Exosomes Highlight Future Directions in the Treatment of Acute Kidney Injury
Abstract
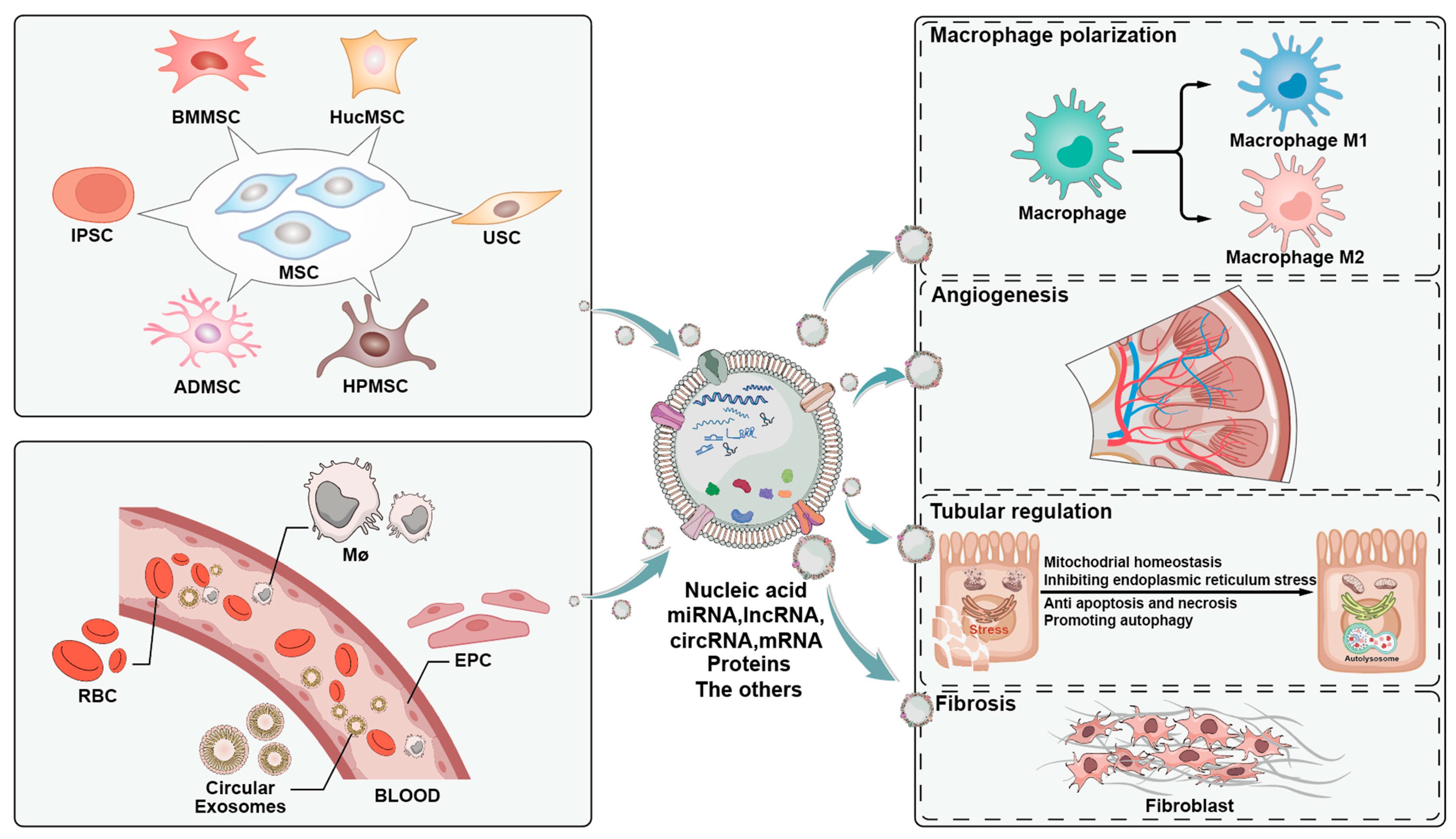
:1. Introduction
2. Role of Exosomes of Different Origins in AKI
2.1. Role of Exosomes of MSCs in AKI
2.2. Role of Exosomes from Other Sources in AKI
3. Role of Exosome Content in AKI
3.1. Reparative Effects
3.2. Damaging Effects
4. Exosomes and Their Contents Act as Biomarkers for AKI
5. Engineering Exosomes
5.1. Isolation and Enrichment
5.2. Targeted Therapy
5.3. Exosome Modifications
6. Future Directions
6.1. Accelerating Clinical Translation
6.2. Elucidating Pathways of Reaching the Renal Tubules
6.3. Exosomes and Multi-Omics Studies
7. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Levey, A.S.; James, M.T. Acute Kidney Injury. Ann. Intern. Med. 2017, 167, ITC66–ITC80. [Google Scholar] [CrossRef]
- Xu, X.; Nie, S.; Liu, Z.; Chen, C.; Xu, G.; Zha, Y.; Qian, J.; Liu, B.; Han, S.; Xu, A.; et al. Epidemiology and Clinical Correlates of AKI in Chinese Hospitalized Adults. Clin. J. Am. Soc. Nephrol. 2015, 10, 1510–1518. [Google Scholar] [CrossRef] [PubMed]
- Zarbock, A.; Nadim, M.K.; Pickkers, P.; Gomez, H.; Bell, S.; Joannidis, M.; Kashani, K.; Koyner, J.L.; Pannu, N.; Meersch, M.; et al. Sepsis-associated acute kidney injury: Consensus report of the 28th Acute Disease Quality Initiative workgroup. Nat. Rev. Nephrol. 2023, 19, 401–417. [Google Scholar] [CrossRef]
- Messerer, D.A.C.; Halbgebauer, R.; Nilsson, B.; Pavenstädt, H.; Radermacher, P.; Huber-Lang, M. Immunopathophysiology of trauma-related acute kidney injury. Nat. Rev. Nephrol. 2021, 17, 91–111. [Google Scholar] [CrossRef] [PubMed]
- Pickkers, P.; Darmon, M.; Hoste, E.; Joannidis, M.; Legrand, M.; Ostermann, M.; Prowle, J.R.; Schneider, A.; Schetz, M. Acute kidney injury in the critically ill: An updated review on pathophysiology and management. Intensive Care Med. 2021, 47, 835–850. [Google Scholar] [CrossRef] [PubMed]
- Peerapornratana, S.; Manrique-Caballero, C.L.; Gómez, H.; Kellum, J.A. Acute kidney injury from sepsis: Current concepts, epidemiology, pathophysiology, prevention and treatment. Kidney Int. 2019, 96, 1083–1099. [Google Scholar] [CrossRef]
- Grange, C.; Bussolati, B. Extracellular vesicles in kidney disease. Nat. Rev. Nephrol. 2022, 18, 499–513. [Google Scholar] [CrossRef] [PubMed]
- Jeppesen, D.K.; Fenix, A.M.; Franklin, J.L.; Higginbotham, J.N.; Zhang, Q.; Zimmerman, L.J.; Liebler, D.C.; Ping, J.; Liu, Q.; Evans, R.; et al. Reassessment of Exosome Composition. Cell 2019, 177, 428–445. [Google Scholar] [CrossRef]
- D’Acunzo, P.; Pérez-González, R.; Kim, Y.; Hargash, T.; Miller, C.; Alldred, M.J.; Erdjument-Bromage, H.; Penikalapati, S.C.; Pawlik, M.; Saito, M.; et al. Mitovesicles are a novel population of extracellular vesicles of mitochondrial origin altered in Down syndrome. Sci. Adv. 2021, 7, eabe5085. [Google Scholar] [CrossRef]
- Kim, H.Y.; Kwon, S.; Um, W.; Shin, S.; Kim, C.H.; Park, J.H.; Kim, B.-S. Functional Extracellular Vesicles for Regenerative Medicine. Small 2022, 18, e2106569. [Google Scholar] [CrossRef]
- Yang, B.; Chen, Y.; Shi, J. Exosome Biochemistry and Advanced Nanotechnology for Next-Generation Theranostic Platforms. Adv. Mater. 2019, 31, e1802896. [Google Scholar] [CrossRef] [PubMed]
- Karpman, D.; Ståhl, A.-L.; Arvidsson, I. Extracellular vesicles in renal disease. Nat. Rev. Nephrol. 2017, 13, 545–562. [Google Scholar] [CrossRef] [PubMed]
- Li, S.-R.; Man, Q.-W.; Gao, X.; Lin, H.; Wang, J.; Su, F.-C.; Wang, H.-Q.; Bu, L.-L.; Liu, B.; Chen, G. Tissue-derived extracellular vesicles in cancers and non-cancer diseases: Present and future. J. Extracell. Vesicles 2021, 10, e12175. [Google Scholar] [CrossRef]
- Mulcahy, L.A.; Pink, R.C.; Carter, D.R.F. Routes and mechanisms of extracellular vesicle uptake. J. Extracell. Vesicles 2014, 3, 24641. [Google Scholar] [CrossRef] [PubMed]
- Hough, K.P.; Trevor, J.L.; Strenkowski, J.G.; Wang, Y.; Chacko, B.K.; Tousif, S.; Chanda, D.; Steele, C.; Antony, V.B.; Dokland, T.; et al. Exosomal transfer of mitochondria from airway myeloid-derived regulatory cells to T cells. Redox. Biol. 2018, 18, 54–64. [Google Scholar] [CrossRef] [PubMed]
- Oh, S.; Kwon, S.-H. Extracellular Vesicles in Acute Kidney Injury and Clinical Applications. Int. J. Mol. Sci. 2021, 22, 8913. [Google Scholar] [CrossRef]
- Huang, Y.; Yang, L. Mesenchymal stem cells and extracellular vesicles in therapy against kidney diseases. Stem Cell Res. Ther. 2021, 12, 219. [Google Scholar] [CrossRef]
- Abumoawad, A.; Saad, A.; Ferguson, C.M.; Eirin, A.; Herrmann, S.M.; Hickson, L.J.; Goksu, B.B.; Bendel, E.; Misra, S.; Glockner, J.; et al. In a Phase 1a escalating clinical trial, autologous mesenchymal stem cell infusion for renovascular disease increases blood flow and the glomerular filtration rate while reducing inflammatory biomarkers and blood pressure. Kidney Int. 2020, 97, 793–804. [Google Scholar] [CrossRef]
- Matsui, F.; Babitz, S.K.; Rhee, A.; Hile, K.L.; Zhang, H.; Meldrum, K.K. Mesenchymal stem cells protect against obstruction-induced renal fibrosis by decreasing STAT3 activation and STAT3-dependent MMP-9 production. Am. J. Physiol. Ren. Physiol. 2017, 312, F25–F32. [Google Scholar] [CrossRef]
- Liu, C.; Wang, J.; Hu, J.; Fu, B.; Mao, Z.; Zhang, H.; Cai, G.; Chen, X.; Sun, X. Extracellular vesicles for acute kidney injury in preclinical rodent models: A meta-analysis. Stem Cell Res. Ther. 2020, 11, 11. [Google Scholar] [CrossRef]
- Dominguez, J.M.; Dominguez, J.H.; Xie, D.; Kelly, K.J. Human extracellular microvesicles from renal tubules reverse kidney ischemia-reperfusion injury in rats. PLoS ONE 2018, 13, e0202550. [Google Scholar] [CrossRef] [PubMed]
- Gurunathan, S.; Kang, M.-H.; Jeyaraj, M.; Qasim, M.; Kim, J.-H. Review of the Isolation, Characterization, Biological Function, and Multifarious Therapeutic Approaches of Exosomes. Cells 2019, 8, 307. [Google Scholar] [CrossRef] [PubMed]
- Kalluri, R.; LeBleu, V.S. The biology, function, and biomedical applications of exosomes. Science 2020, 367, eaau6977. [Google Scholar] [CrossRef] [PubMed]
- Zhu, G.; Pei, L.; Lin, F.; Yin, H.; Li, X.; He, W.; Liu, N.; Gou, X. Exosomes from human-bone-marrow-derived mesenchymal stem cells protect against renal ischemia/reperfusion injury via transferring miR-199a-3p. J. Cell. Physiol. 2019, 234, 23736–23749. [Google Scholar] [CrossRef]
- Menon, S.; Goldstein, S.L.; Mottes, T.; Fei, L.; Kaddourah, A.; Terrell, T.; Arnold, P.; Bennett, M.R.; Basu, R.K. Urinary biomarker incorporation into the renal angina index early in intensive care unit admission optimizes acute kidney injury prediction in critically ill children: A prospective cohort study. Nephrol. Dial. Transpl. 2016, 31, 586–594. [Google Scholar] [CrossRef]
- Heller, F.; Frischmann, S.; Grünbaum, M.; Zidek, W.; Westhoff, T.H. Urinary calprotectin and the distinction between prerenal and intrinsic acute kidney injury. Clin. J. Am. Soc. Nephrol. 2011, 6, 2347–2355. [Google Scholar] [CrossRef]
- Wang, S.-Y.; Xu, Y.; Hong, Q.; Chen, X.-M.; Cai, G.-Y. Mesenchymal stem cells ameliorate cisplatin-induced acute kidney injury via let-7b-5p. Cell Tissue Res. 2023, 392, 517–533. [Google Scholar] [CrossRef]
- Barreto, E.F.; Rule, A.D.; Murad, M.H.; Kashani, K.B.; Lieske, J.C.; Erwin, P.J.; Steckelberg, J.M.; Gajic, O.; Reid, J.M.; Kane-Gill, S.L. Prediction of the Renal Elimination of Drugs with Cystatin C vs. Creatinine: A Systematic Review. Mayo Clin. Proc. 2019, 94, 500–514. [Google Scholar] [CrossRef]
- Basu, R.K.; Kaddourah, A.; Goldstein, S.L. Assessment of a renal angina index for prediction of severe acute kidney injury in critically ill children: A multicentre, multinational, prospective observational study. Lancet Child Adolesc. Health 2018, 2, 112–120. [Google Scholar] [CrossRef]
- De Miguel-Gómez, L.; Ferrero, H.; López-Martínez, S.; Campo, H.; López-Pérez, N.; Faus, A.; Hervás, D.; Santamaría, X.; Pellicer, A.; Cervelló, I. Stem cell paracrine actions in tissue regeneration and potential therapeutic effect in human endometrium: A retrospective study. BJOG 2020, 127, 551–560. [Google Scholar] [CrossRef]
- Huang, T.-Y.; Chien, M.-S.; Su, W.-T. Therapeutic Potential of Pretreatment with Exosomes Derived from Stem Cells from the Apical Papilla against Cisplatin-Induced Acute Kidney Injury. Int. J. Mol. Sci. 2022, 23, 5721. [Google Scholar] [CrossRef]
- Zhang, Q.; Lai, D. Application of human amniotic epithelial cells in regenerative medicine: A systematic review. Stem Cell Res. Ther. 2020, 11, 439. [Google Scholar] [CrossRef] [PubMed]
- Ren, Y.; Chen, Y.; Zheng, X.; Wang, H.; Kang, X.; Tang, J.; Qu, L.; Shao, X.; Wang, S.; Li, S.; et al. Human amniotic epithelial cells ameliorate kidney damage in ischemia-reperfusion mouse model of acute kidney injury. Stem Cell Res. Ther. 2020, 11, 410. [Google Scholar] [CrossRef]
- Kang, X.; Chen, Y.; Xin, X.; Liu, M.; Ma, Y.; Ren, Y.; Ji, J.; Yu, Q.; Qu, L.; Wang, S.; et al. Human Amniotic Epithelial Cells and Their Derived Exosomes Protect Against Cisplatin-Induced Acute Kidney Injury without Compromising Its Antitumor Activity in Mice. Front. Cell Dev. Biol. 2021, 9, 752053. [Google Scholar] [CrossRef] [PubMed]
- Chi, D.; Chen, Y.; Xiang, C.; Yao, W.; Wang, H.; Zheng, X.; Xu, D.; Li, N.; Xie, M.; Wang, S.; et al. Human Amnion Epithelial Cells and Their Derived Exosomes Alleviate Sepsis-Associated Acute Kidney Injury via Mitigating Endothelial Dysfunction. Front. Med. 2022, 9, 829606. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Wang, N.; Huang, Y.; Li, Y.; Li, G.; Lin, Y.; Atala, A.J.; Hou, J.; Zhao, W. Extracellular vesicles from three dimensional culture of human placental mesenchymal stem cells ameliorated renal ischemia/reperfusion injury. Int. J. Artif. Organs 2022, 45, 181–192. [Google Scholar] [CrossRef]
- Zhang, W.; Zhang, J.; Huang, H. Exosomes from adipose-derived stem cells inhibit inflammation and oxidative stress in LPS-acute kidney injury. Exp. Cell Res. 2022, 420, 113332. [Google Scholar] [CrossRef]
- Lin, K.-C.; Yip, H.-K.; Shao, P.-L.; Wu, S.-C.; Chen, K.-H.; Chen, Y.-T.; Yang, C.-C.; Sun, C.-K.; Kao, G.-S.; Chen, S.-Y.; et al. Combination of adipose-derived mesenchymal stem cells (ADMSC) and ADMSC-derived exosomes for protecting kidney from acute ischemia-reperfusion injury. Int. J. Cardiol. 2016, 216, 173–185. [Google Scholar] [CrossRef]
- Gao, F.; Zuo, B.; Wang, Y.; Li, S.; Yang, J.; Sun, D. Protective function of exosomes from adipose tissue-derived mesenchymal stem cells in acute kidney injury through SIRT1 pathway. Life Sci. 2020, 255, 117719. [Google Scholar] [CrossRef]
- Zhang, J.B.; Wang, X.Q.; Lu, G.; Huang, H.S.; Xu, S.Y. Adipose-derived mesenchymal stem cells therapy for acute kidney injury induced by ischemia-reperfusion in a rat model. Clin. Exp. Pharmacol. Physiol. 2017, 44, 1232–1240. [Google Scholar] [CrossRef]
- Liu, W.; Hu, C.; Zhang, B.; Li, M.; Deng, F.; Zhao, S. Exosomal microRNA-342-5p secreted from adipose-derived mesenchymal stem cells mitigates acute kidney injury in sepsis mice by inhibiting TLR9. Biol. Proced. Online 2023, 25, 10. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Wang, W.; He, X.; Liao, Z.; Aierken, A.; Hua, J.; Wang, Y.; Lu, D.; Zhang, S. Rapid recovery of male cats with postrenal acute kidney injury by treating with allogeneic adipose mesenchymal stem cell-derived extracellular vesicles. Stem Cell Res. Ther. 2022, 13, 379. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Xu, H.; Xu, W.; Wang, B.; Wu, H.; Tao, Y.; Zhang, B.; Wang, M.; Mao, F.; Yan, Y.; et al. Exosomes released by human umbilical cord mesenchymal stem cells protect against cisplatin-induced renal oxidative stress and apoptosis in vivo and in vitro. Stem Cell Res. Ther. 2013, 4, 34. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Jia, H.; Zhang, B.; Wang, J.; Ji, C.; Zhu, X.; Yan, Y.; Yin, L.; Yu, J.; Qian, H.; et al. Pre-incubation with hucMSC-exosomes prevents cisplatin-induced nephrotoxicity by activating autophagy. Stem Cell Res. Ther. 2017, 8, 75. [Google Scholar] [CrossRef] [PubMed]
- Viñas, J.L.; Burger, D.; Zimpelmann, J.; Haneef, R.; Knoll, W.; Campbell, P.; Gutsol, A.; Carter, A.; Allan, D.S.; Burns, K.D. Transfer of microRNA-486-5p from human endothelial colony forming cell-derived exosomes reduces ischemic kidney injury. Kidney Int. 2016, 90, 1238–1250. [Google Scholar] [CrossRef]
- Cao, J.-Y.; Wang, B.; Tang, T.-T.; Wen, Y.; Li, Z.-L.; Feng, S.-T.; Wu, M.; Liu, D.; Yin, D.; Ma, K.-L.; et al. Exosomal miR-125b-5p deriving from mesenchymal stem cells promotes tubular repair by suppression of p53 in ischemic acute kidney injury. Theranostics 2021, 11, 5248–5266. [Google Scholar] [CrossRef]
- Ji, C.; Zhang, J.; Zhu, Y.; Shi, H.; Yin, S.; Sun, F.; Wang, Q.; Zhang, L.; Yan, Y.; Zhang, X.; et al. Exosomes derived from hucMSC attenuate renal fibrosis through CK1δ/β-TRCP-mediated YAP degradation. Cell Death Dis. 2020, 11, 327. [Google Scholar] [CrossRef]
- Huang, J.; Cao, H.; Cui, B.; Ma, X.; Gao, L.; Yu, C.; Shen, F.; Yang, X.; Liu, N.; Qiu, A.; et al. Mesenchymal Stem Cells-Derived Exosomes Ameliorate Ischemia/Reperfusion Induced Acute Kidney Injury in a Porcine Model. Front. Cell Dev. Biol. 2022, 10, 899869. [Google Scholar] [CrossRef]
- Takebe, T.; Sekine, K.; Enomura, M.; Koike, H.; Kimura, M.; Ogaeri, T.; Zhang, R.-R.; Ueno, Y.; Zheng, Y.-W.; Koike, N.; et al. Vascularized and functional human liver from an iPSC-derived organ bud transplant. Nature 2013, 499, 481–484. [Google Scholar] [CrossRef]
- Lim, S.W.; Kim, K.W.; Kim, B.M.; Shin, Y.J.; Luo, K.; Quan, Y.; Cui, S.; Ko, E.J.; Chung, B.H.; Yang, C.W. Alleviation of renal ischemia/reperfusion injury by exosomes from induced pluripotent stem cell-derived mesenchymal stem cells. Korean J. Intern. Med. 2022, 37, 411–424. [Google Scholar] [CrossRef]
- Tian, S.-F.; Jiang, Z.-Z.; Liu, Y.-M.; Niu, X.; Hu, B.; Guo, S.-C.; Wang, N.-S.; Wang, Y. Human urine-derived stem cells contribute to the repair of ischemic acute kidney injury in rats. Mol. Med. Rep. 2017, 16, 5541–5548. [Google Scholar] [CrossRef]
- Li, X.; Liao, J.; Su, X.; Li, W.; Bi, Z.; Wang, J.; Su, Q.; Huang, H.; Wei, Y.; Gao, Y.; et al. Human urine-derived stem cells protect against renal ischemia/reperfusion injury in a rat model via exosomal miR-146a-5p which targets IRAK1. Theranostics 2020, 10, 9561–9578. [Google Scholar] [CrossRef]
- Chen, Y.; Zhang, C.; Du, Y.; Yang, X.; Liu, M.; Yang, W.; Lei, G.; Wang, G. Exosomal transfer of microRNA-590-3p between renal tubular epithelial cells after renal ischemia-reperfusion injury regulates autophagy by targeting TRAF6. Chin. Med. J. 2022, 135, 2467–2477. [Google Scholar] [CrossRef]
- Li, L.; Wang, R.; Jia, Y.; Rong, R.; Xu, M.; Zhu, T. Exosomes Derived from Mesenchymal Stem Cells Ameliorate Renal Ischemic-Reperfusion Injury Through Inhibiting Inflammation and Cell Apoptosis. Front. Med. 2019, 6, 269. [Google Scholar] [CrossRef] [PubMed]
- Zou, L.; Ma, X.; Wu, B.; Chen, Y.; Xie, D.; Peng, C. Protective effect of bone marrow mesenchymal stem cell-derived exosomes on cardiomyoblast hypoxia-reperfusion injury through the miR-149/let-7c/Faslg axis. Free Radic. Res. 2020, 54, 722–731. [Google Scholar] [CrossRef]
- Zhou, Y.; Li, P.; Goodwin, A.J.; Cook, J.A.; Halushka, P.V.; Chang, E.; Fan, H. Exosomes from Endothelial Progenitor Cells Improve the Outcome of a Murine Model of Sepsis. Mol. Ther. 2018, 26, 1375–1384. [Google Scholar] [CrossRef] [PubMed]
- Wan, Y.; Yu, Y.; Yu, C.; Luo, J.; Wen, S.; Shen, L.; Wei, G.; Hua, Y. Human umbilical cord mesenchymal stem cell exosomes alleviate acute kidney injury by inhibiting pyroptosis in rats and NRK-52E cells. Ren. Fail. 2023, 45, 2221138. [Google Scholar] [CrossRef] [PubMed]
- Tsugawa-Shimizu, Y.; Fujishima, Y.; Kita, S.; Minami, S.; Sakaue, T.-A.; Nakamura, Y.; Okita, T.; Kawachi, Y.; Fukada, S.; Namba-Hamano, T.; et al. Increased vascular permeability and severe renal tubular damage after ischemia-reperfusion injury in mice lacking adiponectin or T-cadherin. Am. J. Physiol. Endocrinol. Metab. 2021, 320, E179–E190. [Google Scholar] [CrossRef]
- Zhang, Y.; Huang, H.; Liu, W.; Liu, S.; Wang, X.Y.; Diao, Z.L.; Zhang, A.H.; Guo, W.; Han, X.; Dong, X.; et al. Endothelial progenitor cells-derived exosomal microRNA-21-5p alleviates sepsis-induced acute kidney injury by inhibiting RUNX1 expression. Cell Death Dis. 2021, 12, 335. [Google Scholar] [CrossRef]
- Pat, B.; Oh, J.-Y.; Masjoan Juncos, J.X.; Powell, P.C.; Collawn, J.F.; Patel, R.P.; Dell’Italia, L.J. Red blood cell exosome hemoglobin content increases after cardiopulmonary bypass and mediates acute kidney injury in an animal model. J. Thorac. Cardiovasc. Surg. 2022, 164, e289–e308. [Google Scholar] [CrossRef]
- Xiang, H.; Xu, Z.; Zhang, C.; Xiong, J. Macrophage-derived exosomes mediate glomerular endothelial cell dysfunction in sepsis-associated acute kidney injury. Cell Biosci. 2023, 13, 46. [Google Scholar] [CrossRef]
- Xu, X.; Yang, J.; Li, N.; Wu, R.; Tian, H.; Song, H.; Wang, H. Role of Endothelial Progenitor Cell Transplantation in Rats with Sepsis. Transpl. Proc. 2015, 47, 2991–3001. [Google Scholar] [CrossRef]
- Pan, T.; Jia, P.; Chen, N.; Fang, Y.; Liang, Y.; Guo, M.; Ding, X. Delayed Remote Ischemic Preconditioning ConfersRenoprotection against Septic Acute Kidney Injury via Exosomal miR-21. Theranostics 2019, 9, 405–423. [Google Scholar] [CrossRef]
- Ren, G.-L.; Zhu, J.; Li, J.; Meng, X.-M. Noncoding RNAs in acute kidney injury. J. Cell Physiol. 2019, 234, 2266–2276. [Google Scholar] [CrossRef] [PubMed]
- Collino, F.; Bruno, S.; Incarnato, D.; Dettori, D.; Neri, F.; Provero, P.; Pomatto, M.; Oliviero, S.; Tetta, C.; Quesenberry, P.J.; et al. AKI Recovery Induced by Mesenchymal Stromal Cell-Derived Extracellular Vesicles Carrying MicroRNAs. J. Am. Soc. Nephrol. 2015, 26, 2349–2360. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; He, W.; Zheng, D.; He, Q.; Tan, M.; Jin, J. Exosomal-miR-1184 derived from mesenchymal stem cells alleviates cisplatin-associated acute kidney injury. Mol. Med. Rep. 2021, 24, 795. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.; Zhu, Y.; Li, Y.; Liu, W.; Yin, L.; Yin, S.; Ji, C.; Hu, Y.; Wang, Q.; Zhou, X.; et al. Human umbilical cord mesenchymal stem cell exosomes alleviate sepsis-associated acute kidney injury via regulating microRNA-146b expression. Biotechnol. Lett. 2020, 42, 669–679. [Google Scholar] [CrossRef] [PubMed]
- Viñas, J.L.; Spence, M.; Porter, C.J.; Douvris, A.; Gutsol, A.; Zimpelmann, J.A.; Campbell, P.A.; Burns, K.D. micro-RNA-486-5p protects against kidney ischemic injury and modifies the apoptotic transcriptome in proximal tubules. Kidney Int. 2021, 100, 597–612. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.; Chen, M.; Guo, Q.; Shen, L.; Liu, X.; Pan, J.; Zhang, Y.; Xu, T.; Zhang, D.; Wei, G. Human umbilical cord mesenchymal stem cell exosome-derived miR-874-3p targeting RIPK1/PGAM5 attenuates kidney tubular epithelial cell damage. Cell. Mol. Biol. Lett. 2023, 28, 12. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, J.; Yang, B.; Qiao, R.; Li, A.; Guo, H.; Ding, J.; Li, H.; Ye, H.; Wu, D.; et al. Transfer of MicroRNA-216a-5p From Exosomes Secreted by Human Urine-Derived Stem Cells Reduces Renal Ischemia/Reperfusion Injury. Front. Cell Dev. Biol. 2020, 8, 610587. [Google Scholar] [CrossRef]
- Ma, M.; Luo, Q.; Fan, L.; Li, W.; Li, Q.; Meng, Y.; Yun, C.; Wu, H.; Lu, Y.; Cui, S.; et al. The urinary exosomes derived from premature infants attenuate cisplatin-induced acute kidney injury in mice via microRNA-30a-5p/ mitogen-activated protein kinase 8 (MAPK8). Bioengineered 2022, 13, 1650–1665. [Google Scholar] [CrossRef] [PubMed]
- Juan, C.-X.; Mao, Y.; Cao, Q.; Chen, Y.; Zhou, L.-B.; Li, S.; Chen, H.; Chen, J.-H.; Zhou, G.-P.; Jin, R. Exosome-mediated pyroptosis of miR-93-TXNIP-NLRP3 leads to functional difference between M1 and M2 macrophages in sepsis-induced acute kidney injury. J. Cell. Mol. Med. 2021, 25, 4786–4799. [Google Scholar] [CrossRef] [PubMed]
- Yu, W.; Zeng, H.; Chen, J.; Fu, S.; Huang, Q.; Xu, Y.; Xu, A.; Lan, H.-Y.; Tang, Y. miR-20a-5p is enriched in hypoxia-derived tubular exosomes and protects against acute tubular injury. Clin. Sci. 2020, 134, 2223–2234. [Google Scholar] [CrossRef]
- He, Z.; Wang, H.; Yue, L. Endothelial progenitor cells-secreted extracellular vesicles containing microRNA-93-5p confer protection against sepsis-induced acute kidney injury via the KDM6B/H3K27me3/TNF-α axis. Exp. Cell Res. 2020, 395, 112173. [Google Scholar] [CrossRef] [PubMed]
- Li, X.-Y.; Zhang, Y.-Q.; Xu, G.; Li, S.-H.; Li, H. miR-124/MCP-1 signaling pathway modulates the protective effect of itraconazole on acute kidney injury in a mouse model of disseminated candidiasis. Int. J. Mol. Med. 2018, 41, 3468–3476. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Sun, H.; Kong, W.; Zhang, B. Functional role of microRNA-500a-3P-loaded liposomes in the treatment of cisplatin-induced AKI. IET Nanobiotechnol. 2020, 14, 465–469. [Google Scholar] [CrossRef]
- Jiang, L.; Liu, X.-Q.; Ma, Q.; Yang, Q.; Gao, L.; Li, H.-D.; Wang, J.-N.; Wei, B.; Wen, J.; Li, J.; et al. hsa-miR-500a-3P alleviates kidney injury by targeting MLKL-mediated necroptosis in renal epithelial cells. FASEB J. 2019, 33, 3523–3535. [Google Scholar] [CrossRef]
- Shi, L.; Zhang, Y.; Xia, Y.; Li, C.; Song, Z.; Zhu, J. MiR-150-5p protects against septic acute kidney injury via repressing the MEKK3/JNK pathway. Cell. Signal. 2021, 86, 110101. [Google Scholar] [CrossRef]
- Sun, Z.; Wu, J.; Bi, Q.; Wang, W. Exosomal lncRNA TUG1 derived from human urine-derived stem cells attenuates renal ischemia/reperfusion injury by interacting with SRSF1 to regulate ASCL4-mediated ferroptosis. Stem Cell Res. Ther. 2022, 13, 297. [Google Scholar] [CrossRef]
- Chen, L.; Xu, J.-Y.; Tan, H.-B. LncRNA TUG1 regulates the development of ischemia-reperfusion mediated acute kidney injury through miR-494-3p/E-cadherin axis. J. Inflamm. 2021, 18, 12. [Google Scholar] [CrossRef]
- Shen, B.; Liu, J.; Zhang, F.; Wang, Y.; Qin, Y.; Zhou, Z.; Qiu, J.; Fan, Y. CCR2 Positive Exosome Released by Mesenchymal Stem Cells Suppresses Macrophage Functions and Alleviates Ischemia/Reperfusion-Induced Renal Injury. Stem Cells Int. 2016, 2016, 1240301. [Google Scholar] [CrossRef] [PubMed]
- Jia, H.; Liu, W.; Zhang, B.; Wang, J.; Wu, P.; Tandra, N.; Liang, Z.; Ji, C.; Yin, L.; Hu, X.; et al. HucMSC exosomes-delivered 14-3-3ζ enhanced autophagy via modulation of ATG16L in preventing cisplatin-induced acute kidney injury. Am. J. Transl. Res. 2018, 10, 101–113. [Google Scholar] [PubMed]
- Du, J.; Sun, Q.; Wang, Z.; Wang, F.; Chen, F.; Wang, H.; Shang, G.; Chen, X.; Ding, S.; Li, C.; et al. Tubular epithelial cells derived-exosomes containing CD26 protects mice against renal ischemia/reperfusion injury by maintaining proliferation and dissipating inflammation. Biochem. Biophys. Res. Commun. 2021, 553, 134–140. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; George, S.K.; Wu, R.; Thakker, P.U.; Abolbashari, M.; Kim, T.-H.; Ko, I.K.; Zhang, Y.; Sun, Y.; Jackson, J.; et al. Reno-protection of Urine-derived Stem Cells in A Chronic Kidney Disease Rat Model Induced by Renal Ischemia and Nephrotoxicity. Int. J. Biol. Sci. 2020, 16, 435–446. [Google Scholar] [CrossRef] [PubMed]
- Yuan, L.; Yang, J.; Liu, F.; Li, L.; Liu, J.; Chen, Y.; Cheng, J.; Lu, Y.; Yuan, Y. Macrophage-derived exosomal miR-195a-5p impairs tubular epithelial cells mitochondria in acute kidney injury mice. FASEB J. 2023, 37, e22691. [Google Scholar] [CrossRef]
- Zhang, Z.; Chen, H.; Zhou, L.; Li, C.; Lu, G.; Wang, L. Macrophage-derived exosomal miRNA-155 promotes tubular injury in ischemia-induced acute kidney injury. Int. J. Mol. Med. 2022, 50, 116. [Google Scholar] [CrossRef]
- Guan, H.; Peng, R.; Mao, L.; Fang, F.; Xu, B.; Chen, M. Injured tubular epithelial cells activate fibroblasts to promote kidney fibrosis through miR-150-containing exosomes. Exp. Cell Res. 2020, 392, 112007. [Google Scholar] [CrossRef]
- Zhou, X.; Zhao, S.; Li, W.; Ruan, Y.; Yuan, R.; Ning, J.; Jiang, K.; Xie, J.; Yao, X.; Li, H.; et al. Tubular cell-derived exosomal miR-150-5p contributes to renal fibrosis following unilateral ischemia-reperfusion injury by activating fibroblast in vitro and in vivo. Int. J. Biol. Sci. 2021, 17, 4021–4033. [Google Scholar] [CrossRef]
- Guo, Y.; Ni, J.; Chen, S.; Bai, M.; Lin, J.; Ding, G.; Zhang, Y.; Sun, P.; Jia, Z.; Huang, S.; et al. MicroRNA-709 Mediates Acute Tubular Injury through Effects on Mitochondrial Function. J. Am. Soc. Nephrol. 2018, 29, 449–461. [Google Scholar] [CrossRef]
- Ding, C.; Zheng, J.; Wang, B.; Li, Y.; Xiang, H.; Dou, M.; Qiao, Y.; Tian, P.; Ding, X.; Xue, W. Exosomal MicroRNA-374b-5p From Tubular Epithelial Cells Promoted M1 Macrophages Activation and Worsened Renal Ischemia/Reperfusion Injury. Front. Cell Dev. Biol. 2020, 8, 587693. [Google Scholar] [CrossRef]
- Lv, L.-L.; Feng, Y.; Wu, M.; Wang, B.; Li, Z.-L.; Zhong, X.; Wu, W.-J.; Chen, J.; Ni, H.-F.; Tang, T.-T.; et al. Exosomal miRNA-19b-3p of tubular epithelial cells promotes M1 macrophage activation in kidney injury. Cell Death Differ. 2020, 27, 210–226. [Google Scholar] [CrossRef]
- Li, Z.-L.; Lv, L.-L.; Tang, T.-T.; Wang, B.; Feng, Y.; Zhou, L.-T.; Cao, J.-Y.; Tang, R.-N.; Wu, M.; Liu, H.; et al. HIF-1α inducing exosomal microRNA-23a expression mediates the cross-talk between tubular epithelial cells and macrophages in tubulointerstitial inflammation. Kidney Int. 2019, 95, 388–404. [Google Scholar] [CrossRef] [PubMed]
- Jiao, Y.; Zhang, T.; Zhang, C.; Ji, H.; Tong, X.; Xia, R.; Wang, W.; Ma, Z.; Shi, X. Exosomal miR-30d-5p of neutrophils induces M1 macrophage polarization and primes macrophage pyroptosis in sepsis-related acute lung injury. Crit. Care 2021, 25, 356. [Google Scholar] [CrossRef] [PubMed]
- Yin, Q.; Zhao, Y.-J.; Ni, W.-J.; Tang, T.-T.; Wang, Y.; Cao, J.-Y.; Yin, D.; Wen, Y.; Li, Z.-L.; Zhang, Y.-L.; et al. MiR-155 deficiency protects renal tubular epithelial cells from telomeric and genomic DNA damage in cisplatin-induced acute kidney injury. Theranostics 2022, 12, 4753–4766. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.-B.; Chen, X.; Li, D.-J.; Qi, G.-N.; Dai, Y.-Q.; Gu, J.; Chen, M.-Q.; Hu, S.; Liu, Z.-Y.; Yang, Z.-M. Inhibition of miR-155 Ameliorates Acute Kidney Injury by Apoptosis Involving the Regulation on TCF4/Wnt/β-Catenin Pathway. Nephron 2019, 143, 135–147. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Zhang, X.; Wang, P.; Zhou, X.; Liang, H.; Li, C. Knockdown of circ-FANCA alleviates LPS-induced HK2 cell injury via targeting miR-93-5p/OXSR1 axis in septic acute kidney injury. Diabetol. Metab. Syndr. 2021, 13, 7. [Google Scholar] [CrossRef] [PubMed]
- Kuang, F.; Wang, B.; You, T.; Liu, Y.; Li, P.; Wang, J. Circ_0001818 targets miR-136-5p to increase lipopolysaccharide-induced HK2 cell injuries by activating TXNIP/NLRP3 inflammasome pathway. Shock 2023, 60, 110–120. [Google Scholar] [CrossRef]
- Herrera, M.B.; Bussolati, B.; Bruno, S.; Morando, L.; Mauriello-Romanazzi, G.; Sanavio, F.; Stamenkovic, I.; Biancone, L.; Camussi, G. Exogenous mesenchymal stem cells localize to the kidney by means of CD44 following acute tubular injury. Kidney Int. 2007, 72, 430–441. [Google Scholar] [CrossRef] [PubMed]
- Du, J.; Li, Y.; Sun, Q.; Wang, Z.; Wang, F.; Chen, F.; Wang, H.; Liu, Y.; Zhou, H.; Shang, G.; et al. Urinary exosomal CD26 is associated with recovery from acute kidney injury in intensive care units: A prospective cohort study. Clin. Chem. Lab. Med. 2021, 59, 1535–1546. [Google Scholar] [CrossRef]
- Xie, X.; Yang, X.; Wu, J.; Tang, S.; Yang, L.; Fei, X.; Wang, M. Exosome from indoleamine 2,3-dioxygenase-overexpressing bone marrow mesenchymal stem cells accelerates repair process of ischemia/reperfusion-induced acute kidney injury by regulating macrophages polarization. Stem Cell Res. Ther. 2022, 13, 367. [Google Scholar] [CrossRef] [PubMed]
- Succar, L.; Pianta, T.J.; Davidson, T.; Pickering, J.W.; Endre, Z.H. Subclinical chronic kidney disease modifies the diagnosis of experimental acute kidney injury. Kidney Int. 2017, 92, 680–692. [Google Scholar] [CrossRef]
- Yun, S.J.; Jeong, P.; Kim, W.-T.; Kim, T.H.; Lee, Y.-S.; Song, P.H.; Choi, Y.-H.; Kim, I.Y.; Moon, S.-K.; Kim, W.-J. Cell-free microRNAs in urine as diagnostic and prognostic biomarkers of bladder cancer. Int. J. Oncol. 2012, 41, 1871–1878. [Google Scholar] [CrossRef]
- Sonoda, H.; Lee, B.R.; Park, K.-H.; Nihalani, D.; Yoon, J.-H.; Ikeda, M.; Kwon, S.-H. miRNA profiling of urinary exosomes to assess the progression of acute kidney injury. Sci. Rep. 2019, 9, 4692. [Google Scholar] [CrossRef] [PubMed]
- Da-Silva, C.C.S.; Anauate, A.C.; Guirao, T.P.; Novaes, A.D.S.; Maquigussa, E.; Boim, M.A. Analysis of exosome-derived microRNAs as early biomarkers of lipopolysaccharide-induced acute kidney injury in rats. Front. Physiol. 2022, 13, 944864. [Google Scholar] [CrossRef] [PubMed]
- Yun, C.Y.; Lim, J.H.; Oh, J.H.; Cho, A.Y.; Lee, K.Y.; Sun, I.O. Urinary Exosomal microRNA-21 as a Marker for Scrub Typhus-Associated Acute Kidney Injury. Genet. Test. Mol. Biomark. 2021, 25, 140–144. [Google Scholar] [CrossRef]
- Awdishu, L.; Le, A.; Amato, J.; Jani, V.; Bal, S.; Mills, R.H.; Carrillo-Terrazas, M.; Gonzalez, D.J.; Tolwani, A.; Acharya, A.; et al. Urinary Exosomes Identify Inflammatory Pathways in Vancomycin Associated Acute Kidney Injury. Int. J. Mol. Sci. 2021, 22, 2784. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.; Ren, Z.; Xie, A.; Jia, Y.; Xue, Y.; Wang, P.; Ji, D.; Wang, X. Assessment of Urinary Exosomal NHE3 as a Biomarker of Acute Kidney Injury. Diagnostics 2022, 12, 2634. [Google Scholar] [CrossRef] [PubMed]
- Sonoda, H.; Oshikawa-Hori, S.; Ikeda, M. An Early Decrease in Release of Aquaporin-2 in Urinary Extracellular Vesicles After Cisplatin Treatment in Rats. Cells 2019, 8, 139. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Zhu, Q.; Cheng, L.; Wang, Y.; Li, M.; Yang, Q.; Hu, L.; Lou, D.; Li, J.; Dong, X.; et al. Exosome detection via the ultrafast-isolation system: EXODUS. Nat. Methods 2021, 18, 212–218. [Google Scholar] [CrossRef]
- Rayamajhi, S.; Nguyen, T.D.T.; Marasini, R.; Aryal, S. Macrophage-derived exosome-mimetic hybrid vesicles for tumor targeted drug delivery. Acta Biomater. 2019, 94, 482–494. [Google Scholar] [CrossRef]
- Liu, C.; Xu, X.; Li, B.; Situ, B.; Pan, W.; Hu, Y.; An, T.; Yao, S.; Zheng, L. Single-Exosome-Counting Immunoassays for Cancer Diagnostics. Nano Lett. 2018, 18, 4226–4232. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Wang, H.; Fu, J.; Wu, X.; Liang, X.-Y.; Liu, X.-Y.; Wu, X.; Cao, L.-L.; Xu, Z.-Y.; Dong, M. Microfluidic-based exosome isolation and highly sensitive aptamer exosome membrane protein detection for lung cancer diagnosis. Biosens. Bioelectron. 2022, 214, 114487. [Google Scholar] [CrossRef]
- Gardiner, C.; Di Vizio, D.; Sahoo, S.; Théry, C.; Witwer, K.W.; Wauben, M.; Hill, A.F. Techniques used for the isolation and characterization of extracellular vesicles: Results of a worldwide survey. J. Extracell. Vesicles 2016, 5, 32945. [Google Scholar] [CrossRef]
- Rider, M.A.; Hurwitz, S.N.; Meckes, D.G. ExtraPEG: A Polyethylene Glycol-Based Method for Enrichment of Extracellular Vesicles. Sci. Rep. 2016, 6, 23978. [Google Scholar] [CrossRef] [PubMed]
- Zarovni, N.; Corrado, A.; Guazzi, P.; Zocco, D.; Lari, E.; Radano, G.; Muhhina, J.; Fondelli, C.; Gavrilova, J.; Chiesi, A. Integrated isolation and quantitative analysis of exosome shuttled proteins and nucleic acids using immunocapture approaches. Methods 2015, 87, 46–58. [Google Scholar] [CrossRef]
- Ludwig, A.-K.; De Miroschedji, K.; Doeppner, T.R.; Börger, V.; Ruesing, J.; Rebmann, V.; Durst, S.; Jansen, S.; Bremer, M.; Behrmann, E.; et al. Precipitation with polyethylene glycol followed by washing and pelleting by ultracentrifugation enriches extracellular vesicles from tissue culture supernatants in small and large scales. J. Extracell. Vesicles 2018, 7, 1528109. [Google Scholar] [CrossRef]
- Busatto, S.; Vilanilam, G.; Ticer, T.; Lin, W.-L.; Dickson, D.W.; Shapiro, S.; Bergese, P.; Wolfram, J. Tangential Flow Filtration for Highly Efficient Concentration of Extracellular Vesicles from Large Volumes of Fluid. Cells 2018, 7, 273. [Google Scholar] [CrossRef]
- Lee, J.H.; Ha, D.H.; Go, H.-K.; Youn, J.; Kim, H.-K.; Jin, R.C.; Miller, R.B.; Kim, D.-H.; Cho, B.S.; Yi, Y.W. Reproducible Large-Scale Isolation of Exosomes from Adipose Tissue-Derived Mesenchymal Stem/Stromal Cells and Their Application in Acute Kidney Injury. Int. J. Mol. Sci. 2020, 21, 4774. [Google Scholar] [CrossRef]
- Cao, J.; Wang, B.; Tang, T.; Lv, L.; Ding, Z.; Li, Z.; Hu, R.; Wei, Q.; Shen, A.; Fu, Y.; et al. Three-dimensional culture of MSCs produces exosomes with improved yield and enhanced therapeutic efficacy for cisplatin-induced acute kidney injury. Stem Cell Res. Ther. 2020, 11, 206. [Google Scholar] [CrossRef]
- He, C.; Zheng, S.; Luo, Y.; Wang, B. Exosome Theranostics: Biology and Translational Medicine. Theranostics 2018, 8, 237–255. [Google Scholar] [CrossRef]
- Zhang, K.; Li, R.; Chen, X.; Yan, H.; Li, H.; Zhao, X.; Huang, H.; Chen, S.; Liu, Y.; Wang, K.; et al. Renal Endothelial Cell-Targeted Extracellular Vesicles Protect the Kidney from Ischemic Injury. Adv. Sci. 2023, 10, e2204626. [Google Scholar] [CrossRef] [PubMed]
- Tang, T.-T.; Wang, B.; Li, Z.-L.; Wen, Y.; Feng, S.-T.; Wu, M.; Liu, D.; Cao, J.-Y.; Yin, Q.; Yin, D.; et al. Kim-1 Targeted Extracellular Vesicles: A New Therapeutic Platform for RNAi to Treat AKI. J. Am. Soc. Nephrol. 2021, 32, 2467–2483. [Google Scholar] [CrossRef] [PubMed]
- Wu, P.; Tang, Y.; Jin, C.; Wang, M.; Li, L.; Liu, Z.; Shi, H.; Sun, Z.; Hou, X.; Chen, W.; et al. Neutrophil membrane engineered HucMSC sEVs alleviate cisplatin-induced AKI by enhancing cellular uptake and targeting. J. Nanobiotechnol. 2022, 20, 353. [Google Scholar] [CrossRef] [PubMed]
- Hu, S.; Wang, X.; Li, Z.; Zhu, D.; Cores, J.; Wang, Z.; Li, J.; Mei, X.; Cheng, X.; Su, T.; et al. Platelet membrane and stem cell exosome hybrid enhances cellular uptake and targeting to heart injury. Nano Today 2021, 39, 101210. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Huang, Z.; Wang, Q.; Gao, J.; Chen, J.; Tan, H.; Li, S.; Wang, Z.; Weng, X.; Yang, H.; et al. Targeted immunomodulation therapy for cardiac repair by platelet membrane engineering extracellular vesicles via hitching peripheral monocytes. Biomaterials 2022, 284, 121529. [Google Scholar] [CrossRef]
- Kim, M.S.; Haney, M.J.; Zhao, Y.; Yuan, D.; Deygen, I.; Klyachko, N.L.; Kabanov, A.V.; Batrakova, E.V. Engineering macrophage-derived exosomes for targeted paclitaxel delivery to pulmonary metastases: In vitro and in vivo evaluations. Nanomedicine 2018, 14, 195–204. [Google Scholar] [CrossRef]
- Zou, J.; Shi, M.; Liu, X.; Jin, C.; Xing, X.; Qiu, L.; Tan, W. Aptamer-Functionalized Exosomes: Elucidating the Cellular Uptake Mechanism and the Potential for Cancer-Targeted Chemotherapy. Anal. Chem. 2019, 91, 2425–2430. [Google Scholar] [CrossRef]
- Théry, C.; Ostrowski, M.; Segura, E. Membrane vesicles as conveyors of immune responses. Nat. Rev. Immunol. 2009, 9, 581–593. [Google Scholar] [CrossRef]
- Piffoux, M.; Silva, A.K.A.; Wilhelm, C.; Gazeau, F.; Tareste, D. Modification of Extracellular Vesicles by Fusion with Liposomes for the Design of Personalized Biogenic Drug Delivery Systems. ACS Nano 2018, 12, 6830–6842. [Google Scholar] [CrossRef]
- Fazekas, B.; Griffin, M.D. Mesenchymal stromal cell-based therapies for acute kidney injury: Progress in the last decade. Kidney Int. 2020, 97, 1130–1140. [Google Scholar] [CrossRef]
- Shi, H.; Xu, X.; Zhang, B.; Xu, J.; Pan, Z.; Gong, A.; Zhang, X.; Li, R.; Sun, Y.; Yan, Y.; et al. 3,3’-Diindolylmethane stimulates exosomal Wnt11 autocrine signaling in human umbilical cord mesenchymal stem cells to enhance wound healing. Theranostics 2017, 7, 1674–1688. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Shang, Y.; Chen, X.; Midgley, A.C.; Wang, Z.; Zhu, D.; Wu, J.; Chen, P.; Wu, L.; Wang, X.; et al. Supramolecular Nanofibers Containing Arginine-Glycine-Aspartate (RGD) Peptides Boost Therapeutic Efficacy of Extracellular Vesicles in Kidney Repair. ACS Nano 2020, 14, 12133–12147. [Google Scholar] [CrossRef] [PubMed]
- Ji, C.; Zhang, J.; Zhou, Z.; Shi, H.; Liu, W.; Sun, F.; Zhang, C.; Zhang, L.; Sun, Z.; Qian, H. Platelet-rich plasma promotes MSCs exosomes paracrine to repair acute kidney injury via AKT/Rab27 pathway. Am. J. Transl. Res. 2021, 13, 1445–1457. [Google Scholar] [PubMed]
- Ding, Z.-Y.; Tang, T.-T.; Li, Z.-L.; Cao, J.-Y.; Lv, L.-L.; Wen, Y.; Wang, B.; Liu, B.-C. Therapeutic Effect of Extracellular Vesicles Derived from HIF Prolyl Hydroxylase Domain Enzyme Inhibitor-Treated Cells on Renal Ischemia/Reperfusion Injury. Kidney Dis. 2022, 8, 206–216. [Google Scholar] [CrossRef] [PubMed]
| Exo Source | Model | Mechanism | Pathways/Markers | Reference |
|---|---|---|---|---|
| BMMSC | IRI Rats Mice HK-2 (H/R) | Anti-apoptotic Reduces oxidative stress Anti-inflammatory Promote angiogenesis Inhibits endoplasmic reticulum stress | IL-6, TNF-α, NF-κB IFN-γ ↓ caspase-9, cleaved caspase-3, Bax, and Bcl-2 ↓ BIP miR-149/let-7c/Faslg | [24,54,55] |
| BMMSC | UUO Rats | Inhibits fibrosis Epithelial mesenchymal transformation Anti-inflammatory Anti-apoptotic | STAT3 ↓ MMP-9P Colla1,α-SMA ↓ PCNA ↓ | [19] |
| BMMSC | Cisp Mice | Anti-apoptotic Reduces DNA damage | Inhibiting p53 | [27] |
| SCAP | Cisp NRK-52E | Anti-apoptotic Reduces oxidative stress Anti-inflammatory | NF-κβ, IL-1β, p53 ↓ Bcl-2 ↑ Bax, CASP8, CASP9, and CASP3 ↓ | [31] |
| hAEC | IRI mice HK-2 (H/R) | Anti-apoptotic Anti-inflammatory Promote angiogenesis | extracellular matrix growth factor cytokine production, immunomodulation | [33] |
| hAEC | Sepsis LPS-treated HUVECs CLP mice | Anti-inflammatory Maintained endothelial cell adhesion junction Mitigating endothelial dysfunction | phosphor-p65, p65, VCAM-1, ZO-1 | [35] |
| hAEC | Cisp mice | Anti-inflammatory | TNF-α/MAPK and caspase signaling ↓ | [34] |
| ADMSC | IRI | Anti-apoptotic Anti-inflammatory Reduces oxidative stress, DNA damage Promote angiogenesis Inhibits fibrosis Relieves tubular damage | TNF-α/NF-κB/IL-1β/MIF/PAI-1/Cox-2 ↓ NOX-1/NOX-2/oxidized protein ↓ Bax/caspase-3/PARP ↓ Smad3/TGF-β ↓ | [38] |
| ADMSC | Sepsis CLP mice LPS HK-2 | Anti-apoptotic Anti-inflammatory Promote angiogenesis Autophagy | SIRT1 TLR9 | [37,39,41] |
| ADMSC | Postrenal AKI Cats | Metabolome | carnitine, melibiose, D-Glucosamine, cytidine, dihydroorotic acid, stachyose ↑ | [42] |
| USCs | Plasma from AKI patients IRI rats IRI mice HK-2 (H/R) | Anti-inflammatory Inhibits fibrosis Reduces oxidative stress | TRAF6 IRAK1 NF-κB p65 IL10 and TGFβ1 ↑ IFN-γ and IL-1β ↓ | [51,52,53] |
| IPSC | IRI HK-2 (H/R) | Reduce renal damage | ERK 1/2 signaling | [50] |
| HuMSC | Cisp NRK-52E cells | Promotes TEC proliferation Anti-apoptotic Anti-inflammatory Reduces oxidative stress Promotes mitochondrial fusion Autophagy | fibronectin, α-SMA, GSDMD, caspase-1, IL-1β and NLRP3 ↓ | [56] |
| HuMSC | UUO rats | Inhibits fibrosis | CK1δ/β-TRCP inhibited YAP activity | [47] |
| HuMSC | IRI rats | Anti-inflammatory Inhibits fibrosis Reduces oxidative stress | [45,46,57] | |
| Macrophages | Sepsis CLP mice | Relieves endothelial cell dysfunction Reduces tubular cell apoptosis, pyroptosis | HMGB, VCAM1 ↓ | [56] |
| Pericytes | IRI mice | Reduces tubular cell damage Maintain capillary stability | Cadherin APN | [58] |
| EPC | Sepsis CLP rats | Anti-inflammatory | RUNX1 ↓ | [59] |
| Content | Exo Source | Model | Mechanism | Pathways | Reference |
|---|---|---|---|---|---|
| miR-199a-3p | BMMSC | IRI mice HK-2 (H/R) | Anti-apoptotic Reduces oxidative stress | Sema3A ↓—ERK, AKT ↑ | [24] |
| miR-let-7b-5p | BMMSC | Cisp mice | Anti-apoptotic Reduces DNA damage | p53 ↓—DNA damage and apoptosis pathway activity ↓ | [27] |
| miR-1184 | BMMSC | HK-2 by cisp | Anti-apoptotic Anti-inflammatory Break the G1 block | Targeting FOXO4, p27 Kip1 and CDK2 | [66] |
| miR-146b | HucMSC | LPS HK-2 CLP mice | Anti-inflammatory | IRAK1 ↓ NF-κB ↓ | [67] |
| miR-486-5p | HucMSC | IRI mice | Promotes proliferation angiogenesis | PTEN ↓—Akt phosphorylation | [45] |
| miR-486-5p | HucMSC | IRI mice | Anti-apoptotic Anti-inflammatory | protein kinase B ↑ TNF pathway ↓ phosphatase and tensin homolog↓ | [68] |
| miR-125b-5p | HucMSC | IRI mice HK-2 (H/R) | Break the G2/M block Anti-apoptotic | P53 ↓—CDK1 and Cyclin B1 ↑ modulation of Bcl-2 and Bax accumulated in proximal tubules by virtue of the VLA-4 and LFA-1 | [46] |
| miR-874-3p | HucMSC | UUO mice HK-2 by cisp | Reduce necrosis Promotes mitochondrial fusion | RIPK1 PGAM5 ↓—dephosphorylation of the S637 site of the Drp1 gene | [69] |
| miR-342-5p | ADMSC | Sepsis patients with sepsis-associated AKI CLP mice HK-2 by LPS | Enhanced autophagy Anti-inflammatory Reduced BUN and SCr levels | TLR9 ↓—autophagy ↑ BUN, SCr, ↓ | [41] |
| miR-216a-5p | USC | IRI rats HK-2 (H/R) | Promote proliferation Angiogenesis | PTEN ↓—Akt phosphorylation | [70] |
| miR-146a-5p | USC | IRI rats HK-2 (H/R) | Anti-inflammatory Anti-apoptotic | Target 3′UTR of IRAK1—NF-κB ↓ | [52] |
| miR-30a-5p | USCp | Cisp mice HK-2 by cisp | Anti-apoptotic Anti-inflammatory | MAPK8 ↓ | [71] |
| miR-93-5p | Macrophage (M2 > M1) | CLP mice TCMK-1 cells by LPS | Renal epithelial cell pyroptosis Anti-inflammatory Anti-apoptotic | TXNIP—pyroptosis in renal epithelial cell | [72] |
| miR-590-3p | TEC | AKI patients after cardiac surgery HK-2 (H/R) | Autophagy | miR-590-3p was highly enriched in the plasma exosomes of young AKI patients after cardiac surgery Beclin-1 and LC3II ↑ | [53] |
| miR-20a-5p | TEC H | HK-2 (H/R) IRI mice | Promotes TEC proliferation, Promotes mitochondrial fusion, Attenuating necrosis | macrophages infiltration ↓ | [73] |
| miR-21-5p | ECFC | CLP mice | Anti-inflammatory Promotes proliferation angiogenesis | RUNX1 ↓ | [60] |
| miR-486-5p | ECFC | IRI mice | Promotes proliferation angiogenesis | PTEN ↓—Akt phosphorylation | [45] |
| miR-93-5p | EPC | HK-2 by LPS CLP mice | Anti-apoptotic Anti-inflammatory | Regulating KDM6BH/3K27me3/TNF-α axis | [74] |
| miR-124 | Unknown | CIS mice | Anti-inflammatory | MCP-1 ↑ | [75] |
| miR500a3p | Unknown | HK-2 by cisp | Reduce necrosis | RIPK3 and MLKL ↓ | [76,77] |
| miR-150-5p | Unknown | HK-2 by LPS LPS mice | Anti-apoptotic Anti-inflammatory and oxidative stress | MEKK3/JNK pathway | [78] |
| lncRNA TUG1 | USC | HK-2 (H/R) IRI mice | Reduce Ferroptosis | ACSL4 ↑—TUG1’s repression ↓ miR-494-3p—E-cadherin and TUG1 | [79] [80] |
| CCR2 | BMMSC | IRI mice | Inhibits macrophage recruitment activation | Unknown | [81] |
| 14-3-3z | HucMSC | NRK-52E by cisp Cisp Rats | Autophagy | ATG16L↑ | [82] |
| CD26 | TEC | IRI mice | Anti-inflammatory Promotes TEC proliferation | CXCR4, SDF1 ↓ | [83] |
| Klotho | USC | AKI rats | Anti-apoptotic Anti-inflammatory Promotes proliferation angiogenesis | fibrosis, monocyte infiltration ↓ SOD1 ↑ | [84] |
| Content | Exo Source | Model | Mechanism | Pathways | Reference |
|---|---|---|---|---|---|
| miR-195a-5p | Macrophage | Cisp mice | Promote TEC apoptosis and damage TEC mitochondria | unknown | [85] |
| miR-155 | Macrophage | IRI/cisp mice HK-2 (cisp) NRK-52E (H/R) | Promote TEC damage, promote inflammation limiting the telomeric dysfunction and the genomic DNA damage | TRF1, CDK12 ↓ TCF4/Wnt/β-Catenin | [86,94,95] |
| miR-19b-3p | LPS-TEC | Sepsis LPS mice | Promotes inflammation, macrophage 1 polarization | NF-κB/SOCS-1 | [91] |
| miR-150-5p | TEC | IRI UIRI rats NRK-52E (H/R) | Activates fibroblasts and promotes fibrosis | suppressor of cytokine signaling 1 to activate fibroblast ↓ | [88] |
| miR374b-5p | TEC | IRI mice | Promote Macrophage 1 polarization | Transferring miR-374b-5p | [90] |
| miR-709 | PTC | Cisp mice human AKI kidney | Promote TEC apoptosis and mitochondria damage | TFAM ↓ miR-709 in PTCs of patients ↑ | [89] |
| miR-23a | TEC H | UUO/IRI mice and TEC | Promotes inflammation Macrophage activation | suppression of the ubiquitin editor A20 | [92] |
| miR-30d-5p | Neutrophils mice | Sepsis | Promotes inflammation, macrophage 1 polarization Promotes pyroptosis of macrophages | NLRP3—NF-κB ↑ SOCS-1 and SIRT1 ↓ | [93] |
| Circ-FANCA | Sepsis patient LPS-HK2 | Sepsis LPS HK-2 | Promotes inflammation, apoptosis Inhibit proliferation (G0/G1 arrest) | sponging OXSR1 | [96] |
| Circ0001818 | Unknown (Serum) | LPS-HK2 | Promote LPS-HK2 damage | miR-136-5p—TXNIP | [97] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, X.; Wang, J.; Zhang, J.; Tan, Y.; Li, Y.; Peng, Z. Exosomes Highlight Future Directions in the Treatment of Acute Kidney Injury. Int. J. Mol. Sci. 2023, 24, 15568. https://doi.org/10.3390/ijms242115568
Zhang X, Wang J, Zhang J, Tan Y, Li Y, Peng Z. Exosomes Highlight Future Directions in the Treatment of Acute Kidney Injury. International Journal of Molecular Sciences. 2023; 24(21):15568. https://doi.org/10.3390/ijms242115568
Chicago/Turabian StyleZhang, Xiaoyu, Jing Wang, Jing Zhang, Yuwei Tan, Yiming Li, and Zhiyong Peng. 2023. "Exosomes Highlight Future Directions in the Treatment of Acute Kidney Injury" International Journal of Molecular Sciences 24, no. 21: 15568. https://doi.org/10.3390/ijms242115568






