Diarylethene-Based Ionic Liquids: Synthesis and Photo-Driven Solution Properties
Abstract
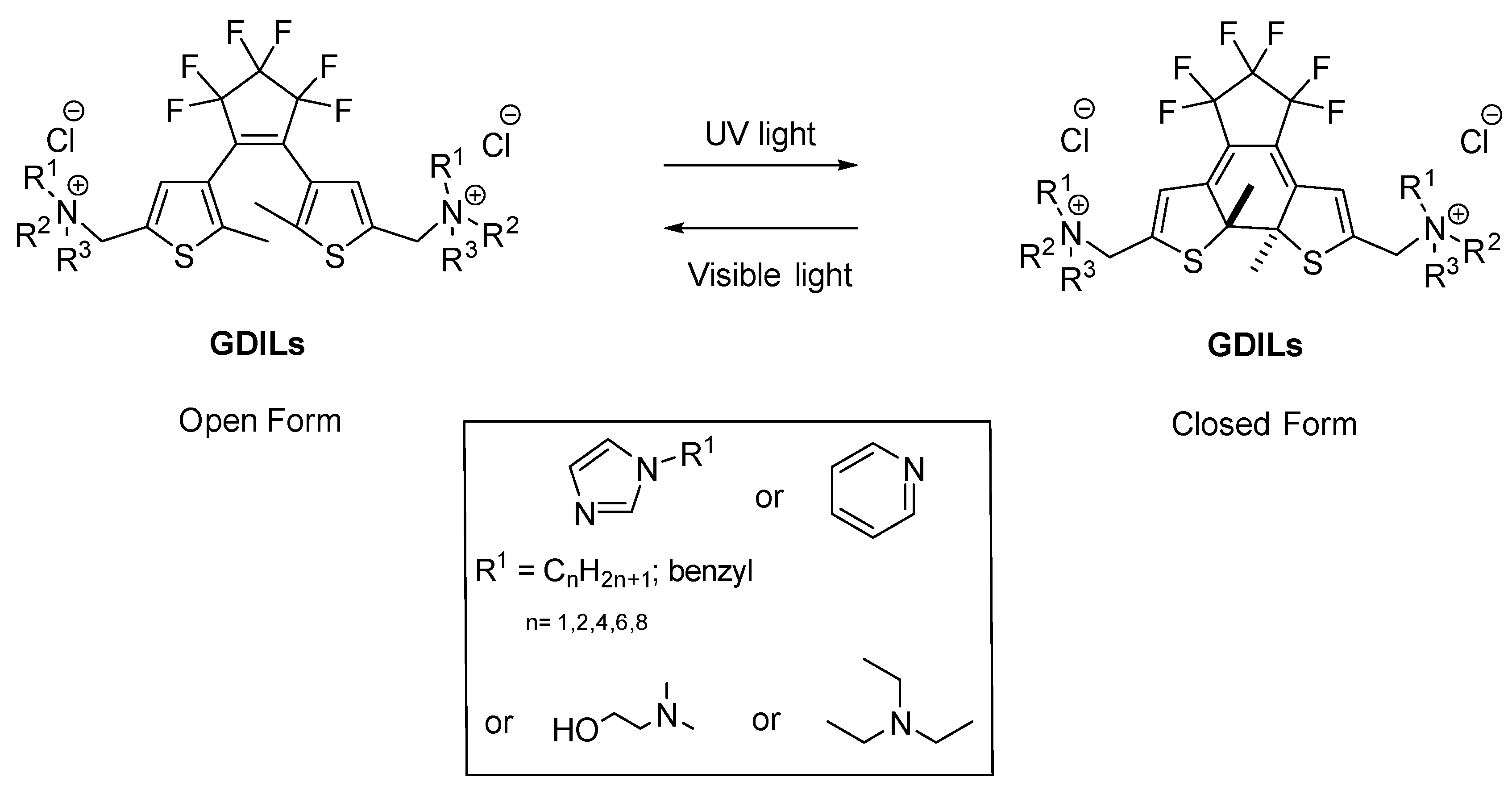
:1. Introduction
2. Results and Discussion
2.1. Influence of GDILs Photoconversion in the Ionic Conductivity in Solution
2.2. Influence of GDILs Photoconversion in the Viscosity of Common Ionic Liquids
2.3. Influence of GDILs Photoconversion in the Density of Common Ionic Liquids
3. Materials and Methods
3.1. Materials
3.2. Methods
Synthesis and Sample Characterization
2-Methylthiophene (1)
5-Methy-2-formylthiophene (2)
3-Bromo-2-methyl-5-formylthiophene (3)
3-Bromo-2-methyl-5-(hydroxymethyl)thiophene (4)
3-Bromo-2-methyl-5-(methoxy(tetrahydro-2H-pyran-2-yl)thiophene (5)
1,2-Bis{2-methyl-5-[2-methoxy(tetrahydro-2H-pyran]-3-thienyl}perfluorocyclopentene (6)
1,2-Bis{2-methyl-5-[2-hydroxymethyl]-3-thienyl}perfluorocyclopentene (7)
1,2-Bis{2-methyl-5-[2-chloromethyl]-3-thienyl}perfluorocyclopentene (8)
1,2-Bis{2-methyl-5-[2-(3-N-methylimidazolium)methyl]-3-thienyl}-perfluorocyclopentene dichloride (IMC1)
1,2-Bis{2-methyl-5-[2-(3-N-ethylimidazolium)methyl]-3-thienyl}-perfluorocyclopentene dichloride (IMC2)
1,2-Bis{2-methyl-5-[2-(3-N-butylimidazolium)methyl]-3-thienyl}-perfluorocyclopentene dichloride (IMC4)
1,2-Bis{2-methyl-5-[2-(3-N-hexylimidazolium)methyl]-3-thienyl}-perfluorocyclopentene dichloride (IMC6)
1,2-Bis{2-methyl-5-[2-(3-N-octylimidazolium)methyl]-3-thienyl}-perfluorocyclopentene dichloride (IMC8)
1,2-Bis{2-methyl-5-[2-(3-N-benzylimidazolium)methyl]-3-thienyl}-perfluorocyclopentene dichloride (IMBENZ)
1,2-Bis{2-methyl-5-[2-(pyridinium)methyl]-3-thienyl}perfluorocyclo-pentene dichloride (PY)
1,2-Bis{2-methyl-5-[2-(2-hydroxy ethyl)dimethylammonium)methyl]-3-thienyl}perfluorocyclopentene dichloride (CH)
1,2-Bis{2-methyl-5-[2-(triethylammonium)methyl]-3-thienyl}perfluoro-cyclopentene dichloride (ET3N)
1H-,13C- and 19F-NMR Spectra Were Obtained on an Ultrashield Bruker Avance II 400 Spectrometer: Chemical Shifts Are Reported in Parts per Million
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Stoll, R.S.; Hecht, S. Artificial Light-Gated Catalyst Systems. Angew. Chem. 2010, 49, 5054–5075. [Google Scholar] [CrossRef] [PubMed]
- Bouas-Laurent, H.; Dürr, H. Organic photochromism (IUPAC Technical Report). Pure Appl. Chem. 2001, 73, 639–665. [Google Scholar] [CrossRef]
- Gust, D.; Moore, T.A.; Moore, A.L. Molecular switches controlled by light. Chem. Commun. 2006, 11, 1169–1178. [Google Scholar] [CrossRef] [PubMed]
- Tian, H.; Feng, Y.J. Next step of photochromic switches? Mater. Chem. 2008, 18, 1617. [Google Scholar] [CrossRef]
- Hanazawa, M.; Sumiya, R.; Horikawa, Y.; Irie, M. Thermally irreversible photochromic systems. Reversible photocyclization of 1,2-bis (2-methylbenzo[b]thiophen-3-yl)perfluorocyclocoalkene derivatives. J. Chem. Soc. Chem. Commun. 1992, 206. [Google Scholar] [CrossRef]
- Peter, G.; Jean-Pierre, H. Photoreflective Materials and Their Application; Springer: Berlin/Heidelberg, Germany, 2002; Volume 2. [Google Scholar]
- Fissi, A.; Pieroni, O. Photoresponsive polymers. Photostimulated aggregation-disaggregation changes and photocontrol of solubility in azo-modified poly(glutamic acid). Macromolecules 1989, 22, 1115. [Google Scholar] [CrossRef]
- Daub, J.; Trieflinger, C.; Kushnir, O.; Procházka, R. Mimicking the Dye Processes of Sensory Photoreceptors. Mol. Cryst. Liq. Cryst. 2005, 430, 115–122. [Google Scholar] [CrossRef]
- Yao, B.; Wang, Y.; Menke, N.; Lei, M.; Zheng, Y.; Ren, L.; Chen, G.; Chen, Y.; Fan, M. Optical Properties and Applications of Photochromic Fulgides. Mol. Cryst. Liq. Cryst. 2005, 430, 211–219. [Google Scholar] [CrossRef]
- Asaka, T.; Akai, N.; Kawai, A.; Shibuya, K.J. Photochromism of 3-butyl-1-methyl-2-phenylazoimidazolium in room temperature ionic liquids. Photochem. Photobiol. A-Chem. 2010, 209, 12–18. [Google Scholar] [CrossRef]
- Toriumi, A.; Kawata, S.; Gu, M. Reflection confocal microscope readout system for three-dimensional photochromic optical data storage. Opt. Lett. 1998, 23, 1924. [Google Scholar] [CrossRef]
- Koçer, A.; Walko, M.; Feringa, B. A light-actuated nanovalve derived from a channel protein. Science 2005, 309, 755. [Google Scholar] [CrossRef]
- Myles, A.J.; Branda, N.R. 1,2-Dithienylethene Photochromes and Non-destructive Erasable Memory. Adv. Funct. Mater. 2002, 12, 167. [Google Scholar] [CrossRef]
- Bens, A.T.; Frewert, D.; Kodatis, K.; Kryschi, C.; Martin, H.-D.; Trommsdorff, H.P. Coupling of Chromophores: Carotenoids and Photoactive Diarylethenes—Photoreactivity versus Radiationless Deactivation. Eur. J. Org. Chem. 1998, 1998, 2333. [Google Scholar] [CrossRef]
- Kawai, T.; Koshido, T.; Yoshino, K. Optical and dielectric properties of photochromic dye in amorphous state and its application. Appl. Phys. Lett. 1995, 67, 795. [Google Scholar] [CrossRef]
- Shishido, A.; Tsutsumi, O.; Kanazawa, A.; Shiono, T.; Ikeda, T.; Tamai, N.J. Distinct Photochemical Phase Transition Behavior of Azobenzene Liquid Crystals Evaluated by Reflection-Mode Analysis. Phys.Chem. B 1997, 101, 2806. [Google Scholar] [CrossRef]
- Kinoshita, K.; Horie, K.; Morino, S.; Nishikubo, T. Large photoinduced refractive index changes of a polymer containing photochromic norbornadiene groups. Appl. Phys. Lett. 1997, 70, 2940. [Google Scholar] [CrossRef]
- Sasaki, K.; Nagamura, T. Ultrafast all-optical switch using complex refractive index changes of thin films containing photochromic dye. Appl. Phys. Lett. 1997, 71, 434. [Google Scholar] [CrossRef]
- Biteau, J.; Chaput, F.; Lahlil, K.; Boilot, J.-P.; Tsivgoulis, G.M.; Lehn, J.-M.; Darracq, B.; Marois, C.; Lévy, Y. Large and Stable Refractive Index Change in Photochromic Hybrid Materials. Chem. Mater. 1998, 10, 1945. [Google Scholar] [CrossRef]
- De Jong, J.; Lucas, L.N.; Kellogg, R.M.; Eschand, J.; Feringa, B.L. Photochromic Heterocycle-Fused Thieno[3,2-b]phosphole Oxides as Visible Light Switches without Sacrificing Photoswitching Efficiency. Science 2004, 304, 278. [Google Scholar] [CrossRef] [PubMed]
- Woodward, R.B.; Hoffmann, R.J. Stereochemistry of Electrocyclic Reactions. Am. Chem. Soc. 1965, 87, 395–397. [Google Scholar] [CrossRef]
- Joule, J.A.; Mills, K. Heterocyclic Chemistry, 5th ed.; John Wiley and Sons Ltd.: Hoboken, NJ, USA, 2010. [Google Scholar]
- Barbarella, G.; Melucci, M.; Sotgiu, G. The Versatile Thiophene: An Overview of Recent Research on Thiophene-Based Materials. Adv. Mater. 2005, 17, 1581–1593. [Google Scholar] [CrossRef]
- King, W.J.; Nord, F.F. Studies in the Thiophene Series. V. Wolff-Kishner Reductions. J. Org. Chem. 1949, 14, 638–642. [Google Scholar] [CrossRef]
- Halik, M.; Klauk, H.; Zschieschang, U.; Schmid, G.; Ponomarenko, S.; Kirchmeyer, S.; Weber, W. Relationship Between Molecular Structure and Electrical Performance of Oligothiophene Organic Thin Film Transistors. Adv. Mater. 2003, 15, 917–922. [Google Scholar] [CrossRef]
- Rost, C.; Karg, S.; Riess, W.; Loi, M.A.; Murgia, M.; Muccini, M. Ambipolar light-emitting organic field-effect transistor. Appl. Phys. Lett. 2004, 85, 1613–1615. [Google Scholar] [CrossRef]
- Vriezema, D.M.; Hoogboom, J.; Velonia, K.; Takazawa, K.; Christianen, P.M.; Maan, J.C.; Rowan, A.E.; Nolte, R.J.M. Vesicles and Polymerized Vesicles from Thiophene-Containing Rod–Coil Block Copolymers. Angew. Chem. Int. Ed. 2003, 42, 772–776. [Google Scholar] [CrossRef]
- Yu, H.H.; Pullen, A.E.; Büschel, M.G.; Swager, T.M. Charge-specific interactions in segmented conducting polymers: An approach to selective ionoresistive responses. Angew. Chem. Int. Ed. 2004, 43, 3700–3703. [Google Scholar] [CrossRef]
- Clot, O.; Wolf, M.O.; Patrick, B.O. Electropolymerization of Pd (II) complexes containing phosphinoterthiophene ligands. J. Am. Chem. Soc. 2001, 123, 9963–9973. [Google Scholar] [CrossRef]
- Tong, Z.; Pu, S.; Xiao, Q.; Lui, G.; Cui, S. Synthesis and photochromism of a novel water-soluble diarylethene with glucosyltriazolyl groups. Tetrahedron Lett. 2013, 54, 474–477. [Google Scholar] [CrossRef]
- Hirose, T.; Matsuda, K.; Irie, M.J. Self-assembly of photochromic diarylethenes with amphiphilic side chains: Reversible thermal and photochemical control. Org. Chem. 2006, 71, 7499–7508. [Google Scholar] [CrossRef]
- Esperança, J.M.S.S.; Tariq, M.; Pereiro, A.B.; Araújo, J.M.M.; Seddon, K.R.; Rebelo, L.P.N. Anomalous and not-so-common behavior in common ionic liquids and ionic liquid-containing systems. Front. Chem. 2019, 7, 450. [Google Scholar] [CrossRef] [Green Version]
- Earle, M.J.; Esperança, J.M.S.S.; Gilea, M.A.; Lopes, J.N.C.; Rebelo, L.P.N.; Magee, J.W.; Seddon, K.R.; Widegren, J.A. The distillation and volatility of ionic liquids. Nature 2006, 439, 831–834. [Google Scholar] [CrossRef]
- Plechkova, N.V.; Seddon, K.R. Applications of ionic liquids in the chemical industry. Chem. Soc. Rev. 2008, 37, 123–150. [Google Scholar] [CrossRef]
- Welton, T. Solvents and sustainable chemistry. Proc. R. Soc. A 2015, 471, 20150502. [Google Scholar] [CrossRef]
- Singh, S.K.; Savoy, A.W. Ionic liquids synthesis and applications: An overview. J. Mol. Liq. 2020, 297, 112038. [Google Scholar] [CrossRef]
- Eshetu, G.G.; Armand, M.; Ohno, H.; Scrosati, B.; Passerini, S. Ionic liquids as tailored media for the synthesis and processing of energy conversion materials. Energy Environ. Sci. 2016, 9, 49–61. [Google Scholar] [CrossRef]
- Huang, W.; Wu, X.; Qi, J.; Zhu, Q.; Wu, W.; Lu, Y.; Chen, Z. Ionic liquids: Green and tailor-made solvents in drug delivery. Drug Discov. Today 2020, 25, 901–908. [Google Scholar] [CrossRef]
- Pardo, R.; Zayat, M.; Levy, D. Photochromic organic–inorganic hybrid materials. Chem. Soc. Rev. 2011, 40, 672–687. [Google Scholar] [CrossRef]
- Cusido, J.; Deniz, E.; Raymo, F.M. Fluorescent switches based on photochromic compounds. Eur. J. Org. Chem. 2009, 2009, 2031–2045. [Google Scholar] [CrossRef]
- Pina, F.; Lima, J.C.; Parola, A.J.; Afonso, C.A.M. Thermal and photochemical properties of 4’,7-dihydroxyflavylium in water-ionic liquid biphasic systems: A write-read-erase molecular switch. Angew. Chem. 2004, 43, 1525–1527. [Google Scholar] [CrossRef]
- Branco, L.C.; Pina, F. Intrinsically photochromic ionic liquids. Chem. Commun. 2009, 6204–6206. [Google Scholar] [CrossRef]
- Soromenho, M.R.C.; Siopa, F.; Afonso, C.A.M.; Esperança, J.M.S.S. Enhanced photoconversion of 1, 2-bis (2-methyl-1-benzothiophene-3-yl) perfluorocyclopentene in ionic liquid solutions. J. Photochem. Photobiol. A Chem. 2018, 358, 44–50. [Google Scholar] [CrossRef]
- Coleman, S.; Byrne, R.; Alhshimy, N.; Fraser, K.J.; MacFarlane, D.R.; Diamond, D. Photochromic imidazolium based ionic liquids based on spiropyran. Phys. Chem. Chem. Phys. 2010, 12, 7009–7017. [Google Scholar] [CrossRef] [PubMed]
- Pu, S.; Zhu, S.; Rao, Y.; Liu, G.; Wei, H.J. Photochromism of 1, 2-bis (2-n-alkyl-5-formyl-3-thienyl) perfluorocyclopentene derivatives. Mol. Struct. 2009, 921, 89–100. [Google Scholar] [CrossRef]
- Oliveira, F.S.; Pereiro, A.B.; Araújo, J.M.M.; Bernardes, C.E.S.; Lopes, J.N.C.; Todorovica, S.; Feioc, G.; Almeida, P.L.; Rebelo, L.P.N.; Marrucho, I.M. High ionicity ionic liquids (HIILs): Comparing the effect of ethylsulfonate and ethylsulfate anions. Phys. Chem. Chem. Phys. 2013, 15, 18138. [Google Scholar] [CrossRef] [PubMed]
- Tariq, M.; Carvalho, P.J.; Coutinho, J.A.P.; Marrucho, I.M.; Lopes, J.N.C.; Rebelo, L.P.N. Viscosity of (C2–C14) 1-alkyl-3-methylimidazolium bis (trifluoromethylsulfonyl) amide ionic liquids in an extended temperature range. Fluid Phase Equilibria 2011, 301, 22–32. [Google Scholar] [CrossRef]
- Paredes, X.; Fandino, O.; Comunas, M.; Pensado, A.; Fernández, J.J. Study of the effects of pressure on the viscosity and density of diisodecyl phthalate. Chem. Thermodyn. 2009, 41, 1007–1015. [Google Scholar] [CrossRef]
- Carvalho, P.; Regueira, T.; Santos, L.; Fernandez, J.; Coutinho, J. Effect of water on the viscosities and densities of 1-butyl-3-methylimidazolium dicyanamide and 1-butyl-3-methylimidazolium tricyanomethane at atmospheric pressure. J. Chem. Eng. Data 2010, 55, 645–652. [Google Scholar] [CrossRef]
- Tariq, M.; Forte, P.A.S.; Gomes, M.F.C.; Lopes, J.N.C.; Rebelo, L.P.N. Densities and refractive indices of imidazolium-and phosphonium-based ionic liquids: Effect of temperature, alkyl chain length, and anion. J. Chem. Thermodyn. 2009, 41, 790–798. [Google Scholar] [CrossRef]
- Lokshin, V.; Khodorkovsky, V. Stereochemistry in control of photochemical reactivity: 2, 6-Diaryl-4H-spiro [cyclohexane-1, 2′-indene]-1′, 3′, 4-triones. Tetrahedron 2018, 74, 418–424. [Google Scholar] [CrossRef]
- Sonawane, H.R.; Nanjundiah, B.S.; Purohit, P.C. Stereochemical control on the photochemistry of α-methoxy-β, γ-cyclopropyl cyclic ketones: Reactions of epimeric 3-methoxy-4-caranones. Tetrahedron Lett. 1983, 24, 3917–3918. [Google Scholar] [CrossRef]
- Dangerfield, E.M.; Stocker, B.L.; Peter, T.; Northcote, P.T.; Harvey, J.E. Stereochemical control in carbohydrate chemistry. J. Chem. Educ. 2008, 85, 689. [Google Scholar] [CrossRef]
- Martins, I.C.B.; Gomes, J.; Duarte, M.; Mafra, L. Understanding polymorphic control of pharmaceuticals using imidazolium-based ionic liquid mixtures as crystallization directing agents. Cryst. Growth Des. 2017, 17, 428–432. [Google Scholar] [CrossRef]
- Kulkarni, S.A.; Meekes, H.; Horst, J.H. Polymorphism Control through a Single Nucleation Event. Cryst. Growth Des. 2014, 14, 1493–1499. [Google Scholar] [CrossRef]
- Barton, B.; Caira, M.R.; Jager, L.; Hosten, E.C. N,N′-Bis(9-phenyl-9-thioxanthenyl)ethylenediamine: Highly Selective Host Behavior in the Presence of Xylene and Ethylbenzene Guest Mixtures. Cryst. Growth Des. 2017, 17, 6660–6667. [Google Scholar] [CrossRef]
- Hariharan, P.S.; Mariyatra, M.B.; Mothi, E.M.; Neels, A.; Rosair, G.; Anthony, S.P. Polymorphism and benzene solvent controlled stimuli responsive reversible fluorescence switching in triphenylphosphoniumfluorenylide crystals. New J. Chem. 2017, 41, 4592–4598. [Google Scholar] [CrossRef]
- Uchida, K.; Yamanoi, Y.; Yonezawa, T.; Nishihara, H. Reversible on/off conductance switching of single diarylethene immobilized on a silicon surface. J. Am. Chem. Soc. 2011, 133, 9239–9241. [Google Scholar] [CrossRef]
- Allen, N.S. Photochemistry and Photophysics of Polymer Materials, 1st ed.; John Wiley & Sons, Inc: Hoboken, NJ, USA, 2010. [Google Scholar]
- Cambié, D.; Bottecchia, C.; Straathof, N.J.W.; Hessel, V.; Noël, T. Applications of continuous-flow photochemistry in organic synthesis, material science, and water treatment. Chem. Rev. 2016, 116, 10276–10341. [Google Scholar] [CrossRef] [Green Version]













| Acronym | mass% | mol% | [C]/M |
|---|---|---|---|
| IMC1 | 5.91 | 1.69 × 10−1 | 9.39 × 10−2 |
| IMC2 | 4.62 | 1.26 × 10−1 | 7.03 × 10−2 |
| IMC4 | 2.82 | 7.10 × 10−2 | 3.95 × 10−2 |
| IMC6 | 1.76 | 4.12 × 10−2 | 2.29 × 10−2 |
| IMC8 | 0.91 | 1.99 × 10−2 | 1.10 × 10−2 |
| IMBENZ | 2.82 | 6.50 × 10−2 | 3.61 × 10−2 |
| PY | 0.02 | 6.73 × 10−4 | 3.74 × 10−4 |
| CH | 31.72 | 8.87 × 10−1 | 4.93 × 10−1 |
| ET3N | 13.58 | 3.66 × 10−1 | 2.03 × 10−1 |
| Acronym | mass% | mol% | [C]/M |
|---|---|---|---|
| IMC1 | 6.00 | 1.61 | 1.06 × 10−1 |
| IMC2 | 4.57 | 1.18 | 7.76 × 10−2 |
| IMC4 | 4.26 | 1.01 | 6.67 × 10−2 |
| IMC6 | 2.35 | 0.52 | 3.40 × 10−2 |
| IMC8 | 2.07 | 0.42 | 2.79 × 10−2 |
| IMBENZ | 2.63 | 0.57 | 3.76 × 10−2 |
| PY | 0.23 | 0.04 | 4.11 × 10−3 |
| CH | 7.00 | 1.84 | 1.21 × 10−1 |
| ET3N | 4.26 | 1.08 | 7.31 × 10−2 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Soromenho, M.R.C.; Afonso, C.A.M.; Esperança, J.M.S.S. Diarylethene-Based Ionic Liquids: Synthesis and Photo-Driven Solution Properties. Int. J. Mol. Sci. 2023, 24, 3533. https://doi.org/10.3390/ijms24043533
Soromenho MRC, Afonso CAM, Esperança JMSS. Diarylethene-Based Ionic Liquids: Synthesis and Photo-Driven Solution Properties. International Journal of Molecular Sciences. 2023; 24(4):3533. https://doi.org/10.3390/ijms24043533
Chicago/Turabian StyleSoromenho, Mário R. C., Carlos A. M. Afonso, and José M. S. S. Esperança. 2023. "Diarylethene-Based Ionic Liquids: Synthesis and Photo-Driven Solution Properties" International Journal of Molecular Sciences 24, no. 4: 3533. https://doi.org/10.3390/ijms24043533






