Treatment of Acute Respiratory Distress Syndrome Caused by COVID-19 with Human Umbilical Cord Mesenchymal Stem Cells
Abstract
1. Introduction
2. Results
2.1. Basic Characteristics of the Patients with COVID-19
2.2. The Main Safety Results
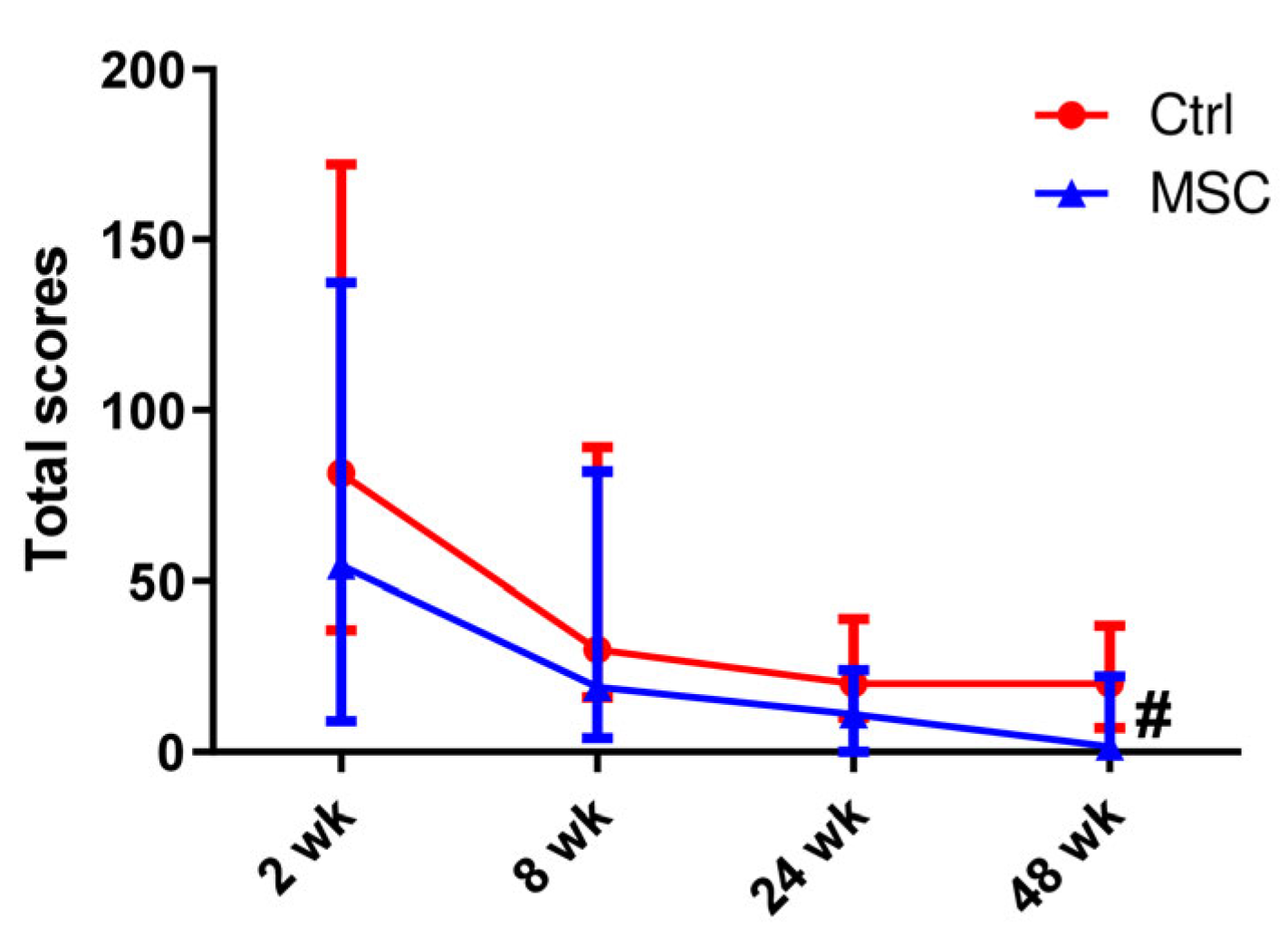
2.3. Computed Tomography of the Lung
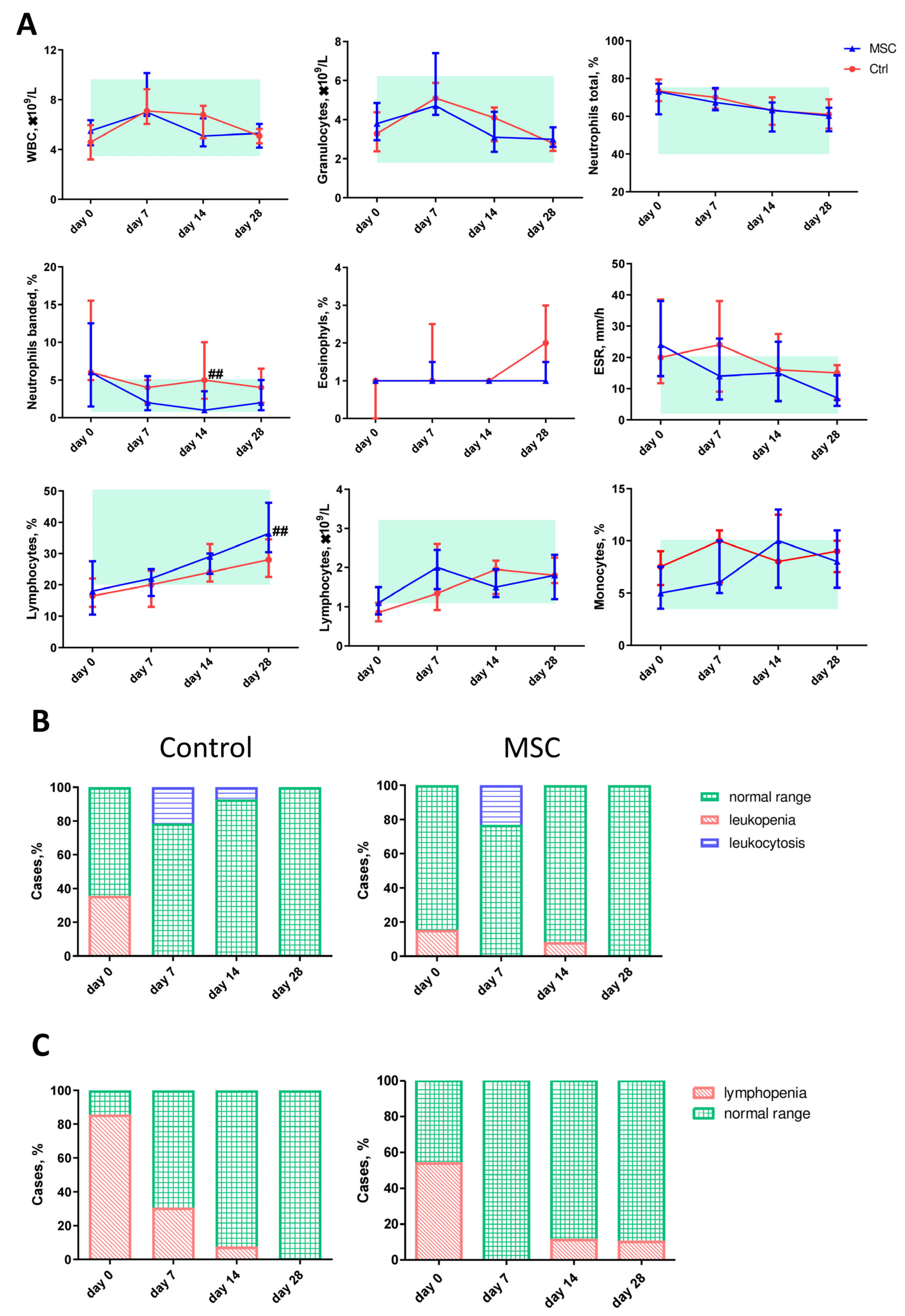
2.4. The Dynamic Profile of Hematological Parameters in Patients with COVID-19
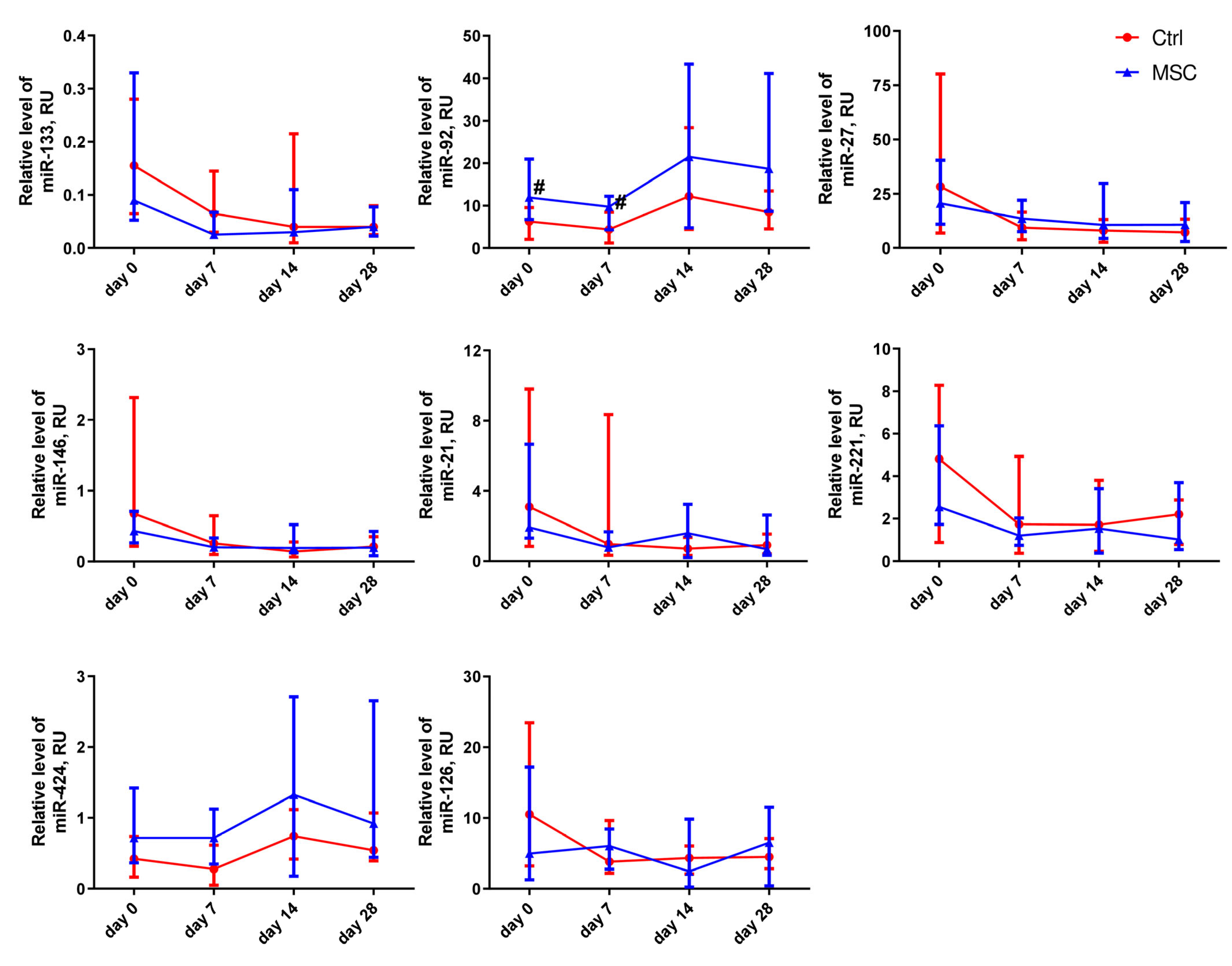
2.5. The Dynamic Changes in Cytokine and miRNA Levels in the Plasma of Patients with COVID-19
2.6. Correlation between Hematological Parameters, Cytokines, and miRNA in the MSC Group
2.7. Correlation between Hematological Parameters, Cytokines, miRNA, and CT in the MSC Group
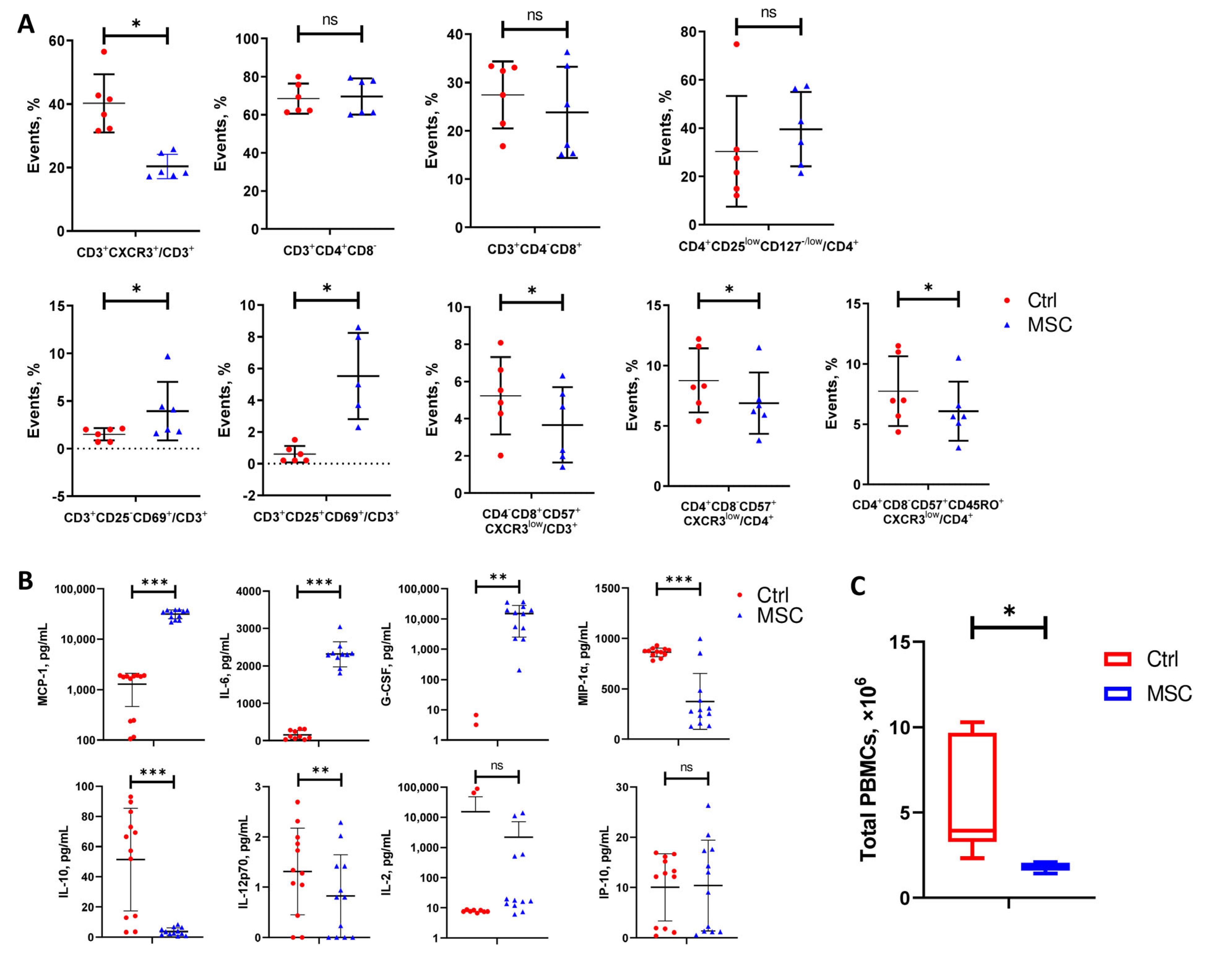
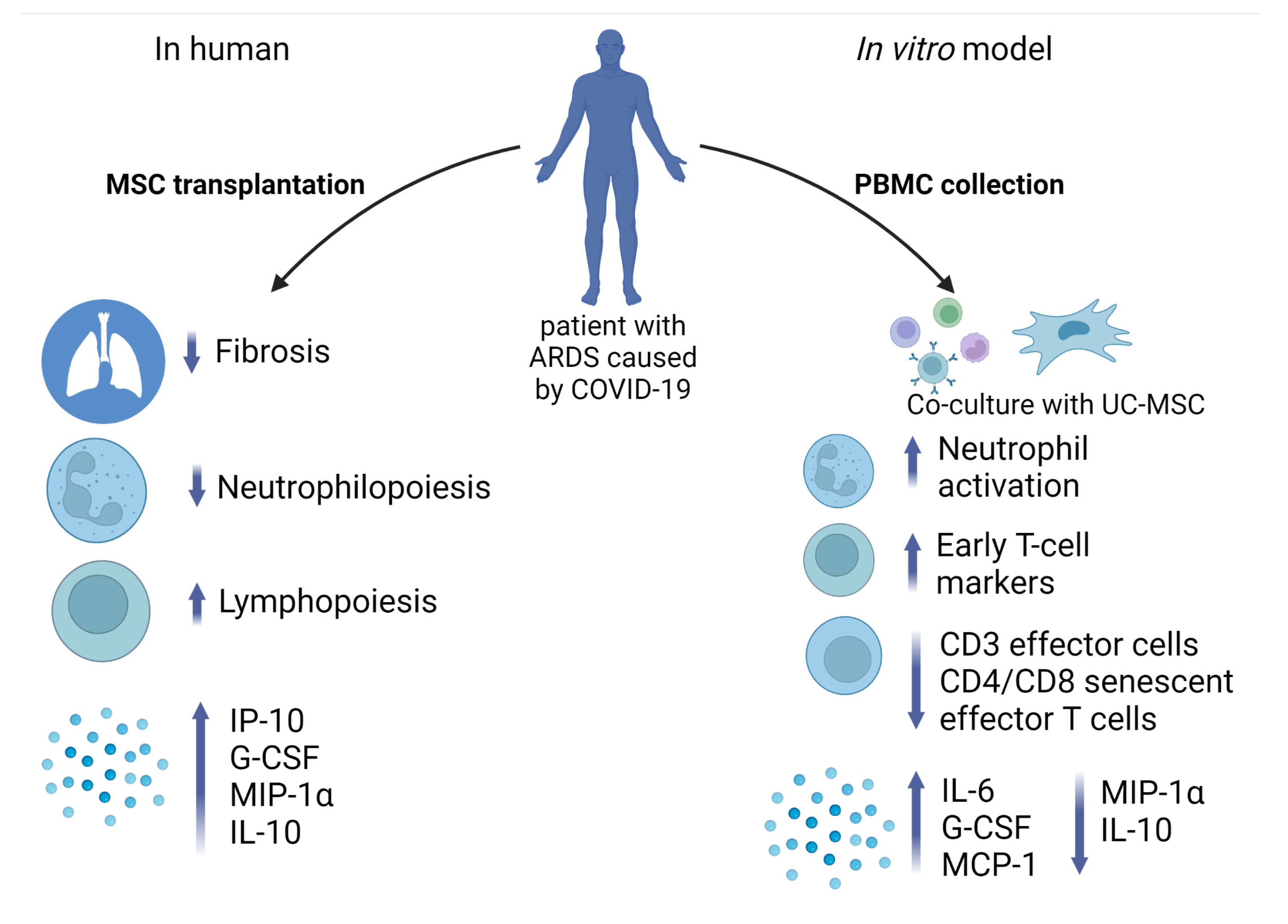
2.8. Effect of UC-MSC on PBMC In Vitro: Transcriptome, Flow Cytometry, and ELISA
3. Discussion
4. Materials and Methods
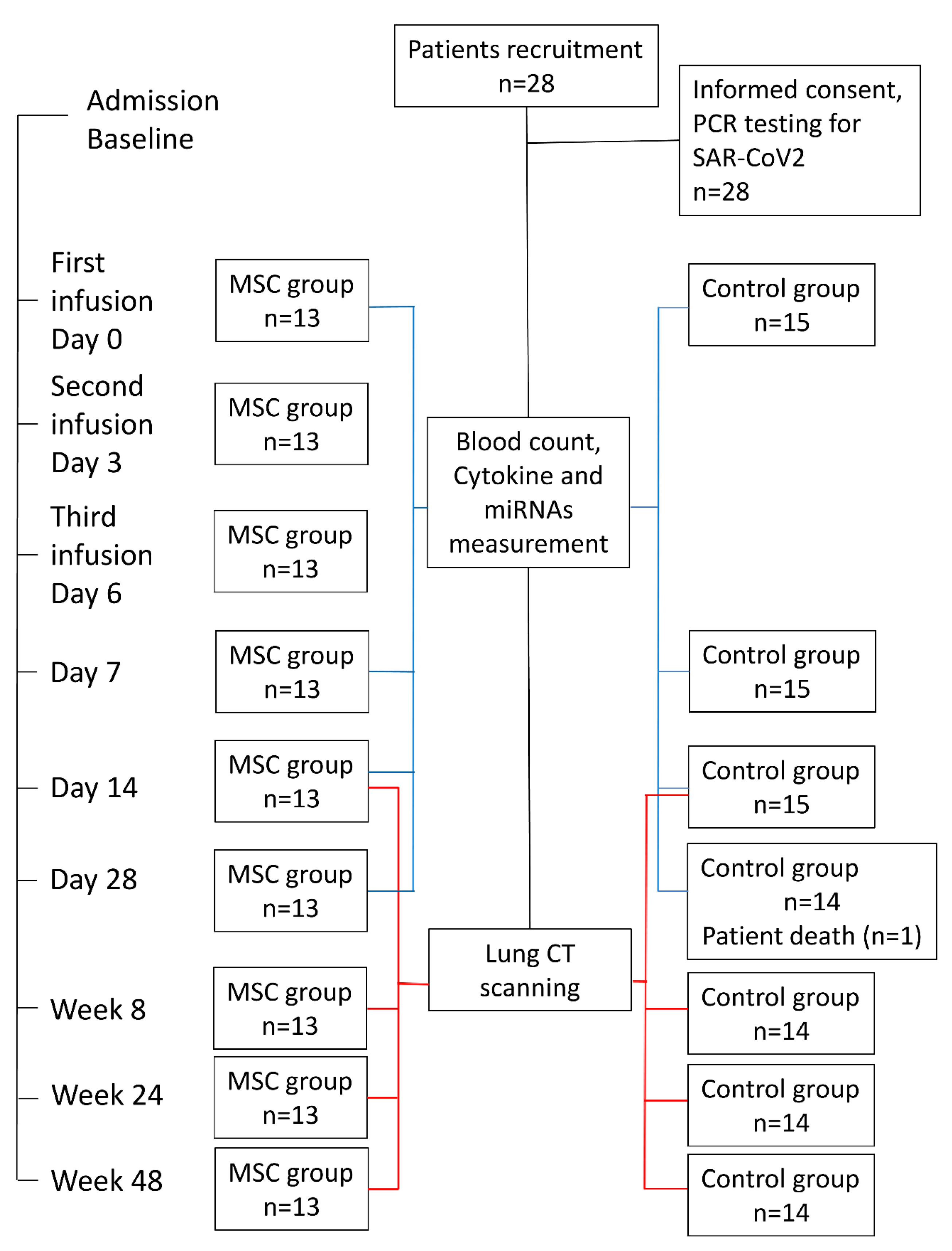
4.1. Participants and Study Design
4.2. Respiratory Pathogen Detection
4.3. UC-MSC Preparation
4.4. UC-MSC Transplantation
4.5. CT Evaluation and Scoring
4.6. Blood Collection
4.7. Cytokine Measurement
4.8. miRNA Expression
4.9. Isolation of PBMC and Co-Culture with UC-MSC
4.10. RNA Isolation, RNA-seq, and Bioinformatics Analysis
4.11. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Rodriguez-Morales, A.J.; Cardona-Ospina, J.A.; Gutiérrez-Ocampo, E.; Villamizar-Peña, R.; Holguin-Rivera, Y.; Escalera-Antezana, J.P.; Alvarado-Arnez, L.E.; Bonilla-Aldana, D.K.; Franco-Paredes, C.; Henao-Martinez, A.F.; et al. Clinical, laboratory and imaging features of COVID-19: A systematic review and meta-analysis. Travel Med. Infect. Dis. 2020, 34, 101623. Available online: https://pubmed.ncbi.nlm.nih.gov/32179124/ (accessed on 17 July 2021). [CrossRef] [PubMed]
- Zhang, J.-Y.; Wang, X.-M.; Xing, X.; Xu, Z.; Zhang, C.; Song, J.-W.; Fan, X.; Xia, P.; Fu, J.L.; Wang, S.Y.; et al. Single-cell landscape of immunological responses in patients with COVID-19. Nat. Immunol. 2020, 21, 1107–1118. Available online: https://pubmed.ncbi.nlm.nih.gov/32788748/ (accessed on 23 June 2022). [CrossRef] [PubMed]
- Mallis, P.; Michalopoulos, E.; Chatzistamatiou, T.; Stavropoulos-Giokas, C. Mesenchymal stromal cells as potential immunomodulatory players in severe acute respiratory distress syndrome induced by SARS-CoV-2 infection. World J. Stem Cells 2020, 12, 731–751. Available online: https://pubmed.ncbi.nlm.nih.gov/32952855/ (accessed on 7 February 2023). [CrossRef]
- Tufan, A.; Güler, A.A.; Matucci-Cerinic, M. COVID-19, immune system response, hyperinflammation and repurposing antirheumatic drugs. Turk. J. Med. Sci. 2020, 50, 620–632. Available online: https://pubmed.ncbi.nlm.nih.gov/32299202/ (accessed on 23 June 2022). [CrossRef]
- Schulte-Schrepping, J.; Reusch, N.; Paclik, D.; Baßler, K.; Schlickeiser, S.; Zhang, B.; Krämer, B.; Krammer, T.; Brumhard, S.; Bonaguro, L.; et al. Severe COVID-19 Is Marked by a Dysregulated Myeloid Cell Compartment. Cell 2020, 182, 1419–1440.e23. Available online: https://pubmed.ncbi.nlm.nih.gov/32810438/ (accessed on 23 June 2022). [CrossRef]
- Rajamanickam, A.; Kumar, N.P.; Pandiarajan, A.N.; Selvaraj, N.; Munisankar, S.; Renji, R.M.; Venkatramani, V.; Murhekar, M.; Thangaraj, J.W.V.; Kumar, M.; et al. Dynamic alterations in monocyte numbers, subset frequencies and activation markers in acute and convalescent COVID-19 individuals. Sci. Rep. 2021, 11, 20254. Available online: https://www.nature.com/articles/s41598-021-99705-y (accessed on 23 June 2022). [CrossRef]
- Geraylow, K.R.; Hemmati, R.; Kadkhoda, S.; Ghafouri-Fard, S. miRNA expression in COVID-19. Gene Rep. 2022, 28, 101641. [Google Scholar] [CrossRef]
- Wang, J.; Huang, R.; Xu, Q.; Zheng, G.; Qiu, G.; Ge, M.; Shu, Q.; Xu, J. Mesenchymal Stem Cell-Derived Extracellular Vesicles Alleviate Acute Lung Injury Via Transfer of miR-27a-3p. Crit. Care Med. 2020, 48, E599–E610. Available online: https://pubmed.ncbi.nlm.nih.gov/32317602/ (accessed on 23 June 2022). [CrossRef]
- Liang, Y.; Fang, D.; Gao, X.; Deng, X.; Chen, N.; Wu, J.; Zeng, M.; Luo, M. Circulating microRNAs as emerging regulators of COVID-19. Theranostics 2023, 13, 125–147. Available online: https://www.thno.org://creativecommons.org/licenses/by/4.0/ (accessed on 7 February 2023). [CrossRef]
- De Melo, P.; Pineros Alvarez, A.R.; Ye, X.; Blackman, A.; Alves-Filho, J.C.; Medeiros, A.I.; Rathmell, J.; Pua, H.; Serezani, C.H. Macrophage-Derived MicroRNA-21 Drives Overwhelming Glycolytic and Inflammatory Response during Sepsis via Repression of the PGE 2/IL-10 Axis. J. Immunol. 2021, 207, 902–912. Available online: https://pubmed.ncbi.nlm.nih.gov/34301845/ (accessed on 23 June 2022). [CrossRef]
- Sengupta, V.; Sengupta, S.; Lazo, A.; Woods, P.; Nolan, A.; Bremer, N. Exosomes Derived from Bone Marrow Mesenchymal Stem Cells as Treatment for Severe COVID-19. Stem Cells Dev. 2020, 29, 747–754. Available online: https://pubmed.ncbi.nlm.nih.gov/32380908/ (accessed on 23 June 2022). [CrossRef]
- Shu, L.; Niu, C.; Li, R.; Huang, T.; Wang, Y.; Huang, M.; Ningfei, J.; Zheng, Y.; Chen, X.; Shi, L.; et al. Treatment of severe COVID-19 with human umbilical cord mesenchymal stem cells. Stem Cell Res. Ther. 2020, 11, 361. Available online: https://pubmed.ncbi.nlm.nih.gov/32811531/ (accessed on 23 June 2022). [CrossRef] [PubMed]
- Lanzoni, G.; Linetsky, E.; Correa, D.; Cayetano, S.M.; Alvarez, R.A.; Kouroupis, D.; Alvarez Gil, A.; Poggioli, R.; Ruiz, P.; Marttos, A.C.; et al. Umbilical cord mesenchymal stem cells for COVID-19 acute respiratory distress syndrome: A double-blind, phase 1/2a, randomized controlled trial. Stem Cells Transl. Med. 2021, 10, 660–673. Available online: https://pubmed.ncbi.nlm.nih.gov/33400390/ (accessed on 23 June 2022). [CrossRef] [PubMed]
- Meng, F.; Xu, R.; Wang, S.; Xu, Z.; Zhang, C.; Li, Y.; Yang, T.; Shi, L.; Fu, J.; Jiang, T.; et al. Human umbilical cord-derived mesenchymal stem cell therapy in patients with COVID-19: A phase 1 clinical trial. Signal Transduct. Target. Ther. 2020, 5, 172. Available online: https://www.nature.com/articles/s41392-020-00286-5 (accessed on 23 June 2022). [CrossRef] [PubMed]
- Tang, L.; Jiang, Y.; Zhu, M.; Chen, L.; Zhou, X.; Zhou, C.; Ye, P.; Chen, X.; Wang, B.; Xu, Z.; et al. Clinical study using mesenchymal stem cells for the treatment of patients with severe COVID-19. Front. Med. 2020, 14, 664–673. [Google Scholar] [CrossRef]
- Feng, Y.; Huang, J.; Wu, J.; Xu, Y.; Chen, B.; Jiang, L.; Xiang, H.; Peng, Z.; Wang, X. Safety and feasibility of umbilical cord mesenchymal stem cells in patients with COVID-19 pneumonia: A pilot study. Cell Prolif. 2020, 53, e12947. Available online: https://pubmed.ncbi.nlm.nih.gov/33205469/ (accessed on 23 June 2022). [CrossRef]
- Gorman, E.; Shankar-Hari, M.; Hopkins, P.; Tunnicliffe, W.S.; Perkins, G.D.; Silversides, J.; McGuigan, P.; Jackson, C.; Boyle, R.; McFerran, J.; et al. Repair of Acute Respiratory Distress Syndrome by Stromal Cell Administration in COVID-19 (REALIST-COVID-19): A structured summary of a study protocol for a randomised, controlled trial. Trials 2020, 21, 462. Available online: https://pubmed.ncbi.nlm.nih.gov/32493473/ (accessed on 23 June 2022). [CrossRef]
- Kouroupis, D.; Lanzoni, G.; Linetsky, E.; Cayetano, S.M.; Metalonis, S.W.; Leñero, C.; Stone, L.D.; Ruiz, P.; Correa, D.; Ricordi, C. Umbilical Cord-derived Mesenchymal Stem Cells modulate TNF and soluble TNF Receptor 2 (sTNFR2) in COVID-19 ARDS patients. Eur. Rev. Med. Pharmacol. Sci. 2021, 25, 4435–4438. Available online: https://pubmed.ncbi.nlm.nih.gov/34227081/ (accessed on 23 June 2022).
- Saleh, M.; Vaezi, A.A.; Aliannejad, R.; Sohrabpour, A.A.; Kiaei, S.Z.F.; Shadnoush, M.; Siavashi, V.; Aghaghazvini, L.; Khoundabi, B.; Abdoli, S.; et al. Cell therapy in patients with COVID-19 using Wharton’s jelly mesenchymal stem cells: A phase 1 clinical trial. Stem Cell Res. Ther. 2021, 25, 4435–4438. Available online: https://pubmed.ncbi.nlm.nih.gov/34271988/ (accessed on 23 June 2022). [CrossRef] [PubMed]
- Shi, L.; Huang, H.; Lu, X.; Yan, X.; Jiang, X.; Xu, R.; Wang, S.; Zhang, C.; Yuan, X.; Xu, Z.; et al. Effect of human umbilical cord-derived mesenchymal stem cells on lung damage in severe COVID-19 patients: A randomized, double-blind, placebo-controlled phase 2 trial. Signal Transduct. Target. Ther. 2021, 6, 58. Available online: https://www.nature.com/articles/s41392-021-00488-5 (accessed on 23 June 2022). [CrossRef]
- Adas, G.; Cukurova, Z.; Yasar, K.K.; Yilmaz, R.; Isiksacan, N.; Kasapoglu, P.; Yesilbag, Z.; Koyuncu, I.D.; Karaoz, E. The Systematic Effect of Mesenchymal Stem Cell Therapy in Critical COVID-19 Patients: A Prospective Double Controlled Trial. Cell Transpl. 2021, 30, 09636897211024942. Available online: https://pubmed.ncbi.nlm.nih.gov/34180719/ (accessed on 23 June 2022).
- Hashemian, S.M.R.; Aliannejad, R.; Zarrabi, M.; Soleimani, M.; Vosough, M.; Hosseini, S.E.; Hossieni, H.; Keshel, S.H.; Naderpour, Z.; Hajizadeh-Saffar, E.; et al. Mesenchymal stem cells derived from perinatal tissues for treatment of critically ill COVID-19-induced ARDS patients: A case series. Stem Cell Res. Ther. 2021, 12, 91. Available online: https://pubmed.ncbi.nlm.nih.gov/33514427/ (accessed on 23 June 2022). [CrossRef] [PubMed]
- Xu, X.; Jiang, W.; Chen, L.; Xu, Z.; Zhang, Q.; Zhu, M.; Ye, P.; Li, H.; Yu, L.; Zhou, X.; et al. Evaluation of the safety and efficacy of using human menstrual blood-derived mesenchymal stromal cells in treating severe and critically ill COVID-19 patients: An exploratory clinical trial. Clin. Transl. Med. 2021, 11, e297. Available online: https://pubmed.ncbi.nlm.nih.gov/33634996/ (accessed on 23 June 2022). [CrossRef] [PubMed]
- Andrzejewska, A.; Lukomska, B.; Janowski, M. Concise Review: Mesenchymal Stem Cells: From Roots to Boost. Stem Cells 2019, 37, 855–864. Available online: https://pubmed.ncbi.nlm.nih.gov/30977255/ (accessed on 23 June 2022). [CrossRef]
- Sharma, A.; Chakraborty, A.; Jaganathan, B.G. Review of the potential of mesenchymal stem cells for the treatment of infectious diseases. World J. Stem Cells 2021, 13, 568–593. Available online: https://pubmed.ncbi.nlm.nih.gov/34249228/. (accessed on 23 June 2022). [CrossRef]
- Akkoc, T. COVID-19 and Mesenchymal Stem Cell Treatment; Mystery or Not. Adv. Exp. Med. Biol. 2020, 1298, 167–176. Available online: https://pubmed.ncbi.nlm.nih.gov/32648245/ (accessed on 23 June 2022).
- Pinky; Gupta, S.; Krishnakumar, V.; Sharma, Y.; AK, D.; Mohanty, S. Mesenchymal Stem Cell Derived Exosomes: A Nano Platform for Therapeutics and Drug Delivery in Combating COVID-19. Stem Cell Rev. Rep. 2021, 17, 33–43. Available online: https://pubmed.ncbi.nlm.nih.gov/32661867/ (accessed on 23 June 2022). [CrossRef]
- Mallis, P.; Michalopoulos, E.; Chatzistamatiou, T.; Giokas, C.S. Interplay between mesenchymal stromal cells and immune system: Clinical applications in immune-related diseases. Explor. Immunol. 2021, 1, 112–139. Available online: https://www.explorationpub.com/Journals/ei/Article/100310 (accessed on 7 February 2023). [CrossRef]
- Le Blanc, K.; Ringdén, O. Immunobiology of Human Mesenchymal Stem Cells and Future Use in Hematopoietic Stem Cell Transplantation. Biol. Blood Marrow Transplant. 2005, 11, 321–334. [Google Scholar] [CrossRef]
- Lee, M.; Jeong, S.Y.; Ha, J.; Kim, M.; Jin, H.J.; Kwon, S.J.; Chang, J.W.; Choi, S.J.; Oh, W.; Yang, Y.S.; et al. Low immunogenicity of allogeneic human umbilical cord blood-derived mesenchymal stem cells in vitro and in vivo. Biochem. Biophys. Res. Commun. 2014, 446, 983–989. [Google Scholar] [CrossRef]
- Abbaspanah, B.; Reyhani, S.; Mousavi, S.H. Applications of Umbilical Cord Derived Mesenchymal Stem Cells in Autoimmune and Immunological Disorders: From Literature to Clinical Practice. Curr. Stem Cell Res. Ther. 2020, 16, 454–464. [Google Scholar] [CrossRef]
- Mebarki, M.; Abadie, C.; Larghero, J.; Cras, A. Human umbilical cord-derived mesenchymal stem/stromal cells: A promising candidate for the development of advanced therapy medicinal products. Stem Cell Res. Ther. 2021, 12, 152. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7907784/ (accessed on 26 September 2022). [CrossRef]
- Kirkham, A.M.; Bailey, A.J.M.; Shorr, R.; Lalu, M.M.; Fergusson, D.A.; Allan, D.S. Systematic review and meta-analysis of randomized controlled trials of mesenchymal stromal cells to treat coronavirus disease 2019: Is it too late? Cytotherapy 2022, 25, 341. [Google Scholar] [CrossRef]
- Xiong, J.; Chen, L.; Zhang, L.; Bao, L.; Shi, Y. Mesenchymal Stromal Cell-Based Therapy: A Promising Approach for Severe COVID-19. Cell Transplant. 2021, 30, 0963689721995455. [Google Scholar] [CrossRef]
- Chaudhary, J.K.; Saini, D.; Chaudhary, P.K.; Maurya, A.; Verma, G.K.; Gupta, A.K.; Roshan, R.; Vats, T.K.; Garg, N.; Yadav, D.; et al. Exploring the Immunomodulatory Aspect of Mesenchymal Stem Cells for Treatment of Severe Coronavirus Disease 19. Cells 2022, 11, 2175. [Google Scholar] [CrossRef]
- Zhu, R.; Yan, T.; Feng, Y.; Liu, Y.; Cao, H.; Peng, G.; Yang, Y.; Xu, Z.; Liu, J.; Hou, W.; et al. Mesenchymal stem cell treatment improves outcome of COVID-19 patients via multiple immunomodulatory mechanisms. Cell Res. 2021, 31, 1244–1262. [Google Scholar] [CrossRef]
- Shi, L.; Yuan, X.; Yao, W.; Wang, S.; Zhang, C.; Zhang, B.; Song, J.; Huang, L.; Xu, Z.; Fu, J.L.; et al. Human mesenchymal stem cells treatment for severe COVID-19: 1-year follow-up results of a randomized, double-blind, placebo-controlled trial. EBioMedicine 2022, 75, 103789. [Google Scholar] [CrossRef] [PubMed]
- Armitage, J.D.; Tan, D.B.A.; Sturm, M.; Moodley, Y.P. Transcriptional Profiling of Circulating Mononuclear Cells from Patients with Chronic Obstructive Pulmonary Disease receiving Mesenchymal Stromal Cell Infusions. Stem Cells Transl. Med. 2021, 10, 1470–1481. Available online: https://academic.oup.com/stcltm/article/10/11/1470/6517846 (accessed on 5 February 2023). [CrossRef]
- Hazeldine, J.; Lord, J.M. Neutrophils and COVID-19: Active Participants and Rational Therapeutic Targets. Front. Immunol. 2021, 12, 680134. [Google Scholar] [CrossRef]
- Morrissey, S.M.; Geller, A.E.; Hu, X.; Tieri, D.; Cooke, E.A.; Ding, C.; Woeste, M.; Zhang, H.; Cavallazzi, R.; Clifford, S.P.; et al. Emergence of Low-density Inflammatory Neutrophils Correlates with Hypercoagulable State and Disease Severity in COVID-19 Patients. MedRxiv 2020. [Google Scholar] [CrossRef]
- Rebelatto, C.L.K.; Senegaglia, A.C.; Franck, C.L.; Daga, D.R.; Shigunov, P.; Stimamiglio, M.A.; Marsaro, D.B.; Schaidt, B.; Micosky, A.; de Azambuja, A.P.; et al. Safety and long-term improvement of mesenchymal stromal cell infusion in critically COVID-19 patients: A randomized clinical trial. Stem Cell Res. Ther. 2022, 13, 122. Available online: https://stemcellres.biomedcentral.com/articles/10.1186/s13287-022-02796-1 (accessed on 23 June 2022). [CrossRef]
- Channappanavar, R.; Perlman, S. Pathogenic human coronavirus infections: Causes and consequences of cytokine storm and immunopathology. Semin. Immunopathol. 2017, 39, 529. [Google Scholar] [CrossRef]
- Irmak, D.K.; Darıcı, H.; Karaöz, E. Stem Cell Based Therapy Option in COVID-19: Is It Really Promising? Aging Dis. 2020, 11, 1174. [Google Scholar] [CrossRef] [PubMed]
- Mardani, R.; Namavar, M.; Ghorbi, E.; Shoja, Z.; Zali, F.; Kaghazian, H.; Aghasadeghi, M.R.; Sadeghi, S.A.; Sabeti, S.; Darazam, I.A.; et al. Association between serum inflammatory parameters and the disease severity in COVID-19 patients. J. Clin. Lab. Anal. 2022, 36, e24162. Available online: https://onlinelibrary.wiley.com/doi/10.1002/jcla.24162 (accessed on 23 June 2022). [CrossRef] [PubMed]
- Leng, Z.; Zhu, R.; Hou, W.; Feng, Y.; Yang, Y.; Han, Q.; Shan, G.; Meng, F.; Du, D.; Wang, S.; et al. Transplantation of ACE2- Mesenchymal Stem Cells Improves the Outcome of Patients with COVID-19 Pneumonia. Aging Dis. 2020, 11, 216. [Google Scholar] [CrossRef]
- Alay, H.; Laloglu, E. The role of angiopoietin-2 and surfactant protein-D levels in SARS-CoV-2-related lung injury: A prospective, observational, cohort study. J. Med. Virol. 2021, 93, 6008–6015. Available online: https://onlinelibrary.wiley.com/doi/10.1002/jmv.27184 (accessed on 25 June 2022). [CrossRef] [PubMed]
- Lim, A.; Radujkovic, A.; Weigand, M.A.; Merle, U. Soluble receptor for advanced glycation end products (sRAGE) as a biomarker of COVID-19 disease severity and indicator of the need for mechanical ventilation, ARDS and mortality. Ann. Intensive Care 2021, 11, 1–13. [Google Scholar] [CrossRef]
- Carcaterra, M.; Caruso, C. Alveolar epithelial cell type II as main target of SARS-CoV-2 virus and COVID-19 development via NF-Kb pathway deregulation: A physio-pathological theory. Med. Hypotheses 2021, 146, 110412. [Google Scholar] [CrossRef]
- Mokra, D.; Mikolka, P.; Kosutova, P.; Mokry, J. Corticosteroids in Acute Lung Injury: The Dilemma Continues. Int. J. MolSci. 2019, 20, 4765. Available online: https://www.mdpi.com/1422-0067/20/19/4765 (accessed on 25 June 2022). [CrossRef]
- Roy, D.; Ramasamy, R.; Schmidt, A.M. Journey to a Receptor for Advanced Glycation End Products Connection in Severe Acute Respiratory Syndrome Coronavirus 2 Infection with Stops along the Way in the Lung, Heart, Blood Vessels, and Adipose Tissue. Arterioscler. Thromb. Vasc. Biol. 2021, 41, 614–627. [Google Scholar] [CrossRef]
- Kimura, D.; Saravia, J.; Rovnaghi, C.R.; Meduri, G.U.; Schwingshackl, A.; Cormier, S.A.; Anand, K.J. Plasma biomarker analysis in pediatric ARDS: Generating future framework from a pilot randomized control trial of methylprednisolone: A framework for identifying plasma biomarkers related to clinical outcomes in pediatric ARDS. Front. Pediatr. 2016, 4, 31. [Google Scholar] [CrossRef]
- Wick, K.D.; Leligdowicz, A.; Zhuo, H.; Ware, L.B.; Matthay, M.A. Mesenchymal stromal cells reduce evidence of lung injury in patients with ARDS. JCI Insight 2021, 6, e87489. [Google Scholar] [CrossRef]
- Yang, Y.; Shen, C.; Li, J.; Yuan, J.; Wei, J.; Huang, F.; Wang, F.; Li, G.; Li, Y.; Xing, L.; et al. Plasma IP-10 and MCP-3 levels are highly associated with disease severity and predict the progression of COVID-19. J. Allergy Clin. Immunol. 2020, 146, 119–127.e4. [Google Scholar] [CrossRef]
- Wang, G.L.; Gao, H.X.; Wang, Y.L.; Wei, X.; Liu, Y.Z.; Lu, J.H.; Li, L.; Wang, H.B.; Zhao, L.; Rong, Y.X.; et al. Serum IP-10 and IL-7 levels are associated with disease severity of coronavirus disease 2019. Cytokine 2021, 142, 155500. [Google Scholar] [CrossRef]
- Kleymenov, D.A.; Bykonia, E.N.; Popova, L.I.; Mazunina, E.P.; Gushchin, V.A.; Kolobukhina, L.V.; Burgasova, O.A.; Kruzhkova, I.S.; Kuznetsova, N.A.; Shidlovskaya, E.V.; et al. A Deep Look Into COVID-19 Severity Through Dynamic Changes in Blood Cytokine Levels. Front. Immunol. 2021, 12, 4508. Available online: https://www.frontiersin.org/articles/10.3389/fimmu.2021.771609/full (accessed on 25 June 2022). [CrossRef]
- Meizlish, M.L.; Pine, A.B.; Bishai, J.D.; Goshua, G.; Nadelmann, E.R.; Simonov, M.; Chang, C.H.; Zhang, H.; Shallow, M.; Bahel, P.; et al. A neutrophil activation signature predicts critical illness and mortality in COVID-19. Blood Adv. 2021, 5, 1164. [Google Scholar] [CrossRef]
- Hasanvand, A. COVID-19 and the role of cytokines in this disease. Inflammopharmacology 2022, 30, 789–798. Available online: https://link.springer.com/article/10.1007/s10787-022-00992-2 (accessed on 15 September 2022). [CrossRef]
- Lu, L.; Zhang, H.; Dauphars, D.J.; He, Y.W. A Potential Role of Interleukin 10 in COVID-19 Pathogenesis. Trends Immunol. 2021, 42, 3–5. Available online: https://pubmed.ncbi.nlm.nih.gov/33214057/ (accessed on 26 September 2022). [CrossRef]
- Islam, H.; Chamberlain, T.C.; Mui, A.L.; Little, J.P. Elevated Interleukin-10 Levels in COVID-19: Potentiation of Pro-Inflammatory Responses or Impaired Anti-Inflammatory Action? Front. Immunol. 2021, 12, 677008. Available online: https://pubmed.ncbi.nlm.nih.gov/34234779/ (accessed on 26 September 2022). [CrossRef]
- Alshammary, A.F.; Alsughayyir, J.M.; Alharbi, K.K.; Al-Sulaiman, A.M.; Alshammary, H.F.; Alshammary, H.F. T-Cell Subsets and Interleukin-10 Levels Are Predictors of Severity and Mortality in COVID-19: A Systematic Review and Meta-Analysis. Front. Med. 2022, 9, 1147. [Google Scholar] [CrossRef]
- Zhang, M.; Yan, X.; Shi, M.; Li, R.; Pi, Z.; Ren, X.; Wang, Y.; Yan, S.; Wang, Y.; Jin, Y.; et al. Safety and efficiency of stem cell therapy for COVID-19: A systematic review and meta-analysis. Glob. Health Res. Policy 2022, 7, 19. Available online: https://ghrp.biomedcentral.com/articles/10.1186/s41256-022-00251-5 (accessed on 28 September 2022). [CrossRef]
- Wei, F.; Kong, D.; Li, T.; Li, A.; Tan, Y.; Fang, J.; Zhuang, X.; Lai, C.; Xu, W.; Dong, H.; et al. Efficacy and safety of umbilical cord mesenchymal stem cells for the treatment of patients with COVID-19. Clinics 2021, 76, e2604. [Google Scholar] [CrossRef]
- Wang, T.; Jiang, L.; Wei, X.; Dong, Z.; Liu, B.; Zhao, J.; Wang, L.; Xie, P.; Wang, Y.; Zhou, S. Inhibition of miR-221 alleviates LPS-induced acute lung injury via inactivation of SOCS1/NF-κB signaling pathway. Cell Cycle 2019, 18, 1893–1907. [Google Scholar] [CrossRef] [PubMed]
- Xu, P.; Xin, J.; Song, L.; Chen, Y.; Ma, J.; Liu, L.; Qi, Z.; Pan, X.; Zhou, S. Serum miR-133 as a Potential Biomarker in Acute Cerebral Infarction Patients. Clin. Lab. 2020, 66, 1923–1928. Available online: https://pubmed.ncbi.nlm.nih.gov/33073947/ (accessed on 19 September 2022). [CrossRef] [PubMed]
- Garg, A.; Seeliger, B.; Derda, A.A.; Xiao, K.; Gietz, A.; Scherf, K.; Sonnenschein, K.; Pink, I.; Hoeper, M.M.; Welte, T.; et al. Circulating cardiovascular microRNAs in critically ill COVID-19 patients. Eur. J. Heart Fail. 2021, 23, 468–475. Available online: https://onlinelibrary.wiley.com/doi/full/10.1002/ejhf.2096 (accessed on 25 July 2021). [CrossRef] [PubMed]
- Fogel, O.; BuggeTinggaard, A.; Fagny, M.; Sigrist, N.; Roche, E.; Leclere, L.; Deleuze, J.F.; Batteux, F.; Dougados, M.; Miceli-Richard, C.; et al. Deregulation of microRNA expression in monocytes and CD4+ T lymphocytes from patients with axial spondyloarthritis. Arthritis Res. Ther. 2019, 21, 51. Available online: https://arthritis-research.biomedcentral.com/articles/10.1186/s13075-019-1829-7 (accessed on 17 August 2022). [CrossRef]
- Zeng, Z.; Gong, H.; Li, Y.; Jie, K.; Ding, C.; Shao, Q.; Liu, F.; Zhan, Y.; Nie, C.; Zhu, W.; et al. Upregulation of miR-146a contributes to the suppression of inflammatory responses in LPS-induced acute lung injury. Exp. Lung Res. 2013, 39, 275–282. Available online: https://pubmed.ncbi.nlm.nih.gov/23848342/ (accessed on 25 July 2021). [CrossRef]
- Lambert, K.A.; Roff, A.N.; Panganiban, R.P.; Douglas, S.; Ishmael, F.T. MicroRNA-146a is induced by inflammatory stimuli in airway epithelial cells and augments the anti-inflammatory effects of glucocorticoids. PLoS ONE 2018, 13, e0205434. [Google Scholar] [CrossRef]
- Han, Y.; Li, Y.; Jiang, Y. The Prognostic Value of Plasma MicroRNA-155 and MicroRNA-146a Level in Severe Sepsis and Sepsis-Induced Acute Lung Injury Patients. Clin. Lab. 2016, 62, 2355–2360. [Google Scholar] [CrossRef]
- Wu, X.; Wu, C.; Gu, W.; Ji, H.; Zhu, L. Serum Exosomal MicroRNAs Predict Acute Respiratory Distress Syndrome Events in Patients with Severe Community-Acquired Pneumonia. Bio. Med. Res. Int. 2019, 2019, 1–11. [Google Scholar] [CrossRef]
- Zhang, S.; Hong, Y.; Liu, H.; Wang, Q.; Xu, J.; Zhang, Y.; Zhao, X.; Yao, Y.; Zhou, K.; Ding, X. miR-584 and miR-146 are candidate biomarkers for acute respiratory distress syndrome. Exp. Ther. Med. 2021, 21, 445. [Google Scholar] [CrossRef]
- Gonzalo-Calvo D de Benitez, I.D.; Pinilla, L.; Carratala, A.; Moncusi-Moix, A.N.N.A.; Gort-Paniello, C.; Molinero, M.; Gonzales, J.; Torres, G.; Bernal, M.; Pico, S. Circulating microRNA profiles predict the severity of COVID-19 in hospitalized patients. Transl. Res. 2021, 236, 147–159. [Google Scholar] [CrossRef]
- Bye, A.; Røsjø, H.; Nauman, J.; Silva, G.J.J.; Follestad, T.; Omland, T.; Wisløff, U. Circulating microRNAs predict future fatal myocardial infarction in healthy individuals-The HUNT study. J. Mol. Cell Cardiol. 2016, 97, 162–168. [Google Scholar] [CrossRef]
- Wang, M.; Liang, L.; Li, L.; Han, K.; Li, Q.; Peng, Y.; Peng, X.; Zeng, K. Increased miR-424-5p expression in peripheral blood mononuclear cells from patients with pemphigus. Mol. Med. Rep. 2017, 15, 3479–3484. [Google Scholar] [CrossRef]
- Fernández-Pato, A.; Virseda-Berdices, A.; Resino, S.; Ryan, P.; Martínez-González, O.; Pérez-García, F.; Martin-Vicente, M.; Valle-Millares, D.; Brochado-Kith, O.; Blancas, R.; et al. Plasma miRNA profile at COVID-19 onset predicts severity status and mortality. Emerg. Microbes Infect. 2022, 11, 676–688. [Google Scholar] [CrossRef]
- Shen, Y.; Lu, H.; Song, G. MiR-221-3p and miR-92a-3p enhances smoking-induced inflammation in COPD. J. Clin. Lab. Anal. 2021, 35, e23857. Available online: https://onlinelibrary.wiley.com/doi/10.1002/jcla.23857 (accessed on 26 June 2022). [CrossRef]
- Fayyad-Kazan, M.; Makki, R.; Skafi, N.; el Homsi, M.; Hamade, A.; el Majzoub, R.; Hamade, E.; Fayyad-Kazan, H.; Badran, B. Circulating miRNAs: Potential diagnostic role for coronavirus disease 2019 (COVID-19). Infect. Genet. Evol. 2021, 94, 105020. [Google Scholar] [CrossRef]
- Fu, L.; Zhu, P.; Qi, S.; Li, C.; Zhao, K. MicroRNA-92a antagonism attenuates lipopolysaccharide (LPS)-induced pulmonary inflammation and injury in mice through suppressing the PTEN/AKT/NF-κB signaling pathway. Biomed. Pharmacother. 2018, 107, 703–711. [Google Scholar] [CrossRef]
- Shang, F.; Wang, S.C.; Hsu, C.Y.; Miao, Y.; Martin, M.; Yin, Y.; Wu, C.C.; Wang, Y.T.; Wu, G.; Chien, S.; et al. MicroRNA-92a mediates endothelial dysfunction in CKD. J. Am. Soc. Nephrol. 2017, 28, 3251–3261. [Google Scholar] [CrossRef]
- Taghavi-Farahabadi, M.; Mahmoudi, M.; Rezaei, N.; Hashemi, S.M. Wharton’s Jelly Mesenchymal Stem Cells Exosomes and Conditioned Media Increased Neutrophil Lifespan and Phagocytosis Capacity. Immunol. Investig. 2021, 50, 1042–1057. [Google Scholar] [CrossRef]
- Mahmoudi, M.; Taghavi-Farahabadi, M.; Namaki, S.; Baghaei, K.; Rayzan, E.; Rezaei, N.; Hashemi, S.M. Exosomes derived from mesenchymal stem cells improved function and survival of neutrophils from severe congenital neutropenia patients in vitro. Hum. Immunol. 2019, 80, 990–998. [Google Scholar] [CrossRef]
- Li, X.; Zhang, Z.; Wang, Z.; Gutiérrez-Castrellón, P.; Shi, H. Cell deaths: Involvement in the pathogenesis and intervention therapy of COVID-19. Signal Transduct. Target. Ther. 2022, 7, 186. [Google Scholar] [CrossRef]
- Fan, X.L.; Zhang, Z.; Ma, C.Y.; Fu, Q.L. Mesenchymal stem cells for inflammatory airway disorders: Promises and challenges. Biosci. Rep. 2019, 39, BSR20182160. [Google Scholar] [CrossRef] [PubMed]
- Mallis, P.; Chatzistamatiou, T.; Dimou, Z.; Sarri, E.F.; Georgiou, E.; Salagianni, M.; Triantafyllia, V.; Andreakos, E.; Stavropoulos-Giokas, C.; Michalopoulos, E. Mesenchymal stromal cell delivery as a potential therapeutic strategy against COVID-19: Promising evidence from in vitro results. World J. Biol. Chem. 2022, 13, 47–65. Available online: https://pubmed.ncbi.nlm.nih.gov/35432769/ (accessed on 7 February 2023). [CrossRef] [PubMed]
- Mareschi, K.; Castiglia, S.; Sanavio, F.; Rustichelli, D.; Muraro, M.; Defedele, D.; Bergallo, M.; Fagioli, F. Immunoregulatory effects on T lymphocytes by human mesenchymal stromal cells isolated from bone marrow, amniotic fluid, and placenta. Exp. Hematol. 2016, 44, 138–150.e1. [Google Scholar] [CrossRef] [PubMed]
- Chinnadurai, R.; Rajan, D.; Qayed, M.; Arafat, D.; Garcia, M.; Liu, Y.; Kugathasan, S.; Anderson, L.J.; Gibson, G.; Galipeau, J. Potency Analysis of Mesenchymal Stromal Cells Using a Combinatorial Assay Matrix Approach. Cell Rep. 2018, 22, 2504–2517. [Google Scholar] [CrossRef] [PubMed]
- Croci, S.; Bonacini, M.; Dolci, G.; Massari, M.; Facciolongo, N.; Pignatti, E.; Pisciotta, A.; Carnevale, G.; Negro, A.; Cassone, G.; et al. Human Dental Pulp Stem Cells Modulate Cytokine Production in vitro by Peripheral Blood Mononuclear Cells From Coronavirus Disease 2019 Patients. Front. Cell Dev. Biol. 2021, 8, 609204. [Google Scholar] [CrossRef]
- Cruz-Barrera, M.; Flórez-Zapata, N.; Lemus-Diaz, N.; Medina, C.; Galindo, C.C.; González-Acero, L.X.; Correa, L.; Camacho, B.; Gruber, J.; Salguero, G. Integrated Analysis of Transcriptome and SecretomeFrom Umbilical Cord Mesenchymal Stromal Cells Reveal New Mechanisms for the Modulation of Inflammation and Immune Activation. Front. Immunol. 2020, 11, 2449. Available online: https://www.frontiersin.org/article/10.3389/fimmu.2020.575488/full (accessed on 5 February 2023). [CrossRef]
- Treatment of Coronavirus COVID-19 Pneumonia (Pathogen SARS-CoV-2) With Cryopreserved Allogeneic P_MMSCs and UC-MMSCs-Full Text View-ClinicalTrials.gov. Available online: https://clinicaltrials.gov/ct2/show/NCT04461925 (accessed on 26 September 2022).
- Clinical Management of Severe Acute Respiratory Infection When Novel Coronavirus (nCoV) Infection Is Suspected. Available online: https://www.who.int/publications/i/item/10665-332299 (accessed on 25 July 2021).
- Dominici, M.; le Blanc, K.; Mueller, I.; Slaper-Cortenbach, I.; Marini, F.; Krause, D.; Deans, R.J.; Keating, A.; Prockop, D.J.; Horwitz, E.M. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy 2006, 8, 315–317. Available online: http://www.ncbi.nlm.nih.gov/pubmed/16923606 (accessed on 3 February 2023). [CrossRef]
- Büttner, L.; Aigner, A.; Fleckenstein, F.N.; Hamper, C.M.; Jonczyk, M.; Hamm, B.; Scholz, O.; Böning, G. Diagnostic Value of Initial Chest CT Findings for the Need of ICU Treatment/Intubation in Patients with COVID-19. Diagnostics 2020, 10, 929. Available online: https://www.mdpi.com/2075-4418/10/11/929/htm (accessed on 3 August 2022). [CrossRef]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 2001, 25, 402–408. Available online: http://www.ncbi.nlm.nih.gov/pubmed/11846609 (accessed on 25 July 2021). [CrossRef]


| Parameter | Control Group | MSC Group | p-Value |
|---|---|---|---|
| (n = 15) | (n = 13) | ||
| Age, years (median, range) | 62 (32–73) | 58 (32–71) | 0.25 |
| Gender | 0.61 | ||
| Male | 11/15 (73.33%) | 8/13 (61.5%) | |
| Female | 4/15 (26.67%) | 5/13 (38.5%) | |
| The interval from illness onset to hospital admission (days) | 11.0 (8.5–12.8) | 12.3 (9–14.5) | 0.29 |
| Underlying diseases—no. (%) | |||
| Hypertension | 9/15 (60.0%) | 5/13 (38.5%) | 0.34 |
| Diabetes | 1/15 (6.67%) | 1/13 (7.7%) | 0.91 |
| Heart disease | 6/15 (40.0%) | 4/13 (30.7%) | 0.68 |
| Symptoms—no. (%) | |||
| Fever | 11/15 (73.33%) | 11/13 (84.6%) | 0.61 |
| Cough | 15/15 (100.0%) | 13/13 (100%) | 1 |
| Shortness of breath | 12/15 (80.0%) | 9/13 (69.2%) | 0.65 |
| Diarrhea | 1/15 (6.67%) | 0/14 (0%) | 0.78 |
| Fatigue | 15/15 (100.0%) | 13/13 (100%) | 1 |
| Myalgia | 3/15 (20.0%) | 6/13 (46.1%) | 0.25 |
| Clinical outcome—no. (%) | |||
| Recovered and discharged | 14/15 (93.33%) | 13/13 (100%) | 0.78 |
| Death | 1/15 (6.67%) | 0/13 (0%) | 0.79 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bukreieva, T.; Svitina, H.; Nikulina, V.; Vega, A.; Chybisov, O.; Shablii, I.; Ustymenko, A.; Nemtinov, P.; Lobyntseva, G.; Skrypkina, I.; et al. Treatment of Acute Respiratory Distress Syndrome Caused by COVID-19 with Human Umbilical Cord Mesenchymal Stem Cells. Int. J. Mol. Sci. 2023, 24, 4435. https://doi.org/10.3390/ijms24054435
Bukreieva T, Svitina H, Nikulina V, Vega A, Chybisov O, Shablii I, Ustymenko A, Nemtinov P, Lobyntseva G, Skrypkina I, et al. Treatment of Acute Respiratory Distress Syndrome Caused by COVID-19 with Human Umbilical Cord Mesenchymal Stem Cells. International Journal of Molecular Sciences. 2023; 24(5):4435. https://doi.org/10.3390/ijms24054435
Chicago/Turabian StyleBukreieva, Tetiana, Hanna Svitina, Viktoriia Nikulina, Alyona Vega, Oleksii Chybisov, Iuliia Shablii, Alina Ustymenko, Petro Nemtinov, Galyna Lobyntseva, Inessa Skrypkina, and et al. 2023. "Treatment of Acute Respiratory Distress Syndrome Caused by COVID-19 with Human Umbilical Cord Mesenchymal Stem Cells" International Journal of Molecular Sciences 24, no. 5: 4435. https://doi.org/10.3390/ijms24054435
APA StyleBukreieva, T., Svitina, H., Nikulina, V., Vega, A., Chybisov, O., Shablii, I., Ustymenko, A., Nemtinov, P., Lobyntseva, G., Skrypkina, I., & Shablii, V. (2023). Treatment of Acute Respiratory Distress Syndrome Caused by COVID-19 with Human Umbilical Cord Mesenchymal Stem Cells. International Journal of Molecular Sciences, 24(5), 4435. https://doi.org/10.3390/ijms24054435







