Pituitary Adenylate Cyclase-Activating Polypeptide (PACAP) and Sudden Infant Death Syndrome: A Potential Model for Investigation
Abstract
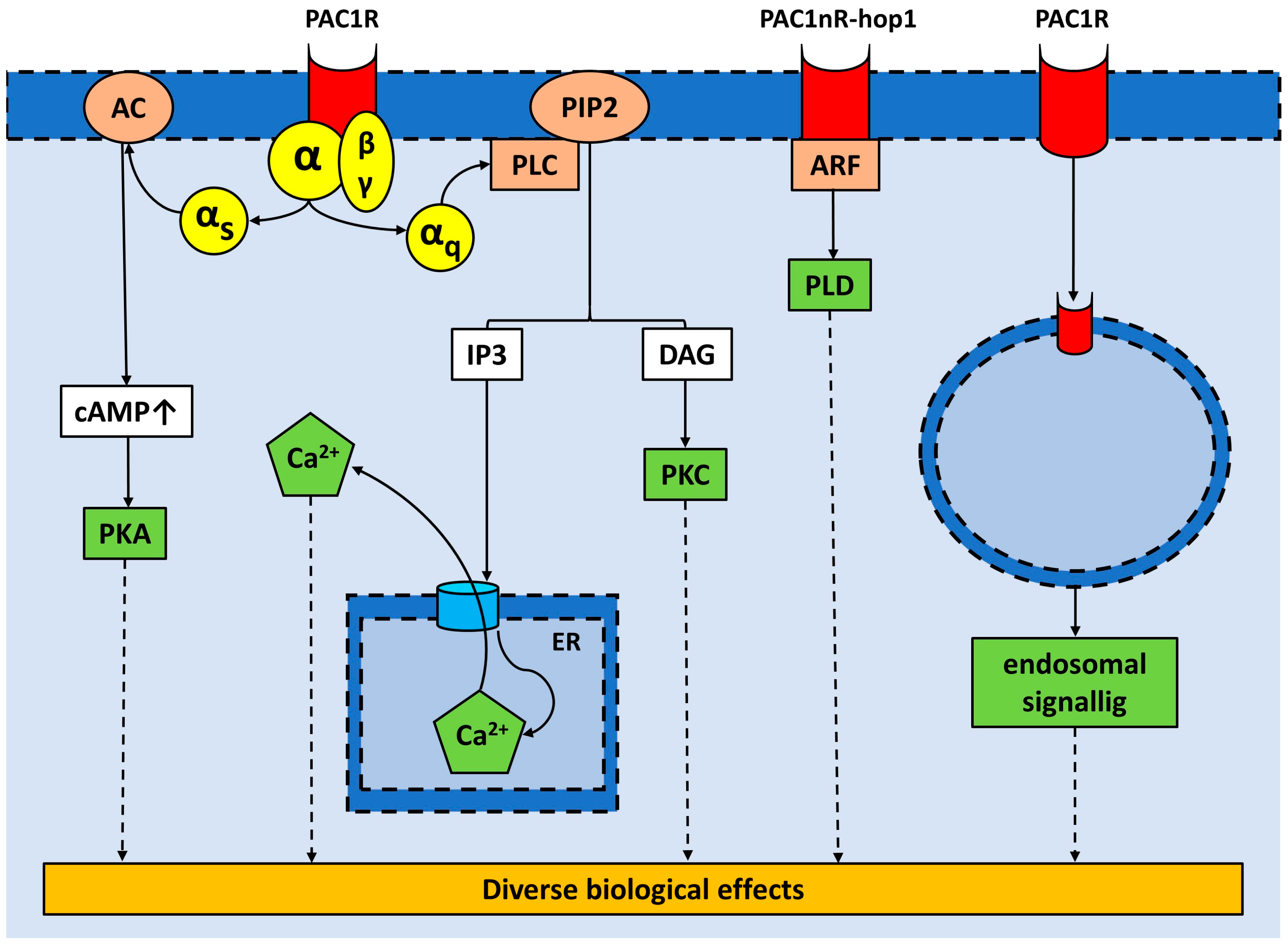
:1. Introduction
2. In Vivo Data
2.1. Knock Out Mouse Models
2.1.1. PACAP Knock Out Mice
2.1.2. PAC1R Knock Out Mice
2.2. Other Observations
3. Human Data
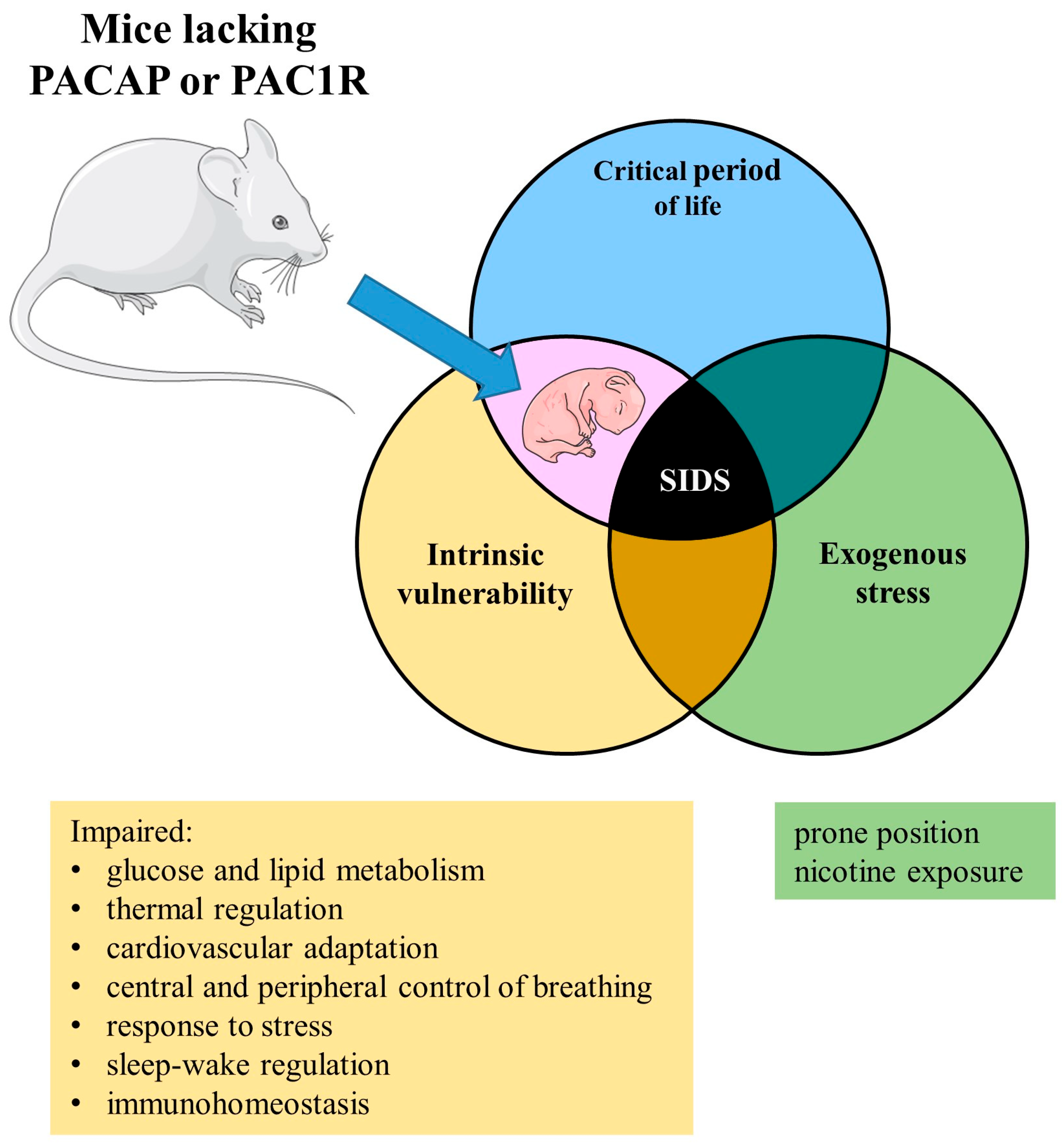
4. Discussion
- PACAP deficiency was found to be associated with impaired lipid and carbohydrate metabolism and an impaired response to metabolic stress [34,36]. For years, it has been known that metabolic diseases can occasionally manifest as SIDS, and human studies indicate that metabolic disorders might be responsible for 1% to 2% of SIDS cases [68,69]. These conditions can lead to severe cardiac failure, shock, cardiac arrest, or acute metabolic crises. Among metabolic disorders, fatty acid oxidation disorders are the most common culprits behind SIDS, and they can often appear with minimal or no preceding clinical symptoms [70,71]. These fatty acid oxidation disorders may be associated with lipid accumulation in various tissues, including skeletal muscle and the liver. Regarding carbohydrate metabolism, genetic deficiencies in the hepatic glucose-6-phosphatase system can lead to fasting hypoglycaemia and, hence, the risk of SIDS. Low hepatic glucose-6-phosphatase catalytic subunit 1 (G6PC1) activities have been previously observed in some full-term SIDS infants. In preterm SIDS infants, there is an indication of disrupted development, as failures in the postnatal activation of G6PC1 expression have been reported [72,73]. The significant downregulation of proteins linked to oxidative stress and antioxidant defence in PACAP-knockout mice [40] underscores that PACAP deficiency influences homeostasis and metabolic activity across multiple domains.
- Thermoregulatory disturbances have been noted in PACAP-deficient mice, where the absence of endogenous PACAP has led to insufficient heat production, primarily due to inadequate norepinephrine stimulation of brown adipose tissue during prolonged but mild cold stress [35]. This altered thermogenesis of PACAP-null mice has also been confirmed by another study [54]. They display hypometabolism and hypothermia under restrained conditions, partially compensated by increased locomotor activity under unrestrained conditions. PACAP is known to inhibit GABAergic neurons in the hypothalamic preoptic area and median preoptic nucleus, which leads to increased body temperature. The absence of this inhibitory mechanism could be the reason behind the decreased temperature and deficient thermoregulation in mice lacking PACAP, although the involvement of other brain structures has also been suggested in this mechanism [54]. During infancy, a hypothesis suggests that brown adipose tissue could play a role in the onset of sudden infant death syndrome (SIDS). This hypothesis is based on the idea that thermal stresses and disrupted thermogenesis are factors in this condition. However, it is important to note that human studies have not confirmed this hypothesis [74].
- In the PACAP knockout mice, there was also evidence of impaired autonomic responses to heat stress, characterized by reduced sympathetic responses. Specifically, the PACAP-null animals exhibited a significantly decreased respiratory rate, tidal volume, and minute ventilation during heat stress, along with slightly longer apnoea durations [41]. Infants are particularly vulnerable to heat stress because their temperature regulation mechanisms are still developing. The interplay between thermal stress and the body’s protective homeostatic responses can lead to potentially life-threatening situations, especially during sleep. It is known that respiration is highly influenced by thermoregulation, so thermal stress can have significant effects on the characteristics of respiratory control. Exposure to heat can negatively affect the autonomic nervous system, potentially disrupting the drive for cardiorespiratory function and hindering the ability to awaken when a vital system is compromised [75]. A study involving sleeping preterm neonates observed that even small thermal loads are associated with reduced overall heart rate variability [76]. Autonomic control also appears to be impaired in SIDS victims [77,78]. Anatomical abnormalities within the intermediolateral nucleus of the spinal cord have been documented in 60% of SIDS cases [79]. This nucleus houses PACAPergic preganglionic sympathetic neurons, which are crucial for regulating cardiorespiratory responses to heat and various physiological stressors. Notably, it is susceptible to pathological changes when exposed to maternal cigarette smoke, a significant risk factor associated with SIDS.
- In mice, PAC1R deficiency has been associated with pulmonary hypertension, which can lead to right heart failure and subsequent death within the second postnatal week [42]. Human data regarding abnormalities in pulmonary vessels observed in cases of SIDS are limited and somewhat controversial. Early observations noted increased muscularity in the pulmonary circulation of SIDS victims [80]. Bradley and colleagues reported a case of SIDS where pulmonary artery thickening and associated pulmonary hypertension played a critical role in the death of a 23-day-old infant [81]. Another study found that the mean relative medial thickness of the alveolar wall arteries did not differ between SIDS cases and age-matched control cases. However, within the SIDS group, the thickest alveolar wall arteries were significantly more likely to be males and premature births [82].
- The PACAP-null mice exhibited higher neonatal mortality primarily due to defects in respiratory control. They displayed reduced baseline minute ventilation and impaired ventilatory responses to hypercapnia and hypoxia. When exposed to hypothermia, these mice experienced prolonged apnoea, and, notably, the P7 PACAP-null mice had respiratory arrest in response to hypoxia [38,39]. PAC1R deficiency also led to impaired cardiorespiratory responses to both hypoxia and hypercapnia. This included blunted respiratory rate and minute ventilation responses to both hypoxia and hypercapnia, along with impaired post-hypoxic and post-hypercapnic cardiorespiratory recovery [43,44]. An investigation into the impact of acute and repeated intermittent hypercapnic hypoxia, simulating rebreathing in the prone position, on PACAP and PAC1R protein expression revealed that intermittent hypercapnic hypoxia led to a decrease in both PACAP and PAC1R expression in the brainstem nuclei responsible for respiratory regulation [45]. Another study indicated that precisely timed PACAP expression in the pre-Bötzinger complex and the RTN/parafacial respiratory group plays a crucial role in providing additional respiratory drive immediately after birth and supporting breathing during a particularly vulnerable phase of life [47]. These findings align with human data since the central nervous system in specific brainstem regions within the ventrolateral medulla controls the cardiorespiratory system. It is worth noting that each of these brainstem regions has specialized functions in regulating breathing and heartbeat: the pre-Bötzinger complex governs inspiration, the post-inspiratory complex controls post-inspiratory activity, and a subset of the parafacial respiratory group manages active expiration [83]. TH immunoreactive catecholaminergic neurons within the ventrolateral medulla oblongata display delayed dendritic development in SIDS individuals [84]. Furthermore, TH immunopositivity experiences a significant decrease in the DMNV and the area reticularis superficialis ventrolateralis of the medulla oblongata in SIDS cases [85].
- Nicotine exposure resulted in decreased PAC1R expression in piglets in the DMNV, while in mice, an increase in PAC1R expression was observed in NTS, CUN, NSTT, and ION [45,46]. Pre- and/or postnatal exposure to cigarette smoke is a factor that increases infant vulnerability to SIDS, with more than 40 studies showing a positive association and risk ratios ranging from 0.7 to 4.85. This heightened risk of SIDS is likely attributed to the effects of nicotine exposure on autonomic control and arousal, affecting, among others, the same brainstem nuclei [86].
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Krous, H.F.; Beckwith, J.B.; Byard, R.W.; Rognum, T.O.; Bajanowski, T.; Corey, T.; Cutz, E.; Hanzlick, R.; Keens, T.G.; Mitchell, E.A. Sudden infant death syndrome and unclassified sudden infant deaths: A definitional and diagnostic approach. Pediatrics 2004, 114, 234–238. [Google Scholar] [CrossRef]
- Matthews, T.J.; MacDorman, M.F.; Thoma, M.E. Infant mortality statistics from the 2013 period linked birth/infant death data set. Natl. Vital. Stat. Rep. 2015, 64, 9. [Google Scholar]
- Hakeem, G.F.; Oddy, L.; Holcroft, C.A.; Abenhaim, H.A. Incidence and determinants of sudden infant death syndrome: A population-based study on 37 million births. World J. Pediatr. 2015, 11, 41–47. [Google Scholar] [CrossRef]
- Shapiro-Mendoza, C.K.; Parks, S.; Lambert, A.E.; Camperlengo, L.; Cottengim, C.; Olson, C. The epidemiology of sudden infant death syndrome and sudden unexpected infant deaths: Diagnostic shift and other temporal changes. In SIDS Sudden Infant and Early Childhood Death: The Past, the Present and the Future; Duncan, J.R., Byard, R.W., Eds.; University of Adelaide Press: Adelaide, Australia, 2018; pp. 257–282. [Google Scholar]
- Goldstein, R.D.; Trachtenberg, F.L.; Sens, M.A.; Harty, B.J.; Kinney, H.C. Overall postneonatal mortality and rates of SIDS. Pediatrics 2016, 137, e20152298. [Google Scholar] [CrossRef]
- Shapiro-Mendoza, C.K.; Palusci, V.J.; Hoffman, B.; Batra, E.; Yester, M.; Corey, T.S.; Sens, M.A.; Moon, R.Y.; Goodstein, M.H. AAP task Force on sudden infant death syndrome, council on child abuse and neglect, council on injury, violence, and poison prevention, section on child death review and prevention, national association of medical examiners; Half century since SIDS: A reappraisal of terminology. Pediatrics 2021, 148, e2021053746. [Google Scholar] [CrossRef]
- Byard, R.W.; Lee, V. A re-audit of the use of definitions of sudden infant death syndrome (SIDS) in peer-reviewed literature. J. Forensic. Leg. Med. 2012, 19, 455–456. [Google Scholar] [CrossRef]
- Filiano, J.J.; Kinney, H.C. A perspective on neuropathologic findings in victims of the sudden infant death syndrome: The triple-risk model. Neonatology 1994, 65, 194–197. [Google Scholar] [CrossRef]
- Carlin, R.F.; Moon, R.Y. Risk factors, protective factors, and current recommendations to reduce sudden infant death syndrome: A review. JAMA Pediatr. 2017, 171, 175. [Google Scholar] [CrossRef]
- Blood-Siegfried, J. Animal models of sudden infant death syndrome. In Sourcebook of Models for Biomedical Research; Conn, P.M., Ed.; Humana Press: Totowa, NJ, USA, 2008; pp. 583–590. [Google Scholar]
- Li, A.; Darnall, R.A.; Dymecki, S.; Leiter, J.C. Animal models: Illuminating the pathogenesis of sudden infant death syndrome. In SIDS Sudden Infant and Early Childhood Death: The Past, the Present and the Future; Duncan, J.R., Byard, R.W., Eds.; University of Adelaide Press: Adelaide, Australia, 2018; pp. 759–828. [Google Scholar]
- Miyata, A.; Arimura, A.; Dahl, R.R.; Minamino, N.; Uehara, A.; Jiang, L.; Culler, M.D.; Coy, D.H. Isolation of a novel 38 residue-hypothalamic polypeptide which stimulates adenylate cyclase in pituitary cells. Biochem. Biophys. Res. Commun. 1989, 164, 567–574. [Google Scholar] [CrossRef]
- Miyata, A.; Jiang, L.; Dahl, R.D.; Kitada, C.; Kubo, K.; Fujino, M.; Minamino, N.; Arimura, A. Isolation of a neuropeptide corresponding to the n-terminal 27 residues of the pituitary adenylate cyclase activating polypeptide with 38 residues (PACAP38). Biochem. Biophys. Res. Commun. 1990, 170, 643–648. [Google Scholar] [CrossRef]
- Vaudry, D.; Falluel-Morel, A.; Bourgault, S.; Basille, M.; Burel, D.; Wurtz, O.; Fournier, A.; Chow, B.K.C.; Hashimoto, H.; Galas, L.; et al. Pituitary adenylate cyclase-activating polypeptide and its receptors: 20 years after the discovery. Pharmacol. Rev. 2009, 61, 283–357. [Google Scholar] [CrossRef]
- Lu, J.; Piper, S.J.; Zhao, P.; Miller, L.J.; Wootten, D.; Sexton, P.M. Targeting VIP and PACAP receptor signaling: New insights into designing drugs for the PACAP subfamily of receptors. Int. J. Mol. Sci. 2022, 23, 8069. [Google Scholar] [CrossRef]
- Langer, I.; Jeandriens, J.; Couvineau, A.; Sanmukh, S.; Latek, D. Signal transduction by VIP and PACAP receptors. Biomedicines 2022, 10, 406. [Google Scholar] [CrossRef]
- McCulloch, D.A.; Lutz, E.M.; Johnson, M.S.; MacKenzie, C.J.; Mitchell, R. Differential activation of phospholipase D by VPAC and PAC1 receptors. Ann. N. Y. Acad. Sci. 2000, 921, 175–185. [Google Scholar] [CrossRef]
- McCulloch, D.A.; Lutz, E.M.; Johnson, M.S.; Robertson, D.N.; MacKenzie, C.J.; Holland, P.J.; Mitchell, R. ADP-ribosylation factor-dependent phospholipase D activation by VPAC receptors and a PAC1 receptor splice variant. Mol. Pharmacol. 2001, 59, 1523–1532. [Google Scholar] [CrossRef]
- May, V.; Lutz, E.; MacKenzie, C.; Schutz, K.C.; Dozark, K.; Braas, K.M. Pituitary adenylate cyclase-activating polypeptide (PACAP)/PAC1HOP1 receptor activation coordinates multiple neurotrophic signaling pathways. J. Biol. Chem. 2010, 285, 9749–9761. [Google Scholar] [CrossRef]
- May, V.; Buttolph, T.R.; Girard, B.M.; Clason, T.A.; Parsons, R.L. PACAP-induced ERK activation in HEK cells expressing PAC1 receptors involves both receptor internalization and PKC signaling. Am. J. Physiol. Cell Physiol. 2014, 306, C1068–C1079. [Google Scholar] [CrossRef]
- Reglodi, D.; Tamas, A.; Koppan, M.; Szogyi, D.; Welke, L. Role of PACAP in female fertility and reproduction at gonadal level–recent advances. Front. Endocrin. 2012, 3, 155. [Google Scholar] [CrossRef]
- Watanabe, J.; Seki, T.; Shioda, S. PACAP and neural development. In Pituitary Adenylate Cyclase Activating Polypeptide—PACAP; Reglodi, D., Tamas, A., Eds.; Current topics in neurotoxicity; Springer International Publishing: Cham, Switzerland, 2016; Volume 11, pp. 65–82. [Google Scholar]
- Reglodi, D.; Atlasz, T.; Szabo, E.; Jungling, A.; Tamas, A.; Juhasz, T.; Fulop, B.D.; Bardosi, A. PACAP deficiency as a model of aging. GeroScience 2018, 40, 437–452. [Google Scholar] [CrossRef]
- Reglodi, D.; Helyes, Z.; Nemeth, J.; Vass, R.A.; Tamas, A. PACAP as a potential biomarker: Alterations of PACAP levels in human physiological and pathological conditions. In Pituitary Adenylate Cyclase Activating Polypeptide—PACAP; Reglodi, D., Tamas, A., Eds.; Current topics in neurotoxicity; Springer International Publishing: Cham, Switzerland, 2016; Volume 11, pp. 815–832. [Google Scholar]
- Toth, D.; Reglodi, D.; Schwieters, L.; Tamas, A. Role of endocrine PACAP in age-related diseases. Front. Endocrinol. 2023, 14, 1118927. [Google Scholar] [CrossRef]
- Toth, D.; Tamas, A.; Reglodi, D. The neuroprotective and biomarker potential of PACAP in human traumatic brain injury. Int. J. Mol. Sci. 2020, 21, 827. [Google Scholar] [CrossRef]
- Tamás, A.; Tóth, D.; Pham, D.; Loibl, C.; Rendeki, S.; Csontos, C.; Rozanovic, M.; Bogár, L.; Polgár, B.; Németh, J.; et al. Changes of pituitary adenylate cyclase activating polypeptide (PACAP) level in polytrauma patients in the early post-traumatic period. Peptides 2021, 146, 170645. [Google Scholar] [CrossRef]
- Staines, D.R. Is sudden infant death syndrome (SIDS) an autoimmune disorder of endogenous vasoactive neuropeptides? Med. Hypotheses 2004, 62, 653–657. [Google Scholar] [CrossRef]
- Hashimoto, H.; Shintani, N.; Tanaka, K.; Mori, W.; Hirose, M.; Matsuda, T.; Sakaue, M.; Miyazaki, J.; Niwa, H.; Tashiro, F.; et al. Altered psychomotor behaviors in mice lacking pituitary adenylate cyclase-activating polypeptide (PACAP). Proc. Natl. Acad. Sci. USA 2001, 98, 13355–13360. [Google Scholar] [CrossRef]
- Jamen, F.; Persson, K.; Bertrand, G.; Rodriguez-Henche, N.; Puech, R.; Bockaert, J.; Ahrén, B.; Brabet, P. PAC1 receptor–deficient mice display impaired insulinotropic response to glucose and reduced glucose tolerance. J. Clin. Investig. 2000, 105, 1307–1315. [Google Scholar] [CrossRef]
- Wilson, R.J.A.; Cummings, K.J. Pituitary adenylate cyclase-activating polypeptide is vital for neonatal survival and the neuronal control of breathing. Respir. Physiol. Neurobiol. 2008, 164, 168–178. [Google Scholar] [CrossRef]
- Salomonis, N. Systems-level perspective of sudden infant death syndrome. Pediatr. Res. 2014, 76, 220–229. [Google Scholar] [CrossRef]
- Stalley, E.; Waters, K.A.; Machaalani, R. Genes involved in paediatric apnoea and death based on knockout animal models: Implications for sudden infant death syndrome (SIDS). Paediatr. Respir. Rev. 2022, 44, 53–60. [Google Scholar] [CrossRef]
- Gray, S.L.; Cummings, K.J.; Jirik, F.R.; Sherwood, N.M. Targeted disruption of the pituitary adenylate cyclase-activating polypeptide gene results in early postnatal death associated with dysfunction of lipid and carbohydrate metabolism. Mol. Endocrinol. 2001, 15, 1739–1747. [Google Scholar] [CrossRef]
- Gray, S.L.; Yamaguchi, N.; Vencová, P.; Sherwood, N.M. Temperature-sensitive phenotype in mice lacking pituitary adenylate cyclase-activating polypeptide. Endocrinology 2002, 143, 3946–3954. [Google Scholar] [CrossRef]
- Hamelink, C.; Tjurmina, O.; Damadzic, R.; Young, W.S.; Weihe, E.; Lee, H.-W.; Eiden, L.E. Pituitary adenylate cyclase-activating polypeptide is a sympathoadrenal neurotransmitter involved in catecholamine regulation and glucohomeostasis. Proc. Natl. Acad. Sci. USA 2002, 99, 461–466. [Google Scholar] [CrossRef] [PubMed]
- Shintani, N.; Hashimoto, H.; Tanaka, K.; Kawaguchi, C.; Tomimoto, S.; Baba, A. Overexpression of PACAP in the pancreas failed to rescue early postnatal mortality in PACAP-null mice. Regul. Pept. 2004, 123, 155–159. [Google Scholar] [CrossRef] [PubMed]
- Cummings, K.J.; Pendlebury, J.D.; Sherwood, N.M.; Wilson, R.J.A. Sudden neonatal death in PACAP-deficient mice is associated with reduced respiratory chemoresponse and susceptibility to apnoea: Breathing abnormalities in PACAP-deficient mice. J. Physiol. 2004, 555, 15–26. [Google Scholar] [CrossRef] [PubMed]
- Arata, S.; Nakamachi, T.; Onimaru, H.; Hashimoto, H.; Shioda, S. Impaired response to hypoxia in the respiratory center is a major cause of neonatal death of the PACAP-knockout mouse. Eur. J. Neurosci. 2013, 37, 407–416. [Google Scholar] [CrossRef]
- Maasz, G.; Pirger, Z.; Reglodi, D.; Petrovics, D.; Schmidt, J.; Kiss, P.; Rivnyak, A.; Hashimoto, H.; Avar, P.; Jambor, E.; et al. Comparative protein composition of the brains of PACAP-deficient mice using mass spectrometry-based proteomic analysis. J. Mol. Neurosci. 2014, 54, 310–319. [Google Scholar] [CrossRef] [PubMed]
- Barrett, K.T.; Daubenspeck, J.A.; Wilson, R.J.A. Pituitary adenylate cyclase-activating polypeptide drives cardiorespiratory responses to heat stress in neonatal mice. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2017, 313, R385–R394. [Google Scholar] [CrossRef] [PubMed]
- Otto, C.; Hein, L.; Brede, M.; Jahns, R.; Engelhardt, S.; Gröne, H.-J.; Schütz, G. Pulmonary hypertension and right heart failure in pituitary adenylate cyclase–activating polypeptide type I receptor–deficient mice. Circulation 2004, 110, 3245–3251. [Google Scholar] [CrossRef]
- Barrett, K.T.; Hasan, S.U.; Scantlebury, M.H.; Wilson, R.J.A. Impaired neonatal cardiorespiratory responses to hypoxia in mice lacking PAC1 or VPAC2 receptors. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2019, 316, R594–R606. [Google Scholar] [CrossRef]
- Barrett, K.T.; Hasan, S.U.; Scantlebury, M.H.; Wilson, R.J.A. Impaired cardiorespiratory responses to hypercapnia in neonatal mice lacking PAC1 but not VPAC2 receptors. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2021, 320, R116–R128. [Google Scholar] [CrossRef]
- Huang, J.; Waters, K.A.; Machaalani, R. Hypoxia and nicotine effects on pituitary adenylate cyclase activating polypeptide (PACAP) and its receptor 1 (PAC1) in the developing piglet brainstem. Neurotoxicology 2017, 62, 30–38. [Google Scholar] [CrossRef]
- Machaalani, R.; Thawley, M.; Huang, J.; Chen, H. Effects of prenatal cigarette smoke exposure on BDNF, PACAP, microglia and gliosis expression in the young male mouse brainstem. Neurotoxicology 2019, 74, 40–46. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y.; Stornetta, D.S.; Reklow, R.J.; Sahu, A.; Wabara, Y.; Nguyen, A.; Li, K.; Zhang, Y.; Perez-Reyes, E.; Ross, R.A.; et al. A brainstem peptide system activated at birth protects postnatal breathing. Nature 2021, 589, 426–430. [Google Scholar] [CrossRef] [PubMed]
- Cummings, K.J.; Klotz, C.; Liu, W.-Q.; Weese-Mayer, D.E.; Marazita, M.L.; Cooper, M.E.; Berry-Kravis, E.M.; Tobias, R.; Goldie, C.; Bech-Hansen, N.T.; et al. Sudden infant death syndrome (SIDS) in african americans: Polymorphisms in the gene encoding the stress peptide pituitary adenylate cyclase-activating polypeptide (PACAP). Acta Paediatr. 2009, 98, 482–489. [Google Scholar] [CrossRef] [PubMed]
- Barrett, K.T.; Rodikova, E.; Weese-Mayer, D.E.; Rand, C.M.; Marazita, M.L.; Cooper, M.E.; Berry-Kravis, E.M.; Bech-Hansen, N.T.; Wilson, R.J. Analysis of PAC1 receptor gene variants in caucasian and african american infants dying of sudden infant death syndrome. Acta Paediatr. 2013, 102, e546–e552. [Google Scholar] [CrossRef]
- Huang, J.; Waters, K.A.; Machaalani, R. Pituitary adenylate cyclase activating polypeptide (PACAP) and its receptor 1 (PAC1) in the human infant brain and changes in the sudden infant death syndrome (SIDS). Neurobiol. Dis. 2017, 103, 70–77. [Google Scholar] [CrossRef]
- Cardoso, J.C.R.; Garcia, M.G.; Power, D.M. Tracing the origins of the pituitary adenylate-cyclase activating polypeptide (PACAP). Front. Neurosci. 2020, 14, 366. [Google Scholar] [CrossRef]
- Sherwood, N.M.; Krueckl, S.L.; McRory, J.E. The origin and function of the pituitary adenylate cyclase-activating polypeptide (PACAP)/glucagon superfamily. Endocr. Rev. 2000, 21, 619–670. [Google Scholar] [CrossRef]
- Adams, B.A.; Gray, S.L.; Isaac, E.R.; Bianco, A.C.; Vidal-Puig, A.J.; Sherwood, N.M. Feeding and metabolism in mice lacking pituitary adenylate cyclase-activating polypeptide. Endocrinology 2008, 149, 1571–1580. [Google Scholar] [CrossRef]
- Garami, A.; Pakai, E.; Rumbus, Z.; Solymar, M. The role of PACAP in the regulation of body temperature. In Pituitary Adenylate Cyclase Activating Polypeptide—PACAP; Reglodi, D., Tamas, A., Eds.; Current topics in neurotoxicity; Springer International Publishing: Cham, Switzerland, 2016; Volume 11, pp. 239–257. [Google Scholar]
- Rudecki, A.P.; Gray, S.L. PACAP in the defense of energy homeostasis. Trends Endocrinol. Metab. 2016, 27, 620–632. [Google Scholar] [CrossRef]
- Farnham, M.M.J.; Li, Q.; Goodchild, A.K.; Pilowsky, P.M. PACAP is expressed in sympathoexcitatory bulbospinal C1 neurons of the brain stem and increases sympathetic nerve activity in vivo. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2008, 294, R1304–R1311. [Google Scholar] [CrossRef]
- Farnham, M.M.J.; Pilowsky, P.M. The role of PACAP in central cardiorespiratory regulation. Respir. Physiol. Neurobiol. 2010, 174, 65–75. [Google Scholar] [CrossRef]
- Liu, Q.; Wong-Riley, M.T.T. Pituitary adenylate cyclase-activating polypeptide: Postnatal development in multiple brain stem respiratory-related nuclei in the rat. Respir. Physiol. Neurobiol. 2019, 259, 149–155. [Google Scholar] [CrossRef]
- Peña, F. PACAP modulates the respiratory rhythm generated in the brainstem slice preparation. In New Frontiers in Respiratory Control; Homma, I., Onimaru, H., Fukuchi, Y., Eds.; Advances in experimental medicine and biology; Springer: New York, NY, USA, 2010; Volume 669, pp. 119–122. [Google Scholar]
- Ferguson, E.V.; Roy, A.; Ciechanski, P.; Wilson, R.J.A. Stress peptide PACAP stimulates and stabilizes neonatal breathing through distinct mechanisms. Respir. Physiol. Neurobiol. 2013, 187, 217–223. [Google Scholar] [CrossRef] [PubMed]
- Eiden, L.E.; Emery, A.C.; Zhang, L.; Smith, C.B. PACAP signaling in stress: Insights from the chromaffin cell. Pflugers Arch. Eur. J. Physiol. 2018, 470, 79–88. [Google Scholar] [CrossRef] [PubMed]
- Mustafa, T. Pituitary adenylate cyclase-activating polypeptide (PACAP): A master regulator in central and peripheral stress responses. In Advances in Pharmacology; Lee, E.E., Ed.; Elsevier: Amsterdam, The Netherlands, 2013; Volume 68, pp. 445–457. [Google Scholar]
- Jiang, S.Z.; Eiden, L.E. PACAPergic synaptic signaling and circuitry mediating mammalian responses to psychogenic and systemic stressors. In Pituitary Adenylate Cyclase Activating Polypeptide—PACAP; Reglodi, D., Tamas, A., Eds.; Current topics in neurotoxicity; Springer International Publishing: Cham, Switzerland, 2016; Volume 11, pp. 711–729. [Google Scholar]
- Nussdorfer, G.G.; Malendowicz, L.K. Role of VIP, PACAP, and related peptides in the regulation of the hypothalamo—Pituitary–adrenal axis. Peptides 1998, 19, 1443–1467. [Google Scholar] [CrossRef] [PubMed]
- Ahnaou, A. Immunocytochemical distribution of VIP and PACAP in the rat brain stem: Implications for REM sleep physiology. Ann. N. Y. Acad. Sci. 2006, 1070, 135–142. [Google Scholar] [CrossRef]
- Murck, H.; Steiger, A.; Frieboes, R.M.; Antonijevic, I.A. Pituitary adenylate cyclase-activating peptide affects homeostatic sleep regulation in healthy young men. Am. J. Physiol. Endocrinol. Metab. 2007, 292, E853–E857. [Google Scholar] [CrossRef]
- Abad, C.; Tan, Y.-V. Immunomodulatory roles of PACAP and VIP: Lessons from knockout mice. J. Mol. Neurosci. 2018, 66, 102–113. [Google Scholar] [CrossRef]
- Pryce, J.W.; Weber, M.A.; Heales, S.; Malone, M.; Sebire, N.J. Tandem mass spectrometry findings at autopsy for detection of metabolic disease in infant deaths: Postmortem changes and confounding factors. J. Clin. Path 2011, 64, 1005–1009. [Google Scholar] [CrossRef]
- van Rijt, W.J.; Koolhaas, G.D.; Bekhof, J.; Fokkema, M.R.H.; de Koning, T.J.; Visser, G.; Schielen, P.C.J.I.; van Spronsen, F.J.; Derks, T.G.J. Inborn errors of metabolism that cause sudden infant death: A systematic review with implications for population neonatal screening programmes. Neonatology 2016, 109, 297–302. [Google Scholar] [CrossRef]
- Olpin, S.E. The metabolic investigation of sudden infant death. Ann. Clin. Biochem. 2004, 41, 282–293. [Google Scholar] [CrossRef] [PubMed]
- Keywan, C.; Poduri, A.H.; Goldstein, R.D.; Holm, I.A. Genetic factors underlying sudden infant death syndrome. Appl. Clin. Genet. 2021, 14, 61–76. [Google Scholar] [CrossRef] [PubMed]
- Forsyth, L.; Hume, R.; Howatson, A.; Busuttil, A.; Burchell, A. Identification of novel polymorphisms in the glucokinase and glucose-6-phosphatase genes in infants who died suddenly and unexpectedly. J. Mol. Med. 2005, 83, 610–618. [Google Scholar] [CrossRef] [PubMed]
- Forsyth, L.; Scott, H.; Howatson, A.; Busuttil, A.; Hume, R.; Burchell, A. Genetic variation in hepatic glucose-6-phosphatase system genes in cases of sudden infant death syndrome. J. Pathol. 2007, 212, 112–120. [Google Scholar] [CrossRef]
- Verduci, E.; Calcaterra, V.; Di Profio, E.; Fiore, G.; Rey, F.; Magenes, V.C.; Todisco, C.F.; Carelli, S.; Zuccotti, G.V. Brown adipose tissue: New challenges for prevention of childhood obesity. A narrative review. Nutrients 2021, 13, 1450. [Google Scholar] [CrossRef]
- Bach, V.; Libert, J.-P. Hyperthermia and heat stress as risk factors for sudden infant death syndrome: A narrative review. Front. Pediatr. 2022, 10, 816136. [Google Scholar] [CrossRef]
- Stéphan-Blanchard, E.; Chardon, K.; Léké, A.; Delanaud, S.; Bach, V.; Telliez, F. Heart rate variability in sleeping preterm neonates exposed to cool and warm thermal conditions. PLoS ONE 2013, 8, e68211. [Google Scholar] [CrossRef]
- Schechtman, V.L.; Harper, R.M.; Kluge, K.A.; Wilson, A.J.; Hoffman, H.J.; Southall, D.P. Heart rate variation in normal infants and victims of the sudden infant death syndrome. Early Hum. Dev. 1989, 19, 167–181. [Google Scholar] [CrossRef]
- Kahn, A.; Groswasser, J.; Franco, P.; Scaillet, S.; Sawaguchi, T.; Kelmanson, I.; Dan, B. Sudden infant deaths: Stress, arousal and SIDS. Early Hum. Dev. 2003, 75, 147–166. [Google Scholar] [CrossRef]
- Lavezzi, A.M.; Corna, M.F.; Mehboob, R.; Matturri, L. Neuropathology of the intermediolateral nucleus of the spinal cord in sudden unexplained perinatal and infant death. Int. J. Dev. Neurosci. 2010, 28, 133–138. [Google Scholar] [CrossRef]
- Williams, A.; Vawter, G.; Reid, L. Increased muscularity of the pulmonary circulation in victims of sudden infant death syndrome. Pediatrics 1979, 63, 18–23. [Google Scholar] [CrossRef] [PubMed]
- Thach, B.T.; Harris, K.A.; Krous, H. Pulmonary arteriolar muscle thickening in sudden infant death syndrome. BMJ Case Rep. 2009, 2009, bcr1020081026. [Google Scholar] [CrossRef] [PubMed]
- Krous, H.F.; Haas, E.; Hampton, C.F.; Chadwick, A.E.; Stanley, C.; Langston, C. Pulmonary arterial medial smooth muscle thickness in sudden infant death syndrome: An analysis of subsets of 73 cases. Forensic. Sci. Med. Pathol. 2009, 5, 261–268. [Google Scholar] [CrossRef] [PubMed]
- Ramirez, J.M.; Ramirez, S.C.; Anderson, T.M. Sudden infant death syndrome, sleep, and the physiology and pathophysiology of the respiratory network. In SIDS Sudden Infant and Early Childhood Death: The Past, the Present and the Future; Duncan, J.R., Byard, R.W., Eds.; University of Adelaide Press: Adelaide, Australia, 2018; pp. 615–641. [Google Scholar]
- Takashima, S.; Becker, L. Delayed dendritic development of catecholaminergic neurons in the ventrolateral medulla of children who died of sudden infant death syndrome. Neuropediatrics 1991, 22, 97–99. [Google Scholar] [CrossRef] [PubMed]
- Obonai, T.; Yasuhara, M.; Nakamura, T.; Takashima, S. Catecholamine neurons alteration in the brainstem of sudden infant death syndrome victims. Pediatrics 1998, 101, 285–288. [Google Scholar] [CrossRef]
- Horne, R.S.C. Autonomic cardiorespiratory physiology and arousal of the fetus and infant. In SIDS Sudden Infant and Early Childhood Death: The Past, the Present and the Future; Duncan, J.R., Byard, R.W., Eds.; University of Adelaide Press: Adelaide, Australia, 2018; pp. 449–491. [Google Scholar]
- Farkas, J.; Kovács, L.Á.; Gáspár, L.; Nafz, A.; Gaszner, T.; Ujvári, B.; Kormos, V.; Csernus, V.; Hashimoto, H.; Reglődi, D.; et al. Construct and face validity of a new model for the three-hit theory of depression using PACAP mutant mice on CD1 background. Neuroscience 2017, 354, 11–29. [Google Scholar] [CrossRef]
| Condition(s) | Observations | Reference | Translational Value | |
| PACAP knock out mice | general |
| [29] | corresponds with epidemiological data related to human SIDS |
| metabolism (lipid and carbohydrate) |
| [34] | can be identified in specific subsets of human SIDS cases. | |
| thermal regulation (cold stress) |
| [35] | not proved in human SIDS cases | |
| metabolism (carbohydrate) |
| [36] | can be identified in specific subsets of human SIDS cases. | |
| general |
| [37] | no human data | |
| respiratory and thermal (cold stress) regulation |
| [38] | corresponds/similarities with data related to human SIDS | |
| respiratory regulation |
| [39] | corresponds/similarities with data related to human SIDS | |
| general |
| [40] | no human data | |
| respiratory and thermal (heat stress) regulation |
| [41] | corresponds/similarities with data related to human SIDS | |
| PAC1R knock out mice | general |
| [30] | corresponds with epidemiological data related to human SIDS |
| general |
| [42] | controversial data in human studies | |
| cardiorespiratory regulation |
| [43] | corresponds/similarities with data related to human SIDS | |
| cardiorespiratory regulation |
| [44] | corresponds/similarities with data related to human SIDS | |
| Other in vivo data | respiratory regulation (prone position, nicotine exposure)—piglet |
| [45] | corresponds/similarities with data related to human SIDS |
| respiratory regulation (cigarette smoke exposure)—mice |
| [46] | corresponds/similarities with data related to human SIDS | |
| respiratory regulation (transgenic mice and viral techniques) |
| [47] | corresponds/similarities with data related to human SIDS | |
| Human data | genetic study |
| [48] | - |
| genetic study |
| [49] | - | |
| immunohistochemical study |
| [50] | - |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tóth, D.; Simon, G.; Reglődi, D. Pituitary Adenylate Cyclase-Activating Polypeptide (PACAP) and Sudden Infant Death Syndrome: A Potential Model for Investigation. Int. J. Mol. Sci. 2023, 24, 15063. https://doi.org/10.3390/ijms242015063
Tóth D, Simon G, Reglődi D. Pituitary Adenylate Cyclase-Activating Polypeptide (PACAP) and Sudden Infant Death Syndrome: A Potential Model for Investigation. International Journal of Molecular Sciences. 2023; 24(20):15063. https://doi.org/10.3390/ijms242015063
Chicago/Turabian StyleTóth, Dénes, Gábor Simon, and Dóra Reglődi. 2023. "Pituitary Adenylate Cyclase-Activating Polypeptide (PACAP) and Sudden Infant Death Syndrome: A Potential Model for Investigation" International Journal of Molecular Sciences 24, no. 20: 15063. https://doi.org/10.3390/ijms242015063







