Markers of Iron Metabolism and Outcomes in Patients with Heart Failure: A Systematic Review
Abstract
1. Introduction
2. Methods
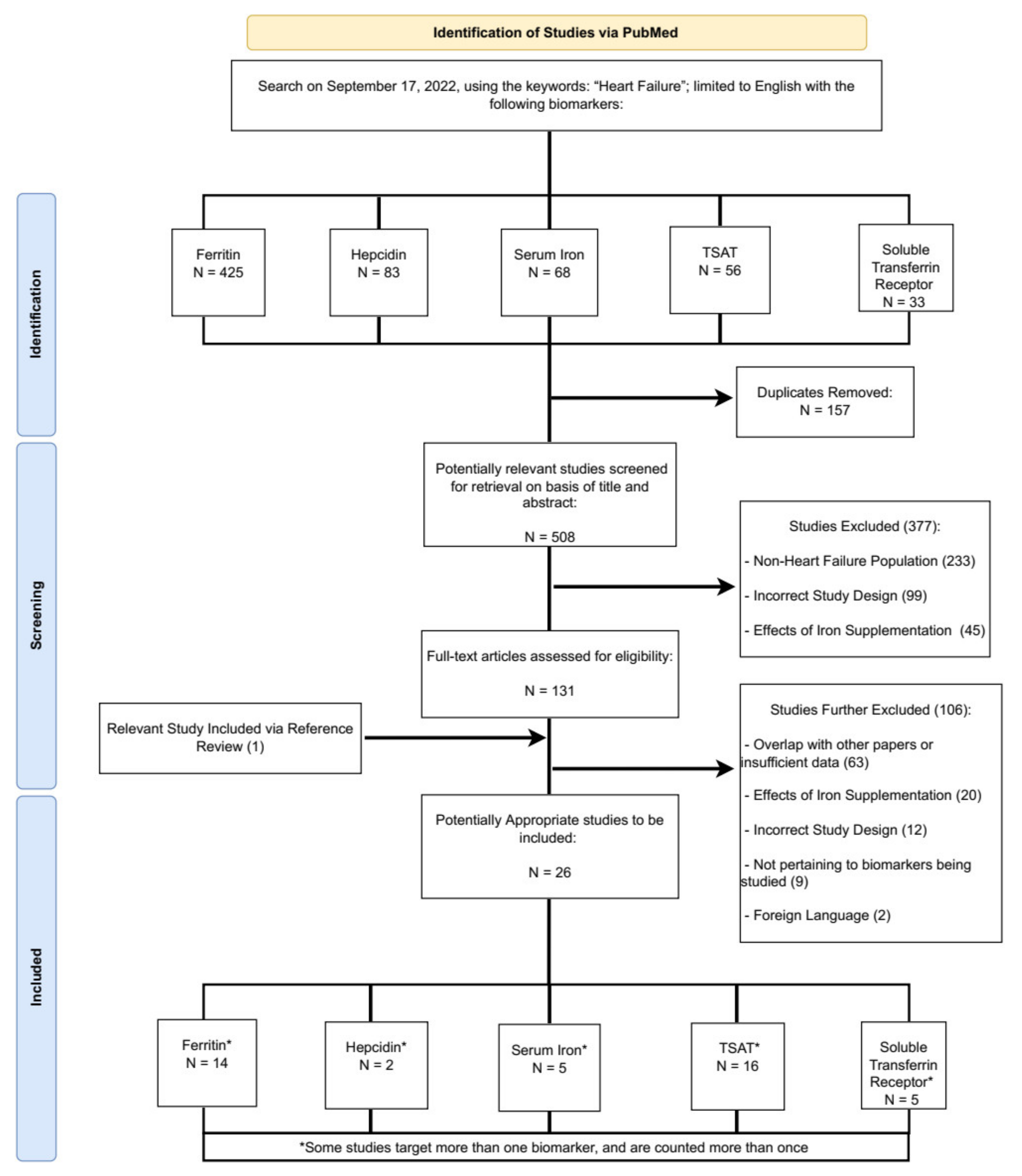
2.1. Data Source/Search Strategy
2.2. Study Selection
2.3. Study Classification
- All-Cause Mortality
- HF hospitalization
- Functional Capacity/Quality of Life
- Additional Adverse Outcomes
2.4. Study Quality/Data Synthesis
2.5. Risk of Bias
3. Results
3.1. All-Cause Mortality
3.2. Heart Failure Hospitalization
3.3. Exercise/Functional Capacity and Quality of Life
3.4. Additional Adverse Outcomes
4. Discussion
4.1. Iron Metabolism and Mortality
4.2. Iron Metabolism and Heart Failure Hospitalizations
4.3. Iron Metabolism and Other Outcomes
4.4. Future Directions
5. Limitations
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| AKI | Acute renal injury |
| BNP | Brain natriuretic peptide |
| CRS | Cardiorenal syndrome |
| ESC | European Society of Cardiology |
| HF | Heart failure |
| HFmrEF | Heart failure with midrange ejection fraction |
| HFpEF | Heart failure with preserved ejection fraction |
| HFrEF | Heart failure with reduced ejection fraction |
| ID | Iron deficiency |
| IV | Intravenous |
| LVEF | Left ventricular ejection fraction |
| QoL | Quality of life |
| STfR | Soluble transferrin receptor |
| TSAT | Transferrin saturation |
References
- Von Haehling, S.; Gremmler, U.; Krumm, M.; Mibach, F.; Schön, N.; Taggeselle, J.; Dahm, J.B.; Angermann, C.E. Prevalence and clinical impact of iron deficiency and anaemia among outpatients with chronic heart failure: The PrEP Registry. Clin. Res. Cardiol. 2017, 106, 436–443. [Google Scholar] [CrossRef]
- Dignass, A.; Farrag, K.; Stein, J. Limitations of Serum Ferritin in Diagnosing Iron Deficiency in Inflammatory Conditions. Int. J. Chronic. Dis. 2018, 2018, 9394060. [Google Scholar] [CrossRef] [PubMed]
- Pezel, T.; Audureau, E.; Mansourati, J.; Baudry, G.; Ben Driss, A.; Durup, F.; Fertin, M.; Godreuil, C.; Jeanneteau, J.; Kloeckner, M.; et al. Diagnosis and Treatment of Iron Deficiency in Heart Failure: OFICSel study by the French Heart Failure Working Group. ESC Heart Fail. 2021, 8, 1509–1521. [Google Scholar] [CrossRef] [PubMed]
- Klip, I.T.; Comin-Colet, J.; Voors, A.A.; Ponikowski, P.; Enjuanes, C.; Banasiak, W.; Lok, D.J.; Rosentryt, P.; Torrens, A.; Polonski, L.; et al. Iron deficiency in chronic heart failure: An international pooled analysis. Am. Heart J. 2013, 165, 575–582.e573. [Google Scholar] [CrossRef] [PubMed]
- Van der Wal, H.H.; Grote Beverborg, N.; Dickstein, K.; Anker, S.D.; Lang, C.C.; Ng, L.L.; van Veldhuisen, D.J.; Voors, A.A.; van der Meer, P. Iron deficiency in worsening heart failure is associated with reduced estimated protein intake, fluid retention, inflammation, and antiplatelet use. Eur. Heart J. 2019, 40, 3616–3625. [Google Scholar] [CrossRef]
- Martens, P. The Effect of Iron Deficiency on Cardiac Function and Structure in Heart Failure with Reduced Ejection Fraction. Card. Fail. Rev. 2022, 8, e06. [Google Scholar] [CrossRef]
- Beale, A.L.; Warren, J.L.; Roberts, N.; Meyer, P.; Townsend, N.P.; Kaye, D. Iron deficiency in heart failure with preserved ejection fraction: A systematic review and meta-analysis. Open Heart 2019, 6, e001012. [Google Scholar] [CrossRef]
- Correale, M.; Paolillo, S.; Mercurio, V.; Limongelli, G.; Barillà, F.; Ruocco, G.; Palazzuoli, A.; Scrutinio, D.; Lagioia, R.; Lombardi, C.; et al. Comorbidities in chronic heart failure: An update from Italian Society of Cardiology (SIC) Working Group on Heart Failure. Eur. J. Intern. Med. 2020, 71, 23–31. [Google Scholar] [CrossRef]
- Nakano, H.; Nagai, T.; Sundaram, V.; Nakai, M.; Nishimura, K.; Honda, Y.; Honda, S.; Iwakami, N.; Sugano, Y.; Asaumi, Y.; et al. Impact of iron deficiency on long-term clinical outcomes of hospitalized patients with heart failure. Int. J. Cardiol. 2018, 261, 114–118. [Google Scholar] [CrossRef]
- Beale, A.; Carballo, D.; Stirnemann, J.; Garin, N.; Agoritsas, T.; Serratrice, J.; Kaye, D.; Meyer, P.; Carballo, S. Iron Deficiency in Acute Decompensated Heart Failure. J. Clin. Med. 2019, 8, 1569. [Google Scholar] [CrossRef]
- Núñez, J.; Comín-Colet, J.; Miñana, G.; Núñez, E.; Santas, E.; Mollar, A.; Valero, E.; García-Blas, S.; Cardells, I.; Bodí, V.; et al. Iron deficiency and risk of early readmission following a hospitalization for acute heart failure. Eur. J. Heart Fail. 2016, 18, 798–802. [Google Scholar] [CrossRef]
- Ghafourian, K.; Shapiro, J.S.; Goodman, L.; Ardehali, H. Iron and Heart Failure: Diagnosis, Therapies, and Future Directions. JACC Basic Transl. Sci. 2020, 5, 300–313. [Google Scholar] [CrossRef]
- Kalra, P.R.; Cleland, J.G.F.; Petrie, M.C.; Thomson, E.A.; Kalra, P.A.; Squire, I.B.; Ahmed, F.Z.; Al-Mohammad, A.; Cowburn, P.J.; Foley, P.W.X.; et al. Intravenous ferric derisomaltose in patients with heart failure and iron deficiency in the UK (IRONMAN): An investigator-initiated, prospective, randomised, open-label, blinded-endpoint trial. Lancet 2022, 400, 2199–2209. [Google Scholar] [CrossRef]
- Graham, F.J.; Pellicori, P.; Kalra, P.; Ford, I.; Cleland, J.G.F. Intravenous iron in patients with heart failure and iron deficiency: An updated metaanalysis. Eur. J. Heart Fail. 2023. online ahead of print. [Google Scholar] [CrossRef] [PubMed]
- Beck-da-Silva, L.; Piardi, D.; Soder, S.; Rohde, L.E.; Pereira-Barretto, A.C.; de Albuquerque, D.; Bocchi, E.; Vilas-Boas, F.; Moura, L.Z.; Montera, M.W.; et al. IRON-HF study: A randomized trial to assess the effects of iron in heart failure patients with anemia. Int. J. Cardiol. 2013, 168, 3439–3442. [Google Scholar] [CrossRef]
- Lewis, G.D.; Malhotra, R.; Hernandez, A.F.; McNulty, S.E.; Smith, A.; Felker, G.M.; Tang, W.H.W.; LaRue, S.J.; Redfield, M.M.; Semigran, M.J.; et al. Effect of Oral Iron Repletion on Exercise Capacity in Patients With Heart Failure With Reduced Ejection Fraction and Iron Deficiency: The IRONOUT HF Randomized Clinical Trial. JAMA 2017, 317, 1958–1966. [Google Scholar] [CrossRef] [PubMed]
- Packer, M. How can sodium-glucose cotransporter 2 inhibitors stimulate erythrocytosis in patients who are iron-deficient? Implications for understanding iron homeostasis in heart failure. Eur. J. Heart Fail. 2022, 24, 2287–2296. [Google Scholar] [CrossRef]
- McDonagh, T.A.; Metra, M.; Adamo, M.; Gardner, R.S.; Baumbach, A.; Böhm, M.; Burri, H.; Butler, J.; Čelutkienė, J.; Chioncel, O.; et al. 2021 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure: Developed by the Task Force for the diagnosis and treatment of acute and chronic heart failure of the European Society of Cardiology (ESC) With the special contribution of the Heart Failure Association (HFA) of the ESC. Eur. Heart J. 2021, 42, 3599–3726. [Google Scholar] [CrossRef] [PubMed]
- Heidenreich, P.A.; Bozkurt, B.; Aguilar, D.; Allen, L.A.; Byun, J.J.; Colvin, M.M.; Deswal, A.; Drazner, M.H.; Dunlay, S.M.; Evers, L.R.; et al. 2022 AHA/ACC/HFSA Guideline for the Management of Heart Failure. J. Am. Coll. Cardiol. 2022, 79, e263–e421. [Google Scholar] [CrossRef] [PubMed]
- Jankowska, E.A.; Rozentryt, P.; Witkowska, A.; Nowak, J.; Hartmann, O.; Ponikowska, B.; Borodulin-Nadzieja, L.; Banasiak, W.; Polonski, L.; Filippatos, G.; et al. Iron deficiency: An ominous sign in patients with systolic chronic heart failure. Eur. Heart J. 2010, 31, 1872–1880. [Google Scholar] [CrossRef]
- Fitzsimons, S.; Poppe, K.K.; Choi, Y.; Devlin, G.; Lund, M.; Lam, C.S.P.; Troughton, R.; Richards, A.M.; Doughty, R.N. Relationship Between Soluble Transferrin Receptor and Clinical Outcomes in Patients With Heart Failure According to Ejection Fraction Phenotype: The New Zealand PEOPLE Study. J. Card. Fail. 2022, 28, 1255–1263. [Google Scholar] [CrossRef]
- Pozzo, J.; Fournier, P.; Delmas, C.; Vervueren, P.L.; Roncalli, J.; Elbaz, M.; Galinier, M.; Lairez, O. Absolute iron deficiency without anaemia in patients with chronic systolic heart failure is associated with poorer functional capacity. Arch. Cardiovasc. Dis. 2017, 110, 99–105. [Google Scholar] [CrossRef]
- Alcaide-Aldeano, A.; Garay, A.; Alcoberro, L.; Jiménez-Marrero, S.; Yun, S.; Tajes, M.; García-Romero, E.; Díez-López, C.; González-Costello, J.; Mateus-Porta, G.; et al. Iron Deficiency: Impact on Functional Capacity and Quality of Life in Heart Failure with Preserved Ejection Fraction. J. Clin. Med. 2020, 9, 1199. [Google Scholar] [CrossRef]
- Yan, J.; Pan, Y.; He, Y.; Wang, R.; Shao, W.; Dong, S. The effects of serum iron level without anemia on long-term prognosis of patients with coronary heart disease complicated with chronic heart failure: A retrospective cohort study. Heart Vessel. 2020, 35, 1419–1428. [Google Scholar] [CrossRef] [PubMed]
- Okonko, D.O.; Mandal, A.K.; Missouris, C.G.; Poole-Wilson, P.A. Disordered iron homeostasis in chronic heart failure: Prevalence, predictors, and relation to anemia, exercise capacity, and survival. J. Am. Coll. Cardiol. 2011, 58, 1241–1251. [Google Scholar] [CrossRef] [PubMed]
- Grote Beverborg, N.; Klip, I.T.; Meijers, W.C.; Voors, A.A.; Vegter, E.L.; van der Wal, H.H.; Swinkels, D.W.; van Pelt, J.; Mulder, A.B.; Bulstra, S.K.; et al. Definition of Iron Deficiency Based on the Gold Standard of Bone Marrow Iron Staining in Heart Failure Patients. Circ. Heart Fail. 2018, 11, e004519. [Google Scholar] [CrossRef] [PubMed]
- Gentil, J.R.S.; Fabricio, C.G.; Tanaka, D.M.; Suen, V.M.M.; Volpe, G.J.; Marchini, J.S.; Simões, M.V. Should we use ferritin in the diagnostic criteria of iron deficiency in heart failure patients? Clin. Nutr. ESPEN 2020, 39, 119–123. [Google Scholar] [CrossRef] [PubMed]
- Kurz, K.; Lanser, L.; Seifert, M.; Kocher, F.; Pölzl, G.; Weiss, G. Anaemia, iron status, and gender predict the outcome in patients with chronic heart failure. ESC Heart Fail. 2020, 7, 1880–1890. [Google Scholar] [CrossRef]
- Fitzsimons, S.; Yeo, T.J.; Ling, L.H.; Sim, D.; Leong, K.T.G.; Yeo, P.S.D.; Ong, H.Y.; Jaufeerally, F.; Ng, T.P.; Poppe, K.; et al. Impact of change in iron status over time on clinical outcomes in heart failure according to ejection fraction phenotype. ESC Heart Fail. 2021, 8, 4572–4583. [Google Scholar] [CrossRef]
- Ambrosy, A.P.; Fitzpatrick, J.K.; Tabada, G.H.; Gurwitz, J.H.; Artz, A.; Schrier, S.L.; Rao, S.V.; Reynolds, K.; Smith, D.H.; Peterson, P.N.; et al. A reduced transferrin saturation is independently associated with excess morbidity and mortality in older adults with heart failure and incident anemia. Int. J. Cardiol. 2020, 309, 95–99. [Google Scholar] [CrossRef]
- Palau, P.; Llàcer, P.; Domínguez, E.; Tormo, J.P.; Zakarne, R.; Mollar, A.; Martínez, A.; Miñana, G.; Santas, E.; Almenar, L.; et al. Iron deficiency and short-term adverse events in patients with decompensated heart failure. Clin. Res. Cardiol. 2021, 110, 1292–1298. [Google Scholar] [CrossRef] [PubMed]
- Masini, G.; Graham, F.J.; Pellicori, P.; Cleland, J.G.F.; Cuthbert, J.J.; Kazmi, S.; Inciardi, R.M.; Clark, A.L. Criteria for Iron Deficiency in Patients With Heart Failure. J. Am. Coll. Cardiol. 2022, 79, 341–351. [Google Scholar] [CrossRef] [PubMed]
- Jankowska, E.A.; Kasztura, M.; Sokolski, M.; Bronisz, M.; Nawrocka, S.; Oleśkowska-Florek, W.; Zymliński, R.; Biegus, J.; Siwołowski, P.; Banasiak, W.; et al. Iron deficiency defined as depleted iron stores accompanied by unmet cellular iron requirements identifies patients at the highest risk of death after an episode of acute heart failure. Eur. Heart J. 2014, 35, 2468–2476. [Google Scholar] [CrossRef]
- Sierpinski, R.; Josiak, K.; Suchocki, T.; Wojtas-Polc, K.; Mazur, G.; Butrym, A.; Rozentryt, P.; van der Meer, P.; Comin-Colet, J.; von Haehling, S.; et al. High soluble transferrin receptor in patients with heart failure: A measure of iron deficiency and a strong predictor of mortality. Eur. J. Heart Fail. 2021, 23, 919–932. [Google Scholar] [CrossRef] [PubMed]
- Jankowska, E.A.; Malyszko, J.; Ardehali, H.; Koc-Zorawska, E.; Banasiak, W.; von Haehling, S.; Macdougall, I.C.; Weiss, G.; McMurray, J.J.; Anker, S.D.; et al. Iron status in patients with chronic heart failure. Eur. Heart J. 2013, 34, 827–834. [Google Scholar] [CrossRef]
- Ueda, T.; Kawakami, R.; Nogi, K.; Nogi, M.; Ishihara, S.; Nakada, Y.; Nakano, T.; Hashimoto, Y.; Nakagawa, H.; Nishida, T.; et al. Serum iron: A new predictor of adverse outcomes independently from serum hemoglobin levels in patients with acute decompensated heart failure. Sci. Rep. 2021, 11, 2395. [Google Scholar] [CrossRef]
- Jankowska, E.A.; Rozentryt, P.; Witkowska, A.; Nowak, J.; Hartmann, O.; Ponikowska, B.; Borodulin-Nadzieja, L.; von Haehling, S.; Doehner, W.; Banasiak, W.; et al. Iron deficiency predicts impaired exercise capacity in patients with systolic chronic heart failure. J. Card. Fail. 2011, 17, 899–906. [Google Scholar] [CrossRef] [PubMed]
- Martens, P.; Nijst, P.; Verbrugge, F.H.; Smeets, K.; Dupont, M.; Mullens, W. Impact of iron deficiency on exercise capacity and outcome in heart failure with reduced, mid-range and preserved ejection fraction. Acta Cardiol. 2018, 73, 115–123. [Google Scholar] [CrossRef] [PubMed]
- Tkaczyszyn, M.; Drozd, M.; Węgrzynowska-Teodorczyk, K.; Flinta, I.; Kobak, K.; Banasiak, W.; Ponikowski, P.; Jankowska, E.A. Depleted iron stores are associated with inspiratory muscle weakness independently of skeletal muscle mass in men with systolic chronic heart failure. J. Cachexia Sarcopenia Muscle 2018, 9, 547–556. [Google Scholar] [CrossRef]
- Bekfani, T.; Pellicori, P.; Morris, D.; Ebner, N.; Valentova, M.; Sandek, A.; Doehner, W.; Cleland, J.G.; Lainscak, M.; Schulze, P.C.; et al. Iron deficiency in patients with heart failure with preserved ejection fraction and its association with reduced exercise capacity, muscle strength and quality of life. Clin. Res. Cardiol. 2019, 108, 203–211. [Google Scholar] [CrossRef] [PubMed]
- Klip, I.T.; Voors, A.A.; Swinkels, D.W.; Bakker, S.J.; Kootstra-Ros, J.E.; Lam, C.S.; van der Harst, P.; van Veldhuisen, D.J.; van der Meer, P. Serum ferritin and risk for new-onset heart failure and cardiovascular events in the community. Eur. J. Heart Fail. 2017, 19, 348–356. [Google Scholar] [CrossRef] [PubMed]
- Ceyhun, G.; Öztürk, M.; Küçüksu, Z.; Aydın, S.; Özkoç, M.; Altınkaya, O.; Ateş, A. B-type natriuretic peptide/ferritin ratio as a predictor of the risk of developing acute renal injury in acute decompensated heart failure. Turk. J. Emerg. Med. 2021, 21, 98–103. [Google Scholar] [CrossRef]
- Naito, Y.; Tsujino, T.; Matsumoto, M.; Sakoda, T.; Ohyanagi, M.; Masuyama, T. Adaptive response of the heart to long-term anemia induced by iron deficiency. Am. J. Physiol. Heart Circ. Physiol. 2009, 296, H585–H593. [Google Scholar] [CrossRef] [PubMed]
- Cohen-Solal, A.; Philip, J.L.; Picard, F.; Delarche, N.; Taldir, G.; Gzara, H.; Korichi, A.; Trochu, J.N.; Cacoub, P. Iron deficiency in heart failure patients: The French CARENFER prospective study. ESC Heart Fail. 2022, 9, 874–884. [Google Scholar] [CrossRef] [PubMed]
- Becher, P.M.; Schrage, B.; Benson, L.; Fudim, M.; Corovic Cabrera, C.; Dahlström, U.; Rosano, G.M.C.; Jankowska, E.A.; Anker, S.D.; Lund, L.H.; et al. Phenotyping heart failure patients for iron deficiency and use of intravenous iron therapy: Data from the Swedish Heart Failure Registry. Eur. J. Heart Fail. 2021, 23, 1844–1854. [Google Scholar] [CrossRef]
- Anker, S.D.; Kirwan, B.A.; van Veldhuisen, D.J.; Filippatos, G.; Comin-Colet, J.; Ruschitzka, F.; Luscher, T.F.; Arutyunov, G.P.; Motro, M.; Mori, C.; et al. Effects of ferric carboxymaltose on hospitalisations and mortality rates in iron-deficient heart failure patients: An individual patient data meta-analysis. Eur. J. Heart Fail. 2018, 20, 125–133. [Google Scholar] [CrossRef]
- Ponikowski, P.; Kirwan, B.A.; Anker, S.D.; McDonagh, T.; Dorobantu, M.; Drozdz, J.; Fabien, V.; Filippatos, G.; Gohring, U.M.; Keren, A.; et al. Ferric carboxymaltose for iron deficiency at discharge after acute heart failure: A multicentre, double-blind, randomised, controlled trial. Lancet 2020, 396, 1895–1904. [Google Scholar] [CrossRef]
- Dallman, P.R. Biochemical basis for the manifestations of iron deficiency. Annu. Rev. Nutr. 1986, 6, 13–40. [Google Scholar] [CrossRef] [PubMed]
- Damman, K.; Testani, J.M. The kidney in heart failure: An update. Eur. Heart J. 2015, 36, 1437–1444. [Google Scholar] [CrossRef]
- Silverberg, D.S.; Wexler, D.; Iaina, A.; Schwartz, D. Correction of iron deficiency in the cardiorenal syndrome. Int. J. Nephrol. 2011, 2011, 365301. [Google Scholar] [CrossRef]
| Author (Year) | N | LVEF (%) | Mean Age (Years) | Male (%) | Follow-Up Period (Days) | Study Type | ID Criteria | Anemia Excluded (Yes/No) | Biomarker(s) | Outcomes | Association of Iron Metabolism Markers with Outcomes |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Jankowska (2010) [20] | 546 | 26 ± 7 | 55 ± 11 | 88 | 731 ± 350 (mean) | P | ESC definitions | No | Ferritin TSAT | Mortality: 28% in 2 years | ↑ morality or HT w/ID, based on ferritin and TSAT (HR 1.58, 95% CI 1.14–2.17, p < 0.01) |
| Okonko (2011) [25] | 157 | 32 ± 9 | 71 ± 12 | 72 | 743 (median) | P | TSAT <20% | No | TSAT | Mortality: 17% in 2 y | ↑ mortality w/TSAT <20% (HR 3.38; 95% CI 1.48−7.72; p = 0.004) |
| Jankowska (2013) [35] | 321 | 31 ± 9 | 61 ± 11 | 84 | 1095 (endpoint) | P | N/A | No | Hepcidin | Mortality: 24% in 3 years | ↑ mortality w/Serum hepcidin ≤31 ng/mL (HR 2.89, 95%CI 1.63–5.05, p < 0.001) |
| Klip (2013) [4] | 1506 | 33 ± 14 | 64 ± 13 | 74 | 701 (median) | P | ESC definitions | Yes (Hg < 8−10 g/dL based on cohort) | Ferritin TSAT | Mortality: 29.2% in 2 years | ↑ morality w/ID, based on ferritin and TSAT (HR 1.42, 95%CI 1.14−1.77, p = 0.002) |
| Jankowska (2014) [33] | 165 | 33 ± 13 | 65 ± 12 | 81 | 323 ± 94 (mean) | P | (a) Hepcidin <14.5 ng/mL; (b) sTfR ≥1.59 mg/L; (c) ESC definitions | No | Hepcidin sTfR | Mortality in low hepcidin AND high sTfR: 41% in 1 year | ↑ morality w/Low Hepcidin <7.84 ng/mL and High sTfR >2.78 mg/L (HR = 6.16, 95% CI = 2.70–14.04, p < 0.001) |
| Grote Beverborg (2018) [26] | 387 | 30.4 ± 9.3 | 66.8 ± 13.4 | 68 | 746 (endpoint) | P | ESC definitions | No | Iron TSAT | Mortality: 25% in 1.5 years | ↑ mortality w/serum iron ≤13 µmol/L (HR 2.39; 95% CI 1.13–5.04; p = 0.022); ↑ mortality w/TSAT <20% (HR 2.78; 95% CI 1.22–6.34, p = 0.015); Ferritin <100 ng/mL not significantly associated w/mortality (HR 1.54; 95% CI 0.45–5.52) |
| Nakano (2018) [9] | 578 | 39 (median) | 78 (median) | 61 | 365 (endpoint) | P | ESC definitions | No | Ferritin | Mortality: 11% in 1 year | ↑ adverse events w/Ferritin <100 μg/L (HR 1.50, 95% CI 1.02–2.21, p = 0.040); FID with neutral effect (HR 0.73, 95% CI 0.39–1.34, p = 0.31) |
| Ambrosy (2020) [30] | 4103 | 35% with LVEF > 50% | 79.2 ± 7 | 51 | 365 (endpoint) | P | ESC definitions | No | TSAT | Mortality and HF Hospitalization: 16.7 per 100 person-years | ↑ mortality w/TSAT < 20% compared to ferritin <100 ng/mL (aHR 1.42, 95% CI:1.20–1.68) and ferritin 100–300 ng/mL (aHR 1.18, 95% CI:1.00–1.38) |
| Kurz (2020) [28] | 2223 | 34 ± 15 | 57.2 ± 14.7 | 72 | 2068 (median) | P | ESC definitions | No | TSAT | Mortality: 8% in 5.6 years | ↑ mortality, HT, VAD implant w/TSAT < 20% (HR 0.608, 95% CI 0.498–0.741, p < 0.001) Neutral Effect when comparing ferritin levels 100–300 μg/L vs. <100 μg/L (HR 0.857, 95% CI 0.643–1.144, p = 0.298) |
| Gentil (2020) [27] | 108 | 28.1 ± 9.2 | 59.5 ± 14.1 | 52.8 | 857.5 (median) | P | ESC definitions | No | TSAT | Mortality: 29% in 2.3 years | ↑ mortality w/TSAT < 20% (HR = 2.3; 95% CI: 1.11–4.85, p = 0.026); Ferritin < 100 μg/L was not related to mortality (Log-rank test, p = 0.439) |
| Sierpinski (2020) [34] | 791 | 28 ± 8 | 58 ± 11 | 84 | 1095 (endpoint) | P | based on ROC for ferritin, TSAT, sTfR | No | sTfR | Mortality: 34% in 3 years | ↓ Survival Rate w/sTfR ≥ 1.25 mg/L: (Z = 4.03, p < 0.001) |
| Fitzsimons (2021) [29] | 1563 | 38 * | 65.6 * | 72 | 730 (endpoint) | P | ESC definitions | No | TSAT Ferritin | Mortality: 19% in 2 years | ↑ mortality w/TSAT < 20% (HR 1.95, CI 95% 1.52−2.51, p < 0.01): ESC definitions had inferior effect (HR 1.21, CI 95% 0.95–1.53, p = 0.12) |
| Palau (2021) [31] | 1701 | 51.7% with LVEF > 50% | 76 (median) | 56 | 30 (endpoint) | P | ESC definitions | No | TSAT | Mortality: 6% in 1 month | ↑ adverse outcomes w/TSAT, exponentially higher if TSAT < 10% (HR 1.75, CI 95% 1.08–2.83; p = 0.024); Ferritin levels, even <54 ug/L, without higher risk (HR 0.97, CI 95% 0.63–1.50; p = 0.906) |
| Ueda (2021) [36] | 615 | 46.7 ± 16.0 | 74.3 ± 12.0 | 57 | 976 (mean) | P | ESC definitions | No | Iron | Mortality: 25% in 2.7 years | ↑ adverse outcomes w/serum iron ≤ 64 µg/dL (irrespective of ferritin/Hg) (HR 1.500; CI 95% 1.128–1.976; p = 0.0044) |
| Fitzsimons (2022) [21] | 788 | 36% with LVEF ≥ 50% | 70.0 * | 69 | 1460 (endpoint) | P | (1) sTfR ≥ 1.59 mg/L, (2) sTfR ≥ 1.76 mg/L (3) ESC definitions (4) TSAT < 20% | No | sTfR TSAT | Mortality and HF hospitalizations: 54% in 4 years | ↑ mortality w/sTfR > 1.76 mg/L in HFpEF (HR 1.84, 95% CI 1.23–2.75); ↑ mortality w/sTfR > 1.59 mg/L regardless LVEF (HR 1.45, 95% CI 1.13–1.86); ↑ mortality w/TSAT < 20% in HFpEF (HR, 1.69, 95% CI 1.10–2.59); ESC definitions were not associated with mortality (HR 1.19, 95% CI 0.77–1.85) |
| Masini (2022) [32] | 4422 | 41% with LVEF ≥ 50% | 75 (median) | 60 | 1490 (median) | P | most accurate biomarker of iron status (ferritin, TSAT, iron) | No | Iron TSAT | Mortality: 34.5% in 5 years | ↑ mortality w/TSAT < 20%(HR: 1.27; 95% CI: 1.14−1.43; p < 0.001); ↑ mortality w/Serum Iron ≤13 μmol/(HR: 1.37; 95% CI: 1.22−1.54; p < 0.001); Serum ferritin < 100 ng/mL insignificantly associated with lower mortality (HR: 0.91; 95% CI: 0.81−1.01; p = 0.09) |
| Author (Year) | N | LVEF (%) | Mean Age (Years) | Male (%) | Follow-Up Period (Days) | Study Type | ID Criteria | Anemia Excluded (Yes/No) | Biomarker(s) | Outcomes | Association of Iron Metabolism Markers with Outcomes |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Núñez (2016) [11] | 626 | 52.1% with LVEF >50% | 73.4 ± 10.4 | 52 | 30 (endpoint) | P | ESC definitions | No | Ferritin | HF Hospitalization: 16.5% in 30 days | ↑ HF Hospitalization w/ferritin < 100 µg/L (sHR 1.72; 95% CI 1.13–2.60, p = 0.011); FID not related to the risk of readmission (HR 0.87; 95% CI 0.46–1.62, p = 0.652) |
| Nakano (2018) [9] | 578 | 39 (median) | 78 (median) | 61 | 365 (endpoint) | P | ESC definitions | No | Ferritin | HF Hospitalization: 19% in 1 year | ↑ Adverse events w/Ferritin < 100 μg/L (HR 1.50, 95% CI 1.02–2.21, p = 0.040) FID with neutral effect (HR 0.73, 95% CI 0.39–1.34, p = 0.31) |
| Ambrosy (2020) [30] | 4103 | 35% with LVEF >50% | 79.2 ± 7 | 51 | 365 (endpoint) | P | ESC definitions | No | TSAT | HF admission plus mortality: 16.7 per 100 person-years | ↑ HF Hospitalization w/TSAT < 20% for ferritin <100 ng/mL (aHR 1.40, 95% CI 1.16–1.70) and 100–300 ng/mL (aHR 1.24, 95% CI 1.01–1.52) |
| Fitzsimons (2021) [29] | 1563 | 38 * | 65.6 * | 72 | 730 (endpoint) | P | ESC definitions OR TSAT< 20% | No | TSAT | Death/HF hospitalization: 43% in 2 years | ↑ Adverse events w/TSAT < 20% (HR 1.19, CI 1.01–1.41, p = 0.03) ESC definitions not a predictor of hospitalization (HR 1.11, CI 95% 0.92–1.33, p = 0.28) |
| Ueda (2021) [36] | 615 | 46.7 ± 16.0 | 74.3 ± 12.0 | 57 | 976 (mean) | P | ESC definitions | No | Iron | HF hospitalization: 29% in 2.7 years | ↑ Adverse events w/Serum iron ≤ 64 µg/dL, irrespective of ferritin/Hg (HR 1.500; 95% CI 1.128–1.976; p = 0.0044) |
| Author (Year) | N | LVEF (%) | Mean Age (Years) | Male (%) | Follow-Up Period (Days) | Study Type | ID Criteria | Anemia Excluded (Yes/No) | Biomarker(s) | Outcomes | Association of Iron Metabolism Markers with Outcomes |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Jankowska (2011) [37] | 443 | 26 ± 7 | 54 ± 10 | 90 | -- | P | ESC definitions | No | Ferritin | Functional Capacity | ↓ VO2 and higher VE/VCO2 slope with serum ferritin (p < 0.05); ESC definitions associated with reduced peak VO2 (β = −0.14, p < 0.01 p < 0.05) and higher VE-VCO2 slope (β = 0.14, p < 0.01 p < 0.05) |
| Okonko (2011) [25] | 157 | 32 ± 9 | 71 ± 12 | 72 | 743 (median) | P | TSAT < 20% | No | sTfR TSAT Ferritin | Functional Capacity Quality of Life | ↑ NYHA class with sTfR ≥1.76 mg/L (ANOVA p < 0.0001) VO2 positively correlated with TSAT (r = 0.71, p < 0.0001) and ferritin (r = 0.48, p = 0.01) levels |
| Pozzo (2017) [22] | 138 | 28.1 * | 61.2 * | 48 | 1825 (mean) | P | ESC definitions | Yes (Hg < 13.0 g/dL in men or <12.0 g/dL in women) | Ferritin | Functional Capacity | ↓ performance in 6MWT (p = 0.03) and ↓ VO2 (p = 0.01) with ferritin < 100 μg/L |
| Martens (2018) [38] | 1197 | 6% with LVEF > 50% | 70 ± 12 | 71 | 1004 (mean) | P | ESC definitions | No | Ferritin TSAT | Functional Capacity | ↓ VO2 with ID, based on ferritin and TSAT (p < 0.001) |
| Tkaczyszyn (2018) [39] | 53 | LVEF ≤ 40 | 64 ± 10 | 100 | -- | P | ESC definitions | No | Ferritin | Functional Capacity | ↓ MIP with ferritin <100 μg/L compared to ferritin ≥100 μg/L (r = 0.42, p < 0.01) |
| Bekfani (2019) [40] | 190 | 58 ± 7 | 71 ± 9 | 67.5 | -- | P | ESC definitions | No | Ferritin TSAT | Functional Capacity Quality of Life | ↓ performance in 6MWT (p = 0.008) and CPX (p = 0.03) with ID, based on ferritin and TSAT ↓ QoL(p = 0.03) with Ferritin < 100 µg/L |
| Alcaide-Aldeano (2020) [23] | 447 | 62 ± 8 | 76 ± 9 | 41 | -- | P | ferritin < 100 ng/mL or TSAT < 20% | Yes (Hg less than < 8.5 g/dL) | sTfR | Functional Capacity Quality of Life | sTfR (overall mean value of 1.92) predictor of functional capacity (β = −63, p < 0.0001, R2 0.39); and QoL (β = 7.95, p < 0.0001, R2 0.14). TSAT < 20% (β = 3.32, p = 0.13, R2 0.10), ferritin (β = 0.64, p = 0.56, R2 0.10) and serum iron (β = −1.50, p = 0.47, R2 0.10) did not have a strong association to QoL |
| Author (Year) | N | LVEF (%) | Mean Age (Years) | Male (%) | Follow-Up Period (Days) | Study Type | ID Criteria | Anemia Excluded (Yes/No) | Biomarker(s) | Outcomes | Association of Iron Metabolism Markers with Outcomes |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Klip (2017) [41] | 6386 | N/A | 53.1 ± 12.0 | 50.3 | 3032 (median) | P | N/A | No | Ferritin | New onset HF | ferritin levels (median: 97 µg/L) predict new-onset HF in women only (p = 0.024). |
| Yan (2020) [24] | 221 | 55.8 ± 9.1 | 68.6 ± 11.8 | 57.5 | 239 (median) | R | Low (7.6 ± 1.6), medium (11.9 ± 1.8) and high (19.4 ± 3.8 serum iron (μmol/L) | Yes | Iron | MACCE | ↑ MACCE with serum iron <7.58 μmol/L or >19.37 μmol/L compared to medium serum iron (p < 0.0001) |
| Ceyhun (2021) [42] | 157 | 30.0 ± 8.1 | 63.9 ± 8.62 | 42 | N/A | R | N/A | No | Ferritin | AKI | ↑ AKI risk with BNP/ferritin, mean value 10.48 ± 2.14 (OR = 3.19; 95% CI 1.92–6.54; p = 0.001) & ferritin, mean value 86.78 ± 57.2 (OR = 0.72; 95% CI, 0.89–0.53; p = 0.028) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dhaliwal, S.; Kalogeropoulos, A.P. Markers of Iron Metabolism and Outcomes in Patients with Heart Failure: A Systematic Review. Int. J. Mol. Sci. 2023, 24, 5645. https://doi.org/10.3390/ijms24065645
Dhaliwal S, Kalogeropoulos AP. Markers of Iron Metabolism and Outcomes in Patients with Heart Failure: A Systematic Review. International Journal of Molecular Sciences. 2023; 24(6):5645. https://doi.org/10.3390/ijms24065645
Chicago/Turabian StyleDhaliwal, Simrat, and Andreas P. Kalogeropoulos. 2023. "Markers of Iron Metabolism and Outcomes in Patients with Heart Failure: A Systematic Review" International Journal of Molecular Sciences 24, no. 6: 5645. https://doi.org/10.3390/ijms24065645
APA StyleDhaliwal, S., & Kalogeropoulos, A. P. (2023). Markers of Iron Metabolism and Outcomes in Patients with Heart Failure: A Systematic Review. International Journal of Molecular Sciences, 24(6), 5645. https://doi.org/10.3390/ijms24065645







