Role and Regulation of Transcription Factors in Osteoclastogenesis
Abstract
:1. Introduction
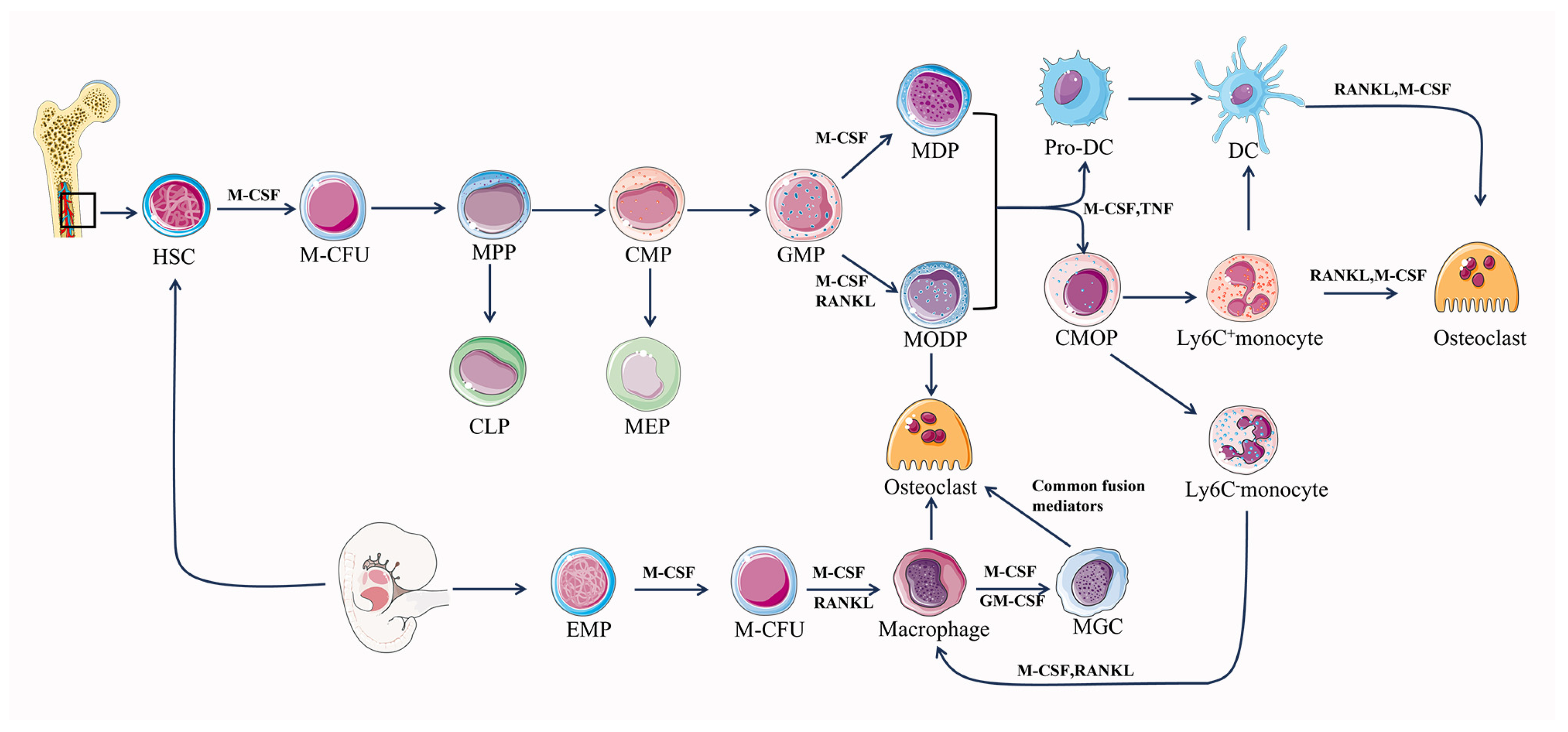
2. The Origins of Osteoclasts
2.1. EMP-Derived Osteoclasts
2.2. HSC-Derived Osteoclasts
3. Osteoclast Differentiation and Maturation
4. Intercellular Communication in Osteoclastogenesis
4.1. Osteoclast–Osteoblast Communication
4.2. Osteoclast–Osteocyte Communication
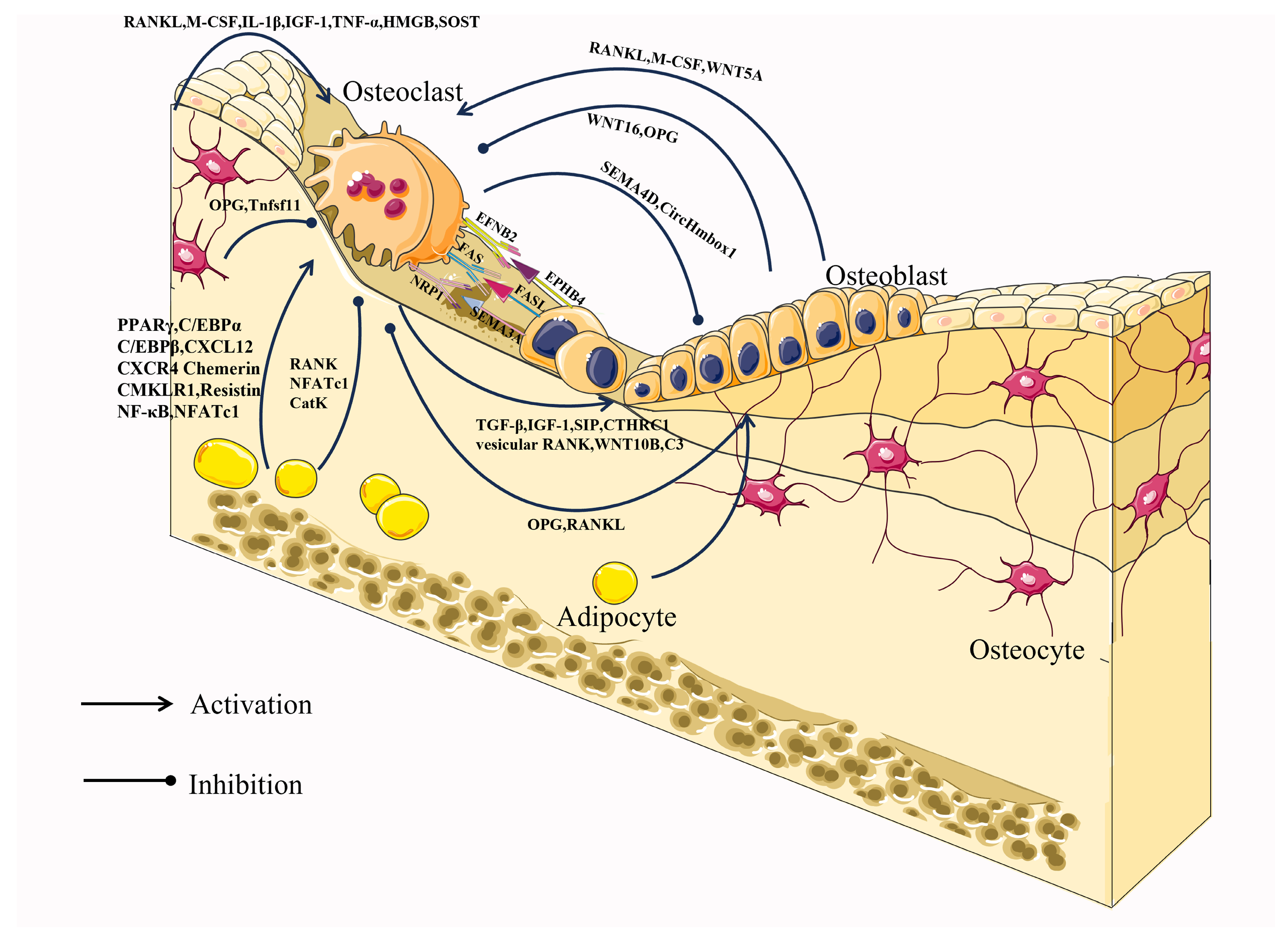
4.3. Osteoclast–Adipocyte Communication
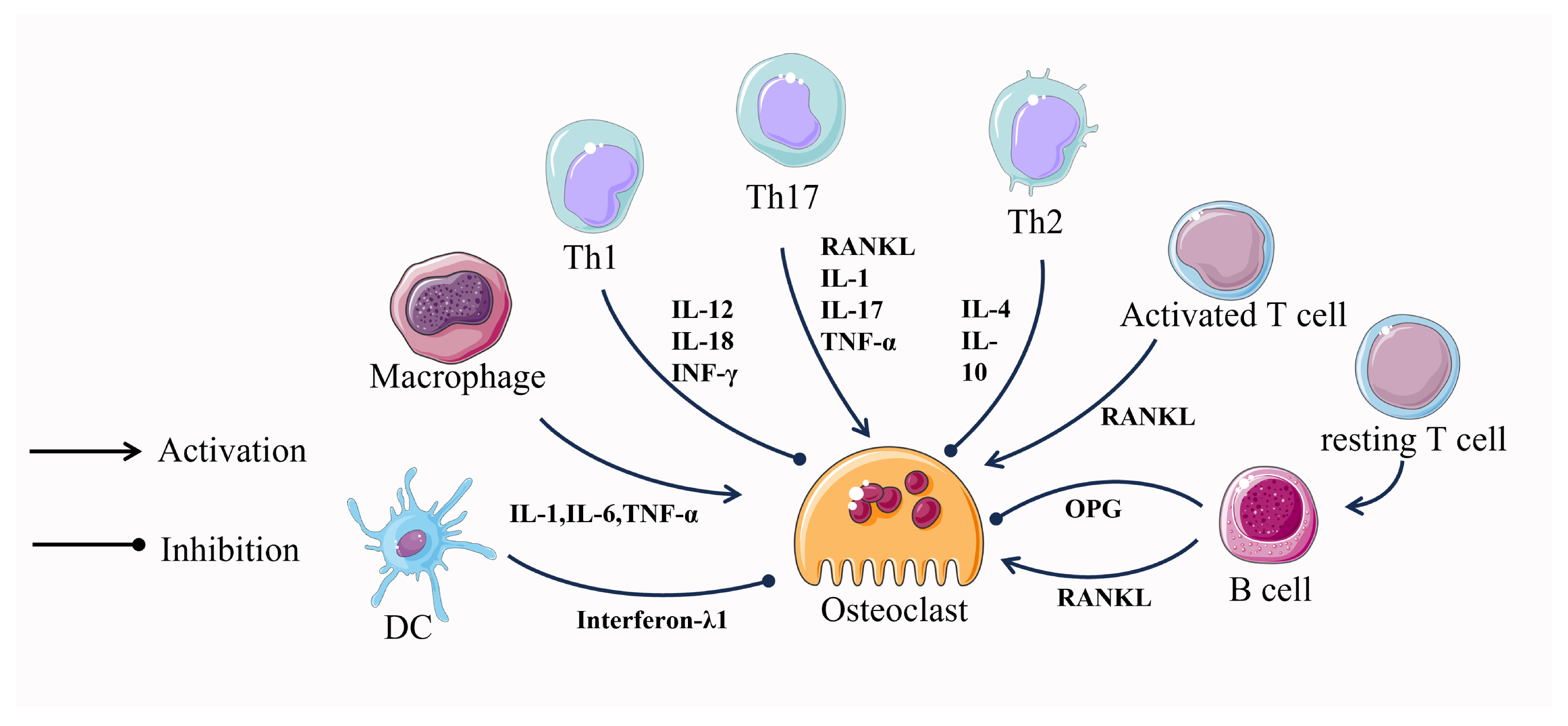
4.4. Osteoclast–Immune Cell Communication
5. Transcriptional Regulation in Osteoclastogenesis
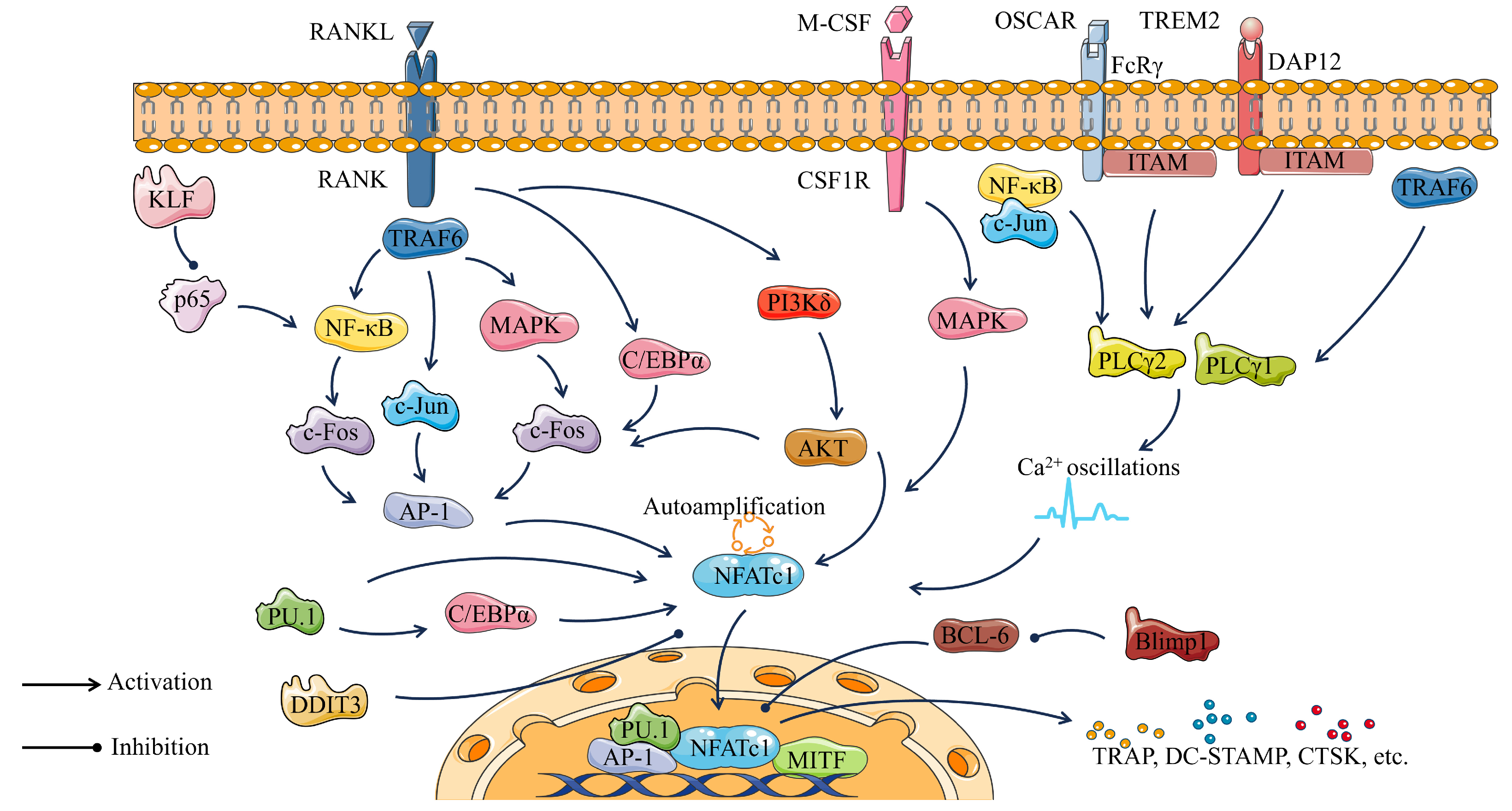
5.1. NFATc1
5.2. MITF
5.3. C/EBPα
5.4. PU.1
5.5. NF-κB
5.6. c-Fos
5.7. DAP12, TREM2, and FcRγ
5.8. Other Transcription Factors Regulating Osteoclast Differentiation
5.8.1. PLC-Gamma
5.8.2. AKT
5.8.3. Src
5.8.4. RIP140
5.8.5. TFE3
5.8.6. BCL-6 and Blimp1
5.8.7. DDIT3
6. Epigenetic Regulation of Transcription Factors in Osteoclastogenesis
6.1. Non-Coding RNAs (ncRNAs))
6.1.1. MiRNA
6.1.2. LncRNA
| LncRNAs | Roles | Target Gene(s) | References |
|---|---|---|---|
| LncRNA TUG1 | Promoting osteoclast differentiation | TRAP, NFATc1, Mafb | [214] |
| LncRNA-AK077216 | Bidirectional regulation of osteoclast differentiation | NFATc1 | [215] |
| lncRNA Bmncr | Inhibiting osteoclast differentiation | RANKL | [222] |
| LncRNA-Jak3 | Promoting osteoclast differentiation | NFATc1, CTSK | [223] |
| LncRNA XIST | Promoting osteoclast differentiation | SPHK1, S1P, ERK | [224] |
| LncRNA Nron | Inhibiting osteoclast differentiation | NF-κb, NFATc1 | [225] |
| lncRNA-MIRG | Promoting osteoclast differentiation and bone resorption function | miR-1897, NFATc1 | [226] |
| lncRNA NEAT1 | Promoting osteoclast differentiation | M-CSF | [227] |
| lncRNA HOTAIR | Promoting osteoclast differentiation | RANKL | [228] |
| lncRNA SNHG15 | Promoting osteoclast differentiation, proliferation, and metastasis | miR-381-3p/ NEK2 | [229] |
6.1.3. CircRNA
| CircRNAs | Roles | Target Gene(s) | References |
|---|---|---|---|
| CircCHEK1_246aa | Promoting osteoclast differentiation | NFATc1 | [233] |
| CircRNA 28313 | Promoting osteoclast differentiation | CSF1, miR-195a | [234] |
| CircRNA 009934 | Promoting osteoclast differentiation | miR-5107, TRAF6 | [235] |
| Circ_0008542 | Promoting osteoclast differentiation and bone resorption function | METTL3, miRNA-185-5p | [236] |
| Circ_0007059 | Inhibiting osteoclast differentiation | miR-378, BMP-2 | [237] |
| CircHmbox1 | Inhibiting osteoclast differentiation and promoting osteoblast differentiation | miRNA-1247-5p | [79] |
6.2. DNA and Histone Modifications
6.3. Modulating Epigenetic Regulators as Therapy for Bone Disorders
7. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Imai, Y.; Youn, M.-Y.; Inoue, K.; Takada, I.; Kouzmenko, A.; Kato, S. Nuclear receptors in bone physiology and diseases. Physiol. Rev. 2013, 93, 481–523. [Google Scholar] [CrossRef]
- Wei, Y.; Sun, Y. Aging of the Bone. Adv. Exp. Med. Biol. 2018, 1086, 189–197. [Google Scholar] [CrossRef]
- van Gastel, N.; Carmeliet, G. Metabolic regulation of skeletal cell fate and function in physiology and disease. Nat. Metab. 2021, 3, 11–20. [Google Scholar] [CrossRef] [PubMed]
- Feng, X.; McDonald, J.M. Disorders of bone remodeling. Annu. Rev. Pathol. 2011, 6, 121–145. [Google Scholar] [CrossRef] [PubMed]
- Gaffney-Stomberg, E. The Impact of Trace Minerals on Bone Metabolism. Biol. Trace Elem. Res. 2019, 188, 26–34. [Google Scholar] [CrossRef] [PubMed]
- Siddiqui, J.A.; Partridge, N.C. Physiological Bone Remodeling: Systemic Regulation and Growth Factor Involvement. Physiology 2016, 31, 233–245. [Google Scholar] [CrossRef]
- Mizoguchi, T.; Ono, N. The diverse origin of bone-forming osteoblasts. J. Bone Miner. Res. 2021, 36, 1432–1447. [Google Scholar] [CrossRef]
- Schaffler, M.B.; Cheung, W.-Y.; Majeska, R.; Kennedy, O. Osteocytes: Master orchestrators of bone. Calcif. Tissue Int. 2014, 94, 5–24. [Google Scholar] [CrossRef]
- Schaffler, M.B.; Kennedy, O.D. Osteocyte signaling in bone. Curr. Osteoporos. Rep. 2012, 10, 118–125. [Google Scholar] [CrossRef]
- Matic, I.; Matthews, B.G.; Wang, X.; Dyment, N.A.; Worthley, D.L.; Rowe, D.W.; Grcevic, D.; Kalajzic, I. Quiescent Bone Lining Cells Are a Major Source of Osteoblasts During Adulthood. Stem Cells 2016, 34, 2930–2942. [Google Scholar] [CrossRef]
- Park-Min, K.H. Metabolic reprogramming in osteoclasts. Semin. Immunopathol. 2019, 41, 565–572. [Google Scholar] [CrossRef] [PubMed]
- Reid, I.R.; Billington, E.O. Drug therapy for osteoporosis in older adults. Lancet 2022, 399, 1080–1092. [Google Scholar] [CrossRef] [PubMed]
- Noh, J.Y.; Yang, Y.; Jung, H. Molecular Mechanisms and Emerging Therapeutics for Osteoporosis. Int. J. Mol. Sci. 2020, 21, 7623. [Google Scholar] [CrossRef] [PubMed]
- Martin, T.J.; Sims, N.A. Osteoclast-derived activity in the coupling of bone formation to resorption. Trends Mol. Med. 2005, 11, 76–81. [Google Scholar] [CrossRef]
- McClung, M.R.; O’Donoghue, M.L.; Papapoulos, S.E.; Bone, H.; Langdahl, B.; Saag, K.G.; Reid, I.R.; Kiel, D.P.; Cavallari, I.; Bonaca, M.P.; et al. Odanacatib for the treatment of postmenopausal osteoporosis: Results of the LOFT multicentre, randomised, double-blind, placebo-controlled trial and LOFT Extension study. Lancet Diabetes Endocrinol. 2019, 7, 899–911. [Google Scholar] [CrossRef]
- Marx, D.; Yazdi, A.R.; Papini, M.; Towler, M. A review of the latest insights into the mechanism of action of strontium in bone. Bone Rep. 2020, 12, 100273. [Google Scholar] [CrossRef]
- Miranda, T.S.; Napimoga, M.H.; De Franco, L.; Marins, L.M.; Malta, F.d.S.; Pontes, L.A.; Morelli, F.M.; Duarte, P.M. Strontium ranelate improves alveolar bone healing in estrogen-deficient rats. J. Periodontol. 2020, 91, 1465–1474. [Google Scholar] [CrossRef]
- Nakamura, T.; Imai, Y.; Matsumoto, T.; Sato, S.; Takeuchi, K.; Igarashi, K.; Harada, Y.; Azuma, Y.; Krust, A.; Yamamoto, Y.; et al. Estrogen prevents bone loss via estrogen receptor alpha and induction of Fas ligand in osteoclasts. Cell 2007, 130, 811–823. [Google Scholar] [CrossRef]
- Trovas, G.P.; Lyritis, G.P.; Galanos, A.; Raptou, P.; Constantelou, E. A randomized trial of nasal spray salmon calcitonin in men with idiopathic osteoporosis: Effects on bone mineral density and bone markers. J. Bone Miner. Res. 2002, 17, 521–527. [Google Scholar] [CrossRef]
- Zhou, J.; Ma, X.; Wang, T.; Zhai, S. Comparative efficacy of bisphosphonates in short-term fracture prevention for primary osteoporosis: A systematic review with network meta-analyses. Osteoporos. Int. 2016, 27, 3289–3300. [Google Scholar] [CrossRef]
- Zhou, J.; Wang, T.; Zhao, X.; Miller, D.R.; Zhai, S. Comparative Efficacy of Bisphosphonates to Prevent Fracture in Men with Osteoporosis: A Systematic Review with Network Meta-Analyses. Rheumatol. Ther. 2016, 3, 117–128. [Google Scholar] [CrossRef] [PubMed]
- Hoppé, E.; Masson, C.; Laffitte, A.; Chappard, D.; Audran, M. Osteomalacia in a patient with Paget’s bone disease treated with long-term etidronate. Morphologie 2012, 96, 40–43. [Google Scholar] [CrossRef] [PubMed]
- Mi, B.; Xiong, W.; Xu, N.; Guan, H.; Fang, Z.; Liao, H.; Zhang, Y.; Gao, B.; Xiao, X.; Fu, J.; et al. Strontium-loaded titania nanotube arrays repress osteoclast differentiation through multiple signalling pathways: In vitro and in vivo studies. Sci. Rep. 2017, 7, 2328. [Google Scholar] [CrossRef] [PubMed]
- Ash, P.; Loutit, J.F.; Townsend, K.M. Osteoclasts derived from haematopoietic stem cells. Nature 1980, 283, 669–670. [Google Scholar] [CrossRef]
- Walker, D.G. Bone resorption restored in osteopetrotic mice by transplants of normal bone marrow and spleen cells. Science 1975, 190, 784–785. [Google Scholar] [CrossRef]
- Scheven, B.A.; Visser, J.W.; Nijweide, P.J. In vitro osteoclast generation from different bone marrow fractions, including a highly enriched haematopoietic stem cell population. Nature 1986, 321, 79–81. [Google Scholar] [CrossRef]
- Yahara, Y.; Barrientos, T.; Tang, Y.J.; Puviindran, V.; Nadesan, P.; Zhang, H.; Gibson, J.R.; Gregory, S.G.; Diao, Y.; Xiang, Y.; et al. Erythromyeloid progenitors give rise to a population of osteoclasts that contribute to bone homeostasis and repair. Nat. Cell Biol. 2020, 22, 49–59. [Google Scholar] [CrossRef]
- Ginhoux, F.; Greter, M.; Leboeuf, M.; Nandi, S.; See, P.; Gokhan, S.; Mehler, M.F.; Conway, S.J.; Ng, L.G.; Stanley, E.R.; et al. Fate mapping analysis reveals that adult microglia derive from primitive macrophages. Science 2010, 330, 841–845. [Google Scholar] [CrossRef]
- Yahara, Y.; Nguyen, T.; Ishikawa, K.; Kamei, K.; Alman, B.A.; Ginhoux, F.; Martin, P. The origins and roles of osteoclasts in bone development, homeostasis and repair. Development 2022, 149, dev199908. [Google Scholar] [CrossRef]
- Perdiguero, E.G.; Klapproth, K.; Schulz, C.; Busch, K.; Azzoni, E.; Crozet, L.; Garner, H.; Trouillet, C.; de Bruijn, M.F.; Geissmann, F.; et al. Tissue-resident macrophages originate from yolk-sac-derived erythro-myeloid progenitors. Nature 2015, 518, 547–551. [Google Scholar] [CrossRef]
- Hoeffel, G.; Wang, Y.; Greter, M.; See, P.; Teo, P.; Malleret, B.; Leboeuf, M.; Low, D.; Oller, G.; Almeida, F.; et al. Adult Langerhans cells derive predominantly from embryonic fetal liver monocytes with a minor contribution of yolk sac-derived macrophages. J. Exp. Med. 2012, 209, 1167–1181. [Google Scholar] [CrossRef] [PubMed]
- Jacome-Galarza, C.E.; Percin, G.I.; Muller, J.T.; Mass, E.; Lazarov, T.; Eitler, J.; Rauner, M.; Yadav, V.K.; Crozet, L.; Bohm, M.; et al. Developmental origin, functional maintenance and genetic rescue of osteoclasts. Nature 2019, 568, 541–545. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Hirschi, K.K. Tissue-Resident Macrophage Development and Function. Front. Cell Dev. Biol. 2020, 8, 617879. [Google Scholar] [CrossRef] [PubMed]
- Gritz, E.; Hirschi, K.K. Specification and function of hemogenic endothelium during embryogenesis. Cell Mol. Life Sci. 2016, 73, 1547–1567. [Google Scholar] [CrossRef]
- Wu, Y.; Hirschi, K.K. Regulation of Hemogenic Endothelial Cell Development and Function. Annu. Rev. Physiol. 2021, 83, 17–37. [Google Scholar] [CrossRef]
- Zovein, A.C.; Turlo, K.A.; Ponec, R.M.; Lynch, M.R.; Chen, K.C.; Hofmann, J.J.; Cox, T.C.; Gasson, J.C.; Iruela-Arispe, M.L. Vascular remodeling of the vitelline artery initiates extravascular emergence of hematopoietic clusters. Blood 2010, 116, 3435–3444. [Google Scholar] [CrossRef]
- Dzierzak, E.; Speck, N.A. Of lineage and legacy: The development of mammalian hematopoietic stem cells. Nat. Immunol. 2008, 9, 129–136. [Google Scholar] [CrossRef]
- Coşkun, S.; Chao, H.; Vasavada, H.; Heydari, K.; Gonzales, N.; Zhou, X.; de Crombrugghe, B.; Hirschi, K.K. Development of the fetal bone marrow niche and regulation of HSC quiescence and homing ability by emerging osteolineage cells. Cell Rep. 2014, 9, 581–590. [Google Scholar] [CrossRef]
- Laurenti, E.; Göttgens, B. From haematopoietic stem cells to complex differentiation landscapes. Nature 2018, 553, 418–426. [Google Scholar] [CrossRef]
- Anesi, A.; Generali, L.; Sandoni, L.; Pozzi, S.; Grande, A. From Osteoclast Differentiation to Osteonecrosis of the Jaw: Molecular and Clinical Insights. Int. J. Mol. Sci. 2019, 20, 4925. [Google Scholar] [CrossRef]
- Søe, K. Osteoclast Fusion: Physiological Regulation of Multinucleation through Heterogeneity-Potential Implications for Drug Sensitivity. Int. J. Mol. Sci. 2020, 21, 7717. [Google Scholar] [CrossRef] [PubMed]
- Hobolt-Pedersen, A.S.; Delaissé, J.M.; Søe, K. Osteoclast fusion is based on heterogeneity between fusion partners. Calcif. Tissue Int. 2014, 95, 73–82. [Google Scholar] [CrossRef] [PubMed]
- Søe, K.; Hobolt-Pedersen, A.S.; Delaisse, J.M. The elementary fusion modalities of osteoclasts. Bone 2015, 73, 181–189. [Google Scholar] [CrossRef] [PubMed]
- Feng, X.; Teitelbaum, S.L. Osteoclasts: New Insights. Bone Res. 2013, 1, 11–26. [Google Scholar] [CrossRef]
- Iwasaki, R.; Ninomiya, K.; Miyamoto, K.; Suzuki, T.; Sato, Y.; Kawana, H.; Nakagawa, T.; Suda, T.; Miyamoto, T. Cell fusion in osteoclasts plays a critical role in controlling bone mass and osteoblastic activity. Biochem. Biophys. Res. Commun. 2008, 377, 899–904. [Google Scholar] [CrossRef] [PubMed]
- Kodama, J.; Kaito, T. Osteoclast Multinucleation: Review of Current Literature. Int. J. Mol. Sci. 2020, 21, 5685. [Google Scholar] [CrossRef]
- Witwicka, H.; Hwang, S.-Y.; Reyes-Gutierrez, P.; Jia, H.; Odgren, P.E.; Donahue, L.R.; Birnbaum, M.J. Studies of OC-STAMP in Osteoclast Fusion: A New Knockout Mouse Model, Rescue of Cell Fusion, and Transmembrane Topology. PLoS ONE 2015, 10, e128275. [Google Scholar] [CrossRef]
- Sterling, H.; Saginario, C.; Vignery, A. CD44 occupancy prevents macrophage multinucleation. J. Cell Biol. 1998, 143, 837–847. [Google Scholar] [CrossRef]
- Takeda, Y.; Tachibana, I.; Miyado, K.; Kobayashi, M.; Miyazaki, T.; Funakoshi, T.; Kimura, H.; Yamane, H.; Saito, Y.; Goto, H.; et al. Tetraspanins CD9 and CD81 function to prevent the fusion of mononuclear phagocytes. J. Cell Biol. 2003, 161, 945–956. [Google Scholar] [CrossRef]
- Cui, W.; Ke, J.Z.; Zhang, Q.; Ke, H.-Z.; Chalouni, C.; Vignery, A. The intracellular domain of CD44 promotes the fusion of macrophages. Blood 2006, 107, 796–805. [Google Scholar] [CrossRef]
- Søe, K.; Andersen, T.L.; Hobolt-Pedersen, A.-S.; Bjerregaard, B.; Larsson, L.-I.; Delaissé, J.-M. Involvement of human endogenous retroviral syncytin-1 in human osteoclast fusion. Bone 2011, 48, 837–846. [Google Scholar] [CrossRef] [PubMed]
- Møller, A.M.; Delaissé, J.M.; Søe, K. Osteoclast Fusion: Time-Lapse Reveals Involvement of CD47 and Syncytin-1 at Different Stages of Nuclearity. J. Cell Physiol. 2017, 232, 1396–1403. [Google Scholar] [CrossRef] [PubMed]
- Yao, Z.; Getting, S.J.; Locke, I.C. Regulation of TNF-Induced Osteoclast Differentiation. Cells 2021, 11, 132. [Google Scholar] [CrossRef] [PubMed]
- Boyle, W.J.; Simonet, W.S.; Lacey, D.L. Osteoclast differentiation and activation. Nature 2003, 423, 337–342. [Google Scholar] [CrossRef]
- Edwards, J.R.; Mundy, G.R. Advances in osteoclast biology: Old findings and new insights from mouse models. Nat. Rev. Rheumatol. 2011, 7, 235–243. [Google Scholar] [CrossRef]
- Sun, Y.; Li, J.; Xie, X.; Gu, F.; Sui, Z.; Zhang, K.; Yu, T. Macrophage-Osteoclast Associations: Origin, Polarization, and Subgroups. Front. Immunol. 2021, 12, 778078. [Google Scholar] [CrossRef]
- Hettinger, J.; Richards, D.M.; Hansson, J.; Barra, M.M.; Joschko, A.-C.; Krijgsveld, J.; Feuerer, M. Origin of monocytes and macrophages in a committed progenitor. Nat. Immunol. 2013, 14, 821–830. [Google Scholar] [CrossRef]
- Mildner, A.; Marinkovic, G.; Jung, S. Murine Monocytes: Origins, Subsets, Fates, and Functions. Microbiol. Spectr. 2016, 4, 10–1128. [Google Scholar] [CrossRef]
- Narasimhan, P.B.; Marcovecchio, P.; Hamers, A.A.; Hedrick, C.C. Nonclassical Monocytes in Health and Disease. Annu. Rev. Immunol. 2019, 37, 439–456. [Google Scholar] [CrossRef]
- Speziani, C.; Rivollier, A.; Gallois, A.; Coury, F.; Mazzorana, M.; Azocar, O.; Flacher, M.; Bella, C.; Tebib, J.; Jurdic, P.; et al. Murine dendritic cell transdifferentiation into osteoclasts is differentially regulated by innate and adaptive cytokines. Eur. J. Immunol. 2007, 37, 747–757. [Google Scholar] [CrossRef]
- Pham, L.; Kaiser, B.; Romsa, A.; Schwarz, T.; Gopalakrishnan, R.; Jensen, E.D.; Mansky, K.C. HDAC3 and HDAC7 have opposite effects on osteoclast differentiation. J. Biol. Chem. 2011, 286, 12056–12065. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.-M.; Lin, C.; Stavre, Z.; Greenblatt, M.B.; Shim, J.-H. Osteoblast-Osteoclast Communication and Bone Homeostasis. Cells 2020, 9, 2073. [Google Scholar] [CrossRef] [PubMed]
- Tonna, S.; Takyar, F.M.; Vrahnas, C.; Crimeen-Irwin, B.; Ho, P.W.M.; Poulton, I.J.; Brennan, H.J.; McGregor, N.E.; Allan, E.H.; Nguyen, H.; et al. EphrinB2 signaling in osteoblasts promotes bone mineralization by preventing apoptosis. FASEB J. 2014, 28, 4482–4496. [Google Scholar] [CrossRef] [PubMed]
- Wajant, H. The Fas signaling pathway: More than a paradigm. Science 2002, 296, 1635–1636. [Google Scholar] [CrossRef] [PubMed]
- Krum, S.A.; Miranda-Carboni, G.A.; Hauschka, P.V.; Carroll, J.S.; Lane, T.F.; Freedman, L.P.; Brown, M. Estrogen protects bone by inducing Fas ligand in osteoblasts to regulate osteoclast survival. EMBO J. 2008, 27, 535–545. [Google Scholar] [CrossRef]
- Hayashi, M.; Nakashima, T.; Taniguchi, M.; Kodama, T.; Kumanogoh, A.; Takayanagi, H. Osteoprotection by semaphorin 3A. Nature 2012, 485, 69–74. [Google Scholar] [CrossRef]
- Boyce, B.F. Advances in the regulation of osteoclasts and osteoclast functions. J. Dent. Res. 2013, 92, 860–867. [Google Scholar] [CrossRef]
- Si, J.; Wang, C.; Zhang, D.; Wang, B.; Hou, W.; Zhou, Y. Osteopontin in Bone Metabolism and Bone Diseases. Med. Sci. Monit. 2020, 26, e919159. [Google Scholar] [CrossRef]
- Walsh, M.C.; Choi, Y. Biology of the RANKL-RANK-OPG System in Immunity, Bone, and Beyond. Front. Immunol. 2014, 5, 511. [Google Scholar] [CrossRef]
- Tang, Y.; Wu, X.; Lei, W.; Pang, L.; Wan, C.; Shi, Z.; Zhao, L.; Nagy, T.R.; Peng, X.; Hu, J.; et al. TGF-beta1-induced migration of bone mesenchymal stem cells couples bone resorption with formation. Nat. Med. 2009, 15, 757–765. [Google Scholar] [CrossRef]
- Xian, L.; Wu, X.; Pang, L.; Lou, M.; Rosen, C.J.; Qiu, T.; Crane, J.; Frassica, F.; Zhang, L.; Rodriguez, J.P.; et al. Matrix IGF-1 maintains bone mass by activation of mTOR in mesenchymal stem cells. Nat. Med. 2012, 18, 1095–1101. [Google Scholar] [CrossRef] [PubMed]
- Ishii, M.; Egen, J.G.; Klauschen, F.; Meier-Schellersheim, M.; Saeki, Y.; Vacher, J.; Proia, R.L.; Germain, R.N. Sphingosine-1-phosphate mobilizes osteoclast precursors and regulates bone homeostasis. Nature 2009, 458, 524–528. [Google Scholar] [CrossRef] [PubMed]
- Takeshita, S.; Fumoto, T.; Matsuoka, K.; Park, K.-A.; Aburatani, H.; Kato, S.; Ito, M.; Ikeda, K. Osteoclast-secreted CTHRC1 in the coupling of bone resorption to formation. J. Clin. Investig. 2013, 123, 3914–3924. [Google Scholar] [CrossRef] [PubMed]
- Ikebuchi, Y.; Aoki, S.; Honma, M.; Hayashi, M.; Sugamori, Y.; Khan, M.; Kariya, Y.; Kato, G.; Tabata, Y.; Penninger, J.M.; et al. Coupling of bone resorption and formation by RANKL reverse signalling. Nature 2018, 561, 195–200. [Google Scholar] [CrossRef] [PubMed]
- Bennett, C.N.; Longo, K.A.; Wright, W.S.; Suva, L.J.; Lane, T.F.; Hankenson, K.D.; MacDougald, O.A. Regulation of osteoblastogenesis and bone mass by Wnt10b. Proc. Natl. Acad. Sci. USA 2005, 102, 3324–3329. [Google Scholar] [CrossRef] [PubMed]
- Negishi-Koga, T.; Shinohara, M.; Komatsu, N.; Bito, H.; Kodama, T.; Friedel, R.H.; Takayanagi, H. Suppression of bone formation by osteoclastic expression of semaphorin 4D. Nat. Med. 2011, 17, 1473–1480. [Google Scholar] [CrossRef]
- O’Brien, J.; Hayder, H.; Zayed, Y.; Peng, C. Overview of MicroRNA Biogenesis, Mechanisms of Actions, and Circulation. Front. Endocrinol. 2018, 9, 402. [Google Scholar] [CrossRef]
- Yang, J.-X.; Xie, P.; Li, Y.-S.; Wen, T.; Yang, X.-C. Osteoclast-derived miR-23a-5p-containing exosomes inhibit osteogenic differentiation by regulating Runx2. Cell Signal 2020, 70, 109504. [Google Scholar] [CrossRef]
- Liu, Z.; Li, C.; Huang, P.; Hu, F.; Jiang, M.; Xu, X.; Li, B.; Deng, L.; Ye, T.; Guo, L. CircHmbox1 Targeting miRNA-1247-5p Is Involved in the Regulation of Bone Metabolism by TNF-α in Postmenopausal Osteoporosis. Front. Cell Dev. Biol. 2020, 8, 594785. [Google Scholar] [CrossRef]
- Delgado-Calle, J.; Bellido, T. The osteocyte as a signaling cell. Physiol. Rev. 2022, 102, 379–410. [Google Scholar] [CrossRef]
- Honma, M.; Ikebuchi, Y.; Kariya, Y.; Hayashi, M.; Hayashi, N.; Aoki, S.; Suzuki, H. RANKL subcellular trafficking and regulatory mechanisms in osteocytes. J. Bone Miner. Res. 2013, 28, 1936–1949. [Google Scholar] [CrossRef] [PubMed]
- Nakashima, T.; Hayashi, M.; Fukunaga, T.; Kurata, K.; Oh-Hora, M.; Feng, J.Q.; Bonewald, L.F.; Kodama, T.; Wutz, A.; Wagner, E.F.; et al. Evidence for osteocyte regulation of bone homeostasis through RANKL expression. Nat. Med. 2011, 17, 1231–1234. [Google Scholar] [CrossRef]
- Robling, A.G.; Bonewald, L.F. The Osteocyte: New Insights. Annu. Rev. Physiol. 2020, 82, 485–506. [Google Scholar] [CrossRef] [PubMed]
- Blanchard, F.; Duplomb, L.; Baud’huin, M.; Brounais, B. The dual role of IL-6-type cytokines on bone remodeling and bone tumors. Cytokine Growth Factor. Rev. 2009, 20, 19–28. [Google Scholar] [CrossRef] [PubMed]
- Harris, S.E.; MacDougall, M.; Horn, D.; Woodruff, K.; Zimmer, S.N.; Rebel, V.I.; Fajardo, R.; Feng, J.Q.; Gluhak-Heinrich, J.; Harris, M.A.; et al. Meox2Cre-mediated disruption of CSF-1 leads to osteopetrosis and osteocyte defects. Bone 2012, 50, 42–53. [Google Scholar] [CrossRef]
- Pathak, J.L.; Bakker, A.D.; Luyten, F.P.; Verschueren, P.; Lems, W.F.; Klein-Nulend, J.; Bravenboer, N. Systemic Inflammation Affects Human Osteocyte-Specific Protein and Cytokine Expression. Calcif. Tissue Int. 2016, 98, 596–608. [Google Scholar] [CrossRef]
- He, F.; Bai, J.; Wang, J.; Zhai, J.; Tong, L.; Zhu, G. Irradiation-induced osteocyte damage promotes HMGB1-mediated osteoclastogenesis in vitro. J. Cell Physiol. 2019, 234, 17314–17325. [Google Scholar] [CrossRef]
- Wang, Y.; Nishida, S.; Elalieh, H.Z.; Long, R.K.; Halloran, B.P.; Bikle, D.D. Role of IGF-I signaling in regulating osteoclastogenesis. J. Bone Miner. Res. 2006, 21, 1350–1358. [Google Scholar] [CrossRef]
- Kitaura, H.; Marahleh, A.; Ohori, F.; Noguchi, T.; Shen, W.-R.; Qi, J.; Nara, Y.; Pramusita, A.; Kinjo, R.; Mizoguchi, I. Osteocyte-Related Cytokines Regulate Osteoclast Formation and Bone Resorption. Int. J. Mol. Sci. 2020, 21, 5169. [Google Scholar] [CrossRef]
- Han, Y.; You, X.; Xing, W.; Zhang, Z.; Zou, W. Paracrine and endocrine actions of bone-the functions of secretory proteins from osteoblasts, osteocytes, and osteoclasts. Bone Res. 2018, 6, 16. [Google Scholar] [CrossRef]
- Muruganandan, S.; Ionescu, A.M.; Sinal, C.J. At the Crossroads of the Adipocyte and Osteoclast Differentiation Programs: Future Therapeutic Perspectives. Int. J. Mol. Sci. 2020, 21, 2277. [Google Scholar] [CrossRef] [PubMed]
- Muruganandan, S.; Sinal, C.J. The impact of bone marrow adipocytes on osteoblast and osteoclast differentiation. IUBMB Life 2014, 66, 147–155. [Google Scholar] [CrossRef] [PubMed]
- Wan, Y.; Chong, L.W.; Evans, R.M. PPAR-gamma regulates osteoclastogenesis in mice. Nat. Med. 2007, 13, 1496–1503. [Google Scholar] [CrossRef] [PubMed]
- Xie, H.; Tang, S.-Y.; Luo, X.-H.; Huang, J.; Cui, R.-R.; Yuan, L.-Q.; Zhou, H.-D.; Wu, X.-P.; Liao, E.-Y. Insulin-like effects of visfatin on human osteoblasts. Calcif. Tissue Int. 2007, 80, 201–210. [Google Scholar] [CrossRef] [PubMed]
- Moschen, A.R.; Geiger, S.; Gerner, R.; Tilg, H. Pre-B cell colony enhancing factor/NAMPT/visfatin and its role in inflammation-related bone disease. Mutat. Res. 2010, 690, 95–101. [Google Scholar] [CrossRef]
- Wu, S.-S.; Liang, Q.-H.; Liu, Y.; Cui, R.-R.; Yuan, L.-Q.; Liao, E.-Y. Omentin-1 Stimulates Human Osteoblast Proliferation through PI3K/Akt Signal Pathway. Int. J. Endocrinol. 2013, 2013, 368970. [Google Scholar] [CrossRef]
- Xie, H.; Xie, P.-L.; Luo, X.-H.; Wu, X.-P.; Zhou, H.-D.; Tang, S.-Y.; Liao, E.-Y. Omentin-1 exerts bone-sparing effect in ovariectomized mice. Osteoporos. Int. 2012, 23, 1425–1436. [Google Scholar] [CrossRef]
- Luo, T.; Liu, H.; Feng, W.; Liu, D.; Du, J.; Sun, J.; Wang, W.; Han, X.; Guo, J.; Amizuka, N.; et al. Adipocytes enhance expression of osteoclast adhesion-related molecules through the CXCL12/CXCR4 signalling pathway. Cell Prolif. 2017, 50, e12317. [Google Scholar] [CrossRef]
- Muruganandan, S.; Dranse, H.J.; Rourke, J.L.; McMullen, N.M.; Sinal, C.J. Chemerin neutralization blocks hematopoietic stem cell osteoclastogenesis. Stem Cells 2013, 31, 2172–2182. [Google Scholar] [CrossRef]
- Thommesen, L.; Stunes, A.K.; Monjo, M.; Grøsvik, K.; Tamburstuen, M.V.; Kjøbli, E.; Lyngstadaas, S.P.; Reseland, J.E.; Syversen, U. Expression and regulation of resistin in osteoblasts and osteoclasts indicate a role in bone metabolism. J. Cell Biochem. 2006, 99, 824–834. [Google Scholar] [CrossRef]
- Arron, J.R.; Choi, Y. Bone versus immune system. Nature 2000, 408, 535–536. [Google Scholar] [CrossRef] [PubMed]
- Okamoto, K.; Nakashima, T.; Shinohara, M.; Negishi-Koga, T.; Komatsu, N.; Terashima, A.; Sawa, S.; Nitta, T.; Takayanagi, H. Osteoimmunology: The Conceptual Framework Unifying the Immune and Skeletal Systems. Physiol. Rev. 2017, 97, 1295–1349. [Google Scholar] [CrossRef] [PubMed]
- Takayanagi, H. New immune connections in osteoclast formation. Ann. N. Y Acad. Sci. 2010, 1192, 117–123. [Google Scholar] [CrossRef] [PubMed]
- Kumar, G.; Roger, P.M. From Crosstalk between Immune and Bone Cells to Bone Erosion in Infection. Int. J. Mol. Sci. 2019, 20, 5154. [Google Scholar] [CrossRef]
- Grcević, D.; Lee, S.K.; Marusić, A.; Lorenzo, J.A. Depletion of CD4 and CD8 T lymphocytes in mice in vivo enhances 1,25-dihydroxyvitamin D3-stimulated osteoclast-like cell formation in vitro by a mechanism that is dependent on prostaglandin synthesis. J. Immunol. 2000, 165, 4231–4238. [Google Scholar] [CrossRef]
- John, V.; Hock, J.M.; Short, L.L.; Glasebrook, A.L.; Galvin, R.J. A role for CD8+ T lymphocytes in osteoclast differentiation in vitro. Endocrinology 1996, 137, 2457–2463. [Google Scholar] [CrossRef]
- Sato, K. Th17 cells and rheumatoid arthritis--from the standpoint of osteoclast differentiation. Allergol. Int. 2008, 57, 109–114. [Google Scholar] [CrossRef]
- Sato, K.; Takayanagi, H. Osteoclasts, rheumatoid arthritis, and osteoimmunology. Curr. Opin. Rheumatol. 2006, 18, 419–426. [Google Scholar] [CrossRef]
- Terheyden, H.; Stadlinger, B.; Sanz, M.; Garbe, A.I.; Meyle, J. Inflammatory reaction—Communication of cells. Clin. Oral. Implants Res. 2014, 25, 399–407. [Google Scholar] [CrossRef]
- Takayanagi, H.; Ogasawara, K.; Hida, S.; Chiba, T.; Murata, S.; Sato, K.; Takaoka, A.; Yokochi, T.; Oda, H.; Tanaka, K.; et al. T-cell-mediated regulation of osteoclastogenesis by signalling cross-talk between RANKL and IFN-gamma. Nature 2000, 408, 600–605. [Google Scholar] [CrossRef]
- Wing, K.; Yamaguchi, T.; Sakaguchi, S. Cell-autonomous and -non-autonomous roles of CTLA-4 in immune regulation. Trends Immunol. 2011, 32, 428–433. [Google Scholar] [CrossRef] [PubMed]
- Adamopoulos, I.E.; Bowman, E.P. Immune regulation of bone loss by Th17 cells. Arthritis Res. Ther. 2008, 10, 225. [Google Scholar] [CrossRef] [PubMed]
- Manabe, N.; Kawaguchi, H.; Chikuda, H.; Miyaura, C.; Inada, M.; Nagai, R.; Nabeshima, Y.-I.; Nakamura, K.; Sinclair, A.M.; Scheuermann, R.H.; et al. Connection between B lymphocyte and osteoclast differentiation pathways. J. Immunol. 2001, 167, 2625–2631. [Google Scholar] [CrossRef] [PubMed]
- Demoersman, J.; Pochard, P.; Framery, C.; Simon, Q.; Boisramé, S.; Soueidan, A.; Pers, J.-O. B cell subset distribution is altered in patients with severe periodontitis. PLoS ONE 2018, 13, e192986. [Google Scholar] [CrossRef]
- Chen, Y.; Wang, Y.; Tang, R.; Yang, J.; Dou, C.; Dong, Y.; Sun, D.; Zhang, C.; Zhang, L.; Tang, Y.; et al. Dendritic cells-derived interferon-λ1 ameliorated inflammatory bone destruction through inhibiting osteoclastogenesis. Cell Death Dis. 2020, 11, 414. [Google Scholar] [CrossRef]
- Zhao, Q.; Wang, X.; Liu, Y.; He, A.; Jia, R. NFATc1: Functions in osteoclasts. Int. J. Biochem. Cell Biol. 2010, 42, 576–579. [Google Scholar] [CrossRef]
- Combs, M.D.; Yutzey, K.E. VEGF and RANKL regulation of NFATc1 in heart valve development. Circ. Res. 2009, 105, 565–574. [Google Scholar] [CrossRef]
- Rana, Z.A.; Gundersen, K.; Buonanno, A. Activity-dependent repression of muscle genes by NFAT. Proc. Natl. Acad. Sci. USA 2008, 105, 5921–5926. [Google Scholar] [CrossRef]
- Klein-Hessling, S.; Muhammad, K.; Klein, M.; Pusch, T.; Rudolf, R.; Flöter, J.; Qureischi, M.; Beilhack, A.; Vaeth, M.; Kummerow, C.; et al. NFATc1 controls the cytotoxicity of CD8(+) T cells. Nat. Commun. 2017, 8, 511. [Google Scholar] [CrossRef]
- Zhou, T.; Gao, B.; Fan, Y.; Liu, Y.; Feng, S.; Cong, Q.; Zhang, X.; Zhou, Y.; Yadav, P.S.; Lin, J.; et al. Piezo1/2 mediate mechanotransduction essential for bone formation through concerted activation of NFAT-YAP1-ß-catenin. Elife 2020, 9, e52779. [Google Scholar] [CrossRef]
- Oikawa, T.; Kuroda, Y.; Matsuo, K. Regulation of osteoclasts by membrane-derived lipid mediators. Cell Mol. Life Sci. 2013, 70, 3341–3353. [Google Scholar] [CrossRef] [PubMed]
- Greenblatt, M.B.; Ritter, S.Y.; Wright, J.; Tsang, K.; Hu, D.; Glimcher, L.H.; Aliprantis, A.O. NFATc1 and NFATc2 repress spontaneous osteoarthritis. Proc. Natl. Acad. Sci. USA 2013, 110, 19914–19919. [Google Scholar] [CrossRef] [PubMed]
- Xu, S.; Cao, X.; Yu, Z.; He, W.; Pang, Y.; Lin, W.; Chen, Z.; Guo, W.; Lu, X.; Lin, C. Nicorandil Inhibits Osteoclast Formation Base on NF-κB and p-38 MAPK Signaling Pathways and Relieves Ovariectomy-Induced Bone Loss. Front. Pharmacol. 2021, 12, 726361. [Google Scholar] [CrossRef] [PubMed]
- Park, J.H.; Lee, N.K.; Lee, S.Y. Current Understanding of RANK Signaling in Osteoclast Differentiation and Maturation. Mol. Cells 2017, 40, 706–713. [Google Scholar] [CrossRef]
- Takayanagi, H.; Kim, S.; Koga, T.; Nishina, H.; Isshiki, M.; Yoshida, H.; Saiura, A.; Isobe, M.; Yokochi, T.; Inoue, J.-I.; et al. Induction and activation of the transcription factor NFATc1 (NFAT2) integrate RANKL signaling in terminal differentiation of osteoclasts. Dev. Cell 2002, 3, 889–901. [Google Scholar] [CrossRef]
- Aliprantis, A.O.; Ueki, Y.; Sulyanto, R.; Park, A.; Sigrist, K.S.; Sharma, S.M.; Ostrowski, M.C.; Olsen, B.R.; Glimcher, L.H. NFATc1 in mice represses osteoprotegerin during osteoclastogenesis and dissociates systemic osteopenia from inflammation in cherubism. J. Clin. Investig. 2008, 118, 3775–3789. [Google Scholar] [CrossRef]
- Miyamoto, T. Regulators of osteoclast differentiation and cell-cell fusion. Keio J. Med. 2011, 60, 101–105. [Google Scholar] [CrossRef]
- Kim, H.-M.; He, L.; Lee, S.; Park, C.; Kim, D.H.; Han, H.-J.; Han, J.; Hwang, J.; Cha-Molstad, H.; Lee, K.H.; et al. Inhibition of osteoclasts differentiation by CDC2-induced NFATc1 phosphorylation. Bone 2020, 131, 115153. [Google Scholar] [CrossRef]
- Yang, Y.; Chung, M.R.; Zhou, S.; Gong, X.; Xu, H.; Hong, Y.; Jin, A.; Huang, X.; Zou, W.; Dai, Q.; et al. STAT3 controls osteoclast differentiation and bone homeostasis by regulating NFATc1 transcription. J. Biol. Chem. 2019, 294, 15395–15407. [Google Scholar] [CrossRef]
- Kim, I.; Kim, J.H.; Kim, K.; Seong, S.; Lee, K.-B.; Kim, N. IRF2 enhances RANKL-induced osteoclast differentiation via regulating NF-κB/NFATc1 signaling. BMB Rep. 2021, 54, 482–487. [Google Scholar] [CrossRef]
- Kim, I.; Kim, J.H.; Kim, K.; Seong, S.; Kim, N. The IRF2BP2-KLF2 axis regulates osteoclast and osteoblast differentiation. BMB Rep. 2019, 52, 469–474. [Google Scholar] [CrossRef] [PubMed]
- Shyu, J.-F.; Liu, W.-C.; Zheng, C.-M.; Fang, T.-C.; Hou, Y.-C.; Chang, C.-T.; Liao, T.-Y.; Chen, Y.-C.; Lu, K.-C. Toxic Effects of Indoxyl Sulfate on Osteoclastogenesis and Osteoblastogenesis. Int. J. Mol. Sci. 2021, 22, 11265. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Mou, Y.; Gong, H.; Chen, H.; Xiao, H. Microphthalmia-Associated Transcription Factor in Senescence and Age-Related Diseases. Gerontology 2021, 67, 708–717. [Google Scholar] [CrossRef] [PubMed]
- Goding, C.R.; Arnheiter, H. MITF-the first 25 years. Genes. Dev. 2019, 33, 983–1007. [Google Scholar] [CrossRef]
- Asai, K.; Funaba, M.; Murakami, M. Enhancement of RANKL-induced MITF-E expression and osteoclastogenesis by TGF-β. Cell Biochem. Funct. 2014, 32, 401–409. [Google Scholar] [CrossRef]
- Pang, M.; Rodríguez-Gonzalez, M.; Hernandez, M.; Recinos, C.C.; Seldeen, K.L.; Troen, B.R. AP-1 and Mitf interact with NFATc1 to stimulate cathepsin K promoter activity in osteoclast precursors. J. Cell Biochem. 2019, 120, 12382–12392. [Google Scholar] [CrossRef]
- Lu, S.Y.; Li, M.; Lin, Y.L. Mitf regulates osteoclastogenesis by modulating NFATc1 activity. Exp. Cell Res. 2014, 328, 32–43. [Google Scholar] [CrossRef]
- Zhang, Y.; Ma, C.; Liu, C.; Wu, W. NF-κB promotes osteoclast differentiation by overexpressing MITF via down regulating microRNA-1276 expression. Life Sci. 2020, 258, 118093. [Google Scholar] [CrossRef]
- Chen, W.; Zhu, G.; Hao, L.; Wu, M.; Ci, H.; Li, Y.-P. C/EBPα regulates osteoclast lineage commitment. Proc. Natl. Acad. Sci. USA 2013, 110, 7294–7299. [Google Scholar] [CrossRef]
- Jules, J.; Chen, W.; Feng, X.; Li, Y.-P. C/EBPα transcription factor is regulated by the RANK cytoplasmic (535) IVVY (538) motif and stimulates osteoclastogenesis more strongly than c-Fos. J. Biol. Chem. 2018, 293, 1480–1492. [Google Scholar] [CrossRef]
- Chen, W.; Zhu, G.; Tang, J.; Zhou, H.-D.; Li, Y.-P. C/ebpα controls osteoclast terminal differentiation, activation, function, and postnatal bone homeostasis through direct regulation of Nfatc1. J. Pathol. 2018, 244, 271–282. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Zhu, G.; Jules, J.; Nguyen, D.; Li, Y.-P. Monocyte-Specific Knockout of C/ebpα Results in Osteopetrosis Phenotype, Blocks Bone Loss in Ovariectomized Mice, and Reveals an Important Function of C/ebpα in Osteoclast Differentiation and Function. J. Bone Miner. Res. 2018, 33, 691–703. [Google Scholar] [CrossRef] [PubMed]
- Jules, J.; Li, Y.P.; Chen, W. C/EBPα and PU.1 exhibit different responses to RANK signaling for osteoclastogenesis. Bone 2018, 107, 104–114. [Google Scholar] [CrossRef] [PubMed]
- Burda, P.; Laslo, P.; Stopka, T. The role of PU.1 and GATA-1 transcription factors during normal and leukemogenic hematopoiesis. Leukemia 2010, 24, 1249–1257. [Google Scholar] [CrossRef]
- Li, G.; Hao, W.; Hu, W. Transcription factor PU.1 and immune cell differentiation (Review). Int. J. Mol. Med. 2020, 46, 1943–1950. [Google Scholar] [CrossRef]
- Dean, A. PU.1 chromosomal dynamics are linked to LDB1. Blood 2018, 132, 2615–2616. [Google Scholar] [CrossRef]
- Carey, H.A.; Hildreth, B.E.; Geisler, J.A.; Nickel, M.C.; Cabrera, J.; Ghosh, S.; Jiang, Y.; Yan, J.; Lee, J.; Makam, S.; et al. Enhancer variants reveal a conserved transcription factor network governed by PU.1 during osteoclast differentiation. Bone Res. 2018, 6, 8. [Google Scholar] [CrossRef]
- de la Rica, L.; Rodríguez-Ubreva, J.; García, M.; Islam, A.B.; Urquiza, J.M.; Hernando, H.; Christensen, J.; Helin, K.; Gómez-Vaquero, C.; Ballestar, E. PU.1 target genes undergo Tet2-coupled demethylation and DNMT3b-mediated methylation in monocyte-to-osteoclast differentiation. Genome Biol. 2013, 14, R99. [Google Scholar] [CrossRef]
- Izawa, N.; Kurotaki, D.; Nomura, S.; Fujita, T.; Omata, Y.; Yasui, T.; Hirose, J.; Matsumoto, T.; Saito, T.; Kadono, Y.; et al. Cooperation of PU.1 With IRF8 and NFATc1 Defines Chromatin Landscapes During RANKL-Induced Osteoclastogenesis. J. Bone Miner. Res. 2019, 34, 1143–1154. [Google Scholar] [CrossRef]
- Tondravi, M.M.; McKercher, S.R.; Anderson, K.; Erdmann, J.M.; Quiroz, M.; Maki, R.; Teitelbaum, S.L. Osteopetrosis in mice lacking haematopoietic transcription factor PU. 1. Nature 1997, 386, 81–84. [Google Scholar] [CrossRef]
- Ishiyama, K.; Yashiro, T.; Nakano, N.; Kasakura, K.; Miura, R.; Hara, M.; Kawai, F.; Maeda, K.; Tamura, N.; Okumura, K.; et al. Involvement of PU.1 in NFATc1 promoter function in osteoclast development. Allergol. Int. 2015, 64, 241–247. [Google Scholar] [CrossRef]
- Jimi, E.; Takakura, N.; Hiura, F.; Nakamura, I.; Hirata-Tsuchiya, S. The Role of NF-κB in Physiological Bone Development and Inflammatory Bone Diseases: Is NF-κB Inhibition "Killing Two Birds with One Stone”? Cells 2019, 8, 1636. [Google Scholar] [CrossRef] [PubMed]
- Yamashita, T.; Yao, Z.; Li, F.; Zhang, Q.; Badell, I.R.; Schwarz, E.M.; Takeshita, S.; Wagner, E.F.; Noda, M.; Matsuo, K.; et al. NF-kappaB p50 and p52 regulate receptor activator of NF-kappaB ligand (RANKL) and tumor necrosis factor-induced osteoclast precursor differentiation by activating c-Fos and NFATc1. J. Biol. Chem. 2007, 282, 18245–18253. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Z.; Hou, X.; Yin, X.; Li, Y.; Duan, R.; Boyce, B.F.; Yao, Z. TNF Induction of NF-κB RelB Enhances RANKL-Induced Osteoclastogenesis by Promoting Inflammatory Macrophage Differentiation but also Limits It through Suppression of NFATc1 Expression. PLoS ONE 2015, 10, e135728. [Google Scholar] [CrossRef]
- Schmidt-Supprian, M.; Bloch, W.; Courtois, G.; Addicks, K.; Israël, A.; Rajewsky, K.; Pasparakis, M. NEMO/IKK gamma-deficient mice model incontinentia pigmenti. Mol. Cell 2000, 5, 981–992. [Google Scholar] [CrossRef]
- Bozec, A.; Bakiri, L.; Jimenez, M.; Schinke, T.; Amling, M.; Wagner, E.F. Fra-2/AP-1 controls bone formation by regulating osteoblast differentiation and collagen production. J. Cell Biol. 2010, 190, 1093–1106. [Google Scholar] [CrossRef]
- Mamun, A.A.; Asim, M.H.; Sahin, A.Z.; Al-Bari, A.A. Flavonoids compounds from Tridax procumbens inhibit osteoclast differentiation by down-regulating c-Fos activation. J. Cell Mol. Med. 2020, 24, 2542–2551. [Google Scholar] [CrossRef]
- Wu, X.; Li, Z.; Yang, Z.; Zheng, C.; Jing, J.; Chen, Y.; Ye, X.; Lian, X.; Qiu, W.; Yang, F.; et al. Caffeic acid 3,4-dihydroxy-phenethyl ester suppresses receptor activator of NF-κB ligand–induced osteoclastogenesis and prevents ovariectomy-induced bone loss through inhibition of mitogen-activated protein kinase/activator protein 1 and Ca2+–nuclear factor of activated T-cells cytoplasmic 1 signaling pathways. J. Bone Miner. Res. 2012, 27, 1298–1308. [Google Scholar] [CrossRef]
- Asagiri, M.; Takayanagi, H. The molecular understanding of osteoclast differentiation. Bone 2007, 40, 251–264. [Google Scholar] [CrossRef]
- Tong, X.; Chen, M.; Song, R.; Zhao, H.; Bian, J.; Gu, J.; Liu, Z. Overexpression of c-Fos reverses osteoprotegerin-mediated suppression of osteoclastogenesis by increasing the Beclin1-induced autophagy. J. Cell Mol. Med. 2021, 25, 937–945. [Google Scholar] [CrossRef]
- Wagner, E.F.; Eferl, R. Fos/AP-1 proteins in bone and the immune system. Immunol. Rev. 2005, 208, 126–140. [Google Scholar] [CrossRef] [PubMed]
- Okada, H.; Tanaka, S. Plasmalemmal interface for calcium signaling in osteoclast differentiation. Curr. Opin. Cell Biol. 2022, 74, 55–61. [Google Scholar] [CrossRef] [PubMed]
- Humphrey, M.B.; Daws, M.R.; Spusta, S.C.; Niemi, E.C.; A Torchia, J.; Lanier, L.L.; E Seaman, W.; Nakamura, M.C. TREM2, a DAP12-associated receptor, regulates osteoclast differentiation and function. J. Bone Miner. Res. 2006, 21, 237–245. [Google Scholar] [CrossRef] [PubMed]
- Cheng, B.; Li, X.; Dai, K.; Duan, S.; Rong, Z.; Chen, Y.; Lü, L.; Liu, Z.; Huang, X.; Xu, H.; et al. Triggering Receptor Expressed on Myeloid Cells-2 (TREM2) Interacts with Colony-Stimulating Factor 1 Receptor (CSF1R) but Is Not Necessary for CSF1/CSF1R-Mediated Microglial Survival. Front. Immunol. 2021, 12, 633796. [Google Scholar] [CrossRef] [PubMed]
- Humphrey, M.B.; Lanier, L.L.; Nakamura, M.C. Role of ITAM-containing adapter proteins and their receptors in the immune system and bone. Immunol. Rev. 2005, 208, 50–65. [Google Scholar] [CrossRef]
- Zou, W.; Teitelbaum, S.L. Absence of Dap12 and the αvβ3 integrin causes severe osteopetrosis. J. Cell Biol. 2015, 208, 125–136. [Google Scholar] [CrossRef]
- Zeng, X.-Z.; Zhang, Y.-Y.; Yang, Q.; Wang, S.; Zou, B.-H.; Tan, Y.-H.; Zou, M.; Liu, S.-W.; Li, X.-J. Artesunate attenuates LPS-induced osteoclastogenesis by suppressing TLR4/TRAF6 and PLCγ1-Ca2+-NFATc1 signaling pathway. Acta Pharmacol. Sin. 2020, 41, 229–236. [Google Scholar] [CrossRef]
- Yang, Z.; Kim, S.; Mahajan, S.; Zamani, A.; Faccio, R. Phospholipase Cγ1 (PLCγ1) Controls Osteoclast Numbers via Colony-stimulating Factor 1 (CSF-1)-dependent Diacylglycerol/β-Catenin/CyclinD1 Pathway. J. Biol. Chem. 2017, 292, 1178–1186. [Google Scholar] [CrossRef]
- Jang, H.-Y.; Kim, J.-M.; Kim, J.-S.; Kim, B.-S.; Lee, Y.-R.; Bae, J.S. Protaetia brevitarsis Extract Attenuates RANKL-Induced Osteoclastogenesis by Inhibiting the JNK/NF-κB/PLCγ2 Signaling Pathway. Nutrients 2023, 15, 3193. [Google Scholar] [CrossRef]
- Xin, Y.; Liu, Y.; Liu, D.; Li, J.; Zhang, C.; Wang, Y.; Zheng, S. New Function of RUNX2 in Regulating Osteoclast Differentiation via the AKT/NFATc1/CTSK Axis. Calcif. Tissue Int. 2020, 106, 553–566. [Google Scholar] [CrossRef]
- Yeon, J.-T.; Kim, K.-J.; Son, Y.-J.; Park, S.-J.; Kim, S.H. Idelalisib inhibits osteoclast differentiation and pre-osteoclast migration by blocking the PI3Kδ-Akt-c-Fos/NFATc1 signaling cascade. Arch. Pharm. Res. 2019, 42, 712–721. [Google Scholar] [CrossRef] [PubMed]
- Yan, D.Y.; Tang, J.; Chen, L.; Wang, B.; Weng, S.; Xie, Z.; Wu, Z.; Shen, Z.; Bai, B.; Yang, L. Imperatorin promotes osteogenesis and suppresses osteoclast by activating AKT/GSK3 β/β-catenin pathways. J. Cell Mol. Med. 2020, 24, 2330–2341. [Google Scholar] [CrossRef] [PubMed]
- Matsubara, T.; Yasuda, K.; Mizuta, K.; Kawaue, H.; Kokabu, S. Tyrosine Kinase Src Is a Regulatory Factor of Bone Homeostasis. Int. J. Mol. Sci. 2022, 23, 5508. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, S.; Nakamura, I.; Inoue, J.-I.; Oda, H.; Nakamura, K. Signal transduction pathways regulating osteoclast differentiation and function. J. Bone Miner. Metab. 2003, 21, 123–133. [Google Scholar] [CrossRef]
- Oikawa, T.; Oyama, M.; Kozuka-Hata, H.; Uehara, S.; Udagawa, N.; Saya, H.; Matsuo, K. Tks5-dependent formation of circumferential podosomes/invadopodia mediates cell-cell fusion. J. Cell Biol. 2012, 197, 553–568. [Google Scholar] [CrossRef]
- Ranea-Robles, P.; Galino, J.; Espinosa, L.; Schlüter, A.; Ruiz, M.; Calingasan, N.Y.; Villarroya, F.; Naudí, A.; Pamplona, R.; Ferrer, I.; et al. Modulation of mitochondrial and inflammatory homeostasis through RIP140 is neuroprotective in an adrenoleukodystrophy mouse model. Neuropathol. Appl. Neurobiol. 2022, 48, e12747. [Google Scholar] [CrossRef]
- Wang, Q.; Wu, Q. Knockdown of receptor interacting protein 140 (RIP140) alleviated lipopolysaccharide-induced inflammation, apoptosis and permeability in pulmonary microvascular endothelial cells by regulating C-terminal binding protein 2 (CTBP2). Bioengineered 2022, 13, 3981–3992. [Google Scholar] [CrossRef]
- Lin, Y.-W.; Liu, P.-S.; Adhikari, N.; Hall, J.L.; Wei, L.-N. RIP140 contributes to foam cell formation and atherosclerosis by regulating cholesterol homeostasis in macrophages. J. Mol. Cell Cardiol. 2015, 79, 287–294. [Google Scholar] [CrossRef]
- Piao, H.; Chu, X.; Lv, W.; Zhao, Y. Involvement of receptor-interacting protein 140 in estrogen-mediated osteoclasts differentiation, apoptosis, and bone resorption. J. Physiol. Sci. 2017, 67, 141–150. [Google Scholar] [CrossRef]
- Lee, B.; Iwaniec, U.T.; Turner, R.T.; Lin, Y.-W.; Clarke, B.L.; Gingery, A.; Wei, L.-N. RIP140 in monocytes/macrophages regulates osteoclast differentiation and bone homeostasis. JCI Insight 2017, 2, e90517. [Google Scholar] [CrossRef]
- Zanocco-Marani, T.; Vignudelli, T.; Parenti, S.; Gemelli, C.; Condorelli, F.; Martello, A.; Selmi, T.; Grande, A.; Ferrari, S. TFE3 transcription factor regulates the expression of MAFB during macrophage differentiation. Exp. Cell Res. 2009, 315, 1798–1808. [Google Scholar] [CrossRef] [PubMed]
- Baba, M.; Endoh, M.; Ma, W.; Toyama, H.; Hirayama, A.; Nishikawa, K.; Takubo, K.; Hano, H.; Hasumi, H.; Umemoto, T.; et al. Folliculin Regulates Osteoclastogenesis Through Metabolic Regulation. J. Bone Miner. Res. 2018, 33, 1785–1798. [Google Scholar] [CrossRef] [PubMed]
- Miyauchi, Y.; Ninomiya, K.; Miyamoto, H.; Sakamoto, A.; Iwasaki, R.; Hoshi, H.; Miyamoto, K.; Hao, W.; Yoshida, S.; Morioka, H.; et al. The Blimp1-Bcl6 axis is critical to regulate osteoclast differentiation and bone homeostasis. J. Exp. Med. 2010, 207, 751–762. [Google Scholar] [CrossRef]
- Yang, B.; Sun, H.; Jia, M.; He, Y.; Luo, Y.; Wang, T.; Wu, Y.; Wang, J. DNA damage-inducible transcript 3 restrains osteoclast differentiation and function. Bone 2021, 153, 116162. [Google Scholar] [CrossRef] [PubMed]
- Husain, A.; Jeffries, M.A. Epigenetics and Bone Remodeling. Curr. Osteoporos. Rep. 2017, 15, 450–458. [Google Scholar] [CrossRef] [PubMed]
- Ko, N.Y.; Chen, L.R.; Chen, K.H. The Role of Micro RNA and Long-Non-Coding RNA in Osteoporosis. Int. J. Mol. Sci. 2020, 21, 4886. [Google Scholar] [CrossRef]
- Hammond, S.M. An overview of microRNAs. Adv. Drug Deliv. Rev. 2015, 87, 3–14. [Google Scholar] [CrossRef]
- Chan, W.C.W.; Tan, Z.; To, M.K.T.; Chan, D. Regulation and Role of Transcription Factors in Osteogenesis. Int. J. Mol. Sci. 2021, 22, 5445. [Google Scholar] [CrossRef]
- Sugatani, T.; Hruska, K.A. Down-regulation of miR-21 biogenesis by estrogen action contributes to osteoclastic apoptosis. J. Cell Biochem. 2013, 114, 1217–1222. [Google Scholar] [CrossRef]
- Sugatani, T.; Vacher, J.; Hruska, K.A. A microRNA expression signature of osteoclastogenesis. Blood 2011, 117, 3648–3657. [Google Scholar] [CrossRef]
- Yuan, X.; Qian, N.; Ling, S.; Li, Y.; Sun, W.; Li, J.; Du, R.; Zhong, G.; Liu, C.; Yu, G.; et al. Breast cancer exosomes contribute to pre-metastatic niche formation and promote bone metastasis of tumor cells. Theranostics 2021, 11, 1429–1445. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.; Su, Y.; Lin, F.; Zhu, S.; Wang, J.; Hou, Y.; Du, J.; Liu, Y.; Guo, L. MicroRNA-21 promotes orthodontic tooth movement by modulating the RANKL/OPG balance in T cells. Oral. Dis. 2020, 26, 370–380. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Yang, Y.; Wang, J.; Yao, S.; Yao, T.; Xu, Y.; Chen, Z.; Yuan, P.; Gao, J.; Shen, S.; et al. miR-21-5p targets SKP2 to reduce osteoclastogenesis in a mouse model of osteoporosis. J. Biol. Chem. 2021, 296, 100617. [Google Scholar] [CrossRef] [PubMed]
- Cong, C.; Tian, J.; Gao, T.; Zhou, C.; Wang, Y.; Cui, X.; Zhu, L. lncRNA GAS5 Is Upregulated in Osteoporosis and Downregulates miR-21 to Promote Apoptosis of Osteoclasts. Clin. Interv. Aging 2020, 15, 1163–1169. [Google Scholar] [CrossRef] [PubMed]
- Guo, S.; Gu, J.; Ma, J.; Xu, R.; Wu, Q.; Meng, L.; Liu, H.; Li, L.; Xu, Y. GATA4-driven miR-206-3p signatures control orofacial bone development by regulating osteogenic and osteoclastic activity. Theranostics 2021, 11, 8379–8395. [Google Scholar] [CrossRef] [PubMed]
- Mizoguchi, F.; Murakami, Y.; Saito, T.; Miyasaka, N.; Kohsaka, H. miR-31 controls osteoclast formation and bone resorption by targeting RhoA. Arthritis Res. Ther. 2013, 15, R102. [Google Scholar] [CrossRef]
- Yu, L.; Sui, B.; Fan, W.; Lei, L.; Zhou, L.; Yang, L.; Diao, Y.; Zhang, Y.; Li, Z.; Liu, J.; et al. Exosomes derived from osteogenic tumor activate osteoclast differentiation and concurrently inhibit osteogenesis by transferring COL1A1-targeting miRNA-92a-1-5p. J. Extracell. Vesicles 2021, 10, e12056. [Google Scholar] [CrossRef]
- Nakao, Y.; Fukuda, T.; Zhang, Q.; Sanui, T.; Shinjo, T.; Kou, X.; Chen, C.; Liu, D.; Watanabe, Y.; Hayashi, C.; et al. Exosomes from TNF-α-treated human gingiva-derived MSCs enhance M2 macrophage polarization and inhibit periodontal bone loss. Acta Biomater. 2021, 122, 306–324. [Google Scholar] [CrossRef]
- Sun, L.; Lian, J.X.; Meng, S. MiR-125a-5p promotes osteoclastogenesis by targeting TNFRSF1B. Cell Mol Biol Lett 2019, 24, 23. [Google Scholar] [CrossRef]
- Wang, Q.; Shen, X.; Chen, Y.; Chen, J.; Li, Y. Osteoblasts-derived exosomes regulate osteoclast differentiation through miR-503-3p/Hpse axis. Acta Histochem. 2021, 123, 151790. [Google Scholar] [CrossRef]
- Inoue, K.; Hu, X.; Zhao, B. Regulatory network mediated by RBP-J/NFATc1-miR182 controls inflammatory bone resorption. FASEB J. 2020, 34, 2392–2407. [Google Scholar] [CrossRef] [PubMed]
- Guo, K.; Zhang, D.; Wu, H.; Zhu, Q.; Yang, C.; Zhu, J. MiRNA-199a-5p positively regulated RANKL-induced osteoclast differentiation by target Mafb protein. J. Cell Biochem. 2019, 120, 7024–7031. [Google Scholar] [CrossRef] [PubMed]
- Dinesh, P.; Kalaiselvan, S.; Sujitha, S.; Rasool, M. miR-506-3p alleviates uncontrolled osteoclastogenesis via repression of RANKL/NFATc1 signaling pathway. J. Cell Physiol. 2020, 235, 9497–9509. [Google Scholar] [CrossRef] [PubMed]
- Yin, Y.; Tang, L.; Chen, J.; Lu, X. MiR-30a attenuates osteoclastogenesis via targeting DC-STAMP-c-Fos-NFATc1 signaling. Am. J. Transl. Res. 2017, 9, 5743–5753. [Google Scholar]
- Li, K.; Chen, S.; Cai, P.; Chen, K.; Li, L.; Yang, X.; Yi, J.; Luo, X.; Du, Y.; Zheng, H. MiRNA-483-5p is involved in the pathogenesis of osteoporosis by promoting osteoclast differentiation. Mol. Cell. Probes 2020, 49, 101479. [Google Scholar] [CrossRef]
- Li, Z.; Zhang, W.; Huang, Y. MiRNA-133a is involved in the regulation of postmenopausal osteoporosis through promoting osteoclast differentiation. Acta Biochim. Biophys. Sin. 2018, 50, 273–280. [Google Scholar] [CrossRef]
- Lee, Y.; Kim, H.J.; Park, C.K.; Kim, Y.-G.; Lee, H.-J.; Kim, J.-Y.; Kim, H.-H. MicroRNA-124 regulates osteoclast differentiation. Bone 2013, 56, 383–389. [Google Scholar] [CrossRef]
- Zhao, C.; Sun, W.; Zhang, P.; Ling, S.; Li, Y.; Zhao, D.; Peng, J.; Wang, A.; Li, Q.; Song, J.; et al. miR-214 promotes osteoclastogenesis by targeting Pten/PI3k/Akt pathway. RNA Biol. 2015, 12, 343–353. [Google Scholar] [CrossRef]
- Zhao, H.; Zhang, J.; Shao, H.; Liu, J.; Jin, M.; Chen, J.; Huang, Y. miRNA-340 inhibits osteoclast differentiation via repression of MITF. Biosci. Rep. 2017, 37. [Google Scholar] [CrossRef]
- Liu, L.-L.; Xiao, Y.-S.; Huang, W.-M.; Liu, S.; Huang, L.-X.; Zhong, J.-H.; Jia, P.; Liu, W.-Y. ATF1/miR-214-5p/ITGA7 axis promotes osteo-clastogenesis to alter OVX-induced bone absorption. Mol. Med. 2022, 28, 56. [Google Scholar] [CrossRef]
- Dou, C.; Zhang, C.; Kang, F.; Yang, X.; Jiang, H.; Bai, Y.; Xiang, J.; Xu, J.; Dong, S. MiR-7b directly targets DC-STAMP causing suppression of NFATc1 and c-Fos signaling during osteoclast fusion and differentiation. Biochim. Biophys. Acta 2014, 1839, 1084–1096. [Google Scholar] [CrossRef] [PubMed]
- Shen, L.; Wu, C.; Zhang, J.; Xu, H.; Liu, X.; Wu, X.; Wang, T.; Mao, L. Roles and potential applications of lncRNAs in HIV infection. Int. J. Infect. Dis. 2020, 92, 97–104. [Google Scholar] [CrossRef]
- Xing, C.; Sun, S.-G.; Yue, Z.-Q.; Bai, F. Role of lncRNA LUCAT1 in cancer. Biomed. Pharmacother. 2021, 134, 111158. [Google Scholar] [CrossRef] [PubMed]
- Du, Y.-J.; Yu, Q.-Q.; Zheng, X.-F.; Wang, S.-P. LncRNA TUG1 positively regulates osteoclast differentiation by targeting v-maf musculoaponeurotic fibrosarcoma oncogene homolog B. Autoimmunity 2020, 53, 443–449. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Cao, Z.; Bai, Y.; Dou, C.; Gong, X.; Liang, M.; Dong, R.; Quan, H.; Li, J.; Dai, J.; et al. LncRNA AK077216 promotes RANKL-induced osteoclastogenesis and bone resorption via NFATc1 by inhibition of NIP45. J. Cell. Physiol. 2019, 234, 1606–1617. [Google Scholar] [CrossRef]
- Ni, J.; Zhang, X.; Li, J.; Zheng, Z.; Zhang, J.; Zhao, W.; Liu, L. Tumour-derived exosomal lncRNA-SOX2OT promotes bone metastasis of non-small cell lung cancer by targeting the miRNA-194-5p/RAC1 signalling axis in osteoclasts. Cell Death Dis. 2021, 12, 662. [Google Scholar] [CrossRef]
- Yu, H.; Zhou, W.; Yan, W.; Xu, Z.; Xie, Y.; Zhang, P. LncRNA CASC11 is upregulated in postmenopausal osteoporosis and is correlated with TNF-α. Clin. Interv. Aging 2019, 14, 1663–1669. [Google Scholar] [CrossRef]
- Li, W.; Zhang, B.; Zhu, H.; Huang, S.; Xu, H. CRNDE impacts the proliferation of osteoclast by estrogen deficiency in postmenopausal osteoporosis. Eur. Rev. Med. Pharmacol. Sci. 2018, 22, 5815–5821. [Google Scholar] [CrossRef]
- Ma, X.; Guo, Z.; Gao, W.; Wang, J.; Liu, Y.; Gao, F.; Sun, S.; Zhou, X.; Yang, Z.; Zheng, W. LncRNA-NEF is downregulated in postmenopausal osteoporosis and is related to course of treatment and recurrence. J. Int. Med. Res. 2019, 47, 3299–3306. [Google Scholar] [CrossRef]
- Huang, S.; Zhu, X.; Xiao, D.; Zhuang, J.; Liang, G.; Liang, C.; Zheng, X.; Ke, Y.; Chang, Y. LncRNA SNHG1 was down-regulated after menopause and participates in postmenopausal osteoporosis. Biosci. Rep. 2019, 39, BSR20190445. [Google Scholar] [CrossRef]
- Zhang, H.L.; Du, X.Y.; Dong, Q.R. LncRNA XIXT promotes osteogenic differentiation of bone mesenchymal stem cells and alleviates osteoporosis progression by targeting miRNA-30a-5p. Eur. Rev. Med. Pharmacol. Sci. 2019, 23, 8721–8729. [Google Scholar] [CrossRef] [PubMed]
- Chen, R.-S.; Zhang, X.-B.; Zhu, X.-T.; Wang, C.-S. LncRNA Bmncr alleviates the progression of osteoporosis by inhibiting RANML-induced osteoclast differentiation. Eur. Rev. Med. Pharmacol. Sci. 2019, 23, 9199–9206. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.P.; Huang, Y.N.; Nithiyanantham, S.; Huang, C.; Ko, Y. LncRNA-Jak3:Jak3 coexpressed pattern regulates monosodium urate crystal-induced osteoclast differentiation through Nfatc1/Ctsk expression. Environ. Toxicol. 2019, 34, 179–187. [Google Scholar] [CrossRef]
- Zhang, D.-W.; Wang, H.-G.; Zhang, K.-B.; Guo, Y.-Q.; Yang, L.-J.; Lv, H. LncRNA XIST facilitates S1P-mediated osteoclast differentiation via interacting with FUS. J. Bone Miner. Metab. 2022, 40, 240–250. [Google Scholar] [CrossRef]
- Li, J.; Jin, F.; Cai, M.; Lin, T.; Wang, X.; Sun, Y. LncRNA Nron Inhibits Bone Resorption in Periodontitis. J. Dent. Res. 2022, 101, 187–195. [Google Scholar] [CrossRef] [PubMed]
- Ling, L.; Hu, H.-L.; Liu, K.-Y.; Ram, Y.-I.; Gao, J.-L.; Cao, Y.-M. Long noncoding RNA MIRG induces osteoclastogenesis and bone resorption in osteoporosis through negative regulation of miR-1897. Eur. Rev. Med. Pharmacol. Sci. 2019, 23, 10195–10203. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Xu, R.; Li, B.; Xin, Z.; Ling, Z.; Zhu, W.; Li, X.; Zhang, P.; Fu, Y.; Chen, J.; et al. LncRNA NEAT1 controls the lineage fates of BMSCs during skeletal aging by impairing mitochondrial function and pluripotency maintenance. Cell Death Differ. 2022, 29, 351–365. [Google Scholar] [CrossRef]
- Zhang, C.; Yang, J.; Zhu, Z.; Qin, J.; Yang, L.; Zhao, X.; Su, W.; Cai, Y.; Yang, J.; Wang, F.; et al. Exosomal lncRNA HOTAIR promotes osteoclast differ-entiation by targeting TGF-β/PTHrP/RANKL pathway. Basic. Clin. Pharmacol. Toxicol. 2023, 132, 242–252. [Google Scholar] [CrossRef]
- Wang, Y.; Zhu, G.; Pei, F.; Zhao, Z. lncRNA SNHG15 as a ceRNA modulates Osteoclast Dif-ferentiation, Proliferation, and Metastasis by Sponging miR-381-3p/NEK2 Axis. J. Immunol. Res. 2022, 2022, 8634820. [Google Scholar] [CrossRef]
- Chen, L.; Wang, C.; Sun, H.; Wang, J.; Liang, Y.; Wang, Y.; Wong, G. The bioinformatics toolbox for circRNA discovery and analysis. Brief. Bioinform. 2021, 22, 1706–1728. [Google Scholar] [CrossRef]
- Zhao, K.; Zhao, Q.; Guo, Z.; Chen, Z.; Hu, Y.; Su, J.; Chen, L.; He, Z.; Cai, X.; Chen, M.; et al. Hsa_Circ_0001275: A Potential Novel Diagnostic Biomarker for Postmenopausal Osteoporosis. Cell. Physiol. Biochem. 2018, 46, 2508–2516. [Google Scholar] [CrossRef] [PubMed]
- Gao, M.; Zhang, Z.; Sun, J.; Li, B.; Li, Y. The roles of circRNA-miRNA-mRNA networks in the development and treatment of osteoporosis. Front. Endocrinol. 2022, 13, 945310. [Google Scholar] [CrossRef] [PubMed]
- Gu, C.; Wang, W.; Tang, X.; Xu, T.; Zhang, Y.; Guo, M.; Wei, R.; Wang, Y.; Jurczyszyn, A.; Janz, S.; et al. CHEK1 and circCHEK1_246aa evoke chromosomal instability and induce bone lesion formation in multiple myeloma. Mol. Cancer 2021, 20, 84. [Google Scholar] [CrossRef]
- Chen, X.; Ouyang, Z.; Shen, Y.; Liu, B.; Zhang, Q.; Wan, L.; Yin, Z.; Zhu, W.; Li, S.; Peng, D. CircRNA_28313/miR-195a/CSF1 axis modulates osteoclast differentiation to affect OVX-induced bone absorption in mice. RNA Biol. 2019, 16, 1249–1262. [Google Scholar] [CrossRef] [PubMed]
- Miao, F.; Yin, B.-H.; Zhang, X.; Xue, D.-D.; Ma, C. CircRNA_009934 induces osteoclast bone resorption via silencing miR-5107. Eur. Rev. Med. Pharmacol. Sci. 2020, 24, 7580–7588. [Google Scholar] [CrossRef]
- Wang, W.; Qiao, S.-C.; Wu, X.-B.; Sun, B.; Yang, J.-G.; Li, X.; Zhang, X.; Qian, S.-J.; Gu, Y.-X.; Lai, H.-C. Circ_0008542 in osteoblast exosomes promotes osteoclast-induced bone resorption through m6A methylation. Cell Death Dis. 2021, 12, 628. [Google Scholar] [CrossRef]
- Liu, S.; Wang, C.; Bai, J.; Li, X.; Yuan, J.; Shi, Z.; Mao, N. Involvement of circRNA_0007059 in the regulation of postmenopausal osteoporosis by promoting the microRNA-378/BMP-2 axis. Cell Biol. Int. 2021, 45, 447–455. [Google Scholar] [CrossRef]
- Liu, J.; Liu, J.; Duan, S.; Liu, L.; Zhang, G.; Peng, X. Reprogrammed Epigenetic Landscape-Prophesied Functions of Bioactive Polysaccharides in Alleviating Diseases: A Pilot Study of DNA Methylome Remodeling in Astragalus Polysaccharide (APS)-Improved Osteoporosis in a Rat Model. J. Agric. Food Chem. 2020, 68, 15449–15459. [Google Scholar] [CrossRef]
- Peng, R.; Dong, Y.; Kang, H.; Guo, Q.; Zhu, M.; Li, F. Identification of Genes with Altered Methylation in Osteoclast Differentiation and Its Roles in Osteoporosis. DNA Cell Biol. 2022, 41, 575–589. [Google Scholar] [CrossRef]
- Møller, A.; Delaissé, J.-M.; Olesen, J.B.; Madsen, J.S.; Canto, L.M.; Bechmann, T.; Rogatto, S.R.; Søe, K. Aging and menopause reprogram osteoclast precursors for aggressive bone resorption. Bone Res. 2020, 8, 27. [Google Scholar] [CrossRef]
- Lawrence, M.; Daujat, S.; Schneider, R. Lateral Thinking: How Histone Modifications Regulate Gene Expression. Trends Genet. 2016, 32, 42–56. [Google Scholar] [CrossRef]
- Füllgrabe, J.; Kavanagh, E.; Joseph, B. Histone onco-modifications. Oncogene 2011, 30, 3391–3403. [Google Scholar] [CrossRef] [PubMed]
- Haberland, M.; Montgomery, R.L.; Olson, E.N. The many roles of histone deacetylases in development and physiology: Implications for disease and therapy. Nat. Rev. Genet. 2009, 10, 32–42. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Kim, K.; Youn, B.U.; Jin, H.M.; Kim, J.-Y.; Moon, J.B.; Ko, A.; Seo, S.-B.; Lee, K.-Y.; Kim, N. RANKL induces NFATc1 acetylation and stability via histone acetyltransferases during osteoclast differentiation. Biochem. J. 2011, 436, 253–262. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.; Lee, J.; Kim, J.H.; Jin, H.M.; Zhou, B.; Lee, S.Y.; Kim, N. Protein inhibitor of activated STAT 3 modulates osteoclastogenesis by down-regulation of NFATc1 and osteoclast-associated receptor. J. Immunol. 2007, 178, 5588–5594. [Google Scholar] [CrossRef] [PubMed]
- Jin, Z.; Wei, W.; Huynh, H.; Wan, Y. HDAC9 Inhibits Osteoclastogenesis via Mutual Suppression of PPARγ/RANKL Signaling. Mol. Endocrinol. 2015, 29, 730–738. [Google Scholar] [CrossRef]
- Blixt, N.C.; Faulkner, B.K.; Astleford, K.; Lelich, R.; Schering, J.; Spencer, E.; Gopalakrishnan, R.; Jensen, E.D.; Mansky, K.C. Class II and IV HDACs function as inhibitors of osteoclast differentiation. PLoS ONE 2017, 12, e185441. [Google Scholar] [CrossRef]
- Shin, Y.; Ghate, N.B.; Moon, B.; Park, K.; Lu, W.; An, W. DNMT and HDAC inhibitors modulate MMP-9-dependent H3 N-terminal tail proteolysis and Osteoclastogenesis. Epigenetics Chromatin 2019, 12, 25. [Google Scholar] [CrossRef]
- Nishikawa, K.; Iwamoto, Y.; Kobayashi, Y.; Katsuoka, F.; Kawaguchi, S.-I.; Tsujita, T.; Nakamura, T.; Kato, S.; Yamamoto, M.; Takayanagi, H.; et al. DNA methyltransferase 3a regulates osteoclast differentiation by coupling to an S-adenosylmethionine-producing metabolic pathway. Nat. Med. 2015, 21, 281–287. [Google Scholar] [CrossRef]
| Drug Names | Functions | Side Effects | References | |
|---|---|---|---|---|
| Anti-resorptive | ||||
| RANKL antibody | Denosumab (Prolia) | Inhibiting the formation, function, and survival of osteoclasts | Rebound vertebral fractures | [12,13] |
| Selective Estrogen Receptor Modulators (SERMs) | Raloxifene (Evista) | Regulating the survival of mature osteoclasts | Venous thromboembolism and stroke | [13,18] |
| Cathepsin K Inhibitors | Odanacatib (III clinical trials) | Inhibiting the late differentiation of osteoclasts | Stroke | [14] |
| Calcitonin | Calcitonin (Miacalcin) | Blocking the maturation of osteoclast precursors and regulating the function of osteoclasts | Rhinitis, nasal irritation, back pain, nosebleed, and headache | [13,19] |
| Bisphosphonates (BPs) | Alendronate (Binosto, Fosamax), risedronate (Actonel, Atelvia), ibandronate (Boniva, Bondronat) | Induction of apoptosis in osteoclasts | Atypical femoral fractures, osteonecrosis of the jaw, gastrointestinal and renal complications, osteomalacia | [20,21,22] |
| Anabolic | ||||
| Strontium | Strontium ranelate (Protelos) | Interfering with osteoclast differentiation and promoting osteoclast differentiation | Heart problems | [23] |
| MiRNAs | Roles | Target Gene(s) | References |
|---|---|---|---|
| MiR-21 | Inhibiting apoptosis of osteoclasts Promoting osteoclast differentiation | FasL PDCD4 | [189,190,191] |
| MiR-21-5p | Inhibiting osteoclast differentiation Promoting apoptosis of osteoclasts | SKP2 | [193,194] |
| MiR-31 | Promoting osteoclast skeleton formation | RhoA | [196] |
| MiR-92a-1-5p | Promoting osteoclast differentiation and repressing osteoblastogenesis | COL1A1 | [197] |
| MiR-1260b | Inhibiting osteoclast differentiation | Wnt5a | [198] |
| MiR-503-3p | Inhibiting osteoclast differentiation | Hpse | [200] |
| MiR-182 | Promoting osteoclast differentiation | RBP-J and NFATc1 | [201] |
| MiR-199a-5p | Promoting osteoclast differentiation | Mafb | [202] |
| MiR-506-3p | Inhibiting osteoclast differentiation and the release of bone-resorbing enzymes | RANKL and NFATc1 | [203] |
| MiR-206-3p | Inhibiting osteoclast differentiation | BMP3 and NFATc1 | [195] |
| MiR-30a | Inhibiting osteoclast differentiation and skeleton formation | DC-STAMP, c-Fos, NFATc1 | [204] |
| MiR-483-5p | Promoting osteoclast differentiation | NFATc1, NFAT2, CTSK | [205] |
| MiR-133a | Promoting osteoclast differentiation | NFATc1, c-Fos, TRAP | [206] |
| MiR-124 | Inhibiting osteoclast differentiation and promoting osteoclast differentiation | NFATc1 Pten/PI3K/Akt | [207,208] |
| MiR-340 | Inhibiting osteoclast differentiation | MITF | [209] |
| MiR-1276 | Inhibiting osteoclast differentiation | MITF | [138] |
| MiR-214-5p | Promoting osteoclast differentiation | ITGA7 | [210] |
| MiR-7b | Inhibiting osteoclast differentiation | DC-STAMP | [211] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jiang, T.; Xia, T.; Qiao, F.; Wang, N.; Jiang, Y.; Xin, H. Role and Regulation of Transcription Factors in Osteoclastogenesis. Int. J. Mol. Sci. 2023, 24, 16175. https://doi.org/10.3390/ijms242216175
Jiang T, Xia T, Qiao F, Wang N, Jiang Y, Xin H. Role and Regulation of Transcription Factors in Osteoclastogenesis. International Journal of Molecular Sciences. 2023; 24(22):16175. https://doi.org/10.3390/ijms242216175
Chicago/Turabian StyleJiang, Tao, Tianshuang Xia, Fangliang Qiao, Nani Wang, Yiping Jiang, and Hailiang Xin. 2023. "Role and Regulation of Transcription Factors in Osteoclastogenesis" International Journal of Molecular Sciences 24, no. 22: 16175. https://doi.org/10.3390/ijms242216175





