Inhibitory Effects of 3-Methylcholanthrene Exposure on Porcine Oocyte Maturation
Abstract
1. Introduction
2. Results
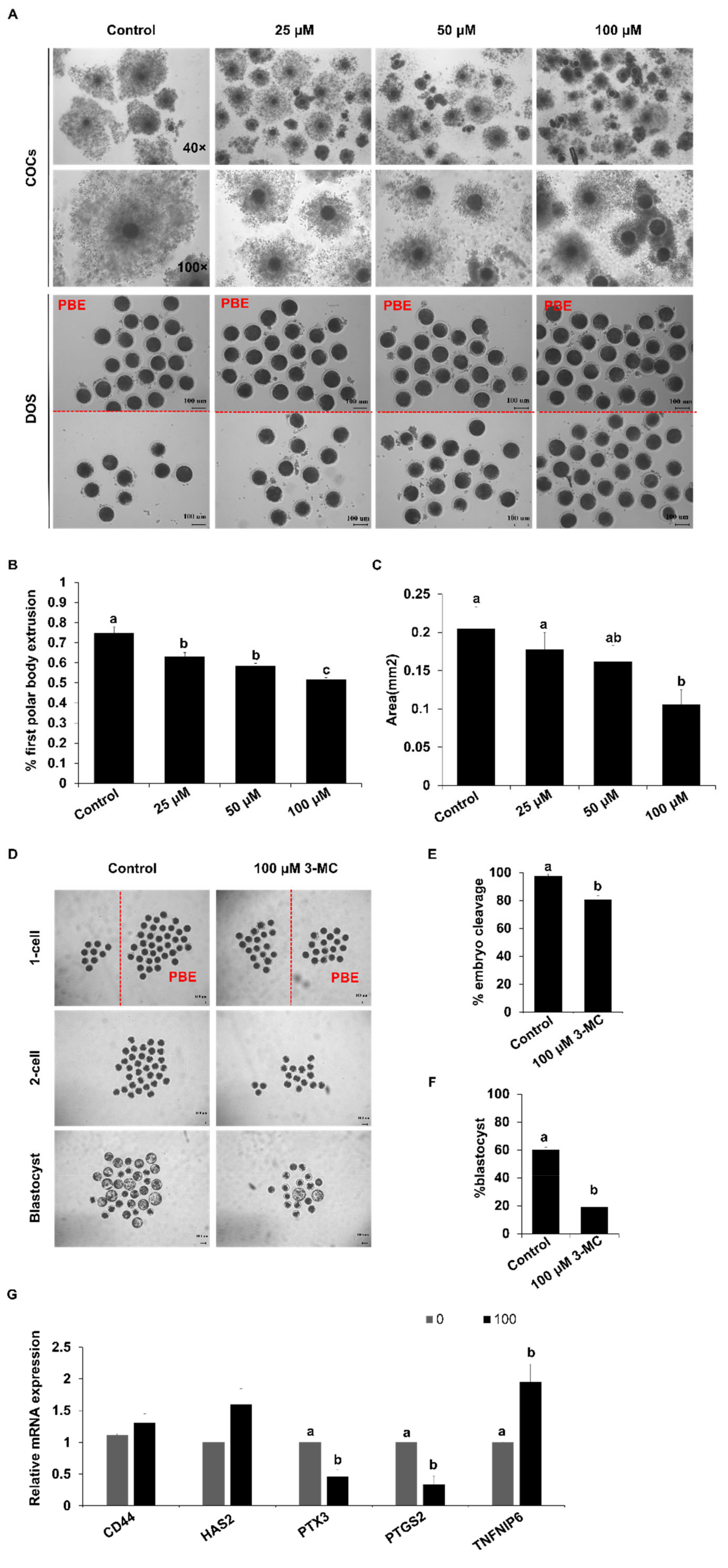
2.1. 3-MC Exposure Inhibited Porcine Oocyte Meiotic Maturation and Early Embryonic Development
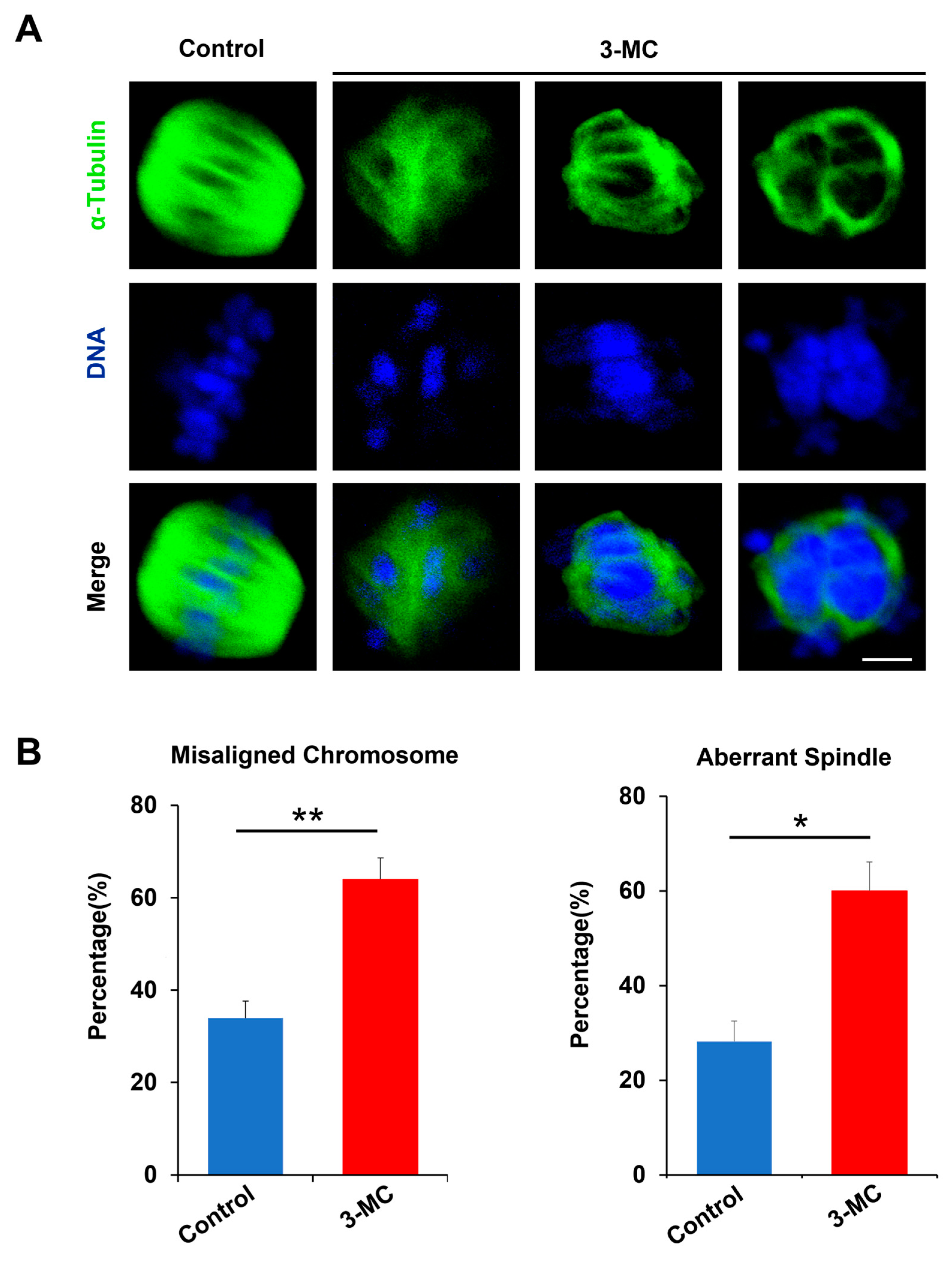
2.2. 3-MC Exposure Disrupted the Spindle Assembly and Chromosome Alignment in Oocytes
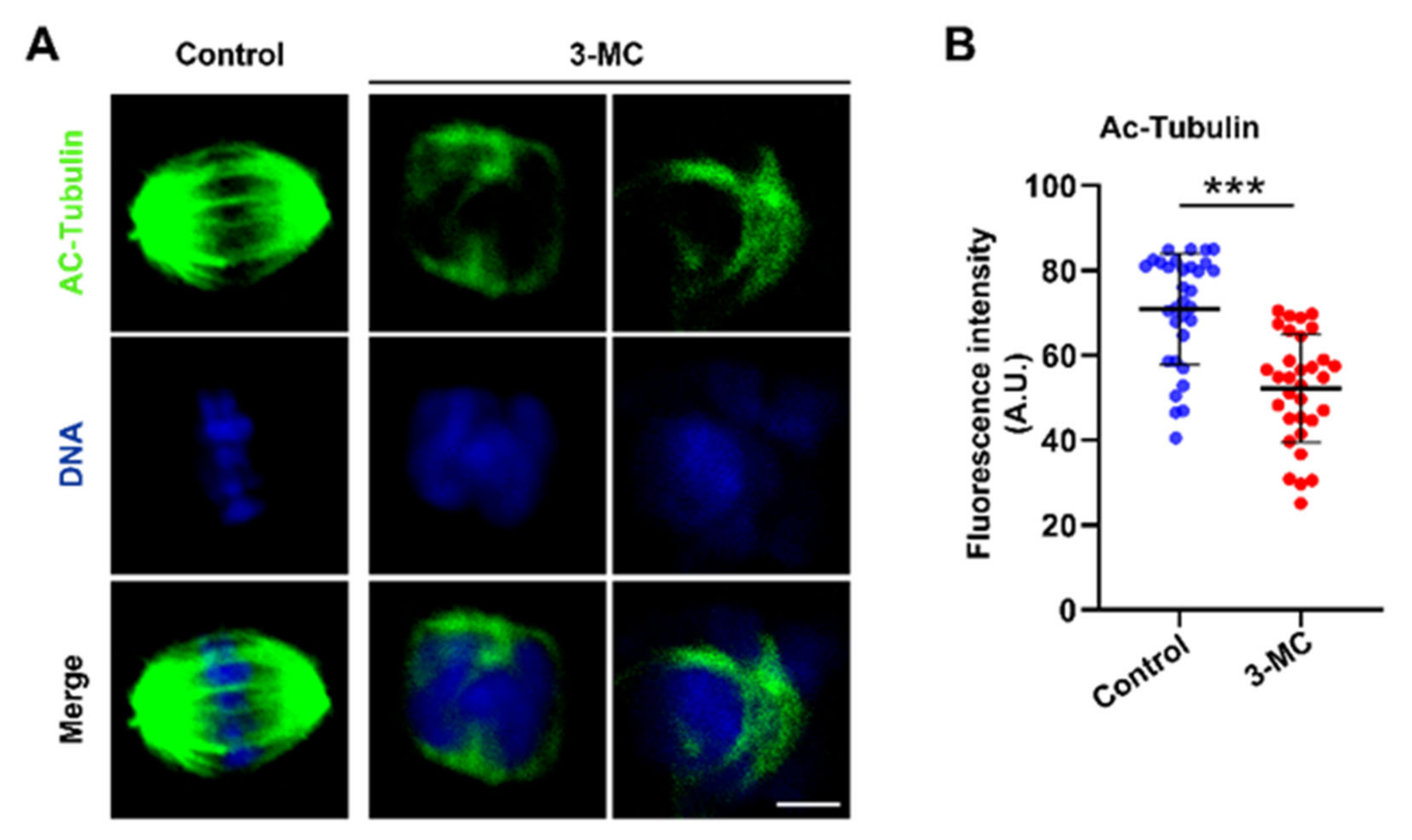
2.3. 3-MC Exposure Inhibited the Acetylation of α-Tubulin in Oocytes
2.4. 3-MC Exposure Disrupted the Polymerization of Actin Filaments in Oocytes
2.5. 3-MC Exposure Disrupted the Distribution of Mitochondria and Cortical Granules in Oocytes
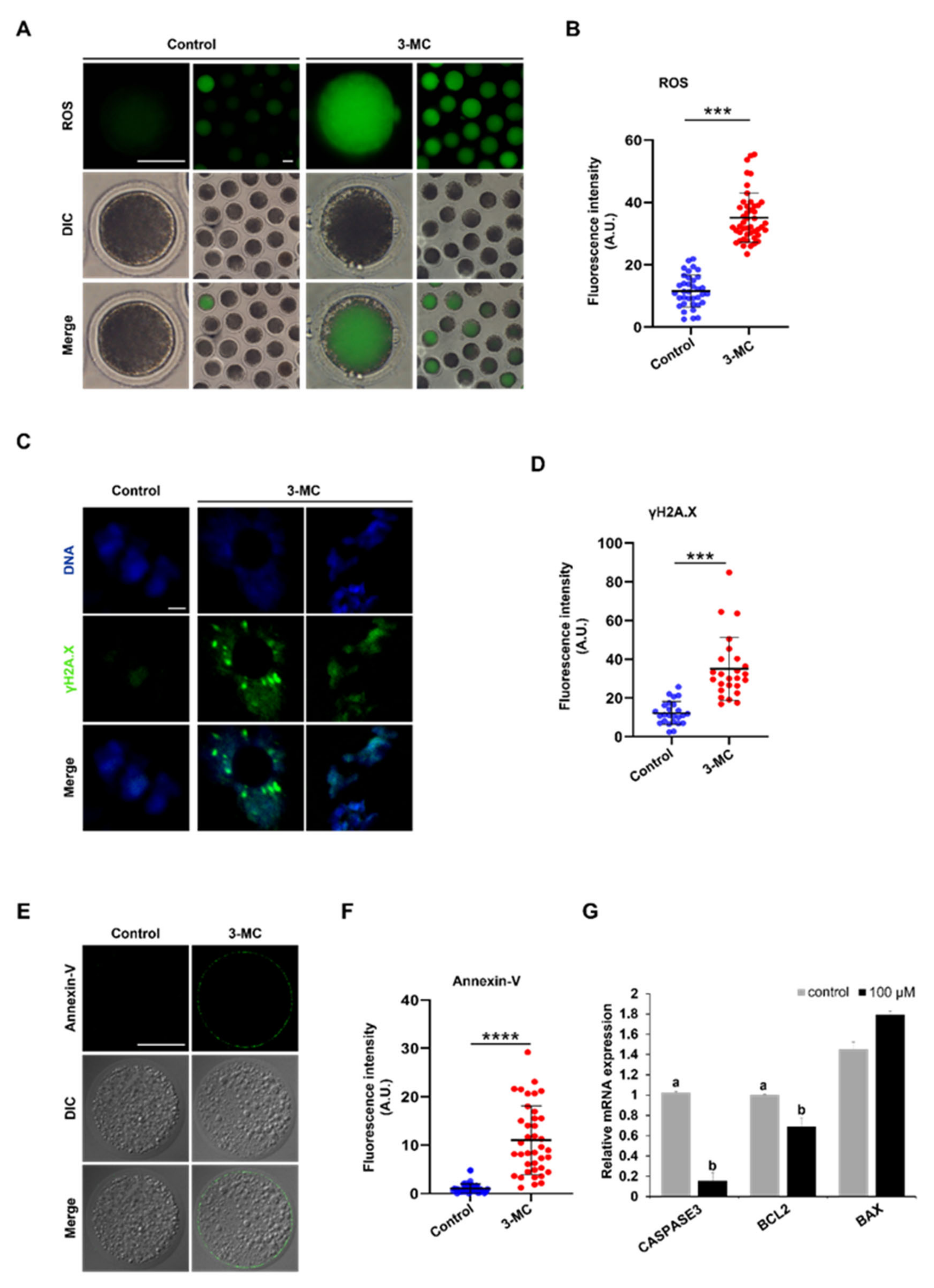
2.6. 3-MC Exposure Increased the Levels of DNA Damage and Apoptosis in Oocytes
3. Discussion
4. Materials and Methods
4.1. Experimental Design
4.2. Preparation of 3-MC
4.3. Oocyte In Vitro Maturation
4.4. Parthenogenetic Activation and Embryo Culture
4.5. Real-Time Quantitative PCR (RT-qPCR)
4.6. Immunofluorescence (IF) Staining
4.7. Determination of Mitochondrial Distribution
4.8. DCFH-DA Staining
4.9. Annexin-V Staining
4.10. Calculation of Expansion Area of Cumulus Cells
4.11. Statistical Analysis of Data
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institution Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Van Grevenynghe, J.; Rion, S.; Le Ferrec, E.; Le Vee, M.; Amiot, L.; Fauchet, R.; Fardel, O. Polycyclic aromatic hydrocarbons inhibit differentiation of human monocytes into macrophages. J. Immunol. 2003, 170, 2374–2381. [Google Scholar] [CrossRef] [PubMed]
- White, A.J.; Bradshaw, P.T.; Herring, A.H.; Teitelbaum, S.L.; Beyea, J.; Stellman, S.D.; Steck, S.E.; Mordukhovich, I.; Eng, S.M.; Engel, L.S.; et al. Exposure to multiple sources of polycyclic aromatic hydrocarbons and breast cancer incidence. Environ. Int. 2016, 89–90, 185–192. [Google Scholar] [CrossRef] [PubMed]
- Hertz-Picciotto, I.; Herr, C.E.; Yap, P.S.; Dostal, M.; Shumway, R.H.; Ashwood, P.; Lipsett, M.; Joad, J.P.; Pinkerton, K.E.; Sram, R.J. Air pollution and lymphocyte phenotype proportions in cord blood. Environ. Health Perspect. 2005, 113, 1391–1398. [Google Scholar] [CrossRef] [PubMed]
- Hertz-Picciotto, I.; Dosta, M.; Dejmek, J.; Selevan, S.G.; Wegienka, G.; Gomez-Caminero, A.; Sram, R.J. Air Pollution and Distributions of Lymphocyte Immunophenotypes in Cord and Maternal Blood at Delivery. Epidemiology 2002, 13, 172–183. [Google Scholar] [CrossRef]
- Jedrychowski, W.A.; Perera, F.P.; Majewska, R.; Camman, D.; Spengler, J.D.; Mroz, E.; Stigter, L.; Flak, E.; Jacek, R. Separate and joint effects of tranplacental and postnatal inhalatory exposure to polycyclic aromatic hydrocarbons: Prospective birth cohort study on wheezing events. Pediatr. Pulmonol. 2014, 49, 162–172. [Google Scholar] [CrossRef]
- Wu, Q.; Ohsako, S.; Baba, T. Effects of 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD) on preimplantation mouse embryos. Toxicology 2002, 174, 119–129. [Google Scholar] [CrossRef]
- Matthews, M.; Heimler, I.; Fahy, M.; Radwanska, E.; Hutz, R.; Trewin, A.; Rawlins, R. Effects of dioxin an environmental pollutant on mouse blastocyst development and apoptosis. Fertil. Steril. 2001, 75, 1159–1162. [Google Scholar] [CrossRef]
- Bruggeman, V.; Swennen, Q.; Ketelaere, B.D.; Onagbesan, O.; Tona, K.; Decuypere, E. Embryonic exposure to 2,3,7,8-tetrachlorodibenzo-p-dioxin in chickens: Effects of dose and embryonic stage on hatchability and growth. Comp. Biochem. Physiol. Part C Toxicol. Pharmacol. 2003, 136, 17–28. [Google Scholar] [CrossRef]
- Johansson, A. Impact of Chemical Warfare with Agent Orange on Women’s Reproductive Lives in Vietnam: A Pilot Study. Reprod. Health Matters 2001, 9, 156–164. [Google Scholar]
- Perera, F.P.; Wang, S.; Rauh, V.; Zhou, H.; Stigter, L.; Camann, D.; Jedrychowski, W.; Mroz, E.; Majewska, R. Prenatal exposure to air pollution, maternal psychological distress, and child behavior. Pediatrics 2013, 132, e1284–e1294. [Google Scholar] [CrossRef]
- Bilbo, S.D.; Schwarz, J.M. The immune system and developmental programming of brain and behavior. Front. Neuroendocrinol. 2012, 33, 267–286. [Google Scholar] [CrossRef]
- Heck, J.E.; Park, A.S.; Qiu, J.; Cockburn, M.; Ritz, B. An exploratory study of ambient air toxics exposure in pregnancy and the risk of neuroblastoma in offspring. Environ. Res. 2013, 127, 1–6. [Google Scholar] [CrossRef]
- Brouwer, A.; Ahlborg, U.G.; Leeuwen, F.X.R.v.; Feeley, M.M. Report of the WHO working group on the assessment of health risks for human infants from exposure to PCDDs, PCDFs and PCBs. Chemosphere 1998, 37, 1627–1643. [Google Scholar] [CrossRef]
- Luebke, R.W.; Copeland, C.B.; Daniels, M.; Lambert, A.L.; Gilmour, M.I. Suppression of allergic immune responses to house dust mite (HDM) in rats exposed to 2,3,7,8-TCDD. Toxicolog. Soc. Toxicol. 2001, 62, 71–79. [Google Scholar] [CrossRef]
- Moser, G.A.; McLachlan, M.S. The influence of dietary concentration on the absorption and excretion of persistent lipophilic organic pollutants in the human intestinal tract. Chemosphere 2001, 45, 201–211. [Google Scholar] [CrossRef]
- Guo, Y.; Hendrickx, A.G.; Overstreet, J.W.; Dieter, J.; Stewart, D.; Tarantal, A.F.; Laughlin, L.; Lasley, B.L. Endocrine Biomarkers of Early Fetal Loss in Cynomolgus Macaques (Macaca fascicularis) Following Exposure to Dioxin1. Biol. Reprod. 1999, 60, 707–713. [Google Scholar] [CrossRef]
- Talcott, P.A.; Exon, J.H.; Mather, G.G.; Koller, L.D. Alterations of rat natural killer (NK) cell cytotoxicity and cytokine production by 3-methylcholanthrene (3-MC). Int. J. Immunopharmacol. 1990, 12, 917–926. [Google Scholar] [CrossRef]
- Devrim, T.; Ekici, H.; Devrim, A.K.; Sozmen, M.; Senol, A.; Bozkur, K.K.; Duru, O.; Yalcin, S. Late effects of cutaneous 3-methylcholanthrene exposure on DNA damage-related pleiotropic growth factors and oxidative stress markers in mice. Bratisl. Med. J. 2020, 121, 325–330. [Google Scholar] [CrossRef]
- Reynaud, S.; Deschaux, P. The effects of 3-methylcholanthrene on lymphocyte proliferation in the common carp (Cyprinus carpio L.). Toxicology 2005, 211, 156–164. [Google Scholar] [CrossRef]
- Laize, V.; Gavaia, P.J.; Tarasco, M.; Viegas, M.N.; Caria, J.; Luis, N.; Cancela, M.L. Osteotoxicity of 3-methylcholanthrene in fish. Ecotoxicol. Environ. Saf. 2018, 161, 721–728. [Google Scholar] [CrossRef]
- Helle, J.; Keiler, A.M.; Zierau, O.; Dorfelt, P.; Vollmer, G.; Lehmann, L.; Chittur, S.V.; Tenniswood, M.; Welsh, J.; Kretzschmar, G. Effects of the aryl hydrocarbon receptor agonist 3-methylcholanthrene on the 17beta-estradiol regulated mRNA transcriptome of the rat uterus. J. Steroid Biochem. Mol. Biol. 2017, 171, 133–143. [Google Scholar] [CrossRef] [PubMed]
- Flisikowska, T.; Kind, A.; Schnieke, A. Genetically modified pigs to model human diseases. J. Appl. Genet. 2014, 55, 53–64. [Google Scholar] [CrossRef] [PubMed]
- Lqbal, M.A.; Hong, K.; Kim, J.H.; Choi, Y. Severe combined immunodeficiency pig as an emerging animal model for human diseases and regenerative medicines. BMB Rep. 2019, 52, 625–634. [Google Scholar] [CrossRef]
- Kobayash, E.; Hishikawa, S.; Teratani, T.; Lefor, A.T. The pig as a model for translational researchv overview of porcine animal models at Jichi Medical University. Transplant. Res. 2012, 16, 8. [Google Scholar] [CrossRef]
- Juan, S.H.; Lee, J.L.; Ho, P.Y.; Lee, Y.H.; Lee, W.S. Antiproliferative and antiangiogenic effects of 3-methylcholanthrene, an aryl-hydrocarbon receptor agonist, in human umbilical vascular endothelial cells. Eur. J. Pharmacol. 2006, 530, 1–8. [Google Scholar] [CrossRef]
- Reynaud, S.; Duchiron, C.; Deschaux, P. 3-Methylcholanthrene induces lymphocyte and phagocyte apoptosis in common carp (Cyprinus carpio L) in vitro. Aquat. Toxicol. 2004, 66, 307–318. [Google Scholar] [CrossRef]
- Swedenborg, E.; Ruegg, J.; Hillenweck, A.; Rehnmark, S.; Faulds, M.H.; Zalko, D.; Pongratz, I.; Pettersson, K. 3-Methylcholanthrene displays dual effects on estrogen receptor (ER) alpha and ER beta signaling in a cell-type specific fashion. Mol. Pharmacol. 2008, 73, 575–586. [Google Scholar] [CrossRef]
- Lasnitzki, I.; Bard, D.R.; Franklin, H.R. 3-Methylcholanthrene uptake and metabolism in organ culture. Br. J. Cancer 1975, 32, 219–229. [Google Scholar] [CrossRef]
- Rhon Calderon, E.A.; Galarza, R.A.; Faletti, A.G. 3-Methylcholanthrene impacts on the female germ cells of rats without causing systemic toxicity. Toxicology 2020, 429, 152328. [Google Scholar] [CrossRef]
- Rhon-Calderon, E.A.; Toro, C.A.; Lomniczi, A.; Galarza, R.A.; Faletti, A.G. Changes in the expression of genes involved in the ovarian function of rats caused by daily exposure to 3-methylcholanthrene and their prevention by alpha-naphthoflavone. Arch. Toxicol. 2018, 92, 907–919. [Google Scholar] [CrossRef]
- Jr, O.B.; Murrill, E.; Fanska, C. Metabolism of 3-methylcholanthrene in rat pancreas. Dig. Dis. Sci. 1981, 26, 219–229. [Google Scholar]
- Zhang, M.; Miao, Y.; Chen, Q.; Cai, M.; Dong, W.; Dai, X.; Lu, Y.; Zhou, C.; Cui, Z.; Xiong, B. BaP exposure causes oocyte meiotic arrest and fertilization failure to weaken female fertility. FASEB J. 2018, 32, 342–352. [Google Scholar] [CrossRef]
- Zhang, L.; Hou, X.; Ma, R.; Moley, K.; Schedl, T.; Wang, Q. Sirt2 functions in spindle organization and chromosome alignment in mouse oocyte meiosis. FASEB J. 2014, 28, 1435–1445. [Google Scholar] [CrossRef]
- Miao, Y.; Zhou, C.; Bai, Q.; Cui, Z.; ShiYang, X.; Lu, Y.; Zhang, M.; Dai, X.; Xiong, B. The protective role of melatonin in porcine oocyte meiotic failure caused by the exposure to benzo(a)pyrene. Hum. Reprod. 2018, 33, 116–127. [Google Scholar] [CrossRef]
- Mikkelsen, A.L.; Lindenberg, S. Morphology of in-vitro matured oocytes:impact on fertility potential and embryo quality. Hum. Reprod. 2001, 16, 1714–1718. [Google Scholar] [CrossRef]
- Cha, K.Y.; Chian, R.C. Maturation in vitro of immature human oocytes for clinical use. Hum. Reprod. 1998, 49, 103–120. [Google Scholar] [CrossRef]
- Tohyama, C. Clarification of Molecular Targets of Dioxin Toxicity. Nihon Eiseigaku Zasshi Jpn. J. Hyg. 2014, 1, 1–7. [Google Scholar] [CrossRef]
- Yoshioka, W.; Tohyama, C. Mechanisms of Developmental Toxicity of Dioxins and Related Compounds. Int. J. Mol. Sci. 2019, 20, 617. [Google Scholar] [CrossRef]
- Han, Y.; Liu, W.; Lei, R.; Wang, M.; Xue, Y. Exposure levels of PCDD/Fs and PCBs in human blood and the transplacental transfer characteristics in cord blood of newborns near the industrialized area. Chemosphere 2022, 303 Pt 1, 134995. [Google Scholar] [CrossRef]
- Petroff, B.K.; Valdez, K.E.; Brown, S.B.; Piasecka, J.; Albertini, D.F. The aryl hydrocarbon receptor agonist 2,3,7,8-tetrachloro-dibenzo-p-dioxin (TCDD) alters early embryonic development in a rat IVF exposure model. Reprod. Toxicol. 2011, 32, 286–292. [Google Scholar] [CrossRef]
- Diaz, F.J.; O’Brien, M.J.; Wigglesworth, K.; Eppig, J.J. The preantral granulosa cell to cumulus cell transition in the mouse ovary: Development of competence to undergo expansion. Develomental Biol. 2006, 299, 91–104. [Google Scholar] [CrossRef] [PubMed]
- Fulop, C.; Szanto, S.; Mukhopadhyay, D.; Bardos, T.; Kamath, R.V.; Rugg, M.S.; Day, A.J.; Salustri, A.; Hascall, V.C.; Glant, T.T.; et al. Impaired cumulus mucification and female sterility in tumor necrosis factor-induced protein-6 deficient mice. Development 2003, 130, 2253–2261. [Google Scholar] [CrossRef] [PubMed]
- Gu, L.; Li, X.; Liu, X.; Gao, M.; He, Y.; Xiong, B.; Liu, H. HDAC3 inhibition disrupts the assembly of meiotic apparatus during porcine oocyte maturation. J. Cell Physiol. 2019, 234, 10178–10183. [Google Scholar] [CrossRef] [PubMed]
- Montagnac, G.; Meas-Yedid, V.; Irondelle, M.; Castro-Castro, A.; Franco, M.; Shida, T.; Nachury, M.V.; Benmerah, A.; Olivo-Marin, J.C.; Chavrier, P. alphaTAT1 catalyses microtubule acetylation at clathrin-coated pits. Nature 2013, 502, 567–570. [Google Scholar] [CrossRef]
- Miao, Y.; Zhou, C.; Cui, Z.; Zhang, M.; ShiYang, X.; Lu, Y.; Xiong, B. Postovulatory aging causes the deterioration of porcine oocytes via induction of oxidative stress. FASEB J. 2018, 32, 1328–1337. [Google Scholar] [CrossRef]
- Lu, Y.; Li, S.; Cui, Z.; Dai, X.; Zhang, M.; Miao, Y.; Zhou, C.; Ou, X.; Xiong, B. The cohesion establishment factor Esco1 acetylates alpha-tubulin to ensure proper spindle assembly in oocyte meiosis. Nucleic Acids Res. 2018, 46, 2335–2346. [Google Scholar] [CrossRef]
- Xu, Z.; Schaedel, L.; Portra, D.; Aguilar, A.; Gaillard, J.; Marinkovich, M.P.; Théry, M.; Nachury, M.V. Microtubules acquire resistance from mechanical breakage through intralumenal acetylation. Science 2017, 356, 328–332. [Google Scholar] [CrossRef]
- Hu, L.L.; Pan, M.H.; Yang, F.L.; Zong, Z.A.; Tang, F.; Pan, Z.N.; Lu, X.; Ren, Y.P.; Wang, J.L.; Sun, S.C. FASCIN regulates actin assembly for spindle movement and polar body extrusion in mouse oocyte meiosis. J. Cell Physiol. 2021, 236, 7725–7733. [Google Scholar] [CrossRef]
- Duan, X.; Sun, S.C. Actin cytoskeleton dynamics in mammalian oocyte meiosis. Biol. Reprod. 2019, 100, 15–24. [Google Scholar] [CrossRef]
- Kunda, P.; Baum, B. The actin cytoskeleton in spindle assembly and positioning. Trends Cell Biol. 2009, 19, 174–179. [Google Scholar] [CrossRef]
- Azoury, J.; Verlhac, M.H.; Dumont, J. Actin filaments: Key players in the control of asymmetric divisions in mouse oocytes. Biol. Cell 2009, 101, 69–76. [Google Scholar] [CrossRef]
- Coticchio, G.; Dal Canto, M.; Mignini Renzini, M.; Guglielmo, M.C.; Brambillasca, F.; Turchi, D.; Novara, P.V.; Fadini, R. Oocyte maturation: Gamete-somatic cells interactions, meiotic resumption, cytoskeletal dynamics and cytoplasmic reorganization. Hum. Reprod. Update 2015, 21, 427–454. [Google Scholar] [CrossRef]
- Mao, L.; Lou, H.; Lou, Y.; Wang, N.; Jin, F. Behaviour of cytoplasmic organelles and cytoskeleton during oocyte maturation. Reprod. Biomed. Online 2014, 28, 284–299. [Google Scholar] [CrossRef]
- Krisher, R.L.; Bavister, B.D. Responses of oocytes and embryos to the culture environment. Theriogenology 1998, 49, 103–114. [Google Scholar] [CrossRef]
- Lemaire, B.; Beck, M.; Jaspart, M.; Debier, C.; Calderon, P.B.; Thome, J.P.; Rees, J.F. Precision-Cut Liver Slices of Salmo salar as a tool to investigate the oxidative impact of CYP1A-mediated PCB 126 and 3-methylcholanthrene metabolism. Toxicol. Vitr. 2011, 25, 335–342. [Google Scholar] [CrossRef]
- Sies, H. Oxidative stress: A concept in redox biology and medicine. Redox Biol. 2015, 4, 180–183. [Google Scholar] [CrossRef]
- Behrman, H.R.; Kodaman, P.H.; Preston, S.L.; Gao, S. Oxidative stress and the ovary. J. Soc. Gynecol. Investig. 2001, 8, S40–S42. [Google Scholar]
- Agarwal, A.; Aponte-Mellado, A.; Premkumar, B.J.; Shaman, A.; Gupta, S. The effects of oxidative stress on female reproduction: A review. Reprod. Biol. Endocrinol. 2012, 10, 49. [Google Scholar] [CrossRef]
- Wang, L.; Tang, J.; Tan, F.; Song, H.; Zhou, J.; Li, F. Oxidative stress in oocyte aging and female reproduction. J. Cell. Physiol. 2021, 236, 7966–7983. [Google Scholar] [CrossRef]
- Raisova, V.; Hossini, A.M.; Eberle, J.; Riebeling, C.; Wieder, T.; Sturm, I.; Daniel, P.T.; Orfanos, C.E.; Geilen, C.C. The Bax/Bcl-2 ratio determines the susceptibility of human melanoma cells to CD95/Fas-mediated apoptosis. J. Investig. Dermatol. 2001, 117, 333–340. [Google Scholar] [CrossRef]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, M.; Wang, X.; Liu, Q.; Yan, Y.; Xu, C.; Zhu, Z.; Wu, S.; Zuo, X.; Zong, Y.; Li, C.; et al. Inhibitory Effects of 3-Methylcholanthrene Exposure on Porcine Oocyte Maturation. Int. J. Mol. Sci. 2023, 24, 5567. https://doi.org/10.3390/ijms24065567
Zhang M, Wang X, Liu Q, Yan Y, Xu C, Zhu Z, Wu S, Zuo X, Zong Y, Li C, et al. Inhibitory Effects of 3-Methylcholanthrene Exposure on Porcine Oocyte Maturation. International Journal of Molecular Sciences. 2023; 24(6):5567. https://doi.org/10.3390/ijms24065567
Chicago/Turabian StyleZhang, Mengya, Xin Wang, Qiuchen Liu, Yelian Yan, Changzhi Xu, Zhihua Zhu, Sucheng Wu, Xiaoyu Zuo, Yanfeng Zong, Chunxiao Li, and et al. 2023. "Inhibitory Effects of 3-Methylcholanthrene Exposure on Porcine Oocyte Maturation" International Journal of Molecular Sciences 24, no. 6: 5567. https://doi.org/10.3390/ijms24065567
APA StyleZhang, M., Wang, X., Liu, Q., Yan, Y., Xu, C., Zhu, Z., Wu, S., Zuo, X., Zong, Y., Li, C., Zhang, Y., & Cao, Z. (2023). Inhibitory Effects of 3-Methylcholanthrene Exposure on Porcine Oocyte Maturation. International Journal of Molecular Sciences, 24(6), 5567. https://doi.org/10.3390/ijms24065567





