M.; Yu, S.; McGeer, P.L. Colocalization of Transactivation-Responsive DNA-Binding Protein 43 and Huntingtin in Inclusions of Huntington Disease. J. Neuropathol. Exp. Neurol. 2008, 67, 1159–1165. [Google Scholar] [CrossRef]
- Sanchez, I.I.; Nguyen, T.B.; England, W.E.; Lim, R.G.; Vu, A.Q.; Miramontes, R.; Byrne, L.M.; Markmiller, S.; Lau, A.L.; Orellana, I.; et al. Huntington’s disease mice and human brain tissue exhibit increased G3BP1 granules and TDP43 mislocalization. J. Clin. Investig. 2021, 131, 140723. [Google Scholar] [CrossRef] [PubMed]
- Johnson, V.E.; Stewart, W.; Trojanowski, J.Q.; Smith, D.H. Acute and chronically increased immunoreactivity to phosphorylation-independent but not pathological TDP-43 after a single traumatic brain injury in humans. Acta Neuropathol. 2011, 122, 715–726. [Google Scholar] [CrossRef] [PubMed]
- Gao, F.; Hu, M.; Zhang, J.; Hashem, J.; Chen, C. TDP-43 drives synaptic and cognitive deterioration following traumatic brain injury. Acta Neuropathol. 2022, 144, 187–210. [Google Scholar] [CrossRef] [PubMed]
- De Boer, E.M.J.; Orie, V.K.; Williams, T.; Baker, M.R.; De Oliveira, H.M.; Polvikoski, T.; Silsby, M.; Menon, P.; van den Bos, M.; Halliday, G.M.; et al. TDP-43 proteinopathies: A new wave of neurodegenerative diseases. J. Neurol. Neurosurg. Psychiatry 2020, 92, 86–95. [Google Scholar] [CrossRef]
- Ou, S.H.; Wu, F.; Harrich, D.; García-Martínez, L.F.; Gaynor, R.B. Cloning and characterization of a novel cellular protein, TDP-43, that binds to human immunodeficiency virus type 1 TAR DNA sequence motifs. J. Virol. 1995, 69, 3584–3596. [Google Scholar] [CrossRef]
- Buratti, E.; Baralle, F.E. Characterization and Functional Implications of the RNA Binding Properties of Nuclear Factor TDP-43, a Novel Splicing Regulator ofCFTR Exon 9. J. Biol. Chem. 2001, 276, 36337–36343. [Google Scholar] [CrossRef]
- Buratti, E.; Dörk, T.; Zuccato, E.; Pagani, F.; Romano, M.; Baralle, F.E. Nuclear factor TDP-43 and SR proteins promote in vitro and in vivo CFTR exon 9 skipping. EMBO J. 2001, 20, 1774–1784. [Google Scholar] [CrossRef]
- Ayala, Y.M.; Pantano, S.; D’Ambrogio, A.; Buratti, E.; Brindisi, A.; Marchetti, C.; Romano, M.; Baralle, F.E. Human, Drosophila, and C. elegans TDP43: Nucleic Acid Binding Properties and Splicing Regulatory Function. J. Mol. Biol. 2005, 348, 575–588. [Google Scholar] [CrossRef]
- Cassel, J.A.; Blass, B.E.; Reitz, A.B.; Pawlyk, A.C. Development of a Novel Nonradiometric Assay for Nucleic Acid Binding to TDP-43 Suitable for High-Throughput Screening Using AlphaScreen® Technology. J. Biomol. Screen. 2010, 15, 1099–1106. [Google Scholar] [CrossRef]
- Kuo, P.-H.; Chiang, C.-H.; Wang, Y.-T.; Doudeva, L.G.; Yuan, H.S. The crystal structure of TDP-43 RRM1-DNA complex reveals the specific recognition for UG- and TG-rich nucleic acids. Nucleic Acids Res. 2014, 42, 4712–4722. [Google Scholar] [CrossRef]
- Qin, H.; Lim, L.-Z.; Wei, Y.; Song, J. TDP-43 N terminus encodes a novel ubiquitin-like fold and its unfolded form in equilibrium that can be shifted by binding to ssDNA. Proc. Natl. Acad. Sci. USA 2014, 111, 18619–18624. [Google Scholar] [CrossRef] [PubMed]
- Furukawa, Y.; Suzuki, Y.; Fukuoka, M.; Nagasawa, K.; Nakagome, K.; Shimizu, H.; Mukaiyama, A.; Akiyama, S. A molecular mechanism realizing sequence-specific recognition of nucleic acids by TDP-43. Sci. Rep. 2016, 6, 20576. [Google Scholar] [CrossRef] [PubMed]
- Mitra, J.; Guerrero, E.N.; Hegde, P.M.; Liachko, N.F.; Wang, H.; Vasquez, V.; Gao, J.; Pandey, A.; Taylor, J.P.; Kraemer, B.C.; et al. Motor neuron disease-associated loss of nuclear TDP-43 is linked to DNA double-strand break repair defects. Proc. Natl. Acad. Sci. USA 2019, 116, 4696–4705. [Google Scholar] [CrossRef] [PubMed]
- Ayala, Y.M.; Zago, P.; D’Ambrogio, A.; Xu, Y.-F.; Petrucelli, L.; Buratti, E.; Baralle, F.E. Structural determinants of the cellular localization and shuttling of TDP-43. J. Cell Sci. 2008, 121, 3778–3785. [Google Scholar] [CrossRef]
- Alami, N.H.; Smith, R.B.; Carrasco, M.A.; Williams, L.A.; Winborn, C.S.; Han, S.S.; Kiskinis, E.; Winborn, B.; Freibaum, B.D.; Kanagaraj, A.; et al. Axonal Transport of TDP-43 mRNA Granules Is Impaired by ALS-Causing Mutations. Neuron 2014, 81, 536–543. [Google Scholar] [CrossRef]
- Briese, M.; Saal-Bauernschubert, L.; Lüningschrör, P.; Moradi, M.; Dombert, B.; Surrey, V.; Appenzeller, S.; Deng, C.; Jablonka, S.; Sendtner, M. Loss of Tdp-43 disrupts the axonal transcriptome of motoneurons accompanied by impaired axonal translation and mitochondria function. Acta Neuropathol. Commun. 2020, 8, 116. [Google Scholar] [CrossRef]
- Nagano, S.; Jinno, J.; Abdelhamid, R.F.; Jin, Y.; Shibata, M.; Watanabe, S.; Hirokawa, S.; Nishizawa, M.; Sakimura, K.; Onodera, O.; et al. TDP-43 transports ribosomal protein mRNA to regulate axonal local translation in neuronal axons. Acta Neuropathol. 2020, 140, 695–713. [Google Scholar] [CrossRef]
- Nagano, S.; Araki, T. Axonal Transport and Local Translation of MRNA in Amyotrophic Lateral Sclerosis. In Amyotrophic Lateral Sclerosis; Araki, T., Ed.; Exon Publications: Brisbane, Australia, 2021; ISBN 978-0-645-00177-8. [Google Scholar]
- Winton, M.J.; Igaz, L.M.; Wong, M.M.; Kwong, L.K.; Trojanowski, J.Q.; Lee, V.M.-Y. Disturbance of Nuclear and Cytoplasmic TAR DNA-binding Protein (TDP-43) Induces Disease-like Redistribution, Sequestration, and Aggregate Formation. J. Biol. Chem. 2008, 283, 13302–13309. [Google Scholar] [CrossRef]
- Johnson, B.S.; Snead, D.; Lee, J.J.; McCaffery, J.M.; Shorter, J.; Gitler, A.D. TDP-43 Is Intrinsically Aggregation-prone, and Amyotrophic Lateral Sclerosis-linked Mutations Accelerate Aggregation and Increase Toxicity. J. Biol. Chem. 2009, 284, 20329–20339. [Google Scholar] [CrossRef]
- Berning, B.A.; Walker, A.K. The Pathobiology of TDP-43 C-Terminal Fragments in ALS and FTLD. Front. Neurosci. 2019, 13, 335. [Google Scholar] [CrossRef]
- Buratti, E.; Baralle, F.E. The multiple roles of TDP-43 in pre-mRNA processing and gene expression regulation. RNA Biol. 2010, 7, 420–429. [Google Scholar] [CrossRef] [PubMed]
- Buratti, E.; De Conti, L.; Stuani, C.; Romano, M.; Baralle, M.; Baralle, F. Nuclear factor TDP-43 can affect selected microRNA levels. FEBS J. 2010, 277, 2268–2281. [Google Scholar] [CrossRef] [PubMed]
- Polymenidou, M.; Lagier-Tourenne, C.; Hutt, K.R.; Huelga, S.C.; Moran, J.; Liang, T.Y.; Ling, S.-C.; Sun, E.; Wancewicz, E.; Mazur, C.; et al. Long pre-mRNA depletion and RNA missplicing contribute to neuronal vulnerability from loss of TDP-43. Nat. Neurosci. 2011, 14, 459–468. [Google Scholar] [CrossRef]
- Tollervey, J.R.; Curk, T.; Rogelj, B.; Briese, M.; Cereda, M.; Kayikci, M.; König, J.; Hortobágyi, T.; Nishimura, A.L.; Župunski, V.; et al. Characterizing the RNA targets and position-dependent splicing regulation by TDP-43. Nat. Neurosci. 2011, 14, 452–458. [Google Scholar] [CrossRef]
- Kawahara, Y.; Mieda-Sato, A. TDP-43 promotes microRNA biogenesis as a component of the Drosha and Dicer complexes. Proc. Natl. Acad. Sci. USA 2012, 109, 3347–3352. [Google Scholar] [CrossRef]
- Rathore, A.; Iketani, S.; Wang, P.; Jia, M.; Sahi, V.; Ho, D.D. CRISPR-based gene knockout screens reveal deubiquitinases involved in HIV-1 latency in two Jurkat cell models. Sci. Rep. 2020, 10, 5350. [Google Scholar] [CrossRef]
- Acharya, K.K.; Govind, C.K.; Shore, A.N.; Stoler, M.H.; Reddi, P.P. cis-Requirement for the maintenance of round spermatid-specific transcription. Dev. Biol. 2006, 295, 781–790. [Google Scholar] [CrossRef]
- Abhyankar, M.M.; Urekar, C.; Reddi, P.P. A Novel CpG-free Vertebrate Insulator Silences the Testis-specific SP-10 Gene in Somatic Tissues: Role for TDP-43 in Insulator Function. J. Biol. Chem. 2007, 282, 36143–36154. [Google Scholar] [CrossRef]
- Lalmansingh, A.S.; Urekar, C.J.; Reddi, P.P. TDP-43 Is a Transcriptional Repressor: The Testis-Specific Mouse Acrv1 Gene Is a TDP-43 Target in Vivo. J. Biol. Chem. 2011, 286, 10970–10982. [Google Scholar] [CrossRef]
- Schwenk, B.M.; Hartmann, H.; Serdaroglu, A.; Schludi, M.H.; Hornburg, D.; Meissner, F.; Orozco, D.; Colombo, A.; Tahirovic, S.; Michaelsen, M.; et al. TDP-43 loss of function inhibits endosomal trafficking and alters trophic signaling in neurons. EMBO J. 2016, 35, 2350–2370. [Google Scholar] [CrossRef]
- Militello, G.; Hosen, M.R.; Ponomareva, Y.; Gellert, P.; Weirick, T.; John, D.; Hindi, S.M.; Mamchaoui, K.; Mouly, V.; Döring, C.; et al. A novel long non-coding RNA Myolinc regulates myogenesis through TDP-43 and Filip1. J. Mol. Cell Biol. 2018, 10, 102–117. [Google Scholar] [CrossRef] [PubMed]
- Keihani, S.; Kluever, V.; Mandad, S.; Bansal, V.; Rahman, R.; Fritsch, E.; Gomes, L.C.; Gärtner, A.; Kügler, S.; Urlaub, H.; et al. The long noncoding RNA neuroLNC regulates presynaptic activity by interacting with the neurodegeneration-associated protein TDP-43. Sci. Adv. 2019, 5, eaay2670. [Google Scholar] [CrossRef] [PubMed]
- Pandya-Jones, A.; Markaki, Y.; Serizay, J.; Chitiashvili, T.; Leon, W.R.M.; Damianov, A.; Chronis, C.; Papp, B.; Chen, C.-K.; McKee, R.; et al. A protein assembly mediates Xist localization and gene silencing. Nature 2020, 587, 145–151. [Google Scholar] [CrossRef]
- Li, P.; Ruan, X.; Yang, L.; Kiesewetter, K.; Zhao, Y.; Luo, H.; Chen, Y.; Gucek, M.; Zhu, J.; Cao, H. A Liver-Enriched Long Non-Coding RNA, lncLSTR, Regulates Systemic Lipid Metabolism in Mice. Cell Metab. 2015, 21, 455–467. [Google Scholar] [CrossRef]
- Murata, H.; Hattori, T.; Maeda, H.; Takashiba, S.; Takigawa, M.; Kido, J.; Nagata, T. Identification of transactivation-responsive DNA-binding protein 43 (TARDBP43; TDP-43) as a novel factor for TNF-α expression upon lipopolysaccharide stimulation in human monocytes. J. Periodontal Res. 2015, 50, 452–460. [Google Scholar] [CrossRef]
- Suzuki, H.; Matsuoka, M. TDP-43 toxicity is mediated by the unfolded protein response-unrelated induction of C/EBP homologous protein expression. J. Neurosci. Res. 2012, 90, 641–647. [Google Scholar] [CrossRef] [PubMed]
- Sanna, S.; Esposito, S.; Masala, A.; Sini, P.; Nieddu, G.; Galioto, M.; Fais, M.; Iaccarino, C.; Cestra, G.; Crosio, C. HDAC1 inhibition ameliorates TDP-43-induced cell death in vitro and in vivo. Cell Death Dis. 2020, 11, 369. [Google Scholar] [CrossRef]
- Li, J.; Zhao, W.; Li, Q.; Huang, Z.; Shi, G.; Li, C. Long Non-Coding RNA H19 Promotes Porcine Satellite Cell Differentiation by Interacting with TDP43. Genes 2020, 11, 259. [Google Scholar] [CrossRef]
- Sreedharan, J.; Neukomm, L.J.; Brown, R.H.; Freeman, M.R. Age-Dependent TDP-43-Mediated Motor Neuron Degeneration Requires GSK3, hat-trick, and xmas-2. Curr. Biol. 2015, 25, 2130–2136. [Google Scholar] [CrossRef]
- Berson, A.; Sartoris, A.; Nativio, R.; Van Deerlin, V.; Toledo, J.B.; Porta, S.; Liu, S.; Chung, C.-Y.; Garcia, B.A.; Lee, V.M.-Y.; et al. TDP-43 Promotes Neurodegeneration by Impairing Chromatin Remodeling. Curr. Biol. 2017, 27, 3579–3590.e6. [Google Scholar] [CrossRef]
- Azpurua, J.; El-Karim, E.G.; Tranquille, M.; Dubnau, J. A behavioral screen for mediators of age-dependent TDP-43 neurodegeneration identifies SF2/SRSF1 among a group of potent suppressors in both neurons and glia. PLoS Genet. 2021, 17, e1009882. [Google Scholar] [CrossRef]
- Saldi, T.K.; Gonzales, P.; Garrido-Lecca, A.; Dostal, V.; Roberts, C.M.; Petrucelli, L.; Link, C.D. The Caenorhabditis elegans Ortholog of TDP-43 Regulates the Chromatin Localization of the Heterochromatin Protein 1 Homolog HPL-2. Mol. Cell. Biol. 2018, 38, e00668-17. [Google Scholar] [CrossRef] [PubMed]
- Moujalled, D.; James, J.L.; Parker, S.J.; Lidgerwood, G.E.; Duncan, C.; Meyerowitz, J.; Nonaka, T.; Hasegawa, M.; Kanninen, K.M.; Grubman, A.; et al. Kinase Inhibitor Screening Identifies Cyclin-Dependent Kinases and Glycogen Synthase Kinase 3 as Potential Modulators of TDP-43 Cytosolic Accumulation during Cell Stress. PLoS ONE 2013, 8, e67433. [Google Scholar] [CrossRef] [PubMed]
- Ambegaokar, S.S.; Jackson, G.R. Functional genomic screen and network analysis reveal novel modifiers of tauopathy dissociated from tau phosphorylation. Hum. Mol. Genet. 2011, 20, 4947–4977. [Google Scholar] [CrossRef] [PubMed]
- Šušnjar, U.; Škrabar, N.; Brown, A.-L.; Abbassi, Y.; Phatnani, H.; NYGC ALS Consortium; Cortese, A.; Cereda, C.; Bugiardini, E.; Cardani, R.; et al. Cell environment shapes TDP-43 function with implications in neuronal and muscle disease. Commun. Biol. 2022, 5, 314. [Google Scholar] [CrossRef]
- Tibshirani, M.; Zhao, B.; Gentil, B.J.; Minotti, S.; Marques, C.; Keith, J.; Rogaeva, E.; Zinman, L.; Rouaux, C.; Robertson, J.; et al. Dysregulation of chromatin remodelling complexes in amyotrophic lateral sclerosis. Hum. Mol. Genet. 2017, 26, 4142–4152. [Google Scholar] [CrossRef]
- Masala, A.; Sanna, S.; Esposito, S.; Rassu, M.; Galioto, M.; Zinellu, A.; Carru, C.; Carrì, M.T.; Iaccarino, C.; Crosio, C. Epigenetic Changes Associated with the Expression of Amyotrophic Lateral Sclerosis (ALS) Causing Genes. Neuroscience 2018, 390, 1–11. [Google Scholar] [CrossRef]
- Fiesel, F.C.; Voigt, A.; Weber, S.S.; Van den Haute, C.; Waldenmaier, A.; Görner, K.; Walter, M.; Anderson, M.L.; Kern, J.V.; Rasse, T.M.; et al. Knockdown of transactive response DNA-binding protein (TDP-43) downregulates histone deacetylase 6. EMBO J. 2010, 29, 209–221. [Google Scholar] [CrossRef]
- Kim, S.H.; Shanware, N.P.; Bowler, M.J.; Tibbetts, R.S. Amyotrophic Lateral Sclerosis-associated Proteins TDP-43 and FUS/TLS Function in a Common Biochemical Complex to Co-regulate HDAC6 mRNA*. J. Biol. Chem. 2010, 285, 34097–34105. [Google Scholar] [CrossRef]
- Cohen, T.J.; Hwang, A.W.; Restrepo, C.R.; Yuan, C.-X.; Trojanowski, J.Q.; Lee, V.M.Y. An acetylation switch controls TDP-43 function and aggregation propensity. Nat. Commun. 2015, 6, 5845. [Google Scholar] [CrossRef]
- Lee, S.; Kwon, Y.; Kim, S.; Jo, M.; Jeon, Y.-M.; Cheon, M.; Lee, S.; Kim, S.R.; Kim, K.; Kim, H.-J. The Role of HDAC6 in TDP-43-Induced Neurotoxicity and UPS Impairment. Front. Cell Dev. Biol. 2020, 8, 581942. [Google Scholar] [CrossRef] [PubMed]
- Igaz, L.M.; Kwong, L.K.; Lee, E.B.; Chen-Plotkin, A.; Swanson, E.; Unger, T.; Malunda, J.; Xu, Y.; Winton, M.J.; Trojanowski, J.Q.; et al. Dysregulation of the ALS-associated gene TDP-43 leads to neuronal death and degeneration in mice. J. Clin. Investig. 2011, 121, 726–738. [Google Scholar] [CrossRef] [PubMed]
- Amlie-Wolf, A.; Ryvkin, P.; Tong, R.; Dragomir, I.; Suh, E.; Xu, Y.; Van Deerlin, V.M.; Gregory, B.D.; Kwong, L.K.; Trojanowski, J.Q.; et al. Transcriptomic Changes Due to Cytoplasmic TDP-43 Expression Reveal Dysregulation of Histone Transcripts and Nuclear Chromatin. PLoS ONE 2015, 10, e0141836. [Google Scholar] [CrossRef]
- Liu, E.Y.; Russ, J.; Cali, C.P.; Phan, J.M.; Amlie-Wolf, A.; Lee, E.B. Loss of Nuclear TDP-43 Is Associated with Decondensation of LINE Retrotransposons. Cell Rep. 2019, 27, 1409–1421.e6. [Google Scholar] [CrossRef] [PubMed]
- Bird, A. DNA methylation patterns and epigenetic memory. Genes Dev. 2002, 16, 6–21. [Google Scholar] [CrossRef]
- Moore, L.D.; Le, T.; Fan, G. DNA Methylation and Its Basic Function. Neuropsychopharmacology 2013, 38, 23–38. [Google Scholar] [CrossRef]
- Gavin, D.P.; Chase, K.A.; Sharma, R.P. Active DNA demethylation in post-mitotic neurons: A reason for optimism. Neuropharmacology 2013, 75, 233–245. [Google Scholar] [CrossRef]
- Guo, J.U.; Su, Y.; Shin, J.H.; Shin, J.; Li, H.; Xie, B.; Zhong, C.; Hu, S.; Le, T.; Fan, G.; et al. Distribution, recognition and regulation of non-CpG methylation in the adult mammalian brain. Nat. Neurosci. 2013, 17, 215–222. [Google Scholar] [CrossRef]
- Robertson, K.D.; Wolffe, A.P. DNA methylation in health and disease. Nat. Rev. Genet. 2000, 1, 11–19. [Google Scholar] [CrossRef]
- Wu, H.; Coskun, V.; Tao, J.; Xie, W.; Ge, W.; Yoshikawa, K.; Li, E.; Zhang, Y.; Sun, Y.E. Dnmt3a-Dependent Nonpromoter DNA Methylation Facilitates Transcription of Neurogenic Genes. Science 2010, 329, 444–448. [Google Scholar] [CrossRef]
- Wang, J.; Zhang, K.X.; Lu, G.Z.; Zhao, X.H. Research progress on 5hmC and TET dioxygenases in neurodevelopment and neurological diseases. Yi Chuan = Hered. 2017, 39, 1138–1149. [Google Scholar]
- Appleby-Mallinder, C.; Schaber, E.; Kirby, J.; Shaw, P.J.; Cooper-Knock, J.; Heath, P.R.; Highley, J.R. TDP43 proteinopathy is associated with aberrant DNA methylation in human amyotrophic lateral sclerosis. Neuropathol. Appl. Neurobiol. 2020, 47, 61–72. [Google Scholar] [CrossRef] [PubMed]
- Catanese, A.; Rajkumar, S.; Sommer, D.; Masrori, P.; Hersmus, N.; Van Damme, P.; Witzel, S.; Ludolph, A.; Ho, R.; Boeckers, T.M.; et al. Multiomics and Machine-Learning Identify Novel Transcriptional and Mutational Signatures in Amyotrophic Lateral Sclerosis. Brain J. Neurol. 2023, awad075. [Google Scholar] [CrossRef]
- Sephton, C.F.; Cenik, C.; Kucukural, A.; Dammer, E.B.; Cenik, B.; Han, Y.; Dewey, C.M.; Roth, F.P.; Herz, J.; Peng, J.; et al. Identification of Neuronal RNA Targets of TDP-43-containing Ribonucleoprotein Complexes. J. Biol. Chem. 2011, 286, 1204–1215. [Google Scholar] [CrossRef]
- Sharifi, O.; Yasui, D.H. The Molecular Functions of MeCP2 in Rett Syndrome Pathology. Front. Genet. 2021, 12, 624290. [Google Scholar] [CrossRef]
- De Conti, L.; Akinyi, M.V.; Mendoza-Maldonado, R.; Romano, M.; Baralle, M.; Buratti, E. TDP-43 affects splicing profiles and isoform production of genes involved in the apoptotic and mitotic cellular pathways. Nucleic Acids Res. 2015, 43, 8990–9005. [Google Scholar] [CrossRef]
- Appocher, C.; Mohagheghi, F.; Cappelli, S.; Stuani, C.; Romano, M.; Feiguin, F.; Buratti, E. Major hnRNP proteins act as general TDP-43 functional modifiers both in Drosophila and human neuronal cells. Nucleic Acids Res. 2017, 45, 8026–8045. [Google Scholar] [CrossRef]
- Chung, C.-Y.; Berson, A.; Kennerdell, J.R.; Sartoris, A.; Unger, T.; Porta, S.; Kim, H.-J.; Smith, E.R.; Shilatifard, A.; Van Deerlin, V.; et al. Aberrant activation of non-coding RNA targets of transcriptional elongation complexes contributes to TDP-43 toxicity. Nat. Commun. 2018, 9, 4406. [Google Scholar] [CrossRef]
- Swarup, V.; Phaneuf, D.; Dupré, N.; Petri, S.; Strong, M.; Kriz, J.; Julien, J.-P. Deregulation of TDP-43 in amyotrophic lateral sclerosis triggers nuclear factor κB–mediated pathogenic pathways. J. Exp. Med. 2011, 208, 2429–2447. [Google Scholar] [CrossRef]
- Janssen, C.; Schmalbach, S.; Boeselt, S.; Sarlette, A.; Dengler, R.; Petri, S. Differential Histone Deacetylase mRNA Expression Patterns in Amyotrophic Lateral Sclerosis. J. Neuropathol. Exp. Neurol. 2010, 69, 573–581. [Google Scholar] [CrossRef]
- Casafont, I.; Bengoechea, R.; Tapia, O.; Berciano, M.T.; Lafarga, M. TDP-43 localizes in mRNA transcription and processing sites in mammalian neurons. J. Struct. Biol. 2009, 167, 235–241. [Google Scholar] [CrossRef] [PubMed]
- Swain, A.; Misulovin, Z.; Pherson, M.; Gause, M.; Mihindukulasuriya, K.; Rickels, R.A.; Shilatifard, A.; Dorsett, D. Drosophila TDP-43 RNA-Binding Protein Facilitates Association of Sister Chromatid Cohesion Proteins with Genes, Enhancers and Polycomb Response Elements. PLOS Genet. 2016, 12, e1006331. [Google Scholar] [CrossRef] [PubMed]
- Davidovich, C.; Cech, T.R. The recruitment of chromatin modifiers by long noncoding RNAs: Lessons from PRC2. RNA 2015, 21, 2007–2022. [Google Scholar] [CrossRef] [PubMed]
- Schuettengruber, B.; Bourbon, H.-M.; Di Croce, L.; Cavalli, G. Genome Regulation by Polycomb and Trithorax: 70 Years and Counting. Cell 2017, 171, 34–57. [Google Scholar] [CrossRef]
- Laugesen, A.; Højfeldt, J.W.; Helin, K. Molecular Mechanisms Directing PRC2 Recruitment and H3K27 Methylation. Mol. Cell 2019, 74, 8–18. [Google Scholar] [CrossRef]
- Owen, B.M.; Davidovich, C. DNA binding by polycomb-group proteins: Searching for the link to CpG islands. Nucleic Acids Res. 2022, 50, 4813–4839. [Google Scholar] [CrossRef]
- Wang, X.; Goodrich, K.J.; Conlon, E.G.; Gao, J.; Erbse, A.H.; Manley, J.L.; Cech, T.R. C9orf72 and triplet repeat disorder RNAs: G-quadruplex formation, binding to PRC2 and implications for disease mechanisms. RNA 2019, 25, 935–947. [Google Scholar] [CrossRef]
- Morera, A.A.; Ahmed, N.S.; Schwartz, J.C. TDP-43 regulates transcription at protein-coding genes and Alu retrotransposons. Biochim. Biophys. Acta Gene Regul. Mech. 2019, 1862, 194434. [Google Scholar] [CrossRef]
- Maor-Nof, M.; Shipony, Z.; Lopez-Gonzalez, R.; Nakayama, L.; Zhang, Y.-J.; Couthouis, J.; Blum, J.A.; Castruita, P.A.; Linares, G.R.; Ruan, K.; et al. p53 is a central regulator driving neurodegeneration caused by C9orf72 poly(PR). Cell 2021, 184, 689–708.e20. [Google Scholar] [CrossRef]
- Nie, X.; Xu, Q.; Xu, C.; Chen, F.; Wang, Q.; Qin, D.; Wang, R.; Gao, Z.; Lu, X.; Yang, X.; et al. Maternal TDP-43 interacts with RNA Pol II and regulates zygotic genome activation. Nat. Commun. 2023, 14, 4275. [Google Scholar] [CrossRef]
- Hill, S.J.; Mordes, D.A.; Cameron, L.A.; Neuberg, D.S.; Landini, S.; Eggan, K.; Livingston, D.M. Two familial ALS proteins function in prevention/repair of transcription-associated DNA damage. Proc. Natl. Acad. Sci. USA 2016, 113, E7701–E7709. [Google Scholar] [CrossRef] [PubMed]
- Guerrero, E.N.; Mitra, J.; Wang, H.; Rangaswamy, S.; Hegde, P.M.; Basu, P.; Rao, K.S.; Hegde, M.L. Amyotrophic lateral sclerosis-associated TDP-43 mutation Q331K prevents nuclear translocation of XRCC4-DNA ligase 4 complex and is linked to genome damage-mediated neuronal apoptosis. Hum. Mol. Genet. 2019, 28, 2459–2476. [Google Scholar] [CrossRef] [PubMed]
- Kawaguchi, T.; Rollins, M.G.; Moinpour, M.; Morera, A.A.; Ebmeier, C.C.; Old, W.M.; Schwartz, J.C. Changes to the TDP-43 and FUS Interactomes Induced by DNA Damage. J. Proteome Res. 2019, 19, 360–370. [Google Scholar] [CrossRef] [PubMed]
- Konopka, A.; Whelan, D.R.; Jamali, S.; Perri, E.; Shahheydari, H.; Toth, R.P.; Parakh, S.; Robinson, T.; Cheong, A.; Mehta, P.; et al. Impaired NHEJ repair in amyotrophic lateral sclerosis is associated with TDP-43 mutations. Mol. Neurodegener. 2020, 15, 51. [Google Scholar] [CrossRef]
- Eldridge, M.J.G.; Hamon, M.A. Histone H3 deacetylation promotes host cell viability for efficient infection by Listeria monocytogenes. PLoS Pathog. 2021, 17, e1010173. [Google Scholar] [CrossRef]
- Giannini, M.; Bayona-Feliu, A.; Sproviero, D.; Barroso, S.I.; Cereda, C.; Aguilera, A. TDP-43 mutations link Amyotrophic Lateral Sclerosis with R-loop homeostasis and R loop-mediated DNA damage. PLoS Genet. 2020, 16, e1009260. [Google Scholar] [CrossRef]
- Sharma, S. Age-related nonhomologous end joining activity in rat neurons. Brain Res. Bull. 2007, 73, 48–54. [Google Scholar] [CrossRef]
- Yu, A.M.; McVey, M. Synthesis-dependent microhomology-mediated end joining accounts for multiple types of repair junctions. Nucleic Acids Res. 2010, 38, 5706–5717. [Google Scholar] [CrossRef]
- Eskandarian, H.A.; Impens, F.; Nahori, M.-A.; Soubigou, G.; Coppée, J.-Y.; Cossart, P.; Hamon, M.A. A Role for SIRT2-Dependent Histone H3K18 Deacetylation in Bacterial Infection. Science 2013, 341, 1238858. [Google Scholar] [CrossRef]
- Chen, X.; Lu, W.; Wu, D. Sirtuin 2 (SIRT2): Confusing Roles in the Pathophysiology of Neurological Disorders. Front. Neurosci. 2021, 15, 614107. [Google Scholar] [CrossRef]
- Yu, Z.; Fan, D.; Gui, B.; Shi, L.; Xuan, C.; Shan, L.; Wang, Q.; Shang, Y.; Wang, Y. Neurodegeneration-associated TDP-43 Interacts with Fragile X Mental Retardation Protein (FMRP)/Staufen (STAU1) and Regulates SIRT1 Expression in Neuronal Cells. J. Biol. Chem. 2012, 287, 22560–22572. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Wu, H.; Yang, M.; Ye, S.; Li, L.; Zhang, H.; Hu, J.; Wang, X.; Xu, J.; Liang, A. SIRT1 inhibition impairs non-homologous end joining DNA damage repair by increasing Ku70 acetylation in chronic myeloid leukemia cells. Oncotarget 2015, 7, 13538–13550. [Google Scholar] [CrossRef]
- Herskovits, A.Z.; Guarente, L. SIRT1 in Neurodevelopment and Brain Senescence. Neuron 2014, 81, 471–483. [Google Scholar] [CrossRef] [PubMed]
- Singh, M.; Zhang, S.; Perez, A.M.; Lee, E.Y.C.; Lee, M.Y.W.T.; Zhang, D. POLDIP3: At the Crossroad of RNA and DNA Metabolism. Genes 2022, 13, 1921. [Google Scholar] [CrossRef]
- Shiga, A.; Ishihara, T.; Miyashita, A.; Kuwabara, M.; Kato, T.; Watanabe, N.; Yamahira, A.; Kondo, C.; Yokoseki, A.; Takahashi, M.; et al. Alteration of POLDIP3 Splicing Associated with Loss of Function of TDP-43 in Tissues Affected with ALS. PLoS ONE 2012, 7, e43120. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.-Y.; Pan, L.; Su, S.C.; Quinn, E.J.; Sasaki, M.; Jimenez, J.C.; Mackenzie, I.R.A.; Huang, E.J.; Tsai, L.-H. Interaction of FUS and HDAC1 regulates DNA damage response and repair in neurons. Nat. Neurosci. 2013, 16, 1383–1391. [Google Scholar] [CrossRef]
- Wang, H.; Guo, W.; Mitra, J.; Hegde, P.M.; Vandoorne, T.; Eckelmann, B.J.; Mitra, S.; Tomkinson, A.E.; Van Den Bosch, L.; Hegde, M.L. Mutant FUS causes DNA ligation defects to inhibit oxidative damage repair in Amyotrophic Lateral Sclerosis. Nat. Commun. 2018, 9, 3683. [Google Scholar] [CrossRef]
- Stirling, P.C.; Hieter, P. Canonical DNA Repair Pathways Influence R-Loop-Driven Genome Instability. J. Mol. Biol. 2017, 429, 3132–3138. [Google Scholar] [CrossRef]
- Cuartas, J.; Gangwani, L. R-loop Mediated DNA Damage and Impaired DNA Repair in Spinal Muscular Atrophy. Front. Cell. Neurosci. 2022, 16, 826608. [Google Scholar] [CrossRef]
- Wood, M.; Quinet, A.; Lin, Y.-L.; Davis, A.A.; Pasero, P.; Ayala, Y.M.; Vindigni, A. TDP-43 dysfunction results in R-loop accumulation and DNA replication defects. J. Cell Sci. 2020, 133, jcs244129. [Google Scholar] [CrossRef]
- Gong, D.; Wang, L.; Zhou, H.; Gao, J.; Zhang, W.; Zheng, P. Long noncoding RNA Lnc530 localizes on R-loops and regulates R-loop formation and genomic stability in mouse embryonic stem cells. Stem Cell Rep. 2023, 18, 952–968. [Google Scholar] [CrossRef] [PubMed]
- Lander, E.S.; Linton, L.M.; Birren, B.; Nusbaum, C.; Zody, M.C.; Baldwin, J.; Devon, K.; Dewar, K.; Doyle, M.; FitzHugh, W.; et al. Initial sequencing and analysis of the human genome. Nature 2001, 409, 860–921. [Google Scholar] [CrossRef] [PubMed]
- Bannert, N.; Kurth, R. The Evolutionary Dynamics of Human Endogenous Retroviral Families. Annu. Rev. Genom. Hum. Genet. 2006, 7, 149–173. [Google Scholar] [CrossRef] [PubMed]
- Villesen, P.; Aagaard, L.; Wiuf, C.; Pedersen, F.S. Identification of endogenous retroviral reading frames in the human genome. Retrovirology 2004, 1, 32. [Google Scholar] [CrossRef] [PubMed]
- Ueda, M.T.; Kryukov, K.; Mitsuhashi, S.; Mitsuhashi, H.; Imanishi, T.; Nakagawa, S. Comprehensive genomic analysis reveals dynamic evolution of endogenous retroviruses that code for retroviral-like protein domains. Mob. DNA 2020, 11, 29. [Google Scholar] [CrossRef]
- Moyes, D.; Griffiths, D.J.; Venables, P.J. Insertional polymorphisms: A new lease of life for endogenous retroviruses in human disease. Trends Genet. 2007, 23, 326–333. [Google Scholar] [CrossRef]
- Wildschutte, J.H.; Williams, Z.H.; Montesion, M.; Subramanian, R.P.; Kidd, J.M.; Coffin, J.M. Discovery of unfixed endogenous retrovirus insertions in diverse human populations. Proc. Natl. Acad. Sci. USA 2016, 113, E2326–E2334. [Google Scholar] [CrossRef]
- Li, W.; Lin, L.; Malhotra, R.; Yang, L.; Acharya, R.; Poss, M. A computational framework to assess genome-wide distribution of polymorphic human endogenous retrovirus-K In human populations. PLoS Comput. Biol. 2019, 15, e1006564. [Google Scholar] [CrossRef]
- Dupressoir, A.; Lavialle, C.; Heidmann, T. From ancestral infectious retroviruses to bona fide cellular genes: Role of the captured syncytins in placentation. Placenta 2012, 33, 663–671. [Google Scholar] [CrossRef]
- Kaneko-Ishino, T.; Ishino, F. The role of genes domesticated from LTR retrotransposons and retroviruses in mammals. Front. Microbiol. 2012, 3, 262. [Google Scholar] [CrossRef]
- Naville, M.; Warren, I.; Haftek-Terreau, Z.; Chalopin, D.; Brunet, F.; Levin, P.; Galiana, D.; Volff, J.-N. Not so bad after all: Retroviruses and long terminal repeat retrotransposons as a source of new genes in vertebrates. Clin. Microbiol. Infect. 2016, 22, 312–323. [Google Scholar] [CrossRef] [PubMed]
- Grandi, N.; Tramontano, E. Human Endogenous Retroviruses Are Ancient Acquired Elements Still Shaping Innate Immune Responses. Front. Immunol. 2018, 9, 2039. [Google Scholar] [CrossRef] [PubMed]
- Mager, D.L.; Stoye, J.P. Mammalian Endogenous Retroviruses. Microbiol. Spectr. 2015, 3, 1079–1100. [Google Scholar] [CrossRef] [PubMed]
- Brouha, B.; Schustak, J.; Badge, R.M.; Lutz-Prigge, S.; Farley, A.H.; Moran, J.V.; Kazazian, H.H. Hot L1s account for the bulk of retrotransposition in the human population. Proc. Natl. Acad. Sci. USA 2003, 100, 5280–5285. [Google Scholar] [CrossRef] [PubMed]
- Dewannieux, M.; Heidmann, T. LINEs, SINEs and processed pseudogenes: Parasitic strategies for genome modeling. Cytogenet. Genome Res. 2005, 110, 35–48. [Google Scholar] [CrossRef]
- Pavlíček, A.; Pačes, J.; Elleder, D.; Hejnar, J. Processed Pseudogenes of Human Endogenous Retroviruses Generated by LINEs: Their Integration, Stability, and Distribution. Genome Res. 2002, 12, 391–399. [Google Scholar] [CrossRef]
- Mouse Genome Sequencing Consortium; Waterston, R.H.; Lindblad-Toh, K.; Birney, E.; Rogers, J.; Abril, J.F.; Agarwal, P.; Agarwala, R.; Ainscough, R.; Alexandersson, M.; et al. Initial sequencing and comparative analysis of the mouse genome. Nature 2002, 420, 520–562. [Google Scholar] [CrossRef]
- Coufal, N.G.; Garcia-Perez, J.L.; Peng, G.E.; Yeo, G.W.; Mu, Y.; Lovci, M.T.; Morell, M.; O’Shea, K.S.; Moran, J.V.; Gage, F.H. L1 retrotransposition in human neural progenitor cells. Nature 2009, 460, 1127–1131. [Google Scholar] [CrossRef]
- Della Valle, F.; Thimma, M.P.; Caiazzo, M.; Pulcrano, S.; Celii, M.; Adroub, S.A.; Liu, P.; Alanis-Lobato, G.; Broccoli, V.; Orlando, V. Transdifferentiation of Mouse Embryonic Fibroblasts into Dopaminergic Neurons Reactivates LINE-1 Repetitive Elements. Stem Cell Rep. 2020, 14, 60–74. [Google Scholar] [CrossRef]
- Richardson, S.R.; Morell, S.; Faulkner, G.J. L1 Retrotransposons and Somatic Mosaicism in the Brain. Annu. Rev. Genet. 2014, 48, 1–27. [Google Scholar] [CrossRef]
- Jern, P.; Sperber, G.O.; Blomberg, J. Use of Endogenous Retroviral Sequences (ERVs) and structural markers for retroviral phylogenetic inference and taxonomy. Retrovirology 2005, 2, 50. [Google Scholar] [CrossRef]
- Groh, S.; Schotta, G. Silencing of endogenous retroviruses by heterochromatin. Cell. Mol. Life Sci. 2017, 74, 2055–2065. [Google Scholar] [CrossRef] [PubMed]
- Geis, F.K.; Goff, S.P. Silencing and Transcriptional Regulation of Endogenous Retroviruses: An Overview. Viruses 2020, 12, 884. [Google Scholar] [CrossRef]
- Goodier, J.L. Restricting retrotransposons: A review. Mob. DNA 2016, 7, 16. [Google Scholar] [CrossRef] [PubMed]
- Savage, A.L.; Lopez, A.I.; Iacoangeli, A.; Bubb, V.J.; Smith, B.; Troakes, C.; Alahmady, N.; Koks, S.; Schumann, G.G.; Al-Chalabi, A.; et al. Frequency and methylation status of selected retrotransposition competent L1 loci in amyotrophic lateral sclerosis. Mol. Brain 2020, 13, 154. [Google Scholar] [CrossRef]
- Li, W.; Jin, Y.; Prazak, L.; Hammell, M.; Dubnau, J. Transposable Elements in TDP-43-Mediated Neurodegenerative Disorders. PLoS ONE 2012, 7, e44099. [Google Scholar] [CrossRef]
- Tam, O.H.; Rozhkov, N.V.; Shaw, R.; Kim, D.; Hubbard, I.; Fennessey, S.; Propp, N.; NYGC ALS Consortium; Fagegaltier, D.; Harris, B.T.; et al. Postmortem Cortex Samples Identify Distinct Molecular Subtypes of ALS: Retrotransposon Activation, Oxidative Stress, and Activated Glia. Cell Rep. 2019, 29, 1164–1177.e5. [Google Scholar] [CrossRef]
- Kelley, D.R.; Hendrickson, D.G.; Tenen, D.; Rinn, J.L. Transposable elements modulate human RNA abundance and splicing via specific RNA-protein interactions. Genome Biol. 2014, 15, 537. [Google Scholar] [CrossRef]
- Douville, R.; Liu, J.; Rothstein, J.; Nath, A. Identification of active loci of a human endogenous retrovirus in neurons of patients with amyotrophic lateral sclerosis. Ann. Neurol. 2011, 69, 141–151. [Google Scholar] [CrossRef]
- Li, W.; Lee, M.-H.; Henderson, L.; Tyagi, R.; Bachani, M.; Steiner, J.; Campanac, E.; Hoffman, D.A.; von Geldern, G.; Johnson, K.; et al. Human endogenous retrovirus-K contributes to motor neuron disease. Sci. Transl. Med. 2015, 7, 307ra153. [Google Scholar] [CrossRef]
- Ibba, G.; Piu, C.; Uleri, E.; Serra, C.; Dolei, A. Disruption by SaCas9 Endonuclease of HERV-Kenv, a Retroviral Gene with Oncogenic and Neuropathogenic Potential, Inhibits Molecules Involved in Cancer and Amyotrophic Lateral Sclerosis. Viruses 2018, 10, 412. [Google Scholar] [CrossRef]
- Fuentes, D.R.; Swigut, T.; Wysocka, J. Systematic perturbation of retroviral LTRs reveals widespread long-range effects on human gene regulation. Elife 2018, 7, e35989. [Google Scholar] [CrossRef] [PubMed]
- Steele, A.J.; Al-Chalabi, A.; Ferrante, K.; Cudkowicz, M.E.; Brown, R.H.; Garson, J.A. Detection of serum reverse transcriptase activity in patients with ALS and unaffected blood relatives. Neurology 2005, 64, 454–458. [Google Scholar] [CrossRef] [PubMed]
- McCormick, A.L.; Brown, R.H.; Cudkowicz, M.E.; Al-Chalabi, A.; Garson, J.A. Quantification of reverse transcriptase in ALS and elimination of a novel retroviral candidate. Neurology 2008, 70, 278–283. [Google Scholar] [CrossRef]
- Li, T.D.; Murano, K.; Kitano, T.; Guo, Y.; Negishi, L.; Siomi, H. TDP-43 safeguards the embryo genome from L1 retrotransposition. Sci. Adv. 2022, 8, eabq3806. [Google Scholar] [CrossRef] [PubMed]
- Della Valle, F.; Reddy, P.; Yamamoto, M.; Liu, P.; Saera-Vila, A.; Bensaddek, D.; Zhang, H.; Martinez, J.P.; Abassi, L.; Celii, M.; et al. LINE-1 RNA causes heterochromatin erosion and is a target for amelioration of senescent phenotypes in progeroid syndromes. Sci. Transl. Med. 2022, 14, eabl6057. [Google Scholar] [CrossRef]
- Krug, L.; Chatterjee, N.; Borges-Monroy, R.; Hearn, S.; Liao, W.-W.; Morrill, K.; Prazak, L.; Rozhkov, N.; Theodorou, D.; Hammell, M.; et al. Retrotransposon activation contributes to neurodegeneration in a Drosophila TDP-43 model of ALS. PLoS Genet. 2017, 13, e1006635. [Google Scholar] [CrossRef] [PubMed]
- Chang, Y.-H.; Dubnau, J. The Gypsy Endogenous Retrovirus Drives Non-Cell-Autonomous Propagation in a Drosophila TDP-43 Model of Neurodegeneration. Curr. Biol. 2019, 29, 3135–3152.e4. [Google Scholar] [CrossRef]
- Romano, G.; Klima, R.; Feiguin, F. TDP-43 prevents retrotransposon activation in the Drosophila motor system through regulation of Dicer-2 activity. BMC Biol. 2020, 18, 82. [Google Scholar] [CrossRef]
- Kim, V.N.; Han, J.; Siomi, M.C. Biogenesis of small RNAs in animals. Nat. Rev. Mol. Cell Biol. 2009, 10, 126–139. [Google Scholar] [CrossRef]
- Pham, J.; Keon, M.; Brennan, S.; Saksena, N. Connecting RNA-Modifying Similarities of TDP-43, FUS, and SOD1 with MicroRNA Dysregulation Amidst A Renewed Network Perspective of Amyotrophic Lateral Sclerosis Proteinopathy. Int. J. Mol. Sci. 2020, 21, 3464. [Google Scholar] [CrossRef] [PubMed]
- Aravin, A.A.; Sachidanandam, R.; Bourc’His, D.; Schaefer, C.; Pezic, D.; Toth, K.F.; Bestor, T.; Hannon, G.J. A piRNA Pathway Primed by Individual Transposons Is Linked to De Novo DNA Methylation in Mice. Mol. Cell 2008, 31, 785–799. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Dahlstrom, J.E.; Lee, S.-H.; Rangasamy, D. Naturally occurring endo-siRNA silences LINE-1 retrotransposons in human cells through DNA methylation. Epigenetics 2012, 7, 758–771. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Hernandez, M.J.; Swanson, M.D.; Contreras-Galindo, R.; Cookinham, S.; King, S.R.; Noel, R.J.; Kaplan, M.H.; Markovitz, D.M. Expression of Human Endogenous Retrovirus Type K (HML-2) Is Activated by the Tat Protein of HIV-1. J. Virol. 2012, 86, 7790–7805. [Google Scholar] [CrossRef] [PubMed]
- Wang, I.-F.; Reddy, N.M.; Shen, C.-K.J. Higher order arrangement of the eukaryotic nuclear bodies. Proc. Natl. Acad. Sci. USA 2002, 99, 13583–13588. [Google Scholar] [CrossRef]
- Wang, H.-Y.; Wang, I.-F.; Bose, J.; Shen, C.-K. Structural diversity and functional implications of the eukaryotic TDP gene family. Genomics 2004, 83, 130–139. [Google Scholar] [CrossRef]
- D’Alton, S.; Altshuler, M.; Lewis, J. Studies of alternative isoforms provide insight into TDP-43 autoregulation and pathogenesis. RNA 2015, 21, 1419–1432. [Google Scholar] [CrossRef]
- Weskamp, K.; Tank, E.M.; Miguez, R.; McBride, J.P.; Gómez, N.B.; White, M.; Lin, Z.; Gonzalez, C.M.; Serio, A.; Sreedharan, J.; et al. Shortened TDP43 isoforms upregulated by neuronal hyperactivity drive TDP43 pathology in ALS. J. Clin. Investig. 2020, 130, 1139–1155. [Google Scholar] [CrossRef]
- Shenouda, M.; Xiao, S.; MacNair, L.; Lau, A.; Robertson, J. A C-Terminally Truncated TDP-43 Splice Isoform Exhibits Neuronal Specific Cytoplasmic Aggregation and Contributes to TDP-43 Pathology in ALS. Front. Neurosci. 2022, 16, 868556. [Google Scholar] [CrossRef]
- Goossens, J.; Vanmechelen, E.; Trojanowski, J.Q.; Lee, V.M.; Van Broeckhoven, C.; van der Zee, J.; Engelborghs, S. TDP-43 as a possible biomarker for frontotemporal lobar degeneration: A systematic review of existing antibodies. Acta Neuropathol. Commun. 2015, 3, 15. [Google Scholar] [CrossRef]
- Yamashita, T.; Akamatsu, M.; Kwak, S. Altered Intracellular Milieu of ADAR2-Deficient Motor Neurons in Amyotrophic Lateral Sclerosis. Genes 2017, 8, 60. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.; Lin, F.; Robertson, C.S.; Wang, K.K. Dual Vulnerability of TDP-43 to Calpain and Caspase-3 Proteolysis after Neurotoxic Conditions and Traumatic Brain Injury. J. Cereb. Blood Flow Metab. 2014, 34, 1444–1452. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Wang, Z.; Liu, L.; Yang, Z.; Liu, S.; Ma, Z.; Liu, Y.; Ma, Y.; Zhang, L.; Zhang, X.; et al. LncRNA Malat1 inhibition of TDP43 cleavage suppresses IRF3-initiated antiviral innate immunity. Proc. Natl. Acad. Sci. USA 2020, 117, 23695–23706. [Google Scholar] [CrossRef]
- Buratti, E. Functional Significance of TDP-43 Mutations in Disease. Adv. Genet. 2015, 91, 1–53. [Google Scholar] [CrossRef] [PubMed]
- Mitra, J.; Hegde, M.L. The Role of TDP-43 in Genome Repair and beyond in Amyotrophic Lateral Sclerosis. In Amyotrophic Lateral Sclerosis—Recent Advances and Therapeutic Challenges; Hegde, M.L., Ed.; IntechOpen: London, UK, 2020; ISBN 978-1-83880-580-7. [Google Scholar]
- Fratta, P.; Sivakumar, P.; Humphrey, J.; Lo, K.; Ricketts, T.; Oliveira, H.; Brito-Armas, J.M.; Kalmar, B.; Ule, A.; Yu, Y.; et al. Mice with endogenous TDP -43 mutations exhibit gain of splicing function and characteristics of amyotrophic lateral sclerosis. EMBO J. 2018, 37, e98684. [Google Scholar] [CrossRef]
- White, M.A.; Kim, E.; Duffy, A.; Adalbert, R.; Phillips, B.U.; Peters, O.M.; Stephenson, J.; Yang, S.; Massenzio, F.; Lin, Z.; et al. TDP-43 gains function due to perturbed autoregulation in a Tardbp knock-in mouse model of ALS-FTD. Nat. Neurosci. 2018, 21, 552–563. [Google Scholar] [CrossRef]
- Arnold, E.S.; Ling, S.-C.; Huelga, S.C.; Lagier-Tourenne, C.; Polymenidou, M.; Ditsworth, D.; Kordasiewicz, H.B.; McAlonis-Downes, M.; Platoshyn, O.; Parone, P.A.; et al. ALS-linked TDP-43 mutations produce aberrant RNA splicing and adult-onset motor neuron disease without aggregation or loss of nuclear TDP-43. Proc. Natl. Acad. Sci. USA 2013, 110, E736–E745. [Google Scholar] [CrossRef]
- Ozyurt, T.; Gautam, M. Differential Epigenetic Signature of Corticospinal Motor Neurons in ALS. Brain Sci. 2021, 11, 754. [Google Scholar] [CrossRef]
- Hasegawa-Ogawa, M.; Okano, H.J. Characterization of the upstream and intron promoters of the gene encoding TAR DNA-binding protein. Sci. Rep. 2021, 11, 8720. [Google Scholar] [CrossRef]
- Luquin, N.; Yu, B.; Saunderson, R.B.; Trent, R.J.; Pamphlett, R. Genetic variants in the promoter of TARDBP in sporadic amyotrophic lateral sclerosis. Neuromuscul. Disord. 2009, 19, 696–700. [Google Scholar] [CrossRef]
- Baralle, M.; Romano, M. Characterization of the human TARDBP gene promoter. Sci. Rep. 2021, 11, 10438. [Google Scholar] [CrossRef]
- Ayala, Y.M.; De Conti, L.; Avendaño-Vázquez, S.E.; Dhir, A.; Romano, M.; D’Ambrogio, A.; Tollervey, J.; Ule, J.; Baralle, M.; Buratti, E.; et al. TDP-43 regulates its mRNA levels through a negative feedback loop. EMBO J. 2011, 30, 277–288. [Google Scholar] [CrossRef]
- Avendaño-Vázquez, S.E.; Dhir, A.; Bembich, S.; Buratti, E.; Proudfoot, N.; Baralle, F.E. Autoregulation of TDP-43 mRNA levels involves interplay between transcription, splicing, and alternative polyA site selection. Genes Dev. 2012, 26, 1679–1684. [Google Scholar] [CrossRef]
- Budini, M.; Buratti, E. TDP-43 Autoregulation: Implications for Disease. J. Mol. Neurosci. 2011, 45, 473–479. [Google Scholar] [CrossRef]
- Koyama, A.; Sugai, A.; Kato, T.; Ishihara, T.; Shiga, A.; Toyoshima, Y.; Koyama, M.; Konno, T.; Hirokawa, S.; Yokoseki, A.; et al. Increased cytoplasmic TARDBP mRNA in affected spinal motor neurons in ALS caused by abnormal autoregulation of TDP-43. Nucleic Acids Res. 2016, 44, 5820–5836. [Google Scholar] [CrossRef] [PubMed]
- Sugai, A.; Kato, T.; Koyama, A.; Koike, Y.; Konno, T.; Ishihara, T.; Onodera, O. Non-genetically modified models exhibit TARDBP mRNA increase due to perturbed TDP-43 autoregulation. Neurobiol. Dis. 2019, 130, 104534. [Google Scholar] [CrossRef] [PubMed]
- Sugai, A.; Kato, T.; Koyama, A.; Koike, Y.; Kasahara, S.; Konno, T.; Ishihara, T.; Onodera, O. Robustness and Vulnerability of the Autoregulatory System That Maintains Nuclear TDP-43 Levels: A Trade-off Hypothesis for ALS Pathology Based on in Silico Data. Front. Neurosci. 2018, 12, 28. [Google Scholar] [CrossRef] [PubMed]
- Koike, Y.; Sugai, A.; Hara, N.; Ito, J.; Yokoseki, A.; Ishihara, T.; Yamagishi, T.; Tsuboguchi, S.; Tada, M.; Ikeuchi, T.; et al. Age-related demethylation of the TDP-43 autoregulatory region in the human motor cortex. Commun. Biol. 2021, 4, 1107. [Google Scholar] [CrossRef] [PubMed]
- Johnston, W.L.; Krizus, A.; Ramani, A.K.; Dunham, W.; Youn, J.Y.; Fraser, A.G.; Gingras, A.-C.; Dennis, J.W.C. elegans SUP-46, an HNRNPM family RNA-binding protein that prevents paternally-mediated epigenetic sterility. BMC Biol. 2017, 15, 61. [Google Scholar] [CrossRef]
- Li, S.; Armstrong, C.M.; Bertin, N.; Ge, H.; Milstein, S.; Boxem, M.; Vidalain, P.-O.; Han, J.-D.J.; Chesneau, A.; Hao, T.; et al. A Map of the Interactome Network of the Metazoan C. elegans. Science 2004, 303, 540–543. [Google Scholar] [CrossRef]
- Cragnaz, L.; Klima, R.; De Conti, L.; Romano, G.; Feiguin, F.; Buratti, E.; Baralle, M.; Baralle, F. An age-related reduction of brain TBPH/TDP-43 levels precedes the onset of locomotion defects in a Drosophila ALS model. Neuroscience 2015, 311, 415–421. [Google Scholar] [CrossRef]
- Huang, C.; Xia, P.Y.; Zhou, H. Sustained Expression of TDP-43 and FUS in Motor Neurons in Rodent’s Lifetime. Int. J. Biol. Sci. 2010, 6, 396–406. [Google Scholar] [CrossRef] [PubMed]
- Sephton, C.F.; Good, S.K.; Atkin, S.; Dewey, C.M.; Mayer, P.; Herz, J.; Yu, G. TDP-43 Is a Developmentally Regulated Protein Essential for Early Embryonic Development. J. Biol. Chem. 2010, 285, 6826–6834. [Google Scholar] [CrossRef] [PubMed]
- Pacetti, M.; De Conti, L.; Marasco, L.E.; Romano, M.; Rashid, M.M.; Nubiè, M.; Baralle, F.E.; Baralle, M. Physiological tissue-specific and age-related reduction of mouse TDP-43 levels is regulated by epigenetic modifications. Dis. Model. Mech. 2022, 15, dmm049032. [Google Scholar] [CrossRef] [PubMed]
- Maunakea, A.K.; Chepelev, I.; Cui, K.; Zhao, K. Intragenic DNA methylation modulates alternative splicing by recruiting MeCP2 to promote exon recognition. Cell Res. 2013, 23, 1256–1269. [Google Scholar] [CrossRef]
- The ENCODE Project Consortium. An Integrated Encyclopedia of DNA Elements in the Human Genome. Nature 2012, 489, 57–74. [Google Scholar] [CrossRef]
- Moujalled, D.; James, J.L.; Yang, S.; Zhang, K.; Duncan, C.; Moujalled, D.M.; Parker, S.J.; Caragounis, A.; Lidgerwood, G.; Turner, B.J.; et al. Phosphorylation of hnRNP K by cyclin-dependent kinase 2 controls cytosolic accumulation of TDP-43. Hum. Mol. Genet. 2014, 24, 1655–1669. [Google Scholar] [CrossRef]
- Moujalled, D.; Grubman, A.; Acevedo, K.; Yang, S.; Ke, Y.D.; Moujalled, D.M.; Duncan, C.; Caragounis, A.; Perera, N.D.; Turner, B.J.; et al. TDP-43 mutations causing amyotrophic lateral sclerosis are associated with altered expression of RNA-binding protein hnRNP K and affect the Nrf2 antioxidant pathway. Hum. Mol. Genet. 2017, 26, 1732–1746. [Google Scholar] [CrossRef]
- Mejzini, R.; Flynn, L.L.; Pitout, I.L.; Fletcher, S.; Wilton, S.D.; Akkari, P.A. ALS Genetics, Mechanisms, and Therapeutics: Where Are We Now? Front. Neurosci. 2019, 13, 1310. [Google Scholar] [CrossRef]
- Ito, D. Promise of Nucleic Acid Therapeutics for Amyotrophic Lateral Sclerosis. Ann. Neurol. 2021, 91, 13–20. [Google Scholar] [CrossRef]
- Pozzi, S.; Thammisetty, S.S.; Codron, P.; Rahimian, R.; Plourde, K.V.; Soucy, G.; Bareil, C.; Phaneuf, D.; Kriz, J.; Gravel, C.; et al. Virus-mediated delivery of antibody targeting TAR DNA-binding protein-43 mitigates associated neuropathology. J. Clin. Investig. 2019, 129, 1581–1595. [Google Scholar] [CrossRef] [PubMed]
- Pozzi, S.; Codron, P.; Soucy, G.; Renaud, L.; Cordeau, P.J.; Dutta, K.; Bareil, C.; Julien, J.-P. Monoclonal full-length antibody against TAR DNA binding protein 43 reduces related proteinopathy in neurons. JCI Insight 2020, 5, e140420. [Google Scholar] [CrossRef] [PubMed]
- Poulin-Brière, A.; Rezaei, E.; Pozzi, S. Antibody-Based Therapeutic Interventions for Amyotrophic Lateral Sclerosis: A Systematic Literature Review. Front. Neurosci. 2021, 15, 790114. [Google Scholar] [CrossRef] [PubMed]
- Bowen, L.N.; Tyagi, R.; Li, W.; Alfahad, T.; Smith, B.; Wright, M.; Singer, E.J.; Nath, A. HIV-associated motor neuron disease: HERV-K Activation and Response to Antiretroviral Therapy. Neurology 2016, 87, 1756–1762. [Google Scholar] [CrossRef]
- Lamers, S.L.; Rose, R.; Maidji, E.; Agsalda-Garcia, M.; Nolan, D.J.; Fogel, G.B.; Salemi, M.; Garcia, D.L.; Bracci, P.; Yong, W.; et al. HIV DNA Is Frequently Present within Pathologic Tissues Evaluated at Autopsy from Combined Antiretroviral Therapy-Treated Patients with Undetectable Viral Loads. J. Virol. 2016, 90, 8968–8983. [Google Scholar] [CrossRef]
- Gold, J.; Rowe, D.B.; Kiernan, M.C.; Vucic, S.; Mathers, S.; van Eijk, R.P.A.; Nath, A.; Montojo, M.G.; Norato, G.; Santamaria, U.A.; et al. Safety and tolerability of Triumeq in amyotrophic lateral sclerosis: The Lighthouse trial. Amyotroph. Lateral Scler. Front. Degener. 2019, 20, 595–604. [Google Scholar] [CrossRef]
- Jones, R.B.; Garrison, K.E.; Wong, J.C.; Duan, E.H.; Nixon, D.F.; Ostrowski, M.A. Nucleoside Analogue Reverse Transcriptase Inhibitors Differentially Inhibit Human LINE-1 Retrotransposition. PLoS ONE 2008, 3, e1547. [Google Scholar] [CrossRef]
- Curty, G.; Marston, J.L.; de Mulder Rougvie, M.; Leal, F.E.; Nixon, D.F.; Soares, M.A. Human Endogenous Retrovirus K in Cancer: A Potential Biomarker and Immunotherapeutic Target. Viruses 2020, 12, 726. [Google Scholar] [CrossRef]
- Kang, B.H.; Momin, N.; Moynihan, K.D.; Silva, M.; Li, Y.; Irvine, D.J.; Wittrup, K.D. Immunotherapy-induced antibodies to endogenous retroviral envelope glycoprotein confer tumor protection in mice. PLoS ONE 2021, 16, e0248903. [Google Scholar] [CrossRef]
- Diebold, M.; Derfuss, T. The monoclonal antibody GNbAC1: Targeting human endogenous retroviruses in multiple sclerosis. Ther. Adv. Neurol. Disord. 2019, 12, 1756286419833574. [Google Scholar] [CrossRef]
- Curtin, F.; Bernard, C.; Levet, S.; Perron, H.; Porchet, H.; Médina, J.; Malpass, S.; Lloyd, D.; Simpson, R.; RAINBOW-T1D investigators. A new therapeutic approach for type 1 diabetes: Rationale for GNbAC1, an anti-HERV-W-Env monoclonal antibody. Diabetes Obes. Metab. 2018, 20, 2075–2084. [Google Scholar] [CrossRef] [PubMed]
- Siklos, M.; Kubicek, S. Therapeutic targeting of chromatin: Status and opportunities. FEBS J. 2021, 289, 1276–1301. [Google Scholar] [CrossRef] [PubMed]
- Vojta, A.; Dobrinić, P.; Tadić, V.; Bočkor, L.; Korać, P.; Julg, B.; Klasić, M.; Zoldoš, V. Repurposing the CRISPR-Cas9 system for targeted DNA methylation. Nucleic Acids Res. 2016, 44, 5615–5628. [Google Scholar] [CrossRef]
- Cali, C.P.; Park, D.; Lee, E.B. Targeted DNA methylation of neurodegenerative disease genes via homology directed repair. Nucleic Acids Res. 2019, 47, 11609–11622. [Google Scholar] [CrossRef] [PubMed]
- Nishiyama, J.; Mikuni, T.; Yasuda, R. Virus-Mediated Genome Editing via Homology-Directed Repair in Mitotic and Postmitotic Cells in Mammalian Brain. Neuron 2017, 96, 755–768.e5. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Duan, Y.; Duan, G.; Wang, Q.; Zhang, K.; Deng, X.; Qian, B.; Gu, J.; Ma, Z.; Zhang, S.; et al. Stress Induces Dynamic, Cytotoxicity-Antagonizing TDP-43 Nuclear Bodies via Paraspeckle LncRNA NEAT1-Mediated Liquid-Liquid Phase Separation. Mol. Cell 2020, 79, 443–458.e7. [Google Scholar] [CrossRef]
- Matsukawa, K.; Kukharsky, M.S.; Park, S.-K.; Park, S.; Watanabe, N.; Iwatsubo, T.; Hashimoto, T.; Liebman, S.W.; Shelkovnikova, T.A. Long non-coding RNA NEAT1_1 ameliorates TDP-43 toxicity in in vivo models of TDP-43 proteinopathy. RNA Biol. 2021, 18, 1546–1554. [Google Scholar] [CrossRef]
- Sekar, D.; Tusubira, D.; Ross, K. TDP-43 and NEAT long non-coding RNA: Roles in neurodegenerative disease. Front. Cell. Neurosci. 2022, 16, 954912. [Google Scholar] [CrossRef]
- Li, Y.; Shen, Y.; Xie, M.; Wang, B.; Wang, T.; Zeng, J.; Hua, H.; Yu, J.; Yang, M. LncRNAs LCETRL3 and LCETRL4 at chromosome 4q12 diminish EGFR-TKIs efficiency in NSCLC through stabilizing TDP43 and EIF2S1. Signal Transduct. Target. Ther. 2022, 7, 30. [Google Scholar] [CrossRef]
- Roman, G.C. Neuroepidemiology of amyotrophic lateral sclerosis: Clues to aetiology and pathogenesis. J. Neurol. Neurosurg. Psychiatry 1996, 61, 131–137. [Google Scholar] [CrossRef]
- Mitsumoto, H.; Przedborski, S.; Gordon, P.H. (Eds.) Amyotrophic Lateral Sclerosis; CRC Press: Boca Raton, FL, USA, 2005; ISBN 978-1-4200-2744-0. [Google Scholar]
- Feldman, E.L.; Goutman, S.A.; Petri, S.; Mazzini, L.; Savelieff, M.G.; Shaw, P.J.; Sobue, G. Amyotrophic lateral sclerosis. Lancet Lond. Engl. 2022, 400, 1363–1380. [Google Scholar] [CrossRef] [PubMed]
- Curtis, A.F.; Masellis, M.; Hsiung, G.-Y.R.; Moineddin, R.; Zhang, K.; Au, B.; Millett, G.; Mackenzie, I.; Rogaeva, E.; Tierney, M.C. Sex differences in the prevalence of genetic mutations in FTD and ALS: A Meta-Analysis. Neurology 2017, 89, 1633–1642. [Google Scholar] [CrossRef] [PubMed]
- Watkins, J.; Ghosh, A.; Keerie, A.F.A.; Alix, J.J.P.; Mead, R.J.; Sreedharan, J. Female sex mitigates motor and behavioural phenotypes in TDP-43Q331K knock-in mice. Sci. Rep. 2020, 10, 19220. [Google Scholar] [CrossRef] [PubMed]
- Kong, S.; Li, R.; Tian, Y.; Zhang, Y.; Lu, Y.; Ou, Q.; Gao, P.; Li, K.; Zhang, Y. Single-cell omics: A new direction for functional genetic research in human diseases and animal models. Front. Genet. 2023, 13, 1100016. [Google Scholar] [CrossRef] [PubMed]






| Gene Symbol | Name | Mammalian Orthologs *, Alias | Gene Ontology Function | Contribution to Chromatin Structure/Transcription | Toxicity Modulation upon Knockdown | Refs. |
|---|---|---|---|---|---|---|
| Chd1 † | Chromodomain-helicase-DNA binding protein 1 | CHD1, CHD2 | Chromatin remodeling | SILENCING | suppresses/ameliorates | [52] |
| e(y)3 † | Enhancer of yellow 3 | PHF10 | Chromatin remodeling | OPENING | suppresses/ameliorates | [52] |
| polybromo † | Polybromo | BAF180 | Chromatin remodeling | OPENING | suppresses/ameliorates | [52] |
| ash1 † | Absent, small, or homeotic discs 1 | ASH1L | Chromatin remodeling | OPENING | suppresses/ameliorates | [52] |
| enok † | Enoki mushroom | KAT6A | Chromatin remodeling | OPENING | suppresses/ameliorates | [52] |
| br † | Broad | - | Chromatin remodeling | OPENING | suppresses/ameliorates | [52] |
| Br140 † | Bromodomain-containing protein, 140kD | BRPF1 | Chromatin remodeling | OPENING | suppresses/ameliorates | [52] |
| mor † | Moira | BAF170S/MARCC2 | Chromatin remodeling | OPENING | suppresses/ameliorates | [52] |
| MED8 † | Mediator complex subunit 8 | MED8 | Mediator complex | TF–RNA Pol interaction | suppresses/ameliorates | [52] |
| MED11 † | Mediator complex subunit 11 | MED11 | Mediator complex | TF–RNA Pol interaction | suppresses/ameliorates | [52] |
| MED15 † | Mediator complex subunit 15 | MED15 | Mediator complex | TF–RNA Pol interaction | suppresses/ameliorates | [52] |
| MED21 † | Mediator complex subunit 21 | MED21 | Mediator complex | TF–RNA Pol interaction | suppresses/ameliorates | [52] |
| MED22 † | Mediator complex subunit 22 | MED22 | Mediator complex | TF–RNA Pol interaction | suppresses/ameliorates | [52] |
| MED27 † | Mediator complex subunit 27 | MED27 | Mediator complex | TF–RNA Pol interaction | suppresses/ameliorates | [52] |
| MED28 † | Mediator complex subunit 28 | MED28 | Mediator complex | TF–RNA Pol interaction | suppresses/ameliorates | [52] |
| MED31 † | Mediator complex subunit 31 | MED31 | Mediator complex | TF–RNA Pol interaction | suppresses/ameliorates | [52] |
| e(y)1 † | Enhancer of yellow 1 | TAF9 | Transcription Factor | Transcription | suppresses/ameliorates | [52] |
| TAF1 † | TBP-associated factor 1 | TAF1 | Transcription Factor | Transcription | suppresses/ameliorates | [52] |
| Tomb † | tombola | LIN54/TESMIN ** | Male meiosis | Transcription | suppresses/ameliorates | [52] |
| Su(Tpl) † | Suppressor of Triplolethal | ELL, ELL2 | Elongator | Transcription | suppresses/ameliorates | [52] |
| SF2 † | Splicing factor 2 | SRSF1 | Splicing | Transcription | suppresses/ameliorates | [52] |
| Hsp70B (Ba/Bb/Bbb/Bc) † | Heat-shock-protein-70B (Ba/Bb/Bbb/Bc) | HSPA1A | Heat shock Factor | Transcription | suppresses/ameliorates | [52] |
| Chd1 † | Chromodomain-helicase-DNA-binding protein 1 | CHD1 | Chromatin remodeling | TDP-1/TDP-43 impedes Chd1 binding | strongly enhances | [51] |
| dom † | Domino | SRCAP ** | Chromatin remodeling | strongly enhances | [51] | |
| mi-2 † | Mi-2 | CHD3; CHD4; CHD5 ** | Chromatin remodeling | enhances | [51] | |
| kis † | Kismet | CHD6, CHD7, CHD8, CHD9 ** | Chromatin remodeling | enhances | [51] | |
| asf1 † | Anti-silencing factor 1 | ASF1A; ASF1B | Chromatin remodeling | suppresses | [51] | |
| Snr1 † | Snf5-related 1 | SMARCB1 | Chromatin remodeling | suppresses | [51] | |
| mor † | Moira | SMARRC1; SMARCC2 ** | Chromatin remodeling | suppresses | [51] | |
| Dalao † | Brahma associated protein 111kD, Bap111 | SMARCE1 ** | Chromatin remodeling | suppresses | [51] | |
| Chd3 † | Chd3 | CHD3 | Chromatin remodeling | suppresses | [51] | |
| Bap60 † | Brahma associated protein 60kD | SMARCD1 | Chromatin remodeling | strongly suppresses | [51] | |
| MBD-R2 † | MBD-R2 | PHF20; PHF20L1 | Histone Lysine acetyltransferase | OPENING | enhances | [51] |
| Tip60 † | Tat interactive protein 60kDa | KAT5; KAT7; KAT8 ** | Histone Lysine acetyltransferase | OPENING | enhances | [51] |
| CG2051 † | Histone acetyltransferase 1 | HAT1 | Histone Lysine acetyltransferase | OPENING | suppresses | [51] |
| HDAC3 † | Histone deacetylase 3 | HDAC3 | Histone Lysine deacetylase | SILENCING | enhances | [51] |
| Mta1-like † | Metastasis associated 1-like | MTA2; MTA3 | Histone Lysine deacetylase | SILENCING | enhances | [51] |
| Sirt4 † | Sirtuin 4 | SIRT4 | Histone Lysine deacetylase | SILENCING | suppresses | [51] |
| HDAC6 † | Histone deacetylase 6 | HDAC6 | Histone Lysine deacetylase | SILENCING | suppresses * | [51] |
| Scm † | Sex comb on midleg | L3MBTL3; PHC2; SCML1; THAP10 | Transcription Factor | Transcription | suppresses | [51] |
| Spt6 † | Spt6 | SUPT6H | Transcription Factor | Transcription | suppresses | [51] |
| lid † | Little imaginal discs | KDM 4A; 4B; 4D; 4E; 4F; 5A; 5B; 5C; 5D; | Histone Lysine demethylase (H3K4) | SILENCING | strongly suppresses | [51] |
| Utx † | Utx histone demethylase | KDM6A; 6B; UTY | Histone Lysine demethylase (H3K27) | OPENING | suppresses | [51] |
| Asx † | Additional sex combs | ASXL1-3 | Chromatin regulator | SILENCING | suppresses | [51] |
| Ncoa6 † | Nuclear receptor coactivator 6 | NCOA6 | co-activator | Transcription | suppresses | [51] |
| Su(var)3-9 † | Suppressor of variegation 3-9 | SUV39H1 | Histone Lysine methyltransferase (H3K9) | SILENCING | suppresses | [51] |
| ash2 † | Absent, small, or homeotic discs 2 | ASH2L | Histone Lysine methyltransferase (H3K4) | OPENING | enhances | [51] |
| Su(z)2 † | Suppressor of zeste 2 | BMI1; PCGF1; 2; 5; 6; RING1B; RNF2 | Histone Lysine methyltransferase, histone monoubiquitination (H2A-K119) | OPENING | enhances | [51] |
| Set1 † | SET domain containing 1 | KMT2B | Histone Lysine methyltransferase (H3K4) | OPENING | enhances | [51] |
| Wdr82 † | WD repeat domain 82 | WDR82 | Histone Lysine methyltransferase (H3K4) | OPENING | enhances | [51] |
| HPL-2 †2 | Heterochromatin Protein-like 2 | HPL1 | Chromatin remodeling | TDP-1 bridges HPL-2 to chromatin | n.d. | [53] |
| nBAF complex | Neuronal Brg1/BRM Associated factor | SS18L1, NBAF; CREST; SYT homolog 1 | Neuronal chromatin remodeling complex nBAF component | n.d. | [57] | |
| sgg † | Shaggy | GSK3 | Control of cellular pathways and metabolism | suppresses | [50] | |
| htk † | Hat-trick | Chromatin remodeling | suppresses | [50] | ||
| xmas-2 † | Xmas-2 | Transcription and RNA export | suppresses | [50] | ||
| H3C14 | H3S10Ph-K14Ac: Histone 3 (serine 10 phosphorylation/lysine 14 acetylation) | H3 | Histone tail PTM | Transcriptional activation/repression | n.d. | [58] |
| H3C14 | H3K9me3: Histone 3 (methylation of lysine 9) | H3 | Histone tail PTM | Transcriptional repression | n.d. | [58] |
| HDAC1 | Histone deacetylase 1 | HDAC1 | Histone/Protein Lysine deacetylase | SILENCING *** | n.d. | [48] |
| H2b | Histone cluster 1, H2bp | HIST1H2BP | Nucleosome assembly | Downregulated in the TDPΔNLS | n.d. | [63] |
| H3d | Histone cluster 1, H3d | HIST1H3D | Nucleosome assembly | Downregulated in the TDPΔNLS | n.d. | [63] |
| H4a/H4b/H4c/H4h | Histone cluster 1, H4a, b, c, h | HIST1H4 | Nucleosome assembly | Downregulated in the TDPΔNLS | n.d. | [63] |
| Nap1l1 | Nucleosome assembly protein 1-like1 | NAP1L1 | Nucleosome assembly | Upregulated in the TDPΔNLS | n.d. | [63] |
| Med20 | Mediator Complex subunit 20 | MED20; SRB2; TRFP; PRO0213 | Mediator Complex/transcriptional coactivator | Transcriptional activation | n.d. | [64] |
| Usp49 | Ubiquitin specific processing protease 49 | USP49 | H2B Histone deubiquitinase | Transcriptional activation/regulation of mRNA splicing | n.d. | [64] |
| HUWE1 | HECT, UBA, WWE domain Containing E3 Ubiquitin Protein ligase 1 | HUWE1 | Histone/protein Ubiquitination | n.d. | [65] | |
| YY1 | Yin and Yang 1 transcription Factor | YY-1; INO80S | Transcription factor | Transcriptional activation/repression | n.d. | [65] |
| MORF4L2 | Mortality Factor 4-like 2 | MRGX; KIAA0026; MORF-Related Gene X | Heterochromatin assembly/histone modification | Transcriptional activation | n.d. | [65] |
| HMGN1 | High-Mobility Group Nucleosome Binding Domain 1 | HMG14; MGC104230; FLJ27265 | Chromatin remodeling | n.d. | [65] | |
| PRKDC | Protein Kinase, DNA-Activated, Catalytic subunit | DNA-PKcs; DNPK1; P460; DNAPKc; XRCC7; P350 | DNA repair and recombination | n.d. | [65] | |
| UIMC1 | Ubiquitin Interaction Motif Containing 1 | RAP80; Retinoid X Receptor-Interacting Protein 110 | DNA repair | Ubiquitination/Transcription repression | n.d. | [65] |
| POLB | DNA Pol Beta | DNA Pol Beta | DNA excision and repair | n.d. | [65] | |
| SFPQ | Splicing Factor Proline and Glutamine Rich | PSF; PPP1R140; HPOMp100; POMP100 | Splicing | Enables DNA binding activity | n.d. | [65] |
| MSH3 | MutS Homolog 3 | MRP1; DUP; HMSH3; Mismatch Repair Protein | DNA mismatch repair system (MMR) | Postreplicative DNA mismatch repair | n.d. | [65] |
| XRCC5 | X-Ray Repair Cross Complementing 5 | Ku86; KARP-1; KU80; KUB2 | DNA repair | DNA DSB repair by NHEJ | n.d. | [65] |
| XRCC6 | X-RAY Repair Cross Complementing 6 | KU70; Cells6; G22P1; ML8 | DNA repair | DNA DSB repair by NHEJ | n.d. | [65] |
| STAG2 | Stromal Antigen 2 | SA2; SCC3B; SA-2; Cohesin Subunit SA-2 | Chromatin remodeling | Cohesion of sister chromatids after DNA replication | n.d. | [77,78] |
| Ell, Su(Tpl) † | Elongation Factor for RNA Pol II | ELL; ELL2; C19orf17; PPP1R68 | Elongator | Transcription regulation | enhances | [52,79] |
| TDP-43 Mutation | Epigenomic Alterations | Cellular Context | Refs. | |
|---|---|---|---|---|
| G294V | DNA damage/DSB TC-DSB | Mislocalization; | SH-S5Y5 | [97] |
| R-loop accumulation; R-loop-dependent increased DSBs | ||||
| Accumulation of FANCD2 repair foci (replication blockage). | ||||
| G298S | DNA methylation | Hyper- and hypo-methylated regions related to controls with common and specific DMRs related to other ALS hIPSCs-derived MNs (C9orf72-, TARDBP-, SOD1-, and FUS-mutation carriers). | hIPSCs-derived MNs from G298S carrier | [74] |
| A315T | Chromatin remodeling | Diminution of nBAF chromatin-remodeling complex components (Brg1, BAF53b, and CREST). | Cultured mouse MNs | [57] |
| DNA methylation | Altered 5mC and 5hmC underlying their increased vulnerability to degeneration. | TDP-43A315T mouse CSMNs | [171] | |
| DNA damage/DSB | Enhanced vulnerability to DNA damages. | MN-like NSC-34 cells | [95] | |
| M323K | Autoregulation | Increased RNA, but not significant for protein. | Mouse TDP-43M323K knock-in | [167] |
| Q331K | Autoregulation | Increase in RNA and nuclear protein in frontal cortex and spinal cords of mutant mice, but not in motor neuron. | Mouse TDP-43Q331K knock-in but not transgene | [168,169] |
| DNA damage/DSB | Increased cytosolic sequestration of the poly-ubiquitinated and aggregated form, nuclear loss of function DNA damage induction, and DSB repair defects (preventing the nuclear translocation of XRCC4); Contribution to oxidative genome damage accumulation via increased reactive oxygen species (ROS). | Spinal cord of ALS Q331K carriers, SH-S5Y5 | [93] | |
| Enhanced vulnerability to DNA damages (increase yH2Ax post damaging agent exposure). | MN-like NSC-34 cells | [95] | ||
| M337V | DNA methylation | No global alteration in 5mC | SH-S5Y5 | [58] |
| Histone PTMs | Significant decrease in global H3S10Ph-K14Ac; No significant increase in H3K9me3. | |||
| DNA damage/DSB | Impairment in the NHEJ DSB repair factors recruitment. | Fibroblasts of human M337V carrier | [95] | |
| G348C | Autoregulation | Upstream intron1 promoter region of TARDBP binding but less efficient than WT for its repression; No significant activation of the intron 1 promoter. | HEK293T | [170] |
| A382T | DNA Methylation | No global alteration in 5mC. | SH-S5Y5 | [58] |
| Histone PTMs | No significant decrease in global H3S10Ph-K14Ac; No significant increase in H3K9me3. | |||
| DNA damage/DSB TC-DSB | Increased yH2Ax; R-loop accumulation; R-loop-dependent increased DSBs accumulation of FANCD2 repair foci (replication blockage). | SH-S5Y5 LCL from A382T carriers | [97] | |
| Isoforms/fragments | Increased TDP-35 form at chromatin at R-loop. | LCL from A382T carriers | [97] | |
| Autoregulation | Upstream intron1 promoter region of TARDBP binding but less efficient than WT for its repression; Able to induce a significant activation of the intron 1 promoter. | HEK293T | [170] | |
| N390D | DNA methylation | Hyper- and hypo-methylated regions related to controls with common and specific DMRs related to other ALS hIPSCs-derived MNs (C9orf72-, TARDBP-, SOD1-, and FUS-mutation carriers). | hIPSCs-derived MNs from N390D carrier | [74] |
| G418C | Chromatin remodeling | Diminution in nBAF chromatin-remodeling complex components (Brg1, BAF53b and CREST). | Cultured mouse MNs | [57] |
| C9orf72 with loss of nuclear TDP-43 | DNA methylation | Altered DNA 5mC and 5hmC in the CSMN without nuclear TDP-43 compared to residual C9orf72 CMSN without TDP-43 nuclear loss. | Residual lower MNs from post-mortem CSMN tissues of C9orf72 carriers | [73] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gimenez, J.; Spalloni, A.; Cappelli, S.; Ciaiola, F.; Orlando, V.; Buratti, E.; Longone, P. TDP-43 Epigenetic Facets and Their Neurodegenerative Implications. Int. J. Mol. Sci. 2023, 24, 13807. https://doi.org/10.3390/ijms241813807
Gimenez J, Spalloni A, Cappelli S, Ciaiola F, Orlando V, Buratti E, Longone P. TDP-43 Epigenetic Facets and Their Neurodegenerative Implications. International Journal of Molecular Sciences. 2023; 24(18):13807. https://doi.org/10.3390/ijms241813807
Chicago/Turabian StyleGimenez, Juliette, Alida Spalloni, Sara Cappelli, Francesca Ciaiola, Valerio Orlando, Emanuele Buratti, and Patrizia Longone. 2023. "TDP-43 Epigenetic Facets and Their Neurodegenerative Implications" International Journal of Molecular Sciences 24, no. 18: 13807. https://doi.org/10.3390/ijms241813807