SARS-CoV-2 Lysate Stimulation Impairs the Release of Platelet-like Particles and Megakaryopoiesis in the MEG-01 Cell Line
Abstract
1. Introduction
2. Results
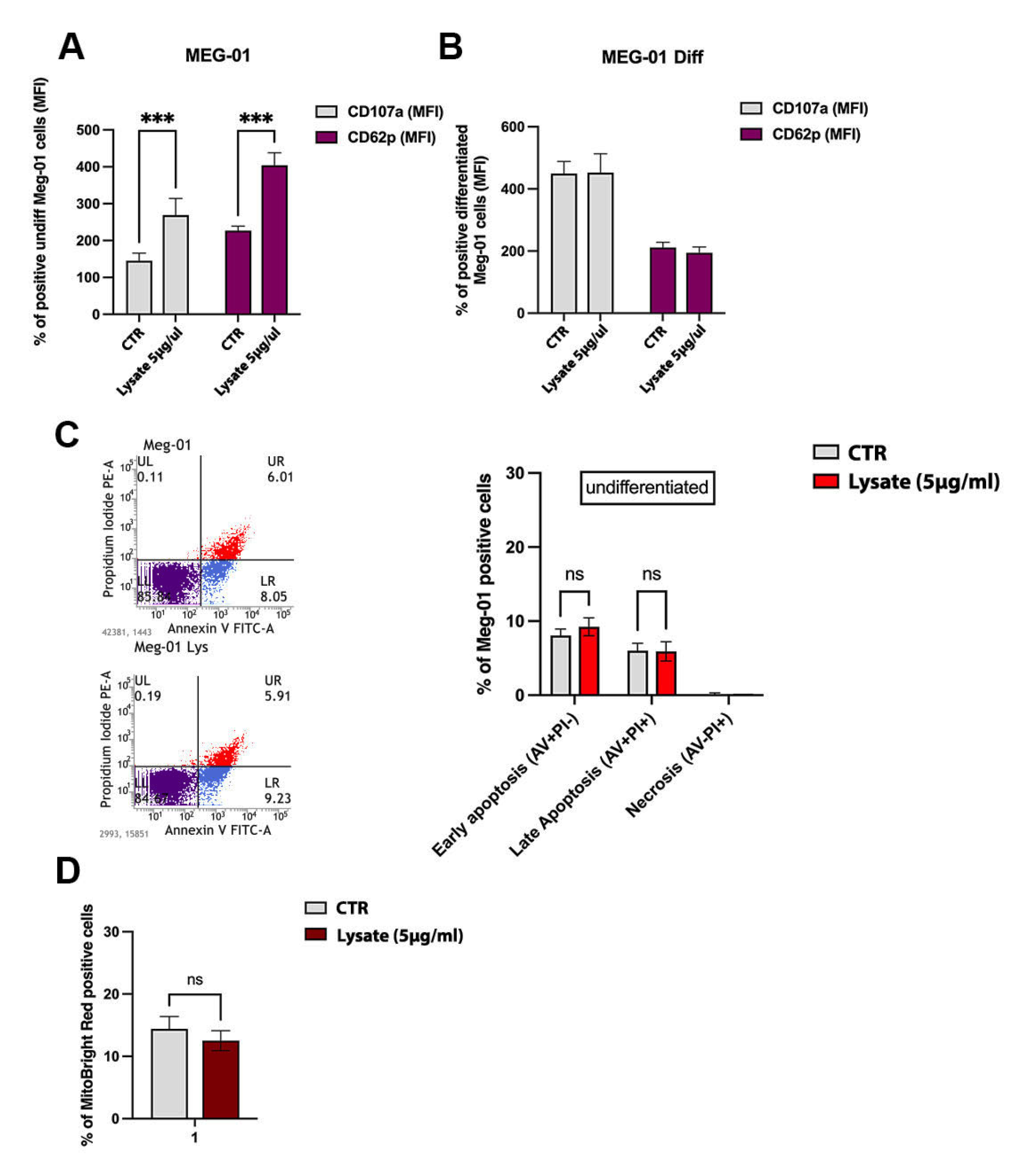
2.1. SARS-CoV-2 Lysate Induces Immature MEG-01 to Release Platelet-like Particles
2.2. SARS-CoV-2 Lysate Interferes with Platelet-like Particle Activation but Not with Cell Viability
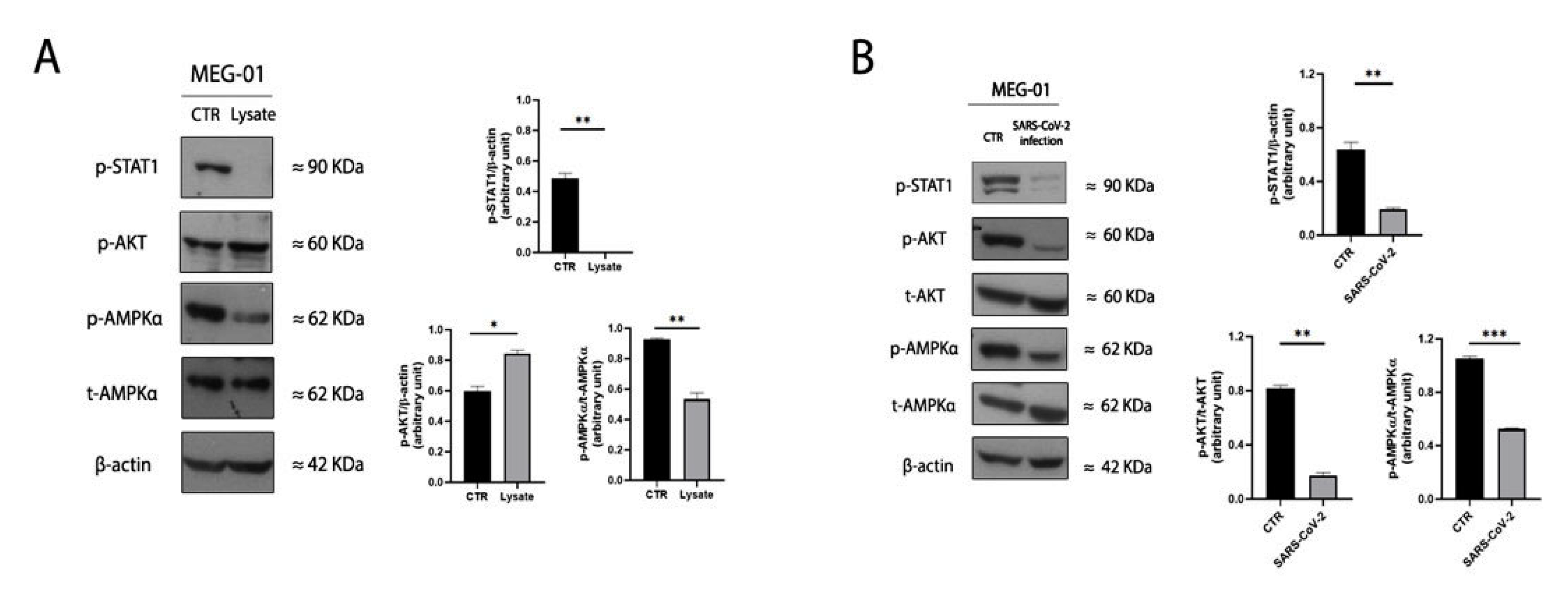
2.3. SARS-CoV-2 Lysate Impairs Megakaryopoiesis through STATs Signaling and AMPK Activity
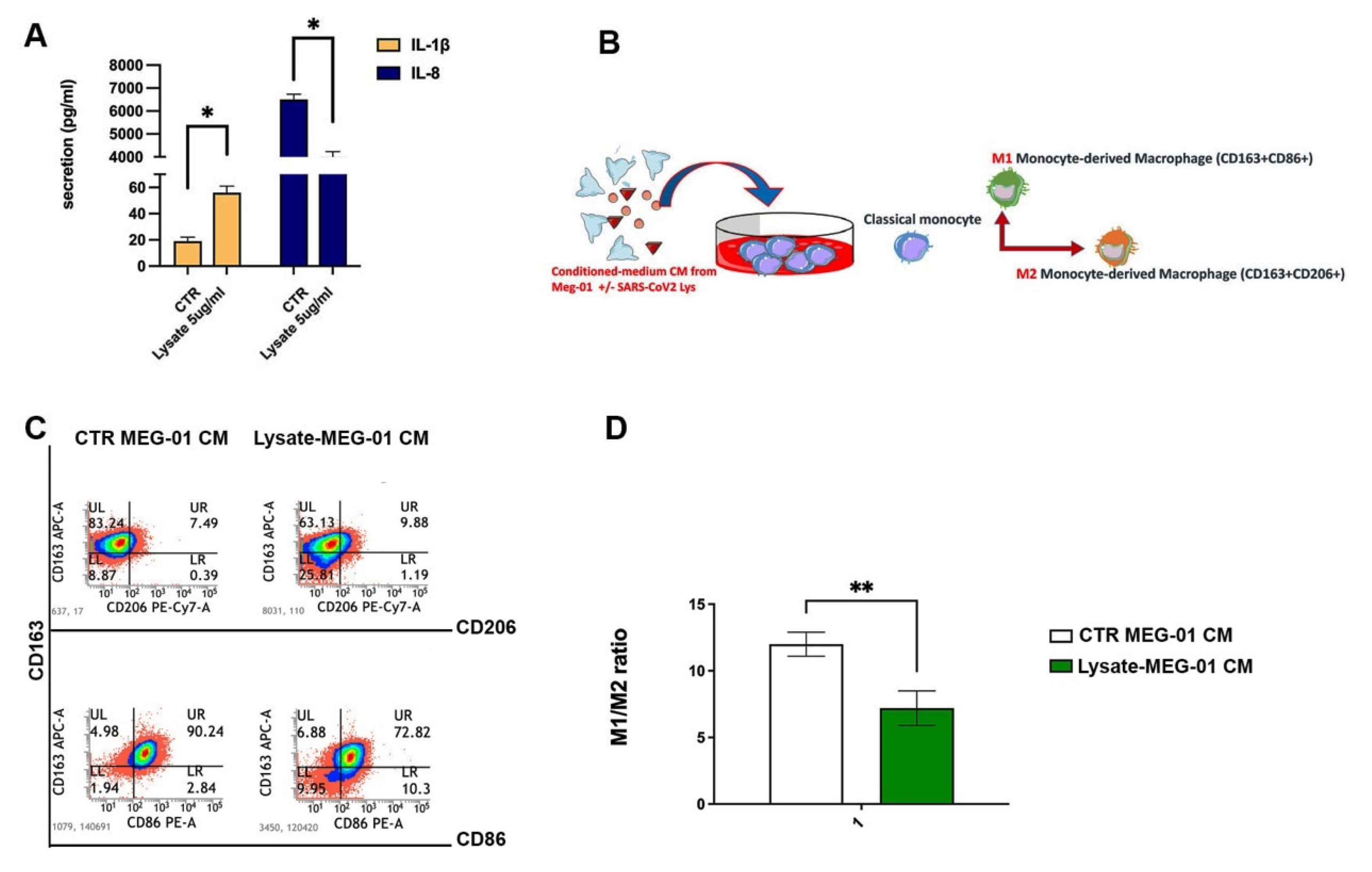
2.4. The Conditioned Medium from SARS-CoV-2-Treated MEG-01 Predisposes Macrophages to Acquire an Anti-Inflammatory Phenotype
3. Discussion
4. Materials and Methods
4.1. SARS-CoV-2 Lysate Production
4.2. Cell Line and Culture Condition
4.3. Platelets Count
4.4. Cytokine Detection
4.5. Flow Cytometry Analysis
4.5.1. Antibodies
4.5.2. Apoptosis Assay
4.5.3. Mitochondrial Activity
4.6. Western Blotting
4.7. Statistical Analysis
Author Contributions
Funding
Conflicts of Interest
References
- Bianchi, E.; Ruggiero, N.; Pennucci, V.; Zini, R.; Manfredini, R. Genomic landscape of megakaryopoiesis and platelet function defects. Blood 2016, 127, 1249–1259. [Google Scholar] [CrossRef] [PubMed]
- Machlus, K.R.; Thon, J.N.; Italiano, J.E., Jr. Interpreting the developmental dance of the megakaryocyte: A review of the cellular and molecular processes mediating platelet formation. Br. J. Haematol. 2014, 165, 227–236. [Google Scholar] [CrossRef] [PubMed]
- Lefrancais, E.; Ortiz-Munoz, G.; Caudrillier, A.; Mallavia, B.; Liu, F.; Sayah, D.M.; Thornton, E.E.; Headley, M.B.; David, T.; Coughlin, S.R.; et al. The lung is a site of platelet biogenesis and a reservoir for haematopoietic progenitors. Nature 2017, 544, 105–109. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Zhang, B.; Li, C.; Huang, X.; Cheng, H.; Bao, X.; Zhao, F.; Cheng, Q.; Yue, S.; Han, J.; et al. Megakaryocytes participate in the occurrence of bleomycin-induced pulmonary fibrosis. Cell Death Dis. 2019, 10, 648. [Google Scholar] [CrossRef]
- Pariser, D.N.; Hilt, Z.T.; Ture, S.K.; Blick-Nitko, S.K.; Looney, M.R.; Cleary, S.J.; Roman-Pagan, E.; Saunders, J., 2nd; Georas, S.N.; Veazey, J.; et al. Lung megakaryocytes are immune modulatory cells. J. Clin. Investig. 2021, 131, e137377. [Google Scholar] [CrossRef]
- Morrell, C.N.; Aggrey, A.A.; Chapman, L.M.; Modjeski, K.L. Emerging roles for platelets as immune and inflammatory cells. Blood 2014, 123, 2759–2767. [Google Scholar] [CrossRef]
- Robier, C. Platelet morphology. J. Lab. Med. 2020, 44, 231–239. [Google Scholar] [CrossRef]
- Handtke, S.; Steil, L.; Palankar, R.; Conrad, J.; Cauhan, S.; Kraus, L.; Ferrara, M.; Dhople, V.; Wesche, J.; Völker, U.; et al. Role of Platelet Size Revisited-Function and Protein Composition of Large and Small Platelets. Thromb. Haemost. 2019, 119, 407–420. [Google Scholar] [CrossRef]
- Miyazaki, R.; Ogata, H.; Kobayashi, Y. Requirement of thrombopoietin-induced activation of ERK for megakaryocyte differentiation and of p38 for erythroid differentiation. Ann. Hematol. 2001, 80, 284–291. [Google Scholar] [CrossRef]
- Lütteke, N.; Raftery, M.J.; Lalwani, P.; Lee, M.H.; Giese, T.; Voigt, S.; Bannert, N.; Schulze, H.; Krüger, D.H.; Schönrich, G. Switch to high-level virus replication and HLA class I upregulation in differentiating megakaryocytic cells after infection with pathogenic hantavirus. Virology 2010, 405, 70–80. [Google Scholar] [CrossRef]
- Gibellini, D.; Clò, A.; Morini, S.; Miserocchi, A.; Ponti, C.; Re, M.C. Effects of human immunodeficiency virus on the erythrocyte and megakaryocyte lineages. World J. Virol. 2013, 2, 91–101. [Google Scholar] [CrossRef]
- Shen, S.; Zhang, J.; Fang, Y.; Lu, S.; Wu, J.; Zheng, X.; Deng, F. SARS-CoV-2 interacts with platelets and megakaryocytes via ACE2-independent mechanism. J. Hematol. Oncol. 2021, 14, 72. [Google Scholar] [CrossRef]
- Campbell, R.A.; Boilard, E.; Rondina, M.T. Is there a role for the ACE2 receptor in SARS-CoV-2 interactions with platelets? J. Thromb. Haemost. 2021, 19, 46–50. [Google Scholar] [CrossRef]
- Zhu, A.; Real, F.; Capron, C.; Rosenberg, A.R.; Silvin, A.; Dunsmore, G.; Zhu, J.; Cottoignies-Callamarte, A.; Massé, J.M.; Moine, P.; et al. Infection of lung megakaryocytes and platelets by SARS-CoV-2 anticipate fatal COVID-19. Cell Mol. Life Sci. 2022, 79, 365. [Google Scholar] [CrossRef]
- Martyanov, A.A.; Boldova, A.E.; Stepanyan, M.G.; An, O.I.; Gur’ev, A.S.; Kassina, D.V.; Volkov, A.Y.; Balatskiy, A.V.; Butylin, A.A.; Karamzin, S.S.; et al. Longitudinal multiparametric characterization of platelet dysfunction in COVID-19: Effects of disease severity, anticoagulation therapy and inflammatory status. Thromb. Res. 2022, 211, 27–37. [Google Scholar] [CrossRef]
- Xiang, M.; Wu, X.; Jing, H.; Liu, L.; Wang, C.; Wang, Y.; Novakovic, V.A.; Shi, J. The impact of platelets on pulmonary microcirculation throughout COVID-19 and its persistent activating factors. Front. Immunol. 2022, 13, 955654. [Google Scholar] [CrossRef]
- Teimury, A.; Khameneh, M.T.; Khaledi, E.M. Major coagulation disorders and parameters in COVID-19 patients. Eur. J. Med. Res. 2022, 27, 25. [Google Scholar] [CrossRef]
- Takeuchi, K.; Satoh, M.; Kuno, H.; Yoshida, T.; Kondo, H.; Takeuchi, M. Platelet-like particle formation in the human megakaryoblastic leukaemia cell lines, MEG-01 and MEG-01s. Br. J. Haematol. 1998, 100, 436–444. [Google Scholar] [CrossRef]
- Banerjee, A.; Tripathi, A.; Duggal, S.; Banerjee, A.; Vrati, S. Dengue virus infection impedes megakaryopoiesis in MEG-01 cells where the virus envelope protein interacts with the transcription factor TAL-1. Sci. Rep. 2020, 10, 19587. [Google Scholar] [CrossRef]
- Roth, H.; Schneider, L.; Eberle, R.; Lausen, J.; Modlich, U.; Blümel, J.; Baylis, S.A. Zika virus infection studies with CD34+ hematopoietic and megakaryocyte-erythroid progenitors, red blood cells and platelets. Transfusion 2020, 60, 561–574. [Google Scholar] [CrossRef]
- Feng, F.E.; Zhang, G.C.; Liu, F.Q.; He, Y.; Zhu, X.L.; Liu, X.; Wang, Y.; Wang, J.Z.; Fu, H.X.; Chen, Y.H.; et al. HCMV modulates c-Mpl/IEX-1 pathway-mediated megakaryo/thrombopoiesis via PDGFRα and αvβ3 receptors after allo-HSCT. J. Cell. Physiol. 2021, 236, 6726–6741. [Google Scholar] [CrossRef] [PubMed]
- Takeuchi, K.; Ogura, M.; Saito, H.; Satoh, M.; Takeuchi, M. Production of platelet-like particles by a human megakaryoblastic leukemia cell line (MEG-01). Exp. Cell. Res. 1991, 193, 223–226. [Google Scholar] [CrossRef] [PubMed]
- Ogura, M.; Morishima, Y.; Okumura, M.; Hotta, T.; Takamoto, S.; Ohno, R.; Hirabayashi, N.; Nagura, H.; Saito, H. Functional and morphological differentiation induction of a human megakaryoblastic leukemia cell line (MEG-01s) by phorbol diesters. Blood 1988, 72, 49–60. [Google Scholar] [CrossRef] [PubMed]
- Hottz, E.D.; Azevedo-Quintanilha, I.G.; Palhinha, L.; Teixeira, L.; Barreto, E.A.; Pão, C.R.R.; Righy, C.; Franco, S.; Souza, T.M.L.; Kurtz, P.; et al. Platelet activation and platelet-monocyte aggregate formation trigger tissue factor expression in patients with severe COVID-19. Blood 2020, 136, 1330–1341. [Google Scholar] [CrossRef] [PubMed]
- Kirito, K.; Osawa, M.; Morita, H.; Shimizu, R.; Yamamoto, M.; Oda, A.; Fujita, H.; Tanaka, M.; Nakajima, K.; Miura, Y.; et al. A functional role of Stat3 in in vivo megakaryopoiesis. Blood 2002, 99, 3220–3227. [Google Scholar] [CrossRef]
- Singh, S.; Singh, P.K.; Suhail, H.; Arumugaswami, V.; Pellett, P.E.; Giri, S.; Kumar, A. AMP-Activated Protein Kinase Restricts Zika Virus Replication in Endothelial Cells by Potentiating Innate Antiviral Responses and Inhibiting Glycolysis. J. Immunol. 2020, 204, 1810–1824. [Google Scholar] [CrossRef]
- Lahon, A.; Arya, R.P.; Banerjea, A.C. Dengue Virus Dysregulates Master Transcription Factors and PI3K/AKT/mTOR Signaling Pathway in Megakaryocytes. Front. Cell. Infect. Microbiol. 2021, 11, 715208. [Google Scholar] [CrossRef]
- Appelberg, S.; Gupta, S.; Svensson Akusjärvi, S.; Ambikan, A.T.; Mikaeloff, F.; Saccon, E.; Végvári, Á.; Benfeitas, R.; Sperk, M.; Ståhlberg, M. Dysregulation in Akt/mTOR/HIF-1 signaling identified by proteo-transcriptomics of SARS-CoV-2 infected cells. Emerg. Microbes Infect. 2020, 9, 1748–1760. [Google Scholar] [CrossRef]
- Alcami, A.; Koszinowski, U.H. Viral mechanisms of immune evasion. Trends Microbiol. 2000, 8, 410–418. [Google Scholar] [CrossRef]
- Tong, X.; Cheng, A.; Yuan, X.; Zhong, X.; Wang, H.; Zhou, W.; Xu, X.; Li, Y. Characteristics of peripheral white blood cells in COVID-19 patients revealed by a retrospective cohort study. BMC Infect. Dis. 2021, 21, 1236. [Google Scholar] [CrossRef]
- Kronstein-Wiedemann, R.; Stadtmüller, M.; Traikov, S.; Georgi, M.; Teichert, M.; Yosef, H.; Wallenborn, J.; Karl, A.; Schütze, K.; Wagner, M.; et al. SARS-CoV-2 Infects Red Blood Cell Progenitors and Dysregulates Hemoglobin and Iron Metabolism. Stem Cell Rev. Rep. 2022, 18, 1809–1821. [Google Scholar] [CrossRef]
- Aliberti, S.M.; De Caro, F.; Boccia, G.; Caruso, R.; Capunzo, M. Dealing with COVID-19: Lessons Learned from the Italian Experience. Coronaviruses 2021, 2, 27–29. [Google Scholar] [CrossRef]
- Wang, Q.; Anang, S.; Iketani, S.; Guo, Y.; Liu, L.; Katsamba, P.S.; Shapiro, L.; Ho, D.D.; Sodroski, J.G. Functional properties of the spike glycoprotein of the emerging SARS-CoV-2 variant B.1.1.529. Cell Rep. 2022, 39, 110924. [Google Scholar] [CrossRef]
- Lavie, M.; Dubuisson, J.; Belouzard, S. SARS-CoV-2 Spike Furin Cleavage Site and S2’ Basic Residues Modulate the Entry Process in a Host Cell-Dependent Manner. J. Virol. 2022, 96, e0047422. [Google Scholar] [CrossRef]
- Ciaglia, E.; Lopardo, V.; Montella, F.; Sellitto, C.; Manzo, V.; De Bellis, E.; Iannaccone, T.; Franci, G.; Zannella, C.; Pagliano, P.; et al. BPIFB4 Circulating Levels and Its Prognostic Relevance in COVID-19. J. Gerontol A Biol. Sci. Med. Sci. 2021, 76, 1775–1783. [Google Scholar] [CrossRef]
- Bester, J.; Pretorius, E. Effects of IL-1β, IL-6 and IL-8 on erythrocytes, platelets and clot viscoelasticity. Sci. Rep. 2016, 6, 32188. [Google Scholar] [CrossRef]
- Amodio, G.; Margarucci, L.; Moltedo, O.; Casapullo, A.; Remondelli, P. Identification of Cysteine Ubiquitylation Sites on the Sec23A Protein of the COPII Complex Required for Vesicle Formation from the ER. Open Biochem. J. 2017, 11, 36–46. [Google Scholar] [CrossRef]
- Heestermans, M.; Poenou, G.; Duchez, A.C.; Hamzeh-Cognasse, H.; Bertoletti, L.; Cognasse, F. Immunothrombosis and the Role of Platelets in Venous Thromboembolic Diseases. Int. J. Mol. Sci. 2022, 23, 13176. [Google Scholar] [CrossRef]
- Barrett, T.J.; Cornwell, M.; Myndzar, K.; Rolling, C.C.; Xia, Y.; Drenkova, K.; Biebuyck, A.; Fields, A.T.; Tawil, M.; Luttrell-Williams, E.; et al. Platelets amplify endotheliopathy in COVID-19. Sci. Adv. 2021, 7, eabh2434. [Google Scholar] [CrossRef]
- Cohen, A.; Harari, E.; Yahud, E.; Cipok, M.; Bryk, G.; Lador, N.K.; Mann, T.; Mayo, A.; Lev, E.I. Immature platelets in patients with COVID-19: Association with disease severity. J. Thromb. Thrombolysis. 2021, 52, 708–714. [Google Scholar] [CrossRef]
- Ranjit Banwait, R.; Marachi, A.; Iyer, U.G. Thrombocytopenia Associated with Increase in Disease Severity and Mortality in COVID-19 Patients. Blood 2020, 136, 1. [Google Scholar] [CrossRef]
- Yang, M.; Liang, E.Y.; Zhou, L.X.; Chong, B.-H.; Li, C. Lung Damage and Thrombocytopenia. Blood 2015, 126, 4638. [Google Scholar] [CrossRef]
- Huang, Z.; Richmond, T.D.; Muntean, A.G.; Barber, D.L.; Weiss, M.J.; Crispino, J.D. STAT1 promotes megakaryopoiesis downstream of GATA-1 in mice. J. Clin. Invest. 2007, 117, 3890–3899. [Google Scholar] [CrossRef] [PubMed]
- Bhutta, M.S.; Gallo, E.S.; Borenstein, R. Multifaceted Role of AMPK in Viral Infections. Cells 2021, 10, 1118. [Google Scholar] [CrossRef] [PubMed]
- Goto, K.; Lin, W.; Zhang, L.; Jilg, N.; Shao, R.X.; Schaefer, E.A.; Zhao, H.; Fusco, D.N.; Peng, L.F.; Kato, N.; et al. The AMPK-related kinase SNARK regulates hepatitis C virus replication and pathogenesis through enhancement of TGF-β signaling. J. Hepatol. 2013, 59, 942–948. [Google Scholar] [CrossRef]
- Zhang, H.S.; Wu, M.R. SIRT1 regulates Tat-induced HIV-1 transactivation through activating AMP-activated protein kinase. Virus Res. 2009, 146, 51–57. [Google Scholar] [CrossRef]
- Xin, L.; Ma, X.; Xiao, Z.; Yao, H.; Liu, Z. Coxsackievirus B3 induces autophagy in HeLa cells via the AMPK/MEK/ERK and Ras/Raf/MEK/ERK signaling pathways. Infect. Genet. Evol. 2015, 36, 46–54. [Google Scholar] [CrossRef]
- Zhao, C.; Chen, J.; Cheng, L.; Xu, K.; Yang, Y.; Su, X. Deficiency of HIF-1α enhances influenza A virus replication by promoting autophagy in alveolar type II epithelial cells. Emerg. Microbes Infect. 2020, 9, 691–706. [Google Scholar] [CrossRef]
- Reuschl, A.; Thorne, L.G.; Whelan, M.V.X.; Mesner, D.; Ragazzini, R.; Dowgier, G.; Bogoda, N.; Turner, J.L.E.; Furnon, W.; Cowton, V.M.; et al. Enhanced innate immune suppression by SARS-CoV-2 Omicron subvariants BA.4 and BA.5. BioRxviv 2022. [Google Scholar] [CrossRef]
- Saichi, M.; Ladjemi, M.Z.; Korniotis, S.; Rousseau, C.; Ait Hamou, Z.; Massenet-Regad, L.; Amblard, E.; Noel, F.; Marie, Y.; Bouteiller, D.; et al. Single-cell RNA sequencing of blood antigen-presenting cells in severe COVID-19 reveals multi-process defects in antiviral immunity. Nat. Cell. Biol. 2021, 23, 538–551. [Google Scholar] [CrossRef]
- Welch, S.R.; Davies, K.A.; Buczkowski, H.; Hettiarachchi, N.; Green, N.; Arnold, U.; Jones, M.; Hannah, M.J.; Evans, R.; Burton, C.; et al. Analysis of Inactivation of SARS-CoV-2 by Specimen Transport Media, Nucleic Acid Extraction Reagents, Detergents, and Fixatives. J. Clin. Microbiol. 2020, 58, e01713–e01720. [Google Scholar] [CrossRef]
- Batéjat, C.; Grassin, Q.; Manuguerra, J.C.; Leclercq, I. Heat inactivation of the severe acute respiratory syndrome coronavirus 2. J. Biosaf. Biosecur. 2021, 3, 1–3. [Google Scholar] [CrossRef]
- Ciaglia, E.; Pisanti, S.; Picardi, P.; Laezza, C.; Sosa, S.; Tubaro, A.; Vitale, M.; Gazzerro, P.; Malfitano, A.M.; Bifulco, M. N6-isopentenyladenosine affects cytotoxic activity and cytokines production by IL-2 activated NK cells and exerts topical anti-inflammatory activity in mice. Pharmacol. Res. 2014, 89, 1–10. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lopardo, V.; Montella, F.; Esposito, R.M.; Zannella, C.; Aliberti, S.M.; Capunzo, M.; Franci, G.; Puca, A.A.; Ciaglia, E. SARS-CoV-2 Lysate Stimulation Impairs the Release of Platelet-like Particles and Megakaryopoiesis in the MEG-01 Cell Line. Int. J. Mol. Sci. 2023, 24, 4723. https://doi.org/10.3390/ijms24054723
Lopardo V, Montella F, Esposito RM, Zannella C, Aliberti SM, Capunzo M, Franci G, Puca AA, Ciaglia E. SARS-CoV-2 Lysate Stimulation Impairs the Release of Platelet-like Particles and Megakaryopoiesis in the MEG-01 Cell Line. International Journal of Molecular Sciences. 2023; 24(5):4723. https://doi.org/10.3390/ijms24054723
Chicago/Turabian StyleLopardo, Valentina, Francesco Montella, Roberta Maria Esposito, Carla Zannella, Silvana Mirella Aliberti, Mario Capunzo, Gianluigi Franci, Annibale Alessandro Puca, and Elena Ciaglia. 2023. "SARS-CoV-2 Lysate Stimulation Impairs the Release of Platelet-like Particles and Megakaryopoiesis in the MEG-01 Cell Line" International Journal of Molecular Sciences 24, no. 5: 4723. https://doi.org/10.3390/ijms24054723
APA StyleLopardo, V., Montella, F., Esposito, R. M., Zannella, C., Aliberti, S. M., Capunzo, M., Franci, G., Puca, A. A., & Ciaglia, E. (2023). SARS-CoV-2 Lysate Stimulation Impairs the Release of Platelet-like Particles and Megakaryopoiesis in the MEG-01 Cell Line. International Journal of Molecular Sciences, 24(5), 4723. https://doi.org/10.3390/ijms24054723








