Photoremoval of Bisphenol A Using Hierarchical Zeolites and Diatom Biosilica
Abstract
:1. Introduction
- electrons in the conduction band initially coming from the valence band (eCB- electron in the conduction band). They act as very good reductants, e.g., reducing molecular oxygen to an anion radical O2.-. On continuing the reaction, it is possible to obtain hydroxyl radicals having one of the highest oxidation potentials;
- hole in the valence band (hVB+), formed when the electrons pair of eCB- disconnects and gain a positive charge. In the reaction, holes act as an oxidant, interacting with organic compounds undergoing degradation, leading to the final yield of mineralized products including CO2 and H2O. The oxidation process can also occur through the reactions with water, resulting in the formation of an electrophilic hydroxyl radical, which is involved in the oxidation of organic pollutants [13,15].
2. Results
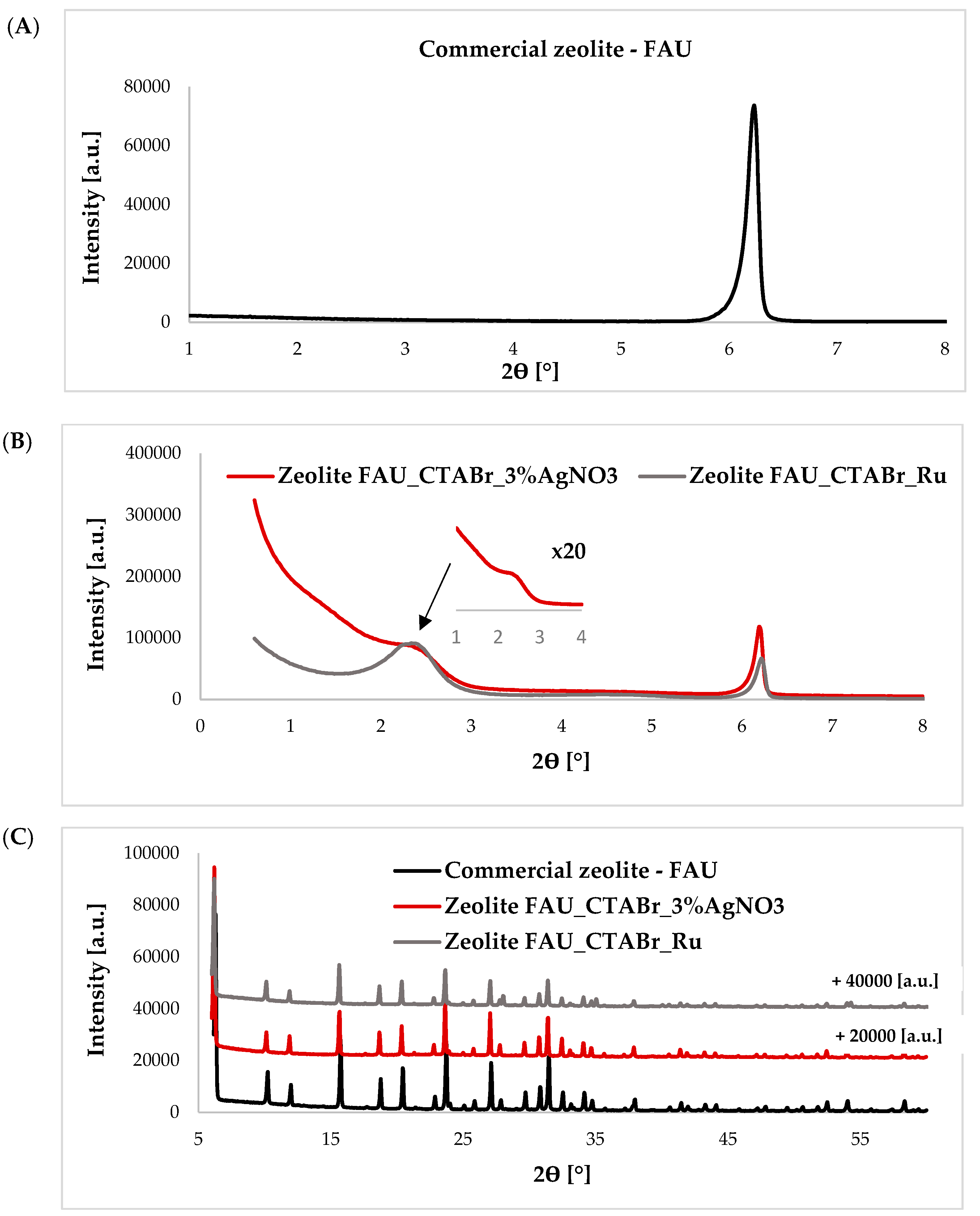
2.1. Characteristics of Materials
2.2. Photocatalytic Removal of Bisphenol A
2.2.1. Effect of Temperature and pH on the Photocatalytic Removal of Bisphenol A for Ruthenium Ion-Modified Diatom Biosilica
2.2.2. Qualitative Analysis of Bisphenol A removal Products by ESI/HPLC-MS Method
3. Materials and Methods
3.1. Synthesis of Hierarchical Zeolites Based on FAU-Type Commercial Zeolite Modified with Silver Ions
3.1.1. STAGE I
3.1.2. STAGE II
3.2. Synthesis of Hierarchical Zeolites Based on FAU-Type Commercial Zeolite Modified with Ruthenium Ions
3.3. Synthesis of Biosilica Modified with Silver Ions or Ruthenium Ions
3.4. Labelling of the Materials Studied
| Zeolite FAU_CTABr_3%AgNO3 | Hierarchical zeolite based on FAU-type commercial zeolite modified with 3 wt. % AgNO3 silver nitrate solution |
| Zeolite FAU_CTABr_Ru | Hierarchical zeolite based on FAU-type commercial zeolite modified with ruthenium |
| Ag Bio | Biosilica modified with silver ions |
| Ru Bio | Biosilica modified with ruthenium ions |
3.5. Characterization of the Materials
3.6. Photoremoval of Bisphenol A
3.7. Blank Tests and Leaching Tests
3.8. Analysis of Bisphenol A Removal Products Using ESI/HPLC-MS
4. Conclusions
- the efficiency of bisphenol A removal was significantly influenced by the modification of the obtained photocatalysts with ruthenium or silver ions, for the materials modified with ruthenium ions, an increase in bisphenol A removal was noted,
- the degree of bisphenol A removal was dependent on the electromagnetic wavelength used, it was the highest at the wavelengths corresponding to blue, green, and cyan light,
- the optimal temperature favorably affecting the removal of bisphenol A is 25 °C. As for the effect of pH values on the photocatalytic removal process, the best results were recorded for the samples whose pH values were 7 and 11,
- the best photocatalyst is ruthenium ion-modified biosilica, in whose presence up to 99% removal of bisphenol A was achieved, the highest degree of bisphenol A d removal was obtained at electromagnetic wavelengths ranging from 450 nm to 525 nm;
- the products of photocatalytic removal of bisphenol A are: 4-hydroxy acetophenone, 4-isopropyl phenol, and monohydroxylated bisphenol A.
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Li, J.; Wang, G. Airborne particulate endocrine disrupting compounds in China: Compositions, size distributions and seasonal variations of phthalate esters and bisphenol A. Atmos. Res. 2015, 154, 138–145. [Google Scholar] [CrossRef]
- Kaneco, S.; Rahman, M.A.; Suzuki, T.; Katsumata, H. Optimization of solar photocatalytic degradation conditions of bisphenol A in water using titanium dioxide. J. Photochem. Photobiol. A Chem. 2004, 163, 419–424. [Google Scholar] [CrossRef]
- Chiang, K.; Lim, T.M.; Tsen, L.; Lee, C. Photocatalytic degradation and mineralization of bisphenol A by TiO2 and platinized TiO2. Appl. Catal. A Gen. 2004, 261, 225–237. [Google Scholar] [CrossRef]
- Wang, R.; Ren, D.; Xia, S.; Zhang, Y.; Zhao, J. Photocatalytic degradation of bisphenol A (BPA) using immobilized TiO2 and UV illumination in a horizontal circulating bed photocatalytic reactor (HCBPR). J. Hazard. Mater. 2009, 169, 926–932. [Google Scholar] [CrossRef]
- Suliza, M.; Rahman, M.A.; Lau, W.J.; Yusop, Z. A review on bisphenol A occurrences, health effects and treatment process via membrane technology for drinking water. Environ. Sci. Pollut. Res. 2016, 23, 11549–11567. [Google Scholar]
- Vasiljevic, T.; Harner, T. Bisphenol A and its analogues in outdoor and indoor air: Properties, sources and global levels. Sci. Total Environ. 2021, 789, 148013–148019. [Google Scholar] [CrossRef] [PubMed]
- Maćczak, A.; Duchnowicz, P.; Sicińska, P.; Koter-Michalak, M.; Bukowska, B.; Michałowicz, J. The in vitro comparative study of the effect of BPA, BPS, BPF and BPAF on human erythrocyte membrane; perturbations in membrane fluidity, alterations in conformational state and damage to proteins, changes in ATP level and Na+/K+ ATPase and AChE activities. Food Chem. Toxicol. 2017, 110, 351–359. [Google Scholar] [CrossRef]
- Ma, Y.; Liu, H.; Wu, J.; Yuan, L.; Wang, Y.; Du, X.; Wang, R.; Marwa, P.W.; Petlulu, P.; Chen, X.; et al. The adverse health effects of bisphenol A and related toxicity mechanisms. Environ. Res. 2019, 176, 108575. [Google Scholar] [CrossRef]
- Colmenares, J.C.; Luque, R. Heterogeneous photocatalytic nanomaterials: Prospects and challenges in selective transformations of biomass-derived compounds. Chem. Soc. Rev. 2014, 43, 765–778. [Google Scholar] [CrossRef]
- Zare, E.N.; Iftekhar, S.; Park, Y.; Joseph, J.; Srivastava, V.; Khan, M.A.; Makvandi, P.; Sillanpaa, M.; Varma, R.S. An overwiev on non-special semiconductors for heterogeneous photocatalytic degradation of organic water contaminants. Chemosphere 2021, 280, 130907. [Google Scholar] [CrossRef]
- Wang, C.Y.; Zhang, X.; Song, X.N.; Wang, W.K.; Yu, H.Q. Novel Bi12O15Cl6 photocatalyst for the degradation of bisphenol A under visible-light irradiation. ACS Appl. Mater. Interfaces 2016, 8, 5320–5326. [Google Scholar] [CrossRef] [PubMed]
- Katsumata, H.; Taniguchi, M.; Kaneco, S.; Suzuki, T. Photocatalytic degradation of bisphenol A by Ag3PO4 under visible light. Catal. Commun. 2013, 34, 30–34. [Google Scholar] [CrossRef]
- Umar, M.; Aziz, H.A. Photocatalytic Degradation of Organic Pollutants in Water. In Organic Pollutants—Monitoring, Risk and Treatment; IntechOpen: London, UK, 2013. [Google Scholar]
- Sprynskyy, M.; Szczyglewska, P.; Wojtczak, I.; Nowak, I.; Witkowski, A.; Buszewski, B.; Feliczak-Guzik, A. Diatom biosilica dopes with palladium (II) chloride nanoparticles as new efficient photocatalysts for methyl orange degradation. Int. J. Mol. Sci. 2021, 22, 6734. [Google Scholar] [CrossRef] [PubMed]
- Nicholas, M.P.; Nolan, T.; Pillai, S.C.; Seery, M.K.; Falaras, P.; Kontos, A.G.; Dunlop, P.S.M.; Hamilton, J.W.J.; Byrne, J.A.; O’Shea, K.; et al. A review on the visible light active titanium dioxide photocatalysts for environmental applications. Appl. Catal. B Environ. 2012, 125, 331–349. [Google Scholar]
- Chen, D.; Cheng, Y.; Zhou, N.; Chen, P.; Wang, Y.; Li, K.; Huo, S.; Cheng, P.; Peng, P.; Zhang, R.; et al. Photocatalytic degradation of organic pollutants usiong TiO2-based photocatalysts: A review. J. Clean. Prod. 2020, 268, 121725. [Google Scholar] [CrossRef]
- Feliczak-Guzik, A.; Sprynskyy, M.; Nowak, I.; Buszewski, B. Synthesis and physicochemical properties of hierarchical zeolites containing ruthenium oxide nanoparticles and their application in the reaction of dihydroxyacetone isomerization. Microporous Mesoporous Mater. 2020, 293, 109787. [Google Scholar] [CrossRef]
- Darvishi Cheshmeh Soltani, R.; Khataee, A.R.; Safari, M.; Joo, S.W. Preparation of bio-silica/chitosan nanocomposite for adsorption of a textile dye in aqueous solutions. Int. Biodeterior. Biodegrad. 2013, 85, 383–391. [Google Scholar] [CrossRef]
- Dang, T.D.; Banerjee, A.N.; Cheney, M.A.; Qian, S.; Joo, S.W.; Min, B.K. Bio-silica coated with amorphous manganese oxide as an efficient catalyst for rapid degradation of organic pollutant. Colloids Surf. B Biointerfaces 2013, 106, 151–157. [Google Scholar] [CrossRef]
- Sprynskyy, M.; Pomastowski, P.; Hornowska, M.; Król, A.; Rafinska, K.; Buszewski, B. Naturally organic functionalized 3D biosilica from diatom microalgae. Mater. Des. 2017, 132, 22–29. [Google Scholar] [CrossRef]
- Mohamed, A.; Ghobara, M.M.; Abdelmaksoud, M.K.; Mohamed, G.G. A novel and highly efficient photocatalytic degradation of malachite green dye via surface modified polyacrylonitrile nanofivers/biogenic silica composite nanofibers. Sep. Purif. Technol. 2019, 210, 935–942. [Google Scholar] [CrossRef]
- Al-Azri, Z.H.N.; Chen, W.T.; Chan, A.; Jovic, V.; Ina, T.; Idriss, H.; Waterhouse, G.I.N. The roles of metal co-catalysts and reaction media in photocatalytic hydrogen production: Performance evaluation of M/TiO2 photocatalysts (M = Pd, Pt, Au) in different alcohol–water mixtures. J. Catal. 2015, 329, 355–367. [Google Scholar] [CrossRef]
- López-Tenllado, F.J.; Hidalgo-Carrillo, J.; Montes, V.; Marinas, A.; Urbano, F.J.; Marinas, J.M.; Ilieva, L.; Tabakova, T.; Reid, F. A comparative study of hydrogen photocatalytic production from glycerol and propan-2-ol on M/TiO2 systems (M=Au, Pt, Pd). Catal. Today 2017, 280, 58–64. [Google Scholar] [CrossRef]
- Pai, M.R.; Banerjee, A.M.; Rawool, S.A.; Singhal, A.; Nayak, C.; Ehrman, S.H.; Tripathi, A.K.; Bharadwaj, S.R. A comprehensive study on sunlight driven photocatalytic hydrogen generation using low cost nanocrystalline Cu-Ti oxides. Sol. Energy Mater. Sol. Cells 2016, 154, 104–120. [Google Scholar] [CrossRef]
- Jung, M.; Hart, J.; Boensch, D.; Scott, J.; Ng, Y.H.; Amal, R. Hydrogen evolution via glycerol photoreforming over Cu–Pt nanoalloys on TiO2. Appl. Catal. A Gen. 2016, 518, 221–230. [Google Scholar] [CrossRef]
- Ananyh, A.; Mok, Y.S. Dielectric Barrier Discharge (DBD) Plasma Assisted Synthesis of Ag2O Nanomaterials and Ag2O/RuO2 Nanocomposites. Nanomaterials 2016, 6, 42. [Google Scholar] [CrossRef]
- Alshahrani, H.; Pathinettampadia, G.; Gujba, A.K.; Prakash, A.; Rethnam, V. Effect of palmyra sprout fiber and biosilica on mechanical, wear, thermal and hydrophobic behavior of epoxy resin composite. J. Ind. Text. 2022, 52, 1–20. [Google Scholar] [CrossRef]
- Thommes, M.; Kaneko, K.; Neimark, A.V.; Olivier, J.P.; Rodriguez-Reinoso, F.; Rouquerol, J.; Sing, K.S.W. Physisorption og gases, with special reference to the evaluation of surface area and pore size distribution (IUPAC Technical Report). Pure Appl. Chem. 2015, 87, 1051–1069. [Google Scholar] [CrossRef]
- Thommes, M. Physical Adsorption Characterization of Nanoporous Materials. Chem. Ing. Tech. 2010, 82, 1059–1073. [Google Scholar] [CrossRef]
- Feliczak-Guzik, A.; Sprynskyy, M.; Nowak, I.; Jaroniec, M.; Buszewski, B. Application of novel hierarchical niobium containing zeolites for synthesis of alkyl lactate and lactic acid. J. Colloid Int. Sci. 2018, 516, 379–383. [Google Scholar] [CrossRef] [PubMed]
- Feliczak-Guzik, A.; Sprynskyy, M.; Nowak, I.; Buszewski, B. Isomerization of dihydroxyacetone to lactic acid and alkyl lactates over hierarchical zeolites containing tin. Catalysts 2018, 8, 31. [Google Scholar] [CrossRef]
- Kruk, M.; Jaroniec, M.; Sayari, A. Application of large pore MCM-41 molecular sieves to improve pore size analysis using nitrogen adsorption measurements. Langmuir 1997, 13, 6267–6273. [Google Scholar] [CrossRef]
- Fukahori, S.; Ichiura, H.; Kitaoka, T.; Tanaka, H. Capturing of bisphenol A photodecomposition intermediates by Composite TiO2-Zeolite Sheets. Appl. Catal. B Environ. 2003, 46, 453–462. [Google Scholar] [CrossRef]
- Guo, C.; Ge, M.; Liu, L.; Gao, G.; Feng, Y.; Wang, Y. Directed Synthesis of Mesoporous TiO2 Microspheres: Catalysts and Their Photocatalysis for Bisphenol A Degradation. Environ. Sci. Technol. 2010, 44, 419–425. [Google Scholar] [CrossRef] [PubMed]
- Tao, H.; Hao, S.; Chang, F.; Wang, L.; Zhang, Y.; Cai, X.; Zeng, J.S.D. Photodegradation of Bisphenol A by Titana Nanoparticles in Mesoporous MCM-41. Water Air Soil Pollut. 2011, 214, 491–498. [Google Scholar] [CrossRef]
- Chalasani, R.; Vasudevan, S. Cyclodextrin-Functionalized Fe3O4@TiO2: Reusable, Magnetic Nanoparticles for Photocatalytic Degradation of Endocrine Disrupting Chemicals in Water Supplies. ACS Nano 2013, 7, 4093–4104. [Google Scholar] [CrossRef]
- Bechambi, O.; Jlaiel, L.; Najjar, W.; Sayadi, S. Photocatalytic Degradation of Bisphenol A in the Presence of Ce-ZnO: Evolution of Kinetics, Toxicity and Photodegradation Mechanism. Mater. Chem. Phys. 2016, 173, 95–105. [Google Scholar] [CrossRef]
- Zhao, L.; Xiao, X.; Peng, L.; Gu, F.L.; Zhang, R.Q. Visible light Photocatalytic Mechanism of Bisphenol-A on NanoBi2O3: A Combined DFT Calculation and Experimental Study. RSC Adv. 2014, 4, 10343–10349. [Google Scholar] [CrossRef]
- Bhunia, S.K.; Jana, N.R. Reduced Graphene Oxide Silver Nanoparticle Composite as Visible Light Photocatalyst for Degradation of Colorless Endocrine Disruptors. ACS Appl. Mater. Interface 2014, 6, 20085–20092. [Google Scholar] [CrossRef]
- Ju, P.; Fan, H.; Ai, S.; Zhang, D.; Wang, Y. Photocatalytic Activity of One-Dimensional Ag2V4O11 Nanowires in the Degradation of Bisphenol A under Visible-Light Irradiation. Res. Chem. Intermed. 2015, 41, 3683–3697. [Google Scholar] [CrossRef]
- Hu, Z.T.; Liu, J.; Yan, X.; Oh, W.D.; Lim, T.T. Low Temperature Synthesis of Graphene/Bi2Fe4O9 Composite for Synergistic Adsorption-Photocatalytic Degradation of Hydrophobic Pollutant under Solar Irradiation. Chem. Eng. J. 2015, 262, 1022–1032. [Google Scholar] [CrossRef]
- Ding, Y.; Zhou, P.; Tang, H. Visible-light Photocatalytic Degradation of Bisphenol A on NaBiO3 Nanosheets in a wide pH Range: A Synergistic Effect between Photocatalytic Oxidation and Chemical Oxidation. Chem. Eng. J. 2016, 291, 149–160. [Google Scholar] [CrossRef]
- Garg, A.; Singhania, T.; Singh, A.; Sharma, S.; Rani, S.; Neogy, A.; Yadav, S.R.; Sangal, V.K.; Garg, N. Photocatalytic Degradation of Bisphenol-A using N, Co Codoped TiO2 Catalyst under Solar Light. Sci. Rep. 2019, 9, 765. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kanakaraju, D.; Jasni, M.A.A.; Pace, A.; Ya, M.H. Enhanced dye-removal performance of Cu-TiO2-fly ash composite by optimized adsorption and photocatalytic activity under visible light irradiation. Environ. Sci. Pollut. Res. 2021, 28, 68834–68845. [Google Scholar] [CrossRef] [PubMed]
- Torres, R.A.; Pétrier, C.; Combet, E.; Carrier, M.; Pulgarin, C. Ultrasonic cavitation applied to the treatment of bisphenol A. Effect of sonochemical parameters and analysis of BPA by-products. Ultrason. Sonochem. 2008, 4, 605–611. [Google Scholar] [CrossRef] [PubMed]







| Sample | Specific Surface Area [m2/g] | Pore Volume [cm3/g] | Average Pore Diameter [nm] | |
|---|---|---|---|---|
| Total | Microporous volume | |||
| Hierarchical zeolites | ||||
| Commercial zeolite—FAU | 718 | 0.37 | 0.34 | - |
| Zeolite FAU_CTABr_3%AgNO3 | 746 | 0.34 | 0.13 | 5.84 |
| Zeolite FAU_CTABr_Ru | 776 | 0.41 | 0.11 | 2.46 |
| Biosilica | ||||
| Biosilica | 30 | 0.43 | - | 3.93 |
| Ag Bio | 104 | 0.25 | - | 2.97 |
| Ru Bio | 39 | 0.27 | - | 4.40 |
| Light Color | Wavelength [nm] | Degree of Bisphenol A Removal [%] |
|---|---|---|
| Zeolite FAU_CTABr_3%AgNO3 | ||
| Green | 525 | 7.3 |
| Blue | 450 | 9.4 |
| UV | 395–405 | 8.3 |
| Yellow | 595 | 8.2 |
| Cyan | 500 | 16.1 |
| Red | 620–630 | 8.1 |
| Green + Blue + Cyan | 450–525 | 20.1 |
| Zeolite FAU_CTABr_Ru | ||
| Green | 525 | 0.2 |
| Blue | 450 | 1.6 |
| UV | 395–405 | 2.2 |
| Yellow | 595 | 6.0 |
| Cyan | 500 | 35.7 |
| Red | 620–630 | 0.0 |
| Green + Blue + Cyan | 450–525 | 1.5 |
| Ag Bio | ||
| Green | 525 | 28.7 |
| Blue | 450 | 8.3 |
| UV | 395–405 | 24.8 |
| Yellow | 595 | 37.2 |
| Cyan | 500 | 11.9 |
| Red | 620–630 | 31.6 |
| Green + Blue + Cyan | 450–525 | 15.1 |
| Ru Bio | ||
| Green | 525 | 70.5 |
| Blue | 450 | 99.6 |
| UV | 395–405 | 41.5 |
| Yellow | 595 | 43.0 |
| Cyan | 500 | 62.4 |
| Red | 620–630 | 34.1 |
| Green + Blue + Cyan | 450–525 | 68.1 |
| Light Color | Wavelength [nm] |
| UV | 395–405 |
| Blue | 450 |
| Cyan | 500 |
| Green | 525 |
| Yellow | 595 |
| Red | 620–630 |
| Green + Blue + Cyan | 450–525 |
| Time [min] | A [%] | B [%] |
|---|---|---|
| 0 | 90 | 10 |
| 1 | 90 | 10 |
| 30 | 30 | 70 |
| 32 | 30 | 70 |
| 34 | 90 | 10 |
| 39 | 90 | 10 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chudzińska, J.; Woźniak, B.; Sprynskyy, M.; Nowak, I.; Feliczak-Guzik, A. Photoremoval of Bisphenol A Using Hierarchical Zeolites and Diatom Biosilica. Int. J. Mol. Sci. 2023, 24, 2878. https://doi.org/10.3390/ijms24032878
Chudzińska J, Woźniak B, Sprynskyy M, Nowak I, Feliczak-Guzik A. Photoremoval of Bisphenol A Using Hierarchical Zeolites and Diatom Biosilica. International Journal of Molecular Sciences. 2023; 24(3):2878. https://doi.org/10.3390/ijms24032878
Chicago/Turabian StyleChudzińska, Jagoda, Bartosz Woźniak, Myroslav Sprynskyy, Izabela Nowak, and Agnieszka Feliczak-Guzik. 2023. "Photoremoval of Bisphenol A Using Hierarchical Zeolites and Diatom Biosilica" International Journal of Molecular Sciences 24, no. 3: 2878. https://doi.org/10.3390/ijms24032878








