Polycystic Ovary Syndrome and Oxidative Stress—From Bench to Bedside
Abstract
:1. Introduction
2. ROS Production
3. Mitochondrial DNA Damage
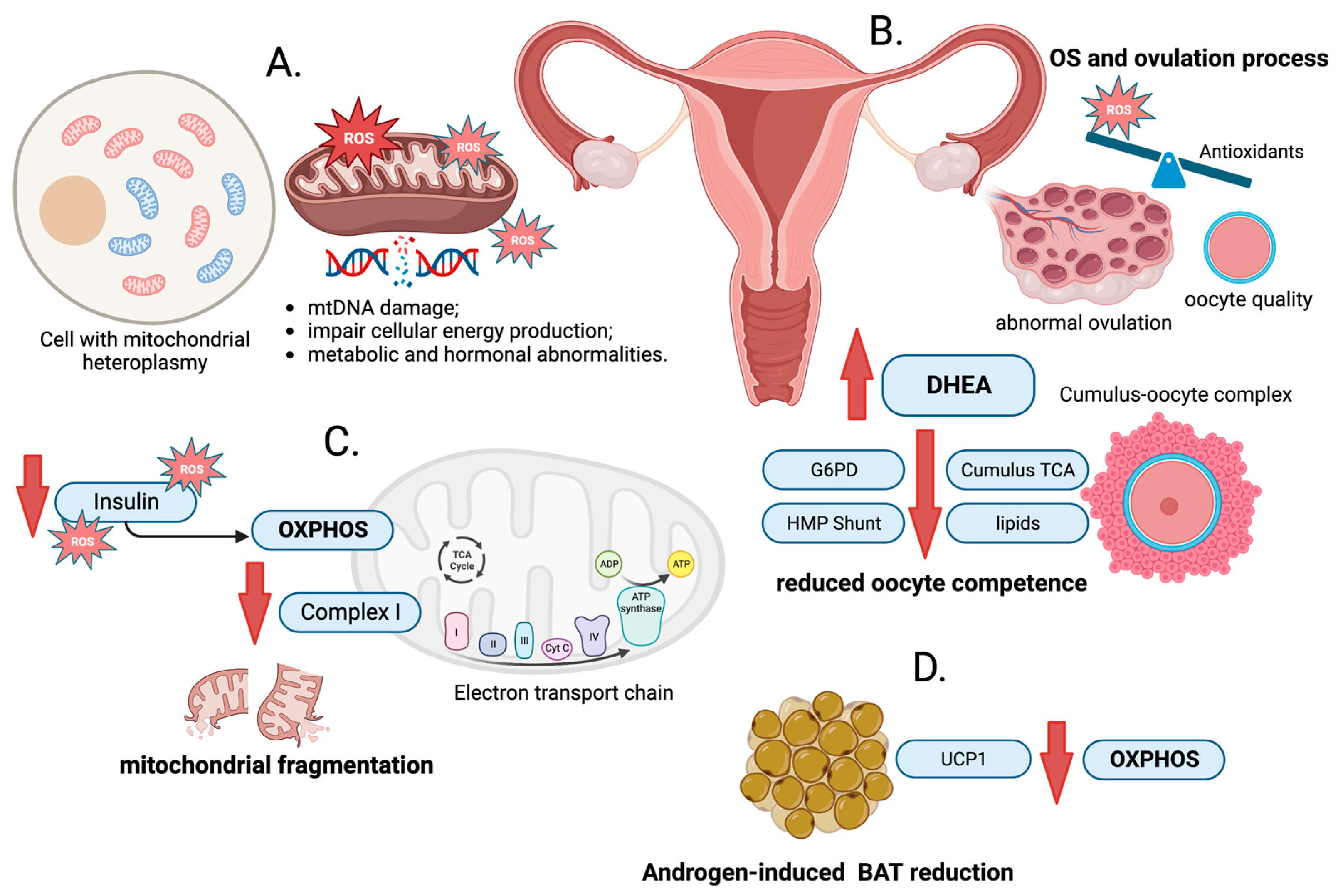
4. Oxidative Stress and PCOS
5. Oxidative Phosphorylation and PCOS
6. Antioxidants as Potential Therapy Methods
7. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Pizzino, G.; Irrera, N.; Cucinotta, M.; Pallio, G.; Mannino, F.; Arcoraci, V.; Squadrito, F.; Altavilla, D.; Bitto, A. Oxidative Stress: Harms and Benefits for Human Health. Oxidative Med. Cell. Longev. 2017, 2017, 8416763. [Google Scholar] [CrossRef] [PubMed]
- Christodoulakos, G.; Augoulea, A.; Lambrinoudaki, I.; Sioulas, V.; Creatsas, G. Pathogenesis of Endometriosis: The Role of Defective ‘Immunosurveillance’. Eur. J. Contracept. Reprod. Health Care 2007, 12, 194–202. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, A.; Aponte-Mellado, A.; Premkumar, B.J.; Shaman, A.; Gupta, S. The Effects of Oxidative Stress on Female Reproduction: A Review. Reprod. Biol. Endocrinol. 2012, 10, 49. [Google Scholar] [CrossRef] [PubMed]
- Polycystic Ovary Syndrome. Available online: https://www.who.int/news-room/fact-sheets/detail/polycystic-ovary-syndrome (accessed on 24 July 2023).
- Witchel, S.F.; Oberfield, S.E.; Peña, A.S. Polycystic Ovary Syndrome: Pathophysiology, Presentation, and Treatment with Emphasis on Adolescent Girls. J. Endocr. Soc. 2019, 3, 1545–1573. [Google Scholar] [CrossRef]
- Chang, S.; Dunaif, A. Diagnosis of Polycystic Ovary Syndrome: Which Criteria to Use When? Endocrinol. Metab. Clin. N. Am. 2021, 50, 11–23. [Google Scholar] [CrossRef]
- Singh, S.; Pal, N.; Shubham, S.; Sarma, D.K.; Verma, V.; Marotta, F.; Kumar, M. Polycystic Ovary Syndrome: Etiology, Current Management, and Future Therapeutics. J. Clin. Med. 2023, 12, 1454. [Google Scholar] [CrossRef]
- Sachdeva, G.; Gainder, S.; Suri, V.; Sachdeva, N.; Chopra, S. Comparison of the Different PCOS Phenotypes Based on Clinical Metabolic, and Hormonal Profile, and Their Response to Clomiphene. Indian. J. Endocrinol. Metab. 2019, 23, 326–331. [Google Scholar] [CrossRef]
- Elsayed, A.M.; Al-Kaabi, L.S.; Al-Abdulla, N.M.; Al-Kuwari, M.S.; Al-Mulla, A.A.; Al-Shamari, R.S.; Alhusban, A.K.; AlNajjar, A.A.; Doi, S.A.R. Clinical Phenotypes of PCOS: A Cross-Sectional Study. Reprod. Sci. 2023; Online ahead of print. [Google Scholar] [CrossRef]
- Akre, S.; Sharma, K.; Chakole, S.; Wanjari, M.B. Recent Advances in the Management of Polycystic Ovary Syndrome: A Review Article. Cureus 2022, 14, e27689. [Google Scholar] [CrossRef]
- Lubrano, V.; Pingitore, A.; Traghella, I.; Storti, S.; Parri, S.; Berti, S.; Ndreu, R.; Andrenelli, A.; Palmieri, C.; Iervasi, G.; et al. Emerging Biomarkers of Oxidative Stress in Acute and Stable Coronary Artery Disease: Levels and Determinants. Antioxidants 2019, 8, 115. [Google Scholar] [CrossRef]
- Yang, B.; Chen, Y.; Shi, J. Reactive Oxygen Species (ROS)-Based Nanomedicine. Chem. Rev. 2019, 119, 4881–4985. [Google Scholar] [CrossRef]
- Ighodaro, O.M.; Akinloye, O.A. First Line Defence Antioxidants-Superoxide Dismutase (SOD), Catalase (CAT) and Glutathione Peroxidase (GPX): Their Fundamental Role in the Entire Antioxidant Defence Grid. Alex. J. Med. 2018, 54, 287–293. [Google Scholar] [CrossRef]
- Gusti, A.M.T.; Qusti, S.Y.; Alshammari, E.M.; Toraih, E.A.; Fawzy, M.S. Antioxidants-Related Superoxide Dismutase (SOD), Catalase (CAT), Glutathione Peroxidase (GPX), Glutathione-S-Transferase (GST), and Nitric Oxide Synthase (NOS) Gene Variants Analysis in an Obese Population: A Preliminary Case-Control Study. Antioxidants 2021, 10, 595. [Google Scholar] [CrossRef] [PubMed]
- Liang, J.; Gao, Y.; Feng, Z.; Zhang, B.; Na, Z.; Li, D. Reactive Oxygen Species and Ovarian Diseases: Antioxidant Strategies. Redox Biol. 2023, 62, 102659. [Google Scholar] [CrossRef] [PubMed]
- Murphy, M.P. How Mitochondria Produce Reactive Oxygen Species. Biochem. J. 2009, 417, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Malin, S.K.; Kirwan, J.P.; Sia, C.L.; González, F. Glucose-Stimulated Oxidative Stress in Mononuclear Cells Is Related to Pancreatic β-Cell Dysfunction in Polycystic Ovary Syndrome. J. Clin. Endocrinol. Metab. 2014, 99, 322–329. [Google Scholar] [CrossRef]
- Lu, J.; Wang, Z.; Cao, J.; Chen, Y.; Dong, Y. A Novel and Compact Review on the Role of Oxidative Stress in Female Reproduction. Reprod. Biol. Endocrinol. 2018, 16, 80. [Google Scholar] [CrossRef]
- Evans, J.L.; Maddux, B.A.; Goldfine, I.D. The Molecular Basis for Oxidative Stress-Induced Insulin Resistance. Antioxid. Redox Signal. 2005, 7, 1040–1052. [Google Scholar] [CrossRef]
- Shan, H.; Luo, R.; Guo, X.; Li, R.; Ye, Z.; Peng, T.; Liu, F.; Yang, Z. Abnormal Endometrial Receptivity and Oxidative Stress in Polycystic Ovary Syndrome. Front. Pharmacol. 2022, 13, 904942. [Google Scholar] [CrossRef]
- DeVallance, E.; Li, Y.; Jurczak, M.J.; Cifuentes-Pagano, E.; Pagano, P.J. The Role of NADPH Oxidases in the Etiology of Obesity and Metabolic Syndrome: Contribution of Individual Isoforms and Cell Biology. Antioxid. Redox Signal. 2019, 31, 687–709. [Google Scholar] [CrossRef]
- Juan, C.A.; Pérez de la Lastra, J.M.; Plou, F.J.; Pérez-Lebeña, E. The Chemistry of Reactive Oxygen Species (ROS) Revisited: Outlining Their Role in Biological Macromolecules (DNA, Lipids and Proteins) and Induced Pathologies. Int. J. Mol. Sci. 2021, 22, 4642. [Google Scholar] [CrossRef]
- Schieber, M.; Chandel, N.S. ROS Function in Redox Signaling and Oxidative Stress. Curr. Biol. 2014, 24, R453–R462. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Zou, Y.; Wu, G.; Zheng, L. Oxidative Stress and Mitochondrial Dysfunction of Granulosa Cells in Polycystic Ovarian Syndrome. Front. Med. 2023, 10, 1193749. [Google Scholar] [CrossRef] [PubMed]
- Sulaiman, M.; Al-Farsi, Y.; Al-Khaduri, M.; Saleh, J.; Waly, M. Polycystic Ovarian Syndrome Is Linked to Increased Oxidative Stress in Omani Women. Int. J. Women’s Health 2018, 10, 763–771. [Google Scholar] [CrossRef] [PubMed]
- Ramya, S.; Poornima, P.; Jananisri, A.; Geofferina, I.P.; Bavyataa, V.; Divya, M.; Priyanga, P.; Vadivukarasi, J.; Sujitha, S.; Elamathi, S.; et al. Role of Hormones and the Potential Impact of Multiple Stresses on Infertility. Stresses 2023, 3, 454–474. [Google Scholar] [CrossRef]
- Ayala, A.; Muñoz, M.F.; Argüelles, S. Lipid Peroxidation: Production, Metabolism, and Signaling Mechanisms of Malondialdehyde and 4-Hydroxy-2-Nonenal. Oxidative Med. Cell. Longev. 2014, 2014, 360438. [Google Scholar] [CrossRef]
- Kolesnikova, L.I.; Kolesnikov, S.I.; Darenskaya, M.A.; Grebenkina, L.A.; Nikitina, O.A.; Lazareva, L.M.; Suturina, L.V.; Danusevich, I.N.; Druzhinina, E.B.; Semendyaev, A.A. Activity of LPO Processes in Women with Polycystic Ovarian Syndrome and Infertility. Bull. Exp. Biol. Med. 2017, 162, 320–322. [Google Scholar] [CrossRef]
- Enechukwu, C.I.; Onuegbu, A.J.; Olisekodiaka, M.J.; Eleje, G.U.; Ikechebelu, J.I.; Ugboaja, J.O.; Amah, U.K.; Okwara, J.E.; Igwegbe, A.O. Oxidative Stress Markers and Lipid Profiles of Patients with Polycystic Ovary Syndrome in a Nigerian Tertiary Hospital. Obs. Gynecol. Sci. 2019, 62, 335. [Google Scholar] [CrossRef]
- Kaltsas, A.; Zikopoulos, A.; Moustakli, E.; Zachariou, A.; Tsirka, G.; Tsiampali, C.; Palapela, N.; Sofikitis, N.; Dimitriadis, F. The Silent Threat to Women’s Fertility: Uncovering the Devastating Effects of Oxidative Stress. Antioxidants 2023, 12, 1490. [Google Scholar] [CrossRef]
- Asally, R.; Markham, R.; Manconi, F. Mitochondrial DNA Haplogroup H Association with Endometriosis and Possible Role in Inflammation and Pain. J. Endometr. Pelvic Pain Disord. 2020, 12, 158–164. [Google Scholar] [CrossRef]
- Habbane, M.; Montoya, J.; Rhouda, T.; Sbaoui, Y.; Radallah, D.; Emperador, S. Human Mitochondrial DNA: Particularities and Diseases. Biomedicines 2021, 9, 1364. [Google Scholar] [CrossRef]
- Schon, E.A.; DiMauro, S.; Hirano, M. Human Mitochondrial DNA: Roles of Inherited and Somatic Mutations. Nat. Rev. Genet. 2012, 13, 878–890. [Google Scholar] [CrossRef] [PubMed]
- Merheb, M.; Matar, R.; Hodeify, R.; Siddiqui, S.S.; Vazhappilly, C.G.; Marton, J.; Azharuddin, S.; Al Zouabi, H. Mitochondrial DNA, a Powerful Tool to Decipher Ancient Human Civilization from Domestication to Music, and to Uncover Historical Murder Cases. Cells 2019, 8, 433. [Google Scholar] [CrossRef] [PubMed]
- Kowalczyk, P.; Sulejczak, D.; Kleczkowska, P.; Bukowska-Ośko, I.; Kucia, M.; Popiel, M.; Wietrak, E.; Kramkowski, K.; Wrzosek, K.; Kaczyńska, K. Mitochondrial Oxidative Stress—A Causative Factor and Therapeutic Target in Many Diseases. Int. J. Mol. Sci. 2021, 22, 13384. [Google Scholar] [CrossRef]
- Hudson, G.; Amati-Bonneau, P.; Blakely, E.L.; Stewart, J.D.; He, L.; Schaefer, A.M.; Griffiths, P.G.; Ahlqvist, K.; Suomalainen, A.; Reynier, P.; et al. Mutation of OPA1 Causes Dominant Optic Atrophy with External Ophthalmoplegia, Ataxia, Deafness and Multiple Mitochondrial DNA Deletions: A Novel Disorder of MtDNA Maintenance. Brain 2008, 131, 329–337. [Google Scholar] [CrossRef]
- Zhuo, G.; Ding, Y.; Feng, G.; Yu, L.; Jiang, Y. Analysis of Mitochondrial DNA Sequence Variants in Patients with Polycystic Ovary Syndrome. Arch. Gynecol. Obs. 2012, 286, 653–659. [Google Scholar] [CrossRef] [PubMed]
- Ding, Y.; Zhuo, G.; Zhang, C.; Leng, J. Point Mutation in Mitochondrial TRNA Gene Is Associated with Polycystic Ovary Syndrome and Insulin Resistance. Mol. Med. Rep. 2016, 13, 3169–3172. [Google Scholar] [CrossRef]
- Ding, Y.; Xia, B.-H.; Zhang, C.-J.; Zhuo, G.-C. Mutations in Mitochondrial TRNA Genes May Be Related to Insulin Resistance in Women with Polycystic Ovary Syndrome. Am. J. Transl. Res. 2017, 9, 2984–2996. [Google Scholar]
- Ding, Y.; Zhuo, G.; Zhang, C. The Mitochondrial TRNALeu(UUR) A3302G Mutation May Be Associated with Insulin Resistance in Woman with Polycystic Ovary Syndrome. Reprod. Sci. 2016, 23, 228–233. [Google Scholar] [CrossRef]
- Reddy, T.V.; Govatati, S.; Deenadayal, M.; Sisinthy, S.; Bhanoori, M. Impact of Mitochondrial DNA Copy Number and Displacement Loop Alterations on Polycystic Ovary Syndrome Risk in South Indian Women. Mitochondrion 2019, 44, 35–40. [Google Scholar] [CrossRef]
- Lee, S.-H.; Chung, D.-J.; Lee, H.-S.; Kim, T.-J.; Kim, M.-H.; Jeong, H.J.; Im, J.-A.; Lee, D.-C.; Lee, J.-W. Mitochondrial DNA Copy Number in Peripheral Blood in Polycystic Ovary Syndrome. Metabolism 2011, 60, 1677–1682. [Google Scholar] [CrossRef]
- Shukla, P.; Mukherjee, S. Mitochondrial Dysfunction: An Emerging Link in the Pathophysiology of Polycystic Ovary Syndrome. Mitochondrion 2020, 52, 24–39. [Google Scholar] [CrossRef]
- Sabuncu, T.; Vural, H.; Harma, M.; Harma, M. Oxidative Stress in Polycystic Ovary Syndrome and Its Contribution to the Risk of Cardiovascular Disease. Clin. Biochem. 2001, 34, 407–413. [Google Scholar] [CrossRef]
- Yilmaz, M.; Bukan, N.; Ayvaz, G.; Karakoç, A.; Törüner, F.; Çakir, N.; Arslan, M. The Effects of Rosiglitazone and Metformin on Oxidative Stress and Homocysteine Levels in Lean Patients with Polycystic Ovary Syndrome. Hum. Reprod. 2005, 20, 3333–3340. [Google Scholar] [CrossRef]
- Siti, H.N.; Kamisah, Y.; Kamsiah, J. The Role of Oxidative Stress, Antioxidants and Vascular Inflammation in Cardiovascular Disease (a Review). Vasc. Pharmacol. 2015, 71, 40–56. [Google Scholar] [CrossRef]
- Mahalingaiah, S.; Diamanti-Kandarakis, E. Targets to Treat Metabolic Syndrome in Polycystic Ovary Syndrome. Expert Opin. Ther. Targets 2015, 19, 1561–1574. [Google Scholar] [CrossRef]
- Piotrowski, P.C.; Rzepczynska, I.J.; Kwintkiewicz, J.; Duleba, A.J. Oxidative Stress Induces Expression of CYP11A, CYP17, Star and 3 Beta HSD in Rat Theca-Interstitial Cells. J. Soc. Gynecol. Investig. 2005, 12, 319A. [Google Scholar]
- Ruggiero, M.; Viana, G.A.; Di Berardino, O.M.; Simi, G.; Papini, F.; Genazzani, A.R.; Cela, V.; Artini, P.G. Comparison between GnRH Agonist and Antagonist Protocols for Severe Endometriosis in Assisted Reproductive Cycles. J. Endometr. 2012, 4, 42–47. [Google Scholar] [CrossRef]
- Richards, J.S.; Russell, D.L.; Robker, R.L.; Dajee, M.; Alliston, T.N. Molecular Mechanisms of Ovulation and Luteinization. Mol. Cell. Endocrinol. 1998, 145, 47–54. [Google Scholar] [CrossRef]
- Holesh, J.E.; Bass, A.N.; Lord, M. Physiology, Ovulation. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2023. [Google Scholar]
- Kicińska, A.M.; Maksym, R.B.; Zabielska-Kaczorowska, M.A.; Stachowska, A.; Babińska, A. Immunological and Metabolic Causes of Infertility in Polycystic Ovary Syndrome. Biomedicines 2023, 11, 1567. [Google Scholar] [CrossRef]
- Yan, F.; Zhao, Q.; Li, Y.; Zheng, Z.; Kong, X.; Shu, C.; Liu, Y.; Shi, Y. The Role of Oxidative Stress in Ovarian Aging: A Review. J. Ovarian Res. 2022, 15, 100. [Google Scholar] [CrossRef]
- Takiguchi, S.; Sugino, N.; Kashida, S.; Yamagata, Y.; Nakamura, Y.; Kato, H. Rescue of the Corpus Luteum and an Increase in Luteal Superoxide Dismutase Expression Induced by Placental Luteotropins in the Rat: Action of Testosterone without Conversion to Estrogen1. Biol. Reprod. 2000, 62, 398–403. [Google Scholar] [CrossRef]
- Shkolnik, K.; Tadmor, A.; Ben-Dor, S.; Nevo, N.; Galiani, D.; Dekel, N. Reactive Oxygen Species Are Indispensable in Ovulation. Proc. Natl. Acad. Sci. USA 2011, 108, 1462–1467. [Google Scholar] [CrossRef]
- Uyanikoglu, H.; Sabuncu, T.; Dursun, H.; Sezen, H.; Aksoy, N. Circulating Levels of Apoptotic Markers and Oxidative Stress Parameters in Women with Polycystic Ovary Syndrome: A Case-Controlled Descriptive Study. Biomarkers 2017, 22, 643–647. [Google Scholar] [CrossRef]
- Yalcinkaya, E.; Cakiroglu, Y.; Doger, E.; Budak, O.; Cekmen, M.; Caliskan, E. Effect of Follicular Fluid NO, MDA and GSH Levels on in Vitro Fertilization Outcomes. J. Turk. Ger. Gynecol. Assoc. 2013, 14, 136–141. [Google Scholar] [CrossRef]
- Nuñez-Calonge, R.; Cortés, S.; Gutierrez Gonzalez, L.M.; Kireev, R.; Vara, E.; Ortega, L.; Caballero, P.; Rancan, L.; Tresguerres, J. Oxidative Stress in Follicular Fluid of Young Women with Low Response Compared with Fertile Oocyte Donors. Reprod. BioMedicine Online 2016, 32, 446–456. [Google Scholar] [CrossRef]
- Sharma, A.; Welt, C.K. Practical Approach to Hyperandrogenism in Women. Med. Clin. N. Am. 2021, 105, 1099–1116. [Google Scholar] [CrossRef]
- Rosenfield, R.L.; Cooke, D.W.; Radovick, S. Puberty and Its Disorders in the Female. In Pediatric Endocrinology; Elsevier: Amsterdam, The Netherlands, 2014; pp. 569–663.e1. ISBN 978-1-4557-4858-7. [Google Scholar]
- Yesiladali, M.; Yazici, M.G.K.; Attar, E.; Kelestimur, F. Differentiating Polycystic Ovary Syndrome from Adrenal Disorders. Diagnostics 2022, 12, 2045. [Google Scholar] [CrossRef]
- Abbott, D.H.; Greinwald, E.P.; Levine, J.E. Developmental Origins of Polycystic Ovary Syndrome: Everything Starts in Utero. In Polycystic Ovary Syndrome; Elsevier: Amsterdam, The Netherlands, 2022; pp. 23–38. ISBN 978-0-12-823045-9. [Google Scholar]
- Turcu, A.F.; Rege, J.; Auchus, R.J.; Rainey, W.E. 11-Oxygenated Androgens in Health and Disease. Nat. Rev. Endocrinol. 2020, 16, 284–296. [Google Scholar] [CrossRef]
- Torchen, L.C.; Sisk, R.; Legro, R.S.; Turcu, A.F.; Auchus, R.J.; Dunaif, A. 11-Oxygenated C19 Steroids Do Not Distinguish the Hyperandrogenic Phenotype of PCOS Daughters from Girls with Obesity. J. Clin. Endocrinol. Metab. 2020, 105, e3903–e3909. [Google Scholar] [CrossRef]
- Saito, K.; Matsuzaki, T.; Iwasa, T.; Miyado, M.; Saito, H.; Hasegawa, T.; Homma, K.; Inoue, E.; Miyashiro, Y.; Kubota, T.; et al. Steroidogenic Pathways Involved in Androgen Biosynthesis in Eumenorrheic Women and Patients with Polycystic Ovary Syndrome. J. Steroid Biochem. Mol. Biol. 2016, 158, 31–37. [Google Scholar] [CrossRef]
- Rosenfield, R.L.; Ehrmann, D.A. The Pathogenesis of Polycystic Ovary Syndrome (PCOS): The Hypothesis of PCOS as Functional Ovarian Hyperandrogenism Revisited. Endocr. Rev. 2016, 37, 467–520. [Google Scholar] [CrossRef]
- Shabbir, S.; Khurram, E.; Moorthi, V.S.; Eissa, Y.T.H.; Kamal, M.A.; Butler, A.E. The Interplay between Androgens and the Immune Response in Polycystic Ovary Syndrome. J. Transl. Med. 2023, 21, 259. [Google Scholar] [CrossRef] [PubMed]
- Silva, J.R.V.; Lima, F.E.O.; Souza, A.L.P.; Silva, A.W.B. Interleukin-1β and TNF-α Systems in Ovarian Follicles and Their Roles during Follicular Development, Oocyte Maturation and Ovulation. Zygote 2020, 28, 270–277. [Google Scholar] [CrossRef] [PubMed]
- Mak, P.J.; Duggal, R.; Denisov, I.G.; Gregory, M.C.; Sligar, S.G.; Kincaid, J.R. Human Cytochrome CYP17A1: The Structural Basis for Compromised Lyase Activity with 17-Hydroxyprogesterone. J. Am. Chem. Soc. 2018, 140, 7324–7331. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Li, S.; Liu, H.; Bai, H.; Hu, K.; Zhang, R.; Liu, Q.; Fan, P. Oxidative Stress Promotes Hyperandrogenism by Reducing Sex Hormone-Binding Globulin in Polycystic Ovary Syndrome. Fertil. Steril. 2021, 116, 1641–1650. [Google Scholar] [CrossRef]
- Li, W.; Liu, C.; Yang, Q.; Zhou, Y.; Liu, M.; Shan, H. Oxidative Stress and Antioxidant Imbalance in Ovulation Disorder in Patients with Polycystic Ovary Syndrome. Front. Nutr. 2022, 9, 1018674. [Google Scholar] [CrossRef]
- Turathum, B.; Gao, E.-M.; Chian, R.-C. The Function of Cumulus Cells in Oocyte Growth and Maturation and in Subsequent Ovulation and Fertilization. Cells 2021, 10, 2292. [Google Scholar] [CrossRef]
- Wang, Q.; Ratchford, A.M.; Chi, M.M.-Y.; Schoeller, E.; Frolova, A.; Schedl, T.; Moley, K.H. Maternal Diabetes Causes Mitochondrial Dysfunction and Meiotic Defects in Murine Oocytes. Mol. Endocrinol. 2009, 23, 1603–1612. [Google Scholar] [CrossRef]
- Jimenez, P.T.; Frolova, A.I.; Chi, M.M.; Grindler, N.M.; Willcockson, A.R.; Reynolds, K.A.; Zhao, Q.; Moley, K.H. DHEA-Mediated Inhibition of the Pentose Phosphate Pathway Alters Oocyte Lipid Metabolism in Mice. Endocrinology 2013, 154, 4835–4844. [Google Scholar] [CrossRef]
- Sarkar, M.; Terrault, N.; Chan, W.; Cedars, M.I.; Huddleston, H.G.; Duwaerts, C.C.; Balitzer, D.; Gill, R.M. Polycystic Ovary Syndrome (PCOS) Is Associated with NASH Severity and Advanced Fibrosis. Liver Int. 2020, 40, 355–359. [Google Scholar] [CrossRef]
- Vassilatou, E. Nonalcoholic Fatty Liver Disease and Polycystic Ovary Syndrome. World J. Gastroenterol. 2014, 20, 8351. [Google Scholar] [CrossRef] [PubMed]
- Gao, S.; Long, F.; Jiang, Z.; Shi, J.; Ma, D.; Yang, Y.; Bai, J.; Han, T.-L. The Complex Metabolic Interactions of Liver Tissue and Hepatic Exosome in PCOS Mice at Young and Middle Age. Front. Physiol. 2022, 13, 990987. [Google Scholar] [CrossRef] [PubMed]
- Bergman, O.; Ben-Shachar, D. Mitochondrial Oxidative Phosphorylation System (OXPHOS) Deficits in Schizophrenia: Possible Interactions with Cellular Processes. Can. J. Psychiatry 2016, 61, 457–469. [Google Scholar] [CrossRef] [PubMed]
- Fernandez-Vizarra, E.; Zeviani, M. Mitochondrial Disorders of the OXPHOS System. FEBS Lett. 2021, 595, 1062–1106. [Google Scholar] [CrossRef]
- Ng, Y.S.; Lim, A.Z.; Panagiotou, G.; Turnbull, D.M.; Walker, M. Endocrine Manifestations and New Developments in Mitochondrial Disease. Endocr. Rev. 2022, 43, 583–609. [Google Scholar] [CrossRef]
- Musumeci, O.; Oteri, R.; Toscano, A. Spectrum of Movement Disorders in Mitochondrial Diseases. J. Transl. Genet. Genom. 2020, 4, 221–237. [Google Scholar] [CrossRef]
- Chen, B.S.; Harvey, J.P.; Gilhooley, M.J.; Jurkute, N.; Yu-Wai-Man, P. Mitochondria and the Eye—Manifestations of Mitochondrial Diseases and Their Management. Eye 2023, 37, 2416–2425. [Google Scholar] [CrossRef]
- Koopman, W.J.H.; Verkaart, S.; Visch, H.J.; Van Emst-de Vries, S.; Nijtmans, L.G.J.; Smeitink, J.A.M.; Willems, P.H.G.M. Human NADH:Ubiquinone Oxidoreductase Deficiency: Radical Changes in Mitochondrial Morphology? Am. J. Physiol. Cell Physiol. 2007, 293, C22–C29. [Google Scholar] [CrossRef]
- Wilcox, G. Insulin and Insulin Resistance. Clin. Biochem. Rev. 2005, 26, 19–39. [Google Scholar]
- Lewandowski, K.C.; Skowrońska-Jóźwiak, E.; Łukasiak, K.; Gałuszko, K.; Dukowicz, A.; Cedro, M.; Lewiński, A. How Much Insulin Resistance in Polycystic Ovary Syndrome? Comparison of HOMA-IR and Insulin Resistance (Belfiore) Index Models. Arch. Med. Sci. 2019, 15, 613–618. [Google Scholar] [CrossRef]
- Hammes, S.R.; Levin, E.R. Impact of Estrogens in Males and Androgens in Females. J. Clin. Investig. 2019, 129, 1818–1826. [Google Scholar] [CrossRef] [PubMed]
- Ashraf, S.; Nabi, M.; Rasool, S.U.A.; Rashid, F.; Amin, S. Hyperandrogenism in Polycystic Ovarian Syndrome and Role of CYP Gene Variants: A Review. Egypt. J. Med. Hum. Genet. 2019, 20, 25. [Google Scholar] [CrossRef]
- Chen, M.-J.; Yang, W.-S.; Yang, J.-H.; Chen, C.-L.; Ho, H.-N.; Yang, Y.-S. Relationship Between Androgen Levels and Blood Pressure in Young Women with Polycystic Ovary Syndrome. Hypertension 2007, 49, 1442–1447. [Google Scholar] [CrossRef] [PubMed]
- Dadachanji, R.; Shaikh, N.; Mukherjee, S. Genetic Variants Associated with Hyperandrogenemia in PCOS Pathophysiology. Genet. Res. Int. 2018, 2018, 7624932. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Bao, Y.; Zhou, X.; Zheng, L. Polycystic Ovary Syndrome and Mitochondrial Dysfunction. Reprod. Biol. Endocrinol. 2019, 17, 67. [Google Scholar] [CrossRef] [PubMed]
- Boirie, Y. Insulin Regulation of Mitochondrial Proteins and Oxidative Phosphorylation in Human Muscle. Trends Endocrinol. Metab. 2003, 14, 393–394. [Google Scholar] [CrossRef] [PubMed]
- Skov, V.; Glintborg, D.; Knudsen, S.; Jensen, T.; Kruse, T.A.; Tan, Q.; Brusgaard, K.; Beck-Nielsen, H.; Højlund, K. Reduced Expression of Nuclear-Encoded Genes Involved in Mitochondrial Oxidative Metabolism in Skeletal Muscle of Insulin-Resistant Women With Polycystic Ovary Syndrome. Diabetes 2007, 56, 2349–2355. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.; Hou, T.; Cheng, H.; Wang, X. NDUFAB1 Protects against Obesity and Insulin Resistance by Enhancing Mitochondrial Metabolism. FASEB J. 2019, 33, 13310–13322. [Google Scholar] [CrossRef]
- Kolodziejczyk, B.; Duleba, A.J.; Spaczynski, R.Z.; Pawelczyk, L. Metformin Therapy Decreases Hyperandrogenism and Hyperinsulinemia in Women with Polycystic Ovary Syndrome. Fertil. Steril. 2000, 73, 1149–1154. [Google Scholar] [CrossRef]
- Hirsch, A.; Hahn, D.; Kempná, P.; Hofer, G.; Nuoffer, J.-M.; Mullis, P.E.; Flück, C.E. Metformin Inhibits Human Androgen Production by Regulating Steroidogenic Enzymes HSD3B2 and CYP17A1 and Complex I Activity of the Respiratory Chain. Endocrinology 2012, 153, 4354–4366. [Google Scholar] [CrossRef]
- Li, Y.; Fromme, T. Uncoupling Protein 1 Does Not Produce Heat without Activation. Int. J. Mol. Sci. 2022, 23, 2406. [Google Scholar] [CrossRef] [PubMed]
- Carpentier, A.C.; Blondin, D.P.; Virtanen, K.A.; Richard, D.; Haman, F.; Turcotte, É.E. Brown Adipose Tissue Energy Metabolism in Humans. Front. Endocrinol. 2018, 9, 447. [Google Scholar] [CrossRef] [PubMed]
- Shorakae, S.; Jona, E.; De Courten, B.; Lambert, G.W.; Lambert, E.A.; Phillips, S.E.; Clarke, I.J.; Teede, H.J.; Henry, B.A. Brown Adipose Tissue Thermogenesis in Polycystic Ovary Syndrome. Clin. Endocrinol. 2018, 90, 425–432. [Google Scholar] [CrossRef] [PubMed]
- Yuan, X.; Hu, T.; Zhao, H.; Huang, Y.; Ye, R.; Lin, J.; Zhang, C.; Zhang, H.; Wei, G.; Zhou, H.; et al. Brown Adipose Tissue Transplantation Ameliorates Polycystic Ovary Syndrome. Proc. Natl. Acad. Sci. USA 2016, 113, 2708–2713. [Google Scholar] [CrossRef]
- Liu, Y.; Jiang, H.; He, L.-Y.; Huang, W.-J.; He, X.-Y.; Xing, F.-Q. Abnormal Expression of Uncoupling Protein-2 Correlates with CYP11A1 Expression in Polycystic Ovary Syndrome. Reprod. Fertil. Dev. 2011, 23, 520. [Google Scholar] [CrossRef]
- Darling, A.M.; Chavarro, J.E.; Malspeis, S.; Harris, H.R.; Missmer, S.A. A Prospective Cohort Study of Vitamins B, C, E, and Multivitamin Intake and Endometriosis. J. Endometr. Pelvic Pain Disord. 2013, 5, 17–26. [Google Scholar] [CrossRef]
- Heidari, H.; Hajhashemy, Z.; Saneei, P. A Meta-Analysis of Effects of Vitamin E Supplementation Alone and in Combination with Omega-3 or Magnesium on Polycystic Ovary Syndrome. Sci. Rep. 2022, 12, 19927. [Google Scholar] [CrossRef]
- Olaniyan, O.T.; Femi, A.; Iliya, G.; Ayobami, D.; Godam, E.; Olugbenga, E.; Bamidele, O.; Chand Mali, P. Vitamin C Suppresses Ovarian Pathophysiology in Experimental Polycystic Ovarian Syndrome. Pathophysiology 2019, 26, 331–341. [Google Scholar] [CrossRef]
- Wong, S.K.; Chin, K.-Y.; Ima-Nirwana, S. Vitamin C: A Review on Its Role in the Management of Metabolic Syndrome. Int. J. Med. Sci. 2020, 17, 1625–1638. [Google Scholar] [CrossRef]
- Mumford, S.L.; Browne, R.W.; Schliep, K.C.; Schmelzer, J.; Plowden, T.C.; Michels, K.A.; Sjaarda, L.A.; Zarek, S.M.; Perkins, N.J.; Messer, L.C.; et al. Serum Antioxidants Are Associated with Serum Reproductive Hormones and Ovulation among Healthy Women. J. Nutr. 2016, 146, 98–106. [Google Scholar] [CrossRef]
- Dludla, P.V.; Nkambule, B.B.; Nyambuya, T.M.; Ziqubu, K.; Mabhida, S.E.; Mxinwa, V.; Mokgalaboni, K.; Ndevahoma, F.; Hanser, S.; Mazibuko-Mbeje, S.E.; et al. Vitamin C Intake Potentially Lowers Total Cholesterol to Improve Endothelial Function in Diabetic Patients at Increased Risk of Cardiovascular Disease: A Systematic Review of Randomized Controlled Trials. Front. Nutr. 2022, 9, 1011002. [Google Scholar] [CrossRef] [PubMed]
- McRae, M.P. Vitamin C Supplementation Lowers Serum Low-Density Lipoprotein Cholesterol and Triglycerides: A Meta-Analysis of 13 Randomized Controlled Trials. J. Chiropr. Med. 2008, 7, 48–58. [Google Scholar] [CrossRef] [PubMed]
- Tenório, M.C.D.S.; Graciliano, N.G.; Moura, F.A.; Oliveira, A.C.M.D.; Goulart, M.O.F. N-Acetylcysteine (NAC): Impacts on Human Health. Antioxidants 2021, 10, 967. [Google Scholar] [CrossRef] [PubMed]
- Nemati, M.; Nemati, S.; Taheri, A.-M.; Heidari, B. Comparison of Metformin and N-Acetyl Cysteine, as an Adjuvant to Clomiphene Citrate, in Clomiphene-Resistant Women with Polycystic Ovary Syndrome. J. Gynecol. Obstet. Hum. Reprod. 2017, 46, 579–585. [Google Scholar] [CrossRef] [PubMed]
- Sandhu, J.K.; Waqar, A.; Jain, A.; Joseph, C.; Srivastava, K.; Ochuba, O.; Alkayyali, T.; Ruo, S.W.; Poudel, S. Oxidative Stress in Polycystic Ovarian Syndrome and the Effect of Antioxidant N-Acetylcysteine on Ovulation and Pregnancy Rate. Cureus 2021, 13, e17887. [Google Scholar] [CrossRef]
- Thakker, D.; Raval, A.; Patel, I.; Walia, R. N-Acetylcysteine for Polycystic Ovary Syndrome: A Systematic Review and Meta-Analysis of Randomized Controlled Clinical Trials. Obstet. Gynecol. Int. 2015, 2015, 817849. [Google Scholar] [CrossRef]
- Gayatri, K.; Kumar, J.S.; Kumar, B.B. Metformin and N-Acetyl Cysteine in Polycystic Ovarian Syndrome—A Comparative Study. Indian J. Clin. Med. 2010, 1, 117739361000100. [Google Scholar] [CrossRef]
- Behrouzi Lak, T.; Hajshafiha, M.; Nanbakhsh, F.; Oshnouei, S. N-Acetyl Cysteine in Ovulation Induction of PCOS Women Underwent Intrauterine Insemination: An RCT. Int. J. Reprod. Biomed. 2017, 15, 203–208. [Google Scholar] [CrossRef]
- Shahveghar Asl, Z.; Parastouei, K.; Eskandari, E. The Effects of N-Acetylcysteine on Ovulation and Sex Hormones Profile in Women with Polycystic Ovary Syndrome: A Systematic Review and Meta-Analysis. Br. J. Nutr. 2023, 130, 202–210. [Google Scholar] [CrossRef]
- Anastasi, E.; Scaramuzzino, S.; Viscardi, M.F.; Viggiani, V.; Piccioni, M.G.; Cacciamani, L.; Merlino, L.; Angeloni, A.; Muzii, L.; Porpora, M.G. Efficacy of N-Acetylcysteine on Endometriosis-Related Pain, Size Reduction of Ovarian Endometriomas, and Fertility Outcomes. Int. J. Environ. Res. Public Health 2023, 20, 4686. [Google Scholar] [CrossRef]
- Dinicola, S.; Minini, M.; Unfer, V.; Verna, R.; Cucina, A.; Bizzarri, M. Nutritional and Acquired Deficiencies in Inositol Bioavailability. Correlations with Metabolic Disorders. Int. J. Mol. Sci. 2017, 18, 2187. [Google Scholar] [CrossRef] [PubMed]
- Greff, D.; Juhász, A.E.; Váncsa, S.; Váradi, A.; Sipos, Z.; Szinte, J.; Park, S.; Hegyi, P.; Nyirády, P.; Ács, N.; et al. Inositol Is an Effective and Safe Treatment in Polycystic Ovary Syndrome: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Reprod. Biol. Endocrinol. 2023, 21, 10. [Google Scholar] [CrossRef] [PubMed]
- Tilak, J.R.; Jain, A.; Wadhwa, N.; Tilak, H.R.; Ahirwar, A.K. The Study of the Role of Insulin Resistance as Etiological Factor in Polycystic Ovarian Syndrome: A Case Control Study. Adv. Lab. Med. Av. Med. Lab. 2022, 3, 201–204. [Google Scholar] [CrossRef] [PubMed]
- March, W.A.; Moore, V.M.; Willson, K.J.; Phillips, D.I.W.; Norman, R.J.; Davies, M.J. The Prevalence of Polycystic Ovary Syndrome in a Community Sample Assessed under Contrasting Diagnostic Criteria. Hum. Reprod. 2010, 25, 544–551. [Google Scholar] [CrossRef] [PubMed]
- Unluhizarci, K.; Karaca, Z.; Kelestimur, F. Role of Insulin and Insulin Resistance in Androgen Excess Disorders. World J. Diabetes 2021, 12, 616–629. [Google Scholar] [CrossRef] [PubMed]
- Kalra, B.; Kalra, S.; Sharma, J. The Inositols and Polycystic Ovary Syndrome. Indian. J. Endocr. Metab. 2016, 20, 720. [Google Scholar] [CrossRef]
- Monastra, G.; Vazquez-Levin, M.; Bezerra Espinola, M.S.; Bilotta, G.; Laganà, A.S.; Unfer, V. D-Chiro-Inositol, an Aromatase down-Modulator, Increases Androgens and Reduces Estrogens in Male Volunteers: A Pilot Study. Basic Clin. Androl. 2021, 31, 13. [Google Scholar] [CrossRef]
- Unfer, V.; Facchinetti, F.; Orrù, B.; Giordani, B.; Nestler, J. Myo-Inositol Effects in Women with PCOS: A Meta-Analysis of Randomized Controlled Trials. Endocr. Connect. 2017, 6, 647–658. [Google Scholar] [CrossRef]
- Facchinetti, F.; Bizzarri, M.; Benvenga, S.; D’Anna, R.; Lanzone, A.; Soulage, C.; Di Renzo, G.C.; Hod, M.; Cavalli, P.; Chiu, T.T.; et al. Results from the International Consensus Conference on Myo-Inositol and d-Chiro-Inositol in Obstetrics and Gynecology: The Link between Metabolic Syndrome and PCOS. Eur. J. Obstet. Gynecol. Reprod. Biol. 2015, 195, 72–76. [Google Scholar] [CrossRef]
- Teede, H.J.; Tay, C.T.; Laven, J.J.E.; Dokras, A.; Moran, L.J.; Piltonen, T.T.; Costello, M.F.; Boivin, J.; Redman, L.M.; Boyle, J.A.; et al. Recommendations From the 2023 International Evidence-Based Guideline for the Assessment and Management of Polycystic Ovary Syndrome. J. Clin. Endocrinol. Metab. 2023, dgad463. [Google Scholar] [CrossRef]
- Teede, H.J.; Misso, M.L.; Costello, M.F.; Dokras, A.; Laven, J.; Moran, L.; Piltonen, T.; Norman, R.J.; Andersen, M.; Azziz, R.; et al. Recommendations from the International Evidence-Based Guideline for the Assessment and Management of Polycystic Ovary Syndrome. Fertil. Steril. 2018, 110, 364–379. [Google Scholar] [CrossRef] [PubMed]
- Chukwunonso Obi, B.; Chinwuba Okoye, T.; Okpashi, V.E.; Nonye Igwe, C.; Olisah Alumanah, E. Comparative Study of the Antioxidant Effects of Metformin, Glibenclamide, and Repaglinide in Alloxan-Induced Diabetic Rats. J. Diabetes Res. 2016, 2016, 1635361. [Google Scholar] [CrossRef] [PubMed]
- Rena, G.; Hardie, D.G.; Pearson, E.R. The Mechanisms of Action of Metformin. Diabetologia 2017, 60, 1577–1585. [Google Scholar] [CrossRef] [PubMed]
- Goel, S.; Singh, R.; Singh, V.; Singh, H.; Kumari, P.; Chopra, H.; Sharma, R.; Nepovimova, E.; Valis, M.; Kuca, K.; et al. Metformin: Activation of 5′ AMP-Activated Protein Kinase and Its Emerging Potential beyond Anti-Hyperglycemic Action. Front. Genet. 2022, 13, 1022739. [Google Scholar] [CrossRef]
- Bai, B.; Chen, H. Metformin: A Novel Weapon Against Inflammation. Front. Pharmacol. 2021, 12, 622262. [Google Scholar] [CrossRef]
- Salvatore, T.; Pafundi, P.C.; Galiero, R.; Rinaldi, L.; Caturano, A.; Vetrano, E.; Aprea, C.; Albanese, G.; Di Martino, A.; Ricozzi, C.; et al. Can Metformin Exert as an Active Drug on Endothelial Dysfunction in Diabetic Subjects? Biomedicines 2020, 9, 3. [Google Scholar] [CrossRef] [PubMed]
- Sam, S.; Ehrmann, D.A. Metformin Therapy for the Reproductive and Metabolic Consequences of Polycystic Ovary Syndrome. Diabetologia 2017, 60, 1656–1661. [Google Scholar] [CrossRef]
- Zhao, J.; Dong, L.; Lin, Z.; Sui, X.; Wang, Y.; Li, L.; Liu, T.; Liu, J. Effects of Selenium Supplementation on Polycystic Ovarian Syndrome: A Systematic Review and Meta-Analysis on Randomized Clinical Trials. BMC Endocr. Disord. 2023, 23, 33. [Google Scholar] [CrossRef]
- Jamilian, M.; Mansury, S.; Bahmani, F.; Heidar, Z.; Amirani, E.; Asemi, Z. The Effects of Probiotic and Selenium Co-Supplementation on Parameters of Mental Health, Hormonal Profiles, and Biomarkers of Inflammation and Oxidative Stress in Women with Polycystic Ovary Syndrome. J. Ovarian Res. 2018, 11, 80. [Google Scholar] [CrossRef]
- Razavi, M.; Jamilian, M.; Kashan, Z.F.; Heidar, Z.; Mohseni, M.; Ghandi, Y.; Bagherian, T.; Asemi, Z. Selenium Supplementation and the Effects on Reproductive Outcomes, Biomarkers of Inflammation, and Oxidative Stress in Women with Polycystic Ovary Syndrome. Horm. Metab. Res. 2016, 48, 185–190. [Google Scholar] [CrossRef]
- Ventura, M.; Melo, M.; Carrilho, F. Selenium and Thyroid Disease: From Pathophysiology to Treatment. Int. J. Endocrinol. 2017, 2017, 1297658. [Google Scholar] [CrossRef] [PubMed]
- Le, S.N.; Porebski, B.T.; McCoey, J.; Fodor, J.; Riley, B.; Godlewska, M.; Góra, M.; Czarnocka, B.; Banga, J.P.; Hoke, D.E.; et al. Modelling of Thyroid Peroxidase Reveals Insights into Its Enzyme Function and Autoantigenicity. PLoS ONE 2015, 10, e0142615. [Google Scholar] [CrossRef] [PubMed]
- Sabatino, L.; Vassalle, C.; Del Seppia, C.; Iervasi, G. Deiodinases and the Three Types of Thyroid Hormone Deiodination Reactions. Endocrinol. Metab. 2021, 36, 952–964. [Google Scholar] [CrossRef] [PubMed]
- Kieliszek, M. Selenium–Fascinating Microelement, Properties and Sources in Food. Molecules 2019, 24, 1298. [Google Scholar] [CrossRef]
- Zeber-Lubecka, N.; Hennig, E.E. Genetic Susceptibility to Joint Occurrence of Polycystic Ovary Syndrome and Hashimoto’s Thyroiditis: How Far Is Our Understanding? Front. Immunol. 2021, 12, 606620. [Google Scholar] [CrossRef]
- Zeber-Lubecka, N.; Suchta, K.; Kulecka, M.; Kluska, A.; Piątkowska, M.; Dabrowski, M.J.; Jankowska, K.; Grymowicz, M.; Smolarczyk, R.; Hennig, E.E. Exome Sequencing to Explore the Possibility of Predicting Genetic Susceptibility to the Joint Occurrence of Polycystic Ovary Syndrome and Hashimoto’s Thyroiditis. Front. Immunol. 2023, 14, 1193293. [Google Scholar] [CrossRef]
- Zhang, J.; Xing, C.; Zhao, H.; He, B. The Effectiveness of Coenzyme Q10, Vitamin E, Inositols, and Vitamin D in Improving the Endocrine and Metabolic Profiles in Women with Polycystic Ovary Syndrome: A Network Meta-Analysis. Gynecol. Endocrinol. 2021, 37, 1063–1071. [Google Scholar] [CrossRef]
- Menichini, D.; Ughetti, C.; Monari, F.; Di Vinci, P.L.; Neri, I.; Facchinetti, F. Nutraceuticals and Polycystic Ovary Syndrome: A Systematic Review of the Literature. Gynecol. Endocrinol. 2022, 38, 623–631. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zeber-Lubecka, N.; Ciebiera, M.; Hennig, E.E. Polycystic Ovary Syndrome and Oxidative Stress—From Bench to Bedside. Int. J. Mol. Sci. 2023, 24, 14126. https://doi.org/10.3390/ijms241814126
Zeber-Lubecka N, Ciebiera M, Hennig EE. Polycystic Ovary Syndrome and Oxidative Stress—From Bench to Bedside. International Journal of Molecular Sciences. 2023; 24(18):14126. https://doi.org/10.3390/ijms241814126
Chicago/Turabian StyleZeber-Lubecka, Natalia, Michał Ciebiera, and Ewa E. Hennig. 2023. "Polycystic Ovary Syndrome and Oxidative Stress—From Bench to Bedside" International Journal of Molecular Sciences 24, no. 18: 14126. https://doi.org/10.3390/ijms241814126






