Amh/Amhr2 Signaling Causes Masculinization by Inhibiting Estrogen Synthesis during Gonadal Sex Differentiation in Japanese Flounder (Paralichthys olivaceus)
Abstract
:1. Introduction
2. Results
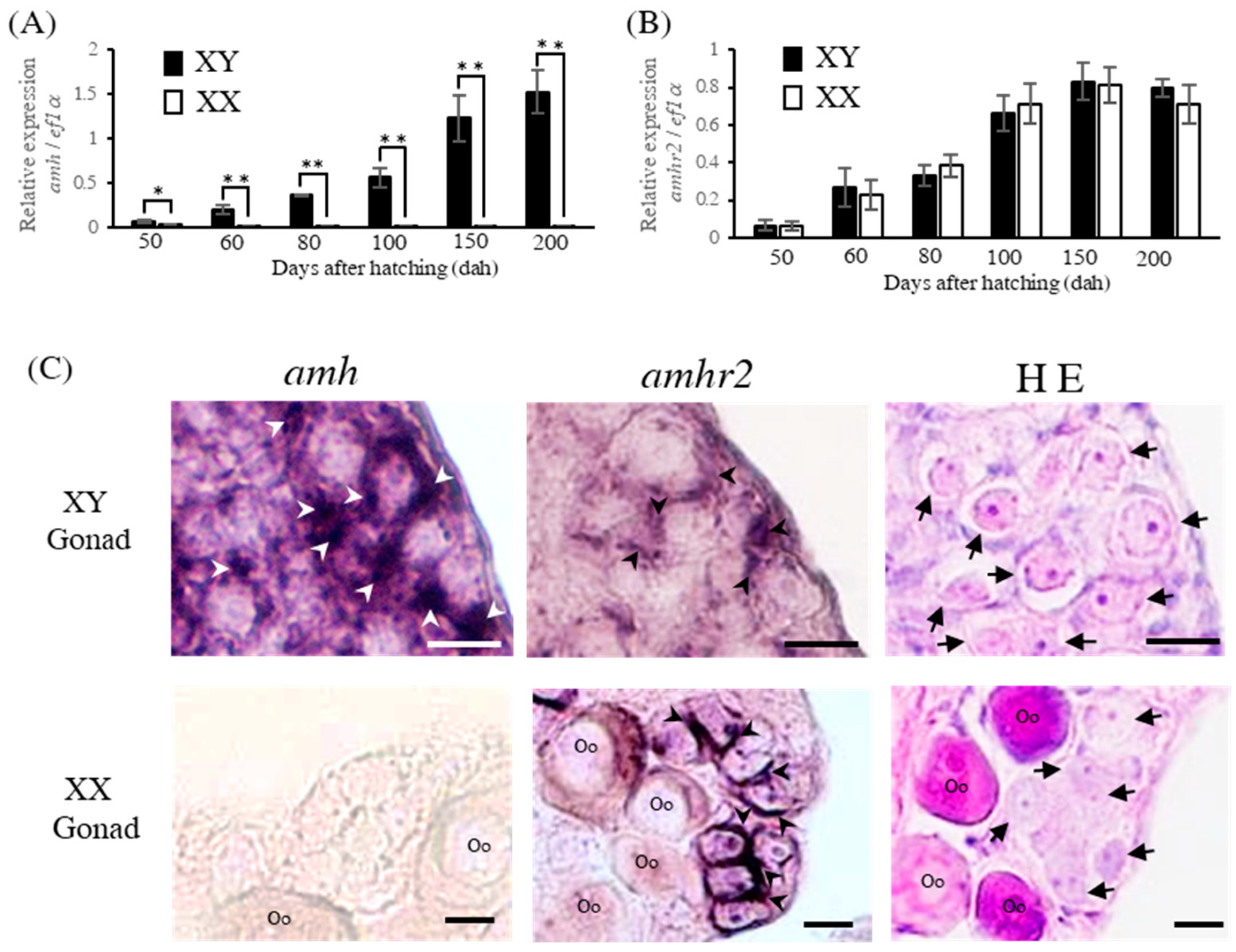
2.1. Expression Analysis of amh and amhr2 in Gonads in XY and XX Flounder
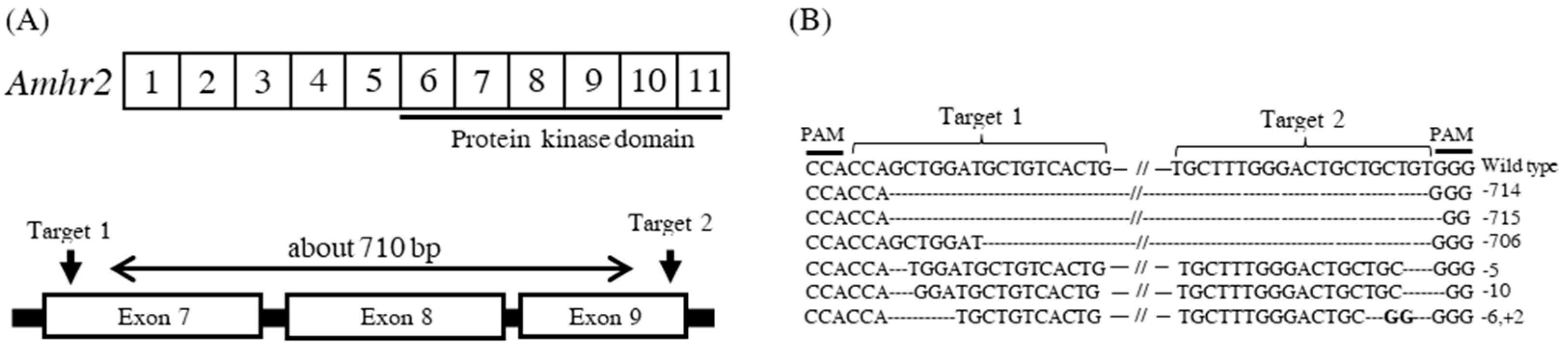
2.2. Generation of amhr2-Deficient Mutant Using CRISPR/Cas9 System in Japanese Flounder
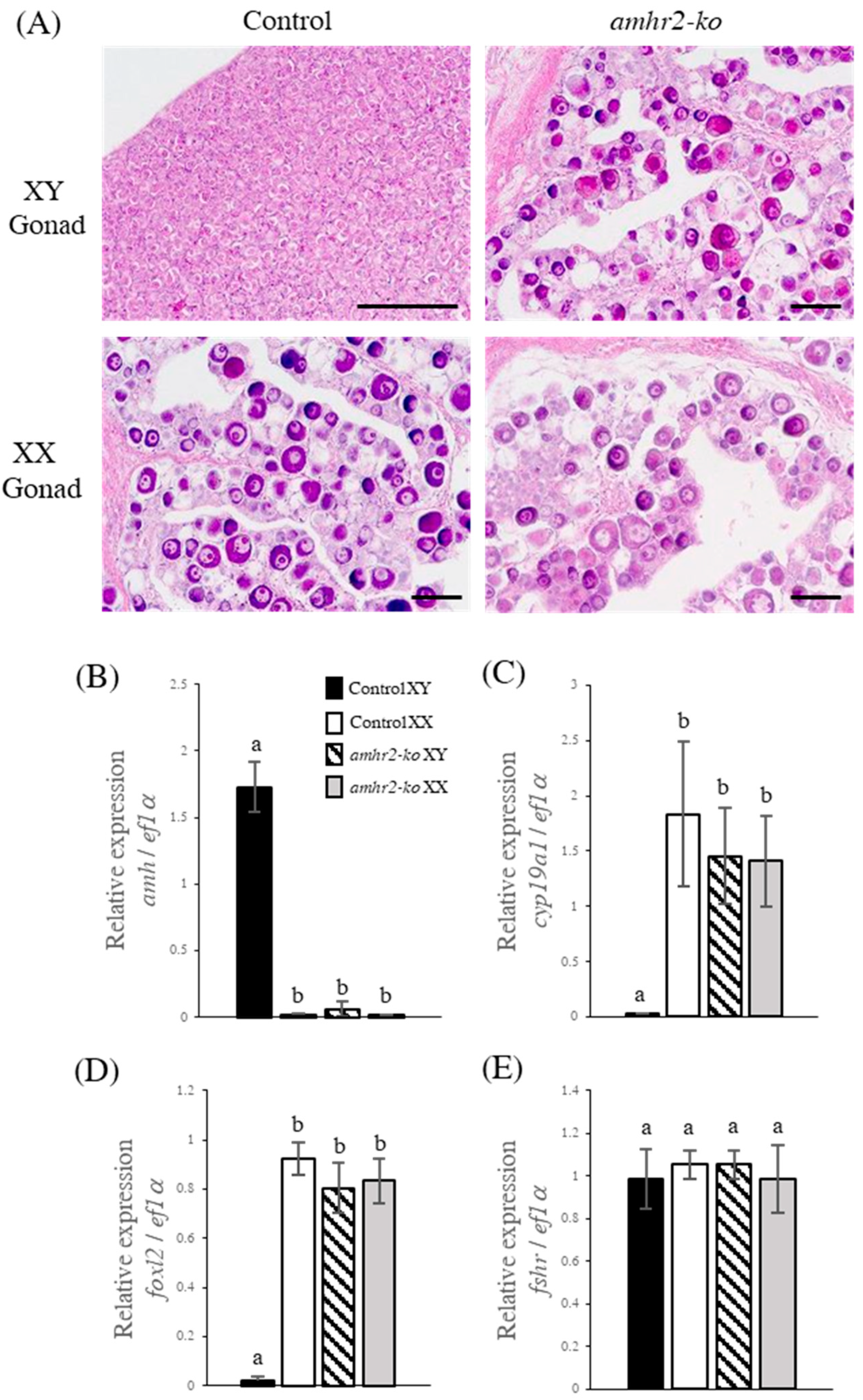
2.3. amhr2 Mutants Showed Phenotypic Sex Reversal from Male to Female
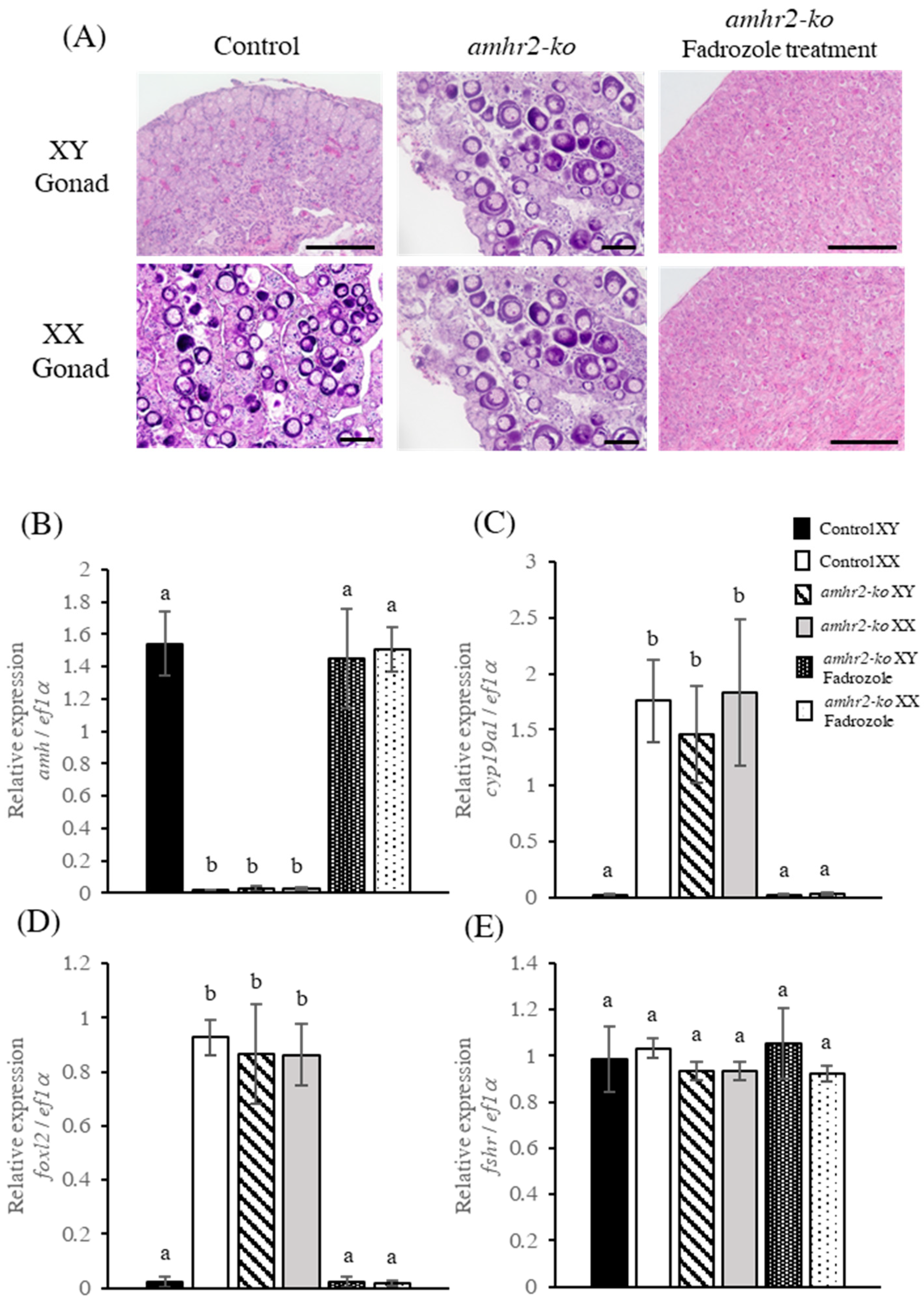
2.4. Effect of Fadrozole Treatment on Gonadal Phenotype of amhr2 Mutants
3. Discussion
4. Materials and Methods
4.1. Experimental Animals
4.2. Determination of Genetic Sex
4.3. Expression Analysis by Real-Time PCR
4.4. Histological Analysis of amhr2 Transcripts Using In Situ Hybridization
4.5. Generation of amhr2 Mutant Using CRISPR/Cas9 System
4.6. Analysis of Genotype in amhr2 Mutants
4.7. Histological Observations
4.8. Experimental Treatment with Fadrozole
4.9. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Baroiller, J.F.; D’Cotta, H. The Reversible Sex of Gonochoristic Fish: Insights and Consequences. Sex Dev. 2016, 10, 242–266. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, E. Studies on sex-manipulation and production of cloned populations in hirame, Paralichthys olivaceus (Temminck et Schlegel). Aquaculture 1999, 173, 235–246. [Google Scholar] [CrossRef]
- Tabata, K. Reduction of female proportion in lower growing fish separated from normal and feminized seedlings of hirame Paralichthys olivaceus. Fish Sci. 1995, 61, 199–201. [Google Scholar] [CrossRef] [Green Version]
- Kitano, T.; Takamune, K.; Nagahama, Y.; Abe, S. Role of P450 aromatase in gonadal sex differentiation in Japanese flounder (Paralichthys olivaceus). Environ. Sci. 2001, 8, 1–11. [Google Scholar]
- Ospina-Alvarez, N.; Piferrer, F. Temperature-dependent sex determination in fish revisited: Prevalence, a single sex ratio response pattern, and possible effects of climate change. PLoS ONE 2008, 3, e2837. [Google Scholar] [CrossRef] [Green Version]
- Rajendiran, P.; Jaafar, F.; Kar, S.; Sudhakumari, C.; Senthilkumaran, B.; Parhar, I.S. Sex Determination and Differentiation in Teleost: Roles of Genetics, Environment, and Brain. Biology 2021, 10, 973. [Google Scholar] [CrossRef]
- Kitano, T.; Takamune, K.; Nagahama, Y.; Abe, S.I. Aromatase inhibitor and 17alpha-methyltestosterone cause sex-reversal from genetical females to phenotypic males and suppression of P450 aromatase gene expression in Japanese flounder (Paralichthys olivaceus). Mol. Reprod. Dev. 2000, 56, 1–5. [Google Scholar] [CrossRef]
- Kitano, T.; Yoshinaga, N.; Shiraishi, E.; Koyanagi, T.; Abe, S. Tamoxifen induces masculinization of genetic females and regulates P450 aromatase and Müllerian inhibiting substance mRNA expression in Japanese flounder (Paralichthys olivaceus). Mol. Reprod. Dev. 2007, 74, 1171–1177. [Google Scholar] [CrossRef]
- Kitano, T.; Hayashi, Y.; Shiraishi, E.; Kamei, Y. Estrogen rescues masculinization of genetically female medaka by exposure to cortisol or high temperature. Mol. Reprod. Dev. 2012, 79, 719–726. [Google Scholar] [CrossRef]
- Yamaguchi, T.; Yamaguchi, S.; Hirai, T.; Kitano, T. Follicle-stimulating hormone signaling and Foxl2 are involved in transcriptional regulation of aromatase gene during gonadal sex differentiation in Japanese flounder, Paralichthys olivaceus. Biochem. Biophys. Res. Commun. 2007, 359, 935–940. [Google Scholar] [CrossRef]
- Yamaguchi, T.; Yoshinaga, N.; Yazawa, T.; Gen, K.; Kitano, T. Cortisol is involved in temperature-dependent sex determination in the Japanese flounder. Endocrinology 2010, 151, 3900–3908. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fitzpatrick, S.L.; Richards, J.S. Identification of a cyclic adenosine 3’,5’-monophosphate-response element in the rat aromatase promoter that is required for transcriptional activation in rat granulosa cells and R2C Leydig cells. Mol. Endocrinol. 1994, 8, 1309–1319. [Google Scholar] [PubMed] [Green Version]
- Guiguen, Y.; Fostier, A.; Piferrer, F.; Chang, C.F. Ovarian aromatase and estrogens: A pivotal role for gonadal sex differentiation and sex change in fish. Gen. Comp. Endocrinol. 2010, 165, 352–366. [Google Scholar] [CrossRef]
- Huang, S.; Ye, L.; Chen, H. Sex determination and maintenance: The role of DMRT1 and FOXL2. Asian J. Androl. 2017, 19, 619–624. [Google Scholar] [PubMed]
- Hayashi, Y.; Kobira, H.; Yamaguchi, T.; Shiraishi, E.; Yazawa, T.; Hirai, T.; Kamei, Y.; Kitano, T. High temperature causes masculinization of genetically female medaka by elevation of cortisol. Mol. Reprod. Dev. 2010, 77, 679–686. [Google Scholar] [CrossRef] [PubMed]
- Hattori, R.S.; Fernandino, J.I.; Kishii, A.; Kimura, H.; Kinno, T.; Oura, M.; Somoza, G.M.; Yokota, M.; Strüssmann, C.A.; Watanabe, S. Cortisol-induced masculinization: Does thermal stress affect gonadal fate in pejerrey, a teleost fish with temperature-dependent sex determination? PLoS ONE 2009, 4, e6548. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yoshinaga, N.; Shiraishi, E.; Yamamoto, T.; Iguchi, T.; Abe, S.; Kitano, T. Sexually dimorphic expression of a teleost homologue of Müllerian inhibiting substance during gonadal sex differentiation in Japanese flounder, Paralichthys olivaceus. Biochem. Biophys. Res. Commun. 2004, 322, 508–513. [Google Scholar] [CrossRef]
- Josso, N. In vitro synthesis of müllerian-inhibiting hormone by seminiferous tubules isolated from the calf fetal testis. Endocrinology 1973, 93, 829–834. [Google Scholar] [CrossRef]
- Tilmann, C.; Capel, B. Cellular and molecular pathways regulating mammalian sex determination. Recent Prog. Horm. Res. 2002, 57, 1–18. [Google Scholar] [CrossRef] [Green Version]
- Park, S.Y.; Jameson, J.L. Minireview: Transcriptional regulation of gonadal development and differentiation. Endocrinology 2005, 146, 1035–1042. [Google Scholar] [CrossRef] [Green Version]
- Josso, N.; Racine, C.; di Clemente, N.; Rey, R.; Xavier, F. The role of anti-Müllerian hormone in gonadal development. Mol. Cell. Endocrinol. 1998, 145, 3–7. [Google Scholar] [CrossRef] [PubMed]
- Behringer, R.R.; Cate, R.L.; Froelick, G.J.; Palmiter, R.D.; Brinster, R.L. Abnormal sexual development in transgenic mice chronically expressing müllerian inhibiting substance. Nature 1990, 345, 167–170. [Google Scholar] [CrossRef] [PubMed]
- Mishina, Y.; Whitworth, D.J.; Racine, C.; Behringer, R.R. High specificity of Müllerian-inhibiting substance signaling in vivo. Endocrinology 1999, 140, 2084–2088. [Google Scholar] [CrossRef] [PubMed]
- Hattori, R.S.; Kumazawa, K.; Nakamoto, M.; Nakano, Y.; Yamaguchi, T.; Kitano, T.; Yamamoto, E.; Fuji, K.; Sakamoto, T. Y-specific amh allele, amhy, is the master sex-determining gene in Japanese flounder Paralichthys olivaceus. Front. Genet. 2022, 13, 1007548. [Google Scholar] [CrossRef] [PubMed]
- Sawamura, R.; Osafune, N.; Murakami, T.; Furukawa, F.; Kitano, T. Generation of biallelic F0 mutants in medaka using the CRISPR/Cas9 system. Genes Cells 2017, 22, 756–763. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rodríguez-Marí, A.; Yan, Y.L.; Bremiller, R.A.; Wilson, C.; Cañestro, C.; Postlethwait, J.H. Characterization and expression pattern of zebrafish Anti-Müllerian hormone (Amh) relative to sox9a, sox9b, and cyp19a1a, during gonad development. Gene Expr. Patterns 2005, 5, 655–667. [Google Scholar] [CrossRef]
- Ijiri, S.; Kaneko, H.; Kobayashi, T.; Wang, D.S.; Sakai, F.; Paul-Prasanth, B.; Nakamura, M.; Nagahama, Y. Sexual dimorphic expression of genes in gonads during early differentiation of a teleost fish, the Nile tilapia Oreochromis niloticus. Biol. Reprod. 2008, 78, 333–341. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mullen, R.D.; Ontiveros, A.E.; Moses, M.M.; Behringer, R.R. AMH and AMHR2 mutations: A spectrum of reproductive phenotypes across vertebrate species. Dev. Biol. 2019, 455, 1–9. [Google Scholar] [CrossRef]
- Oréal, E.; Mazaud, S.; Picard, J.Y.; Magre, S.; Carré-Eusèbe, D. Different patterns of anti-Müllerian hormone expression, as related to DMRT1, SF-1, WT1, GATA-4, Wnt-4, and Lhx9 expression, in the chick differentiating gonads. Dev. Dyn. 2002, 225, 221–232. [Google Scholar] [CrossRef]
- Klüver, N.; Pfennig, F.; Pala, I.; Storch, K.; Schlieder, M.; Froschauer, A.; Gutzeit, H.O.; Schartl, M. Differential expression of anti-Müllerian hormone (amh) and anti-Müllerian hormone receptor type II (amhrII) in the teleost medaka. Dev. Dyn. 2007, 236, 271–281. [Google Scholar] [CrossRef]
- Shiraishi, E.; Yoshinaga, N.; Miura, T.; Yokoi, H.; Wakamatsu, Y.; Abe, S.; Kitano, T. Mullerian inhibiting substance is required for germ cell proliferation during early gonadal differentiation in medaka (Oryzias latipes). Endocrinology 2008, 149, 1813–1819. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kawabe, T.; Kariya, H.; Hara, S.; Shirozu, T.; Shiraishi, E.; Mukai, K.; Yazawa, T.; Inoue, S.; Kitano, T. Transcriptional regulation of Müllerian inhibiting substance (MIS) and establishment of a gonadal somatic cell line using mis-GFP transgenic medaka (Oryzias latipes). Front. Endocrinol. 2020, 11, 578885. [Google Scholar] [CrossRef] [PubMed]
- von Hofsten, J.; Larsson, A.; Olsson, P.E. Novel steroidogenic factor-1 homolog (ff1d) is coexpressed with anti-Mullerian hormone (AMH) in zebrafish. Dev. Dyn. 2005, 233, 595–604. [Google Scholar] [CrossRef] [PubMed]
- Belville, C.; Maréchal, J.D.; Pennetier, S.; Carmillo, P.; Masgrau, L.; Messika-Zeitoun, L.; Galey, J.; Machado, G.; Treton, D.; Gonzalès, J.; et al. Natural mutations of the anti-Mullerian hormone type II receptor found in persistent Mullerian duct syndrome affect ligand binding, signal transduction and cellular transport. Hum. Mol. Genet. 2009, 18, 3002–3013. [Google Scholar] [CrossRef] [Green Version]
- Morinaga, C.; Saito, D.; Nakamura, S.; Sasaki, T.; Asakawa, S.; Shimizu, N.; Mitani, H.; Furutani-Seiki, M.; Tanaka, M.; Kondoh, H. The hotei mutation of medaka in the anti-Mullerian hormone receptor causes the dysregulation of germ cell and sexual development. Proc. Natl. Acad. Sci. USA 2007, 104, 9691–9696. [Google Scholar] [CrossRef] [Green Version]
- Kamiya, T.; Kai, W.; Tasumi, S.; Oka, A.; Matsunaga, T.; Mizuno, N.; Fujita, M.; Suetake, H.; Suzuki, S.; Hosoya, S.; et al. A trans-species missense SNP in Amhr2 is associated with sex determination in the tiger pufferfish, Takifugu rubripes (fugu). PLoS Genet. 2012, 8, e1002798. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Josso, N.; di Clemente, N.; Gouédard, L. Anti-Müllerian hormone and its receptors. Mol. Cell. Endocrinol. 2001, 179, 25–32. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Sun, Y.; Zhao, J.; Shi, H.; Zeng, S.; Ye, K.; Jiang, D.; Zhou, L.; Sun, L.; Tao, W.; et al. A Tandem Duplicate of Anti-Müllerian Hormone with a Missense SNP on the Y Chromosome Is Essential for Male Sex Determination in Nile Tilapia, Oreochromis niloticus. PLoS Genet. 2015, 11, e1005678. [Google Scholar] [CrossRef] [Green Version]
- Wang, D.S.; Kobayashi, T.; Zhou, L.Y.; Paul-Prasanth, B.; Ijiri, S.; Sakai, F.; Okubo, K.; Morohashi, K.; Nagahama, Y. Foxl2 up-regulates aromatase gene transcription in a female-specific manner by binding to the promoter as well as interacting with ad4 binding protein/steroidogenic factor 1. Mol. Endocrinol. 2007, 21, 712–725. [Google Scholar] [CrossRef] [Green Version]
- Okada, E.; Yoshimoto, S.; Ikeda, N.; Kanda, H.; Tamura, K.; Shiba, T.; Takamatsu, N.; Ito, M. Xenopus W-linked DM-W induces Foxl2 and Cyp19 expression during ovary formation. Sex Dev. 2009, 3, 38–42. [Google Scholar] [CrossRef] [PubMed]
- Uhlenhaut, N.H.; Jakob, S.; Anlag, K.; Eisenberger, T.; Sekido, R.; Kress, J.; Treier, A.C.; Klugmann, C.; Klasen, C.; Holter, N.I.; et al. Somatic sex reprogramming of adult ovaries to testes by FOXL2 ablation. Cell 2009, 139, 1130–1142. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hattori, R.S.; Oura, M.; Sakamoto, T.; Yokota, M.; Watanabe, S.; Strüssmann, C.A. Establishment of a strain inheriting a sex-linked SNP marker in Patagonian pejerrey (Odontesthes hatcheri), a species with both genetic and temperature-dependent sex determination. Anim. Genet. 2010, 41, 81–84. [Google Scholar] [CrossRef] [PubMed]
- Xie, F.; Xiao, P.; Chen, D.; Xu, L.; Zhang, B. miRDeepFinder: A miRNA analysis tool for deep sequencing of plant small RNAs. Plant Mol. Biol. 2012, 80, 75–84. [Google Scholar] [CrossRef] [PubMed]
- Silver, N.; Best, S.; Jiang, J.; Thein, S.L. Selection of housekeeping genes for gene expression studies in human reticulocytes using real-time PCR. BMC Mol. Biol. 2006, 7, 1–9. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pfaffl, M.W.; Tichopad, A.; Prgomet, C.; Neuvians, T.P. Determination of stable housekeeping genes, differentially regulated target genes and sample integrity: BestKeeper--Excel-based tool using pair-wise correlations. Biotechnol. Lett. 2004, 26, 509–515. [Google Scholar] [CrossRef]
- Andersen, C.L.; Jensen, J.L.; Ørntoft, T.F. Normalization of real-time quantitative reverse transcription-PCR data: A model-based variance estimation approach to identify genes suited for normalization, applied to bladder and colon cancer data sets. Cancer Res. 2004, 64, 5245–5250. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vandesompele, J.; De Preter, K.; Pattyn, F.; Poppe, B.; Van Roy, N.; De Paepe, A.; Speleman, F. Accurate normalization of real-time quantitative RT-PCR data by geometric averaging of multiple internal control genes. Genome Biol. 2002, 3, 1–12. [Google Scholar] [CrossRef] [Green Version]
| Types of Mutations | ||||||
|---|---|---|---|---|---|---|
| Genetic Sex | Phenotypic Sex (Gonad) | Number of Clones Sequenced | Wide Deletion | Short Deletion | No Mutation | |
| wild-type | XY | ♂ (Testis) | 4 | 0 | 0 | 4 |
| XX | ♀ (Ovary) | 4 | 0 | 0 | 4 | |
| amhr2 mutant | XY | ♀ (Ovary) | 16 | 15 | 1 | 0 |
| XY | ♀ (Ovary) | 16 | 13 | 3 | 0 | |
| XY | ♀ (Ovary) | 16 | 15 | 1 | 0 | |
| XX | ♀ (Ovary) | 8 | 8 | 0 | 0 | |
| XX | ♀ (Ovary) | 8 | 6 | 2 | 0 | |
| Treatment | Genotype | Genetic Sex | ♂ (Testis) | ♀ (Ovary) |
|---|---|---|---|---|
| Control | wild-type | XY | 9 | 0 |
| Control | wild-type | XX | 0 | 11 |
| Control | amhr2 mutant | XY | 0 | 6 |
| Control | amhr2 mutant | XX | 0 | 4 |
| Fadrozole | amhr2 mutant | XY | 5 | 0 |
| Fadrozole | amhr2 mutant | XX | 5 | 0 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yamaguchi, T.; Kitano, T. Amh/Amhr2 Signaling Causes Masculinization by Inhibiting Estrogen Synthesis during Gonadal Sex Differentiation in Japanese Flounder (Paralichthys olivaceus). Int. J. Mol. Sci. 2023, 24, 2480. https://doi.org/10.3390/ijms24032480
Yamaguchi T, Kitano T. Amh/Amhr2 Signaling Causes Masculinization by Inhibiting Estrogen Synthesis during Gonadal Sex Differentiation in Japanese Flounder (Paralichthys olivaceus). International Journal of Molecular Sciences. 2023; 24(3):2480. https://doi.org/10.3390/ijms24032480
Chicago/Turabian StyleYamaguchi, Toshiya, and Takeshi Kitano. 2023. "Amh/Amhr2 Signaling Causes Masculinization by Inhibiting Estrogen Synthesis during Gonadal Sex Differentiation in Japanese Flounder (Paralichthys olivaceus)" International Journal of Molecular Sciences 24, no. 3: 2480. https://doi.org/10.3390/ijms24032480






