Prenatal Sex Hormone Exposure Is Associated with the Development of Autism Spectrum Disorder
Abstract
1. Introduction
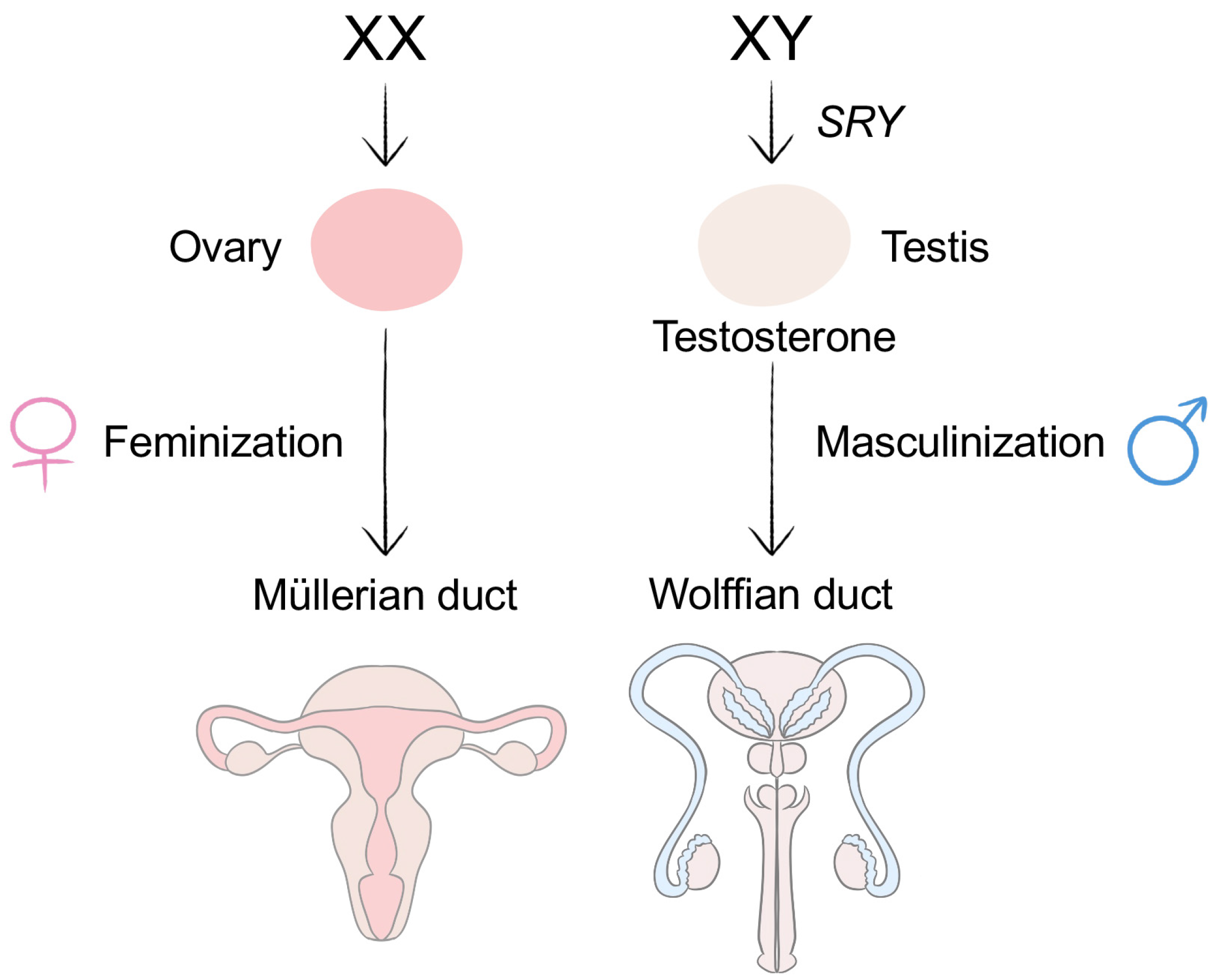
2. Body Development and Sexual Differentiation
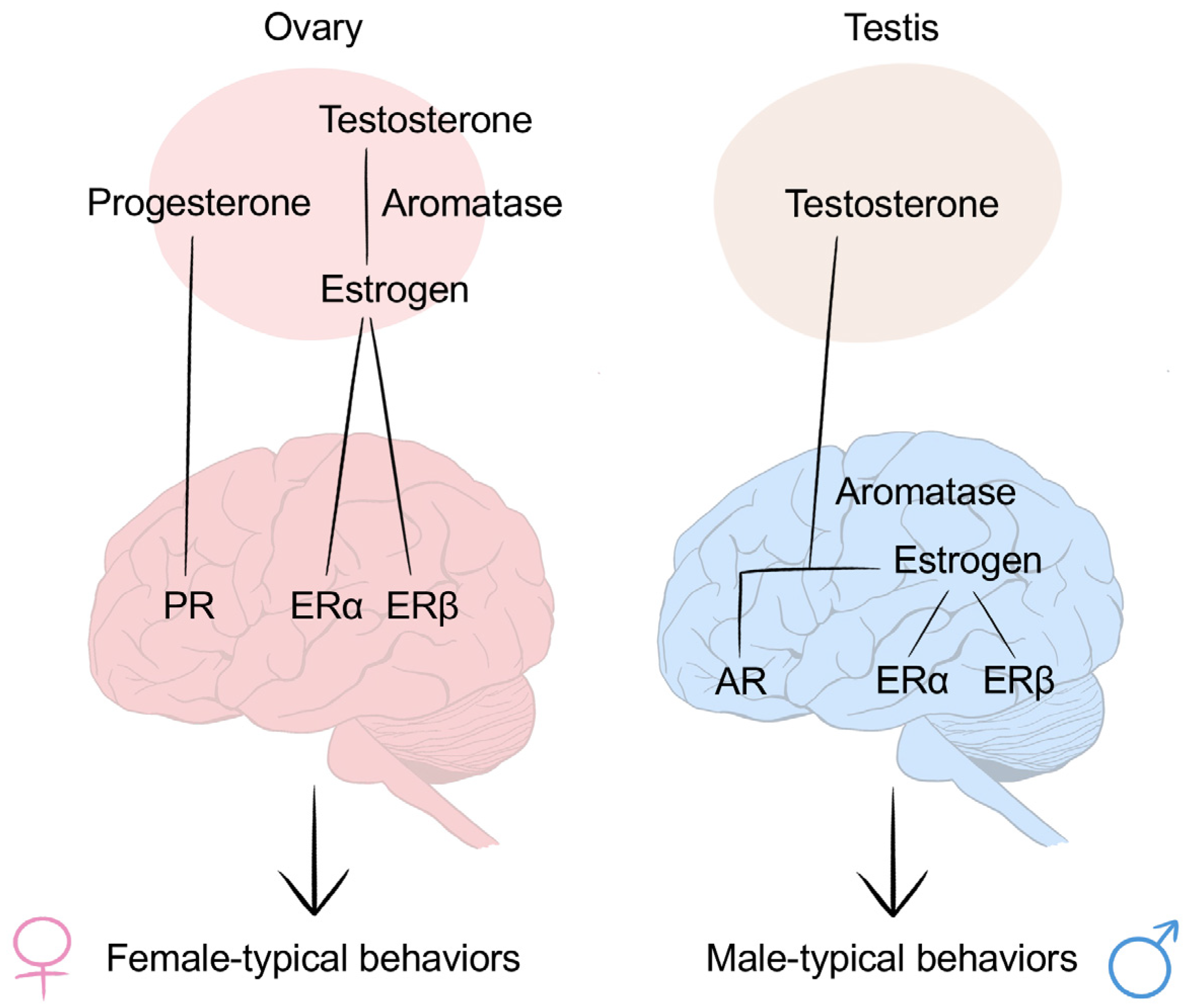
3. Brain Development and Sexual Differentiation
4. Effects of Testosterone in Humans
5. Sex-Biased ASD
6. Testosterone Exposure and the EMB Theory
7. Animal Models of Elevated Prenatal Testosterone Exposure
8. Sex Differences in MIA
9. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Green, T.; Flash, S.; Reiss, A.L. Sex differences in psychiatric disorders: What we can learn from sex chromosome aneuploidies. Neuropsychopharmacology 2019, 44, 9–21. [Google Scholar] [CrossRef] [PubMed]
- Ferri, S.L.; Abel, T.; Brodkin, E.S. Sex Differences in Autism Spectrum Disorder: A Review. Curr. Psychiatry Rep. 2018, 20, 9. [Google Scholar] [CrossRef] [PubMed]
- Dougherty, J.D.; Marrus, N.; Maloney, S.E.; Yip, B.; Sandin, S.; Turner, T.N.; Selmanovic, D.; Kroll, K.L.; Gutmann, D.H.; Constantino, J.N.; et al. Can the “female protective effect” liability threshold model explain sex differences in autism spectrum disorder? Neuron 2022, 110, 3243–3262. [Google Scholar] [CrossRef] [PubMed]
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders (DSM-5), 5th ed.; American Psychiatric Association: Washington, DC, USA, 2013. [Google Scholar] [CrossRef]
- Fombonne, E. Epidemiology of pervasive developmental disorders. Pediatr. Res. 2009, 65, 591–598. [Google Scholar] [CrossRef] [PubMed]
- Loomes, R.; Hull, L.; Mandy, W.P.L. What Is the Male-to-Female Ratio in Autism Spectrum Disorder? A Systematic Review and Meta-Analysis. J. Am. Acad. Child Adolesc. Psychiatry 2017, 56, 466–474. [Google Scholar] [CrossRef]
- Maenner, M.J.; Shaw, K.A.; Bakian, A.V.; Bilder, D.A.; Durkin, M.S.; Esler, A.; Furnier, S.M.; Hallas, L.; Hall-Lande, J.; Hudson, A.; et al. Prevalence and Characteristics of Autism Spectrum Disorder Among Children Aged 8 Years—Autism and Developmental Disabilities Monitoring Network, 11 Sites, United States, 2018. MMWR Surveill. Summ. 2021, 70, 1. [Google Scholar] [CrossRef]
- Szatmari, P.; Liu, X.Q.; Goldberg, J.; Zwaigenbaum, L.; Paterson, A.D.; Woodbury-Smith, M.; Georgiades, S.; Duku, E.; Thompson, A. Sex differences in repetitive stereotyped behaviors in autism: Implications for genetic liability. Am. J. Med. Genet. Part B Neuropsychiatr. Genet. 2012, 159b, 5–12. [Google Scholar] [CrossRef]
- Hu, V.W.; Sarachana, T.; Sherrard, R.M.; Kocher, K.M. Investigation of sex differences in the expression of RORA and its transcriptional targets in the brain as a potential contributor to the sex bias in autism. Mol. Autism 2015, 6, 7. [Google Scholar] [CrossRef]
- Doi, M.; Li, M.; Usui, N.; Shimada, S. Genomic Strategies for Understanding the Pathophysiology of Autism Spectrum Disorder. Front. Mol. Neurosci. 2022, 15, 930941. [Google Scholar] [CrossRef]
- Abrahams, B.S.; Arking, D.E.; Campbell, D.B.; Mefford, H.C.; Morrow, E.M.; Weiss, L.A.; Menashe, I.; Wadkins, T.; Banerjee-Basu, S.; Packer, A. SFARI Gene 2.0: A community-driven knowledgebase for the autism spectrum disorders (ASDs). Mol. Autism 2013, 4, 36. [Google Scholar] [CrossRef]
- Banerjee-Basu, S.; Packer, A. SFARI Gene: An evolving database for the autism research community. Dis. Model. Mech. 2010, 3, 133–135. [Google Scholar] [CrossRef]
- de la Torre-Ubieta, L.; Won, H.; Stein, J.L.; Geschwind, D.H. Advancing the understanding of autism disease mechanisms through genetics. Nat. Med. 2016, 22, 345–361. [Google Scholar] [CrossRef] [PubMed]
- Durand, C.M.; Betancur, C.; Boeckers, T.M.; Bockmann, J.; Chaste, P.; Fauchereau, F.; Nygren, G.; Rastam, M.; Gillberg, I.C.; Anckarsäter, H.; et al. Mutations in the gene encoding the synaptic scaffolding protein SHANK3 are associated with autism spectrum disorders. Nat. Genet. 2007, 39, 25–27. [Google Scholar] [CrossRef] [PubMed]
- Araujo, D.J.; Toriumi, K.; Escamilla, C.O.; Kulkarni, A.; Anderson, A.G.; Harper, M.; Usui, N.; Ellegood, J.; Lerch, J.P.; Birnbaum, S.G.; et al. Foxp1 in Forebrain Pyramidal Neurons Controls Gene Expression Required for Spatial Learning and Synaptic Plasticity. J. Neurosci. Off. J. Soc. Neurosci. 2017, 37, 10917–10931. [Google Scholar] [CrossRef]
- Usui, N.; Araujo, D.J.; Kulkarni, A.; Co, M.; Ellegood, J.; Harper, M.; Toriumi, K.; Lerch, J.P.; Konopka, G. Foxp1 regulation of neonatal vocalizations via cortical development. Genes Dev. 2017, 31, 2039–2055. [Google Scholar] [CrossRef]
- Schaaf, C.P.; Betancur, C.; Yuen, R.K.C.; Parr, J.R.; Skuse, D.H.; Gallagher, L.; Bernier, R.A.; Buchanan, J.A.; Buxbaum, J.D.; Chen, C.A.; et al. A framework for an evidence-based gene list relevant to autism spectrum disorder. Nat. Rev. Genet. 2020, 21, 367–376. [Google Scholar] [CrossRef] [PubMed]
- Hornig, M.; Bresnahan, M.A.; Che, X.; Schultz, A.F.; Ukaigwe, J.E.; Eddy, M.L.; Hirtz, D.; Gunnes, N.; Lie, K.K.; Magnus, P.; et al. Prenatal fever and autism risk. Mol. Psychiatry 2018, 23, 759–766. [Google Scholar] [CrossRef]
- Doi, M.; Usui, N.; Shimada, S. Prenatal Environment and Neurodevelopmental Disorders. Front. Endocrinol. 2022, 13, 860110. [Google Scholar] [CrossRef]
- Haida, O.; Al Sagheer, T.; Balbous, A.; Francheteau, M.; Matas, E.; Soria, F.; Fernagut, P.O.; Jaber, M. Sex-dependent behavioral deficits and neuropathology in a maternal immune activation model of autism. Transl. Psychiatry 2019, 9, 124. [Google Scholar] [CrossRef]
- Estes, M.L.; McAllister, A.K. Maternal immune activation: Implications for neuropsychiatric disorders. Science 2016, 353, 772–777. [Google Scholar] [CrossRef]
- Gore, A.C.; Martien, K.M.; Gagnidze, K.; Pfaff, D. Implications of prenatal steroid perturbations for neurodevelopment, behavior, and autism. Endocr. Rev. 2014, 35, 961–991. [Google Scholar] [CrossRef]
- Ipulan, L.A.; Raga, D.; Suzuki, K.; Murashima, A.; Matsumaru, D.; Cunha, G.; Yamada, G. Investigation of sexual dimorphisms through mouse models and hormone/hormone-disruptor treatments. Differentiation 2016, 91, 78–89. [Google Scholar] [CrossRef] [PubMed]
- Bhargava, A.; Arnold, A.P.; Bangasser, D.A.; Denton, K.M.; Gupta, A.; Hilliard Krause, L.M.; Mayer, E.A.; McCarthy, M.; Miller, W.L.; Raznahan, A.; et al. Considering Sex as a Biological Variable in Basic and Clinical Studies: An Endocrine Society Scientific Statement. Endocr. Rev. 2021, 42, 219–258. [Google Scholar] [CrossRef]
- Sekido, R. The potential role of SRY in epigenetic gene regulation during brain sexual differentiation in mammals. Adv. Genet. 2014, 86, 135–165. [Google Scholar] [CrossRef]
- Goodfellow, P.N.; Lovell-Badge, R. SRY and sex determination in mammals. Annu. Rev. Genet. 1993, 27, 71–92. [Google Scholar] [CrossRef]
- Morris, J.A.; Jordan, C.L.; Breedlove, S.M. Sexual differentiation of the vertebrate nervous system. Nat. Neurosci. 2004, 7, 1034–1039. [Google Scholar] [CrossRef] [PubMed]
- Wilson, J.D. The role of androgens in male gender role behavior. Endocr. Rev. 1999, 20, 726–737. [Google Scholar] [CrossRef] [PubMed]
- Lenz, K.M.; Nugent, B.M.; McCarthy, M.M. Sexual differentiation of the rodent brain: Dogma and beyond. Front. Neurosci. 2012, 6, 26. [Google Scholar] [CrossRef]
- Lenz, K.M.; Pickett, L.A.; Wright, C.L.; Galan, A.; McCarthy, M.M. Prenatal Allergen Exposure Perturbs Sexual Differentiation and Programs Lifelong Changes in Adult Social and Sexual Behavior. Sci. Rep. 2019, 9, 4837. [Google Scholar] [CrossRef]
- Dubois, J.; Benders, M.; Cachia, A.; Lazeyras, F.; Ha-Vinh Leuchter, R.; Sizonenko, S.V.; Borradori-Tolsa, C.; Mangin, J.F.; Hüppi, P.S. Mapping the early cortical folding process in the preterm newborn brain. Cereb. Cortex 2008, 18, 1444–1454. [Google Scholar] [CrossRef]
- Cosgrove, K.P.; Mazure, C.M.; Staley, J.K. Evolving knowledge of sex differences in brain structure, function, and chemistry. Biol. Psychiatry 2007, 62, 847–855. [Google Scholar] [CrossRef] [PubMed]
- Harasty, J.; Double, K.L.; Halliday, G.M.; Kril, J.J.; McRitchie, D.A. Language-associated cortical regions are proportionally larger in the female brain. Arch. Neurol. 1997, 54, 171–176. [Google Scholar] [CrossRef] [PubMed]
- Wood, J.L.; Heitmiller, D.; Andreasen, N.C.; Nopoulos, P. Morphology of the ventral frontal cortex: Relationship to femininity and social cognition. Cereb. Cortex 2008, 18, 534–540. [Google Scholar] [CrossRef] [PubMed]
- Goldstein, J.M.; Seidman, L.J.; Horton, N.J.; Makris, N.; Kennedy, D.N.; Caviness, V.S., Jr.; Faraone, S.V.; Tsuang, M.T. Normal sexual dimorphism of the adult human brain assessed by in vivo magnetic resonance imaging. Cereb. Cortex 2001, 11, 490–497. [Google Scholar] [CrossRef]
- Allen, L.S.; Richey, M.F.; Chai, Y.M.; Gorski, R.A. Sex differences in the corpus callosum of the living human being. J. Neurosci. Off. J. Soc. Neurosci. 1991, 11, 933–942. [Google Scholar] [CrossRef]
- Raz, N.; Gunning-Dixon, F.; Head, D.; Williamson, A.; Acker, J.D. Age and sex differences in the cerebellum and the ventral pons: A prospective MR study of healthy adults. AJNR Am. J. Neuroradiol. 2001, 22, 1161–1167. [Google Scholar]
- Döhler, K.D.; Coquelin, A.; Davis, F.; Hines, M.; Shryne, J.E.; Gorski, R.A. Pre- and postnatal influence of testosterone propionate and diethylstilbestrol on differentiation of the sexually dimorphic nucleus of the preoptic area in male and female rats. Brain Res. 1984, 302, 291–295. [Google Scholar] [CrossRef]
- Ingalhalikar, M.; Smith, A.; Parker, D.; Satterthwaite, T.D.; Elliott, M.A.; Ruparel, K.; Hakonarson, H.; Gur, R.E.; Gur, R.C.; Verma, R. Sex differences in the structural connectome of the human brain. Proc. Natl. Acad. Sci. USA 2014, 111, 823–828. [Google Scholar] [CrossRef]
- Baron-Cohen, S.; Wheelwright, S. The empathy quotient: An investigation of adults with Asperger syndrome or high functioning autism, and normal sex differences. J. Autism Dev. Disord. 2004, 34, 163–175. [Google Scholar] [CrossRef]
- Harrop, C.; Jones, D.; Zheng, S.; Nowell, S.; Schultz, R.; Parish-Morris, J. Visual attention to faces in children with autism spectrum disorder: Are there sex differences? Mol. Autism 2019, 10, 28. [Google Scholar] [CrossRef]
- Antezana, L.; Factor, R.S.; Condy, E.E.; Strege, M.V.; Scarpa, A.; Richey, J.A. Gender differences in restricted and repetitive behaviors and interests in youth with autism. Autism Res. 2019, 12, 274–283. [Google Scholar] [CrossRef] [PubMed]
- Werling, D.M.; Geschwind, D.H. Recurrence rates provide evidence for sex-differential, familial genetic liability for autism spectrum disorders in multiplex families and twins. Mol. Autism 2015, 6, 27. [Google Scholar] [CrossRef] [PubMed]
- Head, A.M.; McGillivray, J.A.; Stokes, M.A. Gender differences in emotionality and sociability in children with autism spectrum disorders. Mol. Autism 2014, 5, 19. [Google Scholar] [CrossRef] [PubMed]
- Crider, A.; Thakkar, R.; Ahmed, A.O.; Pillai, A. Dysregulation of estrogen receptor beta (ERβ), aromatase (CYP19A1), and ER co-activators in the middle frontal gyrus of autism spectrum disorder subjects. Mol. Autism 2014, 5, 46. [Google Scholar] [CrossRef]
- Sarachana, T.; Xu, M.; Wu, R.C.; Hu, V.W. Sex hormones in autism: Androgens and estrogens differentially and reciprocally regulate RORA, a novel candidate gene for autism. PloS ONE 2011, 6, e17116. [Google Scholar] [CrossRef]
- Hutsler, J.J.; Zhang, H. Increased dendritic spine densities on cortical projection neurons in autism spectrum disorders. Brain Res. 2010, 1309, 83–94. [Google Scholar] [CrossRef]
- Kuiri-Hänninen, T.; Seuri, R.; Tyrväinen, E.; Turpeinen, U.; Hämäläinen, E.; Stenman, U.H.; Dunkel, L.; Sankilampi, U. Increased activity of the hypothalamic-pituitary-testicular axis in infancy results in increased androgen action in premature boys. J. Clin. Endocrinol. Metab. 2011, 96, 98–105. [Google Scholar] [CrossRef]
- Roosenboom, J.; Indencleef, K.; Lee, M.K.; Hoskens, H.; White, J.D.; Liu, D.; Hecht, J.T.; Wehby, G.L.; Moreno, L.M.; Hodges-Simeon, C.; et al. SNPs Associated With Testosterone Levels Influence Human Facial Morphology. Front. Genet. 2018, 9, 497. [Google Scholar] [CrossRef]
- Lombardo, M.V.; Ashwin, E.; Auyeung, B.; Chakrabarti, B.; Taylor, K.; Hackett, G.; Bullmore, E.T.; Baron-Cohen, S. Fetal testosterone influences sexually dimorphic gray matter in the human brain. J. Neurosci. Off. J. Soc. Neurosci. 2012, 32, 674–680. [Google Scholar] [CrossRef]
- Auyeung, B.; Knickmeyer, R.; Ashwin, E.; Taylor, K.; Hackett, G.; Baron-Cohen, S. Effects of fetal testosterone on visuospatial ability. Arch. Sex. Behav. 2012, 41, 571–581. [Google Scholar] [CrossRef]
- Auyeung, B.; Taylor, K.; Hackett, G.; Baron-Cohen, S. Foetal testosterone and autistic traits in 18 to 24-month-old children. Mol. Autism 2010, 1, 11. [Google Scholar] [CrossRef]
- Auyeung, B.; Baron-Cohen, S.; Ashwin, E.; Knickmeyer, R.; Taylor, K.; Hackett, G. Fetal testosterone and autistic traits. Br. J. Psychol. 2009, 100, 1–22. [Google Scholar] [CrossRef]
- Pivovarciova, A.; Durdiakova, J.; Babinska, K.; Kubranska, A.; Vokalova, L.; Minarik, G.; Celec, P.; Murin, M.; Ostatnikova, D. Testosterone and Androgen Receptor Sensitivity in Relation to Hyperactivity Symptoms in Boys with Autism Spectrum Disorders. PloS ONE 2016, 11, e0149657. [Google Scholar] [CrossRef] [PubMed]
- Palomba, S.; Marotta, R.; Di Cello, A.; Russo, T.; Falbo, A.; Orio, F.; Tolino, A.; Zullo, F.; Esposito, R.; La Sala, G.B. Pervasive developmental disorders in children of hyperandrogenic women with polycystic ovary syndrome: A longitudinal case-control study. Clin. Endocrinol. 2012, 77, 898–904. [Google Scholar] [CrossRef] [PubMed]
- Cherskov, A.; Pohl, A.; Allison, C.; Zhang, H.; Payne, R.A.; Baron-Cohen, S. Polycystic ovary syndrome and autism: A test of the prenatal sex steroid theory. Transl. Psychiatry 2018, 8, 136. [Google Scholar] [CrossRef] [PubMed]
- Hofman, M.A.; Swaab, D.F. The sexually dimorphic nucleus of the preoptic area in the human brain: A comparative morphometric study. J. Anat. 1989, 164, 55–72. [Google Scholar] [PubMed]
- Edwards, D.A. Neonatal administration of androstenedione, testosterone or testosterone propionate: Effects on ovulation, sexual receptivity and aggressive behavior in female mice. Physiol. Behav. 1971, 6, 223–228. [Google Scholar] [CrossRef] [PubMed]
- Phoenix, C.H.; Goy, R.W.; Gerall, A.A.; Young, W.C. Organizing action of prenatally administered testosterone propionate on the tissues mediating mating behavior in the female guinea pig. Endocrinology 1959, 65, 369–382. [Google Scholar] [CrossRef] [PubMed]
- Geniole, S.N.; Bird, B.M.; McVittie, J.S.; Purcell, R.B.; Archer, J.; Carré, J.M. Is testosterone linked to human aggression? A meta-analytic examination of the relationship between baseline, dynamic, and manipulated testosterone on human aggression. Horm. Behav. 2020, 123, 104644. [Google Scholar] [CrossRef]
- Fusar-Poli, L.; Rodolico, A.; Sturiale, S.; Carotenuto, B.; Natale, A.; Arillotta, D.; Siafis, S.; Signorelli, M.S.; Aguglia, E. Second-to-Fourth Digit Ratio (2D:4D) in Psychiatric Disorders: A Systematic Review of Case-control Studies. Clin. Psychopharmacol. Neurosci. 2021, 19, 26–45. [Google Scholar] [CrossRef]
- Baron-Cohen, S. Autism: The empathizing-systemizing (E-S) theory. Ann. N. Y. Acad. Sc.i 2009, 1156, 68–80. [Google Scholar] [CrossRef] [PubMed]
- Tan, D.W.; Gilani, S.Z.; Maybery, M.T.; Mian, A.; Hunt, A.; Walters, M.; Whitehouse, A.J.O. Hypermasculinised facial morphology in boys and girls with Autism Spectrum Disorder and its association with symptomatology. Sci. Rep. 2017, 7, 9348. [Google Scholar] [CrossRef]
- van Eijk, L.; Zietsch, B.P. Testing the extreme male brain hypothesis: Is autism spectrum disorder associated with a more male-typical brain? Autism Res. 2021, 14, 1597–1608. [Google Scholar] [CrossRef] [PubMed]
- Chew, L.; Sun, K.L.; Sun, W.; Wang, Z.; Rajadas, J.; Flores, R.E.; Arnold, E.; Jo, B.; Fung, L.K. Association of serum allopregnanolone with restricted and repetitive behaviors in adult males with autism. Psychoneuroendocrinology 2021, 123, 105039. [Google Scholar] [CrossRef] [PubMed]
- Baron-Cohen, S. The extreme male brain theory of autism. Trends Cogn. Sci. 2002, 6, 248–254. [Google Scholar] [CrossRef]
- Baron-Cohen, S.; Knickmeyer, R.C.; Belmonte, M.K. Sex differences in the brain: Implications for explaining autism. Science 2005, 310, 819–823. [Google Scholar] [CrossRef]
- Baron-Cohen, S.; Lombardo, M.V.; Auyeung, B.; Ashwin, E.; Chakrabarti, B.; Knickmeyer, R. Why are autism spectrum conditions more prevalent in males? PLoS Biol. 2011, 9, e1001081. [Google Scholar] [CrossRef]
- Lennartsson, A.K.; Kushnir, M.M.; Bergquist, J.; Billig, H.; Jonsdottir, I.H. Sex steroid levels temporarily increase in response to acute psychosocial stress in healthy men and women. Int. J. Psychophysiol. 2012, 84, 246–253. [Google Scholar] [CrossRef]
- Van den Bergh, B.R.H.; van den Heuvel, M.I.; Lahti, M.; Braeken, M.; de Rooij, S.R.; Entringer, S.; Hoyer, D.; Roseboom, T.; Räikkönen, K.; King, S.; et al. Prenatal developmental origins of behavior and mental health: The influence of maternal stress in pregnancy. Neurosci. Biobehav. Rev. 2020, 117, 26–64. [Google Scholar] [CrossRef]
- Azziz, R.; Carmina, E.; Chen, Z.; Dunaif, A.; Laven, J.S.; Legro, R.S.; Lizneva, D.; Natterson-Horowtiz, B.; Teede, H.J.; Yildiz, B.O. Polycystic ovary syndrome. Nat. Rev. Dis. Prim. 2016, 2, 16057. [Google Scholar] [CrossRef] [PubMed]
- Handelsman, D.J.; Teede, H.J.; Desai, R.; Norman, R.J.; Moran, L.J. Performance of mass spectrometry steroid profiling for diagnosis of polycystic ovary syndrome. Hum. Reprod. 2017, 32, 418–422. [Google Scholar] [CrossRef] [PubMed]
- Sir-Petermann, T.; Maliqueo, M.; Angel, B.; Lara, H.E.; Pérez-Bravo, F.; Recabarren, S.E. Maternal serum androgens in pregnant women with polycystic ovarian syndrome: Possible implications in prenatal androgenization. Hum. Reprod. 2002, 17, 2573–2579. [Google Scholar] [CrossRef] [PubMed]
- Filippou, P.; Homburg, R. Is foetal hyperexposure to androgens a cause of PCOS? Hum. Reprod. Update 2017, 23, 421–432. [Google Scholar] [CrossRef] [PubMed]
- Yagishita-Kyo, N.; Ikai, Y.; Uekita, T.; Shinohara, A.; Koshimoto, C.; Yoshikawa, K.; Maruyama, K.; Yagishita, S. Testosterone interrupts binding of Neurexin and Neuroligin that are expressed in a highly socialized rodent, Octodon degus. Biochem. Biophys. Res. Commun. 2021, 551, 54–62. [Google Scholar] [CrossRef] [PubMed]
- Connor, S.A.; Ammendrup-Johnsen, I.; Chan, A.W.; Kishimoto, Y.; Murayama, C.; Kurihara, N.; Tada, A.; Ge, Y.; Lu, H.; Yan, R.; et al. Altered Cortical Dynamics and Cognitive Function upon Haploinsufficiency of the Autism-Linked Excitatory Synaptic Suppressor MDGA2. Neuron 2016, 91, 1052–1068. [Google Scholar] [CrossRef] [PubMed]
- Sanders, S.J.; Ercan-Sencicek, A.G.; Hus, V.; Luo, R.; Murtha, M.T.; Moreno-De-Luca, D.; Chu, S.H.; Moreau, M.P.; Gupta, A.R.; Thomson, S.A.; et al. Multiple recurrent de novo CNVs, including duplications of the 7q11.23 Williams syndrome region, are strongly associated with autism. Neuron 2011, 70, 863–885. [Google Scholar] [CrossRef] [PubMed]
- Feng, J.; Schroer, R.; Yan, J.; Song, W.; Yang, C.; Bockholt, A.; Cook, E.H., Jr.; Skinner, C.; Schwartz, C.E.; Sommer, S.S. High frequency of neurexin 1beta signal peptide structural variants in patients with autism. Neurosci. Lett. 2006, 409, 10–13. [Google Scholar] [CrossRef]
- Krezel, W.; Dupont, S.; Krust, A.; Chambon, P.; Chapman, P.F. Increased anxiety and synaptic plasticity in estrogen receptor beta—Deficient mice. Proc. Natl. Acad. Sci. USA 2001, 98, 12278–12282. [Google Scholar] [CrossRef]
- Gandelman, R.; Simon, N.G.; McDermott, N.J. Prenatal exposure to testosterone and its precursors influences morphology and later behavioral responsiveness to testosterone of female mice. Physiol. Behav. 1979, 23, 23–26. [Google Scholar] [CrossRef] [PubMed]
- vom Saal, F.S. Prenatal exposure to androgen influences morphology and aggressive behavior of male and female mice. Horm. Behav. 1979, 12, 1–11. [Google Scholar] [CrossRef]
- Hatanaka, Y.; Wada, K.; Kabuta, T. Abnormal instability, excess density, and aberrant morphology of dendritic spines in prenatally testosterone-exposed mice. Neurochem. Int. 2015, 85–86, 53–58. [Google Scholar] [CrossRef] [PubMed]
- Archer, J. Testosterone and persistence in mice. Anim. Behav. 1977, 25, 479–488. [Google Scholar] [CrossRef] [PubMed]
- Smith, S.E.; Li, J.; Garbett, K.; Mirnics, K.; Patterson, P.H. Maternal immune activation alters fetal brain development through interleukin-6. J. Neurosci. Off. J. Soc. Neurosci. 2007, 27, 10695–10702. [Google Scholar] [CrossRef]
- Carlezon, W.A., Jr.; Kim, W.; Missig, G.; Finger, B.C.; Landino, S.M.; Alexander, A.J.; Mokler, E.L.; Robbins, J.O.; Li, Y.; Bolshakov, V.Y.; et al. Maternal and early postnatal immune activation produce sex-specific effects on autism-like behaviors and neuroimmune function in mice. Sci. Rep. 2019, 9, 16928. [Google Scholar] [CrossRef]
- Braun, A.E.; Carpentier, P.A.; Babineau, B.A.; Narayan, A.R.; Kielhold, M.L.; Moon, H.M.; Shankar, A.; Su, J.; Saravanapandian, V.; Haditsch, U.; et al. “Females Are Not Just ‘Protected’ Males”: Sex-Specific Vulnerabilities in Placenta and Brain after Prenatal Immune Disruption. ENeuro 2019, 6, ENEURO.0358-19.2019. [Google Scholar] [CrossRef]
- Perez-Pouchoulen, M.; Miquel, M.; Saft, P.; Brug, B.; Toledo, R.; Hernandez, M.E.; Manzo, J. Prenatal exposure to sodium valproate alters androgen receptor expression in the developing cerebellum in a region and age specific manner in male and female rats. Int. J. Dev. Neurosci. Off. J. Int. Soc. Dev. Neurosci. 2016, 53, 46–52. [Google Scholar] [CrossRef] [PubMed]
- Jacquemont, S.; Coe, B.P.; Hersch, M.; Duyzend, M.H.; Krumm, N.; Bergmann, S.; Beckmann, J.S.; Rosenfeld, J.A.; Eichler, E.E. A higher mutational burden in females supports a "female protective model" in neurodevelopmental disorders. Am. J. Hum. Genet. 2014, 94, 415–425. [Google Scholar] [CrossRef]
- Ostatníková, D.; Lakatošová, S.; Babková, J.; Hodosy, J.; Celec, P. Testosterone and the brain: From cognition to autism. Physiol. Res. 2020, 69, S403–S419. [Google Scholar] [CrossRef]
- Quartier, A.; Chatrousse, L.; Redin, C.; Keime, C.; Haumesser, N.; Maglott-Roth, A.; Brino, L.; Le Gras, S.; Benchoua, A.; Mandel, J.L.; et al. Genes and Pathways Regulated by Androgens in Human Neural Cells, Potential Candidates for the Male Excess in Autism Spectrum Disorder. Biol. Psychiatry 2018, 84, 239–252. [Google Scholar] [CrossRef] [PubMed]
- Sedgewick, F.; Hill, V.; Yates, R.; Pickering, L.; Pellicano, E. Gender Differences in the Social Motivation and Friendship Experiences of Autistic and Non-autistic Adolescents. J. Autism Dev. Disord. 2016, 46, 1297–1306. [Google Scholar] [CrossRef]
- Allison, C.; Auyeung, B.; Baron-Cohen, S. Toward brief “Red Flags” for autism screening: The Short Autism Spectrum Quotient and the Short Quantitative Checklist for Autism in toddlers in 1,000 cases and 3,000 controls [corrected]. J. Am. Acad. Child Adolesc. Psychiatry 2012, 51, 202–212. [Google Scholar] [CrossRef] [PubMed]
- Bargiela, S.; Steward, R.; Mandy, W. The Experiences of Late-diagnosed Women with Autism Spectrum Conditions: An Investigation of the Female Autism Phenotype. J. Autism Dev. Disord. 2016, 46, 3281–3294. [Google Scholar] [CrossRef] [PubMed]

| Focus | Subjects | Phenotype | Ref | |
|---|---|---|---|---|
| Sex differences in brain morphology | Sex differences in the absolute and proportional volumes of the main language-associated regions of the cerebral cortex | 21 postmortem brains (10 males, 49–86 y; 11 females, 46–92 y) | Larger volume of the superior temporal cortex and the cortical volume fraction of the Broca’s area in females | [33] |
| The cortical sulci formation in the preterm newborn brain | 35 preterm newborns (16 females, 19 males; 25.6–35.6 w) | Lower cortical surface, smaller cortex, and white matter volumes were observed in females | [31] | |
| The relationship between the structure of the ventral frontal cortex and social cognition in female and male | 60 healthy, right-handed Caucasian participants (30 males, 18–50 y; 30 females, 18–50 y) | Larger SG in females, femininity correlated with greater SG gray matter volume, and surface area and better performance on the IPT | [34] | |
| Normal sexual dimorphisms of cortical and subcortical brain regions | 48 participants from Boston, the United States (27 males, 39.1 ± 12 y; 21 females, 36.3 ± 10.5 y) | Larger volumes of frontal and medial paralimbic cortices in females, larger volumes of frontomedial cortex, the amygdala, and hypothalamus in males | [35] | |
| Sex differences in the corpus callosum | 246 midsagittal images were selected out of approximately 1000 sets of MRIs scanned at four southern California MRI centers | Splenium was bulbous-shaped in females, tubular-shaped in males | [36] | |
| Age and sex differences in the cerebellum and the ventral pons | 190 participants (77 males, 45.74 ± 16.90 y; 113 females, 47.09 ± 16.17 y) | Age-related reduction in the cerebellar hemispheres and the cerebellar vermis; the volume of the cerebellar hemispheres, the vermis, and the ventral pons were larger in males | [37] | |
| Sex differences in SDN-POA | 30 postmortem human brains (13 males and 17 females, 10–93 y) | The volume of the SDN-POA was 2.2 times as large in males than in females and contained 2.1 times as many cells | [38] | |
| Sex differences in the structural connectome | 949 participants (428 males, 521 females) | Greater within-hemispheric connectivity in males; between-hemispheric connectivity in females | [39] | |
| ASD development and sex differences in ASD and ASD-like behaviors | The prevalence and characteristics of ASD | Children aged 8 years in 2018 whose parents or guardians lived in 11 ADDM Network sites in the United States | The overall ASD prevalence estimate was one in 44 children aged 8 years in 2018 | [7] |
| The association of genetic liability and sex biases in ASD | 970 Caucasian families (2028 individuals) with at least two related ASD individuals from the ten AGP sites in North America and Europe | Lower repetitive behavior scores in females; higher scores in males from female containing families that had higher repetitive behavior scores | [8] | |
| ASD individuals and sex differences in the Empathy Quotient score | 90 adults with AS/HFA (65 males, 25 females, 15.4–59.9 y); 71 males (17.4–69.6 y); 126 females (17.7–73.0 y) | Adults with AS or HFA scored significantly lower on the EQ; in a general population, females scored higher than males. | [40] | |
| Sex differences in attention to social stimuli | 74 school-aged males and females with ASD (23 males, 19 females) and without ASD (16 males, 16 females) | ASD females attended more to faces in the socially lean condition; ASD males attended less to faces regardless of social context | [41] | |
| Sex differences in restricted and repetitive behaviors and interests in ASD youths | 615 youth with ASD (507 boys, 108 girls, 3–18 y) | Increased self-injurious, compulsive, restricted, and insistence on sameness behaviors in females; stereotyped and restricted behaviors in males | [42] | |
| To determine if the risk in these families follows a female protective model by assessing recurrence patterns in multiplex families | 12,260 individuals from 2278 families acquired from Autism Genetics Resource Exchange | Higher recurrence in males; interbirth interval negatively correlated with ASD recurrence that is driven by children in male-only families | [43] | |
| To examine whether females with ASD would display better social skills than males with ASD on a test of friendship and social function | 101 participants in total; 50 ASD adolescents (25 males, 25 females), 51 typically developing participants (26 males, 25 females) | Females demonstrated higher scores on the friendship questionnaire than males | [44] | |
| Sex differences in the expression of RORA | Human neuroblastoma cells SH-SY5Y | Sex differences were observed in the expression of Rora/RORA and its transcriptional targets in the human brain | [9] | |
| Dysregulation of ERβ, aromatase, and ER co-activators in the middle frontal gyrus of ASD individuals | 13 postmortem middle frontal gyrus tissues of ASD and 13 control subjects | Decrease in ERβ mRNA expression in the middle frontal gyrus of ASD subjects; a reduction in CYP19A1 mRNA expression was observed in ASD subjects | [45] | |
| Differences in male and female hormones in the regulation of RORA, a novel candidate ASD gene | The human neuroblastoma cells SH-SY5Y | Male and female hormones differentially regulate RORA | [46] | |
| Dendritic spines on Golgi-impregnated cortical pyramidal cells in the cortex of ASD subjects and age-matched control cases. | Cortical tissue samples were acquired from 10 postmortem ASD males and 15 male control subjects | Greater spine densities were observed in ASD subjects | [47] | |
| Effects of testosterone or other sex hormones | The differences in reproductive hormone levels and their biological effects between full-term and preterm infant boys | 25 full-term and 25 preterm boys (gestational age 24.7–36.6 w) | Postnatal HPG axis activation in infancy is increased in preterm boys and associated with faster testicular and penile growth | [48] |
| The effects of testosterone-related genetic variants on facial morphology | 7418 individuals belonging to three separate cohorts: 2297 participants (3–40 y, Western-European descent); 1555 participants (18–83 y; European descent); 3566 participants (14–17 y, Western–European descent) | Two intronic variants of SHBG, rs12150660 and rs1799941, showed an effect on mandible shape; Rs8023580 showed an association with the total and upper facial width-to-height ratios | [49] | |
| The effects of fetal testosterone on organizing the human brain for expression of sexual dimorphism later in life | 28 boys (8 –11 y); 101 males (8–11 y), and 116 females (8–11 years) from the NIH Pediatric MRI Repository | Gray matter volume in RTPJ/pSTS was greater in males, positively predicted by fetal testosterone; gray matter volume in PT/PO and plOFC was greater in females, negatively predicted by fetal testosterone | [50] | |
| Effects of fetal testosterone on visuospatial ability in children | 64 children (35 boys, 29 girls, 7–10 y) | Fetal testosterone was a significant predictor for Embedded Figures Test scores in both boys and girls | [51] | |
| The association of fetal testosterone with ASD traits in children | 129 participants (66 boys, 63 girls) | Boys scoring significantly higher in the Q-CHAT | [52] | |
| The association of fetal testosterone and ASD traits | 235 (118 boys, 117 girls) | Fetal testosterone levels were positively associated with higher scores on the CAST and the AQ-Child | [53] | |
| The relationship between parameters of androgenicity (plasmatic testosterone levels and androgen receptor sensitivity) and hyperactivity in boys | 60 ASD boys (3–15 y) | No association between actual plasmatic testosterone levels and hyperactivity symptoms; the number of CAG triplets was negatively correlated with hyperactivity symptoms | [54] | |
| The effects of MIA or PCOS on offspring | The association of MIA and ASD development in the offspring | 114,500 children born in 1999–2009 from The Norwegian Mother and Child Cohort Study | Prenatal fever was associated with increased ASD risk | [18] |
| The risk of developing PDDs in the offspring of women with PCOS | 30 pregnant PCOS patients with hyperandrogenemia and 45 other pregnant healthy women | Children of PCOS patients scored higher on the AQC; daughters of PCOS patients scored higher on communication and attention switching | [55] | |
| The risk of developing ASD in the offspring of women with PCOS | 971 women with autism were included in Study 1; 26,263 women with PCOS phenotype were included in Study 2; 8588 children of women with the PCOS phenotype in Study 2 who had first-born children linked in the database | Increased prevalence of PCOS in women with ASD and elevated rates of ASD in women with PCOS | [56] | |
| Focus | Species and Strain | Method | Phenotype | Ref | |
|---|---|---|---|---|---|
| Effects of T or other sex hormones | Effects of neonatal androgen exposure on ovulation, sexual receptivity, and aggressive behaviors in female mice | Swiss Webster mice | Either 100μg TP, 100μg T, or 100μg AD on PN0–3 | Females given either TP, T, or AD neonatally were anovulatory; more aggression in females given either TP or T neonatally | [58] |
| Effects of prenatal T exposure on morphology and later behavior | Rockland Swiss albino mice | Either 1 mg T, 1 mg DHE, 1 mg PRO, or 1 mg PREG s.c. on E11–17 (1 mg/0.1 m) | Prenatal T, DHE, or PRO exposure increased anogenital distance of female mice | [80] | |
| Effects of prenatal T exposure on morphology and aggressive behavior | Female Rockland Swiss mice | 1.5 mg TP s.c. on E12–16 | Prenatal T exposure increased aggression in female offspring | [81] | |
| Effects of pre- and post-natal T exposure on differentiation of SDN-POA | Sprague Dawley rats | 2 mg TP or DES s.c. from E16 until delivery | Larger SDN-POA in female rats treated with T propionate or diethylstilbestrol | [38] | |
| Effects of prenatal T exposure on dendritic spines | Mice expressing YFP in layer 5 pyramidal neurons (C57BL/6J background) | 0.1 mg TP s.c. on E16–18 | Instability and excess density of dendritic spines were observed in prenatally T-exposed mice | [82] | |
| Effects of T on the persistence of food searching | 64 mice (48 males, 16 females)Half of the mice were BALB/c strain, and the other half were ‘porton’ mice | 7.5 mg T enanthate | T produced increased persistence in castrated mice | [83] | |
| To establish degus as a highly socialized animal model and the effects of T on sociality-related genes | Octodon degus | - | Neurexin and neuroligin in degus brain are highly conserved to that of human sequences; T directly binds to degus neurexin and intercepts intercellular neurexin–neuroligin binding | [75] | |
| Effects of MIA induced by poly(I:C) or LPS | Sex differences in behavioral deficits and neuropathology of ASD | C57BL/6J mice | poly(I:C) (20 mg/kg) i.p. on E12.5 | Reduced social interactions and motor development and coordination deficits; reduced numbers of Purkinje cells in the cerebellum were more widespread in males | [20] |
| Effects of prenatal allergen exposure on sexual differentiation, social, and sexual behaviors | Sprague Dawley rats | LPS (200 μ/kg) i.p. on PN0; Prior to pregnancy: 1 mg valbumin (4 mg/mL) subcutaneous injection; after 2 weeks: 1 mg ovalbumin-alum adjuvant injection | Increased mast cell and microglia activation in the neonatal brain; an increase in male-typical mounting behavior in adult females | [30] | |
| The mechanism by which MIA causes long-term behavioral deficits in the offspring | Female C57BL/6J mice; IL-6 KO mice (C57BL/6J background) | Cytokine injection: Either 5 μg carrierprotein-free recombinant mouse IL-6 or carrier-free recombinant mouse IFNγ i.p. Cytokine blocking: 20 mg/kg poly(I:C) i.p. | IL-6 caused prepulse inhibition and latent inhibition deficits in the adult offspring; MIA in IL-6 KO mice does not result in several behavioral changes | [84] | |
| Effects of MIA and EIA on ASD-like behaviors and neuroimmune function | C57BL/6J mice | 20 mg/kg poly(I:C) i.p. on E12.510 mg/kg LPS on PN9 | EIA-produced sex-specific behavioral effects and immune responses in the brain | [85] | |
| Sex differences in immune-driven alterations in fetal brain development and related outcomes in female and male mice | C57BL/6J mice | 30 or 60 µg/kg LPS i.p. on E12.5 | A unique set of vulnerabilities and developmental consequences only in females | [86] | |
| Studies using mutant mice and others | Effects of Mdga2 haploinsufficiency on behavioral phenotypes related to ASD | Mdga2 null mice | - | Increased excitatory synapses and function, altered LTP, and ASD-like phenotype | [76] |
| Effects of increased anxiety on synaptic plasticity in ERβ-deficient mice | ERα and ERβ mutant mice | - | Increased anxiety, reduced threshold for the induction of synaptic plasticity in the amygdala in ERβ mutant females | [79] | |
| Effects of prenatal VPA exposure on AR | Male and female Wistar rats | 600 mg.kg VPA i.p. on E12 | VPA exposure alters AR expression in the postnatal developing cerebellum in both male and female rats | [87] | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, M.; Usui, N.; Shimada, S. Prenatal Sex Hormone Exposure Is Associated with the Development of Autism Spectrum Disorder. Int. J. Mol. Sci. 2023, 24, 2203. https://doi.org/10.3390/ijms24032203
Li M, Usui N, Shimada S. Prenatal Sex Hormone Exposure Is Associated with the Development of Autism Spectrum Disorder. International Journal of Molecular Sciences. 2023; 24(3):2203. https://doi.org/10.3390/ijms24032203
Chicago/Turabian StyleLi, Mengwei, Noriyoshi Usui, and Shoichi Shimada. 2023. "Prenatal Sex Hormone Exposure Is Associated with the Development of Autism Spectrum Disorder" International Journal of Molecular Sciences 24, no. 3: 2203. https://doi.org/10.3390/ijms24032203
APA StyleLi, M., Usui, N., & Shimada, S. (2023). Prenatal Sex Hormone Exposure Is Associated with the Development of Autism Spectrum Disorder. International Journal of Molecular Sciences, 24(3), 2203. https://doi.org/10.3390/ijms24032203





