Small Organic Compounds Mimicking the Effector Domain of Myristoylated Alanine-Rich C-Kinase Substrate Stimulate Female-Specific Neurite Outgrowth
Abstract
:1. Introduction
2. Results
2.1. Identification of a MARCKS Antibody That Binds to the ED Peptide
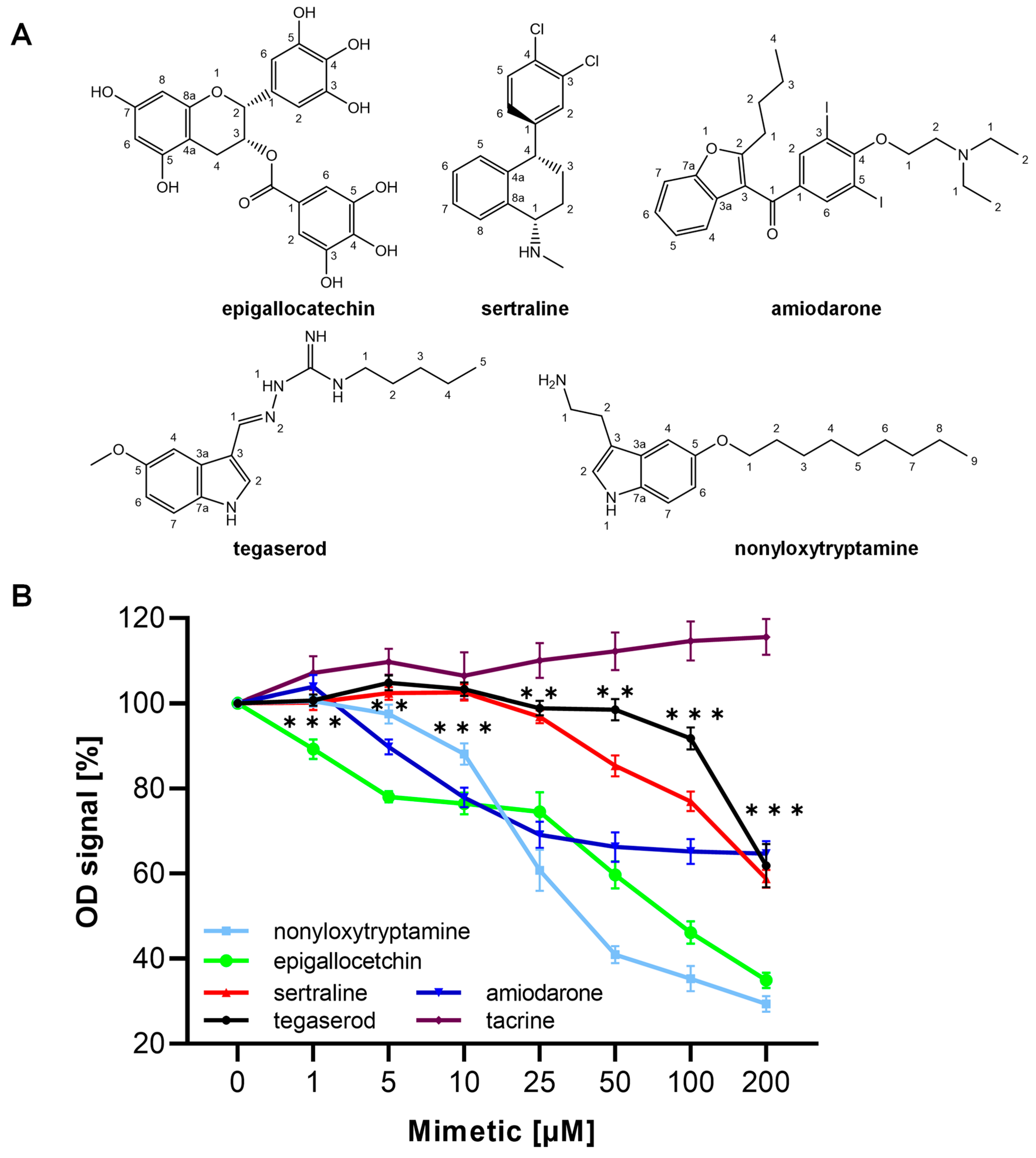
2.2. ED Peptide Mimetics Inhibited the Binding of ED Peptide to the MARCKS Antibody
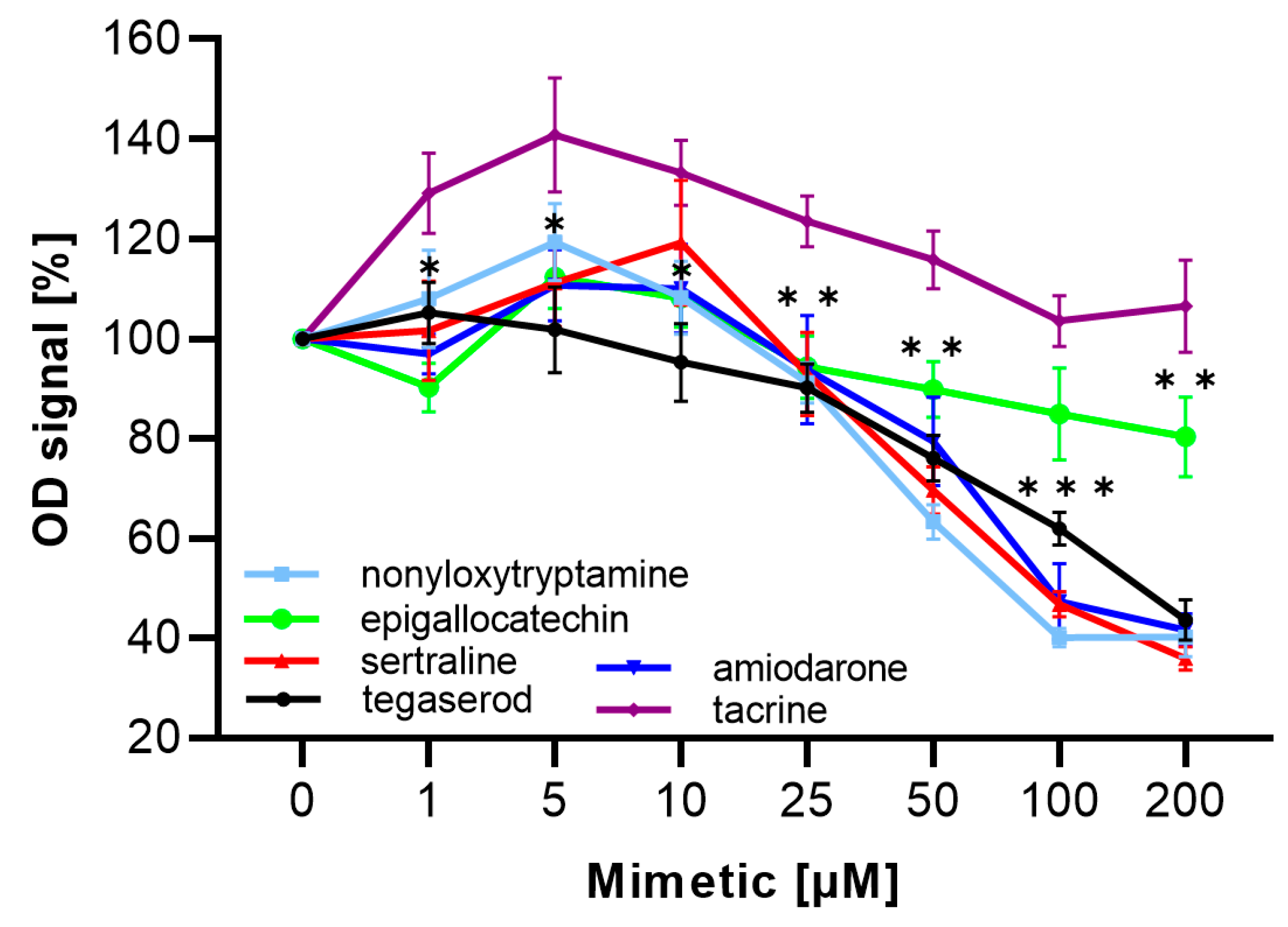
2.3. ED Peptide Mimetics Bound to PSA
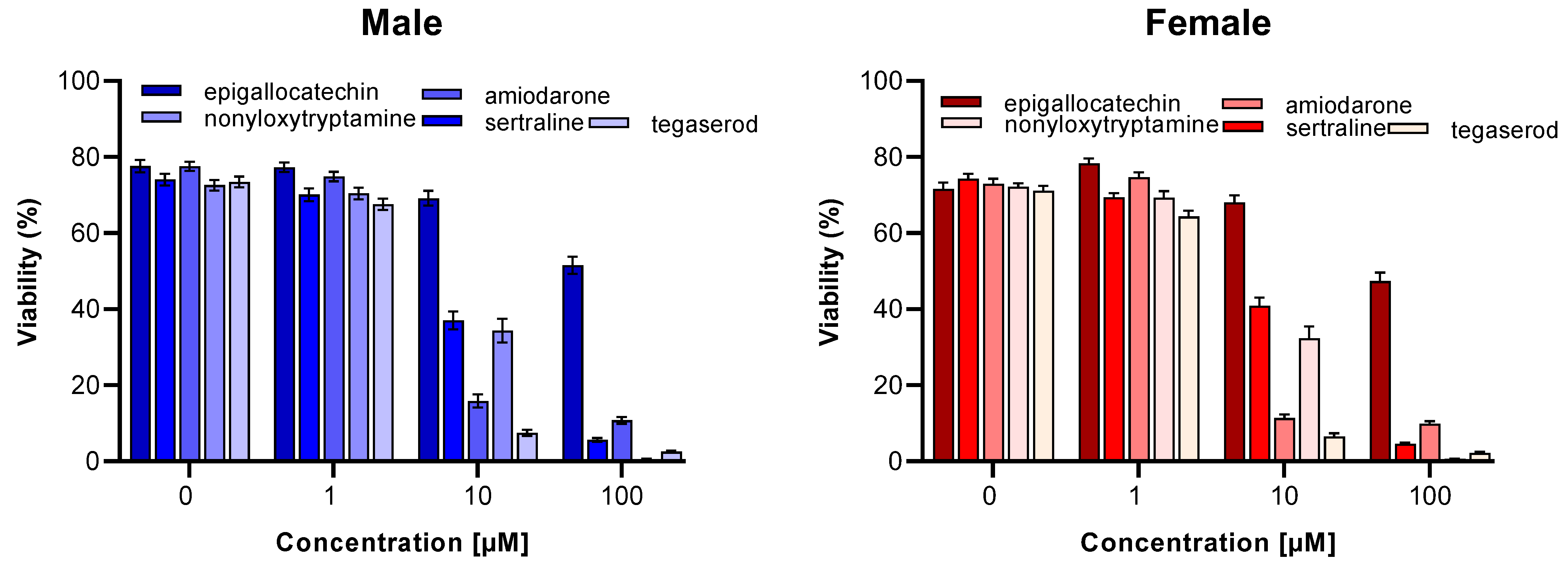
2.4. Effect of ED Peptide Mimetics on Neuronal Viability
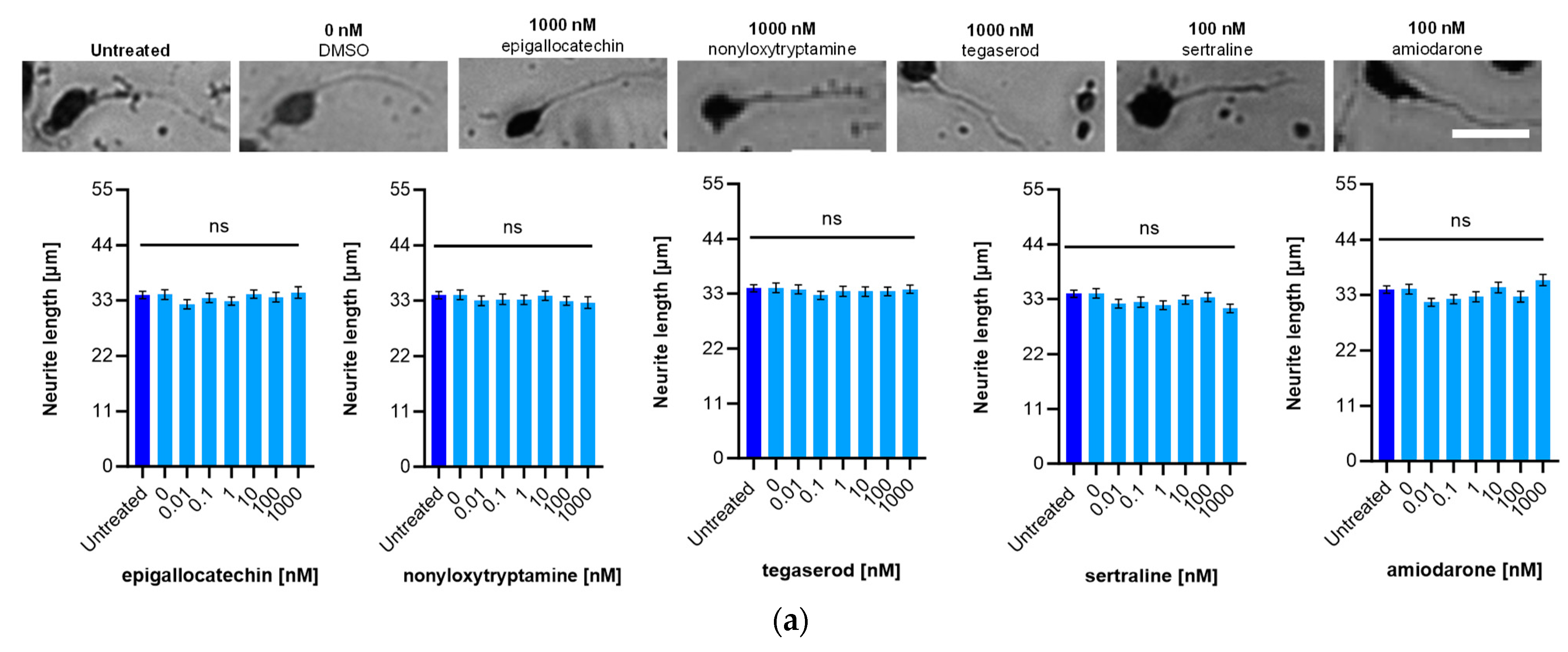
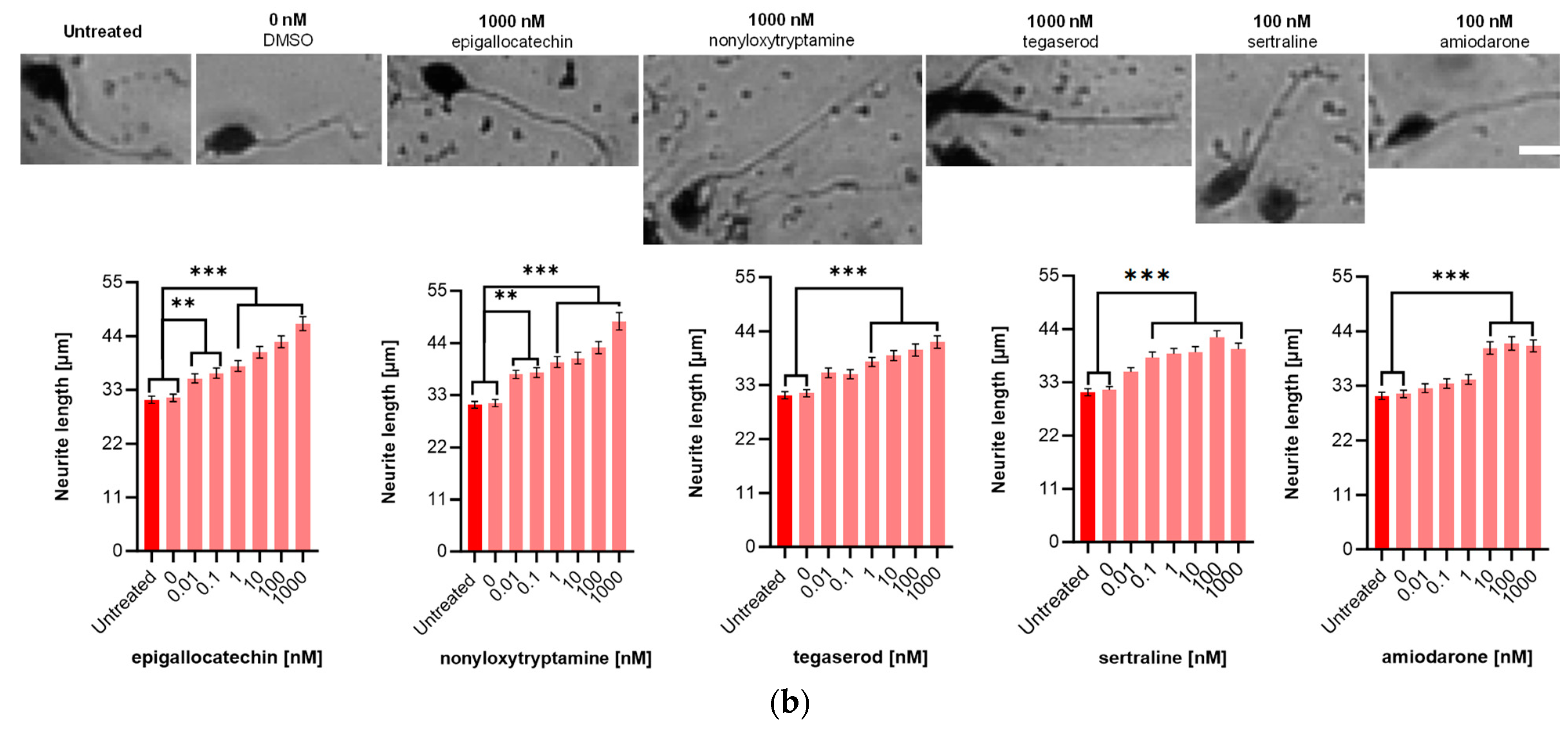
2.5. Effect of ED Peptide Mimetics on Neurite Outgrowth
3. Discussion
4. Materials and Methods
4.1. Animals
4.2. Antibodies and Small Compounds
4.3. Enzyme-Linked Immunosorbent Assay (ELISA) to Screen for ED Peptide Mimetics
4.4. Cerebellar Granule Cell Culture
4.5. Toxicity Assay
4.6. Neurite Outgrowth
4.7. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Loers, G.; Astafiev, S.; Hapiak, Y.; Saini, V.; Mishra, B.; Gul, S.; Kaur, G.; Schachner, M.; Theis, T. The Polysialic Acid Mimetics Idarubicin and Irinotecan Stimulate Neuronal Survival and Neurite Outgrowth and Signal via Protein Kinase C. J. Neurochem. 2017, 142, 392–406. [Google Scholar] [CrossRef]
- Theis, T.; Johal, A.S.; Kabat, M.; Basak, S.; Schachner, M. Enhanced Neuronal Survival and Neurite Outgrowth Triggered by Novel Small Organic Compounds Mimicking the LewisX Glycan. Mol. Neurobiol. 2018, 55, 8203–8215. [Google Scholar] [CrossRef]
- Wang, M.; Theis, T.; Kabat, M.; Loers, G.; Agre, L.A.; Schachner, M. Functions of Small Organic Compounds That Mimic the HNK-1 Glycan. Int. J. Mol. Sci. 2020, 21, 7018. [Google Scholar] [CrossRef]
- Sytnyk, V.; Leshchyns’ka, I.; Schachner, M. Neural Cell Adhesion Molecules of the Immunoglobulin Superfamily Regulate Synapse Formation, Maintenance, and Function. Trends Neurosci. 2017, 40, 295–308. [Google Scholar] [CrossRef]
- Sytnyk, V.; Leshchyns’ka, I.; Schachner, M. Neural Glycomics: The Sweet Side of Nervous System Functions. Cell. Mol. Life Sci. 2021, 78, 93–116. [Google Scholar] [CrossRef]
- Hildebrandt, H.; Dityatev, A. Polysialic Acid in Brain Development and Synaptic Plasticity. Top Curr. Chem. 2015, 366, 55–96. [Google Scholar] [CrossRef]
- Rutishauser, U. Polysialic Acid in the Plasticity of the Developing and Adult Vertebrate Nervous System. Nat. Rev. Neurosci. 2008, 9, 26–35. [Google Scholar] [CrossRef]
- Kiermaier, E.; Moussion, C.; Veldkamp, C.T.; Gerardy-Schahn, R.; de Vries, I.; Williams, L.G.; Chaffee, G.R.; Phillips, A.J.; Freiberger, F.; Imre, R.; et al. Polysialylation Controls Dendritic Cell Trafficking by Regulating Chemokine Recognition. Science 2016, 351, 186–190. [Google Scholar] [CrossRef]
- Mühlenhoff, M.; Rollenhagen, M.; Werneburg, S.; Gerardy-Schahn, R.; Hildebrandt, H. Polysialic Acid: Versatile Modification of NCAM, SynCAM 1 and Neuropilin-2. Neurochem. Res. 2013, 38, 1134–1143. [Google Scholar] [CrossRef]
- Kleene, R.; Schachner, M. Glycans and Neural Cell Interactions. Nat. Rev. Neurosci. 2004, 5, 195–208. [Google Scholar] [CrossRef]
- A Polysialic Acid Mimetic Peptide Promotes Functional Recovery in a Mouse Model of Spinal Cord Injury—PubMed. Available online: https://pubmed.ncbi.nlm.nih.gov/19445935/ (accessed on 26 July 2023).
- Mehanna, A.; Jakovcevski, I.; Acar, A.; Xiao, M.; Loers, G.; Rougon, G.; Irintchev, A.; Schachner, M. Polysialic Acid Glycomimetic Promotes Functional Recovery and Plasticity after Spinal Cord Injury in Mice. Mol. Ther. 2010, 18, 34–43. [Google Scholar] [CrossRef] [PubMed]
- Mehanna, A.; Mishra, B.; Kurschat, N.; Schulze, C.; Bian, S.; Loers, G.; Irintchev, A.; Schachner, M. Polysialic Acid Glycomimetics Promote Myelination and Functional Recovery after Peripheral Nerve Injury in Mice. Brain 2009, 132, 1449–1462. [Google Scholar] [CrossRef] [PubMed]
- Pan, H.-C.; Shen, Y.-Q.; Loers, G.; Jakovcevski, I.; Schachner, M. Tegaserod, a Small Compound Mimetic of Polysialic Acid, Promotes Functional Recovery after Spinal Cord Injury in Mice. Neuroscience 2014, 277, 356–366. [Google Scholar] [CrossRef] [PubMed]
- Amoureux, M.-C.; Coulibaly, B.; Chinot, O.; Loundou, A.; Metellus, P.; Rougon, G.; Figarella-Branger, D. Polysialic Acid Neural Cell Adhesion Molecule (PSA-NCAM) Is an Adverse Prognosis Factor in Glioblastoma, and Regulates Olig2 Expression in Glioma Cell Lines. BMC Cancer 2010, 10, 91. [Google Scholar] [CrossRef] [PubMed]
- Elkashef, S.M.; Allison, S.J.; Sadiq, M.; Basheer, H.A.; Ribeiro Morais, G.; Loadman, P.M.; Pors, K.; Falconer, R.A. Polysialic Acid Sustains Cancer Cell Survival and Migratory Capacity in a Hypoxic Environment. Sci. Rep. 2016, 6, 33026. [Google Scholar] [CrossRef]
- Guo, X.; Elkashef, S.M.; Patel, A.; Ribeiro Morais, G.; Shnyder, S.D.; Loadman, P.M.; Patterson, L.H.; Falconer, R.A. An Assay for Quantitative Analysis of Polysialic Acid Expression in Cancer Cells. Carbohydr. Polym. 2021, 259, 117741. [Google Scholar] [CrossRef]
- Hromatka, B.S.; Drake, P.M.; Kapidzic, M.; Stolp, H.; Goldfien, G.A.; Shih, I.-M.; Fisher, S.J. Polysialic Acid Enhances the Migration and Invasion of Human Cytotrophoblasts. Glycobiology 2013, 23, 593–602. [Google Scholar] [CrossRef]
- Villanueva-Cabello, T.M.; Gutiérrez-Valenzuela, L.D.; Salinas-Marín, R.; López-Guerrero, D.V.; Martínez-Duncker, I. Polysialic Acid in the Immune System. Front. Immunol. 2021, 12, 823637. [Google Scholar] [CrossRef]
- Polysialyltransferase: A New Target in Metastatic Cancer—PubMed. Available online: https://pubmed.ncbi.nlm.nih.gov/22463390/ (accessed on 26 July 2023).
- Saini, V.; Kaur, T.; Kalotra, S.; Kaur, G. The Neuroplasticity Marker PSA-NCAM: Insights into New Therapeutic Avenues for Promoting Neuroregeneration. Pharmacol. Res. 2020, 160, 105186. [Google Scholar] [CrossRef]
- Kanato, Y.; Kitajima, K.; Sato, C. Direct Binding of Polysialic Acid to a Brain-Derived Neurotrophic Factor Depends on the Degree of Polymerization. Glycobiology 2008, 18, 1044–1053. [Google Scholar] [CrossRef]
- Ono, S.; Hane, M.; Kitajima, K.; Sato, C. Novel Regulation of Fibroblast Growth Factor 2 (FGF2)-Mediated Cell Growth by Polysialic Acid. J. Biol. Chem. 2012, 287, 3710–3722. [Google Scholar] [CrossRef]
- Mishra, B.; von der Ohe, M.; Schulze, C.; Bian, S.; Makhina, T.; Loers, G.; Kleene, R.; Schachner, M. Functional Role of the Interaction between Polysialic Acid and Extracellular Histone H1. J. Neurosci. 2010, 30, 12400–12413. [Google Scholar] [CrossRef]
- Westphal, N.; Theis, T.; Loers, G.; Schachner, M.; Kleene, R. Nuclear Fragments of the Neural Cell Adhesion Molecule NCAM with or without Polysialic Acid Differentially Regulate Gene Expression. Sci. Rep. 2017, 7, 13631. [Google Scholar] [CrossRef]
- Theis, T.; Mishra, B.; von der Ohe, M.; Loers, G.; Prondzynski, M.; Pless, O.; Blackshear, P.J.; Schachner, M.; Kleene, R. Functional Role of the Interaction between Polysialic Acid and Myristoylated Alanine-Rich C Kinase Substrate at the Plasma Membrane. J. Biol. Chem. 2013, 288, 6726–6742. [Google Scholar] [CrossRef] [PubMed]
- Albert, K.A.; Nairn, A.C.; Greengard, P. The 87-KDa Protein, a Major Specific Substrate for Protein Kinase C: Purification from Bovine Brain and Characterization. Proc. Natl. Acad. Sci. USA 1987, 84, 7046–7050. [Google Scholar] [CrossRef]
- Swierczynski, S.L.; Siddhanti, S.R.; Tuttle, J.S.; Blackshear, P.J. Nonmyristoylated MARCKS Complements Some but Not All of the Developmental Defects Associated with MARCKS Deficiency in Mice. Dev. Biol. 1996, 179, 135–147. [Google Scholar] [CrossRef]
- Vergères, G.; Ramsden, J.J. Binding of MARCKS (Myristoylated Alanine-Rich C Kinase Substrate)-Related Protein (MRP) to Vesicular Phospholipid Membranes. Biochem. J. 1998, 330 Pt 1, 5–11. [Google Scholar] [CrossRef]
- Swierczynski, S.L.; Blackshear, P.J. Membrane Association of the Myristoylated Alanine-Rich C Kinase Substrate (MARCKS) Protein. Mutational Analysis Provides Evidence for Complex Interactions. J. Biol. Chem. 1995, 270, 13436–13445. [Google Scholar] [CrossRef]
- Theis, T.; Kumar, S.; Wei, E.; Nguyen, J.; Glynos, V.; Paranjape, N.; Askarifirouzjaei, H.; Khajouienejad, L.; Berthiaume, F.; Young, W.; et al. Myristoylated Alanine-Rich C-Kinase Substrate Effector Domain Peptide Improves Sex-Specific Recovery and Axonal Regrowth after Spinal Cord Injury. FASEB J. 2020, 34, 12677–12690. [Google Scholar] [CrossRef]
- Uhlén, M.; Fagerberg, L.; Hallström, B.M.; Lindskog, C.; Oksvold, P.; Mardinoglu, A.; Sivertsson, Å.; Kampf, C.; Sjöstedt, E.; Asplund, A.; et al. Proteomics. Tissue-Based Map of the Human Proteome. Science 2015, 347, 1260419. [Google Scholar] [CrossRef]
- Kataria, H.; Lutz, D.; Chaudhary, H.; Schachner, M.; Loers, G. Small Molecule Agonists of Cell Adhesion Molecule L1 Mimic L1 Functions In Vivo. Mol. Neurobiol. 2016, 53, 4461–4483. [Google Scholar] [CrossRef] [PubMed]
- Bushman, J.; Mishra, B.; Ezra, M.; Gul, S.; Schulze, C.; Chaudhury, S.; Ripoll, D.; Wallqvist, A.; Kohn, J.; Schachner, M.; et al. Tegaserod Mimics the Neurostimulatory Glycan Polysialic Acid and Promotes Nervous System Repair. Neuropharmacology 2014, 79, 456–466. [Google Scholar] [CrossRef] [PubMed]
- Loers, G.; Saini, V.; Mishra, B.; Gul, S.; Chaudhury, S.; Wallqvist, A.; Kaur, G.; Schachner, M. Vinorelbine and Epirubicin Share Common Features with Polysialic Acid and Modulate Neuronal and Glial Functions. J. Neurochem. 2016, 136, 48–62. [Google Scholar] [CrossRef] [PubMed]
- Saini, V.; Lutz, D.; Kataria, H.; Kaur, G.; Schachner, M.; Loers, G. The Polysialic Acid Mimetics 5-Nonyloxytryptamine and Vinorelbine Facilitate Nervous System Repair. Sci. Rep. 2016, 6, 26927. [Google Scholar] [CrossRef]
- Nagaraj, V.; Kim, R.; Martianou, T.; Kurian, S.; Nayak, A.; Patel, M.; Schachner, M.; Theis, T. Effects of L1 Adhesion Molecule Agonistic Mimetics on Signal Transduction in Neuronal Functions. Biochem Biophys. Res. Commun. 2023, 642, 27–34. [Google Scholar] [CrossRef]
- DeVane, C.L.; Liston, H.L.; Markowitz, J.S. Clinical Pharmacokinetics of Sertraline. Clin. Pharmacokinet. 2002, 41, 1247–1266. [Google Scholar] [CrossRef]
- Evans, B.W.; Clark, W.K.; Moore, D.J.; Whorwell, P.J. Tegaserod for the Treatment of Irritable Bowel Syndrome and Chronic Constipation. Cochrane Database Syst. Rev. 2007, CD003960. [Google Scholar] [CrossRef]
- Freedman, M.D.; Somberg, J.C. Pharmacology and Pharmacokinetics of Amiodarone. J. Clin. Pharmacol. 1991, 31, 1061–1069. [Google Scholar] [CrossRef]
- Glennon, R.A.; Hong, S.S.; Dukat, M.; Teitler, M.; Davis, K. 5-(Nonyloxy)Tryptamine: A Novel High-Affinity 5-HT1D Beta Serotonin Receptor Agonist. J. Med. Chem. 1994, 37, 2828–2830. [Google Scholar] [CrossRef]
- Pervin, M.; Unno, K.; Takagaki, A.; Isemura, M.; Nakamura, Y. Function of Green Tea Catechins in the Brain: Epigallocatechin Gallate and Its Metabolites. Int. J. Mol. Sci. 2019, 20, 3630. [Google Scholar] [CrossRef]
- Al-Amri, J.S.; Hagras, M.M.; Mohamed, I.M. Effect of Epigallocatechin-3-Gallate on Inflammatory Mediators Release in LPS-Induced Parkinson’s Disease in Rats. Indian J. Exp. Biol. 2013, 51, 357–362. [Google Scholar] [PubMed]
- Renaud, J.; Nabavi, S.F.; Daglia, M.; Nabavi, S.M.; Martinoli, M.-G. Epigallocatechin-3-Gallate, a Promising Molecule for Parkinson’s Disease? Rejuvenation Res. 2015, 18, 257–269. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Zhang, Y.; Quan, Z.; Wong, W.; Guo, J.; Zhang, R.; Yang, Q.; Dai, R.; McGeer, P.L.; Qing, H. Epigallocatechin Gallate (EGCG) Inhibits Alpha-Synuclein Aggregation: A Potential Agent for Parkinson’s Disease. Neurochem. Res. 2016, 41, 2788–2796. [Google Scholar] [CrossRef] [PubMed]
- (-)-Epigallocatechin-3-Gallate Modulates Peripheral Immunity in the MPTP-Induced Mouse Model of Parkinson’s Disease—PubMed. Available online: https://pubmed.ncbi.nlm.nih.gov/29363729/ (accessed on 26 July 2023).
- Lin, X.; Liu, W.; Dong, X.; Sun, Y. Epigallocatechin Gallate-Derived Carbonized Polymer Dots: A Multifunctional Scavenger Targeting Alzheimer’s β-Amyloid Plaques. Acta Biomater. 2023, 157, 524–537. [Google Scholar] [CrossRef]
- Nan, S.; Wang, P.; Zhang, Y.; Fan, J. Epigallocatechin-3-Gallate Provides Protection Against Alzheimer’s Disease-Induced Learning and Memory Impairments in Rats. Drug Des. Dev. Ther. 2021, 15, 2013–2024. [Google Scholar] [CrossRef]
- Cai, F.; Liu, S.; Lei, Y.; Jin, S.; Guo, Z.; Zhu, D.; Guo, X.; Zhao, H.; Niu, X.; Xi, Y.; et al. Epigallocatechin-3 Gallate Regulates Macrophage Subtypes and Immunometabolism to Ameliorate Experimental Autoimmune Encephalomyelitis. Cell. Immunol. 2021, 368, 104421. [Google Scholar] [CrossRef]
- Wu, D. Green Tea EGCG, T-Cell Function, and T-Cell-Mediated Autoimmune Encephalomyelitis. J. Investig. Med. 2016, 64, 1213–1219. [Google Scholar] [CrossRef]
- Ahadi, S.; Zargari, M.; Khalatbary, A.R. Assessment of the Neuroprotective Effects of (-)-Epigallocatechin-3-Gallate on Spinal Cord Ischemia-Reperfusion Injury in Rats. J. Spinal Cord. Med. 2021, 44, 725–732. [Google Scholar] [CrossRef]
- Ge, R.; Zhu, Y.; Diao, Y.; Tao, L.; Yuan, W.; Xiong, X. Anti-Edema Effect of Epigallocatechin Gallate on Spinal Cord Injury in Rats. Brain Res. 2013, 1527, 40–46. [Google Scholar] [CrossRef]
- Lu, F.; Zhang, G.; Zhu, Y.; Liu, Z. (-)-Epigallocatechin Gallate Attenuates Spinal Motoneuron Death Induced by Brachial Plexus Root Avulsion in Rats. Curr. Med. Chem. 2022, 29, 5139–5154. [Google Scholar] [CrossRef]
- Eustace, N.J.; Anderson, J.C.; Warram, J.M.; Widden, H.N.; Pedersen, R.T.; Alrefai, H.; Patel, Z.; Hicks, P.H.; Placzek, W.J.; Gillespie, G.Y.; et al. A Cell-Penetrating MARCKS Mimetic Selectively Triggers Cytolytic Death in Glioblastoma. Oncogene 2020, 39, 6961–6974. [Google Scholar] [CrossRef]
- Sheats, M.K.; Yin, Q.; Fang, S.; Park, J.; Crews, A.L.; Parikh, I.; Dickson, B.; Adler, K.B. MARCKS and Lung Disease. Am. J. Respir. Cell. Mol. Biol. 2019, 60, 16–27. [Google Scholar] [CrossRef]
- Chen, P.B.; Kim, J.H.; Young, L.; Clark, J.M.; Park, Y. Epigallocatechin Gallate (EGCG) Alters Body Fat and Lean Mass through Sex-Dependent Metabolic Mechanisms in Drosophila Melanogaster. Int. J. Food Sci. Nutr. 2019, 70, 959–969. [Google Scholar] [CrossRef]
- Wellens, H.J.; Lie, K.I.; Bär, F.W.; Wesdorp, J.C.; Dohmen, H.J.; Düren, D.R.; Durrer, D. Effect of Amiodarone in the Wolff-Parkinson-White Syndrome. Am. J. Cardiol. 1976, 38, 189–194. [Google Scholar] [CrossRef]
- Traupe, T.; Keller, M.; Fojtu, E.; Bhattacharya, I.; Lang, M.; Ha, H.R.; Jochum, W.; Mundy, A.L.; Barton, M. Antioxidant Activity and Sex Differences of Acute Vascular Effects of Amiodarone in Advanced Atherosclerosis. J. Cardiovasc. Pharmacol. 2007, 50, 578–584. [Google Scholar] [CrossRef]
- Essebag, V.; Reynolds, M.R.; Hadjis, T.; Lemery, R.; Olshansky, B.; Buxton, A.E.; Josephson, M.E.; Zimetbaum, P. Sex Differences in the Relationship between Amiodarone Use and the Need for Permanent Pacing in Patients with Atrial Fibrillation. Arch. Intern. Med. 2007, 167, 1648–1653. [Google Scholar] [CrossRef] [PubMed]
- Ma, L.; Xu, Y.; Jiang, W.; Li, Y.; Zhang, X.; Wang, G.; Li, R. Sex Differences in Antidepressant Effect of Sertraline in Transgenic Mouse Models. Front. Cell. Neurosci. 2019, 13, 24. [Google Scholar] [CrossRef]
- Baca, E.; Garcia-Garcia, M.; Porras-Chavarino, A. Gender Differences in Treatment Response to Sertraline versus Imipramine in Patients with Nonmelancholic Depressive Disorders. Prog. Neuropsychopharmacol. Biol. Psychiatry. 2004, 28, 57–65. [Google Scholar] [CrossRef] [PubMed]
- Loughlin, J.; Quinn, S.; Rivero, E.; Wong, J.; Huang, J.; Kralstein, J.; Earnest, D.L.; Seeger, J.D. Tegaserod and the Risk of Cardiovascular Ischemic Events: An Observational Cohort Study. J. Cardiovasc. Pharmacol. Ther. 2010, 15, 151–157. [Google Scholar] [CrossRef]
- Madia, V.N.; Messore, A.; Saccoliti, F.; Tudino, V.; De Leo, A.; De Vita, D.; Bortolami, M.; Scipione, L.; Pindinello, I.; Costi, R.; et al. Tegaserod for the Treatment of Irritable Bowel Syndrome. Antiinflamm. Antiallergy Agents Med. Chem. 2020, 19, 342–369. [Google Scholar] [CrossRef] [PubMed]
- Degen, L.; Petrig, C.; Studer, D.; Schroller, S.; Beglinger, C. Effect of Tegaserod on Gut Transit in Male and Female Subjects. Neurogastroenterol. Motil. 2005, 17, 821–826. [Google Scholar] [CrossRef]
- Loers, G.; Saini, V.; Mishra, B.; Papastefanaki, F.; Lutz, D.; Chaudhury, S.; Ripoll, D.R.; Wallqvist, A.; Gul, S.; Schachner, M.; et al. Nonyloxytryptamine Mimics Polysialic Acid and Modulates Neuronal and Glial Functions in Cell Culture. J. Neurochem. 2014, 128, 88–100. [Google Scholar] [CrossRef]
- Al-Shboul, O.A.; Al-Dwairi, A.N.; Alqudah, M.A.; Mustafa, A.G. Gender Differences in the Regulation of MLC20 Phosphorylation and Smooth Muscle Contraction in Rat Stomach. Biomed. Rep. 2018, 8, 283–288. [Google Scholar] [CrossRef] [PubMed]
- Zangar, R.C.; Reiners, J.J.; Novak, R.F. Gender-Specific and Developmental Differences in Protein Kinase C Isozyme Expression in Rat Liver. Carcinogenesis 1995, 16, 2593–2597. [Google Scholar] [CrossRef] [PubMed]
- Nagaraj, V.; Mikhail, M.; Baronio, M.; Gatto, A.; Nayak, A.; Theis, T.; Cavallaro, U.; Schachner, M. Antagonistic L1 Adhesion Molecule Mimetic Compounds Inhibit Glioblastoma Cell Migration In Vitro. Biomolecules 2022, 12, 439. [Google Scholar] [CrossRef] [PubMed]
- Greenham, L.W.; Greenham, V. Sexing Mouse Pups. Lab. Anim. 1977, 11, 181–184. [Google Scholar] [CrossRef]
- Westphal, N.; Kleene, R.; Lutz, D.; Theis, T.; Schachner, M. Polysialic Acid Enters the Cell Nucleus Attached to a Fragment of the Neural Cell Adhesion Molecule NCAM to Regulate the Circadian Rhythm in Mouse Brain. Mol. Cell. Neurosci. 2016, 74, 114–127. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tschang, M.; Kumar, S.; Young, W.; Schachner, M.; Theis, T. Small Organic Compounds Mimicking the Effector Domain of Myristoylated Alanine-Rich C-Kinase Substrate Stimulate Female-Specific Neurite Outgrowth. Int. J. Mol. Sci. 2023, 24, 14271. https://doi.org/10.3390/ijms241814271
Tschang M, Kumar S, Young W, Schachner M, Theis T. Small Organic Compounds Mimicking the Effector Domain of Myristoylated Alanine-Rich C-Kinase Substrate Stimulate Female-Specific Neurite Outgrowth. International Journal of Molecular Sciences. 2023; 24(18):14271. https://doi.org/10.3390/ijms241814271
Chicago/Turabian StyleTschang, Monica, Suneel Kumar, Wise Young, Melitta Schachner, and Thomas Theis. 2023. "Small Organic Compounds Mimicking the Effector Domain of Myristoylated Alanine-Rich C-Kinase Substrate Stimulate Female-Specific Neurite Outgrowth" International Journal of Molecular Sciences 24, no. 18: 14271. https://doi.org/10.3390/ijms241814271







