1. Introduction
Head and neck squamous cell carcinomas (HNSCC) are a heterogeneous but common malignancy with a worldwide incidence of more than 800,000 cases every year [
1]. The disease arises from the mucosal epithelia of the oral cavity, pharynx, and larynx. The therapy of choice primarily depends on tumor stage and tumor localization and comprises surgery, radiotherapy, and systemic therapy. Tumors in the oral cavity are predominantly treated surgically, while radiotherapy is commonly used as adjuvant therapy and for unresectable tumors [
2]. Despite multimodal treatment regimens, 5-year survival rates have remained around 40–60% over the past decades [
3,
4,
5,
6,
7]. Locoregional disease recurrence and metastasis are the main predicting factors for adverse clinical outcomes in affected patients, as prospects for curative treatment are limited [
8,
9]. Immunotherapy with the programmed death 1 inhibitor pembrolizumab has been established as the first-line therapy for recurrent and metastatic disease and as a monotherapy for programmed death ligand 1 (PD-L1)–positive disease or with platinum plus fluorouracil independent of PD-L1 status [
10,
11]. Together with nivolumab, pembrolizumab is recommended for second-line treatment of recurrent and metastatic HNSCC after progression on or after platinum-containing therapy [
11]. Therefore, immune checkpoint inhibition targeting the PD-1/PD-L1 axis has brought a significant improvement in disease control and long-term outcome for a relevant number of patients [
10,
12,
13,
14,
15,
16].
The up-regulation of PD-L1 has been demonstrated for a variety of malignancies including HNSCC and the expression of PD-L1 in tumor cells has been identified as part of an immune escape mechanism to evade detection by effector cells of the immune system [
17,
18]. Several authors have described a correlation between high PD-L1 expression with the development of cervical metastases and poor clinical outcome in HNSCC [
17,
19,
20,
21]. Furthermore, it has been shown that PD-L1 expression is predictive of treatment response on immune checkpoint inhibition with increased efficacy of therapy in patients with higher PD-L1 expression [
15,
22].
In a previous study, we demonstrated higher PD-L1 levels in lymph node metastases compared to their corresponding primary tumors [
19]. Since the main application of immune checkpoint inhibition is for recurrent or metastatic HNSCC, there is an urgent need to investigate the dynamics of PD-L1 expression during disease progression. Therefore, we analyzed local PD-L1 expression level recurrence and their corresponding primary tumors in a large cohort of surgically treated patients with oral squamous cell carcinomas. In addition, we investigated the resulting impact of PD-L1 expression on survival.
4. Discussion
In the present study, we investigated the expression of PD-L1 in primary and recurrent oral squamous cell carcinoma in a cohort of patients who underwent primary surgical treatment followed by radio(chemo)therapy, if required. The role of immune-modulatory checkpoint mechanisms in oral carcinomas is not yet fully understood. Their regulation in the recurrence of disease has not yet been clarified, and the data regarding their predictive value are controversial. This study aimed to further characterize the dynamic expression profile of PD-L1 in the disease progression of oral squamous cell carcinoma and assess its role as a prognosticator.
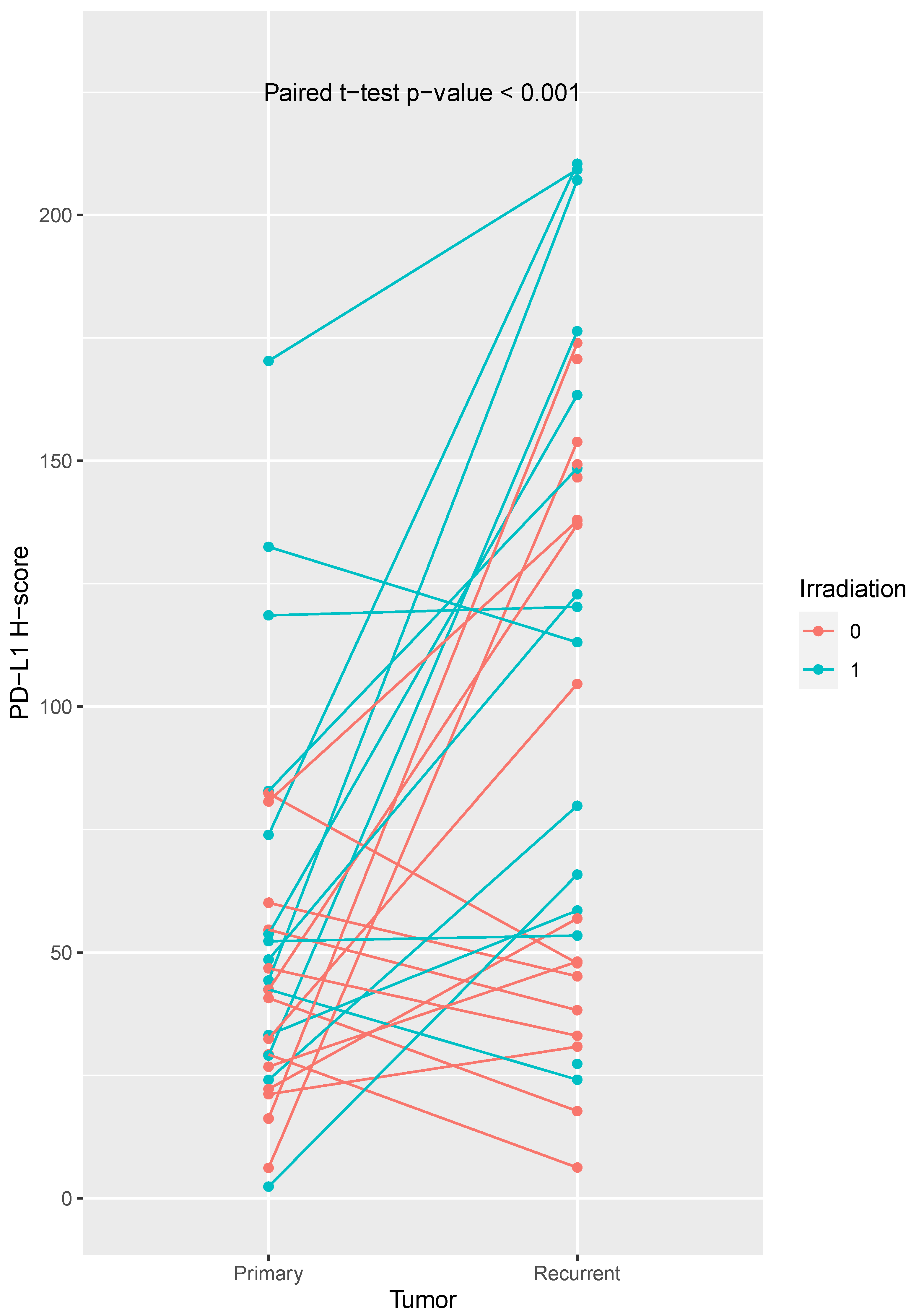
In this study, we found a mean PD-L1 H-score in primary tumors of 47.1 ± 31.4, while the mean PD-L1 H-Score in recurrent tumors was 103.53 ± 62.78. The PD-L1 expression differed between primary tumors and their matched recurrent tumors (paired t-test: p < 0.001).
We demonstrated a longitudinal heterogeneity of PD-L1 expression with higher expression levels in local and cervical recurrences of oral squamous cell carcinoma compared to their matched primary tumor. Our findings are consistent with a previous assessment of longitudinal PD-L1 expression in head and neck cancer, where a 33–36% temporal heterogeneity of PD-L1 expression has been reported depending on the chosen threshold used for CPS scoring [
26]. In this context, we want to emphasize that head and neck cancer summarizes different tumor entities of distinct localizations with relevant tumor heterogeneity and wide disparity regarding their prognosis. Importantly, qualitative differences in PD-L1 expression between oral and oropharyngeal squamous cell carcinoma have been presented [
27]. We previously reported a PD-L1 up-regulation in lymph node metastases in the primary setting, although we used a different methodology at that time [
19]. These observations regarding a heterogeneous PD-L1 expression in the context of lymph node metastasis have been similarly reported by others [
28,
29].
In addition, we observed an increased PD-L1 expression in recurrent tumors from irradiated patients, which has also been reported by others [
30,
31,
32]. A recent study investigated the modulation of PD-L1 expression in irradiated head and neck cancer cell lines [
33]. Affolter et al. reported a significant and dose-dependent increase in PD-L1 expression levels in irradiated cell lines, which was further enhanced after chemoradiation. The authors hypothesized a co-regulation between ERK1/2 activation and PD-L1 expression as the underlying mode of action in the irradiated tumor cells. Moreover, exosomes derived from irradiated cells demonstrated a protective effect on the unirradiated tumor cells, suppressing apoptosis through downregulation of Caspase 3/7. This study proposes a tumor cell-mediated regulation of PD-L1 in response to platinum-based chemoradiotherapy and clearly demonstrates that chemoradiotherapy can influence immune checkpoint regulation in HNSCC.
Overall, PD-L1 appears to exhibit a dynamic expression in both primary metastasis and secondary recurrence. Therefore, we argue that an adequate biopsy should be taken from the current tumor tissue before initiation of anti-PD-L1 therapy. This should be done independently of PD-L1 expression in the primary tumor to be able to consider PD-L1 expression at the time of the application of immune checkpoint blockade. At present, immune checkpoint blockade is used in R/M disease, but a curative efficacy has not been proven so far [
34,
35]. Determining PD-L1 expression at relapse could potentially identify more patients suitable for immune checkpoint blockade thereby, improving patients prognosis.
The risk factors or clinical variables, which are associated with PD-L1 expression are still not clear. We found a positive correlation between f PD-L1 expression with increasing tumor size and clinical stage. However, there was no significant correlation with the presence of cervical lymph node metastases, sex, and age. Although, Schneider et al. reported consistent results for sex and age [
28], Lin et al. demonstrated a positive correlation between male sex and positive smoking history [
20], while others identified female sex as a risk factor for increased PD-L1 expression [
36]. Thus, there is no consensus on risk factors within the literature. In addition, we could not detect clinical risk factors for high PD-L1 expression in recurrent tumors.
Overall survival was worse for patients with high PD-L1 expression within the primary tumor (log-rank test,
p = 0.043). However, we did not detect any effect of PD-L1 expression on overall and progression-free survival in this cohort using multivariate analysis. This may be explained by the positive correlation between PD-L1 expression with tumor size and tumor stage in our cohort and is contrary to our previous observations [
19]. However, in this cohort, additional patients were included, and were followed up over a longer period, which might explain the disparities. The predictive value of PD-L1 expression has not been clarified and recently published meta-analyses come to different conclusions [
37,
38,
39]. PD-L1 expression appears to be associated with several clinicopathological factors (e.g., sex, tumor stage), which makes interpretation as a predictor difficult [
37,
38,
39].
As this study examined retrospective data and was conducted at a single center, we cannot exclude the possibility of selection bias. As PD-L1 expression varies within head and neck carcinoma due to tumor heterogeneity [
40], a potential weakness of this study is the use of tissue microarrays. In the present study, tissue microarrays of primary tumors were compared with whole-slide sections from the recurrent tumors. This is a weakness of the study design. As both tissue microarrays and whole-slide tissue sections do not represent overall tumor heterogeneity [
40], we considered the study to be feasible under the study design. Our results are solely based on IHC; however, previous studies showed a strong association between
CD274 amplification and PD-L1 IHC positivity [
26,
30].