Pharmacokinetic Model-Based Control across the Blood–Brain Barrier for Circadian Entrainment
Abstract
:1. Introduction
- To develop two passive and three active transport models that describe drug concentration profiles of a circadian pharmaceutical at the brain level under oral administration;
- To design an MPC-based control framework based on each PK profile for phase resetting.
2. Material and Methods
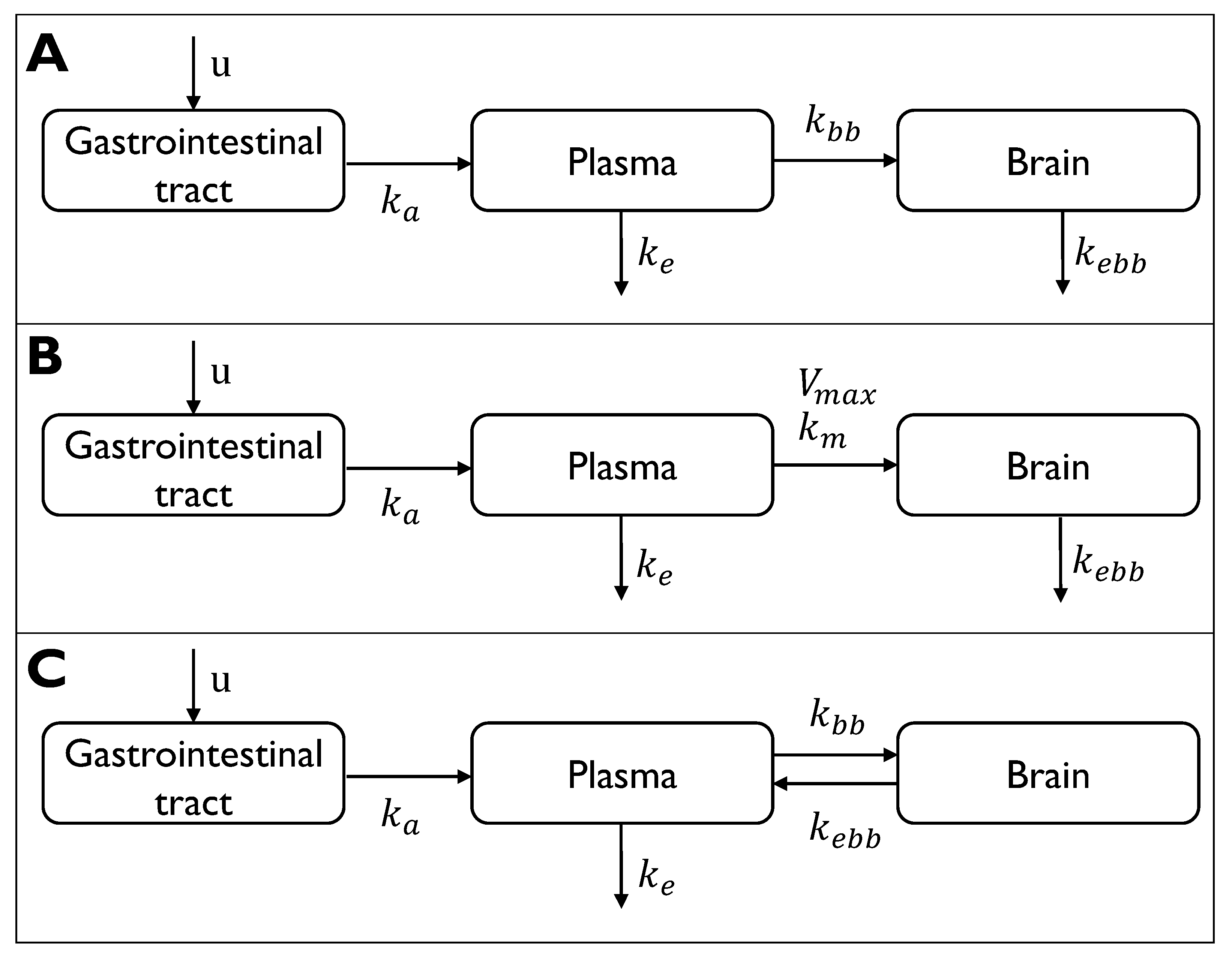
2.1. Pharmacokinetic Models
- Simple passive diffusion: linear distribution from the plasma compartment to the brain compartment (Figure 1A);
- Carrier-mediated transport: saturable transport from the plasma compartment to the brain compartment (Figure 1B);
- Polymeric nanoparticle transcytosis: reversible kinetics between the plasma and the brain compartments (Figure 1C).
2.1.1. Passive Diffusion
2.1.2. Carrier-Mediated Transport
2.1.3. Polymeric Nanoparticle Transcytosis
2.2. Model Parameters Identification
Extended Kalman Filter
2.3. Circadian Oscillator Model
2.4. MPC Formulation
3. Results
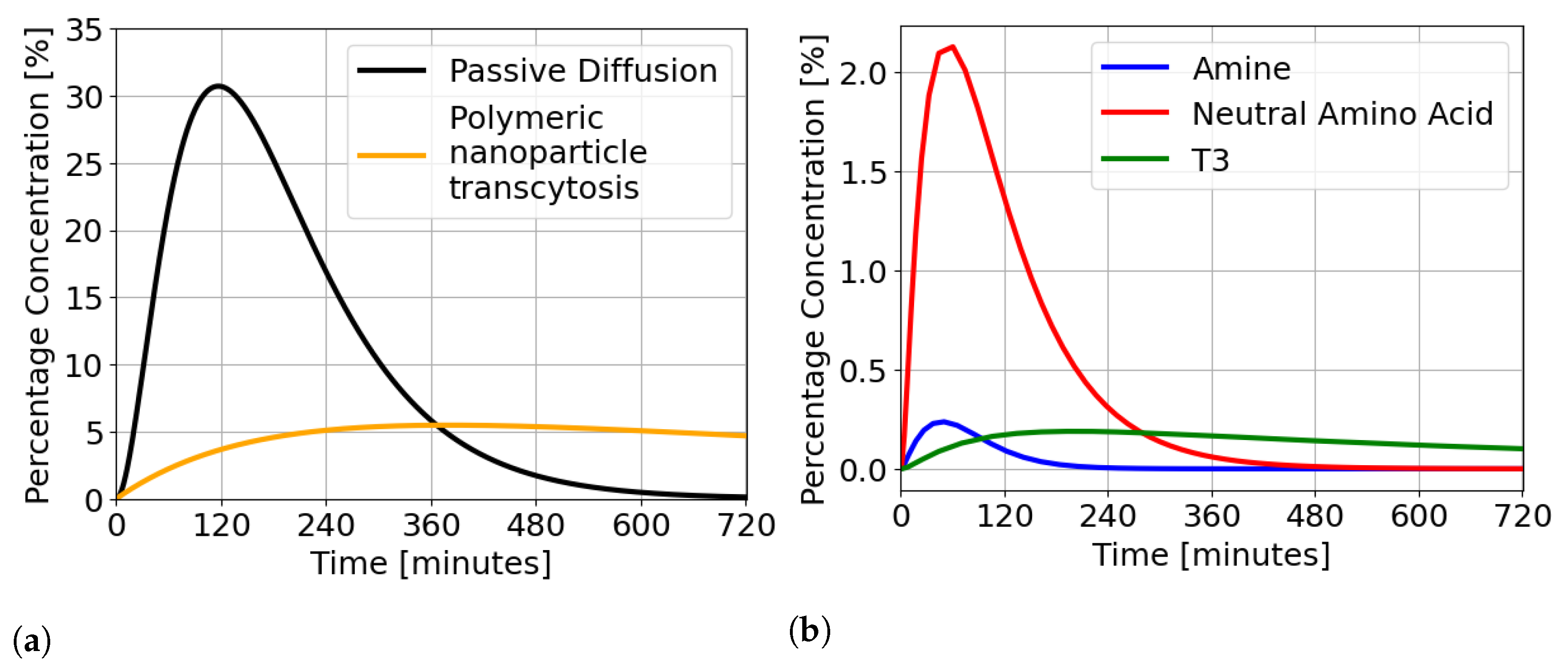
3.1. Pharmacokinetic Models
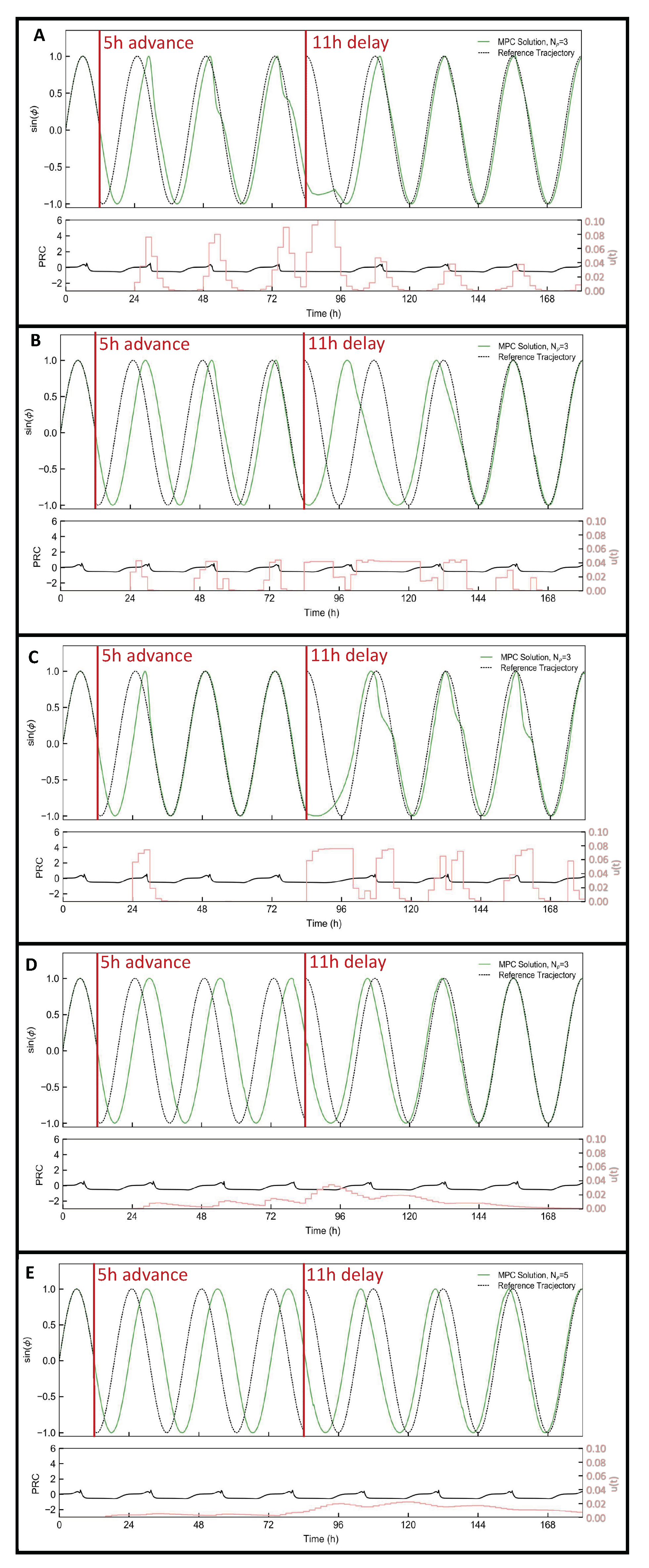
3.2. Controller Performance
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Czeisler, C.A.; Buxton, O.M. The human circadian timing system and sleep-wake regulation. In Principles and Practice of Sleep Medicine, 5th ed.; Elsevier Inc.: Amsterdam, The Netherlands, 2010; pp. 402–419. [Google Scholar]
- Skeldon, A.C.; Phillips, A.J.; Dijk, D.J. The effects of self-selected light-dark cycles and social constraints on human sleep and circadian timing: A modeling approach. Sci. Rep. 2017, 7, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Dunlap, J.C.; Loros, J.J.; DeCoursey, P.J. Chronobiology: Biological Timekeeping; Sinauer Associates: Sunderland, MA, USA, 2004. [Google Scholar]
- Ayyar, V.S.; Sukumaran, S. Circadian rhythms: Influence on physiology, pharmacology, and therapeutic interventions. J. Pharmacokinet. Pharmacodyn. 2021, 48, 321–338. [Google Scholar] [CrossRef] [PubMed]
- Baron, K.G.; Reid, K.J. Circadian misalignment and health. Int. Rev. Psychiatry 2014, 26, 139–154. [Google Scholar] [CrossRef]
- Spoelstra, K.; Albrecht, U.; Van Der Horst, G.T.; Brauer, V.; Daan, S. Phase responses to light pulses in mice lacking functional per or cry genes. J. Biol. Rhythm. 2004, 19, 518–529. [Google Scholar] [CrossRef]
- Ono, D.; Honma, S.; Nakajima, Y.; Kuroda, S.; Enoki, R.; Honma, K.i. Dissociation of Per1 and Bmal1 circadian rhythms in the suprachiasmatic nucleus in parallel with behavioral outputs. Proc. Natl. Acad. Sci. USA 2017, 114, E3699–E3708. [Google Scholar] [CrossRef] [PubMed]
- Biggi, N.; Consonni, D.; Galluzzo, V.; Sogliani, M.; Costa, G. Metabolic syndrome in permanent night workers. Chronobiol. Int. 2008, 25, 443–454. [Google Scholar] [CrossRef]
- Cho, K. Chronic’jet lag’produces temporal lobe atrophy and spatial cognitive deficits. Nat. Neurosci. 2001, 4, 567–568. [Google Scholar] [CrossRef] [PubMed]
- Masri, S.; Sassone-Corsi, P. The emerging link between cancer, metabolism, and circadian rhythms. Nat. Med. 2018, 24, 1795–1803. [Google Scholar] [CrossRef] [PubMed]
- Maury, E.; Ramsey, K.M.; Bass, J. Circadian rhythms and metabolic syndrome: From experimental genetics to human disease. Circ. Res. 2010, 106, 447–462. [Google Scholar] [CrossRef]
- Castanon-Cervantes, O.; Wu, M.; Ehlen, J.C.; Paul, K.; Gamble, K.L.; Johnson, R.L.; Besing, R.C.; Menaker, M.; Gewirtz, A.T.; Davidson, A.J. Dysregulation of inflammatory responses by chronic circadian disruption. J. Immunol. 2010, 185, 5796–5805. [Google Scholar] [CrossRef]
- Julius, A.A.; Yin, J.; Wen, J.T. Time optimal entrainment control for circadian rhythm. PLoS ONE 2019, 14, e0225988. [Google Scholar] [CrossRef] [PubMed]
- Slaby, O.; Sager, S.; Shaik, O.; Kummer, U.; Lebiedz, D. Optimal control of self-organized dynamics in cellular signal transduction. Math. Comput. Model. Dyn. Syst. 2007, 13, 487–502. [Google Scholar] [CrossRef]
- Shaik, O.; Sager, S.; Slaby, O.; Lebiedz, D. Phase tracking and restoration of circadian rhythms by model-based optimal control. IET Syst. Biol. 2008, 2, 16–23. [Google Scholar] [CrossRef] [PubMed]
- Bagheri, N.; Stelling, J.; Doyle, F.J., III. Circadian phase entrainment via nonlinear model predictive control. Int. J. Robust Nonlinear Control 2007, 17, 1555–1571. [Google Scholar] [CrossRef]
- Zhang, J.; Qiao, W.; Wen, J.T.; Julius, A. Light-based circadian rhythm control: Entrainment and optimization. Automatica 2016, 68, 44–55. [Google Scholar] [CrossRef]
- Serkh, K.; Forger, D.B. Optimal schedules of light exposure for rapidly correcting circadian misalignment. PLoS Comput. Biol. 2014, 10, e1003523. [Google Scholar] [CrossRef]
- Bagheri, N.; Stelling, J.; Doyle, F.J., III. Circadian phase resetting via single and multiple control targets. PLoS Comput. Biol. 2008, 4, e1000104. [Google Scholar] [CrossRef]
- Hirota, T.; Lee, J.W.; St. John, P.C.; Sawa, M.; Iwaisako, K.; Noguchi, T.; Pongsawakul, P.Y.; Sonntag, T.; Welsh, D.K.; Brenner, D.A.; et al. Identification of small molecule activators of cryptochrome. Science 2012, 337, 1094–1097. [Google Scholar] [CrossRef]
- Sacre, P.; Sepulchre, R. Sensitivity analysis of oscillator models in the space of phase-response curves: Oscillators as open systems. IEEE Control Syst. Mag. 2014, 34, 50–74. [Google Scholar]
- St John, P.C.; Taylor, S.R.; Abel, J.H.; Doyle, F.J., III. Amplitude metrics for cellular circadian bioluminescence reporters. Biophys. J. 2014, 107, 2712–2722. [Google Scholar] [CrossRef]
- Abel, J.H.; Chakrabarty, A.; Doyle, F.J., III. Nonlinear model predictive control for circadian entrainment using small-molecule pharmaceuticals. Proc. IFAC World Congr. 2017, 50, 9864–9870. [Google Scholar] [CrossRef] [PubMed]
- Abel, J.H.; Chakrabarty, A.; Klerman, E.B.; Doyle, F.J., III. Pharmaceutical-based entrainment of circadian phase via nonlinear model predictive control. Automatica 2019, 100, 336–348. [Google Scholar] [CrossRef] [PubMed]
- St. John, P.C.; Hirota, T.; Kay, S.A.; Doyle, F.J., III. Spatiotemporal separation of PER and CRY posttranslational regulation in the mammalian circadian clock. Proc. Natl. Acad. Sci. USA 2014, 111, 2040–2045. [Google Scholar] [CrossRef] [PubMed]
- Harpsøe, N.G.; Andersen, L.P.H.; Gögenur, I.; Rosenberg, J. Clinical pharmacokinetics of melatonin: A systematic review. Eur. J. Clin. Pharmacol. 2015, 71, 901–909. [Google Scholar] [CrossRef]
- Le Bars, D.; Thivolle, P.; Vitte, P.; Bojkowski, C.; Chazot, G.; Arendt, J.; Frackowiak, R.; Claustrat, B. PET and plasma pharmacokinetic studies after bolus intravenous administration of [11C] melatonin in humans. Int. J. Radiat. Appl. Instrum. 1991, 18, 357–362. [Google Scholar] [CrossRef]
- Van Den Berg, M.P.; Merkus, P.; Romeijn, S.G.; Verhoef, J.C.; Merkus, F.W. Uptake of melatonin into the cerebrospinal fluid after nasal and intravenous delivery: Studies in rats and comparison with a human study. Pharm. Res. 2004, 21, 799–802. [Google Scholar] [CrossRef]
- Senek, M.; Nyholm, D.; Nielsen, E.I. Population pharmacokinetics of levodopa/carbidopa microtablets in healthy subjects and Parkinson’s disease patients. Eur. J. Clin. Pharmacol. 2018, 74, 1299–1307. [Google Scholar] [CrossRef]
- Pardridge, W.M. Transport of nutrients and hormones through the blood-brain barrier. Diabetologia 1981, 20, 246–254. [Google Scholar] [CrossRef]
- DeMuro, R.L.; Nafziger, A.N.; Blask, D.E.; Menhinick, A.M.; Bertino, J.S., Jr. The absolute bioavailability of oral melatonin. J. Clin. Pharmacol. 2000, 40, 781–784. [Google Scholar] [CrossRef]
- Jonklaas, J.; Burman, K.D.; Wang, H.; Latham, K.R. Single dose T3 administration: Kinetics and effects on biochemical and physiologic parameters. Ther. Drug Monit. 2015, 37, 110. [Google Scholar] [CrossRef]
- Böckmann, K.A.; Franz, A.R.; Minarski, M.; Shunova, A.; Maiwald, C.A.; Schwarz, J.; Gross, M.; Poets, C.F.; Bernhard, W. Differential metabolism of choline supplements in adult volunteers. Eur. J. Nutr. 2022, 61, 219–230. [Google Scholar] [CrossRef] [PubMed]
- Nyholm, D.; Lewander, T.; Gomes-Trolin, C.; Bäckström, T.; Panagiotidis, G.; Ehrnebo, M.; Nyström, C.; Aquilonius, S.M. Pharmacokinetics of levodopa/carbidopa microtablets versus levodopa/benserazide and levodopa/carbidopa in healthy volunteers. Clin. Neuropharmacol. 2012, 35, 111–117. [Google Scholar] [CrossRef] [PubMed]
- Khan, A.I.; Lu, Q.; Du, D.; Lin, Y.; Dutta, P. Quantification of kinetic rate constants for transcytosis of polymeric nanoparticle through blood-brain barrier. Biochim. Et Biophys. Acta (BBA)-Gen. Subj. 2018, 1862, 2779–2787. [Google Scholar] [CrossRef] [PubMed]
- Skene, D.; Deacon, S.; Arendt, J. Use of melatonin in circadian rhythm disorders and following phase shifts. Acta Neurobiol. Exp. 1996, 56, 359–362. [Google Scholar]
- Arendt, J.; Skene, D.J.; Middleton, B.; Lockley, S.W.; Deacon, S. Efficacy of melatonin treatment in jet lag, shift work, and blindness. J. Biol. Rhythm. 1997, 12, 604–617. [Google Scholar] [CrossRef]
- Auld, F.; Maschauer, E.L.; Morrison, I.; Skene, D.J.; Riha, R.L. Evidence for the efficacy of melatonin in the treatment of primary adult sleep disorders. Sleep Med. Rev. 2017, 34, 10–22. [Google Scholar] [CrossRef]
- Burgess, H.J.; Sharkey, K.M.; Eastman, C.I. Bright light, dark and melatonin can promote circadian adaptation in night shift workers. Sleep Med. Rev. 2002, 6, 407–420. [Google Scholar] [CrossRef]
- Li, T.; Jiang, S.; Han, M.; Yang, Z.; Lv, J.; Deng, C.; Reiter, R.J.; Yang, Y. Exogenous melatonin as a treatment for secondary sleep disorders: A systematic review and meta-analysis. Front. Neuroendocrinol. 2019, 52, 22–28. [Google Scholar] [CrossRef]
- Sack, R.L.; Lewy, A.J. Melatonin as a chronobiotic: Treatment of circadian desynchrony in night workers and the blind. J. Biol. Rhythm. 1997, 12, 595–603. [Google Scholar] [CrossRef]
- Yu, H.; Dickson, E.; Jung, S.R.; Koh, D.S.; Hille, B. High membrane permeability for melatonin. Biophys. J. 2016, 110, 605a. [Google Scholar] [CrossRef]
- Reiter, R.J.; Tan, D.X.; Kim, S.J.; Cruz, M.H.C. Delivery of pineal melatonin to the brain and SCN: Role of canaliculi, cerebrospinal fluid, tanycytes and Virchow–Robin perivascular spaces. Brain Struct. Funct. 2014, 219, 1873–1887. [Google Scholar] [CrossRef] [PubMed]
- Abbott, N.J.; Patabendige, A.A.; Dolman, D.E.; Yusof, S.R.; Begley, D.J. Structure and function of the blood–brain barrier. Neurobiol. Dis. 2010, 37, 13–25. [Google Scholar] [CrossRef] [PubMed]
- Wong, A.D.; Ye, M.; Levy, A.F.; Rothstein, J.D.; Bergles, D.E.; Searson, P.C. The blood-brain barrier: An engineering perspective. Front. Neuroeng. 2013, 6, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Fu, M.B. Experimental methods and transport models for drug delivery across the blood-brain barrier. Curr. Pharm. Biotechnol. 2012, 13, 1346–1359. [Google Scholar] [CrossRef]
- Pardridge, W.M. The blood-brain barrier: Bottleneck in brain drug development. NeuroRx 2005, 2, 3–14. [Google Scholar] [CrossRef] [PubMed]
- Pardridge, W.M. Drug transport across the blood–brain barrier. J. Bral. Blood Flow Metab. 2012, 32, 1959–1972. [Google Scholar] [CrossRef]
- Chow, B.W.; Gu, C. The molecular constituents of the blood–brain barrier. Trends Neurosci. 2015, 38, 598–608. [Google Scholar] [CrossRef]
- Naik, P.; Cucullo, L. In vitro blood–brain barrier models: Current and perspective technologies. J. Pharm. Sci. 2012, 101, 1337–1354. [Google Scholar] [CrossRef]
- Vendel, E.; Rottschäfer, V.; de Lange, E.C. The need for mathematical modelling of spatial drug distribution within the brain. Fluids Barriers CNS 2019, 16, 1–33. [Google Scholar] [CrossRef]
- Shargel, L.; Andrew, B.; Wu-Pong, S. Applied Biopharmaceutics & Pharmacokinetics; Appleton & Lange: Stamford, CT, USA, 1999; Volume 264. [Google Scholar]
- Rawlings, J.B.; Mayne, D.Q.; Diehl, M. Model predictive control: Theory, Computation, and Design; Nob Hill Publishing: Madison, WI, USA, 2017; Volume 2. [Google Scholar]
- Leloup, J.C.; Goldbeter, A. Toward a detailed computational model for the mammalian circadian clock. Proc. Natl. Acad. Sci. USA 2003, 100, 7051–7056. [Google Scholar] [CrossRef]
- Kronauer, R.E.; Forger, D.B.; Jewett, M.E. Quantifying human circadian pacemaker response to brief, extended, and repeated light stimuli over the phototopic range. J. Biol. Rhythm. 1999, 14, 501–516. [Google Scholar] [CrossRef]
- Taylor, S.R.; Doyle, F.J., III; Petzold, L.R. Oscillator model reduction preserving the phase response: Application to the circadian clock. Biophys. J. 2008, 95, 1658–1673. [Google Scholar] [CrossRef]
- Johns, J. Estimation of melatonin blood brain barrier permeability. J. Bioanal. Biomed. 2011, 3, 64–69. [Google Scholar] [CrossRef]
- Hammarlund-Udenaes, M.; Friden, M.; Syvnen, M.; Gupta, A. On the rate and extent of drug delivery to the brain. Pharm. Res. 2008, 25, 1737–1750. [Google Scholar] [CrossRef] [PubMed]
- LeGates, T.A.; Fernandez, D.C.; Hattar, S. Light as a central modulator of circadian rhythms, sleep and affect. Nat. Rev. Neurosci. 2014, 15, 443–454. [Google Scholar] [CrossRef]
- Brown, L.S. A Systems Biology Approach to the Modeling and Control of Circadian Rhythms. Ph.D. Thesis, Harvard University, Cambridge, MA, USA, 2021. [Google Scholar]
| Transport System | F [%] | [min] | [min] | [min] | [min] | [mL] | [mL] | [mg/min] | [mg/mL] |
|---|---|---|---|---|---|---|---|---|---|
| Passive diffusion [26,27,28] | 0.14 | 0.11 | 0.11 | 4.53 | 2.82 | 1.80 | 3.30 | - | - |
| Carrier-mediated transport (Amine) [29,30] | 0.80 | 2.90 | 0.10 | - | 3.38 | 3.00 | 0.90 | 10.00 | 0.44 |
| Carrier-mediated transport (Neutral Amino Acid) [29,30] | 0.30 | 1.40 | 1.30 | - | 2.82 | 1.00 | 0.30 | 30.00 | 0.12 |
| Carrier-mediated transport (T3) [29,30] | 0.75 | 4.00 | 0.02 | - | 1.30 | 1.50 | 3.00 | 0.10 | 1.00 |
| Polymeric nanoparticle transcytosis [35] | 0.14 | 0.11 | 0.11 | 6.10 | 8.30 | 1.80 | 3.30 | - | - |
| Reference | Sample Size | Trasport System (Substrate) | Dose [mg] | [mg/mL/min] | [mg/mL/min] | [mg/mL] | [mg/mL] |
|---|---|---|---|---|---|---|---|
| De Muro et al. [31] | 12 | Passive diffusion | 2.00 | 2.37 | 2.31 | 3.40 | 4.67 |
| Jonklass et al. [32] | 12 | T3 (Triiodothyronine) | 0.05 | 1.12 | 1.94 | 3.46 | 3.81 |
| Bockmann et al. [33] | 6 | Amine (Choline) | 550.00 | 0.40 | 0.30 | 1.04 | 2.30 |
| Nyholm et al. [34] | 19 | Neutral amino acid (L-Dopa) | 100.00 | 8.48 | 0.12 | 9.46 | 1.13 |
| Transport System | % Administered Dose | [min] |
|---|---|---|
| Passive diffusion | 30.70 | 117 |
| Carrier-mediated transport (Amine) | 0.23 | 50 |
| Carrier-mediated transport (Neutral Amino Acid) | 2.12 | 61 |
| Carrier-mediated transport (T3) | 0.18 | 193 |
| Polymeric nanoparticle transcytosis | 5.48 | 380 |
| Transport System | [mg] | Time Required for 5-h Phase Advance [h] | Time Required for 11-h Phase Delay [h] | |
|---|---|---|---|---|
| Passive diffusion | 2.00 | 1.00 | 60 | 66 |
| Carrier-mediated transport (Amine) | 16.50 | 1.00 | 50 | 54 |
| Carrier-mediated transport (Neutral Amino Acid) | 3.00 | 0.10 | 30 | 48 |
| Carrier-mediated transport (T3) | 4.30 | 0.10 | N/A | 72 |
| Polymeric nanoparticle transcytosis | 0.06 | 1.00 | N/A | 84 |
| Baseline [24] | 0.06 | 1.00 | 30 | 36 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Murdoch, S.Ó.; Aiello, E.M.; Doyle, F.J., III. Pharmacokinetic Model-Based Control across the Blood–Brain Barrier for Circadian Entrainment. Int. J. Mol. Sci. 2023, 24, 14830. https://doi.org/10.3390/ijms241914830
Murdoch SÓ, Aiello EM, Doyle FJ III. Pharmacokinetic Model-Based Control across the Blood–Brain Barrier for Circadian Entrainment. International Journal of Molecular Sciences. 2023; 24(19):14830. https://doi.org/10.3390/ijms241914830
Chicago/Turabian StyleMurdoch, Síofra Ó., Eleonora M. Aiello, and Francis J. Doyle, III. 2023. "Pharmacokinetic Model-Based Control across the Blood–Brain Barrier for Circadian Entrainment" International Journal of Molecular Sciences 24, no. 19: 14830. https://doi.org/10.3390/ijms241914830





