Environmental Enrichment Protects against Neurotoxic Effects of Lipopolysaccharide: A Comprehensive Overview
Abstract
:1. Introduction
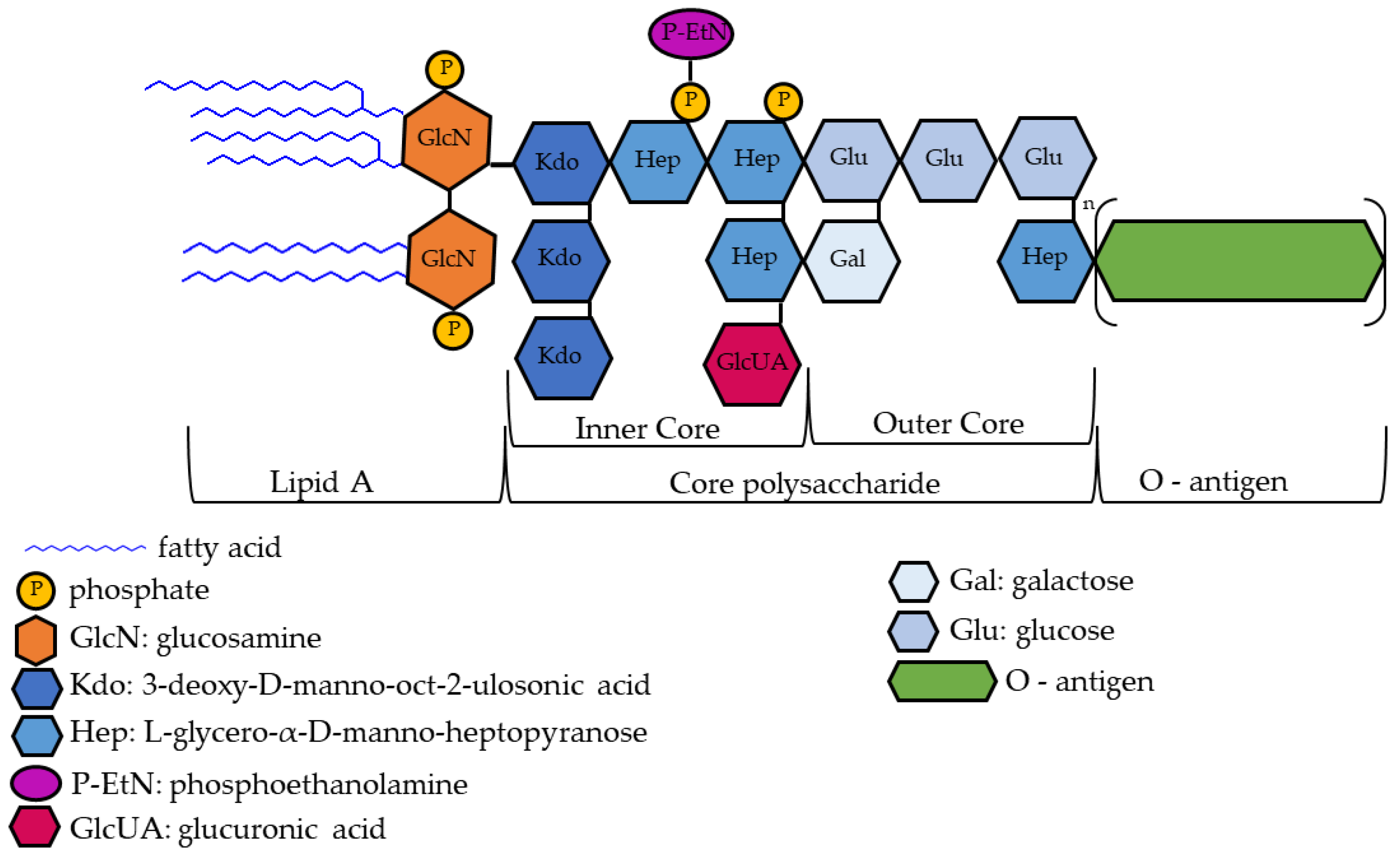
1.1. Lipopolysaccharide (LPS)
1.2. Environmental Enrichment (EE)
2. Methodology of the Literature Search
- As the population of interest, we selected rodents, both healthy and pathological models, with no sex or age restrictions.
- As the intervention of interest, we selected multidimensional EE exposure of animals submitted to LPS treatment carried out in any period of life, before or after the exposure to EE. For models based on transgenerational and MIA models, studies on enriched mothers and LPS-injected pups, and vice versa, were also considered.
- As the control group of interest, we selected animals submitted to LPS treatment and reared under standard laboratory conditions.
- As outcomes of interest, we selected cognitive, emotional, motor, and social effects and related structural, physiological, or biochemical processes.
3. Beneficial Effects of Exposure to EE on Neuroinflammation Induced by LPS Injections
- The first section reports the findings related to the beneficial effects of EE in case of prenatal neuroinflammation induced by LPS injections. The beneficial effects provided by exposure to EE for mother and offspring, respectively, are described.
- The second section is devoted to the beneficial effects of EE in case of neuroinflammation induced by LPS injections in early age.
- Finally, the third section includes the beneficial effects of EE in case of neuroinflammation induced by LPS injections in adult age.
3.1. Beneficial Effects of Exposure to EE on Mothers and Offspring in Case of Gestational Neuroinflammation Induced by LPS Injection
3.1.1. Beneficial Effects of the Mother’s Exposure to EE in Case of Gestational Inflammation Induced by LPS Injection
3.1.2. Beneficial Effects of the Offspring’s Exposure of EE in Case of Gestational Inflammation Induced by LPS Injection
3.2. Beneficial Effects of Exposure to EE in Case of Early Postnatal Neuroinflammation Induced by LPS Injection
3.3. Beneficial Effects of Exposure to EE in case of Neuroinflammation Induced in Adulthood by LPS Injection
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Spencer, P.S.; Lein, P.J. Neurotoxicity. In Encyclopedia of Toxicology, 3rd ed.; Wexler, P., Ed.; Elsevier Inc.; Academic Press: Amsterdam, The Netherlands, 2014; Volume 3, pp. 489–500. ISBN 9780123864543. [Google Scholar] [CrossRef]
- Bilge, S. Neurotoxicity, Types, Clinical Manifestations, Diagnosis and Treatment. In Neurotoxicity-New Advances; IntechOpen: London, UK, 2022; ISBN 978-1-83968-970-3. [Google Scholar]
- Dorman, D.C. An Integrative Approach to Neurotoxicology. Toxicol. Pathol. 2000, 28, 37–42. [Google Scholar] [CrossRef] [Green Version]
- Popoff, M.R.; Poulain, B. Bacterial Toxins and the Nervous System: Neurotoxins and Multipotential Toxins Interacting with Neuronal Cells. Toxins 2010, 2, 683–737. [Google Scholar] [CrossRef] [Green Version]
- Van Belleghem, J.D.; Dąbrowska, K.; Vaneechoutte, M.; Barr, J.J.; Bollyky, P.L. Interactions between Bacteriophage, Bacteria, and the Mammalian Immune System. Viruses 2018, 11, 10. [Google Scholar] [CrossRef] [Green Version]
- Bertani, B.; Ruiz, N. Function and Biogenesis of Lipopolysaccharides. EcoSal Plus 2018, 8. [Google Scholar] [CrossRef]
- Lukiw, W.J.; Arceneaux, L.; Li, W.; Bond, T.; Zhao, Y. Gastrointestinal (GI)-Tract Microbiome Derived Neurotoxins and Their Potential Contribution to Inflammatory Neurodegeneration in Alzheimer’s Disease (AD). J. Alzheimers Dis. Parkinsonism 2021, 11, 525. [Google Scholar]
- Zhao, Y.; Jaber, V.R.; Pogue, A.I.; Sharfman, N.M.; Taylor, C.; Lukiw, W.J. Lipopolysaccharides (LPSs) as Potent Neurotoxic Glycolipids in Alzheimer’s Disease (AD). Int. J. Mol. Sci. 2022, 23, 12671. [Google Scholar] [CrossRef]
- Raetz, C.R.H.; Whitfield, C. Lipopolysaccharide Endotoxins. Annu. Rev. Biochem. 2002, 71, 635–700. [Google Scholar] [CrossRef] [Green Version]
- Maldonado, R.F.; Sá-Correia, I.; Valvano, M.A. Lipopolysaccharide Modification in Gram-Negative Bacteria during Chronic Infection. FEMS Microbiol. Rev. 2016, 40, 480–493. [Google Scholar] [CrossRef] [Green Version]
- Farhana, A.; Khan, Y.S. Biochemistry, Lipopolysaccharide. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2022. [Google Scholar]
- Allen, K.N.; Imperiali, B. Structural and Mechanistic Themes in Glycoconjugate Biosynthesis at Membrane Interfaces. Curr. Opin. Struct. Biol. 2019, 59, 81–90. [Google Scholar] [CrossRef]
- Gorzelak, P.; Klein, G.; Raina, S. Molecular Basis of Essentiality of Early Critical Steps in the Lipopolysaccharide Biogenesis in Escherichia coli K-12: Requirement of MsbA, Cardiolipin, LpxL, LpxM and GcvB. Int. J. Mol. Sci. 2021, 22, 5099. [Google Scholar] [CrossRef]
- Holst, O. Chemical Structure of the Core Region of Lipopolysaccharides; CRC Press: Boca Raton, FL, USA, 2020; pp. 115–154. ISBN 978-1-00-306496-1. [Google Scholar]
- Whitfield, C.; Williams, D.M.; Kelly, S.D. Lipopolysaccharide O-Antigens-Bacterial Glycans Made to Measure. J. Biol. Chem. 2020, 295, 10593–10609. [Google Scholar] [CrossRef]
- Klein, G.; Raina, S. Regulated Assembly of LPS, Its Structural Alterations and Cellular Response to LPS Defects. Int. J. Mol. Sci. 2019, 20, 356. [Google Scholar] [CrossRef] [Green Version]
- Han, V.X.; Patel, S.; Jones, H.F.; Nielsen, T.C.; Mohammad, S.S.; Hofer, M.J.; Gold, W.; Brilot, F.; Lain, S.J.; Nassar, N.; et al. Maternal Acute and Chronic Inflammation in Pregnancy Is Associated with Common Neurodevelopmental Disorders: A Systematic Review. Transl. Psychiatry 2021, 11, 71. [Google Scholar] [CrossRef]
- Bossù, P.; Cutuli, D.; Palladino, I.; Caporali, P.; Angelucci, F.; Laricchiuta, D.; Gelfo, F.; De Bartolo, P.; Caltagirone, C.; Petrosini, L. A Single Intraperitoneal Injection of Endotoxin in Rats Induces Long-Lasting Modifications in Behavior and Brain Protein Levels of TNF-α and IL-18. J. Neuroinflam. 2012, 9, 101. [Google Scholar] [CrossRef]
- Ferrucci, L.; Fabbri, E. Inflammageing: Chronic Inflammation in Ageing, Cardiovascular Disease, and Frailty. Nat. Rev. Cardiol. 2018, 15, 505–522. [Google Scholar] [CrossRef]
- Tsukamoto, H.; Takeuchi, S.; Kubota, K.; Kobayashi, Y.; Kozakai, S.; Ukai, I.; Shichiku, A.; Okubo, M.; Numasaki, M.; Kanemitsu, Y.; et al. Lipopolysaccharide (LPS)-Binding Protein Stimulates CD14-Dependent Toll-like Receptor 4 Internalization and LPS-Induced TBK1-IKKϵ-IRF3 Axis Activation. J. Biol. Chem. 2018, 293, 10186–10201. [Google Scholar] [CrossRef] [Green Version]
- Beutler, B.; Cerami, A. Tumor Necrosis, Cachexia, Shock, and Inflammation: A Common Mediator. Annu. Rev. Biochem. 1988, 57, 505–518. [Google Scholar] [CrossRef]
- Dinarello, C.A. Interleukin-1 and Interleukin-1 Antagonism. Blood 1991, 77, 1627–1652. [Google Scholar] [CrossRef] [Green Version]
- Medzhitov, R.; Janeway, C. Innate Immunity. N. Engl. J. Med. 2000, 343, 338–344. [Google Scholar] [CrossRef]
- Dantzer, R. Cytokine-Induced Sickness Behavior: Where Do We Stand? Brain Behav. Immun. 2001, 15, 7–24. [Google Scholar] [CrossRef] [Green Version]
- Giandomenico, S.L.; Sutcliffe, M.; Lancaster, M.A. Generation and Long-Term Culture of Advanced Cerebral Organoids for Studying Later Stages of Neural Development. Nat. Protoc. 2021, 16, 579–602. [Google Scholar] [CrossRef] [PubMed]
- Guzzetta, A.; Murray, L.; Montirosso, R.; Ferrari, P.F. The Role of the Social Environment on Adaptive Neuroplasticity in Early Development. Neural Plast 2019, 2019, 5375849. [Google Scholar] [CrossRef] [PubMed]
- Serra, L.; Gelfo, F.; Petrosini, L.; Di Domenico, C.; Bozzali, M.; Caltagirone, C. Rethinking the Reserve with a Translational Approach: Novel Ideas on the Construct and the Interventions. J. Alzheimers Dis. 2018, 65, 1065–1078. [Google Scholar] [CrossRef]
- Stern, Y.; Arenaza-Urquijo, E.M.; Bartrés-Faz, D.; Belleville, S.; Cantilon, M.; Chetelat, G.; Ewers, M.; Franzmeier, N.; Kempermann, G.; Kremen, W.S.; et al. Whitepaper: Defining and Investigating Cognitive Reserve, Brain Reserve, and Brain Maintenance. Alzheimers Dement 2020, 16, 1305–1311. [Google Scholar] [CrossRef] [PubMed]
- Cutuli, D.; Landolfo, E.; Petrosini, L.; Gelfo, F. Environmental Enrichment Effects on the Brain-Derived Neurotrophic Factor Expression in Healthy Condition, Alzheimer’s Disease, and Other Neurodegenerative Disorders. J. Alzheimers Dis. 2022, 85, 975–992. [Google Scholar] [CrossRef]
- Gelfo, F.; Petrosini, L. Environmental Enrichment Enhances Cerebellar Compensation and Develops Cerebellar Reserve. Int. J. Environ. Res. Public Health 2022, 19, 5697. [Google Scholar] [CrossRef]
- Hebb, H.D. The Effects of Early Experience on Problem-Solving at Maturity. Am. Psychol. 1947, 2, 306–307. [Google Scholar]
- Rosenzweig, M.R.; Bennett, E.L.; Hebert, M.; Morimoto, H. Social Grouping Cannot Account for Cerebral Effects of Enriched Environments. Brain Res. 1978, 153, 563–576. [Google Scholar] [CrossRef]
- Redolat, R.; Mesa-Gresa, P. Potential Benefits and Limitations of Enriched Environments and Cognitive Activity on Age-Related Behavioural Decline. Curr. Top. Behav. Neurosci. 2012, 10, 293–316. [Google Scholar] [CrossRef]
- van Praag, H.; Kempermann, G.; Gage, F.H. Neural Consequences of Environmental Enrichment. Nat. Rev. Neurosci. 2000, 1, 191–198. [Google Scholar] [CrossRef]
- Liew, A.K.Y.; Teo, C.H.; Soga, T. The Molecular Effects of Environmental Enrichment on Alzheimer’s Disease. Mol. Neurobiol. 2022, 59, 7095–7118. [Google Scholar] [CrossRef]
- Balietti, M.; Conti, F. Environmental Enrichment and the Aging Brain: Is It Time for Standardization? Neurosci. Biobehav. Rev. 2022, 139, 104728. [Google Scholar] [CrossRef]
- Fratiglioni, L.; Paillard-Borg, S.; Winblad, B. An Active and Socially Integrated Lifestyle in Late Life Might Protect against Dementia. Lancet Neurol. 2004, 3, 343–353. [Google Scholar] [CrossRef]
- Karp, A.; Paillard-Borg, S.; Wang, H.-X.; Silverstein, M.; Winblad, B.; Fratiglioni, L. Mental, Physical and Social Components in Leisure Activities Equally Contribute to Decrease Dementia Risk. Dement Geriatr. Cogn. Disord. 2006, 21, 65–73. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mangialasche, F.; Kivipelto, M.; Solomon, A.; Fratiglioni, L. Dementia Prevention: Current Epidemiological Evidence and Future Perspective. Alzheimers Res. Ther. 2012, 4, 6. [Google Scholar] [CrossRef] [PubMed]
- Gelfo, F.; Mandolesi, L.; Serra, L.; Sorrentino, G.; Caltagirone, C. The Neuroprotective Effects of Experience on Cognitive Functions: Evidence from Animal Studies on the Neurobiological Bases of Brain Reserve. Neuroscience 2018, 370, 218–235. [Google Scholar] [CrossRef]
- Qiu, C.; Kivipelto, M.; von Strauss, E. Epidemiology of Alzheimer’s Disease: Occurrence, Determinants, and Strategies toward Intervention. Dialogues Clin. Neurosci. 2009, 11, 111–128. [Google Scholar] [CrossRef]
- Fratiglioni, L.; Wang, H.-X. Brain Reserve Hypothesis in Dementia. J. Alzheimers Dis. 2007, 12, 11–22. [Google Scholar] [CrossRef]
- Wang, H.-X.; Xu, W.; Pei, J.-J. Leisure Activities, Cognition and Dementia. Biochim Biophys Acta 2012, 1822, 482–491. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Caracciolo, B.; Xu, W.; Collins, S.; Fratiglioni, L. Cognitive Decline, Dietary Factors and Gut-Brain Interactions. Mech. Ageing Dev. 2014, 136–137, 59–69. [Google Scholar] [CrossRef] [PubMed]
- Cutuli, D. Functional and Structural Benefits Induced by Omega-3 Polyunsaturated Fatty Acids During Aging. Curr. Neuropharmacol. 2017, 15, 534–542. [Google Scholar] [CrossRef] [Green Version]
- Mandolesi, L.; Polverino, A.; Montuori, S.; Foti, F.; Ferraioli, G.; Sorrentino, P.; Sorrentino, G. Effects of Physical Exercise on Cognitive Functioning and Wellbeing: Biological and Psychological Benefits. Front. Psychol. 2018, 9, 509. [Google Scholar] [CrossRef] [Green Version]
- Serra, L.; Gelfo, F. What Good Is the Reserve? A Translational Perspective for the Managing of Cognitive Decline. Neural Regen Res. 2019, 14, 1219–1220. [Google Scholar] [CrossRef]
- Serra, L.; Petrosini, L.; Mandolesi, L.; Bonarota, S.; Balsamo, F.; Bozzali, M.; Caltagirone, C.; Gelfo, F. Walking, Running, Swimming: An Analysis of the Effects of Land and Water Aerobic Exercises on Cognitive Functions and Neural Substrates. Int. J. Environ. Res. Public Health 2022, 19, 16310. [Google Scholar] [CrossRef] [PubMed]
- Markham, J.A.; Greenough, W.T. Experience-Driven Brain Plasticity: Beyond the Synapse. Neuron Glia Biol. 2004, 1, 351–363. [Google Scholar] [CrossRef] [Green Version]
- Simpson, J.; Kelly, J.P. The Impact of Environmental Enrichment in Laboratory Rats--Behavioural and Neurochemical Aspects. Behav. Brain Res. 2011, 222, 246–264. [Google Scholar] [CrossRef]
- Cintoli, S.; Cenni, M.C.; Pinto, B.; Morea, S.; Sale, A.; Maffei, L.; Berardi, N. Environmental Enrichment Induces Changes in Long-Term Memory for Social Transmission of Food Preference in Aged Mice through a Mechanism Associated with Epigenetic Processes. Neural Plast 2018, 2018, 3725087. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gelfo, F. Does Experience Enhance Cognitive Flexibility? An Overview of the Evidence Provided by the Environmental Enrichment Studies. Front. Behav. Neurosci. 2019, 13, 150. [Google Scholar] [CrossRef] [PubMed]
- Kentner, A.C.; Lambert, K.G.; Hannan, A.J.; Donaldson, S.T. Editorial: Environmental Enrichment: Enhancing Neural Plasticity, Resilience, and Repair. Front. Behav. Neurosci. 2019, 13, 75. [Google Scholar] [CrossRef]
- Petrosini, L.; De Bartolo, P.; Foti, F.; Gelfo, F.; Cutuli, D.; Leggio, M.G.; Mandolesi, L. On Whether the Environmental Enrichment May Provide Cognitive and Brain Reserves. Brain Res. Rev. 2009, 61, 221–239. [Google Scholar] [CrossRef]
- Caporali, P.; Cutuli, D.; Gelfo, F.; Laricchiuta, D.; Foti, F.; De Bartolo, P.; Mancini, L.; Angelucci, F.; Petrosini, L. Pre-Reproductive Maternal Enrichment Influences Offspring Developmental Trajectories: Motor Behavior and Neurotrophin Expression. Front. Behav. Neurosci. 2014, 8, 195. [Google Scholar] [CrossRef] [Green Version]
- Cutuli, D.; Caporali, P.; Gelfo, F.; Angelucci, F.; Laricchiuta, D.; Foti, F.; De Bartolo, P.; Bisicchia, E.; Molinari, M.; Farioli Vecchioli, S.; et al. Pre-Reproductive Maternal Enrichment Influences Rat Maternal Care and Offspring Developmental Trajectories: Behavioral Performances and Neuroplasticity Correlates. Front. Behav. Neurosci. 2015, 9, 66. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cutuli, D.; Berretta, E.; Pasqualini, G.; De Bartolo, P.; Caporali, P.; Laricchiuta, D.; Sampedro-Piquero, P.; Gelfo, F.; Pesoli, M.; Foti, F.; et al. Influence of Pre-Reproductive Maternal Enrichment on Coping Response to Stress and Expression of c-Fos and Glucocorticoid Receptors in Adolescent Offspring. Front. Behav. Neurosci. 2017, 11, 73. [Google Scholar] [CrossRef] [Green Version]
- Cutuli, D.; Berretta, E.; Laricchiuta, D.; Caporali, P.; Gelfo, F.; Petrosini, L. Pre-Reproductive Parental Enriching Experiences Influence Progeny’s Developmental Trajectories. Front. Behav. Neurosci. 2018, 12, 254. [Google Scholar] [CrossRef]
- Riyahi, J.; Abdoli, B.; Gelfo, F.; Petrosini, L.; Rezaei, R.; Haghparast, A. Maternal Spatial Training before Fertilization Improves the Spatial Learning Process in Female Offspring. Neuroreport 2021, 32, 1106–1112. [Google Scholar] [CrossRef] [PubMed]
- Kempermann, G.; Kuhn, H.G.; Gage, F.H. More Hippocampal Neurons in Adult Mice Living in an Enriched Environment. Nature 1997, 386, 493–495. [Google Scholar] [CrossRef]
- Kempermann, G.; Kuhn, H.G.; Gage, F.H. Experience-Induced Neurogenesis in the Senescent Dentate Gyrus. J. Neurosci. 1998, 18, 3206–3212. [Google Scholar] [CrossRef] [Green Version]
- Kempermann, G.; Gast, D.; Gage, F.H. Neuroplasticity in Old Age: Sustained Fivefold Induction of Hippocampal Neurogenesis by Long-Term Environmental Enrichment. Ann. Neurol. 2002, 52, 135–143. [Google Scholar] [CrossRef]
- Segovia, G.; Yagüe, A.G.; García-Verdugo, J.M.; Mora, F. Environmental Enrichment Promotes Neurogenesis and Changes the Extracellular Concentrations of Glutamate and GABA in the Hippocampus of Aged Rats. Brain Res. Bull. 2006, 70, 8–14. [Google Scholar] [CrossRef]
- Leal-Galicia, P.; Castañeda-Bueno, M.; Quiroz-Baez, R.; Arias, C. Long-Term Exposure to Environmental Enrichment since Youth Prevents Recognition Memory Decline and Increases Synaptic Plasticity Markers in Aging. Neurobiol. Learn. Mem. 2008, 90, 511–518. [Google Scholar] [CrossRef] [PubMed]
- Speisman, R.B.; Kumar, A.; Rani, A.; Pastoriza, J.M.; Severance, J.E.; Foster, T.C.; Ormerod, B.K. Environmental Enrichment Restores Neurogenesis and Rapid Acquisition in Aged Rats. Neurobiol. Aging 2013, 34, 263–274. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 Statement: An Updated Guideline for Reporting Systematic Reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef]
- Boulanger-Bertolus, J.; Pancaro, C.; Mashour, G.A. Increasing Role of Maternal Immune Activation in Neurodevelopmental Disorders. Front. Behav. Neurosci. 2018, 12, 230. [Google Scholar] [CrossRef] [Green Version]
- Knuesel, I.; Chicha, L.; Britschgi, M.; Schobel, S.A.; Bodmer, M.; Hellings, J.A.; Toovey, S.; Prinssen, E.P. Maternal Immune Activation and Abnormal Brain Development across CNS Disorders. Nat. Rev. Neurol. 2014, 10, 643–660. [Google Scholar] [CrossRef]
- Paraschivescu, C.; Barbosa, S.; Van Steenwinckel, J.; Gressens, P.; Glaichenhaus, N.; Davidovic, L. Early Life Exposure to Tumor Necrosis Factor Induces Precocious Sensorimotor Reflexes Acquisition and Increases Locomotor Activity During Mouse Postnatal Development. Front. Behav. Neurosci. 2022, 16, 845458. [Google Scholar] [CrossRef] [PubMed]
- Kępińska, A.P.; MacCabe, J.H.; Cadar, D.; Steptoe, A.; Murray, R.M.; Ajnakina, O. Schizophrenia Polygenic Risk Predicts General Cognitive Deficit but Not Cognitive Decline in Healthy Older Adults. Transl. Psychiatry 2020, 10, 422. [Google Scholar] [CrossRef] [PubMed]
- Purves-Tyson, T.D.; Brown, A.M.; Weissleder, C.; Rothmond, D.A.; Shannon Weickert, C. Reductions in Midbrain GABAergic and Dopamine Neuron Markers Are Linked in Schizophrenia. Mol. Brain 2021, 14, 96. [Google Scholar] [CrossRef]
- Davis, J.; Mire, E. Maternal Obesity and Developmental Programming of Neuropsychiatric Disorders: An Inflammatory Hypothesis. Brain Neurosci. Adv. 2021, 5, 23982128211003484. [Google Scholar] [CrossRef]
- Chen, S.-W.; Zhong, X.-S.; Jiang, L.-N.; Zheng, X.-Y.; Xiong, Y.-Q.; Ma, S.-J.; Qiu, M.; Huo, S.-T.; Ge, J.; Chen, Q. Maternal Autoimmune Diseases and the Risk of Autism Spectrum Disorders in Offspring: A Systematic Review and Meta-Analysis. Behav. Brain Res. 2016, 296, 61–69. [Google Scholar] [CrossRef]
- Gong, T.; Lundholm, C.; Rejnö, G.; Bölte, S.; Larsson, H.; D’Onofrio, B.M.; Lichtenstein, P.; Almqvist, C. Parental Asthma and Risk of Autism Spectrum Disorder in Offspring: A Population and Family-Based Case-Control Study. Clin. Exp. Allergy 2019, 49, 883–891. [Google Scholar] [CrossRef] [Green Version]
- Careaga, M.; Murai, T.; Bauman, M.D. Maternal Immune Activation and Autism Spectrum Disorder: From Rodents to Nonhuman and Human Primates. Biol. Psychiatry 2017, 81, 391–401. [Google Scholar] [CrossRef] [Green Version]
- Baines, K.J.; Hillier, D.M.; Haddad, F.L.; Rajakumar, N.; Schmid, S.; Renaud, S.J. Maternal Immune Activation Alters Fetal Brain Development and Enhances Proliferation of Neural Precursor Cells in Rats. Front. Immunol. 2020, 11, 1145. [Google Scholar] [CrossRef]
- Cheslack-Postava, K.; Brown, A.S. Prenatal Infection and Schizophrenia: A Decade of Further Progress. Schizophr. Res. 2022, 247, 7–15. [Google Scholar] [CrossRef]
- Massrali, A.; Adhya, D.; Srivastava, D.P.; Baron-Cohen, S.; Kotter, M.R. Virus-Induced Maternal Immune Activation as an Environmental Factor in the Etiology of Autism and Schizophrenia. Front. Neurosci. 2022, 16, 834058. [Google Scholar] [CrossRef]
- Patel, S.; Dale, R.C.; Rose, D.; Heath, B.; Nordahl, C.W.; Rogers, S.; Guastella, A.J.; Ashwood, P. Maternal Immune Conditions Are Increased in Males with Autism Spectrum Disorders and Are Associated with Behavioural and Emotional but Not Cognitive Co-Morbidity. Transl. Psychiatry 2020, 10, 286. [Google Scholar] [CrossRef]
- Mueller, F.S.; Scarborough, J.; Schalbetter, S.M.; Richetto, J.; Kim, E.; Couch, A.; Yee, Y.; Lerch, J.P.; Vernon, A.C.; Weber-Stadlbauer, U.; et al. Behavioral, Neuroanatomical, and Molecular Correlates of Resilience and Susceptibility to Maternal Immune Activation. Mol. Psychiatry 2021, 26, 396–410. [Google Scholar] [CrossRef]
- Langdon, K.D.; Corbett, D. Improved Working Memory Following Novel Combinations of Physical and Cognitive Activity. Neurorehabil. Neural Repair. 2012, 26, 523–532. [Google Scholar] [CrossRef]
- Hegaard, H.K.; Pedersen, B.K.; Nielsen, B.B.; Damm, P. Leisure Time Physical Activity during Pregnancy and Impact on Gestational Diabetes Mellitus, Pre-Eclampsia, Preterm Delivery and Birth Weight: A Review. Acta Obstet. Gynecol. Scand. 2007, 86, 1290–1296. [Google Scholar] [CrossRef]
- Hegaard, H.K.; Hedegaard, M.; Damm, P.; Ottesen, B.; Petersson, K.; Henriksen, T.B. Leisure Time Physical Activity Is Associated with a Reduced Risk of Preterm Delivery. Am. J. Obstet. Gynecol. 2008, 198, 180.e1–180.e5. [Google Scholar] [CrossRef]
- Schander, J.A.; Aisemberg, J.; Correa, F.; Wolfson, M.L.; Juriol, L.; Cymeryng, C.; Jensen, F.; Franchi, A.M. The Enrichment of Maternal Environment Prevents Pre-Term Birth in a Mice Model. Reproduction 2020, 159, 479–492. [Google Scholar] [CrossRef]
- Schander, J.A.; Marvaldi, C.; Correa, F.; Wolfson, M.L.; Cella, M.; Aisemberg, J.; Jensen, F.; Franchi, A.M. Maternal Environmental Enrichment Modulates the Immune Response against an Inflammatory Challenge during Gestation and Protects the Offspring. J. Reprod. Immunol. 2021, 144, 103273. [Google Scholar] [CrossRef] [PubMed]
- Zhuang, Z.-Q.; Zhang, Z.-Z.; Zhang, Y.-M.; Ge, H.-H.; Sun, S.-Y.; Zhang, P.; Chen, G.-H. A Long-Term Enriched Environment Ameliorates the Accelerated Age-Related Memory Impairment Induced by Gestational Administration of Lipopolysaccharide: Role of Plastic Mitochondrial Quality Control. Front. Cell Neurosci. 2021, 14, 559182. [Google Scholar] [CrossRef]
- Connors, E.J.; Shaik, A.N.; Migliore, M.M.; Kentner, A.C. Environmental Enrichment Mitigates the Sex-Specific Effects of Gestational Inflammation on Social Engagement and the Hypothalamic Pituitary Adrenal Axis-Feedback System. Brain Behav. Immun. 2014, 42, 178–190. [Google Scholar] [CrossRef]
- Rajesh, T.; Tantradi, R.R.; Rai, K.S.; Gopalakrishnan, S.; Ramesh Babu, M.G.; Adiga, M.; Huban Thomas, R.; Bhaktavatchalam, P. Prenatal Inflammation Induced Alterations in Spatial Learning and Memory Abilities in Adult Offspring: Mitigated by Physical Exercise and Environmental Enrichment. Res. J. Pharm. Biol. Chem. Sci. 2016, 7, 1681–1688. [Google Scholar]
- Rajesh, T.; Tantradi, R.R.; Rai, K.S.; Thomas, R.H.; Konuri, A. Additive Effects of Physical Exercise and Environmental Enrichment in Attenuating Alterations in the Hippocampal Neuronal Morphology of Adult Wistar Rats Induced by Prenatal Inflammations. Jordan J. Biol. Sci. 2018, 11, 563–570. [Google Scholar]
- Bakos, J.; Duncko, R.; Makatsori, A.; Pirnik, Z.; Kiss, A.; Jezova, D. Prenatal Immune Challenge Affects Growth, Behavior, and Brain Dopamine in Offspring. Ann. N. Y. Acad. Sci. 2004, 1018, 281–287. [Google Scholar] [CrossRef]
- Kentner, A.C.; Khoury, A.; Lima Queiroz, E.; MacRae, M. Environmental Enrichment Rescues the Effects of Early Life Inflammation on Markers of Synaptic Transmission and Plasticity. Brain Behav. Immun. 2016, 57, 151–160. [Google Scholar] [CrossRef]
- Wu, Y.-F.; Zhang, Y.-M.; Ge, H.-H.; Ren, C.-Y.; Zhang, Z.-Z.; Cao, L.; Wang, F.; Chen, G.-H. Effects of Embryonic Inflammation and Adolescent Psychosocial Environment on Cognition and Hippocampal Staufen in Middle-Aged Mice. Front. Aging Neurosci. 2020, 12, 578719. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.-Z.; Zeng, L.-P.; Chen, J.; Wu, Y.-F.; Wang, Y.-T.; Xia, L.; Yang, Q.-G.; Wang, F.; Chen, G.-H. Long-Term Environmental Enrichment Relieves Dysfunctional Cognition and Synaptic Protein Levels Induced by Prenatal Inflammation in Older CD-1 Mice. Neural Plast 2022, 2022, 1483101. [Google Scholar] [CrossRef]
- Zhao, X.; Rondón-Ortiz, A.N.; Lima, E.P.; Puracchio, M.; Roderick, R.C.; Kentner, A.C. Therapeutic Efficacy of Environmental Enrichment on Behavioral, Endocrine, and Synaptic Alterations in an Animal Model of Maternal Immune Activation. Brain Behav. Immun. Health 2020, 3, 100043. [Google Scholar] [CrossRef]
- Zeraati, M.; Najdi, N.; Mosaferi, B.; Salari, A.-A. Environmental Enrichment Alters Neurobehavioral Development Following Maternal Immune Activation in Mice Offspring with Epilepsy. Behav. Brain Res 2021, 399, 112998. [Google Scholar] [CrossRef]
- Latendresse, G. The Interaction between Chronic Stress and Pregnancy: Preterm Birth from a Biobehavioral Perspective. J. Midwifery Womens Health 2009, 54, 8–17. [Google Scholar] [CrossRef] [Green Version]
- Grote, N.K.; Bridge, J.A.; Gavin, A.R.; Melville, J.L.; Iyengar, S.; Katon, W.J. A Meta-Analysis of Depression during Pregnancy and the Risk of Preterm Birth, Low Birth Weight, and Intrauterine Growth Restriction. Arch. Gen. Psychiatry 2010, 67, 1012–1024. [Google Scholar] [CrossRef]
- Romero, R.; Dey, S.K.; Fisher, S.J. Preterm Labor: One Syndrome, Many Causes. Science 2014, 345, 760–765. [Google Scholar] [CrossRef] [Green Version]
- Giller, A.; Andrawus, M.; Gutman, D.; Atzmon, G. Pregnancy as a Model for Aging. Ageing Res. Rev. 2020, 62, 101093. [Google Scholar] [CrossRef] [PubMed]
- Diz-Chaves, Y.; Astiz, M.; Bellini, M.J.; Garcia-Segura, L.M. Prenatal Stress Increases the Expression of Proinflammatory Cytokines and Exacerbates the Inflammatory Response to LPS in the Hippocampal Formation of Adult Male Mice. Brain Behav. Immun. 2013, 28, 196–206. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Z.-Z.; Zhuang, Z.-Q.; Sun, S.-Y.; Ge, H.-H.; Wu, Y.-F.; Cao, L.; Xia, L.; Yang, Q.-G.; Wang, F.; Chen, G.-H. Effects of Prenatal Exposure to Inflammation Coupled With Stress Exposure During Adolescence on Cognition and Synaptic Protein Levels in Aged CD-1 Mice. Front. Aging Neurosci. 2020, 12, 157. [Google Scholar] [CrossRef]
- McCreary, J.K.; Metz, G.A.S. Environmental Enrichment as an Intervention for Adverse Health Outcomes of Prenatal Stress. Environ. Epigenet. 2016, 2, dvw013. [Google Scholar] [CrossRef] [PubMed]
- Dandi, E.; Kalamari, A.; Touloumi, O.; Lagoudaki, R.; Nousiopoulou, E.; Simeonidou, C.; Spandou, E.; Tata, D.A. Beneficial Effects of Environmental Enrichment on Behavior, Stress Reactivity and Synaptophysin/BDNF Expression in Hippocampus Following Early Life Stress. Int. J. Dev. Neurosci. 2018, 67, 19–32. [Google Scholar] [CrossRef] [PubMed]
- Corredor, K.; Duran, J.M.; Herrera-Isaza, L.; Forero, S.; Quintanilla, J.P.; Gomez, A.; Martínez, G.S.; Cardenas, F.P. Behavioral Effects of Environmental Enrichment on Male and Female Wistar Rats with Early Life Stress Experiences. Front. Physiol. 2022, 13, 837661. [Google Scholar] [CrossRef]
- Li, X.-Y.; Wang, F.; Chen, G.-H.; Li, X.-W.; Yang, Q.-G.; Cao, L.; Yan, W.-W. Inflammatory Insult during Pregnancy Accelerates Age-Related Behavioral and Neurobiochemical Changes in CD-1 Mice. Age 2016, 38, 59. [Google Scholar] [CrossRef] [Green Version]
- Höltge, J.; Mc Gee, S.L.; Thoma, M.V. The Curvilinear Relationship of Early-Life Adversity and Successful Aging: The Mediating Role of Mental Health. Aging Ment. Health 2019, 23, 608–617. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Vestergaard, M.; Christensen, J.; Nahmias, A.J.; Olsen, J. Prenatal Exposure to Maternal Infections and Epilepsy in Childhood: A Population-Based Cohort Study. Pediatrics 2008, 121, e1100–e1107. [Google Scholar] [CrossRef] [PubMed]
- Jurgens, H.A.; Johnson, R.W. Environmental Enrichment Attenuates Hippocampal Neuroinflammation and Improves Cognitive Function during Influenza Infection. Brain Behav. Immun. 2012, 26, 1006–1016. [Google Scholar] [CrossRef] [Green Version]
- Fares, R.P.; Belmeguenai, A.; Sanchez, P.E.; Kouchi, H.Y.; Bodennec, J.; Morales, A.; Georges, B.; Bonnet, C.; Bouvard, S.; Sloviter, R.S.; et al. Standardized Environmental Enrichment Supports Enhanced Brain Plasticity in Healthy Rats and Prevents Cognitive Impairment in Epileptic Rats. PLoS ONE 2013, 8, e53888. [Google Scholar] [CrossRef] [Green Version]
- Morelli, E.; Ghiglieri, V.; Pendolino, V.; Bagetta, V.; Pignataro, A.; Fejtova, A.; Costa, C.; Ammassari-Teule, M.; Gundelfinger, E.D.; Picconi, B.; et al. Environmental Enrichment Restores CA1 Hippocampal LTP and Reduces Severity of Seizures in Epileptic Mice. Exp. Neurol. 2014, 261, 320–327. [Google Scholar] [CrossRef] [PubMed]
- Koe, A.S.; Ashokan, A.; Mitra, R. Short Environmental Enrichment in Adulthood Reverses Anxiety and Basolateral Amygdala Hypertrophy Induced by Maternal Separation. Transl. Psychiatry 2016, 6, e729. [Google Scholar] [CrossRef] [Green Version]
- Dinel, A.-L.; Joffre, C.; Trifilieff, P.; Aubert, A.; Foury, A.; Le Ruyet, P.; Layé, S. Inflammation Early in Life Is a Vulnerability Factor for Emotional Behavior at Adolescence and for Lipopolysaccharide-Induced Spatial Memory and Neurogenesis Alteration at Adulthood. J. Neuroinflam. 2014, 11, 155. [Google Scholar] [CrossRef] [PubMed]
- Vorhees, C.V.; Graham, D.L.; Braun, A.A.; Schaefer, T.L.; Skelton, M.R.; Richtand, N.M.; Williams, M.T. Prenatal Immune Challenge in Rats: Effects of Polyinosinic-Polycytidylic Acid on Spatial Learning, Prepulse Inhibition, Conditioned Fear, and Responses to MK-801 and Amphetamine. Neurotoxicol. Teratol. 2015, 47, 54–65. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, Z.; van Praag, H. Maternal Immune Activation Differentially Impacts Mature and Adult-Born Hippocampal Neurons in Male Mice. Brain Behav. Immun. 2015, 45, 60–70. [Google Scholar] [CrossRef]
- Khantakova, J.N.; Bondar, N.P.; Sapronova, A.A.; Reshetnikov, V.V. Delayed Effects of Neonatal Immune Activation on Brain Neurochemistry and Hypothalamic-Pituitary-Adrenal Axis Functioning. Eur. J. Neurosci. 2022, 56, 5931–5951. [Google Scholar] [CrossRef] [PubMed]
- Baldini, S.; Restani, L.; Baroncelli, L.; Coltelli, M.; Franco, R.; Cenni, M.C.; Maffei, L.; Berardi, N. Enriched Early Life Experiences Reduce Adult Anxiety-like Behavior in Rats: A Role for Insulin-like Growth Factor 1. J. Neurosci. 2013, 33, 11715–11723. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kiss, P.; Vadasz, G.; Kiss-Illes, B.; Horvath, G.; Tamas, A.; Reglodi, D.; Koppan, M. Environmental Enrichment Decreases Asphyxia-Induced Neurobehavioral Developmental Delay in Neonatal Rats. Int. J. Mol. Sci. 2013, 14, 22258–22273. [Google Scholar] [CrossRef] [PubMed]
- Schuch, C.P.; Diaz, R.; Deckmann, I.; Rojas, J.J.; Deniz, B.F.; Pereira, L.O. Early Environmental Enrichment Affects Neurobehavioral Development and Prevents Brain Damage in Rats Submitted to Neonatal Hypoxia-Ischemia. Neurosci. Lett. 2016, 617, 101–107. [Google Scholar] [CrossRef] [PubMed]
- Durán-Carabali, L.E.; Arcego, D.M.; Sanches, E.F.; Odorcyk, F.K.; Marques, M.R.; Tosta, A.; Reichert, L.; Carvalho, A.S.; Dalmaz, C.; Netto, C.A. Preventive and Therapeutic Effects of Environmental Enrichment in Wistar Rats Submitted to Neonatal Hypoxia-Ischemia. Behav. Brain Res. 2019, 359, 485–497. [Google Scholar] [CrossRef]
- MacRae, M.; Macrina, T.; Khoury, A.; Migliore, M.M.; Kentner, A.C. Tracing the Trajectory of Behavioral Impairments and Oxidative Stress in an Animal Model of Neonatal Inflammation. Neuroscience 2015, 298, 455–466. [Google Scholar] [CrossRef]
- Wu, X.-M.; Ji, M.-H.; Yin, X.-Y.; Gu, H.-W.; Zhu, T.-T.; Wang, R.-Z.; Yang, J.-J.; Shen, J.-C. Reduced Inhibition Underlies Early Life LPS Exposure Induced-Cognitive Impairment: Prevention by Environmental Enrichment. Int. Immunopharmacol. 2022, 108, 108724. [Google Scholar] [CrossRef]
- Pavlova, I.V.; Broshevitskaya, N.D.; Zaichenko, M.I.; Grigoryan, G.A. Effects of Social Isolation and an Enriched Environment on Anxious-Depressive Behavior in Rats in Normal Conditions and after Early Proinflammatory Stress. Neurosci. Behav. Phys. 2022, 52, 684–697. [Google Scholar] [CrossRef]
- Mlynarik, M.; Johansson, B.B.; Jezova, D. Enriched Environment Influences Adrenocortical Response to Immune Challenge and Glutamate Receptor Gene Expression in Rat Hippocampus. Ann. N. Y. Acad. Sci. 2004, 1018, 273–280. [Google Scholar] [CrossRef]
- Williamson, L.L.; Chao, A.; Bilbo, S.D. Environmental Enrichment Alters Glial Antigen Expression and Neuroimmune Function in the Adult Rat Hippocampus. Brain Behav. Immun. 2012, 26, 500–510. [Google Scholar] [CrossRef] [Green Version]
- Kawano, T.; Morikawa, A.; Imori, S.; Waki, S.; Tamura, T.; Yamanaka, D.; Yamazaki, F.; Yokoyama, M. Preventive Effects of Multisensory Rehabilitation on Development of Cognitive Dysfunction Following Systemic Inflammation in Aged Rats. J. Anesth. 2014, 28, 780–784. [Google Scholar] [CrossRef] [PubMed]
- Keymoradzadeh, A.; Hedayati Ch, M.; Abedinzade, M.; Gazor, R.; Rostampour, M.; Taleghani, B.K. Enriched Environment Effect on Lipopolysaccharide-Induced Spatial Learning, Memory Impairment and Hippocampal Inflammatory Cytokine Levels in Male Rats. Behav. Brain Res. 2020, 394, 112814. [Google Scholar] [CrossRef] [PubMed]
- Keymoradzadeh, A.; Hedayati Ch, M.; Abedinzade, M.; Khakpour-Taleghani, B. Enriched Environment Restores Passive Avoidance Memory Impairment in a Rat Model of Neuroinflammation. Physiol. Pharmacol. 2022, 26, 127–137. [Google Scholar] [CrossRef]
- Ji, M.-H.; Tang, H.; Luo, D.; Qiu, L.-L.; Jia, M.; Yuan, H.-M.; Feng, S.-W.; Yang, J.-J. Environmental Conditions Differentially Affect Neurobehavioral Outcomes in a Mouse Model of Sepsis-Associated Encephalopathy. Oncotarget 2017, 8, 82376–82389. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Aranda, M.L.; González Fleitas, M.F.; Dieguez, H.H.; Milne, G.A.; Devouassoux, J.D.; Keller Sarmiento, M.I.; Chianelli, M.; Sande, P.H.; Dorfman, D.; Rosenstein, R.E. Therapeutic Benefit of Environmental Enrichment on Optic Neuritis. Neuropharmacology 2019, 145, 87–98. [Google Scholar] [CrossRef]
- Iwashyna, T.J.; Ely, E.W.; Smith, D.M.; Langa, K.M. Long-Term Cognitive Impairment and Functional Disability among Survivors of Severe Sepsis. JAMA 2010, 304, 1787–1794. [Google Scholar] [CrossRef] [Green Version]
- Sanders, R.D.; Pandharipande, P.P.; Davidson, A.J.; Ma, D.; Maze, M. Anticipating and Managing Postoperative Delirium and Cognitive Decline in Adults. BMJ 2011, 343, d4331. [Google Scholar] [CrossRef]
- Murray, C.; Sanderson, D.J.; Barkus, C.; Deacon, R.M.J.; Rawlins, J.N.P.; Bannerman, D.M.; Cunningham, C. Systemic Inflammation Induces Acute Working Memory Deficits in the Primed Brain: Relevance for Delirium. Neurobiol. Aging 2012, 33, 603–616.e3. [Google Scholar] [CrossRef] [Green Version]
- Witlox, J.; Eurelings, L.S.M.; de Jonghe, J.F.M.; Kalisvaart, K.J.; Eikelenboom, P.; van Gool, W.A. Delirium in Elderly Patients and the Risk of Postdischarge Mortality, Institutionalization, and Dementia: A Meta-Analysis. JAMA 2010, 304, 443–451. [Google Scholar] [CrossRef]
- Steinmetz, J.; Christensen, K.B.; Lund, T.; Lohse, N.; Rasmussen, L.S. ISPOCD Group Long-Term Consequences of Postoperative Cognitive Dysfunction. Anesthesiology 2009, 110, 548–555. [Google Scholar] [CrossRef] [Green Version]
- Perry, V.H.; Cunningham, C.; Holmes, C. Systemic Infections and Inflammation Affect Chronic Neurodegeneration. Nat. Rev. Immunol. 2007, 7, 161–167. [Google Scholar] [CrossRef] [PubMed]
- Terrando, N.; Monaco, C.; Ma, D.; Foxwell, B.M.J.; Feldmann, M.; Maze, M. Tumor Necrosis Factor-Alpha Triggers a Cytokine Cascade Yielding Postoperative Cognitive Decline. Proc. Natl. Acad. Sci. USA 2010, 107, 20518–20522. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhao, J.; Bi, W.; Xiao, S.; Lan, X.; Cheng, X.; Zhang, J.; Lu, D.; Wei, W.; Wang, Y.; Li, H.; et al. Neuroinflammation Induced by Lipopolysaccharide Causes Cognitive Impairment in Mice. Sci. Rep. 2019, 9, 5790. [Google Scholar] [CrossRef] [Green Version]
- Manickavasagam, D.; Lin, L.; Oyewumi, M.O. Nose-to-Brain Co-Delivery of Repurposed Simvastatin and BDNF Synergistically Attenuates LPS-Induced Neuroinflammation. Nanomedicine 2020, 23, 102107. [Google Scholar] [CrossRef] [PubMed]
- Glass, C.K.; Saijo, K.; Winner, B.; Marchetto, M.C.; Gage, F.H. Mechanisms Underlying Inflammation in Neurodegeneration. Cell 2010, 140, 918–934. [Google Scholar] [CrossRef] [Green Version]
- Wang, W.-Y.; Tan, M.-S.; Yu, J.-T.; Tan, L. Role of Pro-Inflammatory Cytokines Released from Microglia in Alzheimer’s Disease. Ann. Transl. Med. 2015, 3, 136. [Google Scholar] [CrossRef]
- Phillips, C.; Baktir, M.A.; Srivatsan, M.; Salehi, A. Neuroprotective Effects of Physical Activity on the Brain: A Closer Look at Trophic Factor Signaling. Front. Cell Neurosci. 2014, 8, 170. [Google Scholar] [CrossRef] [Green Version]
- Boehme, F.; Gil-Mohapel, J.; Cox, A.; Patten, A.; Giles, E.; Brocardo, P.S.; Christie, B.R. Voluntary Exercise Induces Adult Hippocampal Neurogenesis and BDNF Expression in a Rodent Model of Fetal Alcohol Spectrum Disorders. Eur. J. Neurosci. 2011, 33, 1799–1811. [Google Scholar] [CrossRef]
- Giridharan, V.V.; Masud, F.; Petronilho, F.; Dal-Pizzol, F.; Barichello, T. Infection-Induced Systemic Inflammation Is a Potential Driver of Alzheimer’s Disease Progression. Front. Aging Neurosci. 2019, 11, 122. [Google Scholar] [CrossRef]
- Girard, T.D.; Jackson, J.C.; Pandharipande, P.P.; Pun, B.T.; Thompson, J.L.; Shintani, A.K.; Gordon, S.M.; Canonico, A.E.; Dittus, R.S.; Bernard, G.R.; et al. Delirium as a Predictor of Long-Term Cognitive Impairment in Survivors of Critical Illness. Crit Care Med. 2010, 38, 1513–1520. [Google Scholar] [CrossRef]
- Pandharipande, P.P.; Girard, T.D.; Jackson, J.C.; Morandi, A.; Thompson, J.L.; Pun, B.T.; Brummel, N.E.; Hughes, C.G.; Vasilevskis, E.E.; Shintani, A.K.; et al. Long-Term Cognitive Impairment after Critical Illness. N. Engl. J. Med. 2013, 369, 1306–1316. [Google Scholar] [CrossRef] [Green Version]
- Aranda, M.L.; Dorfman, D.; Sande, P.H.; Rosenstein, R.E. Experimental Optic Neuritis Induced by the Microinjection of Lipopolysaccharide into the Optic Nerve. Exp. Neurol. 2015, 266, 30–41. [Google Scholar] [CrossRef]
- Chao, C.C.; Hu, S.; Close, K.; Choi, C.S.; Molitor, T.W.; Novick, W.J.; Peterson, P.K. Cytokine Release from Microglia: Differential Inhibition by Pentoxifylline and Dexamethasone|The Journal of Infectious Diseases|Oxford Academic. Available online: https://academic.oup.com/jid/article-abstract/166/4/847/837965 (accessed on 18 January 2023).
- Font-Nieves, M.; Sans-Fons, M.G.; Gorina, R.; Bonfill-Teixidor, E.; Salas-Pérdomo, A.; Márquez-Kisinousky, L.; Santalucia, T.; Planas, A.M. Induction of COX-2 Enzyme and down-Regulation of COX-1 Expression by Lipopolysaccharide (LPS) Control Prostaglandin E2 Production in Astrocytes. J. Biol. Chem. 2012, 287, 6454–6468. [Google Scholar] [CrossRef] [Green Version]
- Badshah, H.; Ali, T.; Rehman, S.U.; Amin, F.U.; Ullah, F.; Kim, T.H.; Kim, M.O. Protective Effect of Lupeol Against Lipopolysaccharide-Induced Neuroinflammation via the P 38/c-Jun N-Terminal Kinase Pathway in the Adult Mouse Brain. J. Neuroimmune Pharmacol. 2016, 11, 48–60. [Google Scholar] [CrossRef] [PubMed]
- Sheppard, O.; Coleman, M.P.; Durrant, C.S. Lipopolysaccharide-Induced Neuroinflammation Induces Presynaptic Disruption through a Direct Action on Brain Tissue Involving Microglia-Derived Interleukin 1 Beta. J. Neuroinflam. 2019, 16, 106. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, J.W.; Lee, Y.K.; Yuk, D.Y.; Choi, D.Y.; Ban, S.B.; Oh, K.W.; Hong, J.T. Neuro-Inflammation Induced by Lipopolysaccharide Causes Cognitive Impairment through Enhancement of Beta-Amyloid Generation. J. Neuroinflam. 2008, 5, 37. [Google Scholar] [CrossRef] [Green Version]
- Zhan, X.; Stamova, B.; Sharp, F.R. Lipopolysaccharide Associates with Amyloid Plaques, Neurons and Oligodendrocytes in Alzheimer’s Disease Brain: A Review. Front. Aging Neurosci. 2018, 10, 42. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Arango, C.; Díaz-Caneja, C.M.; McGorry, P.D.; Rapoport, J.; Sommer, I.E.; Vorstman, J.A.; McDaid, D.; Marín, O.; Serrano-Drozdowskyj, E.; Freedman, R.; et al. Preventive Strategies for Mental Health. Lancet Psychiatry 2018, 5, 591–604. [Google Scholar] [CrossRef]
- Sampedro-Piquero, P.; Begega, A. Environmental Enrichment as a Positive Behavioral Intervention Across the Lifespan. Curr. Neuropharmacol. 2017, 15, 459–470. [Google Scholar] [CrossRef] [Green Version]
- Mandolesi, L.; Gelfo, F.; Serra, L.; Montuori, S.; Polverino, A.; Curcio, G.; Sorrentino, G. Environmental Factors Promoting Neural Plasticity: Insights from Animal and Human Studies. Neural Plast. 2017, 2017, 7219461. [Google Scholar] [CrossRef] [Green Version]
- Vakharia, K.; Hinson, J.P. Lipopolysaccharide Directly Stimulates Cortisol Secretion by Human Adrenal Cells by a Cyclooxygenase-Dependent Mechanism. Endocrinology 2005, 146, 1398–1402. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Maikoo, S.; Wilkins, A.; Qulu, L. The Effect of Oxytocin and an Enriched Environment on Anxiety-like Behaviour and Corticosterone Levels in a Prenatally Stressed Febrile Seizure Rat Model. IBRO Neurosci. Rep. 2022, 13, 47–56. [Google Scholar] [CrossRef] [PubMed]
- Freret, T.; Billard, J.-M.; Schumann-Bard, P.; Dutar, P.; Dauphin, F.; Boulouard, M.; Bouet, V. Rescue of Cognitive Aging by Long-Lasting Environmental Enrichment Exposure Initiated before Median Lifespan. Neurobiol. Aging 2012, 33, 1005.e1–1005.e10. [Google Scholar] [CrossRef] [PubMed]
- Leger, M.; Quiedeville, A.; Paizanis, E.; Natkunarajah, S.; Freret, T.; Boulouard, M.; Schumann-Bard, P. Environmental Enrichment Enhances Episodic-Like Memory in Association with a Modified Neuronal Activation Profile in Adult Mice. PLoS ONE 2012, 7, e48043. [Google Scholar] [CrossRef]
- Clemenson, G.; Gage, F.; Stark, C. Environmental Enrichment and Neuronal Plasticity. In The Oxford Handbook of Developmental Neural Plasticity; Oxford University Press: Oxford, UK, 2018. [Google Scholar]
- Bolós, M.; Terreros-Roncal, J.; Perea, J.R.; Pallas-Bazarra, N.; Ávila, J.; Llorens-Martín, M. Maturation Dynamics of the Axon Initial Segment (AIS) of Newborn Dentate Granule Cells in Young Adult C57BL/6J Mice. J. Neurosci. 2019, 39, 1605–1620. [Google Scholar] [CrossRef]


| References | Species | Environmental Enrichment | LPS Injection(s) | ||
|---|---|---|---|---|---|
| (Age and Timing of Evaluations) | Type of Enrichment | Subjects and Duration | (Time and Dose) | ||
| EE on dams | Schander et al., 2020 [84] | BALB/c mice Dams: 6 weeks (Effects on: mothers (pregnancy)) | Large cage (640 × 420 × 200 mm) with running wheels and objects (toys, tunnels, shelters, and stairs) of different shapes, textures, and colors changed once a week. Every 3–4 days, one object removed, moved, or added. Ten animals per cage. | On dams: EE for 6 weeks; mating (regular cages); EE until G15. | On dams: G15, two i.p. injections of LPS (1°—0.13 mg/kg; 2°—0.39 mg/kg). (LPS after EE protocol) |
| Schander et al., 2021 [85] | BALB/c mice Dams: 6 weeks (Effects on: mothers (pregnancy); offspring (P 1–P 22) | Large cage (640 × 420 × 200 mm) with running wheels and objects (toys, tunnels, shelters and stairs) of different shapes, textures, and colors changed once a week. Every 3–4 days, one object removed, moved or added. Ten animals per cage. | On dams: EE for 6 weeks; mating (regular cages); EE until G15. | On dams: G15, two i.p. injections of LPS (1°—0.13 mg/kg; 2°—0.39 mg/kg). (LPS after EE protocol) | |
| Zhuang et al., 2021 [86] | CD-1 mice Dams: 7/8 weeks (Effects on: mothers (after pregnancy)) | Large cage (52 × 40 × 20 cm3) with toys (running wheels, tunnels, poplar wood block toys, and rings). Ten to fifteen animals per cage. | On dams: EE from weaning of pups until about 6 or 18 months of age of the mothers. | On dams: G15–17, daily i.p. injection of LPS (50 μg/kg). (LPS before EE protocol) | |
| EE on offspring | Connors et al., 2014 [87] | Sprague–Dawley rats Dams: age not specified (Effects on: offspring (P 36–P 40)) | Multi-level cage with toys changed two times weekly, tubes, chew bone, Nestlets©, and ramps. Pair-housed animals. | On dams: EE for about 2 months. On pups: EE from P 1 to P 40. | On dams: G11, i.p. injection of LPS (100 μg/kg). (LPS during EE protocol) |
| Rajesh et al., 2016 [88] | Wistar rats Dams: 3 months (Effects on: male offspring (P 61–P 66)) | EE: Large cage (120 × 100 × 100 cm) with plastic tunnels, raised platform with ladder, various size metal balls, toys, and steel swing. Objects were changed on alternate days. Treadmill running exercise: 1.5 m/min–10.9 m/min; 15 min/day. Number of animals per cage not specified. | On pups: EE (4 h/day) or/and treadmill from P 15 to P 60. | On dams: From G14 till parturition on alternate days i.p. injection of LPS (0.5 mg/Kg). | |
| Rajesh et al., 2018 [89] | Wistar rats Dams: 3 months (Effects on: male offspring (P 67)) | EE: Large cage (120 × 100 × 100 cm) with plastic tunnels, raised platform with ladder, various size metal balls, toys, and steel swing. Objects were changed on alternate days. Treadmill running exercise: 1.5 m/min -10.9 m/min; 15 min/day. Number of animals per cage not specified. | On pups: EE (4 h/day) or/and treadmill from P 15 to P 60. | On dams: From G14 till parturition on alternate days i.p. injection of LPS (0.5 mg/Kg). | |
| Bakos et al., 2004 [90] | Wistar rats Dams: 8th day-pregnant—age not specified (Effects on: male offspring (~P 83)) | Large cage (1 m × 0.5 m × 0.5 m) with bedding changed once a week, running wheels, wooden swings, plastic tubes, and beams changed three times a week. Occasionally added pieces of food (nuts, apples). Six animals per cage. | On pups: EE after weaning, for 2 months. | On dams: G15–19, daily subcutaneous injection of LPS in increasing doses of 20, 20, 40, 40, 80 μg/kg/mL. | |
| Kentner et al., 2016 [91] | Sprague-Dawley rats Dams: 12th day- pregnant—age not specified (Effects on: male offspring (~P 92)). | Communal nesting: larger-style one-level cage with tube, chew bone, and Nestlets©. Four animals per cage. EE: multi-level cage with chew bone, Nestlets©, ramps, novel toys, and tubes. Toys and tubes changed twice a week. Four animals per cage. | On pups: EE from P 50, for 6 weeks. | On dams: G15 i.p. injection of LPS (100 μg/kg). | |
| Wu et al., 2020 [92] | CD-1 mice Dams: 8 weeks (Effects on: male offspring (3 months old; 15 months old)) | Large cage with pipes, plastic running wheels, balls, and toys changed every week. Number of animals per cage not specified. | On pups: EE from 2 months of age until 3 or 15 months of age. | On dams: G15–17, daily i.p. injection of LPS (50 μg/kg). | |
| Zhang et al., 2022 [93] | CD-1 mice Dams: 6/8 weeks (Effects on: male offspring (3 months old; 15 months old)) | Large cage (36 × 23 × 18 cm) with climbing ladders, running wheel, ball, plastic and wooden objects suspended from the cage top, paper, cardboard boxes, nesting material, and toys (changed every 1–2 days). Eight animals per cage. | On pups: EE after weaning until 3 or 15 months of age. | On dams: G15–17, daily i.p. injection of LPS (50 μg/kg). | |
| Zhao et al., 2020 [94] | Sprague-Dawley rats Dams: 12th day- pregnant—age not specified (Effects on: female offspring (~P 92)) | Communal nesting: larger-style one-level cage with tube, chew bone, and Nestlets©. Four animals per cage. EE: multi-level cage with chew bone, Nestlets©, ramps, novel toys, and tubes. Toys and tubes changed twice a week. Four animals per cage. | On pups: EE from P 50, for 6 weeks. | On dams: G15 i.p. injection of LPS (100 μg/kg). | |
| Zeraati et al., 2021 [95] | NMRI mice Dams: 70–80 days (Effects on: offspring—(P 121–P 142)) | Cage (58 × 38 × 20 cm) with one running wheel, two plastic shelters, four tunnels, and two cotton nestlets replaced once a week. Four animals per cage. | On pups: EE from P 21 to P 120. | On dams: G17, i.p. injection of LPS (300 μg/kg/100 μL). | |
| References | Main Effects of Environmental Enrichment | ||
|---|---|---|---|
| On Mothers | On Offspring | ||
| Schander et al., 2020 [84] | EE reduced - body weight gain in nonpregnant mice; - cholesterol and triglycerides serum levels in pregnant mice;- LPS-induced preterm birth rate and offspring perinatal death (by 40%); - expression of TLR4 and CD14 in the uterus of LPS-challenged mice. EE prevented - LPS-induced increase in corticosterone serum levels; - LPS-induced neutrophil infiltration into the cervix as well as metalloprotease activity in this tissue. EE induced molecular changes in uterus and cervix of LPS-induced preterm birth mice. | ||
| Schander et al., 2021 [85] | EE modulated - white blood cell count and its response to systemic LPS administration; - amniotic fluid response to LPS administration, promoting a tolerogenic microenvironment. | EE prevented the negative influence of intrauterine exposure to an inflammatory environment on physical landmarks (pinnae detachment, lower incisors eruption, eye opening) of the offspring’s development during lactation. | |
| Zhuang et al., 2021 [86] | Long-term exposure to EE - reduced spatial learning and memory impairment (MWM) in aged dams resulting from LPS-induced gestational inflammation; - improved dynamics in hippocampi of LPS-injected dams; - alleviated the accelerated changes in mitochondrial biogenesis and mitophagy resulting from gestational inflammation in aged mice. | ||
| EE on offspring / EE on dams | Connors et al., 2014 [87] | EE prevented - elevations in hippocampal corticosterone level; - decreased glucocorticoid receptor expression in hippocampus; - reductions in social contact (SI) in juvenile male rats treated with LPS in utero. | |
| Rajesh et al., 2016 [88] | Rats of LPS + EE + exercise group showed a significant enhancement in learning and memory performance (MWM), compared with other groups. | ||
| Rajesh et al., 2018 [89] | Rats of LPS + EE + exercise group showed a significant enhancement in dendritic arborization of CA3 hippocampal neurons, compared with other groups. | ||
| Bakos et al., 2004 [90] | EE reversed LPS-induced nucleus accumbens dopamine level decrement. | ||
| Kentner et al., 2016 [91] | EE prevented the LPS-induced reduction of the expression of the EAAT-2 gene in the PFC; EE mitigated LPS-induced downregulation of BDNF in the hippocampus and neurotrophic tyrosine kinase receptor type 2 genes in PFC; colony nesting mitigated LPS-induced spatial and object memory impairment (object-in-place test). | ||
| Wu et al., 2020 [92] | EE alleviated LPS-induced memory impairments (MWM) in middle-aged mice. | ||
| Zhang et al., 2022 [93] | EE improved learning and memory performance (MWM) deteriorated by LPS injection; EE reduced protein and mRNA levels of Arc and Syt1 genes in the hippocampus increased by LPS injection. | ||
| Zhao et al., 2020 [94] | EE attenuated - recognition memory (object-in-place test) deficits observed in MIA offspring; - social (SI) deficits observed in MIA offspring; - the elevation of plasma corticosterone in MIA offspring. | ||
| Zeraati et al., 2021 [95] | EE improved - anxiety-like behavior in LPS offspring treated with water or PTZ compared to control groups (open field; light–dark box); - spatial working memory in LPS offspring treated with PTZ compared to control groups (Y-maze); - recognition ratio in LPS offspring treated with PTZ compared to control groups (novel object recognition test). EE decreased - Seizure scores; - TNF-α and IL-10 levels in the hippocampus of LPS offspring treated with PTZ compared to normal-PTZ LPS offspring. | ||
| References | Species | Environmental Enrichment | LPS Injection(s) | |
|---|---|---|---|---|
| (Age and Timing of Evaluations) | Type of Enrichment | Subjects and Duration | (Time and Dose) | |
| MacRae et al., 2015 [120] | Sprague–Dawley rats Dams: age not specified Pups: newborn (Evaluation of effects in male and female offspring (P 40–P 90)) | Large multi-level cage with toys and tubes changed twice weekly, chew bone, Nestlets©, and ramps. Four to six animals per cage. | On dams: EE was conducted from breeding to weaning of pups. On pups: EE was conducted from birth to the end of the experiment (P 1–P 90). | On pups: P 3 and P 5, 2 i.p. injections of LPS (50 μg/kg). (LPS administered when EE protocol is already ongoing) |
| Wu et al., 2022 [121] | C57BL/6 mice. Pups: 10 days (Evaluation of effects in male offspring P 39–P 42) | Cage (60 × 32 × 38 cm) with running wheel, swing, platforms and toys. Objects were changed twice a week. Number of animals per cage not specified. | On pups: EE was conducted four hours per day during P 10–P 38. | On pups: P 10, i.p. injection of LPS (100 μg/kg). (LPS before EE protocol) |
| Pavlova et al., 2022 [122] | Wistar rats Pups: newborn (Evaluation of effects in male and female offspring P 90–P 105) | Plastic box (51 × 40.5 × 30 cm) containing stairs, a running wheel, tubes, and materials for burying (sawdust, foam filler, paper, etc.). Four to six animals per cage. | On pups: EE was conducted for 20 min every other day, from the age of 45 days until the end of experiment (~3.5 months). | On pups: P 3 and P 5, 2 i.p. injection of LPS (dose of 50 μg/kg in a volume of 10 μL/g). (LPS before EE protocol) |
| References | Main Effects of Environmental Enrichment | |
|---|---|---|
| On Mothers | On Offspring | |
| MacRae et al., 2015 [120] | EE dams started retrieving pups earlier and reunited the entire litter more quickly than standard-housed rats. | EE partially protected against the consequences of inflammation on juvenile social (social interaction test) behavior. EE prevented LPS-induced glutathione level reduction in the juvenile PFC. |
| Wu et al., 2022 [121] | EE improved LPS-induced working memory impairment (Y-maze). EE attenuated - LPS-induced microglial activation in the hippocampus and mPFC; - LPS-induced loss of dendritic spines in the hippocampus. EE reversed LPS-induced decrease in parvalbumin expression in the hippocampus and mPFC. | |
| Pavlova et al., 2022 [122] | EE increased anxiety (open field) and depression-like behavior (sucrose preference test) in female LPS group. | |
| References | Species | Environmental Enrichment | LPS Injection(s) |
|---|---|---|---|
| (Age) | (Type and Duration) | (Time and Dose) | |
| Mlynarik et al., 2004 [123] | Male Wistar rats 2 months | Plexiglas box (1 × 0.5 × 0.5 m) with sawdust straw bedding, several platforms, swings, and a variety of miscellaneous objects (plastic boxes, tunnels, cartons, iron ladders, glass bottles, laboratory beakers, natural branches, water pools, spin wheels). The floor configuration was changed every second working day and aforementioned items were randomly put in or taken out at the same time. A small amount of extra food (e.g., pieces of apples or oranges, peanuts, dried bread, and curd) were occasionally served or hidden in the straw bedding. Ten animals per cage. EE was conducted for ~5 weeks. | Daily i.p. injection of LPS in increasing doses of 10, 10, 20, 20, and 40 μg/kg/mL for 5 consecutive days. (LPS after EE protocol) |
| Williamson et al., 2012 [124] | Male Sprague-Dawley rats 60 days | Boxes (55.9 × 35.6 × 30.5 cm) contained quarter-inch corn-cob bedding, a running wheel, a PVC tube, and various small objects and toys. Pair-housed animals. EE was conducted for 7 weeks for 12 h per day. | i.p. injection of LPS (100 μg/kg). (LPS after EE protocol) |
| Kawano et al., 2014 [125] | Male Wistar rats 24–25 months | Large cage equipped with plastic toys, a tunnel, a ladder, a platform, nesting material, and a running wheel. Items were routinely rearranged during the experimental period. The multi-modal sensory stimulations were conducted by a buzzer sound (167 Hz), a blinking light-emitting diode (LED) light (3 Hz), and vibration (60 dB) for 1 min using a multi-digital tuner three times daily during the active phase. Pair-housed animals. EE was conducted for 7 days. | i.p. injection of LPS (5 mg/kg). (LPS before EE protocol) |
| Keymoradzadeh et al., 2020 [126] | Male Wistar rats 7 weeks | Large cage (96 × 49 × 38 cm) containing running wheels, a tunnel, a small compartment, stairs, and many other colorful objects (e.g., colorful plastic plates, wooden disks of varied colors and sizes, plastic cups, and hanging cubes). Objects were changed every day. Number of animals per cage not specified. EE was conducted for 21 days (3 weeks). | i.p. injection of LPS (1 mg/kg) days 1, 3, 5, and 7. (LPS before EE protocol) |
| Keymoradzadehet al., 2022 [127] | Male Wistar rats 7 weeks | Large cage (96 × 49 × 38 cm) containing running wheels, a tunnel, a small compartment, stairs, and many other colorful objects (e.g., colorful plastic plates, wooden disks of varied colors and sizes, plastic cups, and hanging cubes). Objects were changed every day. Number of animals per cage not specified. EE was conducted for 21 days (3 weeks). | i.p. injection of LPS (1 mg/kg) days 1, 3, 5, and 7. (LPS before EE protocol) |
| Ji et al., 2017 [128] | Male C57BL/6 mice 3-4 months | Large cage (60 cm × 35 cm × 20 cm) containing a small house, a running wheel for voluntary exercise, and four to five toys that were exchanged three times a week for new toys of different shape and colors. Four to six animals per cage. EE was conducted for 4 weeks. | i.p. injection of LPS (5 mg/kg). (LPS before EE protocol) |
| Aranda et al., 2019 [129] | Male Wistar rats 2 months | Big cages (46.5 × 78 × 95 cm) containing 4 floors and several food hoppers, water bottles, running wheels, tubes, ramps, and differently shaped objects (balls, ropes, stones) repositioned once a day and fully substituted once a week. Six animals per cage. Animals were continuously exposed to standard enrichment or EE during different intervals (21, 17, 14, or 3 days). | An amount of 1 μL of 4.5 μg/μL LPS in pyrogen-free saline locally injected in the optic nerve. (LPS before EE protocol) |
| References | Main Effects of Environmental Enrichment |
|---|---|
| Mlynarik et al., 2004 [123] | EE prevented - LPS-induced transient decrease in body weight; - LPS-induced increase in plasma and adrenal corticosterone levels. |
| Williamson et al., 2012 [124] | EE increased - density of glial markers within the DG altered by LPS injection; - BDNF mRNA in the hippocampus decreased by LPS injection. EE attenuated hippocampal response to LPS for a subset of cytokines and chemokines. |
| Kawano et al., 2014 [125] | Multisensory early rehabilitation environment reverted - LPS-induced memory impairment (novel object recognition test); - LPS-induced elevation of TNF-a and IL-1b levels in hippocampus. |
| Keymoradzadeh et al., 2020 [126] | EE ameliorated LPS-induced spatial learning and memory impairment (Morris water maze). EE counteracted LPS-induced increase of hippocampal IL-1β levels. EE further augmented LPS-induced increase of the hippocampal IL-10 levels. |
| Keymoradzadeh et al., 2022 [127] | EE counteracted LPS-induced memory impairment (passive avoidance test). EE counteracted LPS-induced increase of IL-6 levels and decrease of BDNF levels in hippocampus. |
| Ji et al., 2017 [128] | EE reverted - LPS-induced hippocampal and non-hippocampal-dependent cognitive impairment (fear conditioning test); - LPS-induced increased hippocampal IL-6 expression. |
| Aranda et al., 2019 [129] | EE rescued - LPS-induced decrement in pupil light reflex, visual evoked potentials, retinal anterograde transport, phosphorylated neurofilament immunoreactivity, myelination, axon and retinal ganglion cell number; - LPS-induced optic nerve oxidative damage and LPS-increased optic nerve inducible nitric oxide synthase, cyclooxygenase-2, and IL-1b and TNF-α mRNA. EE counteracted the microglial/macrophage reactivity and astrocytosis observed in LPS group. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Landolfo, E.; Cutuli, D.; Decandia, D.; Balsamo, F.; Petrosini, L.; Gelfo, F. Environmental Enrichment Protects against Neurotoxic Effects of Lipopolysaccharide: A Comprehensive Overview. Int. J. Mol. Sci. 2023, 24, 5404. https://doi.org/10.3390/ijms24065404
Landolfo E, Cutuli D, Decandia D, Balsamo F, Petrosini L, Gelfo F. Environmental Enrichment Protects against Neurotoxic Effects of Lipopolysaccharide: A Comprehensive Overview. International Journal of Molecular Sciences. 2023; 24(6):5404. https://doi.org/10.3390/ijms24065404
Chicago/Turabian StyleLandolfo, Eugenia, Debora Cutuli, Davide Decandia, Francesca Balsamo, Laura Petrosini, and Francesca Gelfo. 2023. "Environmental Enrichment Protects against Neurotoxic Effects of Lipopolysaccharide: A Comprehensive Overview" International Journal of Molecular Sciences 24, no. 6: 5404. https://doi.org/10.3390/ijms24065404





