1. Introduction
The disruption of the epithelial barrier function (EBF) with increased intestinal permeability is a common finding in several gastrointestinal disorders, including inflammatory bowel disease (IBD) and irritable bowel syndrome (IBS) [
1,
2,
3,
4]. The intestinal epithelium represents a functional barrier between the lumen and the internal milieu, allowing selective exchange processes (secretory and absorptive) and acting at the same time as a protective barrier against luminal antigens [
5]. Gut commensal microbiota, considered for long time to have been a passive component of the gastrointestinal tract, is now considered as a dynamic player in intestinal homeostasis [
6,
7,
8,
9]. Gut microbiota and microbial-derived products act as luminal factors that interact continuously with the host. In physiological conditions, these interactions contribute to the maintenance of intestinal homeostasis. However, in states of dysbiosis, host–microbial interactions change and might lead to the activation of the immune system and the generation of a persistent inflammatory state with functional changes, including alterations in the EBF [
10]. In this sense, dysbiosis and altered epithelial permeability are co-existent features of IBS and IBD [
1,
3,
11,
12,
13,
14,
15].
Host–microbial interactions are largely mediated through pattern recognition receptors (PRRs). PRRs are expressed within the gut and recognize conserved microbial components named pathogen-associated molecular patterns (PAMPs) [
16]. In particular, recognition of gut microbiota depends largely on the interaction with a subgroup of PRRs named Toll-like receptors (TLRs), with up to 13 members described in mammals (TLR1–13) [
17,
18]. The relative expression of the different TLRs differs depending upon the cell type and the cellular localization [
18,
19]. Within the gut, some of the predominant TLRs are TLR2, TLR3, TLR4, TLR5 and TLR7, being present in several cell types, including the epithelial cells [
17]. TLRs directly interact with luminal microbial components and, upon activation, elicit neuroimmune responses that affect intestinal functions, including the EBF [
20,
21,
22,
23,
24,
25]. For instance, we have shown that during antibiotic-induced dysbiosis, host–bacterial interactions are altered, including changes in the expression of TLRs, and there are local changes in immune- and sensory-related systems within the gut [
26,
27]. Similar alterations might be elicited when TLRs are over-stimulated, mimicking a state of dysbiosis with altered host–bacterial interactions. In this sense, we have shown that direct stimulation of colonic TLR7 with the selective agonist imiquimod leads to immune- and sensory-related changes similar to those observed during intestinal dysbiosis [
28,
29]. Moreover, imiquimod reduced the serum levels of inflammatory cytokines and ameliorated clinical signs and intestinal lesions during DSS-induced colitis in mice [
30].
Taking these observations into account, the aim of the present work was to directly assess the effects of colonic TLR7 stimulation on the EBF in rats. For this, we assessed the effects of the over-stimulation of colonic TLR7 with the selective agonist imiquimod on epithelial electrical parameters and on epithelial permeability to macromolecules in in vitro (the Ussing chamber system) and in vivo conditions. In order to characterize the underlying mechanisms mediating potential TLR7-dependent modulation of the EBF, we also assessed changes in the expression and epithelial distribution of the main tight-junction (TJ)-related proteins (occludin, tricellulin, claudin-2, claudin-3, junctional adhesion molecule 1 -JAM-1- and zonula occludens-1 -ZO-1-). Moreover, changes in the expression of the barrier-enhancer factor glucagon-like peptide 2 (GLP-2, assessed through the expression of its precursor, proglucagon) [
31] were also assessed. Finally, the induction of a colonic inflammatory-like state associated with the over-stimulation of TLR7 was assessed at the macroscopical and molecular levels.
3. Discussion
In this study we show that the local, colonic, over-stimulation of TLR7 with the selective agonist imiquimod modulates the colonic EBF in rats. In vivo over-stimulation of TLR7 led to an enhancement of colonic epithelial barrier function, while acute, in vitro, stimulation was without effect. In order to verify these TLR7-dependent pro-barrier effects, over-stimulation of TLR7 was combined with the intracolonic administration of DMSO as a stimulus to alter epithelial permeability. Under these conditions, the opposite effect was observed, with an increase in epithelial permeability upon TLR7 stimulation. The effects of TLR7 on the EBF seem to be independent of the modulation of the expression or the distribution of the main tight-junction-related proteins or the modulation of the expression levels of the barrier-enhancer factor GLP-2.
Alterations of the colonic EBF are a component of several gastrointestinal diseases and are a common finding in IBD and IBS [
1,
2,
3,
4,
5]. Although the pathophysiological mechanisms underlying these alterations are not completely understood, compelling evidence suggests the presence of a sustained enhancement of epithelial paracellular permeability facilitating the exposure to luminal antigens, triggering innate mucosal immune system responses, thus perpetuating a state of persistent, and abnormal, immune activation leading to the development of intestinal inflammation. Several studies suggest that gut-commensal microbiota and host–microbial interactions might be important pathogenic factors in this process [
6,
10,
11,
33,
34,
35,
36,
37,
38,
39]. Indeed, epithelial interactions with the microbiota and microbial-derived products, as well as the increased entrance within the intestinal wall, are altered in states of dysbiosis, which are likely contributing to the process of immune activation [
3,
24,
40,
41]. Nevertheless, the microbial contribution and the exact mechanisms involved are still unclear. In this context, in the present report, we explored the possibility that TLR7-dependent host–bacterial interactions might be part of the mechanisms through which the microbiota modulates epithelial barrier function. For this, we simulated a state of dysbiosis with over-stimulation of TLR7-dependent host–bacterial interactions through direct stimulation of TLR7 with a selective agonist, imiquimod, administered locally (intracolonically). We have previously used this approach to study TLR-dependent host–bacterial interactions in rats and mice [
28,
29].
The results obtained show that the local (colonic) over-stimulation of TLR7 with imiquimod leads to a reduction in the epithelial conductance and paracellular permeability to macromolecules in in vitro conditions (Ussing chamber), thus indicating an improvement of the EBF. These observations suggest that the over-stimulation of TLR7 associated with a dysbiotic state might have a defensive function, increasing the tightness of the epithelium and, therefore, preventing an increased passage of luminal, microbial-related antigens during dysbiosis. This protective role of TLR7 is in line with that observed in mice during DSS-induced colitis, where epithelial lesions, clinical signs and serum levels of pro-inflammatory markers were ameliorated after TLR7 stimulation with imiquimod [
30]. A similar protective role has been postulated for other TLRs, mainly TLR2. Indeed, data obtained in murine models of dysbiosis indicate that TLR2 is a receptor that, upon activation by luminal microbial products, enhances the EBF as a protective response to dysbiosis, preventing the excessive passage of luminal antigens and an aberrant activation of the local immune system [
24,
42,
43]. Altogether, these observations suggest that several TLRs, including at least TLR2 and TLR7, might share a similar protective role during states of over-stimulation (as potentially occurring during dysbiosis). Considering that that several TLRs have been largely associated with the activation of intestinal immune responses and the induction of inflammation and secretomotor alterations [
23,
44,
45], these observations support a dual role for TLRs, mediating both pathogenic and protective responses in a TLR-specific manner. The balance between protective and damaging signals will lead to a final state in which homeostasis is maintained or functional alterations appear.
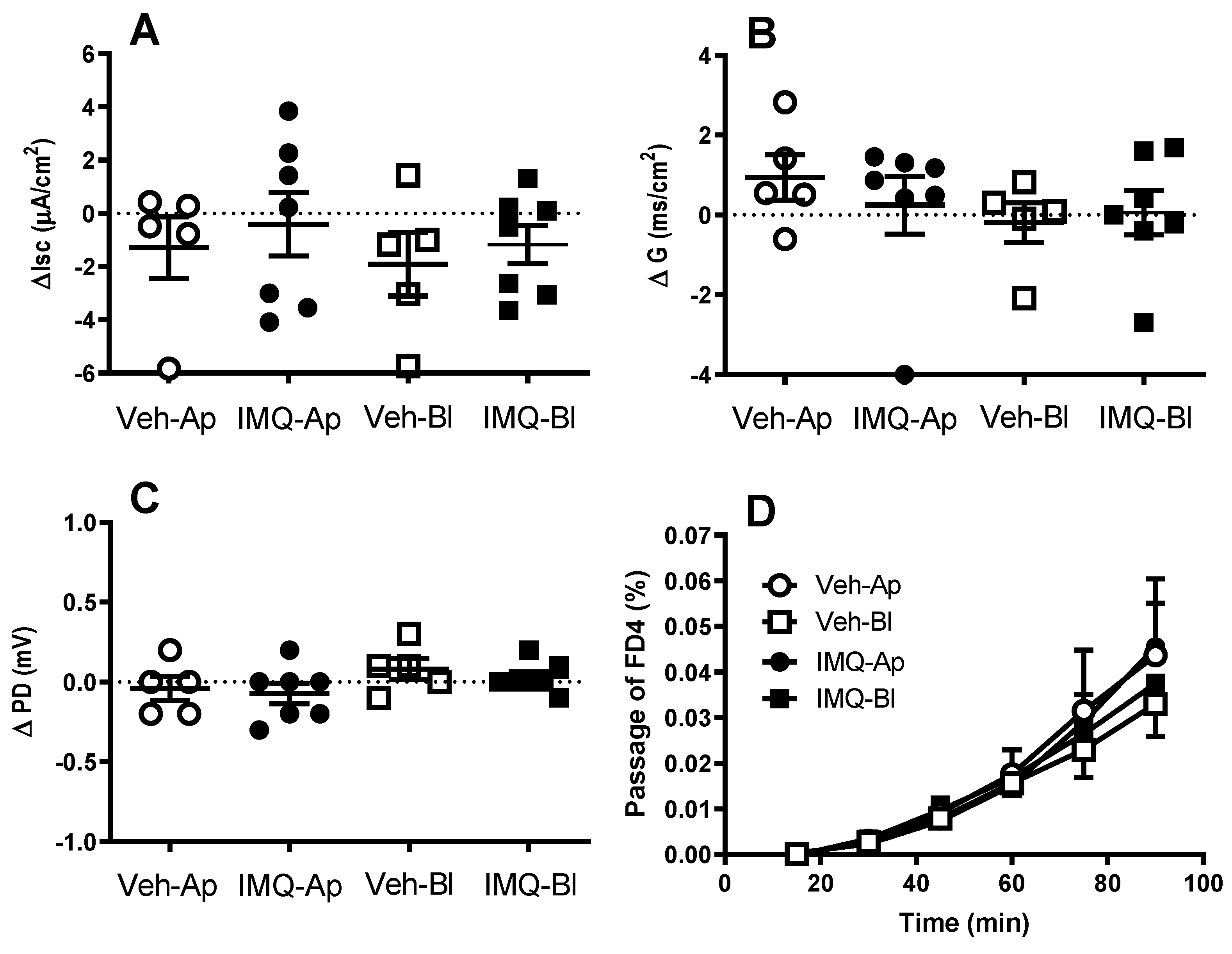
Interestingly, the pro-barrier effects of TLR7 were not observed when the receptor was acutely stimulated in vitro (the direct addition of imiquimod in the Ussing chamber). This apparent discrepancy vs. the observation after the in vivo stimulation might suggest that the activation of TLR7 triggers a signaling cascade that requires a relatively long time-frame to generate functional changes. In this respect, the technical limitations of the technique used, since the viability of the mucosal sheets in the Ussing chamber is limited, might not allow the development of the full response observed after the in vivo stimulation. In addition, the recruitment of extra-intestinal neuroendocrine mechanisms, not preserved in in vitro conditions, might be necessary to elicit the full effects associated with TLR7 stimulation, which consequently, cannot be manifested during the acute in vitro stimulation.
Considering the potential protective role of TLR7, we hypothesized that its effects would manifest in states of barrier alteration in which the epithelial entrance of luminal factors might be facilitated. In these conditions, a more effective activation of TLR-dependent signaling mechanisms, including TLR7-mediated pro-barrier responses, might occur. To test this hypothesis, we assessed the effects of TLR7 over-stimulation during the simulation of a state of favored epithelial permeability, such as during DMSO exposure. In this sense, DMSO is an aprotic solvent that permeabilizes the cell membrane acting as a non-selective penetration enhancer [
46]. In the presence of DMSO, we should expect an increased entrance of luminally-administered imiquimod and, therefore, an enhanced stimulation of intracellular TLR7 and the consequent activation of barrier protective mechanisms. However, against this hypothesis, the combined imiquimod—DMSO resulted in a deterioration of the EBF, with an increased passage of luminal macromolecules to blood and urine, as assessed in vivo. These effects are likely to be TLR7-dependent and not secondary to the permeabilization of the epithelium, since DMSO,
per se, did not increase epithelial permeability to macromolecules. Currently, we cannot explain the differences observed between the stimulation of TLR7 by imiquimod alone or with the combination imiquimod–DMSO (a TLR7-mediated enhancement of the barrier vs. a TLR7-mediated worsening of the barrier, respectively). We can speculate that after DMSO administration the passage of luminal factors leads to the simultaneous activation of multiple mechanisms, including barrier protective and damaging pathways, with a final outcome depending upon the balance between both effects.
In any case, the results obtained suggest that the TLR7-mediated effects on the EBF are mainly associated with a modulation of the permeability to macromolecules, which predominantly occurs through paracellular pathways. Since paracellular permeability to macromolecules depends largely on the TJ organized among the epithelial cells we also assessed if TLR7 over-stimulation could modify the expression of the main TJ-related proteins (occludin, tricellulin, claudin-2, claudin-3, JAM-1 and ZO-1). Overall, no consistent change in the expression of these proteins was observed following imiquimod administration, either alone or combined with DMSO. In spite of these negative findings, we cannot exclude conformational changes of the already available proteins that could lead to a reorganization of the TJ at a molecular level, thus explaining our functional findings. For instance, Cario et al. showed that the stimulation of TLR2 enhances the transepithelial resistance of the colonic epithelial barrier through the apical redistribution of ZO-1 [
43]. Therefore, similar mechanisms cannot be discarded for TLR7. However, when assessing the epithelial distribution of ZO-1, claudin-2 and claudin-3 using immunohistochemistry, no consistent treatment-related change in the distribution or intensity of staining was observed during the stimulation TLR7 (with or without DMSO). Overall, these observations indicate that the TLR7-dependent modulation of the colonic EBF is independent of the changes in TJ-related proteins, at least as it relates to occludin, tricellulin, claudin-2, claudin-3, JAM-1 and ZO-1. Nevertheless, extended ultrastructural studies addressing the organization of TJ are necessary to further address the potential implication of TJ in the TLR7-mediated modulation of the EBF.
GLP-2 has been postulated as a barrier-enhancer factor. Indeed, a positive correlation between intestinal proglucagon levels, the precursor of GLP-2, and the expression of the TJ-related proteins (ZO-1 and occludin) has been observed [
31]. Similarly, a reduction in proglucagon expression has been described in states of increased epithelial permeability associated with the down-regulation of TJ-related proteins [
12]. Despite this evidence, in our studies, no change in proglucagon gene expression was detected upon the activation of TLR7, regardless of the presence of changes in the EBF. This suggests that GLP-2 is not implicated in the barrier modulatory effect of TLR7.
In previous studies, we observed that intracolonic imiquimod had minor effects on immune activation in rats, with a moderate up-regulation of pro-inflammatory cytokines, observed only after repeated treatment [
28,
29]. In agreement with these observations, a single treatment with imiquimod resulted in neither the up-regulation of pro-inflammatory cytokines nor the induction of inflammatory like-changes within the colon, at either the macroscopical or microscopical levels. This indicates that the TLR7-mediated modulation of the EBF is not secondary to a local immune activation within the colon. These observations contrast with previous data showing that the TLR7/8 agonist resiquimod elicited a colitic-like state upon intracolonic administration in mice [
47]. However, this effect is probably associated with the stimulation of TLR8 [
48].
4. Materials and Methods
4.1. Chemicals
Imiquimod [R-837, 1-(2-Methylpropyl)-1H-imidazole [4,5-c]quinoline-4-amine]; Enzo Life Sciences, Farmingdale, NY, USA] was dissolved in 0.5% hydroxypropylmethyl cellulose (Sigma-Aldrich, Saint Louis, MO, USA). Carbachol (CCh, Sigma-Aldrich) and tetrodotoxin (TTX, Latoxan, Valence, France) were dissolved in distilled water as stock solutions of 10−1 M (CCh) and 10−4 M (TTX), respectively, and further dilutions were performed in distilled water. All stock solutions were aliquoted and stored at −80 °C until their use. Dimethyl sulfoxide (DMSO) was obtained from Panreac (Barcelona, Spain). Fluorescein isothiocyanate (FITC)-labeled dextran (FD) with a mean molecular weight of 4 kDa (FD4; TdB Consultancy AB. Uppsala, Sweden) was stored at 5 °C and was dissolved with Krebs solution (20 mg/mL) at the time of use.
4.2. Animals
Adult male Sprague–Dawley rats (6-week-old at arrival; 250–280 g; Charles-River Laboratories, Lyon, France) were used. A total of 109 animals were used for all the experiments described in this report, including the pilot studies and animals that were excluded because of technical problems (approximately 10%). On arrival, the animals were housed in pairs in standard plastic cages under conventional controlled environmental conditions (20–22 °C, 40–70% humidity and 12 h light/dark cycle) and fed with a standard pellet diet (Panlab SL, Barcelona, Spain) and tap water ad libitum. All experimental procedures were approved by the ethics committees of the Universitat Autònoma de Barcelona and the Generalitat de Catalunya (protocols 1420 and 6333, respectively).
4.3. Tissue Sampling
At the time of the experiments, animals were euthanized by decapitation, except when otherwise stated. A laparotomy was performed and the colon gently dissected and placed in ice-cold oxygenated Krebs buffer [(in mM) 115.48 NaCl, 21.90 NaHCO3; 4.61 KCl; 1.14 NaH2PO4; 2.50 CaCl2; 1.16 MgSO4 (pH: 7.3–7.4)] containing 10 mM glucose. Colonic segments were used to perform in vitro epithelial barrier function studies (mid colon) or preserved for morphological or molecular biology studies (mid-distal colon). Samples for histological and immunostaining studies were fixed in 4% paraformaldehyde in a phosphate buffer for 24 h. Thereafter, fixed samples were processed routinely for paraffin embedding and 5 µm sections were obtained for hematoxylin and eosin (H&E) staining or immunohistochemistry. Samples for molecular biology studies were immediately frozen in liquid nitrogen and stored at −80 °C until processed.
4.4. Measurement of Electrophysiological Parameters (Ussing Chambers)
Colonic segments were stripped of the outer muscle layers and myenteric plexus, opened along the mesenteric border, and divided into 1 cm2 flat segments, approximately. Epithelial sheets were mounted in Ussing chambers (World Precision Instruments, Aston, UK) with an exposed window surface area of 0.67 cm2. The tissues were bathed bilaterally with 5 mL of oxygenated and warmed (37 °C) Krebs buffer (in mmol/L: 115.48 NaCl, 21.90 NaHCO3, 4.61 KCl, 1.14 NaH2PO4, 2.50 CaCl2 and 1.16 MgSO4; pH: 7.3–7.4). The buffer also contained 10 mmol/L glucose as an energy source. The electrical equipment of the Ussing chambers consisted of two voltage-sensitive electrodes (EKV; World Precision Instruments) to monitor the potential difference (PD) across the tissue, and two Ag–AgCl current-passing electrodes (EKC; World Precision Instruments) to inject the required short-circuit current (Isc) to maintain a zero potential difference, as registered via an automated voltage/current clamp (DVC-1000; World Precision Instruments). Ohm’s law was used to calculate the tissue conductance (G), using the change in Isc when a voltage step of 1 mV was applied at 5 min intervals. The tissues were allowed to stabilize for 15–25 min before the baseline values for PD, Isc and G were recorded. The data were digitized with an analog-to-digital converter (MP150; Biopac Systems, Goleta, CA, USA) and measurements were recorded and analyzed with Acqknowledge computer software (version 3.8.1; Biopac Systems). The short circuit current and G were normalized for the mucosal surface area.
4.5. Epithelial Paracellular Permeability In Vitro
Paracellular permeability was evaluated following protocols previously described by us [
12]. The mucosal-to-basolateral flux of fluorescein isothiocyanate (FITC)-labeled dextran (FD) with a mean molecular weight of 4 kDa was assessed in colonic epithelial sheets mounted in Ussing chambers. After stabilization (20–30 min), baseline electrophysiological parameters were assessed and FD4 were added to the mucosal reservoir to a final concentration of 2.5 × 10
−4 M. Basolateral samples (250 µL, replaced by 250 µL of buffer solution with glucose) were taken at 15-min intervals during the following 60 min for the measurement of FD4. The concentration of fluorescein in the samples was determined by fluorometry (Infinite F200; Tecan, Crailsheim, Germany) with an excitation wavelength of 485 nm (20 nm band width) and an emission wavelength of 535 nm (25 nm band width), against a standard curve. Readings are expressed as percentage (%) of the total amount of FD4 added to the mucosal reservoir.
4.6. The In Vivo Colonic Permeability
To assess the colonic permeability in in vivo conditions, the passage of intracolonically administered FD4 to blood and urine, as well as the accumulation of FD4 in the colonic wall, was determined. For this, the animals were anesthetized with isoflurane (Isoflo®) and FD4 was administered intracolonically (rectal enema, 10 mg/animal, 0.2 mL) with a plastic cannula (8 cm from the anus, corresponding to the mid colon). FD4 administration was performed slowly (30 s–1 min) to avoid any reflux. Thereafter, the animals were returned to their home cages and 30 min later were deeply anesthetized with isoflurane and blood collected via intracardiac puncture. Subsequently, the animals were euthanized by a thoracotomy, and urine (intravesically) and colon samples were collected. The fluorescence was determined in urine, serum (obtained from blood centrifuged at 3000× g for 10 min, 4 °C) and colon homogenates as described above. When assessing the fluorescence in colonic samples, the tissues were washed thoroughly with a saline solution to eliminate any luminal remains of FD4.
4.7. Experimental Protocols
4.7.1. The Effects of Imiquimod on Epithelial Electrical Parameters
The animals were deeply anesthetized with isoflurane (Isoflo®, Esteve Veterinaria, Barcelona, Spain) and treated with intracolonic imiquimod (rectal enema, 100 or 300 µg/rat) or vehicle (hydroxypropylmethyl cellulose; 5 mg/mL; 0.2 mL/rat). Five hours later, the animals were euthanized and colonic sheets were obtained and mounted in Ussing chambers as described above. After a 20 min stabilization period, the electrical parameters (Isc, PD and G) were assessed for an additional 60 min period.
In some cases, the effects of the acute exposure to imiquimod (as a direct addition to the Ussing chambers) on the electrical parameters were assessed in the colonic sheets obtained from a naïve animal. In this case, after a 20–30 min stabilization period the basal electrical parameters were recorded for a 10 min period, and thereafter, imiquimod (300 µg) was added to either the apical or the basolateral side and the changes in electrical parameters were recorded for an additional period of 60 min. The addition of the vehicle in some tissues served as a control.
At the end of the experiments, a single concentration of CCh (100 µM) was added to the basolateral side of the chamber in order to assess the viability of the tissues.
4.7.2. The Effects of Imiquimod on Epithelial Permeability In Vitro
The isoflurane-anesthetized animals were treated with intracolonic imiquimod (100 or 300 µg/rat) or the vehicle (hydroxypropylmethyl cellulose; 5 mg/mL; 0.2 mL/rat). Five hours later, the animals were euthanized and the colonic sheets were obtained and mounted in Ussing chambers as described above. After a 20–30 min stabilization period, baseline electrophysiological parameters were assessed and FD4 was added to the mucosal reservoir (2.5 × 10−4 M) and the passage of the marker evaluated, as described above, for the following 60 min.
4.7.3. The Effects of Imiquimod on Epithelial Permeability In Vivo
The rats were anesthetized with isoflurane and treated intracolonically with imiquimod (300 µg/rat) or its vehicle (0.2 mL) and returned to their home cages. Six hours later, the animals were anesthetized again and FD4 (10 mg/rat, 0.2 mL) was administered intracolonically and permeability tested 30 min later, as described above.
In some cases, the imiquimod treatment was combined with a permeabilization of the colonic epithelium. In these cases, 4 h after the imiquimod administration, animals were anesthetized with isoflurane and the colonic epithelium was challenged with dimethyl sulfoxide [
46] (DMSO, 100%, 0.2 mL) and 2 h later FD4 was administered intracolonically to assess permeability.
In all cases, the animals received a single intracolonic treatment. For this, the animals were deeply anesthetized with isoflurane (Isoflo®, Esteve Veterinaria, Barcelona, Spain) and treatments were applied intracolonically with a plastic cannula (8 cm from the anus, corresponding to the mid-distal colon). Thereafter, the animals were returned to their home cages and 3 h (gene expression), 5 h (Ussing chamber studies) or 6.5 h later (in vivo permeability) were euthanized and tissue samples were obtained as described above.
4.8. The Quantitative Real Time Reverse Transcription Polymerase Chain Reaction (RT-qPCR)
The total RNA was extracted from colonic tissue samples using Ribopure RNA Isolation Kit (Applied Biosystems, CA, USA) and quantified with a Nanodrop (ND-100 spectrophotometer, Nanodrop Technologies, Rockland, DE, USA). For cDNA synthesis, 1 µg of RNA was reverse-transcribed in a 20 µLl reaction volume using a high capacity cDNA reverse transcription kit (Applied Biosystems, Foster City, CA, USA). TaqMan gene expression assays for occludin (Rn00580064_m1), tricellulin (Rn01494284_m1), claudin-2 (Rn02063575_s1), claudin-3 (Rn00581751_s1), JAM-1 (Rn00587389_m1), ZO-1 (Rn02116071_s1), IL-6 (Rn01410330_m1), IFNα (Rn02395770_g1), TLR 7 (Rn01771083_s1) and proglucagon (Rn00562293_m1) were used (all from Applied Biosystems). Actin-β (Rn00667869_m1) was used as an endogenous reference gene. The PCR reaction mixture was incubated on a 7500 Fast Real Time PCR system (Applied Biosystems). All samples, as well as the negative controls, were assayed in triplicates. The cycle threshold for each sample was obtained and, thereafter, all data were analyzed with the comparative 2
−∆∆CT method, with the control group serving as the calibrator [
49].
4.9. Immunohistochemistry for Tight-Junction-Related Proteins
Paraffin embedded tissue sections (5 µm thick) were deparaffinized and rehydrated with a battery gradient of alcohols. The antigen retrieval for claudin-2 and claudin-3 was achieved by processing the slides in a microwave (2 cycles of 5 min, 800 W) in a 10 mM Tris Base, 1 mM EDTA solution (pH 9). The epitope retrieval for ZO-1 was performed using a pressure cooker (at full pressure for 6 min) in 10 mM citrate buffer (pH 6). Thereafter, samples were incubated for 40 min in H2O2 (5% in distilled water) for inhibiting endogenous peroxidases and with the reagents of the Avidin/Biotin Blocking Kit (SP-2001; Vector Laboratories, Burlingame, CA, USA) for inhibiting the endogenous avidin and biotin. Finally, a 1 h incubation at room temperature with horse, rabbit or goat serum, as appropriate, was performed for blocking unspecific unions before incubating the slides (overnight at 4 °C) with their respective primary antibodies: mouse monoclonal anti-Claudin-2 antibody (1:2000; ref.: 32–5600. Invitrogen, Camarillo, CA, USA), goat polyclonal anti-Claudin-3 antibody (1:500; ref.: SC-17662. Santa Cruz, Dallas, TX, USA), rabbit polyclonal anti-Occludin (1:1000; ref.: SC-5562. Santa Cruz) and rabbit polyclonal anti-ZO-1 (1:500; ref.: SC-10804. Santa Cruz). The following day, sections were incubated with the respective secondary antibody for 1 h at room temperature: biotinylated horse anti-mouse IgG (1:200, ref.: BA-2000, Vector Laboratories), biotinylated rabbit anti-goat IgG (1:200, ref.: SC2774. Santa Cruz) or Biotin-XX Goat Anti-Rabbit IgG (H + L) (1:200, ref.: B2770. Invitrogen). In all cases, an avidin/peroxidase kit (Vectastain ABC kit; Vector Laboratories, Burlingame, CA, USA) was used for detection, and the antigen–antibody complexes were revealed using 3,3′-diaminobenzi-dine (SK-4100 DAB; Vector Laboratories). The slides were counterstained with toluidine blue or with hematoxylin. In all cases, the specificity of the staining was confirmed by omitting the primary antibody.
For the claudin-2 and ZO-1 immunohistochemistry results, 10–20 representative microphotographs (×200) were taken per animal with a Nikon Eclipse 90i microscope. The intensity of the staining was measured using the software Image J (National Institute of Health, Bethesda, MD, USA) and a mean value obtained. Claudin-3 immunoreactivity at the epithelial surface was quantified by applying a semi-quantitative score (0: no immunoreactivity; 1: scarce, low intensity, granules in some epithelial cells; 2: clear, but not-organized granules in abundant epithelial cells; 3: clear and well-organized granules, drawing a chain in the zone corresponding to the epithelial TJs, in most cells). In this case, for quantification, 15 randomly selected fields covering the whole thickness of the jejunal mucosa, from at least two tissue sections for each animal, were scored by two independent observers. A final score (0–3) for each animal was calculated as the mean of the scores assigned by each observer. All procedures were performed on coded slides to avoid any bias.
4.10. Statistical Analysis
All data are expressed as a mean ± SEM. A robust analysis (one interaction) was used to obtain the mean ± SEM for the RT-qPCR data. Comparisons between two groups were performed using Student’s unpaired t test. Comparisons between multiple groups were performed using a one-way or a two-way ANOVA, as appropriate; followed, when necessary, by a Newman–Keuls’ multiple comparisons test. In all cases, the results were considered statistically significant when p < 0.05. A GraphPad Prism 4 (GraphPad Software, La Jolla, CA, USA) was used to perform all statistical analyses.