Hyponatremia Promotes Cancer Growth in a Murine Xenograft Model of Neuroblastoma
Abstract
:1. Introduction
2. Results
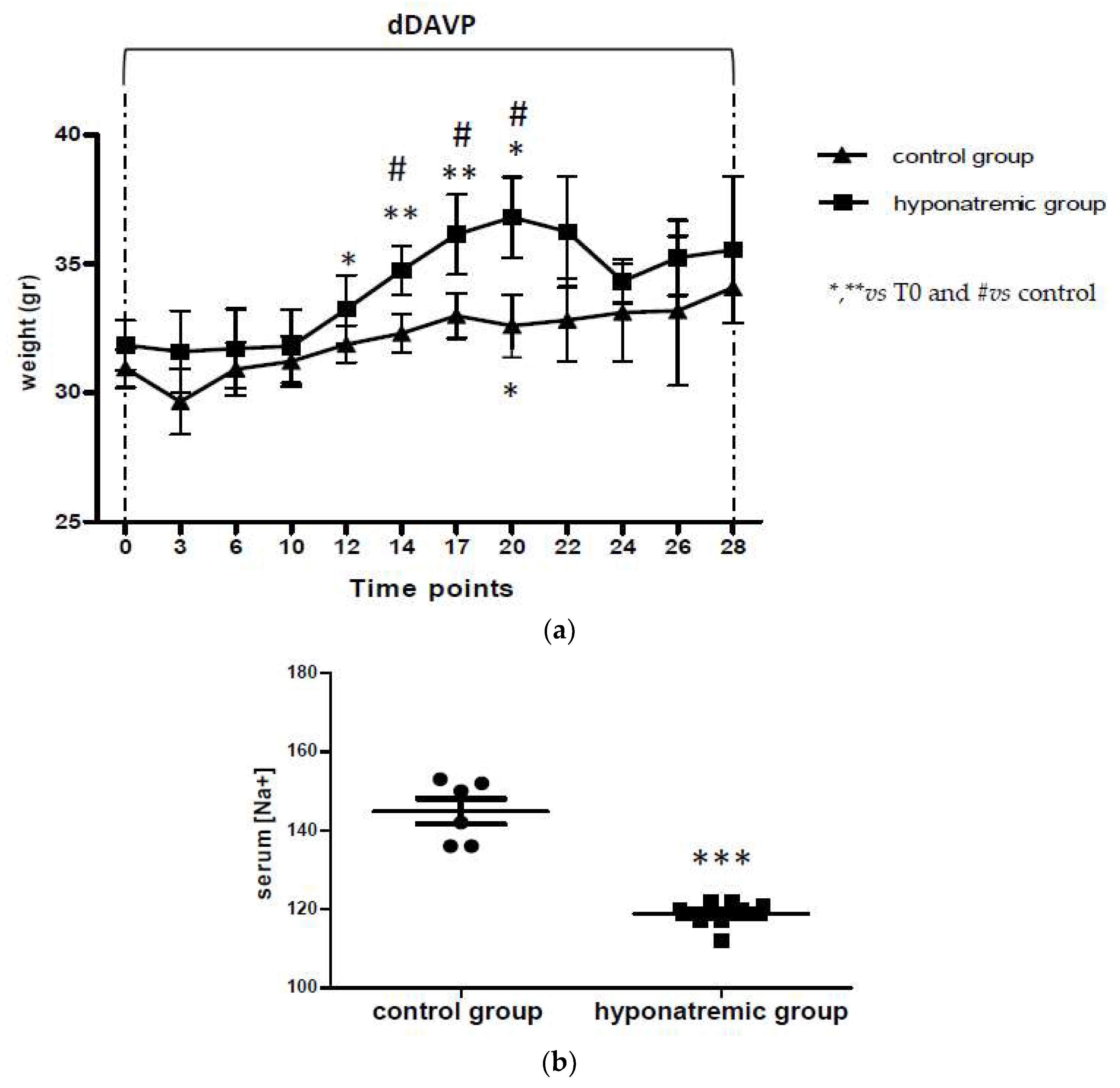
2.1. Induction of Hyponatremia in Nude Mice
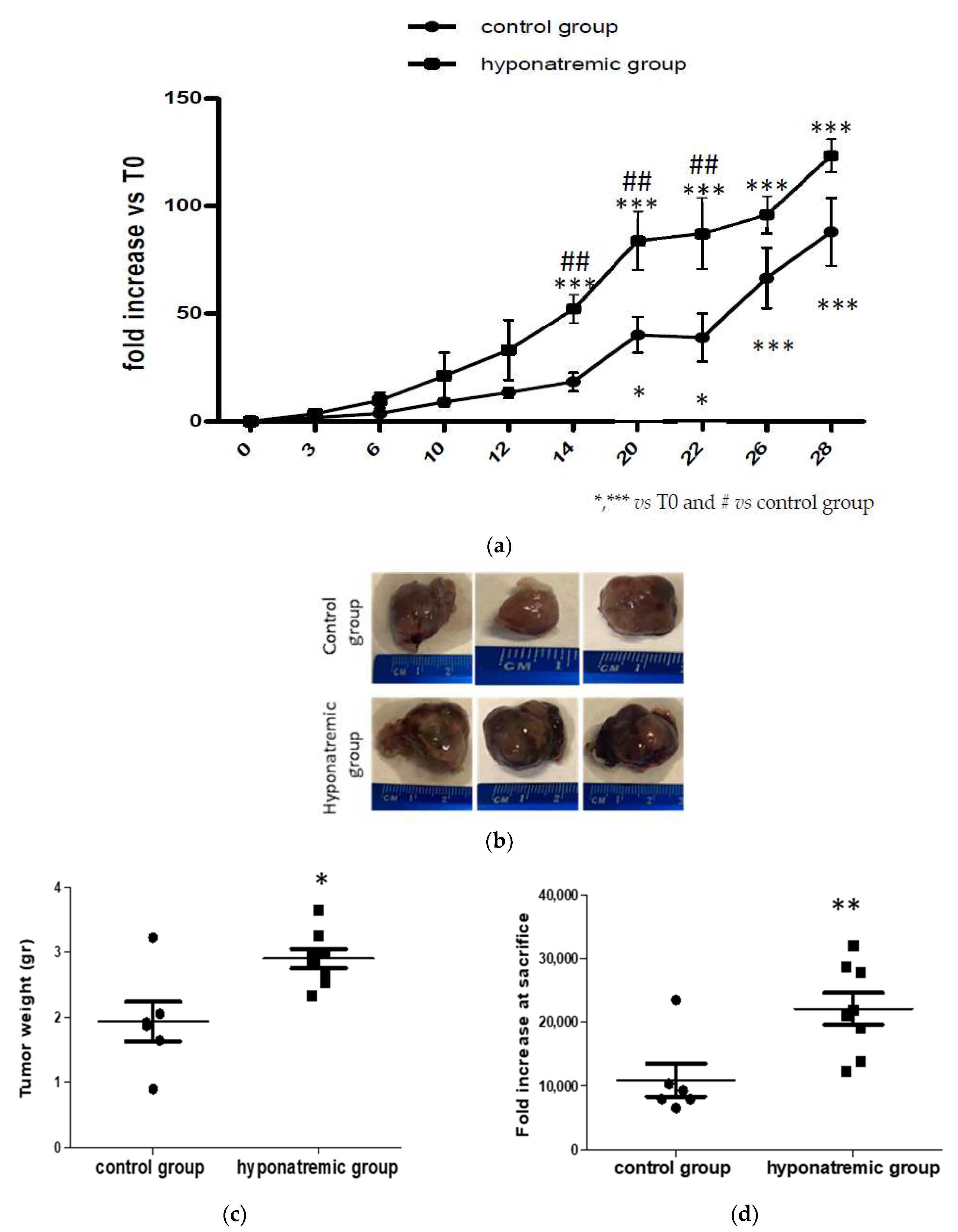
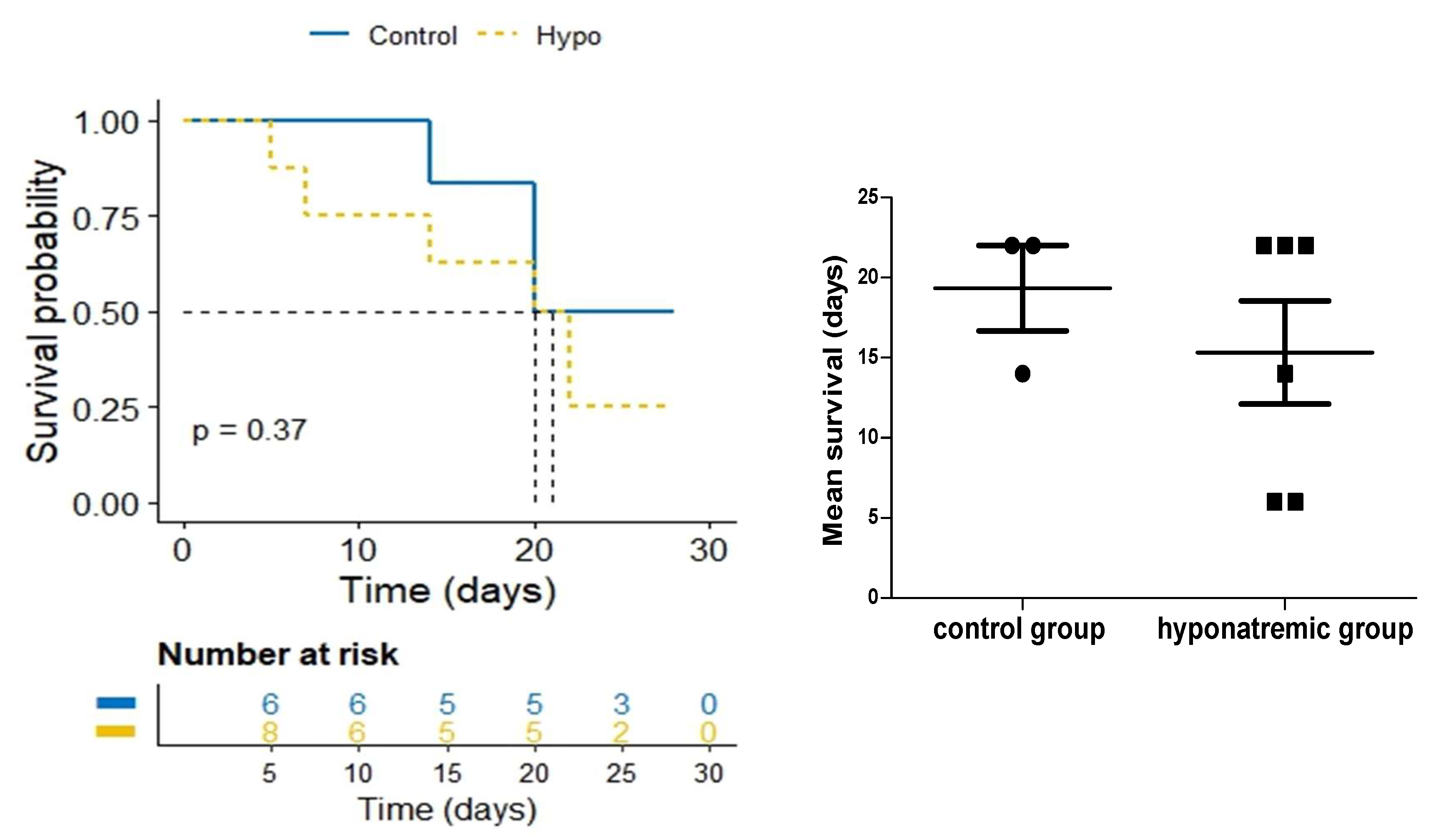
2.2. Analysis of Tumor Masses and Survival
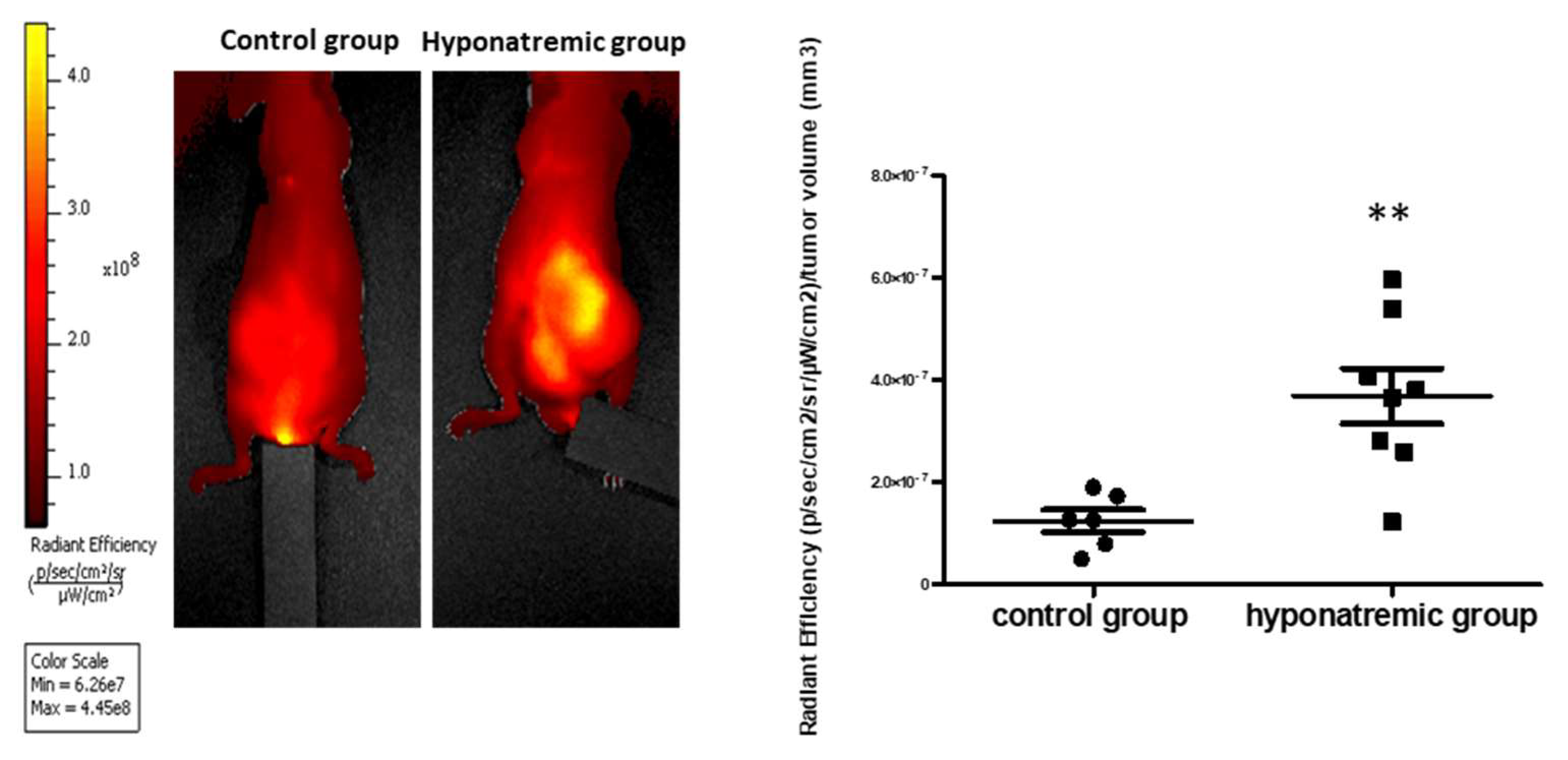
2.3. Analysis of Tumor Progression with IVIS Lumina 5 System
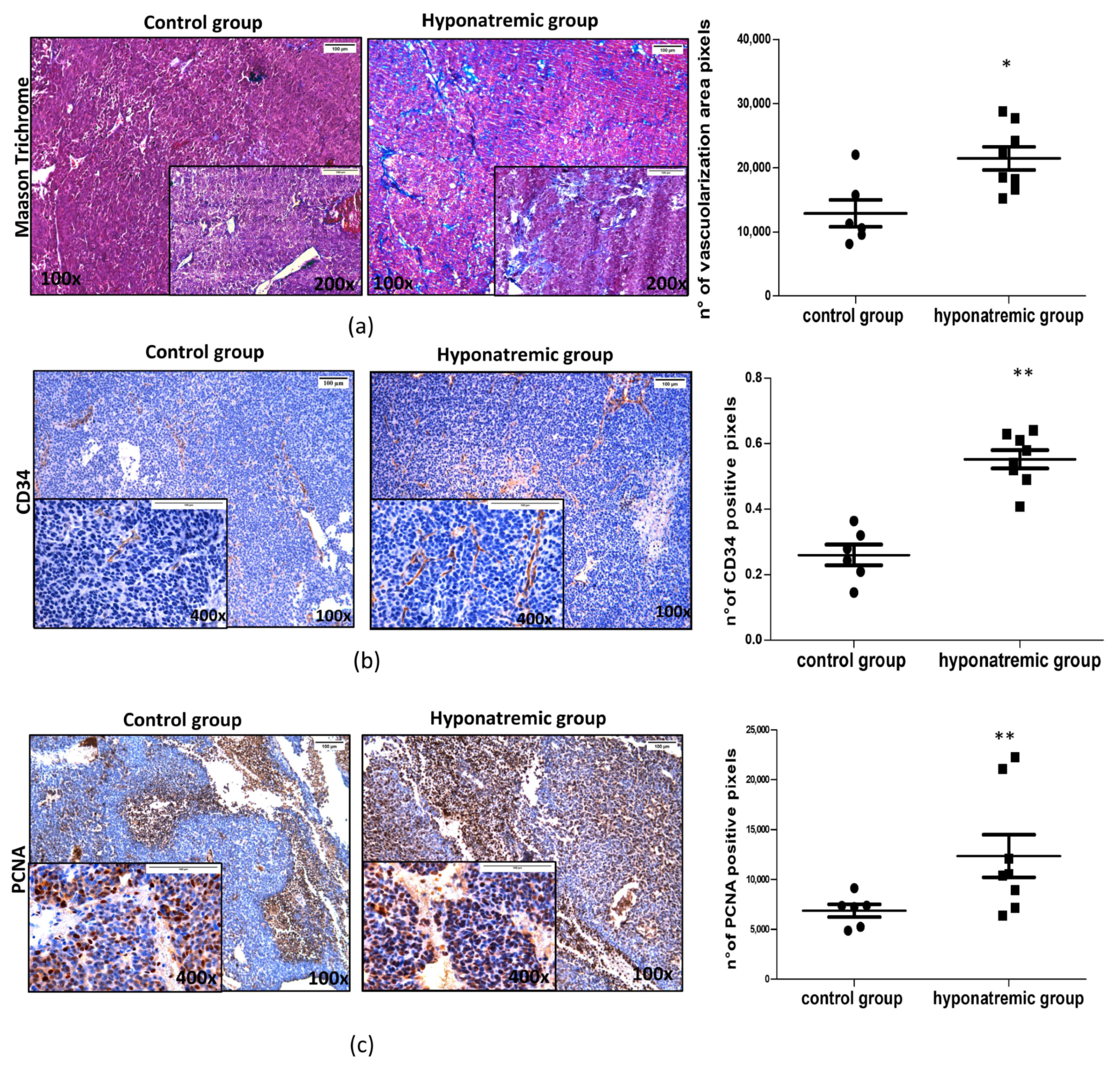
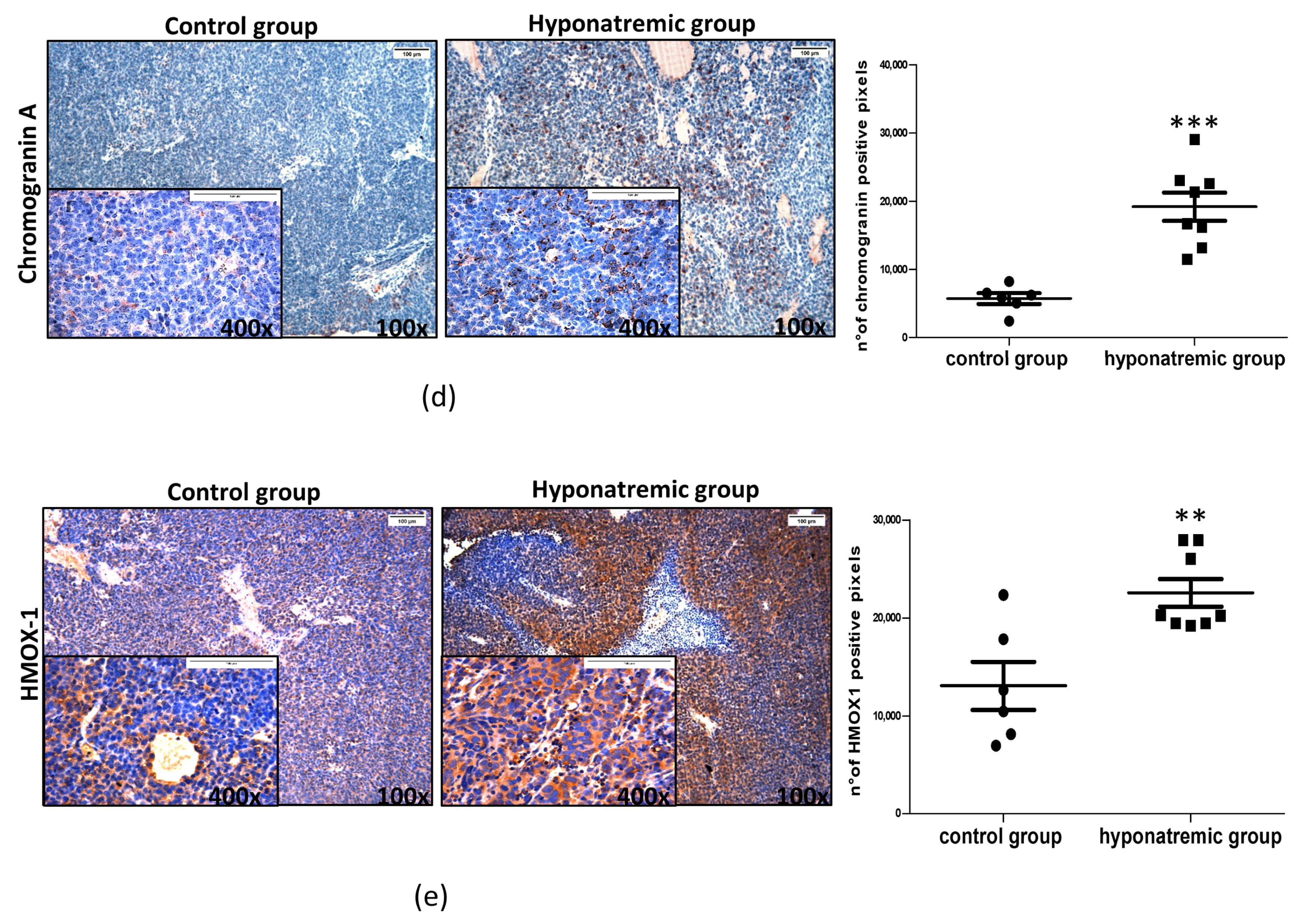
2.4. Histological and Immunohistochemical Analysis
2.5. Analysis of MMPs Activity In Vivo with IVIS Lumina S5
3. Discussion
4. Materials and Methods
4.1. Chemicals and Reagents
4.2. Cell Cultures and Cell Transfection
4.3. A Murine Xenograft Model of Neuroblastoma
4.4. In Vivo Imaging: IVIS Lumina S5 Imaging System
4.5. Serum [Na+] Analysis
4.6. Tissues Preparation and Morphological Characterization
4.7. Masson’s Trichrome Analysis
4.8. Immunohistochemical Analysis
4.9. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Fibbi, B.; Marroncini, G.; Naldi, L.; Anceschi, C.; Errico, A.; Norello, D.; Peri, A. Hyponatremia and Cancer: From Bedside to Benchside. Cancers 2023, 15, 1197. [Google Scholar] [CrossRef] [PubMed]
- Castillo, J.J.; Glezerman, I.G.; Boklage, S.H.; Chiodo, J., 3rd; Tidwell, B.A.; Lamerato, L.E.; Schulman, K.L. The Occurrence of Hyponatremia and Its Importance as a Prognostic Factor in a Cross-Section of Cancer Patients. BMC Cancer 2016, 16, 564. [Google Scholar] [CrossRef] [PubMed]
- Sbardella, E.; Isidori, A.M.; Arnaldi, G.; Arosio, M.; Barone, C.; Benso, A.; Berardi, R.; Capasso, G.; Caprio, M.; Ceccato, F.; et al. Approach to Hyponatremia According to the Clinical Setting: Consensus Statement from the Italian Society of Endocrinology (SIE), Italian Society of Nephrology (SIN), and Italian Association of Medical Oncology (AIOM). J. Endocrinol. Investig. 2018, 41, 3–19. [Google Scholar] [CrossRef] [PubMed]
- Grohé, C. Hyponatremia in Oncology Patients. Front. Horm. Res. 2019, 52, 161–166. [Google Scholar] [CrossRef] [PubMed]
- Doshi, S.M.; Shah, P.; Lei, X.; Lahoti, A.; Salahudeen, A.K. Hyponatremia in Hospitalized Cancer Patients and Its Impact on Clinical Outcomes. Am. J. Kidney Dis. Off. J. Natl. Kidney Found. 2012, 59, 222–228. [Google Scholar] [CrossRef] [PubMed]
- Hansen, O.; Sørensen, P.; Hansen, K.H. The Occurrence of Hyponatremia in SCLC and the Influence on Prognosis: A Retrospective Study of 453 Patients Treated in a Single Institution in a 10-Year Period. Lung Cancer 2010, 68, 111–114. [Google Scholar] [CrossRef] [PubMed]
- Berardi, R.; Santoni, M.; Newsom-Davis, T.; Caramanti, M.; Rinaldi, S.; Tiberi, M.; Morgese, F.; Torniai, M.; Pistelli, M.; Onofri, A.; et al. Hyponatremia Normalization as an Independent Prognostic Factor in Patients with Advanced Non-Small Cell Lung Cancer Treated with First-Line Therapy. Oncotarget 2017, 8, 23871–23879. [Google Scholar] [CrossRef]
- Balachandran, K.; Okines, A.; Gunapala, R.; Morganstein, D.; Popat, S. Resolution of Severe Hyponatraemia Is Associated with Improved Survival in Patients with Cancer. BMC Cancer 2015, 15, 163. [Google Scholar] [CrossRef]
- Benvenuti, S.; Deledda, C.; Luciani, P.; Modi, G.; Bossio, A.; Giuliani, C.; Fibbi, B.; Peri, A. Low Extracellular Sodium Causes Neuronal Distress Independently of Reduced Osmolality in an Experimental Model of Chronic Hyponatremia. Neuromolecular Med. 2013, 15, 493–503. [Google Scholar] [CrossRef]
- Marroncini, G.; Fibbi, B.; Errico, A.; Grappone, C.; Maggi, M.; Peri, A. Effects of Low Extracellular Sodium on Proliferation and Invasive Activity of Cancer Cells in Vitro. Endocrine 2020, 67, 473–484. [Google Scholar] [CrossRef]
- Marroncini, G.; Anceschi, C.; Naldi, L.; Fibbi, B.; Baldanzi, F.; Martinelli, S.; Polvani, S.; Maggi, M.; Peri, A. Low Sodium and Tolvaptan Have Opposite Effects in Human Small Cell Lung Cancer Cells. Mol. Cell. Endocrinol. 2021, 537, 111419. [Google Scholar] [CrossRef] [PubMed]
- Marroncini, G.; Anceschi, C.; Naldi, L.; Fibbi, B.; Brogi, M.; Lanzilao, L.; Fanelli, A.; Maggi, M.; Peri, A. Hyponatremia-Related Liver Steatofibrosis and Impaired Spermatogenesis: Evidence from a Mouse Model of the Syndrome of Inappropriate Antidiuresis. J. Endocrinol. Investig. 2023, 46, 967–983. [Google Scholar] [CrossRef] [PubMed]
- Corona, G.; Giuliani, C.; Parenti, G.; Norello, D.; Verbalis, J.G.; Forti, G.; Maggi, M.; Peri, A. Moderate Hyponatremia Is Associated with Increased Risk of Mortality: Evidence from a Meta-Analysis. PLoS ONE 2013, 8, e80451. [Google Scholar] [CrossRef] [PubMed]
- Svaton, M.; Fiala, O.; Pesek, M.; Bruha, F.; Mukensnabl, P.; Racek, J.; Minarik, M.; Bortlicek, Z. Predictive and Prognostic Significance of Sodium Levels in Patients with NSCLC Treated by Erlotinib. Anticancer Res. 2014, 34, 7461–7465. [Google Scholar]
- Doshi, K.H.; Shriyan, B.; Nookala, M.K.; Kannan, S.; Joshi, A.; Noronha, V.; Gota, V.; Prabhash, K. Prognostic Significance of Pretreatment Sodium Levels in Patients of Nonsmall Cell Lung Cancer Treated with Pemetrexed-Platinum Doublet Chemotherapy. J. Cancer Res. Ther. 2018, 14, 1049–1053. [Google Scholar] [CrossRef] [PubMed]
- Berardi, R.; Caramanti, M.; Fiordoliva, I.; Morgese, F.; Savini, A.; Rinaldi, S.; Torniai, M.; Tiberi, M.; Ferrini, C.; Castagnani, M.; et al. Hyponatraemia Is a Predictor of Clinical Outcome for Malignant Pleural Mesothelioma. Support. Care Cancer 2015, 23, 621–626. [Google Scholar] [CrossRef] [PubMed]
- Folkman, J. Tumor Angiogenesis: Therapeutic Implications. N. Engl. J. Med. 1971, 285, 1182–1186. [Google Scholar] [CrossRef]
- Carmeliet, P.; Jain, R.K. Angiogenesis in Cancer and Other Diseases. Nature 2000, 407, 249–257. [Google Scholar] [CrossRef]
- Jain, R.K. Antiangiogenesis Strategies Revisited: From Starving Tumors to Alleviating Hypoxia. Cancer Cell 2014, 26, 605–622. [Google Scholar] [CrossRef]
- Civin, C.I.; Strauss, L.C.; Brovall, C.; Fackler, M.J.; Schwartz, J.F.; Shaper, J.H. Antigenic Analysis of Hematopoiesis. III. A Hematopoietic Progenitor Cell Surface Antigen Defined by a Monoclonal Antibody Raised against KG-1a Cells. J. Immunol. 1984, 133, 157–165. [Google Scholar] [CrossRef]
- Fina, L.; Molgaard, H.V.; Robertson, D.; Bradley, N.J.; Monaghan, P.; Delia, D.; Sutherland, D.R.; Baker, M.A.; Greaves, M.F. Expression of the CD34 Gene in Vascular Endothelial Cells. Blood 1990, 75, 2417–2426. [Google Scholar] [CrossRef] [PubMed]
- Kapoor, S.; Shenoy, S.P.; Bose, B. CD34 Cells in Somatic, Regenerative and Cancer Stem Cells: Developmental Biology, Cell Therapy, and Omics Big Data Perspective. J. Cell. Biochem. 2020, 121, 3058–3069. [Google Scholar] [CrossRef] [PubMed]
- Aravindan, N.; Somasundaram, D.B.; Herman, T.S.; Aravindan, S. Significance of Hematopoietic Surface Antigen CD34 in Neuroblastoma Prognosis and the Genetic Landscape of CD34-Expressing Neuroblastoma CSCs. Cell Biol. Toxicol. 2021, 37, 461–478. [Google Scholar] [CrossRef] [PubMed]
- González-Magaña, A.; Blanco, F.J. Human PCNA Structure, Function and Interactions. Biomolecules 2020, 10, 570. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Xiong, Y.; Beach, D. Proliferating Cell Nuclear Antigen and P21 Are Components of Multiple Cell Cycle Kinase Complexes. Mol. Biol. Cell 1993, 4, 897–906. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Das Kurmi, B.; Singh, A.; Singh, D. Potential Role of Resveratrol and Its Nano-Formulation as Anti-Cancer Agent. Explor. Target. Anti-Tumor Ther. 2022, 3, 643–658. [Google Scholar] [CrossRef] [PubMed]
- Begum, S.N.; Ray, A.S.; Rahaman, C.H. A Comprehensive and Systematic Review on Potential Anticancer Activities of Eugenol: From Pre-Clinical Evidence to Molecular Mechanisms of Action. Phytomedicine 2022, 107, 154456. [Google Scholar] [CrossRef]
- Song, B.; Wang, W.; Tang, X.; Goh, R.M.W.-J.; Thuya, W.L.; Ho, P.C.L.; Chen, L.; Wang, L. Inhibitory Potential of Resveratrol in Cancer Metastasis: From Biology to Therapy. Cancers 2023, 15, 2758. [Google Scholar] [CrossRef] [PubMed]
- Bartolomucci, A.; Possenti, R.; Mahata, S.K.; Fischer-Colbrie, R.; Loh, Y.P.; Salton, S.R.J. The Extended Granin Family: Structure, Function, and Biomedical Implications. Endocr. Rev. 2011, 32, 755–797. [Google Scholar] [CrossRef]
- Modlin, I.M.; Gustafsson, B.I.; Moss, S.F.; Pavel, M.; Tsolakis, A.V.; Kidd, M. Chromogranin A--Biological Function and Clinical Utility in Neuro Endocrine Tumor Disease. Ann. Surg. Oncol. 2010, 17, 2427–2443. [Google Scholar] [CrossRef] [PubMed]
- Hsiao, R.J.; Seeger, R.C.; Yu, A.L.; O’Connor, D.T. Chromogranin A in Children with Neuroblastoma. Serum Concentration Parallels Disease Stage and Predicts Survival. J. Clin. Investig. 1990, 85, 1555–1559. [Google Scholar] [CrossRef] [PubMed]
- Pagani, A.; Macri, L.; Faulkner, L.B.; Tintori, V.; Paoli, A.; Garaventa, A.; Bussolati, G. Detection Procedures for Neuroblastoma Cells Metastatic to Blood and Bone Marrow: Blinded Comparison of Chromogranin A Heminested Reverse Transcription Polymerase Chain Reaction to Tyrosine Hydroxylase Nested Reverse Transcription Polymerase Chain Reaction and to Anti-GD2 Immunocytology. Diagnostic Mol. Pathol. Am. J. Surg. Pathol. Part B 2002, 11, 98–106. [Google Scholar] [CrossRef]
- Zhang, D.; Babayan, L.; Ho, H.; Heaney, A.P. Chromogranin A Regulates Neuroblastoma Proliferation and Phenotype. Biol. Open 2019, 8, bio036566. [Google Scholar] [CrossRef]
- Hemmati, M.; Yousefi, B.; Bahar, A.; Eslami, M. Importance of Heme Oxygenase-1 in Gastrointestinal Cancers: Functions, Inductions, Regulations, and Signaling. J. Gastrointest. Cancer 2021, 52, 454–461. [Google Scholar] [CrossRef]
- Wang, H.; Cheng, Q.; Bao, L.; Li, M.; Chang, K.; Yi, X. Cytoprotective Role of Heme Oxygenase-1 in Cancer Chemoresistance: Focus on Antioxidant, Antiapoptotic, and Pro-Autophagy Properties. Antioxidants 2023, 12, 1217. [Google Scholar] [CrossRef]
- Abdalla, M.Y.; Ahmad, I.M.; Rachagani, S.; Banerjee, K.; Thompson, C.M.; Maurer, H.C.; Olive, K.P.; Bailey, K.L.; Britigan, B.E.; Kumar, S. Enhancing Responsiveness of Pancreatic Cancer Cells to Gemcitabine Treatment under Hypoxia by Heme Oxygenase-1 Inhibition. Transl. Res. 2019, 207, 56–69. [Google Scholar] [CrossRef] [PubMed]
- Najafi, M.; Farhood, B.; Mortezaee, K. Extracellular Matrix (ECM) Stiffness and Degradation as Cancer Drivers. J. Cell. Biochem. 2019, 120, 2782–2790. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.; Xuan, Z.; Liu, X.; Zheng, M.; Yang, C.; Wang, H. Immunomodulatory Role of Metalloproteinase ADAM17 in Tumor Development. Front. Immunol. 2022, 13, 1059376. [Google Scholar] [CrossRef] [PubMed]
- Selmer, C.; Madsen, J.C.; Torp-Pedersen, C.; Gislason, G.H.; Faber, J. Hyponatremia, All-Cause Mortality, and Risk of Cancer Diagnoses in the Primary Care Setting: A Large Population Study. Eur. J. Intern. Med. 2016, 36, 36–43. [Google Scholar] [CrossRef] [PubMed]
- Holland-Bill, L.; Christiansen, C.F.; Farkas, D.K.; Donskov, F.; Jørgensen, J.O.L.; Sørensen, H.T. Diagnosis of Hyponatremia and Increased Risk of a Subsequent Cancer Diagnosis: Results from a Nationwide Population-Based Cohort Study. Acta Oncol. 2018, 57, 522–527. [Google Scholar] [CrossRef] [PubMed]

| Time Point of Sacrifice | T6 | T14 | T22 | T28 |
|---|---|---|---|---|
| Number of control mice sacrificed | / | n = 1 | n = 2 | n = 3 |
| [Na+] serum of control mice | / | 153 | 136 150 | 152 142 136 |
| Number of hyponatremic mice sacrificed | n = 2 | n = 1 | n = 3 | n = 2 |
| [Na+] serum of hyponatremic mice | 122 122 | 119.88 | 121 120 117 | 117 112 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Marroncini, G.; Naldi, L.; Fibbi, B.; Errico, A.; Polvani, S.; Brogi, M.; Fanelli, A.; Maggi, M.; Peri, A. Hyponatremia Promotes Cancer Growth in a Murine Xenograft Model of Neuroblastoma. Int. J. Mol. Sci. 2023, 24, 16680. https://doi.org/10.3390/ijms242316680
Marroncini G, Naldi L, Fibbi B, Errico A, Polvani S, Brogi M, Fanelli A, Maggi M, Peri A. Hyponatremia Promotes Cancer Growth in a Murine Xenograft Model of Neuroblastoma. International Journal of Molecular Sciences. 2023; 24(23):16680. https://doi.org/10.3390/ijms242316680
Chicago/Turabian StyleMarroncini, Giada, Laura Naldi, Benedetta Fibbi, Alice Errico, Simone Polvani, Marco Brogi, Alessandra Fanelli, Mario Maggi, and Alessandro Peri. 2023. "Hyponatremia Promotes Cancer Growth in a Murine Xenograft Model of Neuroblastoma" International Journal of Molecular Sciences 24, no. 23: 16680. https://doi.org/10.3390/ijms242316680
APA StyleMarroncini, G., Naldi, L., Fibbi, B., Errico, A., Polvani, S., Brogi, M., Fanelli, A., Maggi, M., & Peri, A. (2023). Hyponatremia Promotes Cancer Growth in a Murine Xenograft Model of Neuroblastoma. International Journal of Molecular Sciences, 24(23), 16680. https://doi.org/10.3390/ijms242316680





