Navigating Like a Fly: Drosophila melanogaster as a Model to Explore the Contribution of Serotonergic Neurotransmission to Spatial Navigation
Abstract
1. Introduction
2. A Brief Description of the Serotonergic System in Drosophila melanogaster
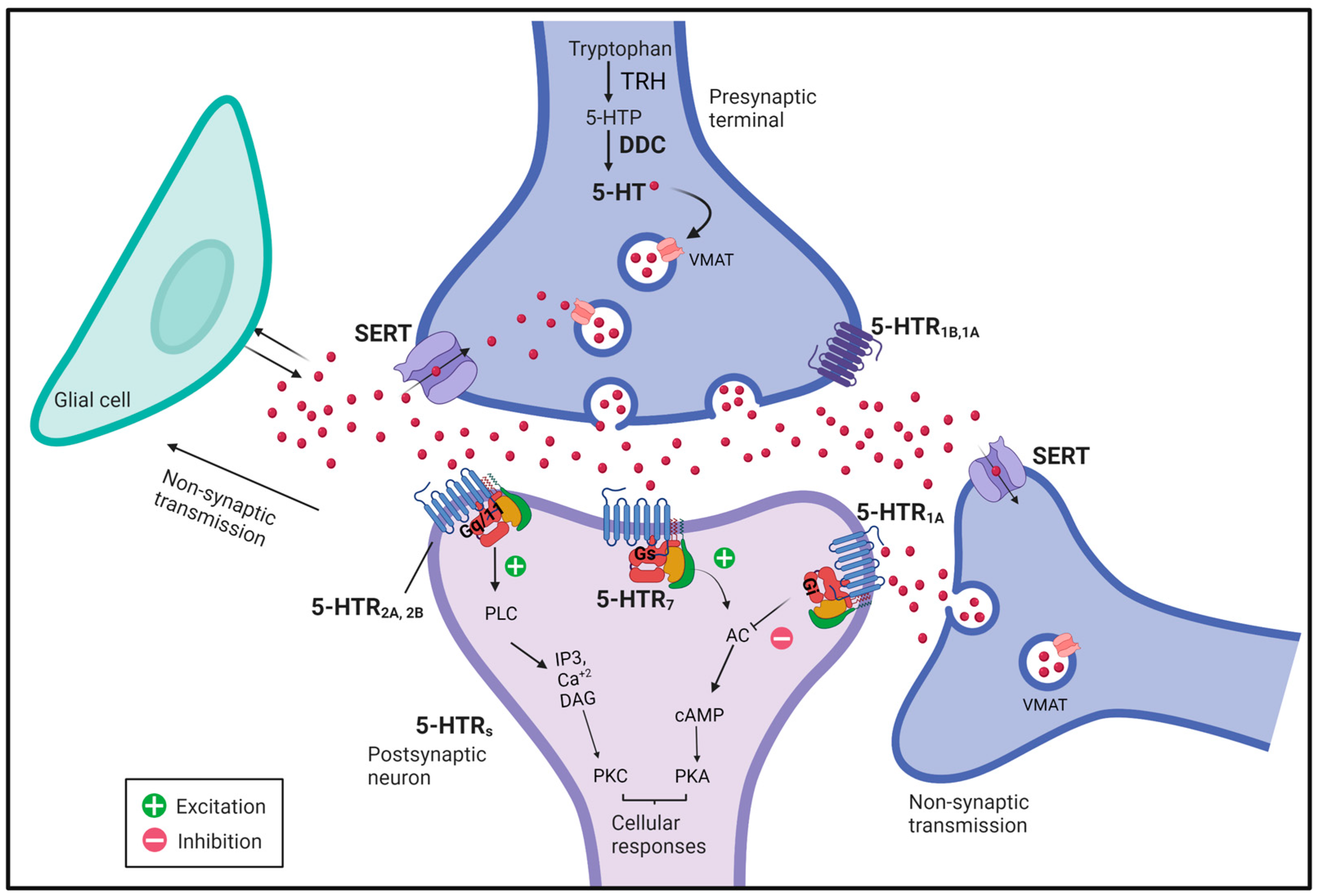
2.1. Molecular Organization of Serotonergic Components in the Adult Fly
2.2. Anatomical Organization of Serotonin-Releasing Neurons and Their Projections in the Adult Brain
3. Serotonergic Neural Circuits on Different Aspects of Spatial Navigation
4. Conclusions and Future Directions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sizemore, T.R.; Dacks, A.M. Serotonergic Modulation Differentially Targets Distinct Network Elements within the Antennal Lobe of Drosophila melanogaster. Sci. Rep. 2016, 6, 37119. [Google Scholar] [CrossRef] [PubMed]
- Glikmann-Johnston, Y.; Saling, M.M.; Reutens, D.C.; Stout, J.C. Hippocampal 5-HT1A Receptor and Spatial Learning and Memory. Front. Pharmacol. 2015, 6, 289. [Google Scholar] [CrossRef] [PubMed]
- Wert-Carvajal, C.; Reneaux, M.; Tchumatchenko, T.; Clopath, C. Dopamine and Serotonin Interplay for Valence-Based Spatial Learning. Cell Rep. 2022, 39, 110645. [Google Scholar] [CrossRef]
- Cools, R.; Roberts, A.C.; Robbins, T.W. Serotoninergic Regulation of Emotional and Behavioural Control Processes. Trends Cogn. Sci. 2008, 12, 31–40. [Google Scholar] [CrossRef]
- Blenau, W.; Thamm, M. Distribution of Serotonin (5-HT) and Its Receptors in the Insect Brain with Focus on the Mushroom Bodies. Lessons from Drosophila melanogaster and Apis Mellifera. Arthropod Struct. Dev. 2011, 40, 381–394. [Google Scholar] [CrossRef]
- Lesch, K.-P.; Waider, J. Serotonin in the Modulation of Neural Plasticity and Networks: Implications for Neurodevelopmental Disorders. Neuron 2012, 76, 175–191. [Google Scholar] [CrossRef]
- Albin, S.D.; Kaun, K.R.; Knapp, J.-M.; Chung, P.; Heberlein, U.; Simpson, J.H. A Subset of Serotonergic Neurons Evokes Hunger in Adult Drosophila. Curr. Biol. 2015, 25, 2435–2440. [Google Scholar] [CrossRef]
- Ries, A.-S.; Hermanns, T.; Poeck, B.; Strauss, R. Serotonin Modulates a Depression-like State in Drosophila Responsive to Lithium Treatment. Nat. Commun. 2017, 8, 15738. [Google Scholar] [CrossRef]
- Howard, C.E.; Chen, C.-L.; Tabachnik, T.; Hormigo, R.; Ramdya, P.; Mann, R.S. Serotonergic Modulation of Walking in Drosophila. Curr. Biol. 2019, 29, 4218–4230.e8. [Google Scholar] [CrossRef]
- Alekseyenko, O.V.; Lee, C.; Kravitz, E.A. Targeted Manipulation of Serotonergic Neurotransmission Affects the Escalation of Aggression in Adult Male Drosophila melanogaster. PLoS ONE 2010, 5, e10806. [Google Scholar] [CrossRef]
- Alekseyenko, O.V.; Kravitz, E.A. Serotonin and the Search for the Anatomical Substrate of Aggression. Fly 2014, 8, 200–205. [Google Scholar] [CrossRef]
- Pooryasin, A.; Fiala, A. Identified Serotonin-Releasing Neurons Induce Behavioral Quiescence and Suppress Mating in Drosophila. J. Neurosci. 2015, 35, 12792–12812. [Google Scholar] [CrossRef] [PubMed]
- Sitaraman, D.; Kramer, E.F.; Kahsai, L.; Ostrowski, D.; Zars, T. Discrete Serotonin Systems Mediate Memory Enhancement and Escape Latencies after Unpredicted Aversive Experience in Drosophila Place Memory. Front. Syst. Neurosci. 2017, 11, 92. [Google Scholar] [CrossRef]
- Scheunemann, L.; Plaçais, P.-Y.; Dromard, Y.; Schwärzel, M.; Preat, T. Dunce Phosphodiesterase Acts as a Checkpoint for Drosophila Long-Term Memory in a Pair of Serotonergic Neurons. Neuron 2018, 98, 350–365.e5. [Google Scholar] [CrossRef] [PubMed]
- De-Miguel, F.F.; Trueta, C. Synaptic and Extrasynaptic Secretion of Serotonin. Cell Mol. Neurobiol. 2005, 25, 297–312. [Google Scholar] [CrossRef] [PubMed]
- Kaushalya, S.K.; Nag, S.; Balaji, J.; Maiti, S. Serotonin: Multiphoton Imaging and Relevant Spectral Data. In Proceedings of the Multiphoton Microscopy in the Biomedical Sciences VIII, San Jose, CA, USA, 15 February 2008; Periasamy, A., So, P.T.C., Eds.; SPIE: Bellingham, DC, USA, 2008; p. 68601C. [Google Scholar] [CrossRef]
- De-Miguel, F.F.; Leon-Pinzon, C.; Noguez, P.; Mendez, B. Serotonin Release from the Neuronal Cell Body and Its Long-Lasting Effects on the Nervous System. Philos. Trans. R. Soc. B Biol. Sci. 2015, 370, 20140196. [Google Scholar] [CrossRef]
- Coleman, C.M.; Neckameyer, W.S. Serotonin Synthesis by Two Distinct Enzymes in Drosophila melanogaster. Arch. Insect Biochem. Physiol. 2005, 59, 12–31. [Google Scholar] [CrossRef]
- Tempel, B.L.; Livingstone, M.S.; Quinn, W.G. Mutations in the Dopa Decarboxylase Gene Affect Learning in Drosophila. Proc. Natl. Acad. Sci. USA 1984, 81, 3577–3581. [Google Scholar] [CrossRef]
- Beall, C.J.; Hirsh, J. Regulation of the Drosophila dopa decarboxylase gene in neuronal and glial cells. Genes Dev. 1987, 1, 510–520. [Google Scholar] [CrossRef]
- Greer, C.L.; Grygoruk, A.; Patton, D.E.; Ley, B.; Romero-Calderon, R.; Chang, H.-Y.; Houshyar, R.; Bainton, R.J.; DiAntonio, A.; Krantz, D.E. A Splice Variant of TheDrosophila Vesicular Monoamine Transporter Contains a Conserved Trafficking Domain and Functions in the Storage of Dopamine, Serotonin, and Octopamine. J. Neurobiol. 2005, 64, 239–258. [Google Scholar] [CrossRef]
- Park, S.K.; George, R.; Cai, Y.; Chang, H.Y.; Krantz, D.E.; Friggi-Grelin, F.; Birman, S.; Hirsh, J. Cell-Type-Specific Limitation Onin Vivo Serotonin Storage Following Ectopic Expression of TheDrosophila Serotonin Transporter, DSERT. J. Neurobiol. 2006, 66, 452–462. [Google Scholar] [CrossRef]
- Roelofs, J.; Van Haastert, P. Genes lost during evolution. Nature 2001, 411, 1013–1014. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Zhang, C.-W.; Chen, G.Y.J.; Zhu, B.; Chai, C.; Xu, Q.-H.; Tan, E.-K.; Zhu, Q.; Lim, K.-L.; Yao, S.Q. A Sensitive Two-Photon Probe to Selectively Detect Monoamine Oxidase B Activity in Parkinson’s Disease Models. Nat. Commun. 2014, 5, 3276. [Google Scholar] [CrossRef]
- Neckameyer, W.S.; Coleman, C.M.; Eadie, S.; Goodwin, S.F. Compartmentalization of Neuronal and Peripheral Serotonin Synthesis in Drosophila melanogaster. Genes Brain Behav. 2007, 6, 756–769. [Google Scholar] [CrossRef] [PubMed]
- Bao, X.; Wang, B.; Zhang, J.; Yan, T.; Yang, W.; Jiao, F.; Liu, J.; Wang, S. Localization of Serotonin/Tryptophan-Hydroxylase-Immunoreactive Cells in the Brain and Suboesophageal Ganglion of Drosophila melanogaster. Cell Tissue Res. 2010, 340, 51–59. [Google Scholar] [CrossRef]
- Deshpande, S.A.; Freyberg, Z.; Lawal, H.O.; Krantz, D.E. Vesicular Neurotransmitter Transporters in Drosophila melanogaster. Biochim. Biophys. Acta BBA-Biomembr. 2020, 1862, 183308. [Google Scholar] [CrossRef] [PubMed]
- Demchyshyn, L.L.; Pristupa, Z.B.; Sugamori, K.S.; Barker, E.L.; Blakely, R.D.; Wolfgang, W.J.; Forte, M.A.; Niznik, H.B. Cloning, Expression, and Localization of a Chloride-Facilitated, Cocaine-Sensitive Serotonin Transporter from Drosophila melanogaster. Proc. Natl. Acad. Sci. USA 1994, 91, 5158–5162. [Google Scholar] [CrossRef]
- Corey, J.L.; Quick, M.W.; Davidson, N.; Lester, H.A.; Guastella, J. A Cocaine-Sensitive Drosophila Serotonin Transporter: Cloning, Expression, and Electrophysiological Characterization. Proc. Natl. Acad. Sci. USA 1994, 91, 1188–1192. [Google Scholar] [CrossRef] [PubMed]
- Giang, T.; Rauchfuss, S.; Ogueta, M.; Scholz, H. The Serotonin Transporter Expression in Drosophila melanogaster. J. Neurogenet. 2011, 25, 17–26. [Google Scholar] [CrossRef]
- Becnel, J.; Johnson, O.; Luo, J.; Nässel, D.R.; Nichols, C.D. The Serotonin 5-HT7Dro Receptor Is Expressed in the Brain of Drosophila, and Is Essential for Normal Courtship and Mating. PLoS ONE 2011, 6, e20800. [Google Scholar] [CrossRef]
- Kahsai, L.; Carlsson, M.A.; Winther, Å.M.E.; Nässel, D.R. Distribution of Metabotropic Receptors of Serotonin, Dopamine, GABA, Glutamate, and Short Neuropeptide F in the Central Complex of Drosophila. Neuroscience 2012, 208, 11–26. [Google Scholar] [CrossRef] [PubMed]
- Blenau, W.; Daniel, S.; Balfanz, S.; Thamm, M.; Baumann, A. Dm5-HT2B: Pharmacological Characterization of the Fifth Serotonin Receptor Subtype of Drosophila melanogaster. Front. Syst. Neurosci. 2017, 11, 28. [Google Scholar] [CrossRef] [PubMed]
- Vogt, K. Towards a Functional Connectome in Drosophila. J. Neurogenet. 2020, 34, 156–161. [Google Scholar] [CrossRef]
- Bose, S.K.; Mehta, M.A.; Selvaraj, S.; Howes, O.D.; Hinz, R.; Rabiner, E.A.; Grasby, P.M.; Turkheimer, F.E.; Murthy, V. Presynaptic 5-HT1A Is Related to 5-HTT Receptor Density in the Human Brain. Neuropsychopharmacology 2011, 36, 2258–2265. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Garcia, A.L.; Newman-Tancredi, A.; Leonardo, E.D. P5-HT1A Receptors in Mood and Anxiety: Recent Insights into Autoreceptor versus Heteroreceptor Function. Psychopharmacology 2014, 231, 623–636. [Google Scholar] [CrossRef]
- Yuan, Q.; Lin, F.; Zheng, X.; Sehgal, A. Serotonin Modulates Circadian Entrainment in Drosophila. Neuron 2005, 47, 115–127. [Google Scholar] [CrossRef]
- Johnson, O.; Becnel, J.; Nichols, C.D. Serotonin 5-HT2 and 5-HT1A-like Receptors Differentially Modulate Aggressive Behaviors in Drosophila melanogaster. Neuroscience 2009, 158, 1292–1300. [Google Scholar] [CrossRef]
- Sampson, M.M.; Myers Gschweng, K.M.; Hardcastle, B.J.; Bonanno, S.L.; Sizemore, T.R.; Arnold, R.C.; Gao, F.; Dacks, A.M.; Frye, M.A.; Krantz, D.E. Serotonergic Modulation of Visual Neurons in Drosophila melanogaster. PLoS Genet. 2020, 16, e1009003. [Google Scholar] [CrossRef]
- Gramates, L.S.; Agapite, J.; Attrill, H.; Calvi, B.R.; Crosby, M.A.; dos Santos, G.; Goodman, J.L.; Goutte-Gattat, D.; Jenkins, V.K.; Kaufman, T.; et al. FlyBase: A Guided Tour of Highlighted Features. Genetics 2022, 220, iyac035. [Google Scholar] [CrossRef]
- Saudou, F.; Boschert, U.; Amlaiky, N.; Plassat, J.L.; Hen, R. A Family of Drosophila Serotonin Receptors with Distinct Intracellular Signalling Properties and Expression Patterns. EMBO J. 1992, 11, 7–17. [Google Scholar] [CrossRef]
- Clarke, H.F. Prefrontal Serotonin Depletion Affects Reversal Learning But Not Attentional Set Shifting. J. Neurosci. 2005, 25, 532–538. [Google Scholar] [CrossRef]
- Gasque, G.; Conway, S.; Huang, J.; Rao, Y.; Vosshall, L.B. Small Molecule Drug Screening in Drosophila Identifies the 5HT2A Receptor as a Feeding Modulation Target. Sci. Rep. 2013, 3, srep02120. [Google Scholar] [CrossRef] [PubMed]
- Colas, J.F.; Launay, J.M.; Kellermann, O.; Rosay, P.; Maroteaux, L. Drosophila 5-HT2 Serotonin Receptor: Coexpression with Fushi-Tarazu during Segmentation. Proc. Natl. Acad. Sci. USA 1995, 92, 5441–5445. [Google Scholar] [CrossRef] [PubMed]
- Witz, P.; Amlaiky, N.; Plassat, J.L.; Maroteaux, L.; Borrelli, E.; Hen, R. Cloning and Characterization of a Drosophila Serotonin Receptor That Activates Adenylate Cyclase. Proc. Natl. Acad. Sci. USA 1990, 87, 8940–8944. [Google Scholar] [CrossRef]
- Dahlström, A.; Fuxe, K. Evidence for the existence of monoamine-containing neurons in_the central nervous system. I. Demonstration of monoamines in the cell bodies of brain_stem neurons. Acta Physiol. Scand Suppl. 1964, 232, 1–55. Available online: https://pubmed.ncbi.nlm.nih.gov/14229500/ (accessed on 29 December 2022).
- Hökfelt, T. Early Attempts to Visualize Cortical Monoamine Nerve Terminals. Brain Res. 2016, 1645, 8–11. [Google Scholar] [CrossRef]
- Baker, K.G.; Halliday, G.M.; Hornung, J.-P.; Geffen, L.B.; Cotton, R.G.H.; To¨rk, I. Distribution, Morphology and Number of Monoamine-Synthesizing and Substance P-Containing Neurons in the Human Dorsal Raphe Nucleus. Neuroscience 1991, 42, 757–775. [Google Scholar] [CrossRef]
- Vertes, R.P.; Fortin, W.J.; Crane, A.M. Projections of the Median Raphe Nucleus in the Rat. J. Comp. Neurol. 1999, 407, 555–582. [Google Scholar] [CrossRef]
- Ishimura, K.; Takeuchi, Y.; Fujiwara, K.; Tominaga, M.; Yoshioka, H.; Sawada, T. Quantitative Analysis of the Distribution of Serotonin-Immunoreactive Cell Bodies in the Mouse Brain. Neurosci. Lett. 1988, 91, 265–270. [Google Scholar] [CrossRef]
- Jacobs, B.L.; Azmitia, E.C. Structure and Function of the Brain Serotonin System. Physiol. Rev. 1992, 72, 165–229. [Google Scholar] [CrossRef]
- Calizo, L.H.; Akanwa, A.; Ma, X.; Pan, Y.; Lemos, J.C.; Craige, C.; Heemstra, L.A.; Beck, S.G. Raphe Serotonin Neurons Are Not Homogenous: Electrophysiological, Morphological and Neurochemical Evidence. Neuropharmacology 2011, 61, 524–543. [Google Scholar] [CrossRef]
- Vallés, A.M.; White, K. Serotonin-Containing Neurons in Drosophila melanogaster: Development and Distribution: SEROTONIN IN DROSOPHILA NERVOUS SYSTEM. J. Comp. Neurol. 1988, 268, 414–428. [Google Scholar] [CrossRef] [PubMed]
- Sitaraman, D.; LaFerriere, H.; Birman, S.; Zars, T. Serotonin Is Critical for Rewarded Olfactory Short-Term Memory in Drosophila. J. Neurogenet. 2012, 26, 238–244. [Google Scholar] [CrossRef]
- Brand, A.H.; Perrimon, N. Targeted Gene Expression as a Means of Altering Cell Fates and Generating Dominant Phenotypes. Development 1993, 118, 401–415. [Google Scholar] [CrossRef]
- Duffy, J.B. GAL4 System In Drosophila: A Fly Geneticist’s Swiss Army Knife. Genesis 2002, 34, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Chiang, A.S.; Lin, C.Y.; Chuang, C.C.; Chang, H.M.; Hsieh, C.H.; Yeh, C.W.; Shih, C.T.; Wu, J.J.; Wang, G.T.; Chen, Y.C.; et al. Three-dimensional reconstruction of brain-wide wiring networks in Drosophila at single-cell resolution. Curr. Biol. 2011, 21, 1–11. [Google Scholar] [CrossRef]
- Zárate, R.V.; Hidalgo, S.; Navarro, N.; Molina-Mateo, D.; Arancibia, D.; Rojo-Cortés, F.; Oliva, C.; Andrés, M.E.; Zamorano, P.; Campusano, J.M. An Early Disturbance in Serotonergic Neurotransmission Contributes to the Onset of Parkinsonian Phenotypes in Drosophila melanogaster. Cells 2022, 11, 1544. [Google Scholar] [CrossRef] [PubMed]
- Sitaraman, D.; Zars, M.; LaFerriere, H.; Chen, Y.-C.; Sable-Smith, A.; Kitamoto, T.; Rottinghaus, G.E.; Zars, T. Serotonin Is Necessary for Place Memory in Drosophila. Proc. Natl. Acad. Sci. USA 2008, 105, 5579–5584. [Google Scholar] [CrossRef]
- Niens, J.; Reh, F.; Çoban, B.; Cichewicz, K.; Eckardt, J.; Liu, Y.-T.; Hirsh, J.; Riemensperger, T.D. Dopamine Modulates Serotonin Innervation in the Drosophila Brain. Front. Syst. Neurosci. 2017, 11, 76. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Lindsey, J.W.; Marin, E.C.; Otto, N.; Dreher, M.; Dempsey, G.; Stark, I.; Bates, A.S.; Pleijzier, M.W.; Schlegel, P.; et al. The Connectome of the Adult Drosophila Mushroom Body Provides Insights into Function. eLife 2020, 9, e62576. [Google Scholar] [CrossRef]
- Hulse, B.K.; Haberkern, H.; Franconville, R.; Turner-Evans, D.; Takemura, S.; Wolff, T.; Noorman, M.; Dreher, M.; Dan, C.; Parekh, R.; et al. A Connectome of the Drosophila Central Complex Reveals Network Motifs Suitable for Flexible Navigation and Context-Dependent Action Selection. eLife 2021, 10, e66039. [Google Scholar] [CrossRef] [PubMed]
- Coates, K.E.; Majot, A.T.; Zhang, X.; Michael, C.T.; Spitzer, S.L.; Gaudry, Q.; Dacks, A.M. Identified Serotonergic Modulatory Neurons Have Heterogeneous Synaptic Connectivity within the Olfactory System of Drosophila. J. Neurosci. 2017, 37, 7318–7331. [Google Scholar] [CrossRef] [PubMed]
- Coates, K.E.; Calle-Schuler, S.A.; Helmick, L.M.; Knotts, V.L.; Martik, B.N.; Salman, F.; Warner, L.T.; Valla, S.V.; Bock, D.D.; Dacks, A.M. The Wiring Logic of an Identified Serotonergic Neuron That Spans Sensory Networks. J. Neurosci. 2020, 40, 6309–6327. [Google Scholar] [CrossRef] [PubMed]
- Dacks, A.M.; Green, D.S.; Root, C.M.; Nighorn, A.J.; Wang, J.W. Serotonin modulates olfactory processing in the antennal lobe of Drosophila. J. Neurogenet. 2009, 23, 366–377. [Google Scholar] [CrossRef]
- Lee, P.-T.; Lin, H.-W.; Chang, Y.-H.; Fu, T.-F.; Dubnau, J.; Hirsh, J.; Lee, T.; Chiang, A.-S. Serotonin–Mushroom Body Circuit Modulating the Formation of Anesthesia-Resistant Memory in Drosophila. Proc. Natl. Acad. Sci. USA 2011, 108, 13794–13799. [Google Scholar] [CrossRef] [PubMed]
- Hidalgo, S.; Fuenzalida-Uribe, N.; Molina-Mateo, D.; Escobar, A.P.; Oliva, C.; España, R.A.; Andrés, M.E.; Campusano, J.M. Study of the release of endogenous amines in Drosophila brain in vivo in response to stimuli linked to aversive olfactory conditioning. J. Neurochem. 2021, 156, 337–351. [Google Scholar] [CrossRef]
- Owald, D.; Lin, S.; Waddell, S. Light, Heat, Action: Neural Control of Fruit Fly Behaviour. Philos. Trans. R. Soc. B Biol. Sci. 2015, 370, 20140211. [Google Scholar] [CrossRef]
- Martín, F.; Alcorta, E. Novel Genetic Approaches to Behavior in Drosophila. J. Neurogenet. 2017, 31, 288–299. [Google Scholar] [CrossRef]
- Pfeiffer, B.D.; Ngo, T.T.; Hibbard, K.L.; Murphy, C.; Jenett, A.; Truman, J.W.; Rubin, G.M. Refinement of tools for targeted gene expression in Drosophila. Genetics 2010, 186, 735–755. [Google Scholar] [CrossRef]
- Jenett, A.; Rubin, G.M.; Ngo, T.T.; Shepherd, D.; Murphy, C.; Dionne, H.; Pfeiffer, B.D.; Cavallaro, A.; Hall, D.; Jeter, J.; et al. A GAL4-driver line resource for Drosophila neurobiology. Cell Rep. 2012, 2, 991–1001. [Google Scholar] [CrossRef]
- Tirian, L.; Dickson, B.J. The VT GAL4, LexA, and Split-GAL4 Driver Line Collections for Targeted Expression in the Drosophila Nervous System. Neuroscience 2017. preprint. [Google Scholar] [CrossRef]
- Qian, Y.; Cao, Y.; Deng, B.; Yang, G.; Li, J.; Xu, R.; Zhang, D.; Huang, J.; Rao, Y. Sleep Homeostasis Regulated by 5HT2b Receptor in a Small Subset of Neurons in the Dorsal Fan-Shaped Body of Drosophila. eLife 2017, 6, e26519. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Meng, Z.; Wiggin, T.D.; Yu, J.; Reed, M.L.; Guo, F.; Zhang, Y.; Rosbash, M.; Griffith, L.C. A Serotonin-Modulated Circuit Controls Sleep Architecture to Regulate Cognitive Function Independent of Total Sleep in Drosophila. Curr. Biol. 2019, 29, 3635–3646.e5. [Google Scholar] [CrossRef]
- Cao, H.; Tang, J.; Liu, Q.; Huang, J.; Xu, R. Autism-like Behaviors Regulated by the Serotonin Receptor 5-HT2B in the Dorsal Fan-Shaped Body Neurons of Drosophila melanogaster. Eur. J. Med. Res. 2022, 27, 203. [Google Scholar] [CrossRef]
- Alekseyenko, O.V.; Chan, Y.-B.; Fernandez, M.d.l.P.; Bülow, T.; Pankratz, M.J.; Kravitz, E.A. Single Serotonergic Neurons That Modulate Aggression in Drosophila. Curr. Biol. 2014, 24, 2700–2707. [Google Scholar] [CrossRef] [PubMed]
- Alekseyenko, O.V.; Chan, Y.-B.; Okaty, B.W.; Chang, Y.; Dymecki, S.M.; Kravitz, E.A. Serotonergic Modulation of Aggression in Drosophila Involves GABAergic and Cholinergic Opposing Pathways. Curr. Biol. 2019, 29, 2145–2156.e5. [Google Scholar] [CrossRef]
- Hu, S.W.; Yang, Y.T.; Sun, Y.; Zhan, Y.P.; Zhu, Y. Serotonin Signals Overcome Loser Mentality in Drosophila. iScience 2020, 23, 101651. [Google Scholar] [CrossRef]
- Sun, Y.; Qiu, R.; Li, X.; Cheng, Y.; Gao, S.; Kong, F.; Liu, L.; Zhu, Y. Social Attraction in Drosophila Is Regulated by the Mushroom Body and Serotonergic System. Nat. Commun. 2020, 11, 5350. [Google Scholar] [CrossRef]
- Hinman, J.R.; Dannenberg, H.; Alexander, A.S.; Hasselmo, M.E. Neural Mechanisms of Navigation Involving Interactions of Cortical and Subcortical Structures. J Neurophysiol. 2018, 119, 2007–2029. [Google Scholar] [CrossRef]
- Hartley, T.; Lever, C.; Burgess, N.; O’Keefe, J. Space in the Brain: How the Hippocampal Formation Supports Spatial Cognition. Philos. Trans. R Soc. B Biol. Sci. 2014, 369, 20120510. [Google Scholar] [CrossRef]
- Moser, M.-B.; Rowland, D.C.; Moser, E.I. Place Cells, Grid Cells, and Memory. Cold Spring Harb. Perspect. Biol. 2015, 7, a021808. [Google Scholar] [CrossRef] [PubMed]
- Muzerelle, A.; Scotto-Lomassese, S.; Bernard, J.F.; Soiza-Reilly, M.; Gaspar, P. Conditional Anterograde Tracing Reveals Distinct Targeting of Individual Serotonin Cell Groups (B5–B9) to the Forebrain and Brainstem. Brain Struct. Funct. 2016, 221, 535–561. [Google Scholar] [CrossRef]
- Buhot, M.C.; Patra, S.K.; Naïli, S. Spatial memory deficits following stimulation of hippocampal 5-HT1B receptors in the rat. Eur. J. Pharmacol. 1995, 285, 221–228. [Google Scholar] [CrossRef]
- Rogers, J.; Churilov, L.; Hannan, A.J.; Renoir, T. Search Strategy Selection in the Morris Water Maze Indicates Allocentric Map Formation during Learning That Underpins Spatial Memory Formation. Neurobiol. Learn. Mem. 2017, 139, 37–49. [Google Scholar] [CrossRef] [PubMed]
- Beaudet, G.; Jozet-Alves, C.; Asselot, R.; Schumann-Bard, P.; Freret, T.; Boulouard, M.; Paizanis, E. Deletion of the Serotonin Receptor Type 7 Disrupts the Acquisition of Allocentric but Not Egocentric Navigation Strategies in Mice. Behav. Brain Res. 2017, 320, 179–185. [Google Scholar] [CrossRef]
- Ryu, L.; Kim, S.Y.; Kim, A.J. From Photons to Behaviors: Neural Implementations of Visual Behaviors in Drosophila. Front. Neurosci. 2022, 16, 883640. [Google Scholar] [CrossRef]
- Turner-Evans, D.B.; Jensen, K.T.; Ali, S.; Paterson, T.; Sheridan, A.; Ray, R.P.; Wolff, T.; Lauritzen, J.S.; Rubin, G.M.; Bock, D.D.; et al. The Neuroanatomical Ultrastructure and Function of a Biological Ring Attractor. Neuron 2020, 108, 145–163.e10. [Google Scholar] [CrossRef]
- Namiki, S.; Dickinson, M.H.; Wong, A.M.; Korff, W.; Card, G.M. The Functional Organization of Descending Sensory-Motor Pathways in Drosophila. eLife 2018, 7, e34272. [Google Scholar] [CrossRef]
- Xu, L.; He, J.; Kaiser, A.; Gräber, N.; Schläger, L.; Ritze, Y.; Scholz, H. A Single Pair of Serotonergic Neurons Counteracts Serotonergic Inhibition of Ethanol Attraction in Drosophila. PLoS ONE 2016, 11, e0167518. [Google Scholar] [CrossRef]
- Nichols, C.D. 5-HT2 Receptors in Drosophila Are Expressed in the Brain and Modulate Aspects of Circadian Behaviors. Dev. Neurobiol. 2007, 67, 752–763. [Google Scholar] [CrossRef]
- Sizemore, T.R.; Hurley, L.M.; Dacks, A.M. Serotonergic Modulation across Sensory Modalities. J. Neurophysiol. 2020, 123, 2406–2425. [Google Scholar] [CrossRef]
- Marin, E.C.; Büld, L.; Theiss, M.; Sarkissian, T.; Roberts, R.J.V.; Turnbull, R.; Tamimi, I.F.M.; Pleijzier, M.W.; Laursen, W.J.; Drummond, N.; et al. Connectomics Analysis Reveals First-, Second-, and Third-Order Thermosensory and Hygrosensory Neurons in the Adult Drosophila Brain. Curr. Biol. 2020, 30, 3167–3182.e4. [Google Scholar] [CrossRef]
- Putz, G.; Heisenberg, M. Memories in Drosophila Heat-Box Learning. Learn. Mem. 2002, 9, 349–359. [Google Scholar] [CrossRef]
- Wu, M.; Nern, A.; Williamson, W.R.; Morimoto, M.M.; Reiser, M.B.; Card, G.M.; Rubin, G.M. Visual Projection Neurons in the Drosophila Lobula Link Feature Detection to Distinct Behavioral Programs. eLife 2016, 5, e21022. [Google Scholar] [CrossRef]
- Keleş, M.F.; Frye, M.A. Object-Detecting Neurons in Drosophila. Curr. Biol. 2017, 27, 680–687. [Google Scholar] [CrossRef]
- Foucaud, J.; Philippe, A.-S.; Moreno, C.; Mery, F. A Genetic Polymorphism Affecting Reliance on Personal versus Public Information in a Spatial Learning Task in Drosophila melanogaster. Proc. R Soc. B Biol. Sci. 2013, 280, 20130588. [Google Scholar] [CrossRef]
- Foucaud, J.; Burns, J.G.; Mery, F. Use of Spatial Information and Search Strategies in a Water Maze Analog in Drosophila melanogaster. PLoS ONE 2010, 5, e15231. [Google Scholar] [CrossRef]
- Ofstad, T.A.; Zuker, C.S.; Reiser, M.B. Visual Place Learning in Drosophila melanogaster. Nature 2011, 474, 204–207. [Google Scholar] [CrossRef]
- Melnattur, K.; Kirszenblat, L.; Morgan, E.; Militchin, V.; Sakran, B.; English, D.; Patel, R.; Chan, D.; van Swinderen, B.; Shaw, P.J. A conserved role for sleep in supporting Spatial Learning in Drosophila. Sleep 2021, 44, zsaa197. [Google Scholar] [CrossRef]
- Omoto, J.J.; Nguyen, B.-C.M.; Kandimalla, P.; Lovick, J.K.; Donlea, J.M.; Hartenstein, V. Neuronal Constituents and Putative Interactions Within the Drosophila Ellipsoid Body Neuropil. Front. Neural Circuits 2018, 12, 103. [Google Scholar] [CrossRef]
- Kong, E.C.; Woo, K.; Li, H.; Lebestky, T.; Mayer, N.; Sniffen, M.R.; Heberlein, U.; Bainton, R.J.; Hirsh, J.; Wolf, F.W. A Pair of Dopamine Neurons Target the D1-Like Dopamine Receptor DopR in the Central Complex to Promote Ethanol-Stimulated Locomotion in Drosophila. PLoS ONE 2010, 5, e9954. [Google Scholar] [CrossRef]
- Monastirioti, M. Biogenic Amine Systems in the Fruit Fly Drosophila melanogaster. Microsc. Res. Techol. 1999, 45, 106–121. [Google Scholar] [CrossRef]
- Kahsai, L.; Winther, Å.M.E. Chemical Neuroanatomy of the Drosophila Central Complex: Distribution of Multiple Neuropeptides in Relation to Neurotransmitters. J. Comp. Neurol. 2011, 519, 290–315. [Google Scholar] [CrossRef]


| Cluster Number | Vallés and White, 1998 [53] | Aleksenyenko et al., 2010 [10] | Pooryasin and Fiala, 2015 [12] | 5-HT neurons | Trh-Gal4 > UAS-GFP |
|---|---|---|---|---|---|
| 1 | None | ALP | ALP | 6 ± 0 | 4 ± 1 |
| 2 | None | AMP | AMP | 2 ± 0 | 2 ± 0 |
| 3 | None | None | ADMP | 2 ± 0 | 2 ± 0 |
| 4 | LP2a, b | LP2 | LP | 24 ± 3 | 18 ± 3 |
| 5 | SE1 and SE2 | SE1 and SE2 | SEL | 10 ± 2 | 11 ± 1 |
| 6 | SE3 | SE3 | SEM | 10 ± 3 | 9 ± 3 |
| 7 | SE3 | SE3 | SEM | ||
| 8 | LP1 | PLP | PLP | 4 ± 0 | 4 ± 0 |
| 9 | SP1 | PMP | PMPD | 6 ± 0 | 6 ± 0 |
| 10 | SP2 | PMP | PMPM | 13 ± 2 | 12 ± 3 |
| 11 | IP | PMP | PMPV | 14 ± 3 | 8 ± 3 |
| Σ 5HT neurons | 91 ± 13 | 76 ± 14 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gajardo, I.; Guerra, S.; Campusano, J.M. Navigating Like a Fly: Drosophila melanogaster as a Model to Explore the Contribution of Serotonergic Neurotransmission to Spatial Navigation. Int. J. Mol. Sci. 2023, 24, 4407. https://doi.org/10.3390/ijms24054407
Gajardo I, Guerra S, Campusano JM. Navigating Like a Fly: Drosophila melanogaster as a Model to Explore the Contribution of Serotonergic Neurotransmission to Spatial Navigation. International Journal of Molecular Sciences. 2023; 24(5):4407. https://doi.org/10.3390/ijms24054407
Chicago/Turabian StyleGajardo, Ivana, Simón Guerra, and Jorge M. Campusano. 2023. "Navigating Like a Fly: Drosophila melanogaster as a Model to Explore the Contribution of Serotonergic Neurotransmission to Spatial Navigation" International Journal of Molecular Sciences 24, no. 5: 4407. https://doi.org/10.3390/ijms24054407
APA StyleGajardo, I., Guerra, S., & Campusano, J. M. (2023). Navigating Like a Fly: Drosophila melanogaster as a Model to Explore the Contribution of Serotonergic Neurotransmission to Spatial Navigation. International Journal of Molecular Sciences, 24(5), 4407. https://doi.org/10.3390/ijms24054407






