Sex Differences in Brain Region-Specific Activation of c-Fos following Kappa Opioid Receptor Stimulation or Acute Stress in Mice
Abstract
:1. Introduction
2. Results
2.1. Effects of Systemic Administration of U50,488 on c-Fos Expression in the PFCx of Male and Female Mice
2.2. Effects of Acute FSS on c-Fos Expression in the PFCx of Male and Female Mice
2.3. Effects of Activating KOPr and Acute FSS on c-Fos Expression in the NAcc
2.4. Effects of Activating KOPr and Acute FSS on c-Fos Expression in the Hippocampus
2.5. Effects of Activating KOPr and Acute FSS on c-Fos Expression in the Amygdala
2.6. KOPr- and Acute FSS-Activated Cells Are Predominantly Non-GABAergic Neurons
3. Discussion
3.1. Sex Differences following Systemic KOPr Agonist Administration
3.2. Sex Differences following an Acute In Vivo Stressor
3.3. Functional Role of Sex-Dependent c-Fos Expression following KOPr Agonist Administration and In Vivo Stress
3.4. Conclusions
4. Materials and Methods
4.1. Animals
4.2. Drugs
4.3. Acute Forced Swim Stress (FSS)
4.4. Immunohistochemistry
4.5. Quantification and Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Daviu, N.; Bruchas, M.R.; Moghaddam, B.; Sandi, C.; Beyeler, A. Neurobiological links between stress and anxiety. Neurobiol. Stress 2019, 11, 100191. [Google Scholar] [CrossRef] [PubMed]
- Kendler, K.S.; Karkowski, L.M.; Prescott, C.A. Causal relationship between stressful life events and the onset of major depression. Am. J. Psychiatry 1999, 156, 837–841. [Google Scholar] [CrossRef]
- López, J.F.; Akil, H.; Watson, S.J. Neural circuits mediating stress. Biol. Psychiatry 1999, 46, 1461–1471. [Google Scholar] [CrossRef] [PubMed]
- Mobbs, D.; Petrovic, P.; Marchant, J.L.; Hassabis, D.; Weiskopf, N.; Seymour, B.; Dolan, R.J.; Frith, C.D. When Fear Is Near: Threat Imminence Elicits Prefrontal-Periaqueductal Gray Shifts in Humans. Science 2007, 317, 1079. [Google Scholar] [CrossRef]
- Moghaddam, B. The Complicated Relationship of Stress and Prefrontal Cortex. Biol. Psychiatry 2016, 80, 728–729. [Google Scholar] [CrossRef]
- Belujon, P.; Grace, A.A. Regulation of dopamine system responsivity and its adaptive and pathological response to stress. Proc. Biol. Sci. 2015, 282, 20142516. [Google Scholar] [CrossRef] [PubMed]
- Tejeda, H.A.; Shippenberg, T.S.; Henriksson, R. The dynorphin/κ-opioid receptor system and its role in psychiatric disorders. Cell. Mol. Life Sci. 2012, 69, 857–896. [Google Scholar] [CrossRef]
- Henriksen, G.; Willoch, F. Imaging of opioid receptors in the central nervous system. Brain 2008, 131, 1171–1196. [Google Scholar] [CrossRef] [PubMed]
- Land, B.B.; Bruchas, M.R.; Lemos, J.C.; Xu, M.; Melief, E.J.; Chavkin, C. The dysphoric component of stress is encoded by activation of the dynorphin kappa-opioid system. J. Neurosci. 2008, 28, 407–414. [Google Scholar] [CrossRef]
- Shirayama, Y.; Ishida, H.; Iwata, M.; Hazama, G.I.; Kawahara, R.; Duman, R.S. Stress increases dynorphin immunoreactivity in limbic brain regions and dynorphin antagonism produces antidepressant-like effects. J. Neurochem. 2004, 90, 1258–1268. [Google Scholar] [CrossRef]
- Pfeiffer, A.; Brantl, V.; Herz, A.; Emrich, H.M. Psychotomimesis mediated by kappa opiate receptors. Science 1986, 233, 774–776. [Google Scholar] [CrossRef]
- Carlezon, W.A.; Beguin, C.; DiNieri, J.A.; Baumann, M.H.; Richards, M.R.; Todtenkopf, M.S.; Rothman, R.B.; Ma, Z.; Lee, D.Y.W.; Cohen, B.M. Depressive-like effects of the kappa-opioid receptor agonist salvinorin A on behavior and neurochemistry in rats. J. Pharmacol. Exp. Ther. 2006, 316, 440–447. [Google Scholar] [CrossRef] [PubMed]
- Casal-Dominguez, J.J.; Clark, M.; Traynor, J.R.; Husbands, S.M.; Bailey, S.J. In Vivo and In Vitro characterization of naltrindole-derived ligands at the κ-opioid receptor. J. Psychopharmacol. 2013, 27, 192–202. [Google Scholar] [CrossRef] [PubMed]
- McLaughlin, J.P.; Marton-Popovici, M.; Chavkin, C. Kappa opioid receptor antagonism and prodynorphin gene disruption block stress-induced behavioral responses. J. Neurosci. 2003, 23, 5674–5683. [Google Scholar] [CrossRef]
- Wittmann, W.; Schunk, E.; Rosskothen, I.; Gaburro, S.; Singewald, N.; Herzog, H.; Schwarzer, C. Prodynorphin-Derived Peptides Are Critical Modulators of Anxiety and Regulate Neurochemistry and Corticosterone. Neuropsychopharmacology 2009, 34, 775–785. [Google Scholar] [CrossRef] [PubMed]
- Bailey, S.J.; Husbands, S.M. Targeting opioid receptor signaling in depression: Do we need selective κ opioid receptor antagonists? Neuronal Signal. 2018, 2, NS20170145. [Google Scholar] [CrossRef] [PubMed]
- Carlezon, W.A.; Krystal, A.D. Kappa-Opioid Antagonists for Psychiatric Disorders: From Bench to Clinical Trials. Depress. Anxiety 2016, 33, 895–906. [Google Scholar] [CrossRef]
- McHugh, K.L.; Kelly, J.P. Modulation of the central opioid system as an antidepressant target in rodent models. Prog. Brain Res. 2018, 239, 49–87. [Google Scholar]
- Bolea-Alamanac, B.; Bailey, S.J.; Lovick, T.A.; Scheele, D.; Valentino, R. Female psychopharmacology matters! Towards a sex-specific psychopharmacology. J. Psychopharmacol. 2018, 32, 125–133. [Google Scholar] [CrossRef]
- Hodes, G.E.; Kropp, D.R. Sex as a biological variable in stress and mood disorder research. Nat. Ment. Health 2023, 1, 453–461. [Google Scholar] [CrossRef]
- Altemus, M.; Sarvaiya, N.; Neill Epperson, C. Sex differences in anxiety and depression clinical perspectives. Front. Neuroendocr. 2014, 35, 320–330. [Google Scholar] [CrossRef] [PubMed]
- Kessler, R.C. Gender differences in major depression. In Gender and Its Effects on Psychopathology; American Psychiatric Press, Inc.: Washington, DC, USA, 2000; pp. 61–84. [Google Scholar]
- Becker, J.B.; Hu, M. Sex differences in drug abuse. Front. Neuroendocr. 2008, 29, 36–47. [Google Scholar] [CrossRef]
- Kudielka, B.M.; Kirschbaum, C. Sex differences in HPA axis responses to stress: A review. Biol. Psychol. 2005, 69, 113–132. [Google Scholar] [CrossRef]
- Chartoff, E.H.; Mavrikaki, M. Sex Differences in Kappa Opioid Receptor Function and Their Potential Impact on Addiction. Front. Neurosci. 2015, 9, 466. [Google Scholar] [CrossRef]
- Kavaliers, M.; Innes, D.G.L. Sex and day-night differences in opiate-induced responses of insular wild deer mice, Peromyscus maniculatus triangularis. Pharmacol. Biochem. Behav. 1987, 27, 477–482. [Google Scholar] [CrossRef] [PubMed]
- Liu, N.-J.; Schnell, S.; Wessendorf, M.W.; Gintzler, A.R. Sex, Pain, and Opioids: Interdependent Influences of Sex and Pain Modality on Dynorphin-Mediated Antinociception in Rats. J. Pharmacol. Exp. Ther. 2013, 344, 522–530. [Google Scholar] [CrossRef]
- Abraham, A.D.; Schattauer, S.S.; Reichard, K.L.; Cohen, J.H.; Fontaine, H.M.; Song, A.J.; Johnson, S.D.; Land, B.B.; Chavkin, C. Estrogen Regulation of GRK2 Inactivates Kappa Opioid Receptor Signaling Mediating Analgesia, But Not Aversion. J. Neurosci. 2018, 38, 8031–8043. [Google Scholar] [CrossRef]
- Lawson, K.P.; Nag, S.; Thompson, A.D.; Mokha, S.S. Sex-specificity and estrogen-dependence of kappa opioid receptor-mediated antinociception and antihyperalgesia. Pain 2010, 151, 806–815. [Google Scholar] [CrossRef]
- Russell, S.E.; Rachlin, A.B.; Smith, K.L.; Muschamp, J.; Berry, L.; Zhao, Z.; Chartoff, E.H. Sex Differences in Sensitivity to the Depressive-like Effects of the Kappa Opioid Receptor Agonist U-50488 in Rats. Biol. Psych. 2014, 76, 213–222. [Google Scholar] [CrossRef] [PubMed]
- Robles, C.F.; McMackin, M.Z.; Campi, K.L.; Doig, I.E.; Takahashi, E.Y.; Pride, M.C.; Trainor, B.C. Effects of kappa opioid receptors on conditioned place aversion and social interaction in males and females. Behav. Brain Res. 2014, 262, 84–93. [Google Scholar] [CrossRef] [PubMed]
- Huang, P.; Chen, C.; Cao, D.; Huang, M.; Liu-Chen, L.Y. Agonist-promoted kappa opioid receptor (KOR) phosphorylation has behavioural endpoint-dependent and sex-specific effects. Neuropharmacology 2022, 202, 108860. [Google Scholar] [CrossRef]
- Jacobson, M.L.; Wulf, H.A.; Tsuda, M.C.; Browne, C.A.; Lucki, I. Sex differences in the modulation of mouse nest building behavior by kappa opioid receptor signaling. Neuropharmacology 2020, 177, 108254. [Google Scholar] [CrossRef] [PubMed]
- Laman-Maharg, A.; Williams, A.V.; Zufelt, M.D.; Minie, V.A.; Ramos-Maciel, S.; Hao, R.; Sanchez, E.O.; Copeland, T.; Silverman, J.L.; Leigh, A.; et al. Sex Differences in the Effects of a Kappa Opioid Receptor Antagonist in the Forced Swim Test. Front. Pharmacol. 2018, 9, 93. [Google Scholar] [CrossRef] [PubMed]
- McReynolds, J.R.; Christianson, J.P.; Blacktop, J.M.; Mantsch, J.R. What does the Fos say? Using Fos-based approaches to understand the contribution of stress to substance use disorders. Neurobiol. Stress 2018, 9, 271–285. [Google Scholar] [CrossRef] [PubMed]
- Corish, P.; Tyler-Smith, C. Attenuation of green fluorescent protein half-life in mammalian cells. Protein Eng. 1999, 12, 1035–1040. [Google Scholar] [CrossRef]
- Kovács, K.J. c-Fos as a transcription factor: A stressful (re)view from a functional map. Neurochem. Int. 1998, 33, 287–297. [Google Scholar] [CrossRef]
- Paxinos, G.; Franklin, K.B.J. The Mouse Brain in Stereotaxic Coordinates; Academic Press: San Diego, CA, USA; London, UK, 2001. [Google Scholar]
- Cruz-Mendoza, F.; Jauregui-Huerta, F.; Aguilar-Delgadillo, A.; Garcia-Estrada, J.; Luquin, S. Immediate early gene c-fos in the brain: Focus on glial cells. Brain Sci. 2022, 12, 687. [Google Scholar] [CrossRef]
- Tamamaki, N.; Yanagawa, Y.; Tomioka, R.; Miyazaki, J.I.; Obata, K.; Kaneko, T. Green fluorescent protein expression and colocalization with calretinin, parvalbumin and somatostatin in the GAD67-GFP knock-in mouse. J. Comp. Neurol. 2003, 467, 60–79. [Google Scholar] [CrossRef]
- Barth, A.L.; Gerkin, R.C.; Dean, K.L. Alteration of Neuronal Firing Properties after In Vivo Experience in a FosGFP Transgenic Mouse. J. Neurosci. 2004, 24, 6466–6475. [Google Scholar] [CrossRef]
- Wang, Y.J.; Rasakham, K.; Huang, P.; Chudnovskaya, D.; Cowan, A.; Liu-Chen, L.Y. Sex difference in kappa-opioid receptor (KOPR)-mediated behaviors, brain region KOPR level and KOPR-mediated guanosine 5′-O-(3-[35S]thiotriphosphate) binding in the guinea pig. J. Pharmacol. Exp. Ther. 2011, 339, 438–450. [Google Scholar] [CrossRef]
- Vijay, A.; Wang, A.; Worhunsky, P.; Zheng, M.Q.; Nabulsi, N.; Ropchan, J.; Krishnan-Sarin, S.; Huang, Y.; Morris, E.D. PET imaging reveals sex differences in kappa opioid receptor availability in humans, in vivo. Am. J. Nucl. Med. Mol. Imaging 2016, 6, 205–214. [Google Scholar]
- Heijna, M.H.; Padt, M.; Hogenbook, F.; Portoghese, P.S.; Mulder, A.H.; Schoffelmeer, A.N. Opioid receptor-mediated inhibition of dopamine and acetylcholine release from slices of rat nucleus accumbens, olfactory tubercle and frontal cortex. Eur. J. Pharmacol. 1990, 181, 267–278. [Google Scholar] [CrossRef]
- Shippenberg, T.S.; Chefer, V.I.; Zapata, A.; Heidbreder, C.A. Modulation of the behavioral and neurochemical effects of psychostimulants by kappa-opioid receptor systems. Ann. N. Y. Acad. Sci. 2001, 937, 50–73. [Google Scholar] [CrossRef]
- Hjelmstad, G.O.; Fields, H.L. Kappa opioid receptor activation in the nucleus accumbens inhibits glutamate and GABA release through different mechanisms. J. Neurophysiol. 2003, 89, 2389–2395. [Google Scholar] [CrossRef] [PubMed]
- Britt, J.P.; McGehee, D.S. Presynaptic opioid and nicotinic receptor modulation of dopamine overflow in the nucleus accumbens. J. Neurosci. 2008, 28, 1672–1681. [Google Scholar] [CrossRef]
- Tejeda, H.A.; Counotte, D.S.; Oh, E.; Ramamoorthy, S.; Schultz-Kuszak, K.N.; Backman, C.M.; Chefer, V.; O’Donnell, P.; Shippenberg, T.S. Prefrontal cortical kappa-opioid receptor modulation of local neurotransmission and conditioned place aversion. Neuropsychopharmacology 2013, 38, 1770–1779. [Google Scholar] [CrossRef]
- Tejeda, H.A.; Wu, J.; Kornspun, A.R.; Pignatelli, M.; Kashtelyan, V.; Krashes, M.J.; Lowell, B.B.; Carlezon, W.A.; Bonci, A. Pathway- and Cell-Specific Kappa- Opioid Receptor Modulation of Excitation-Inhibition Balance differentially gates D1 and D2 accumbens neuron activity. Neuron 2017, 93, 147–163. [Google Scholar] [CrossRef]
- Eriksson, P.S.; Nilsson, M.; Wagberg, M.; Hansson, E.; Ronnback, L. Kappa-opioid receptors on astrocytes stimulate L-type Ca2+ channels. Neuroscience 1993, 54, 401–407. [Google Scholar] [CrossRef] [PubMed]
- Ruzicka, B.B.; Fox, C.A.; Thompson, R.C.; Meng, F.; Watson, S.J.; Akil, H. Primary astroglial cultures derived from several rat brain regions differentially express mu, delta and kappa opioid receptor mRNA. Brain Res. Mol. Brain Res. 1995, 34, 209–220. [Google Scholar] [CrossRef]
- Gurwell, J.A.; Duncan, M.J.; Maderspach, K.; Steine-Martin, A.; Elde, R.P.; Hauser, K.F. Kappa-opioid receptor expression defines a phenotypically distinct subpopulation of astroglia: Relationship to Ca2+ mobilization, development, and the antiproliferative effect of opioids. Brain Res. 1996, 737, 175–187. [Google Scholar] [CrossRef] [PubMed]
- Bruchas, M.R.; Macey, T.A.; Lowe, J.D.; Chavkin, C. Kappa opioid receptor activation of p38 MAPK is GRK3- and arrestin-dependent in neurons and astrocytes. J. Biol. Chem. 2006, 281, 18081–18089. [Google Scholar] [CrossRef] [PubMed]
- Ons, S.; Marti, O.; Armario, A. Stress-induced activation of the immediate early gene Arc (activity-regulated cytoskeleton-associated protein) is restricted to telencephalic areas in the rat brain: Relationship to cFos mRNA. J. Neurochem. 2004, 89, 1111–1118. [Google Scholar] [CrossRef] [PubMed]
- Funk, D.; Li, Z.; Le, A.D. Effects of environmental and pharmacological stressors on c-Fos and corticotropin-releasing factor mRNA in rat brain: Relationship to the reinstatement of alcohol seeking. Neuroscience 2006, 138, 236–243. [Google Scholar] [CrossRef]
- Briand, L.A.; Vassoler, F.M.; Pierce, R.C.; Valentino, R.J.; Blendy, J.A. Ventral tegmental afferents in stress-induced reinstatement: The role of cAMP response element-binding protein. J. Neurosci. 2010, 30, 16149–16159. [Google Scholar] [CrossRef] [PubMed]
- Chavkin, C.; Goldstein, A. Demonstration of a specific dynorphin receptor in guinea pig ileum myenteric plexus. Nature 1981, 291, 591–593. [Google Scholar] [CrossRef]
- McEwen, B.S.; Bowles, N.P.; Gray, J.D.; Hill, M.N.; Hunter, R.G.; Karatsoreos, I.N.; Nasca, C. Mechanisms of stress in the brain. Nat. Neurosci. 2015, 18, 1353–1363. [Google Scholar] [CrossRef]
- Escudero-Lara, A.; Cabanero, D.; Maldonado, R. Kappa opioid receptor modulation of endometriosis pain in mice. Neuropharmacology 2021, 195, 108677. [Google Scholar] [CrossRef]
- Carr, G.V.; Bangasser, D.A.; Bethea, T.; Young, M.; Valentino, R.J.; Lucki, I. Antidepressant-like effects of kappa-opioid receptor antagonists in Wistar Kyoto rats. Neuropsychopharmacology 2010, 35, 752–763. [Google Scholar] [CrossRef]
- Newton, S.S.; Thome, J.; Wallace, T.L.; Shirayama, Y.; Schlesinger, L.; Sakai, N.; Chen, J.; Neve, R.; Nestler, E.J.; Duman, R.S. Inhibition of cAMP response element-binding protein or dynorphin in the nucleus accumbens produces an antidepressant-like effect. J. Neurosci. 2002, 22, 10883–10890. [Google Scholar] [CrossRef]
- Shoda, T.; Fukuda, K.; Uga, H.; Mima, H.; Morikawa, H. Activation of mu-opioid receptor induces expression of c-Fos and junB via mitogen-activated protein kinase cascade. Anesthesiology 2001, 95, 983–989. [Google Scholar] [CrossRef]
- Varastehmoradi, B.; Smith, K.L.; Kaastrup Muller, H.; Elfving, B.; Sanchez, C.; Wegener, G. Kappa opioid activation changes protein profiles in different regions of the brain relevant to depression. Eur. Neuropsychopharmacol. 2023, 72, 9–17. [Google Scholar] [CrossRef]
- Casello, S.M.; Flores, R.J.; Yarur, H.E.; Wang, H.; Awanyai, M.; Arenivar, M.A.; Jaime-Lara, R.B.; Bravo-Rivera, H.; Tejeda, H.A. Neuropeptide system regulation of prefrontal cortex circuitry: Implications for neuropsychiatric disorders. Front. Neural Circuits 2022, 16, 796443. [Google Scholar] [CrossRef]
- Sohn, J.; Hioki, H.; Okamoto, S.; Kaneko, T. Preprodynorphin-expressing neurons constitute a large subgroup of somatostatin-expressing GABAergic interneurons in the mouse neocortex. J. Comp. Neurol. 2014, 522, 1506–1526. [Google Scholar] [CrossRef] [PubMed]
- McEwen, B.S. Stress, sex, and neural adaptation to a changing environment: Mechanisms of neuronal remodeling. Ann. N. Y. Acad. Sci. 2010, 1204, 38–59. [Google Scholar] [CrossRef]
- Endoh, T.; Matsuura, H.; Tanaka, C.; Nagase, H. Nor-binaltorphimine: A potent and selective kappa-opioid receptor antagonist with long-lasting activity in vivo. Arch. Int. Pharmacodyn. Ther. 1992, 316, 30–42. [Google Scholar] [PubMed]
- Biland-Bleuel, A.; Rech, J.; de Carli, S.; Holsboer, F.; Reul, J.M.H.M. Forced swimming evoked a biphasic response in CREB phosphorylation in extrahypothalamic limbic and neocortical brain structures in the rat. Eur. J. Neurosci. 2002, 15, 1048–1060. [Google Scholar] [CrossRef]
- Karandrea, D.; Kittas, C.; Kitraki, E. Forced Swimming Differentially Affects Male and Female Brain Corticosteroid Receptors. Neuroendocrinology 2002, 75, 217–226. [Google Scholar] [CrossRef] [PubMed]
- Marrone, G.F.; Grinnell, S.G.; Lu, Z.; Rossi, G.C.; Le Rouzic, V.L.; Xu, J.; Majumdar, S.; Pan, Y.X.; Pasternak, G.W. Truncated Mu Opioid GPCR Variant Involvement in Opioid-Dependent and Opioid-Independent Pain Modulatory Systems within the CNS. Proc. Natl. Acad. Sci. USA 2016, 113, 3663–3668. [Google Scholar] [CrossRef]
- Sadler, A.M.; Bailey, S.J. Validation of a refined technique for taking repeated blood samples from juvenile and adult mice. Lab. Anim. 2013, 47, 316–319. [Google Scholar] [CrossRef]
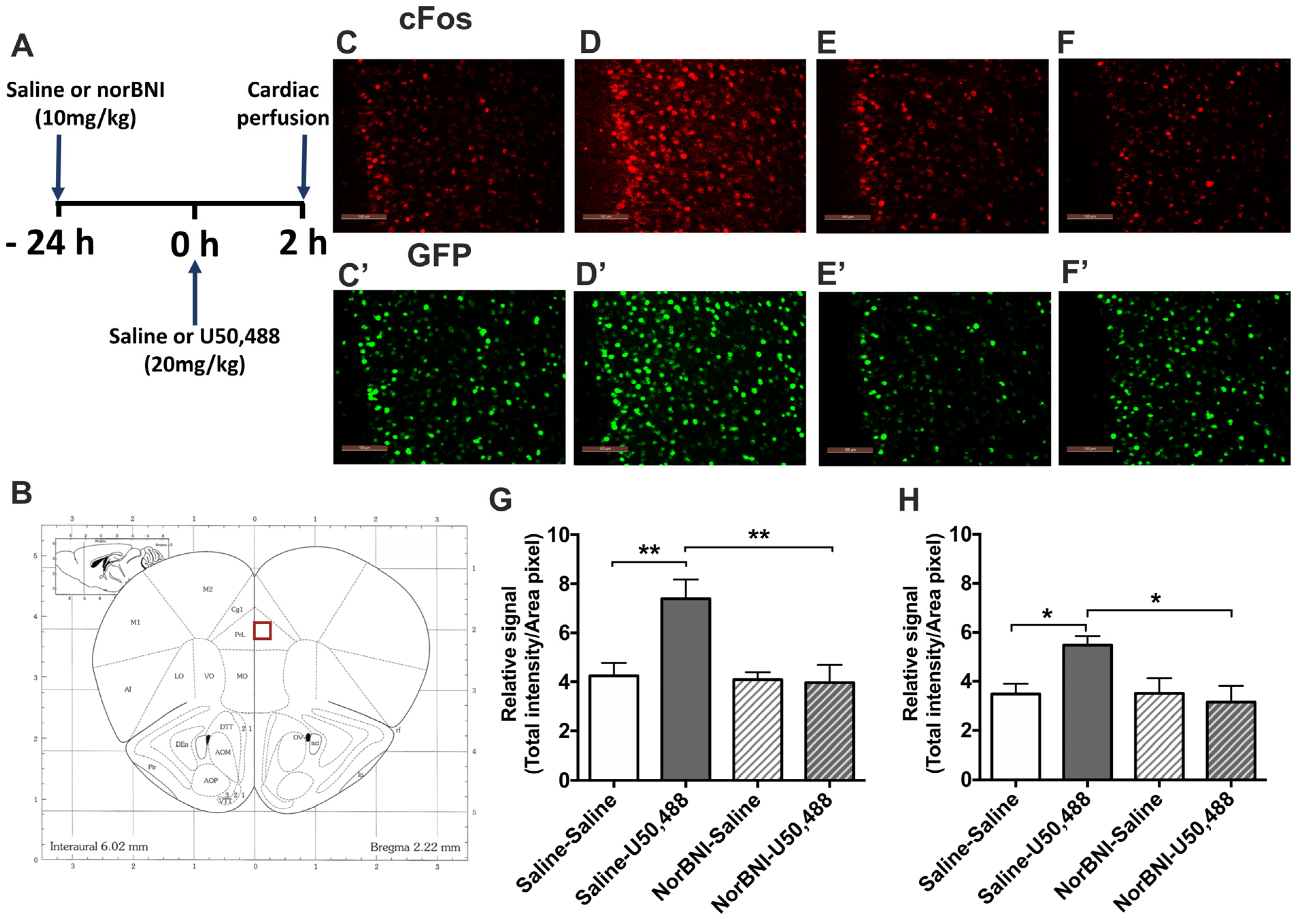
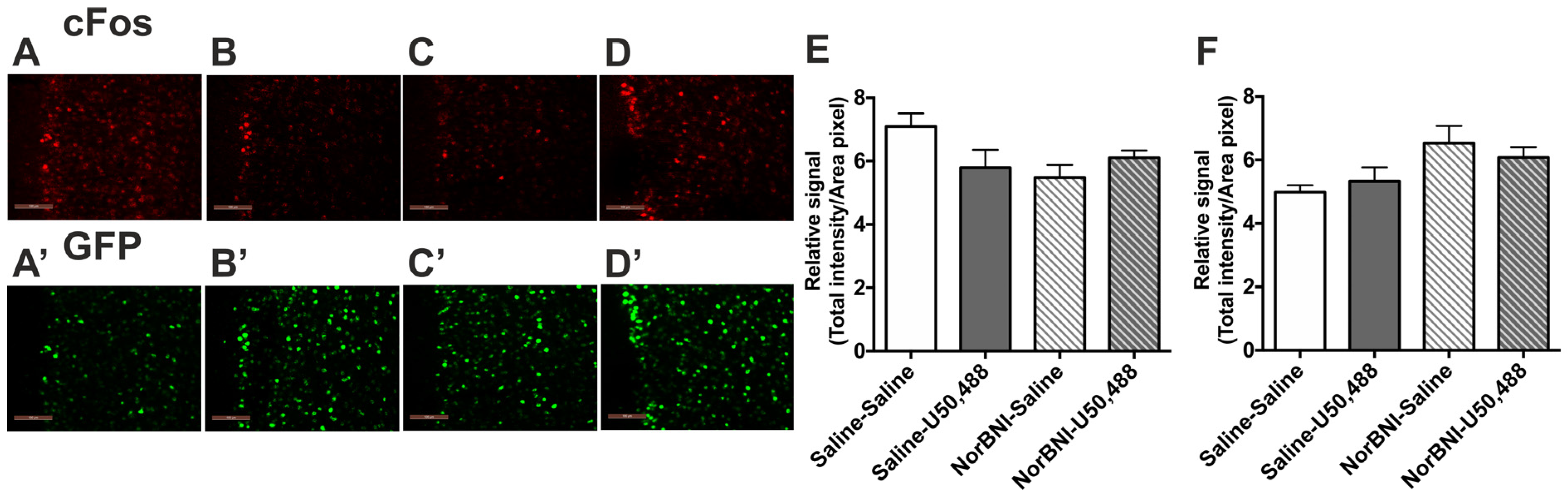







| Male | U50,488 | Forced Swim Stress | ||||||
|---|---|---|---|---|---|---|---|---|
| c-Fos | c-Fos-GFP | c-Fos | c-Fos-GFP | |||||
| Saline | norBNI | Saline | norBNI | Saline | norBNI | Saline | norBNI | |
| PFCx | ↑↑ | ** | ↑ | * | ↑↑ | ns | ↑↑ | ns |
| NAc | ↑↑ | ** | ↑ | ** | ↑↑ | * | ↑↑ | ns |
| CA1 | ↑ | * | ↑↑ | ** | ns | ns | ns | ns |
| DG | ns | ns | ns | Ns | ns | ns | ns | ns |
| CeA | ↑ | * | ↑ | * | ns | ns | ns | ns |
| BLA | ↑ | * | ↑↑ | ** | ↑ | ns | ↑ | ns |
| Female | U50,488 | Forced swim stress | ||||||
| c-Fos | c-Fos-GFP | c-Fos | c-Fos-GFP | |||||
| Saline | norBNI | Saline | norBNI | Saline | norBNI | Saline | norBNI | |
| PFCx | ns | ns | ns | ns | ns | ns | ns | ns |
| NAc | ns | ns | ns | ns | ↑↑ | ns | ↑ | ns |
| CA1 | ↑↑↑ | ** | ns | ns | ns | ns | ns | ns |
| DG | ns | ns | ns | ns | ns | ns | ns | ns |
| CeA | ↑ | * | ↑↑ | * | ns | ns | ns | ns |
| BLA | ↑↑↑ | ** | ↑↑ | * | ↑↑↑ | ns | ↑ | ns |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ma, Q.; Wonnacott, S.; Bailey, S.J.; Bailey, C.P. Sex Differences in Brain Region-Specific Activation of c-Fos following Kappa Opioid Receptor Stimulation or Acute Stress in Mice. Int. J. Mol. Sci. 2023, 24, 15098. https://doi.org/10.3390/ijms242015098
Ma Q, Wonnacott S, Bailey SJ, Bailey CP. Sex Differences in Brain Region-Specific Activation of c-Fos following Kappa Opioid Receptor Stimulation or Acute Stress in Mice. International Journal of Molecular Sciences. 2023; 24(20):15098. https://doi.org/10.3390/ijms242015098
Chicago/Turabian StyleMa, Qianhan, Susan Wonnacott, Sarah J. Bailey, and Christopher P. Bailey. 2023. "Sex Differences in Brain Region-Specific Activation of c-Fos following Kappa Opioid Receptor Stimulation or Acute Stress in Mice" International Journal of Molecular Sciences 24, no. 20: 15098. https://doi.org/10.3390/ijms242015098






