Overexpression of Potential Markers of Regulatory and Exhausted CD8+ T Cells in the Peripheral Blood Mononuclear Cells of Patients with B-Acute Lymphoblastic Leukemia
Abstract
:1. Introduction
2. Results
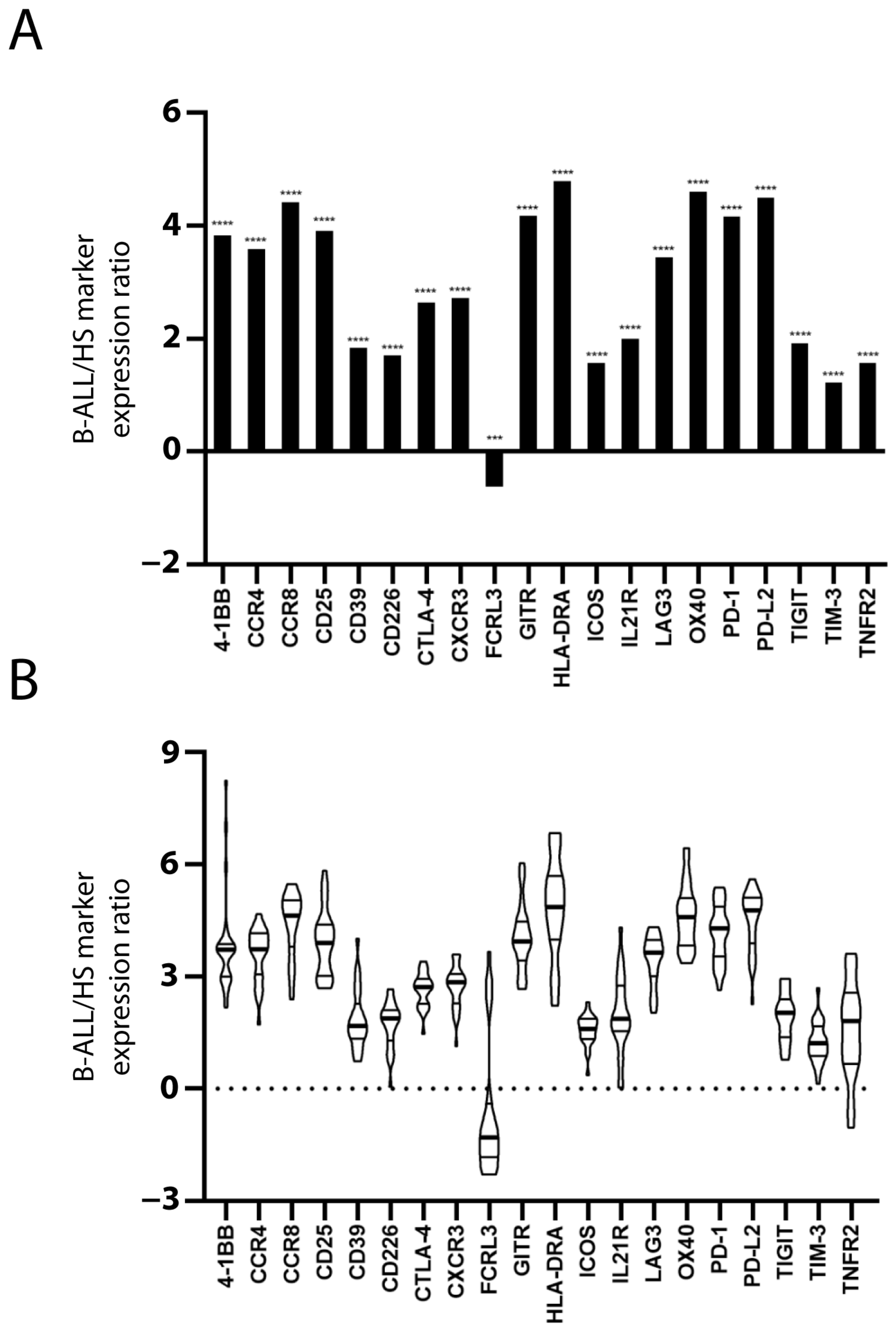
2.1. Treg/CD8 Exhaustion Marker Overexpression in Patients with B-ALL
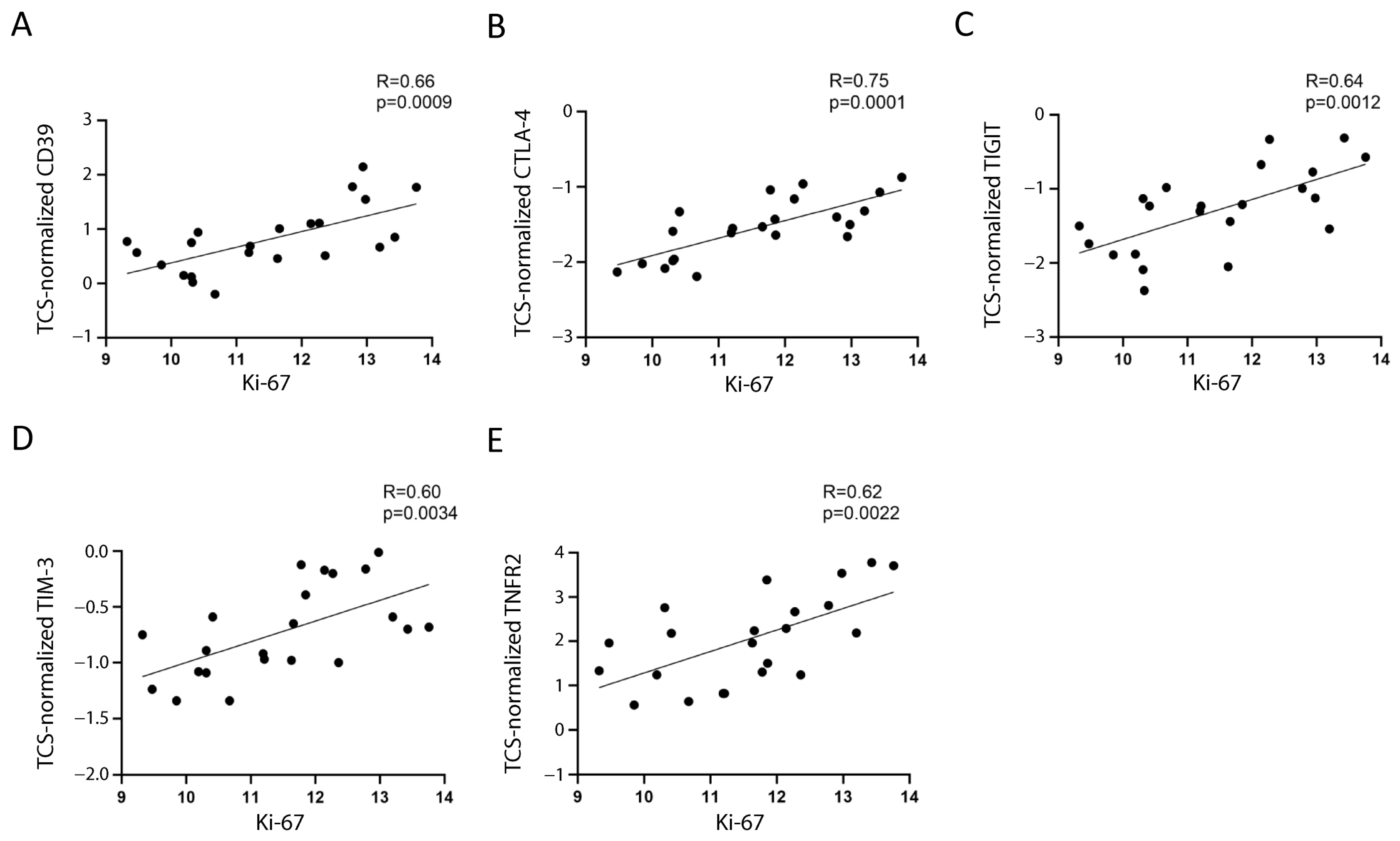
2.2. Correlation of Treg/CD8 Exhaustion Marker Overexpression with Higher Leukemic Cell Proliferation Rate
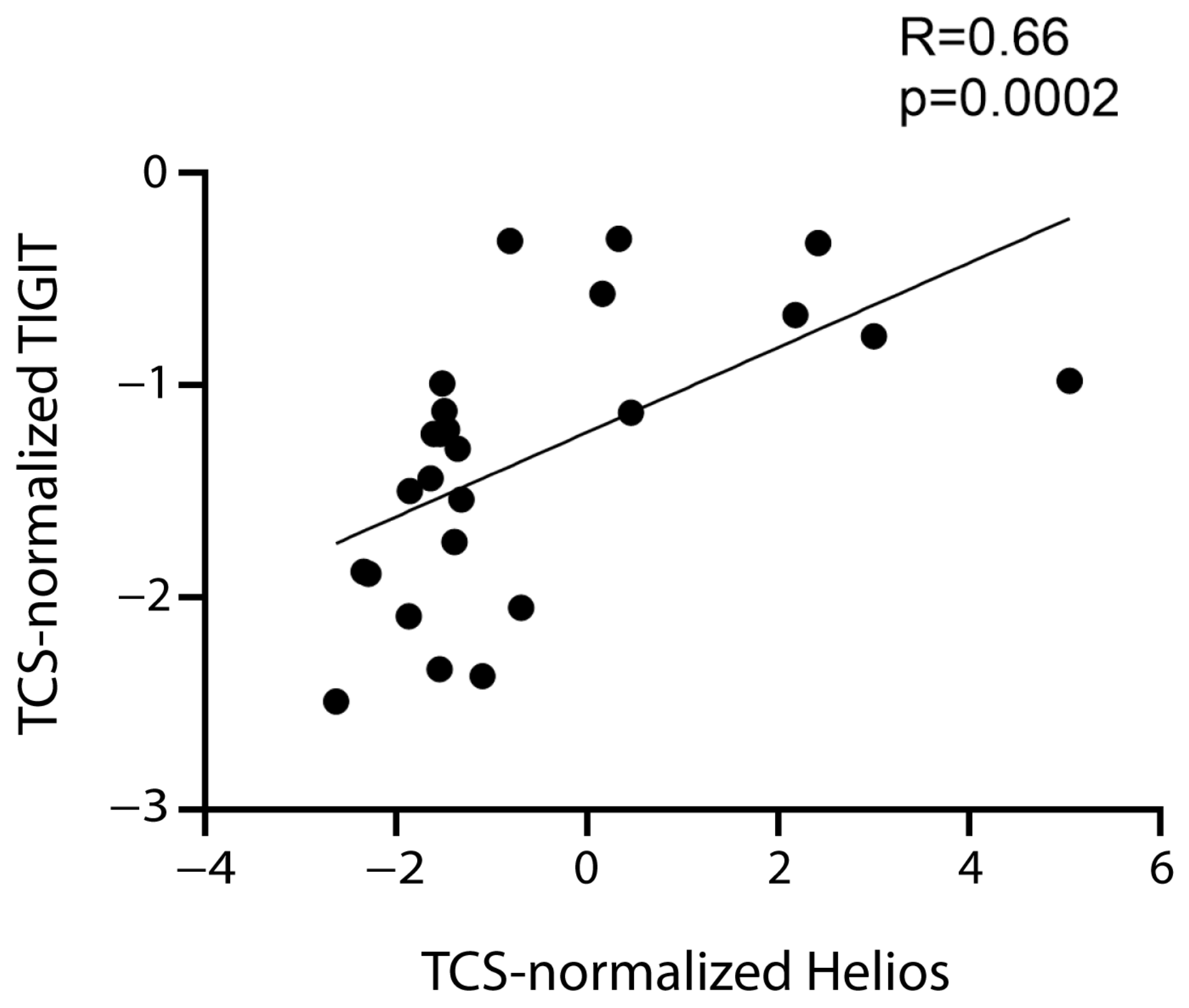
2.3. Treg Phenotypes of Cells Expressing Treg/CD8 Exhaustion Markers
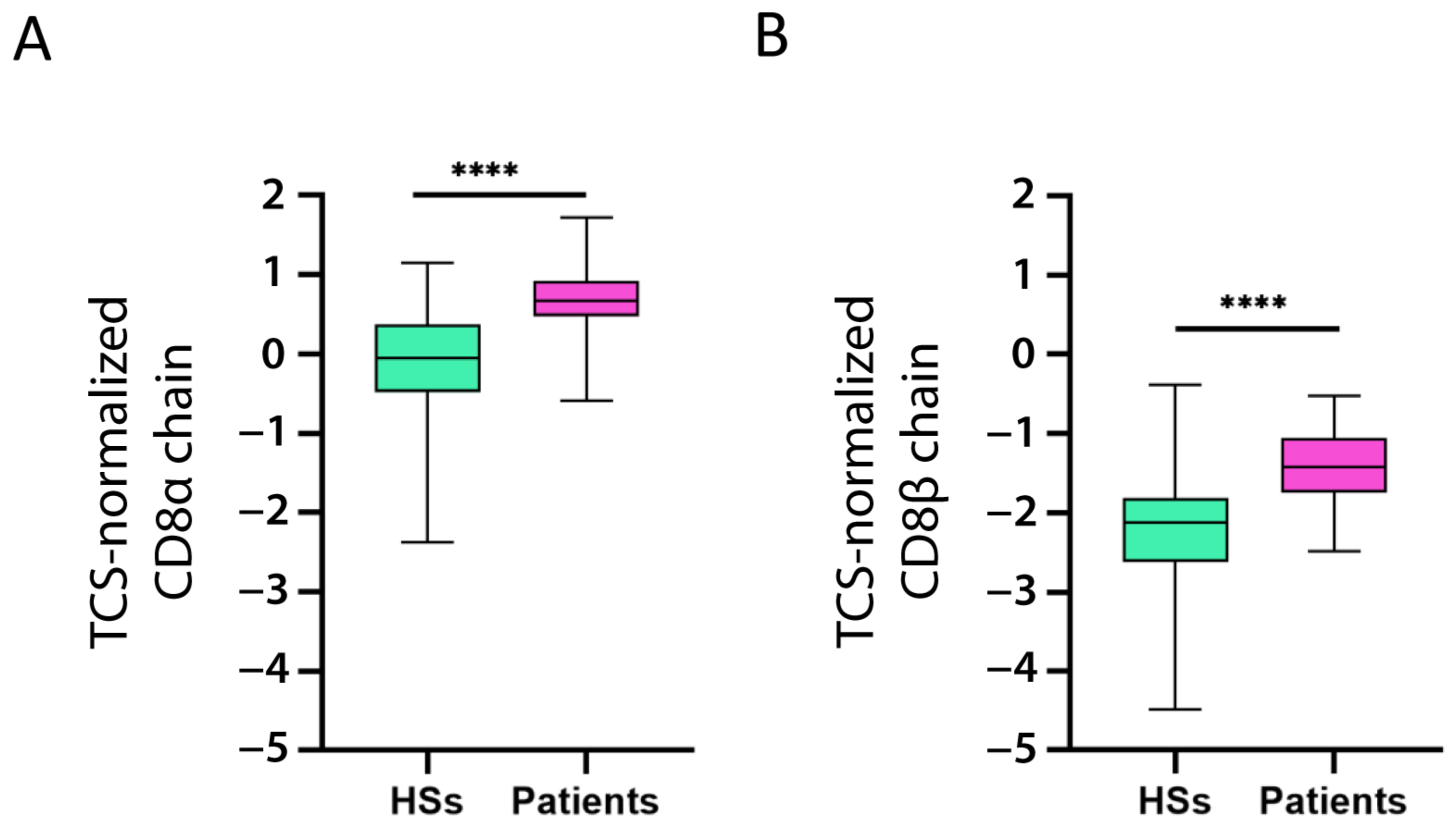
2.4. Treg/CD8 Exhaustion Marker Expression by Exhausted CD8+ T Cells
3. Discussion
4. Materials and Methods
4.1. Data Source, Tumor Type, and Controls
4.2. Genes of Interest
4.3. Use of T Cell Signature to Normalize Gene Expression
4.4. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Sakaguchi, S.; Sakaguchi, N.; Asano, M.; Itoh, M.; Toda, M. Immunologic self-tolerance maintained by activated T cells expressing IL-2 receptor alpha-chains (CD25). Breakdown of a single mechanism of self-tolerance causes various autoimmune diseases. J. Immunol. 1995, 155, 1151–1164. [Google Scholar] [CrossRef] [PubMed]
- Hori, S.; Nomura, T.; Sakaguchi, S. Control of regulatory T cell development by the transcription factor Foxp3. Science 2003, 299, 1057–1061. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, W.; Putnam, A.L.; Xu-Yu, Z.; Szot, G.L.; Lee, M.R.; Zhu, S.; Gottlieb, P.A.; Kapranov, P.; Gingeras, T.R.; de St. Groth, B.F. CD127 expression inversely correlates with FoxP3 and suppressive function of human CD4+ T reg cells. J. Exp. Med. 2006, 203, 1701–1711. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Seddiki, N.; Santner-Nanan, B.; Martinson, J.; Zaunders, J.; Sasson, S.; Landay, A.; Solomon, M.; Selby, W.; Alexander, S.I.; Nanan, R. Expression of interleukin (IL)-2 and IL-7 receptors discriminates between human regulatory and activated T cells. J. Exp. Med. 2006, 203, 1693–1700. [Google Scholar] [CrossRef]
- Wolf, D.; Sopper, S.; Pircher, A.; Gastl, G.; Wolf, A.M. Treg (s) in cancer: Friends or foe? J. Cell. Physiol. 2015, 230, 2598–2605. [Google Scholar] [CrossRef]
- Tanaka, A.; Sakaguchi, S. Regulatory T cells in cancer immunotherapy. Cell Res. 2017, 27, 109–118. [Google Scholar] [CrossRef] [Green Version]
- Caramalho, Í.; Nunes-Cabaço, H.; Foxall, R.B.; Sousa, A.E. Regulatory T-cell development in the human thymus. Front. Immunol. 2015, 6, 395. [Google Scholar] [CrossRef] [Green Version]
- De Lafaille, M.A.C.; Lafaille, J.J. Natural and adaptive foxp3+ regulatory T cells: More of the same or a division of labor? Immunity 2009, 30, 626–635. [Google Scholar] [CrossRef] [Green Version]
- Itahashi, K.; Irie, T.; Nishikawa, H. Regulatory T-cell development in the tumor microenvironment. Eur. J. Immunol. 2022, 52, 1216–1227. [Google Scholar] [CrossRef]
- Chen, B.-J.; Zhao, J.-W.; Zhang, D.-H.; Zheng, A.-H.; Wu, G.-Q. Immunotherapy of Cancer by Targeting Regulatory T cells. Int. Immunopharmacol. 2022, 104, 108469. [Google Scholar] [CrossRef]
- Thornton, A.M.; Korty, P.E.; Tran, D.Q.; Wohlfert, E.A.; Murray, P.E.; Belkaid, Y.; Shevach, E.M. Expression of Helios, an Ikaros transcription factor family member, differentiates thymic-derived from peripherally induced Foxp3+ T regulatory cells. J. Immunol. 2010, 184, 3433–3441. [Google Scholar] [CrossRef] [Green Version]
- Yu, W.-q.; Ji, N.-f.; Gu, C.-j.; Wang, Y.-l.; Huang, M.; Zhang, M.-s. Coexpression of Helios in Foxp3+ Regulatory T Cells and Its Role in Human Disease. Dis. Markers 2021, 2021, 5574472. [Google Scholar] [CrossRef]
- Cari, L.; Nocentini, G.; Migliorati, G.; Riccardi, C. Potential effect of tumor-specific Treg-targeted antibodies in the treatment of human cancers: A bioinformatics analysis. Oncoimmunology 2018, 7, e1387705. [Google Scholar] [CrossRef] [Green Version]
- Curiel, T.J.; Coukos, G.; Zou, L.; Alvarez, X.; Cheng, P.; Mottram, P.; Evdemon-Hogan, M.; Conejo-Garcia, J.R.; Zhang, L.; Burow, M.; et al. Specific recruitment of regulatory T cells in ovarian carcinoma fosters immune privilege and predicts reduced survival. Nat. Med. 2004, 10, 942–949. [Google Scholar] [CrossRef]
- Halvorsen, E.C.; Hamilton, M.J.; Young, A.; Wadsworth, B.J.; LePard, N.E.; Lee, H.N.; Firmino, N.; Collier, J.L.; Bennewith, K.L. Maraviroc decreases CCL8-mediated migration of CCR5+ regulatory T cells and reduces metastatic tumor growth in the lungs. Oncoimmunology 2016, 5, e1150398. [Google Scholar] [CrossRef] [Green Version]
- Muto, S.; Owada, Y.; Inoue, T.; Watanabe, Y.; Yamaura, T.; Fukuhara, M.; Okabe, N.; Matsumura, Y.; Hasegawa, T.; Osugi, J.; et al. Clinical significance of expanded Foxp3(+) Helios(-) regulatory T cells in patients with non-small cell lung cancer. Int. J. Oncol. 2015, 47, 2082–2090. [Google Scholar] [CrossRef] [Green Version]
- Kurachi, M. CD8+ T cell exhaustion. In Proceedings of the Seminars in Immunopathology; Springer: Berlin/Heidelberg, Germany, 2019; pp. 327–337. [Google Scholar]
- Jiang, W.; He, Y.; He, W.; Wu, G.; Zhou, X.; Sheng, Q.; Zhong, W.; Lu, Y.; Ding, Y.; Lu, Q. Exhausted CD8+ T cells in the tumor immune microenvironment: New pathways to therapy. Front. Immunol. 2021, 11, 622509. [Google Scholar] [CrossRef]
- Canale, F.P.; Ramello, M.C.; Núñez, N.; Furlan, C.L.A.; Bossio, S.N.; Serrán, M.G.; Boari, J.T.; Del Castillo, A.; Ledesma, M.; Sedlik, C. CD39 Expression Defines Cell Exhaustion in Tumor-Infiltrating CD8+ T CellsCD39 Defines Exhaustion in CD8+ TILs. Cancer Res. 2018, 78, 115–128. [Google Scholar] [CrossRef] [Green Version]
- Franzin, R.; Netti, G.S.; Spadaccino, F.; Porta, C.; Gesualdo, L.; Stallone, G.; Castellano, G.; Ranieri, E. The use of immune checkpoint inhibitors in oncology and the occurrence of AKI: Where do we stand? Front. Immunol. 2020, 11, 574271. [Google Scholar] [CrossRef]
- Swerdlow, S.H. WHO classification of tumours of haematopoietic and lymphoid tissues. WHO Classif. Tumours 2008, 22008, 439. [Google Scholar]
- Rowley, J.D. Chromosome translocations: Dangerous liaisons revisited. Nat. Rev. Cancer 2001, 1, 245–250. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.; Urayama, K.; Chang, J.; Wiemels, J.L.; Buffler, P.A. Infection and pediatric acute lymphoblastic leukemia. Blood Cells Mol. Dis. 2009, 42, 117–120. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Greaves, M.; Muschen, M. Infection and the Perils of B-cell Activation. Cancer Discov. 2015, 5, 1244–1246. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Idris, S.-Z.; Hassan, N.; Lee, L.-J.; Md Noor, S.; Osman, R.; Abdul-Jalil, M.; Nordin, A.-J.; Abdullah, M. Increased regulatory T cells in acute lymphoblastic leukemia patients. Hematology 2015, 20, 523–529. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Traxel, S.; Schadt, L.; Eyer, T.; Mordasini, V.; Gysin, C.; Munthe, L.A.; Niggli, F.; Nadal, D.; Burgler, S. Bone marrow T helper cells with a Th1 phenotype induce activation and proliferation of leukemic cells in precursor B acute lymphoblastic leukemia patients. Oncogene 2019, 38, 2420–2431. [Google Scholar] [CrossRef]
- Wang, R.; Feng, W.; Wang, H.; Wang, L.; Yang, X.; Yang, F.; Zhang, Y.; Liu, X.; Zhang, D.; Ren, Q. Blocking migration of regulatory T cells to leukemic hematopoietic microenvironment delays disease progression in mouse leukemia model. Cancer Lett. 2020, 469, 151–161. [Google Scholar] [CrossRef]
- Lindqvist, C.A.; Loskog, A.S. T regulatory cells in B-cell malignancy–tumour support or kiss of death? Immunology 2012, 135, 255–260. [Google Scholar] [CrossRef]
- Bhattacharya, K.; Chandra, S.; Mandal, C. Critical stoichiometric ratio of CD4+ CD25+ FoxP3+ regulatory T cells and CD4+ CD25− responder T cells influence immunosuppression in patients with B-cell acute lymphoblastic leukaemia. Immunology 2014, 142, 124–139. [Google Scholar] [CrossRef]
- Liu, S.X.; Xiao, H.R.; Wang, G.B.; Chen, X.W.; Li, C.G.; Mai, H.R.; Yuan, X.L.; Liu, G.S.; Wen, F.Q. Preliminary investigation on the abnormal mechanism of CD4+ FOXP3+ CD25high regulatory T cells in pediatric B-cell acute lymphoblastic leukemia. Exp. Ther. Med. 2018, 16, 1433–1441. [Google Scholar] [CrossRef] [Green Version]
- Wu, Z.-L.; Hu, G.-Y.; Chen, F.-X.; Lu, H.-M.; Li, H.; Wei, F.; Guan, J.; Wu, L. Change of CD4+ CD25+ regulatory T cells and NK Cells in peripheral blood of children with acute leukemia and its possible significance in tumor immunity. Zhongguo Shi Yan Xue Ye Xue Za Zhi 2010, 18, 709–713. [Google Scholar]
- Hodi, F.S.; Butler, M.; Oble, D.A.; Seiden, M.V.; Haluska, F.G.; Kruse, A.; MacRae, S.; Nelson, M.; Canning, C.; Lowy, I. Immunologic and clinical effects of antibody blockade of cytotoxic T lymphocyte-associated antigen 4 in previously vaccinated cancer patients. Proc. Natl. Acad. Sci. USA 2008, 105, 3005–3010. [Google Scholar] [CrossRef] [Green Version]
- Kang, D.H.; Chung, C.; Sun, P.; Lee, D.H.; Lee, S.-I.; Park, D.; Koh, J.S.; Kim, Y.; Yi, H.-S.; Lee, J.E. Circulating regulatory T cells predict efficacy and atypical responses in lung cancer patients treated with PD-1/PD-L1 inhibitors. Cancer Immunol. Immunother. 2022, 71, 579–588. [Google Scholar] [CrossRef]
- H Hiroki, C.; P Erthal, R.; PL Pereira, A.; M Pacholak, L.; C Fujita, T.; C Marinello, P.; N Costa, I.; PD da Rocha, S.; AE Watanabe, M.; EC de Oliveira, C. Acute lymphoblastic leukemia and regulatory T cells: Biomarkers and immunopathogenesis. Curr. Immunol. Rev. 2016, 12, 14–19. [Google Scholar] [CrossRef]
- D’Arena, G.; Vitale, C.; Coscia, M.; Festa, A.; Di Minno, N.M.D.; De Feo, V.; Caraglia, M.; Calapai, G.; Laurenti, L.; Musto, P. Regulatory T cells and their prognostic relevance in hematologic malignancies. J. Immunol. Res. 2017, 2017, 1832968. [Google Scholar] [CrossRef] [Green Version]
- Saleh, R.; Elkord, E. FoxP3+ T regulatory cells in cancer: Prognostic biomarkers and therapeutic targets. Cancer Lett. 2020, 490, 174–185. [Google Scholar] [CrossRef]
- Dolina, J.S.; Braeckel-Budimir, V.; Thomas, G.D.; Salek-Ardakani, S. CD8+ T Cell Exhaustion in Cancer. Front. Immunol. 2021, 12, 715234. [Google Scholar] [CrossRef]
- Thornton, A.M.; Korty, P.E.; Kim, Y.C.; Martens, C.; Shevach, E.M. Helios expression defines a phenotypically distinct population of Treg cells. J. Immunol. 2018, 200 (Suppl. 1), 116–119. [Google Scholar] [CrossRef]
- Huang, Y.; Jia, A.; Wang, Y.; Liu, G. CD8+ T cell exhaustion in anti-tumour immunity: The new insights for cancer immunotherapy. Immunology 2022, 168, 30–48. [Google Scholar] [CrossRef]
- Timperi, E.; Barnaba, V. CD39 regulation and functions in T cells. Int. J. Mol. Sci. 2021, 22, 8068. [Google Scholar] [CrossRef]
- Mehta, A.K.; Gracias, D.T.; Croft, M. TNF activity and T cells. Cytokine 2018, 101, 14–18. [Google Scholar] [CrossRef]
- Medler, J.; Kucka, K.; Wajant, H. Tumor Necrosis Factor Receptor 2 (TNFR2): An Emerging Target in Cancer Therapy. Cancers 2022, 14, 2603. [Google Scholar] [CrossRef] [PubMed]
- Bruey, J.-M.; Kantarjian, H.; Estrov, Z.; Zhang, Z.; Ma, W.; Albitar, F.; Abdool, A.; Thomas, D.; Yeh, C.; O’Brien, S. Circulating Ki-67 protein in plasma as a biomarker and prognostic indicator of acute lymphoblastic leukemia. Leuk. Res. 2010, 34, 173–176. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, M.; Wang, L.-R. Reseach progress of Ki-67 in acute myeloid leukemia-review. Zhongguo Shi Yan Xue Ye Xue Za Zhi 2016, 24, 1264–1268. [Google Scholar] [PubMed]
- Zahran, A.M.; Shibl, A.; Rayan, A.; Mohamed, M.A.E.H.; Osman, A.M.; Saad, K.; Mahmoud, K.H.; Ghandour, A.M.; Elsayh, K.I.; El-Badawy, O. Increase in polymorphonuclear myeloid-derived suppressor cells and regulatory T-cells in children with B-cell acute lymphoblastic leukemia. Sci. Rep. 2021, 11, 15039. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.-J.; Barnitz, R.A.; Kreslavsky, T.; Brown, F.D.; Moffett, H.; Lemieux, M.E.; Kaygusuz, Y.; Meissner, T.; Holderried, T.A.; Chan, S. Stable inhibitory activity of regulatory T cells requires the transcription factor Helios. Science 2015, 350, 334–339. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, X.; Li, D.; Huang, X.; Zhou, P.; Shi, Q.; Zhang, B.; Ju, X. Helios expression in regulatory T cells promotes immunosuppression, angiogenesis and the growth of leukemia cells in pediatric acute lymphoblastic leukemia. Leuk. Res. 2018, 67, 60–66. [Google Scholar] [CrossRef]
- Ahlmanner, F.; Sundström, P.; Akeus, P.; Eklöf, J.; Börjesson, L.; Gustavsson, B.; Lindskog, E.B.; Raghavan, S.; Quiding-Järbrink, M. CD39+ regulatory T cells accumulate in colon adenocarcinomas and display markers of increased suppressive function. Oncotarget 2018, 9, 36993. [Google Scholar] [CrossRef] [Green Version]
- Cai, X.Y.; Wang, X.F.; Li, J.; Dong, J.N.; Liu, J.Q.; Li, N.P.; Yun, B.; Xia, R.L.; Qin, J.; Sun, Y.H. High expression of CD39 in gastric cancer reduces patient outcome following radical resection. Oncol. Lett. 2016, 12, 4080–4086. [Google Scholar] [CrossRef] [Green Version]
- Jin, Z.; Ye, W.; Lan, T.; Zhao, Y.; Liu, X.; Chen, J.; Lai, J.; Chen, S.; Zhong, X.; Wu, X. Characteristic of TIGIT and DNAM-1 Expression on Foxp3+ γδ T Cells in AML Patients. BioMed Res. Int. 2020, 2020, 4612952. [Google Scholar] [CrossRef]
- Zhang, X.; Zhang, H.; Chen, L.; Feng, Z.; Gao, L.; Li, Q. TIGIT expression is upregulated in T cells and causes T cell dysfunction independent of PD-1 and Tim-3 in adult B lineage acute lymphoblastic leukemia. Cell. Immunol. 2019, 344, 103958. [Google Scholar] [CrossRef]
- Swann, J.B.; Smyth, M.J. Immune surveillance of tumors. J. Clin. Investig. 2007, 117, 1137–1146. [Google Scholar] [CrossRef] [Green Version]
- Xu, Y.; Mou, J.; Wang, Y.; Zhou, W.; Rao, Q.; Xing, H.; Tian, Z.; Tang, K.; Wang, M.; Wang, J. Regulatory T cells promote the stemness of leukemia stem cells through IL10 cytokine-related signaling pathway. Leukemia 2022, 36, 403–415. [Google Scholar] [CrossRef]
- Gholipour, E.; Kahroba, H.; Soltani, N.; Samadi, P.; Sarvarian, P.; Vakili-Samiani, S.; Hosein Pour Feizi, A.A.; Soltani-Zangbar, M.S.; Baghersalimi, A.; Darbandi, B.; et al. Paediatric pre-B acute lymphoblastic leukaemia-derived exosomes regulate immune function in human T cells. J. Cell. Mol. Med. 2022, 26, 4566–4576. [Google Scholar] [CrossRef]
- Awwad, M.H.; Mahmoud, A.; Bruns, H.; Echchannaoui, H.; Kriegsmann, K.; Lutz, R.; Raab, M.S.; Bertsch, U.; Munder, M.; Jauch, A. Selective elimination of immunosuppressive T cells in patients with multiple myeloma. Leukemia 2021, 35, 2602–2615. [Google Scholar] [CrossRef]
- Dahlhoff, J.; Manz, H.; Steinfatt, T.; Delgado-Tascon, J.; Seebacher, E.; Schneider, T.; Wilnit, A.; Mokhtari, Z.; Tabares, P.; Böckle, D. Transient regulatory T-cell targeting triggers immune control of multiple myeloma and prevents disease progression. Leukemia 2022, 36, 790–800. [Google Scholar] [CrossRef]
- Yano, M.; Nunes, J.; Mo, X.; Rogers, K.A.; Woyach, J.A.; Byrd, J.C.; Muthusamy, N. Differential regulation of CTLA4 expression through BTK-dependent and independent mechanisms in CLL. Blood Adv. 2022, 6, 5440–5448. [Google Scholar] [CrossRef]
- Lamble, A.J.; Kosaka, Y.; Laderas, T.; Maffit, A.; Kaempf, A.; Brady, L.K.; Wang, W.; Long, N.; Saultz, J.N.; Mori, M. Reversible suppression of T cell function in the bone marrow microenvironment of acute myeloid leukemia. Proc. Natl. Acad. Sci. USA 2020, 117, 14331–14341. [Google Scholar] [CrossRef]
- Salik, B.; Smyth, M.J.; Nakamura, K. Targeting immune checkpoints in hematological malignancies. J. Hematol. Oncol. 2020, 13, 111. [Google Scholar] [CrossRef]
- Gupta, P.K.; Godec, J.; Wolski, D.; Adland, E.; Yates, K.; Pauken, K.E.; Cosgrove, C.; Ledderose, C.; Junger, W.G.; Robson, S.C. CD39 expression identifies terminally exhausted CD8+ T cells. PLoS Pathog. 2015, 11, e1005177. [Google Scholar] [CrossRef] [Green Version]
- Noble, A.; Mehta, H.; Lovell, A.; Papaioannou, E.; Fairbanks, L. IL-12 and IL-4 activate a CD39-dependent intrinsic peripheral tolerance mechanism in CD8+ T cells. Eur. J. Immunol. 2016, 46, 1438–1448. [Google Scholar] [CrossRef] [Green Version]
- Vaisitti, T.; Arruga, F.; Guerra, G.; Deaglio, S. Ectonucleotidases in blood malignancies: A tale of surface markers and therapeutic targets. Front. Immunol. 2019, 10, 2301. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Islam, M.S.; Hu, Y.; Chen, X. TNFR2: Role in cancer immunology and immunotherapy. ImmunoTargets Ther. 2021, 10, 103. [Google Scholar] [CrossRef] [PubMed]
- Edgar, R.; Domrachev, M.; Lash, A.E. Gene Expression Omnibus: NCBI gene expression and hybridization array data repository. Nucleic Acids Res. 2002, 30, 207–210. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kolesnikov, N.; Hastings, E.; Keays, M.; Melnichuk, O.; Tang, Y.A.; Williams, E.; Dylag, M.; Kurbatova, N.; Brandizi, M.; Burdett, T. ArrayExpress update—Simplifying data submissions. Nucleic Acids Res. 2015, 43, D1113–D1116. [Google Scholar] [CrossRef]
- Cari, L.; De Rosa, F.; Petrillo, M.G.; Migliorati, G.; Nocentini, G.; Riccardi, C. Identification of 15 T cell restricted genes evaluates T cell infiltration of human healthy tissues and cancers and shows prognostic and predictive potential. Int. J. Mol. Sci. 2019, 20, 5242. [Google Scholar] [CrossRef] [Green Version]


| CD39 | CTLA-4 | TIGIT | TIM-3 | TNFR2 | |
|---|---|---|---|---|---|
| FoxP3 | 0.66 | 0.73 | 0.86 | 0.56 | ns |
| Helios | ns | ns | 0.66 | ns | ns |
| IL-10 | 0.62 | 0.47 | 0.52 | 0.51 | 0.43 |
| TGF-β | ns | ns | ns | 0.40 | 0.66 |
| TIM-3 | CD39 | TIGIT | CTLA-4 | TNFR2 | |
|---|---|---|---|---|---|
| mean of CD8α chain + CD8β chain | 0.50 | 0.50 | 0.56 | 0.61 | 0.58 |
| mean of Granzyme B + Granulysin | −0.55 | −0.58 | −0.76 | −0.58 | ns |
| (mean of CD8α chain + CD8β chain)—(mean of Granzyme B + Granulysin) | 0.68 | 0.67 | 0.81 | 0.73 | ns |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Naghavi Alhosseini, M.; Palazzo, M.; Cari, L.; Ronchetti, S.; Migliorati, G.; Nocentini, G. Overexpression of Potential Markers of Regulatory and Exhausted CD8+ T Cells in the Peripheral Blood Mononuclear Cells of Patients with B-Acute Lymphoblastic Leukemia. Int. J. Mol. Sci. 2023, 24, 4526. https://doi.org/10.3390/ijms24054526
Naghavi Alhosseini M, Palazzo M, Cari L, Ronchetti S, Migliorati G, Nocentini G. Overexpression of Potential Markers of Regulatory and Exhausted CD8+ T Cells in the Peripheral Blood Mononuclear Cells of Patients with B-Acute Lymphoblastic Leukemia. International Journal of Molecular Sciences. 2023; 24(5):4526. https://doi.org/10.3390/ijms24054526
Chicago/Turabian StyleNaghavi Alhosseini, Mahdieh, Marianna Palazzo, Luigi Cari, Simona Ronchetti, Graziella Migliorati, and Giuseppe Nocentini. 2023. "Overexpression of Potential Markers of Regulatory and Exhausted CD8+ T Cells in the Peripheral Blood Mononuclear Cells of Patients with B-Acute Lymphoblastic Leukemia" International Journal of Molecular Sciences 24, no. 5: 4526. https://doi.org/10.3390/ijms24054526





