Histone Deacetylases (HDACs): Promising Biomarkers and Potential Therapeutic Targets in Thymic Epithelial Tumors
Abstract
:1. Introduction
2. Results
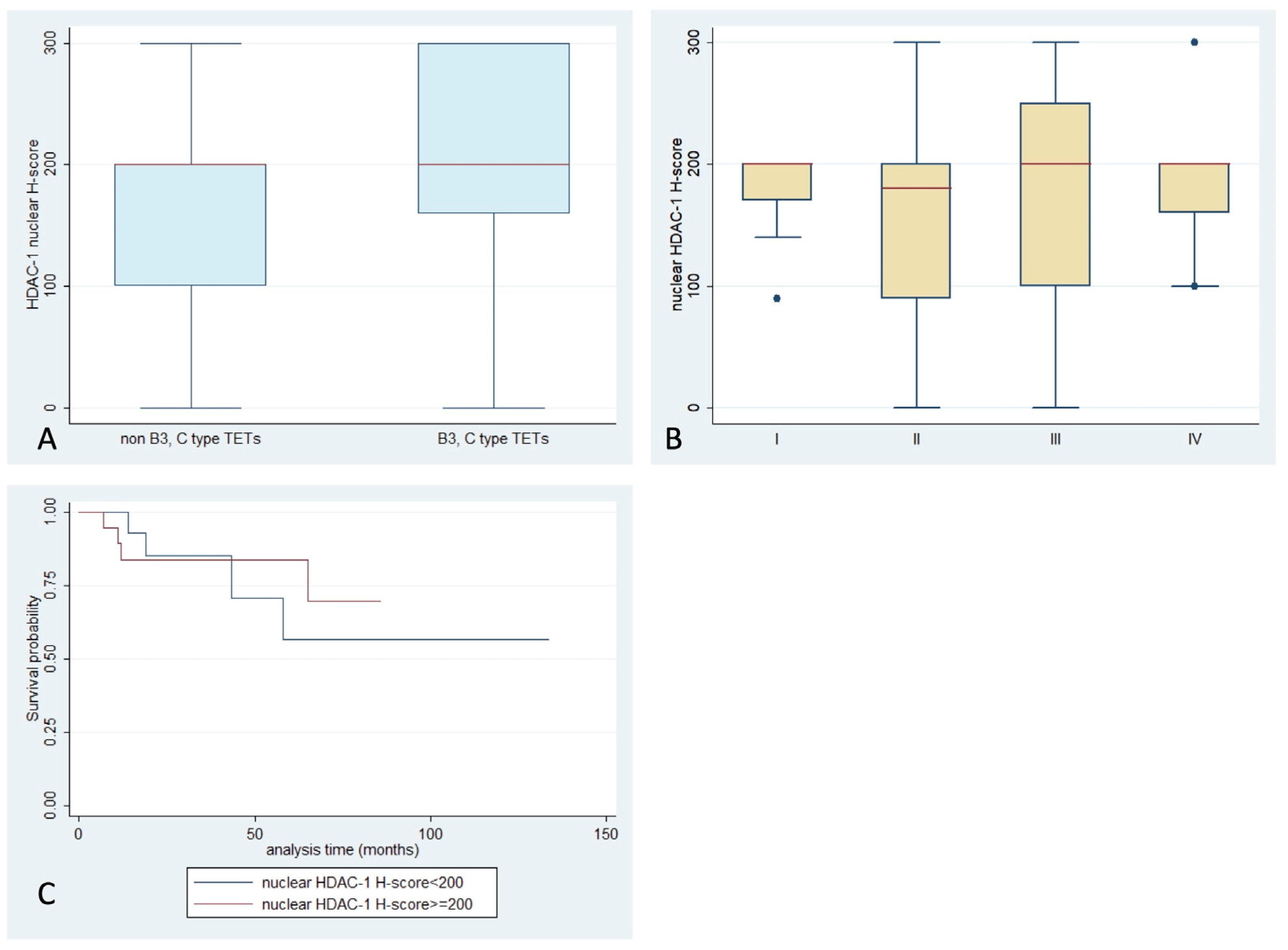
2.1. Expression of HDAC1 in TETs and Associations with Clinicopathological Characteristics
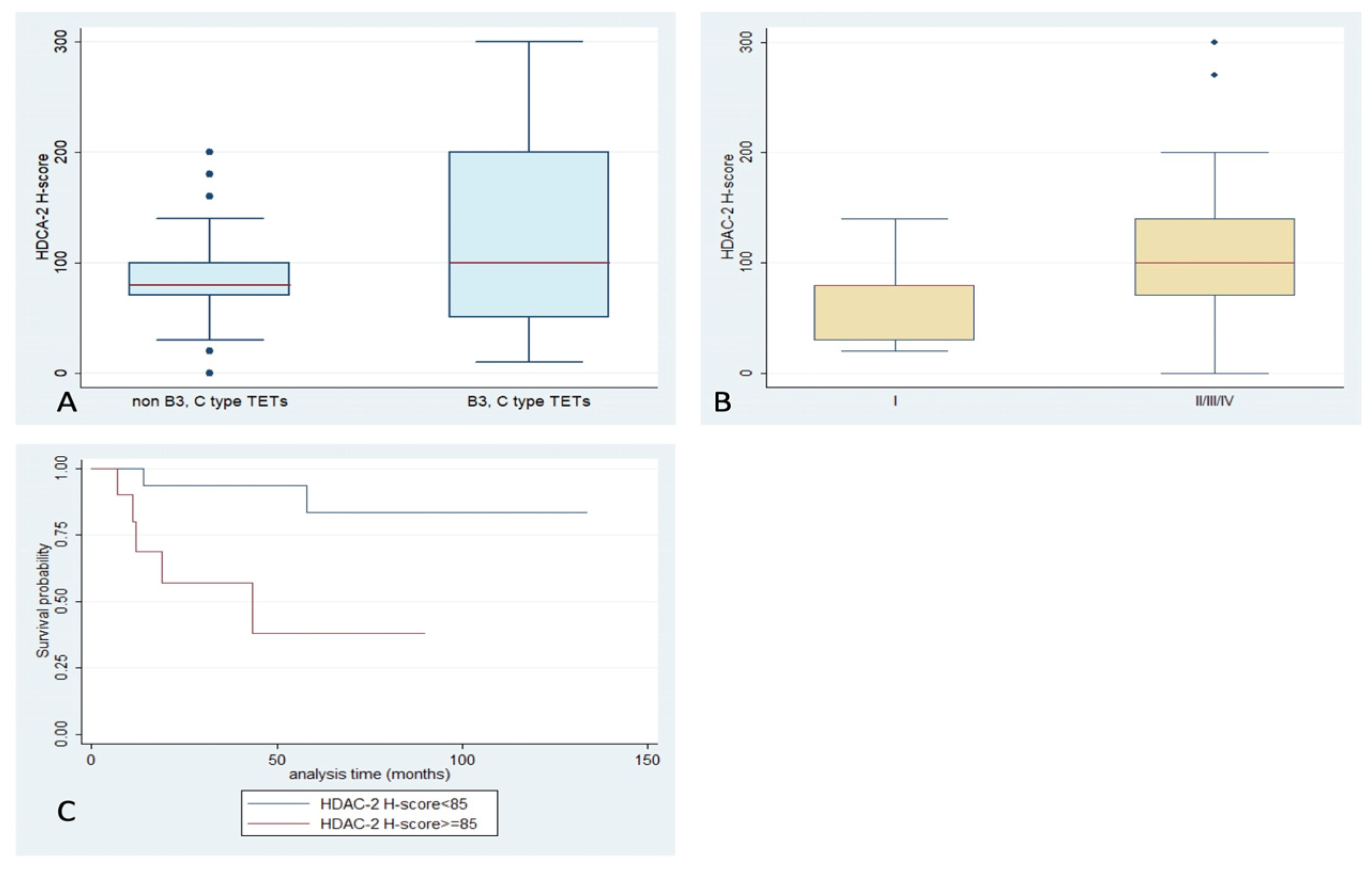
2.2. Expression of HDAC2 in TETs and Associations with Clinicopathological Characteristics
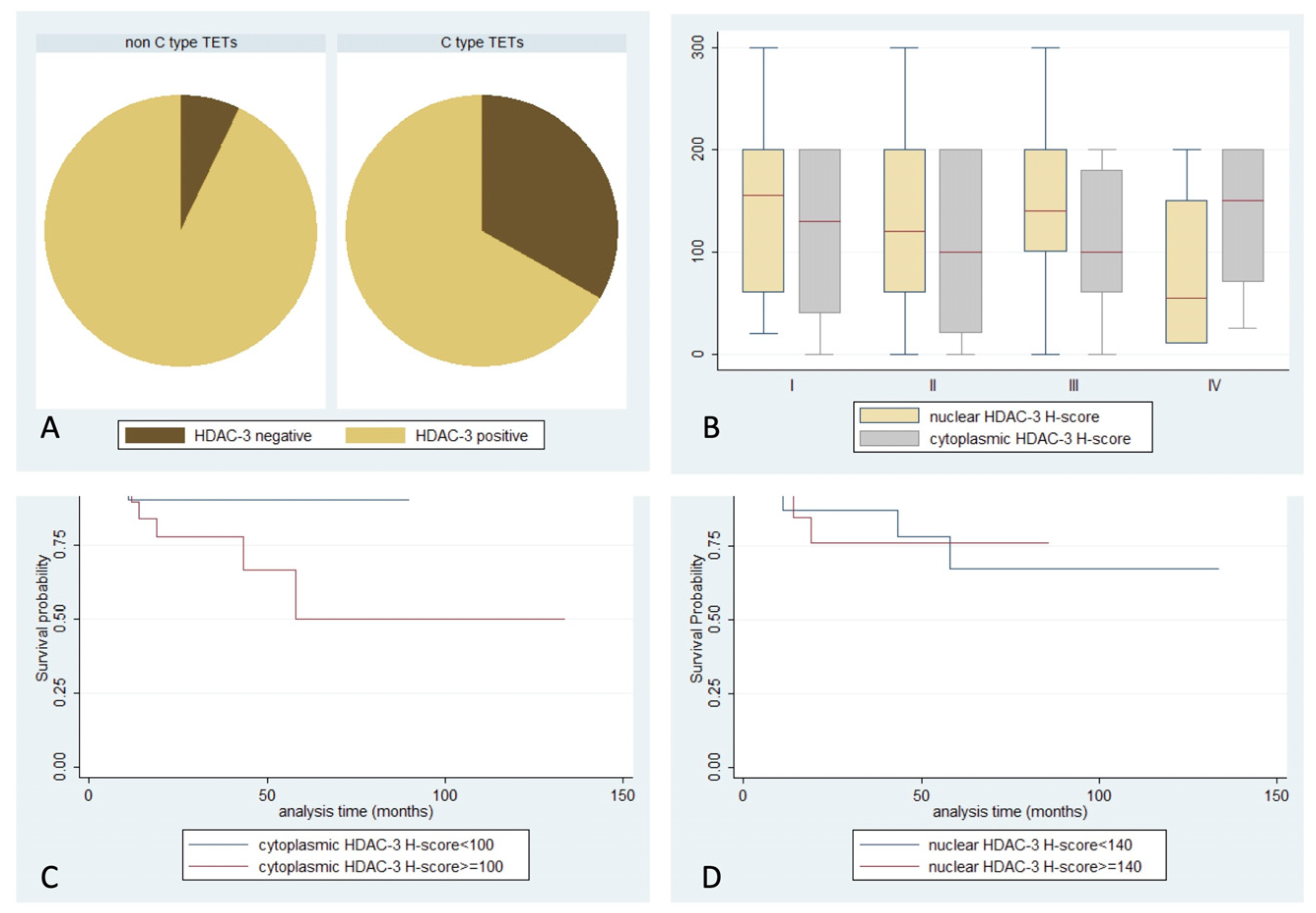
2.3. Expression of HDAC3 in TETs and Associations with Clinicopathological Characteristics
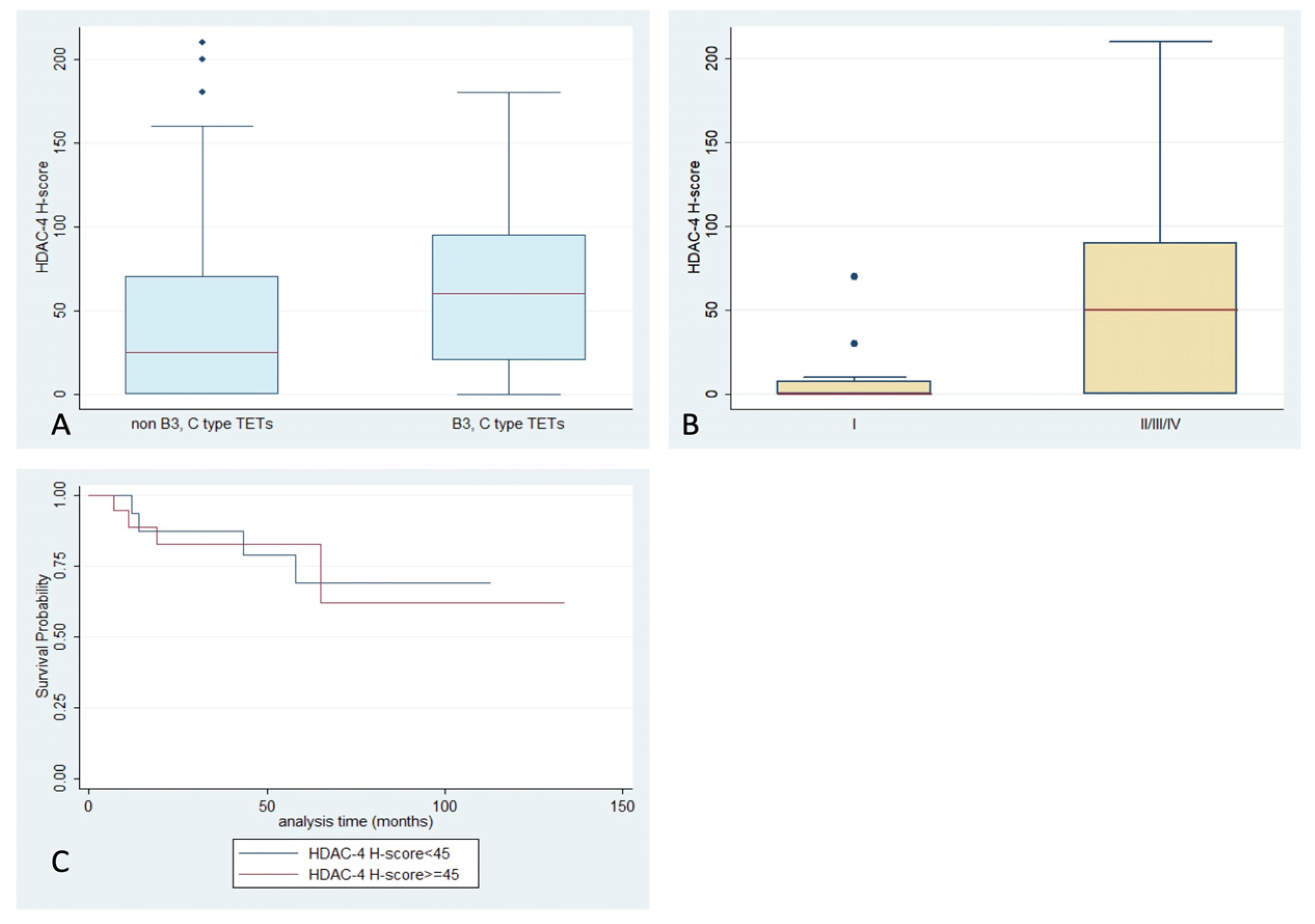
2.4. Expression of HDAC4 in TETs and Associations with Clinicopathological Characteristics
2.5. Expression of HDAC5 in TETs and Associations with Clinicopathological Characteristics
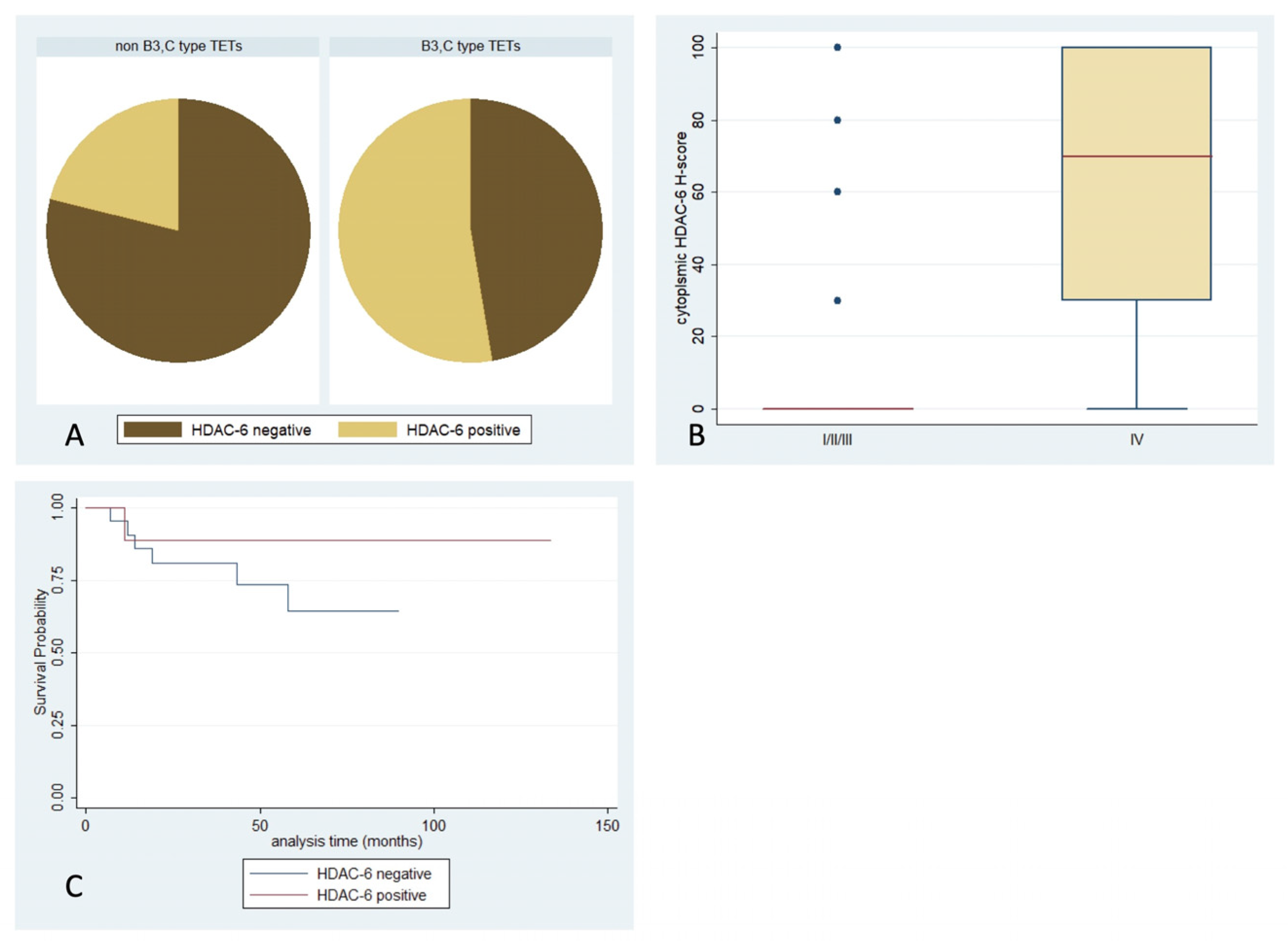
2.6. Expression of HDAC6 in TETs and Associations with Clinicopathological Characteristics
3. Discussion
4. Materials and Methods
4.1. Patients’ Characteristics
4.2. Immunohistochemistry
4.3. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Marx, A.; Ströbel, P.; Badve, S.S.; Chalabreysse, L.; Chan, J.K.C.; Chen, G.; de Leval, L.; Detterbeck, F.; Girard, N.; Huang, J.; et al. ITMIG Consensus Statement on the Use of the WHO Histological Classification of Thymoma and Thymic Carcinoma: Refined Definitions, Histological Criteria, and Reporting. J. Thorac. Oncol. 2014, 9, 596–611. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Radovich, M.; Pickering, C.R.; Felau, I.; Ha, G.; Zhang, H.; Jo, H.; Hoadley, K.A.; Anur, P.; Zhang, J.; McLellan, M.; et al. The Integrated Genomic Landscape of Thymic Epithelial Tumors. Cancer Cell 2018, 33, 244–258.e10. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, H.S.; Jang, H.J.; Shah, R.; Yoon, D.; Hamaji, M.; Wald, O.; Lee, J.S.; Sugarbaker, D.J.; Burt, B.M. Genomic Analysis of Thymic Epithelial Tumors Identifies Novel Subtypes Associated with Distinct Clinical Features. Clin. Cancer Res. 2017, 23, 4855–4864. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Psilopatis, I.; Pergaris, A.; Vrettou, K.; Theocharis, S.; Troungos, C. Thymic Epithelial Neoplasms: Focusing on the Epigenetic Alterations. Int. J. Mol. Sci. 2022, 23, 4045. [Google Scholar] [CrossRef] [PubMed]
- Ganci, F.; Vico, C.; Korita, E.; Sacconi, A.; Gallo, E.; Mori, F.; Cambria, A.; Russo, E.; Anile, M.; Vitolo, D.; et al. MicroRNA Expression Profiling of Thymic Epithelial Tumors. Lung Cancer 2014, 85, 197–204. [Google Scholar] [CrossRef] [PubMed]
- Wei, J.; Liu, Z.; Wu, K.; Yang, D.; He, Y.; Chen, G.G.; Zhang, J.; Lin, J. Identification of Prognostic and Subtype-Specific Potential MiRNAs in Thymoma. Epigenomics 2017, 9, 647–657. [Google Scholar] [CrossRef]
- Ji, G.; Ren, R.; Fang, X. Identification and Characterization of Non-Coding RNAs in Thymoma. Med. Sci. Monit. 2021, 27, e929727-1. [Google Scholar] [CrossRef]
- Su, Y.; Chen, Y.; Tian, Z.; Lu, C.; Chen, L.; Ma, X. LncRNAs Classifier to Accurately Predict the Recurrence of Thymic Epithelial Tumors. Thorac. Cancer 2020, 11, 1773–1783. [Google Scholar] [CrossRef]
- Chen, C.; Yin, B.; Wei, Q.; Li, D.; Hu, J.; Yu, F.; Lu, Q. Aberrant DNA Methylation in Thymic Epithelial Tumors. Cancer Investig. 2009, 27, 582–591. [Google Scholar] [CrossRef]
- Suzuki, M.; Chen, H.; Shigematsu, H.; Ando, S.; Iida, T.; Nakajima, T.; Fujisawa, T.; Kimura, H. Aberrant Methylation: Common in Thymic Carcinomas, Rare in Thymomas. Oncol. Rep. 2005, 14, 1621–1624. [Google Scholar] [CrossRef]
- Hirose, Y.; Kondo, K.; Takizawa, H.; Nagao, T.; Nakagawa, Y.; Fujino, H.; Toba, H.; Kenzaki, K.; Sakiyama, S.; Tangoku, A. Aberrant Methylation of Tumour-Related Genes in Thymic Epithelial Tumours. Lung Cancer 2009, 64, 155–159. [Google Scholar] [CrossRef]
- Shvedunova, M.; Akhtar, A. Modulation of Cellular Processes by Histone and Non-Histone Protein Acetylation. Nat. Rev. Mol. Cell Biol. 2022, 23, 329–349. [Google Scholar] [CrossRef]
- Marmorstein, R.; Zhou, M.M. Writers and Readers of Histone Acetylation: Structure, Mechanism, and Inhibition. Cold Spring Harb Perspect Biol. 2014, 6, a018762. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Seto, E.; Yoshida, M. Erasers of Histone Acetylation: The Histone Deacetylase Enzymes. Cold Spring Harb. Perspect. Biol. 2014, 6, a018713. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ropero, S.; Esteller, M. The Role of Histone Deacetylases (HDACs) in Human Cancer. Mol. Oncol. 2007, 1, 19–25. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Goutas, D.; Theocharis, S.; Tsourouflis, G. Unraveling the Epigenetic Role and Clinical Impact of Histone Deacetylases in Neoplasia. Diagnostics 2021, 11, 1346. [Google Scholar] [CrossRef]
- Theocharis, S.; Klijanienko, J.; Giaginis, C.; Rodriguez, J.; Jouffroy, T.; Girod, A.; Alexandrou, P.; Sastre-Garau, X. Histone Deacetylase-1 and -2 Expression in Mobile Tongue Squamous Cell Carcinoma: Associations with Clinicopathological Parameters and Patients Survival. J. Oral Pathol. Med. 2011, 40, 706–714. [Google Scholar] [CrossRef]
- Giaginis, C.; Damaskos, C.; Koutsounas, I.; Zizi-Serbetzoglou, A.; Tsoukalas, N.; Patsouris, E.; Kouraklis, G.; Theocharis, S. Histone Deacetylase (HDAC)-1, -2, -4 and -6 Expression in Human Pancreatic Adenocarcinoma: Associations with Clinicopathological Parameters, Tumor Proliferative Capacity and Patients’ Survival. BMC Gastroenterol. 2015, 15, 148. [Google Scholar] [CrossRef] [Green Version]
- Fritzsche, F.R.; Weichert, W.; Röske, A.; Gekeler, V.; Beckers, T.; Stephan, C.; Jung, K.; Scholman, K.; Denkert, C.; Dietel, M.; et al. Class I Histone Deacetylases 1, 2 and 3 Are Highly Expressed in Renal Cell Cancer. BMC Cancer 2008, 8, 381. [Google Scholar] [CrossRef] [Green Version]
- Pacheco, M.; Nielsen, T.O. Histone Deacetylase 1 and 2 in Mesenchymal Tumors. Mod. Pathol. 2012, 25, 222–230. [Google Scholar] [CrossRef] [Green Version]
- Ler, S.Y.; Leung, C.H.W.; Khin, L.W.; Lu, G.D.; Salto-Tellez, M.; Hartman, M.; Iau, P.T.C.; Yap, C.T.; Hooi, S.C. HDAC1 and HDAC2 Independently Predict Mortality in Hepatocellular Carcinoma by a Competing Risk Regression Model in a Southeast Asian Population. Oncol. Rep. 2015, 34, 2238–2250. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Weichert, W.; Röske, A.; Gekeler, V.; Beckers, T.; Ebert, M.P.A.; Pross, M.; Dietel, M.; Denkert, C.; Röcken, C. Association of Patterns of Class I Histone Deacetylase Expression with Patient Prognosis in Gastric Cancer: A Retrospective Analysis. Lancet Oncol. 2008, 9, 139–148. [Google Scholar] [CrossRef] [PubMed]
- Osada, H.; Tatematsu, Y.; Saito, H.; Yatabe, Y.; Mitsudomi, T.; Takahashi, T. Reduced Expression of Class II Histone Deacetylase Genes Is Associated with Poor Prognosis in Lung Cancer Patients. Int. J. Cancer 2004, 112, 26–32. [Google Scholar] [CrossRef] [PubMed]
- Chaiyawat, P.; Pruksakorn, D.; Phanphaisarn, A.; Teeyakasem, P.; Klangjorhor, J.; Settakorn, J. Expression Patterns of Class i Histone Deacetylases in Osteosarcoma: A Novel Prognostic Marker with Potential Therapeutic Implications. Mod. Pathol. 2018, 31, 264–274. [Google Scholar] [CrossRef] [Green Version]
- Langer, R.; Mutze, K.; Becker, K.; Feith, M.; Ott, K.; Höfler, H.; Keller, G. Expression of Class I Histone Deacetylases (HDAC1 and HDAC2) in Oesophageal Adenocarcinomas: An Immunohistochemical Study. J. Clin. Pathol. 2010, 63, 994–998. [Google Scholar] [CrossRef] [Green Version]
- Klieser, E.; Urbas, R.; Stättner, S.; Primavesi, F.; Jäger, T.; Dinnewitzer, A.; Mayr, C.; Kiesslich, T.; Holzmann, K.; di Fazio, P.; et al. Comprehensive Immunohistochemical Analysis of Histone Deacetylases in Pancreatic Neuroendocrine Tumors: HDAC5 as a Predictor of Poor Clinical Outcome. Hum. Pathol. 2017, 65, 41–52. [Google Scholar] [CrossRef]
- Krusche, C.A.; Vloet, A.J.; Classen-Linke, I.; von Rango, U.; Beier, H.M.; Alfer, J. Class I Histone Deacetylase Expression in the Human Cyclic Endometrium and Endometrial Adenocarcinomas. Hum. Reprod. 2007, 22, 2956–2966. [Google Scholar] [CrossRef] [Green Version]
- Poyet, C.; Jentsch, B.; Hermanns, T.; Schweckendiek, D.; Seifert, H.H.; Schmidtpeter, M.; Sulser, T.; Moch, H.; Wild, P.J.; Kristiansen, G. Expression of Histone Deacetylases 1, 2 and 3 in Urothelial Bladder Cancer. BMC Clin. Pathol. 2014, 14, 10. [Google Scholar] [CrossRef] [Green Version]
- Li, H.; Li, H.; Waresijiang, Y.; Chen, Y.; Li, Y.; Yu, L.; Li, Y.; Liu, L. Clinical Significance of HDAC1, -2 and -3 Expression Levels in Esophageal Squamous Cell Carcinoma. Exp. Ther. Med. 2020, 20, 315–324. [Google Scholar] [CrossRef]
- Wang, L.; Zou, X.; Berger, A.D.; Twiss, C.; Peng, Y.; Li, Y.; Chiu, J.; Guo, H.; Satagopan, J.; Wilton, A.; et al. Increased Expression of Histone Deacetylaces (HDACs) and Inhibition of Prostate Cancer Growth and Invasion by HDAC Inhibitor SAHA. Am. J. Transl. Res. 2009, 1, 62. [Google Scholar]
- Maria Müller, B.; Jana, L.; Kasajima, A.; Lehmann, A.; Prinzler, J.; Budczies, J.; Winzer, K.-J.; Dietel, M.; Weichert, W.; Denkert, C. Differential Expression of Histone Deacetylases HDAC1, 2 and 3 in Human Breast Cancer-Overexpression of HDAC2 and HDAC3 Is Associated with Clinicopathological Indicators of Disease Progression. BMC Cancer 2013, 13, 215. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lucio-Eterovic, A.K.B.; Cortez, M.A.A.; Valera, E.T.; Motta, F.J.N.; Queiroz, R.G.P.; Machado, H.R.; Carlotti, C.G.; Neder, L.; Scrideli, C.A.; Tone, L.G. Differential Expression of 12 Histone Deacetylase (HDAC) Genes in Astrocytomas and Normal Brain Tissue: Class II and IV Are Hypoexpressed in Glioblastomas. BMC Cancer 2008, 8, 243. [Google Scholar] [CrossRef] [PubMed]
- Kouraklis, G.; Theocharis, S. Histone Deacetylase Inhibitors and Anticancer Therapy. Curr. Med. Chem. Anticancer. Agents 2002, 2, 477–484. [Google Scholar] [CrossRef] [PubMed]
- Palamaris, K.; Moutafi, M.; Gakiopoulou, H.; Theocharis, S. Histone Deacetylase (HDAC) Inhibitors: A Promising Weapon to Tackle Therapy Resistance in Melanoma. Int. J. Mol. Sci. 2022, 23, 3660. [Google Scholar] [CrossRef]
- McClure, J.J.; Li, X.; Chou, C.J. Advances and Challenges of HDAC Inhibitors in Cancer Therapeutics. In Advances in Cancer Research; Academic Press Inc.: Cambridge, MA, USA, 2018; Volume 138, pp. 183–211. [Google Scholar]
- Chang, H.H.; Chiang, C.P.; Hung, H.C.; Lin, C.Y.; Deng, Y.T.; Kuo, M.Y.P. Histone Deacetylase 2 Expression Predicts Poorer Prognosis in Oral Cancer Patients. Oral. Oncol. 2009, 45, 610–614. [Google Scholar] [CrossRef]
- He, J.C.; Yao, W.; Wang, J.M.; Schemmer, P.; Yang, Y.; Liu, Y.; Qian, Y.W.; Qi, W.P.; Zhang, J.; Shen, Q.; et al. TACC3 Overexpression in Cholangiocarcinoma Correlates with Poor Prognosis and Is a Potential Anti-Cancer Molecular Drug Target for HDAC Inhibitors. Oncotarget 2016, 7, 75441–75456. [Google Scholar] [CrossRef] [Green Version]
- Song, J.; Noh, J.H.; Lee, J.H.; Eun, J.W.; Ahn, Y.M.; Kim, S.Y.; Hyung Lee, S.; Park, W.S.; Yoo, N.J.; Lee, J.Y.; et al. Increased Expression of Histone Deacetylase 2 Is Found in Human Gastric Cancer. APMIS 2005, 113, 264–268. [Google Scholar] [CrossRef]
- Weichert, W.; Röske, A.; Niesporek, S.; Noske, A.; Buckendahl, A.C.; Dietel, M.; Gekeler, V.; Boehm, M.; Beckers, T.; Denkert, C. Class I Histone Deacetylase Expression Has Independent Prognostic Impact in Human Colorectal Cancer: Specific Role of Class I Histone Deacetylases In Vitro and In Vivo. Clin. Cancer Res. 2008, 14, 1669–1677. [Google Scholar] [CrossRef] [Green Version]
- Weichert, W.; Denkert, C.; Noske, A.; Darb-Esfahani, S.; Dietel, M.; Kalloger, S.E.; Huntsman, D.G.; Köbel, M. Expression of Class I Histone Deacetylases Indicates Poor Prognosis in Endometrioid Subtypes of Ovarian and Endometrial Carcinomas. Neoplasia 2008, 10, 1021–1027. [Google Scholar] [CrossRef] [Green Version]
- Nemati, M.; Ajami, N.; Estiar, M.A.; Rezapour, S.; Gavgani, R.R.; Hashemzadeh, S.; Kafil, H.S.; Sakhinia, E. Deregulated Expression of HDAC3 in Colorectal Cancer and Its Clinical Significance. Adv. Clin. Exp. Med. 2018, 27, 305–311. [Google Scholar] [CrossRef] [Green Version]
- Cao, L.L.; Song, X.; Pei, L.; Liu, L.; Wang, H.; Jia, M. Histone Deacetylase HDAC1 Expression Correlates with the Progression and Prognosis of Lung Cancer. Medicine 2017, 96, e7663. [Google Scholar] [CrossRef] [PubMed]
- Zeng, L.S.; Yang, X.Z.; Wen, Y.F.; Mai, S.J.; Wang, M.H.; Zhang, M.Y.; Steven Zheng, X.F.; Wang, H.Y. Overexpressed HDAC4 Is Associated with Poor Survival and Promotes Tumor Progression in Esophageal Carcinoma. Aging 2016, 8, 1236. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cheng, C.; Yang, J.; Li, S.W.; Huang, G.; Li, C.; Min, W.P.; Sang, Y. HDAC4 Promotes Nasopharyngeal Carcinoma Progression and Serves as a Therapeutic Target. Cell Death Dis. 2021, 12, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Li, A.; Liu, Z.; Li, M.; Zhou, S.; Xu, Y.; Xiao, Y.; Yang, W. HDAC5, a Potential Therapeutic Target and Prognostic Biomarker, Promotes Proliferation, Invasion and Migration in Human Breast Cancer. Oncotarget 2016, 7, 37966–37978. [Google Scholar] [CrossRef] [PubMed]
- Pouloudi, D.; Manou, M.; Sarantis, P.; Tsoukalas, N.; Tsourouflis, G.; Dana, E.; Karamouzis, M.V.; Klijanienko, J.; Theocharis, S. Clinical Significance of Histone Deacetylase (HDAC)-1, -2, -4 and -6 Expression in Salivary Gland Tumors. Diagn 2021, 11, 517. [Google Scholar] [CrossRef] [PubMed]
- Giaginis, C.; Alexandrou, P.; Delladetsima, I.; Giannopoulou, I.; Patsouris, E.; Theocharis, S. Clinical Significance of Histone Deacetylase (HDAC)-1, HDAC-2, HDAC-4, and HDAC-6 Expression in Human Malignant and Benign Thyroid Lesions. Tumour Biol. 2014, 35, 61–71. [Google Scholar] [CrossRef] [PubMed]
- Sakuma, T.; Uzawa, K.; Onda, T.; Shiiba, M.; Yokoe, H.; Shibahara, T.; Tanzawa, H. Aberrant Expression of Histone Deacetylase 6 in Oral Squamous Cell Carcinoma. Int. J. Oncol. 2006, 29, 117–124. [Google Scholar] [CrossRef]
- Yang, W.M.; Tsai, S.C.; Wen, Y.D.; Fejé, G.; Seto, E. Functional Domains of Histone Deacetylase-3. J. Biol. Chem. 2002, 277, 9447–9454. [Google Scholar] [CrossRef] [Green Version]
- Viatour, P.; Legrand-Poels, S.; van Lint, C.; Warnier, M.; Merville, M.P.; Gielen, J.; Piette, J.; Bours, V.; Chariot, A. Cytoplasmic IκBα Increases NF-ΚB-Independent Transcription through Binding to Histone Deacetylase (HDAC) 1 and HDAC3. J. Biol. Chem. 2003, 278, 46541–46548. [Google Scholar] [CrossRef] [Green Version]
- Krämer, O.H.; Knauer, S.K.; Greiner, G.; Jandt, E.; Reichardt, S.; Gührs, K.H.; Stauber, R.H.; Böhmer, F.D.; Heinzel, T. A Phosphorylation-Acetylation Switch Regulates STAT1 Signaling. Genes Dev. 2009, 23, 223–235. [Google Scholar] [CrossRef] [Green Version]
- Yuan, Z.L.; Guan, Y.J.; Chatterjee, D.; Chin, Y.E. Stat3 Dimerization Regulated by Reversible Acetylation of a Single Lysine Residue. Science 2005, 307, 269–273. [Google Scholar] [CrossRef]
- Chen, L.F.; Fischle, W.; Verdin, E.; Greene, W.C. Duration of Nuclear NF-ΚB Action Regulated by Reversible Acetylation. Science 2001, 293, 1653–1657. [Google Scholar] [CrossRef] [Green Version]
- Kiernan, R.; Brès, V.; Ng, R.W.M.; Coudart, M.P.; el Messaoudi, S.; Sardet, C.; Jin, D.Y.; Emiliani, S.; Benkirane, M. Post-Activation Turn-off of NF-ΚB-Dependent Transcription Is Regulated by Acetylation of P65. J. Biol. Chem. 2003, 278, 2758–2766. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vidal-Laliena, M.; Gallastegui, E.; Mateo, F.; Martínez-Balbás, M.; Pujol, M.J.; Bachs, O. Histone Deacetylase 3 Regulates Cyclin A Stability. J. Biol. Chem. 2013, 288, 21096. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nishino, T.G.; Miyazaki, M.; Hoshino, H.; Miwa, Y.; Horinouchi, S.; Yoshida, M. 14-3-3 Regulates the Nuclear Import of Class IIa Histone Deacetylases. Biochem. Biophys. Res. Commun. 2008, 377, 852–856. [Google Scholar] [CrossRef] [PubMed]
- Basile, V.; Mantovani, R.; Imbriano, C. DNA Damage Promotes Histone Deacetylase 4 Nuclear Localization and Repression of G2/M Promoters, via P53 C-Terminal Lysines. J. Biol. Chem. 2006, 281, 2347–2357. [Google Scholar] [CrossRef] [Green Version]
- Jeong, J.W.; Bae, M.K.; Ahn, M.Y.; Kim, S.H.; Sohn, T.K.; Bae, M.H.; Yoo, M.A.; Song, E.J.; Lee, K.J.; Kim, K.W. Regulation and Destabilization of HIF-1α by ARD1-Mediated Acetylation. Cell 2002, 111, 709–720. [Google Scholar] [CrossRef] [Green Version]
- Li, Y.; Zhang, X.; Polakiewicz, R.D.; Yao, T.P.; Comb, M.J. HDAC6 Is Required for Epidermal Growth Factor-Induced β-Catenin Nuclear Localization. J. Biol. Chem. 2008, 283, 12686–12690. [Google Scholar] [CrossRef] [Green Version]
- Karagiannis, D.; Rampias, T. HDAC Inhibitors: Dissecting Mechanisms of Action to Counter Tumor Heterogeneity. Cancers 2021, 13, 3575. [Google Scholar] [CrossRef]
- Arjonen, A.; Mäkelä, R.; Härmä, V.; Rintanen, N.; Kuopio, T.; Kononen, J.; Rantala, J.K. Image-Based Ex Vivo Drug Screen to Assess Targeted Therapies in Recurrent Thymoma. Lung Cancer 2020, 145, 27–32. [Google Scholar] [CrossRef]
- Bellissimo, T.; Ganci, F.; Gallo, E.; Sacconi, A.; Tito, C.; de Angelis, L.; Pulito, C.; Masciarelli, S.; Diso, D.; Anile, M.; et al. Thymic Epithelial Tumors Phenotype Relies on MiR-145-5p Epigenetic Regulation. Mol. Cancer 2017, 16, 1–15. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Giaccone, G.; Rajan, A.; Berman, A.; Kelly, R.J.; Szabo, E.; Lopez-Chavez, A.; Trepel, J.; Lee, M.J.; Cao, L.; Espinoza-Delgado, I.; et al. Phase II Study of Belinostat in Patients with Recurrent or Refractory Advanced Thymic Epithelial Tumors. J. Clin. Oncol. 2011, 29, 2052–2059. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Thomas, A.; Rajan, A.; Szabo, E.; Tomita, Y.; Carter, C.A.; Scepura, B.; Lopez-Chavez, A.; Lee, M.J.; Redon, C.E.; Frosch, A.; et al. A Phase I/II Trial of Belinostat in Combination with Cisplatin, Doxorubicin, and Cyclophosphamide in Thymic Epithelial Tumors: A Clinical and Translational Study. Clin. Cancer Res. 2014, 20, 5392–5402. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Marx, A.; Chan, J.K.C.; Chalabreysse, L.; Dacic, S.; Detterbeck, F.; French, C.A.; Hornick, J.L.; Inagaki, H.; Jain, D.; Lazar, A.J.; et al. The 2021 WHO Classification of Tumors of the Thymus and Mediastinum: What Is New in Thymic Epithelial, Germ Cell, and Mesenchymal Tumors? J. Thorac. Oncol. 2022, 17, 200–213. [Google Scholar] [CrossRef] [PubMed]
- Koga, K.; Matsuno, Y.; Noguchi, M.; Mukai, K.; Goya, T.; Shimosato, Y. A Review of 79 Thymomas: Modification of Staging System and Reappraisal of Conventional Division into Invasive and Non-Invasive Thymoma. Pathol. Int. 1994, 44, 359–367. [Google Scholar] [CrossRef] [PubMed]


| Parameter | Median | Range |
|---|---|---|
| Age | 62 | 27–88 years |
| Number | % | |
| Gender | ||
| Male | 39/91 | 43% |
| Female | 52/91 | 57% |
| WHO subtypes | ||
| Type A | 12/91 | 13.6% |
| Type AB | 19/91 | 20.8% |
| Type B1 | 14/91 | 15.4% |
| Type B2 | 19/91 | 20.8% |
| Type B3 | 14/91 | 15.4% |
| Micronodular with lymphoid stroma | 2/91 | 2.2% |
| Thymic carcinoma | 11/91 | 12.1% |
| Masaoka–Koga stage | ||
| I | 13/81 | 16% |
| IIa | 32/81 | 39.5% |
| IIb | 14/81 | 17.3% |
| III | 64/81 | 19.7% |
| IVa | 3/81 | 3.7% |
| IVb | 3/81 | 3.7% |
| Presence of myasthenia gravis | 35/59 | 59.3% |
| Presence of chemotherapy | 11/39 | 28% |
| Presence of radiotherapy | 19/38 | 50% |
| Event | ||
| Alive | 29/40. follow-up 5–134 months | 72.5% |
| Dead of disease | 11/40. within 7–65 months | 27.5% |
| Presence of relapse | 4/35. within 58–65 months | 11% |
| Positivity Rate | H-Score, Median | H-Score, Range | |
|---|---|---|---|
| HDAC1 nuclear expression | 96.5% | 200 | 0–300 |
| HDAC1 cytoplasmic expression | 3.5% | 0 | 0–40 |
| HDAC2 nuclear/cytoplasmic expression | 97% | 85 | 0–300 |
| HDAC3 nuclear expression | 94% | 140 | 0–300 |
| HDAC3 cytoplasmic expression | 77% | 100 | 0–200 |
| HDAC4 cytoplasmic expression | 70% | 45 | 0–210 |
| HDAC5 nuclear expression | 13% | 0 | 0–20 |
| HDAC5 cytoplasmic expression | 62% | 45 | 0–300 |
| HDAC6 cytoplasmic expression | 29.6% | 0 | 0–100 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Palamaris, K.; Tzimou, L.-M.; Levidou, G.; Masaoutis, C.; Theochari, I.; Rontogianni, D.; Theocharis, S. Histone Deacetylases (HDACs): Promising Biomarkers and Potential Therapeutic Targets in Thymic Epithelial Tumors. Int. J. Mol. Sci. 2023, 24, 4263. https://doi.org/10.3390/ijms24054263
Palamaris K, Tzimou L-M, Levidou G, Masaoutis C, Theochari I, Rontogianni D, Theocharis S. Histone Deacetylases (HDACs): Promising Biomarkers and Potential Therapeutic Targets in Thymic Epithelial Tumors. International Journal of Molecular Sciences. 2023; 24(5):4263. https://doi.org/10.3390/ijms24054263
Chicago/Turabian StylePalamaris, Kostas, Luisa-Maria Tzimou, Georgia Levidou, Christos Masaoutis, Irene Theochari, Dimitra Rontogianni, and Stamatios Theocharis. 2023. "Histone Deacetylases (HDACs): Promising Biomarkers and Potential Therapeutic Targets in Thymic Epithelial Tumors" International Journal of Molecular Sciences 24, no. 5: 4263. https://doi.org/10.3390/ijms24054263





