Effects of GRP78 on Endoplasmic Reticulum Stress and Inflammatory Response in Macrophages of Large Yellow Croaker (Larimichthys crocea)
Abstract
:1. Introduction
2. Results
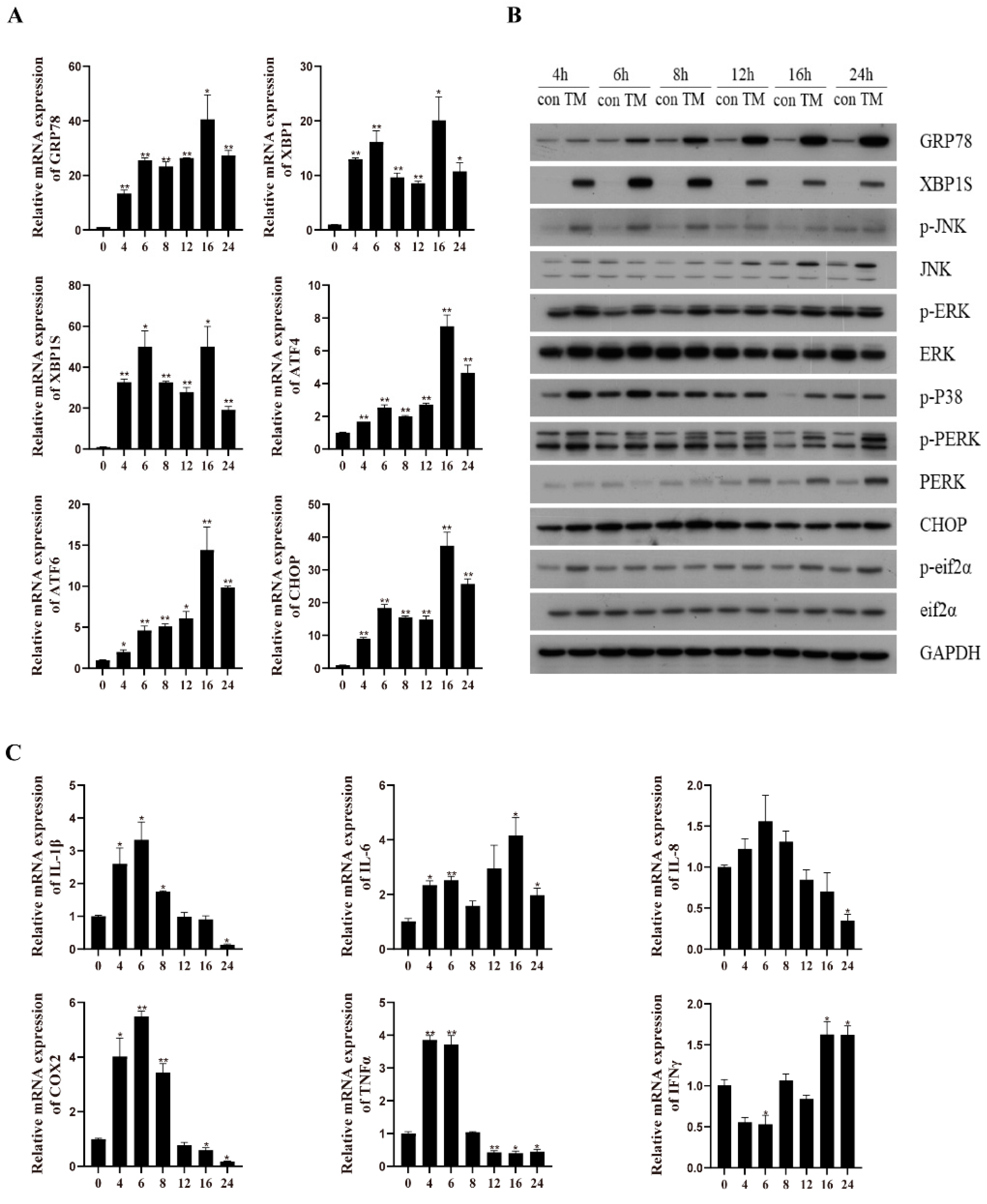
2.1. TM Treatment Induces ER Stress and an Inflammatory Response in the Macrophages of Large Yellow Croakers
2.2. BiP Inducer X (GRP78 Agonist) Treatment Induces ER Stress and an Inflammatory Response in Macrophages
2.3. HM03 (GRP78 Inhibitor) Treatment Induces ER Stress and an Inflammatory Response in Macrophages
2.4. Effect of GRP78 on TM-Induced ER Stress and Inflammation in Macrophages
2.5. PA Treatment Induces ER Stress and an Inflammatory Response in Macrophages
2.6. Effect of GRP78 on PA-Induced ER Stress and Inflammation in Macrophages
3. Discussion
4. Materials and Methods
4.1. Cell Culture and Treatment
4.2. RNA Extraction and RT-qPCR
4.3. Western Blot
4.4. Statistical Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kozutsumi, Y.; Segal, M.; Normington, K.; Gething, M.J.; Sambrook, J. The presence of malfolded proteins in the endoplasmic reticulum signals the induction of glucose-regulated proteins. Nature 1988, 332, 462–464. [Google Scholar] [CrossRef] [PubMed]
- Høyer-Hansen, M.; Jäättelä, M.J.C.D. Connecting endoplasmic reticulum stress to autophagy by unfolded protein response and calcium. Cell Death Differ. 2007, 14, 1576–1582. [Google Scholar] [CrossRef] [PubMed]
- Hendershot, M. The Unfolding Tale of the Unfolded Protein Response. Cell 2001, 107, 827–830. [Google Scholar] [CrossRef]
- Ron, D.; Walter, P. Signal integration in the endoplasmic reticulum unfolded protein response. Nat. Rev. Mol. Cell Biol. 2007, 8, 519–529. [Google Scholar] [CrossRef]
- Bertolotti, A.; Zhang, Y.; Hendershot, L.M.; Harding, H.P.; Ron, D. Dynamic interaction of BiP and ER stress transducers in the unfolded-protein response. Nat. Cell Biol. 2000, 2, 326–332. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.; Zhao, Y.; Zhou, K.; Freeze, H.H.; Xu, H. Insufficient ER-stress response causes selective mouse cerebellar granule cell degeneration resembling that seen in congenital disorders of glycosylation. Mol. Brain 2013, 6, 52. [Google Scholar] [CrossRef] [Green Version]
- Pfaffenbach, K.T.; Lee, A.S. The critical role of GRP78 in physiologic and pathologic stress. Curr. Opin. Cell Biol. 2011, 23, 150–156. [Google Scholar] [CrossRef] [Green Version]
- Urano, F. Coupling of Stress in the ER to Activation of JNK Protein Kinases by Transmembrane Protein Kinase IRE1. Science 2000, 287, 664–666. [Google Scholar] [CrossRef] [Green Version]
- Hu, P.; Han, Z.; Couvillon, A.D.; Kaufman, R.J.; Exton, J.H. Autocrine tumor necrosis factor alpha links endoplasmic reticulum stress to the membrane death receptor pathway through IRE1 alpha-mediated NF-kappa B activation and down-regulation of TRAF2 expression. Mol. Cell. Biol. 2006, 26, 3071–3084. [Google Scholar] [CrossRef] [Green Version]
- Deng, J.; Lu, P.D.; Zhang, Y.; Scheuner, D.; Kaufman, R.J.; Sonenberg, N.; Harding, H.P.; Ron, D. Translational Repression Mediates Activation of Nuclear Factor Kappa B by Phosphorylated Translation Initiation Factor 2. Mol. Cell. Biol. 2004, 24, 10161–10168. [Google Scholar] [CrossRef] [Green Version]
- Yamazaki, H.; Hiramatsu, N.; Hayakawa, K.; Tagawa, Y.; Okamura, M.; Ogata, R.; Huang, T.; Nakajima, S.; Yao, J.; Paton, A.W.; et al. Activation of the Akt-NF-kappaB pathway by subtilase cytotoxin through the ATF6 branch of the unfolded protein response. J. Immunol. (1950) 2009, 183, 1480. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shields, A.M.; Panayi, G.S.; Corrigall, V.M. Resolution-associated molecular patterns (RAMP): RAMParts defending immunological homeostasis? Clin. Exp. Immunol. 2011, 165, 292–300. [Google Scholar] [CrossRef] [PubMed]
- Qin, K.; Ma, S.; Li, H.; Wu, M.; Sun, Y.; Fu, M.; Guo, Z.; Zhu, H.; Gong, F.; Lei, P.; et al. GRP78 Impairs Production of Lipopolysaccharide-Induced Cytokines by Interaction with CD14. Front. Immunol. 2017, 8, 579. [Google Scholar] [CrossRef] [Green Version]
- Zhou, X.; Yang, M.; Lv, Y.; Li, H.; Wu, S.; Min, J.; Shen, G.; He, Y.; Lei, P. Adoptive transfer of GRP78-treated dendritic cells alleviates insulitis in NOD mice. J. Leukoc. Biol. 2021, 110, 1023–1031. [Google Scholar] [CrossRef] [PubMed]
- Luo, C.; Xiong, H.; Chen, L.; Liu, X.; Zou, S.; Guan, J.; Wang, K. GRP78 Promotes Hepatocellular Carcinoma proliferation by increasing FAT10 expression through the NF-κB pathway. Exp. Cell Res. 2018, 365, 1–11. [Google Scholar] [CrossRef]
- Misra, U.K.; Kaczowka, S.; Pizzo, S.V. Inhibition of NF-κB1 and NF-κB2 activation in prostate cancer cells treated with antibody against the carboxyl terminal domain of GRP78: Effect of p53 upregulation. Biochem. Biophys. Res. Commun. 2010, 392, 538–542. [Google Scholar] [CrossRef]
- Natsume, Y.; Ito, S.; Satsu, H.; Shimizu, M. Protective effect of quercetin on ER stress caused by calcium dynamics dysregulation in intestinal epithelial cells. Toxicology 2009, 258, 164–175. [Google Scholar] [CrossRef]
- Abdullahi, A.; Stanojcic, M.; Parousis, A.; Patsouris, D.; Jeschke, M.G. Modeling Acute ER Stress In Vivo and In Vitro. Shock 2017, 47, 506–513. [Google Scholar] [CrossRef] [Green Version]
- Fang, W.; Chen, Q.; Li, J.; Liu, Y.; Zhao, Z.; Shen, Y.; Mai, K.; Ai, Q. Endoplasmic Reticulum Stress Disturbs Lipid Homeostasis and Augments Inflammation in the Intestine and Isolated Intestinal Cells of Large Yellow Croaker (Larimichthys crocea). Front. Immunol. 2021, 12, 738143. [Google Scholar] [CrossRef]
- Chen, Q.; Fang, W.; Cui, K.; Chen, Q.; Xiang, X.; Zhang, J.; Zhang, Y.; Mai, K.; Ai, Q. Endoplasmic reticulum stress induces hepatic steatosis by transcriptional upregulating lipid droplet protein perilipin2. FASEB J. 2021, 35, e21900. [Google Scholar] [CrossRef]
- Guo, Q.; Li, H.; Liu, J.; Xu, L.; Yang, L.; Sun, Z.; Zhou, B. Tunicamycin aggravates endoplasmic reticulum stress and airway inflammation via PERK-ATF4-CHOP signaling in a murine model of neutrophilic asthma. J. Asthma 2017, 54, 125–133. [Google Scholar] [CrossRef] [PubMed]
- Ren, F.; Zhou, L.; Zhang, X.; Wen, T.; Shi, H.; Xie, B.; Li, Z.; Chen, D.; Wang, Z.; Duan, Z. Endoplasmic Reticulum Stress-Activated Glycogen Synthase Kinase 3 beta Aggravates Liver Inflammation and Hepatotoxicity in Mice with Acute Liver Failure. Inflammation 2015, 38, 1151–1165. [Google Scholar] [CrossRef] [PubMed]
- Mondal, A.K.; Das, S.K.; Varma, V.; Nolen, G.T.; McGehee, R.E.; Elbein, S.C.; Wei, J.Y.; Ranganathan, G. Effect of endoplasmic reticulum stress on inflammation and adiponectin regulation in human adipocytes. Metab. Syndr. Relat. Disord. 2012, 10, 297–306. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Korbecki, J.B.R.K. The effect of palmitic acid on inflammatory response in macrophages: An overview of molecular mechanisms. Inflamm. Res. Off. J. Eur. Histamine Res. Soc. 2019, 68, 915–932. [Google Scholar] [CrossRef] [Green Version]
- Bell, J.G.; Henderson, R.J.; Tocher, D.R.; Mcghee, F.; Dick, J.R.; Porter, A.; Smullen, R.P.; Sargent, J.R. Substituting fish oil with crude palm oil in the diet of Atlantic salmon (Salmo salar) affects muscle fatty acid composition and hepatic fatty acid metabolism. J. Nutr. 2002, 132, 222–230. [Google Scholar] [CrossRef] [Green Version]
- Bahurmiz, O.M.; Ng, W.K. Effects of dietary palm oil source on growth, tissue fatty acid composition and nutrient digestibility of red hybrid tilapia, Oreochromis sp., raised from stocking to marketable size. Aquaculture 2007, 262, 382–392. [Google Scholar] [CrossRef]
- Li, X.; Ji, R.; Cui, K.; Chen, Q.; Chen, Q.; Fang, W.; Mai, K.; Zhang, Y.; Xu, W.; Ai, Q. High percentage of dietary palm oil suppressed growth and antioxidant capacity and induced the inflammation by activation of TLR NF-κB signaling pathway in large yellow croaker (Larimichthys crocea). Fish Shellfish Immunol. 2019, 87, 600–608. [Google Scholar] [CrossRef]
- Fang, W.; Liu, Y.; Chen, Q.; Xu, D.; Liu, Q.; Cao, X.; Hao, T.; Zhang, L.; Mai, K.; Ai, Q. Palmitic acid induces intestinal lipid metabolism disorder, endoplasmic reticulum stress and inflammation by affecting phosphatidylethanolamine content in large yellow croaker Larimichthys crocea. Front. Immunol. 2022, 13, 984508. [Google Scholar] [CrossRef]
- Zhang, J.; Liu, Q.; Pang, Y.; Xu, X.; Cui, K.; Zhang, Y.; Mai, K.; Ai, Q. Molecular cloning and the involvement of IRE1α-XBP1s signaling pathway in palmitic acid induced—Inflammation in primary hepatocytes from large yellow croaker (Larimichthys crocea). Fish Shellfish Immunol. 2020, 98, 112–121. [Google Scholar] [CrossRef]
- Xu, D.; Cui, K.; Li, Q.; Zhu, S.; Zhang, J.; Gao, S.; Hao, T.; Mai, K.; Ai, Q. Docosahexaenoic Acid Alleviates Palmitic Acid-Induced Inflammation of Macrophages via TLR22-MAPK-PPARγ/Nrf2 Pathway in Large Yellow Croaker (Larimichthys crocea). Antioxidants 2022, 11, 682. [Google Scholar] [CrossRef]
- Hotamisligil, G.S. Inflammation and metabolic disorders. Nature 2006, 444, 860–867. [Google Scholar] [CrossRef]
- Gillon, A.D.; Latham, C.F.; Miller, E.A. Vesicle-mediated ER export of proteins and lipids. Biochim. Biophys. Acta 2012, 1821, 1040–1049. [Google Scholar] [CrossRef] [Green Version]
- Guzel, E.; Arlier, S.; Guzeloglu-Kayisli, O.; Tabak, M.S.; Ekiz, T.; Semerci, N.; Larsen, K.; Schatz, F.; Lockwood, C.J.; Kayisli, U.A. Endoplasmic Reticulum Stress and Homeostasis in Reproductive Physiology and Pathology. Int. J. Mol. Sci. 2017, 18, 792. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Puri, P.; Mirshahi, F.; Cheung, O.; Natarajan, R.; Maher, J.W.; Kellum, J.M.; Sanyal, A.J. Activation and Dysregulation of the Unfolded Protein Response in Nonalcoholic Fatty Liver Disease. Gastroenterology 2008, 134, 568–576. [Google Scholar] [CrossRef]
- Malhi, H.; Kaufman, R.J. Endoplasmic reticulum stress in liver disease. J. Hepatol. 2011, 54, 795–809. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wei, Y.; Wang, D.; Topczewski, F.; Pagliassotti, M.J. Saturated fatty acids induce endoplasmic reticulum stress and apoptosis independently of ceramide in liver cells. Am. J. Physiol.-Endocrinol. Metab. 2006, 291, E275–E281. [Google Scholar] [CrossRef] [PubMed]
- Borradaile, N.M.; Han, X.; Harp, J.D.; Gale, S.E.; Ory, D.S.; Schaffer, J.E. Disruption of endoplasmic reticulum structure and integrity in lipotoxic cell death. J. Lipid Res. 2006, 47, 2726–2737. [Google Scholar] [CrossRef] [Green Version]
- Yang, L.; Guan, G.; Lei, L.; Lv, Q.; Liu, S.; Zhan, X.; Jiang, Z.; Gu, X. Palmitic acid induces human osteoblast-like Saos-2 cell apoptosis via endoplasmic reticulum stress and autophagy. Cell Stress Chaperones 2018, 23, 1283–1294. [Google Scholar] [CrossRef]
- Ariyama, H.; Kono, N.; Matsuda, S.; Inoue, T.; Arai, H. Decrease in Membrane Phospholipid Unsaturation Induces Unfolded Protein Response*. J. Biol. Chem. 2010, 285, 22027–22035. [Google Scholar] [CrossRef] [Green Version]
- Fu, S.; Yang, L.; Li, P.; Hofmann, O.; Dicker, L.; Hide, W.; Lin, X.; Watkins, S.M.; Ivanov, A.R.; Hotamisligil, G.S. Aberrant lipid metabolism disrupts calcium homeostasis causing liver endoplasmic reticulum stress in obesity. Nature 2011, 473, 528–531. [Google Scholar] [CrossRef] [Green Version]
- Volmer, R.; Ron, D. Lipid-dependent regulation of the unfolded protein response. Curr. Opin. Cell Biol. 2015, 33, 67–73. [Google Scholar] [CrossRef] [Green Version]
- Kudo, T.; Kanemoto, S.; Hara, H.; Morimoto, N.; Morihara, T.; Kimura, R.; Tabira, T.; Imaizumi, K.; Takeda, M. A molecular chaperone inducer protects neurons from ER stress. Cell Death Differ. 2008, 15, 364–375. [Google Scholar] [CrossRef] [Green Version]
- Ha, T.K.; Hansen, A.H.; Kildegaard, H.F.; Lee, G.M. BiP Inducer X: An ER Stress Inhibitor for Enhancing Recombinant Antibody Production in CHO Cell Culture. Biotechnol. J. 2019, 14, e1900130. [Google Scholar] [CrossRef] [PubMed]
- Inokuchi, Y.; Nakajima, Y.; Shimazawa, M.; Kurita, T.; Kubo, M.; Saito, A.; Sajiki, H.; Kudo, T.; Aihara, M.; Imaizumi, K.; et al. Effect of an Inducer of BiP, a Molecular Chaperone, on Endoplasmic Reticulum (ER) Stress-Induced Retinal Cell Death. Investig. Opthalmology Vis. Sci. 2009, 50, 334. [Google Scholar] [CrossRef] [PubMed]
- Oida, Y.; Izuta, H.; Oyagi, A.; Shimazawa, M.; Kudo, T.; Imaizumi, K.; Hara, H. Induction of BiP, an ER-resident protein, prevents the neuronal death induced by transient forebrain ischemia in gerbil. Brain Res. 2008, 1208, 217–224. [Google Scholar] [CrossRef]
- Contreras, C.; González-García, I.; Martínez-Sánchez, N.; Seoane-Collazo, P.; Jacas, J.; Morgan, D.A.; Serra, D.; Gallego, R.; Gonzalez, F.; Casals, N.; et al. Central Ceramide-Induced Hypothalamic Lipotoxicity and ER Stress Regulate Energy Balance. Cell Rep. 2014, 9, 366–377. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Contreras, C.; Gonzalez-Garcia, I.; Seoane-Collazo, P.; Martinez-Sanchez, N.; Linares-Pose, L.; Rial-Pensado, E.; Ferno, J.; Tena-Sempere, M.; Casals, N.; Dieguez, C.; et al. Reduction of Hypothalamic Endoplasmic Reticulum Stress Activates Browning of White Fat and Ameliorates Obesity. Diabetes 2017, 66, 87–99. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Suzuki, T.; Lu, J.; Zahed, M.; Kita, K.; Suzuki, N. Reduction of GRP78 expression with siRNA activates unfolded protein response leading to apoptosis in HeLa cells. Arch. Biochem. Biophys. 2007, 468, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Ni, M.; Lee, B.; Barron, E.; Hinton, D.R.; Lee, A.S. The unfolded protein response regulator GRP78/BiP is required for endoplasmic reticulum integrity and stress-induced autophagy in mammalian cells. Cell Death Differ. 2008, 15, 1460–1471. [Google Scholar] [CrossRef]
- Cui, K.; Li, Q.; Xu, D.; Zhang, J.; Gao, S.; Xu, W.; Mai, K.; Ai, Q. Establishment and characterization of two head kidney macrophage cell lines from large yellow croaker (Larimichthys crocea). Dev. Comp. Immunol. 2020, 102, 103477. [Google Scholar] [CrossRef]
- Livak, K.J.; Schmittgen, T.D.L. Analysis of relative gene expression data using real-time quantitative PCR and the 2-ΔΔct method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Gan, L.; Liu, G.; Chen, Y.; Wu, T.; Feng, F.; Sun, C. Sirt1 decreased adipose inflammation by interacting with Akt2 and inhibiting mTOR/S6K1 pathway in mice. J. Lipid Res. 2016, 57, 1373–1381. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, X.; He, G.; Mai, K.; Xu, W.; Zhou, H. Differential regulation of taurine biosynthesis in rainbow trout and Japanese flounder. Sci. Rep. 2016, 6, 21231. [Google Scholar] [CrossRef] [PubMed] [Green Version]











Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sun, J.; Mai, K.; Ai, Q. Effects of GRP78 on Endoplasmic Reticulum Stress and Inflammatory Response in Macrophages of Large Yellow Croaker (Larimichthys crocea). Int. J. Mol. Sci. 2023, 24, 5855. https://doi.org/10.3390/ijms24065855
Sun J, Mai K, Ai Q. Effects of GRP78 on Endoplasmic Reticulum Stress and Inflammatory Response in Macrophages of Large Yellow Croaker (Larimichthys crocea). International Journal of Molecular Sciences. 2023; 24(6):5855. https://doi.org/10.3390/ijms24065855
Chicago/Turabian StyleSun, Jie, Kangsen Mai, and Qinghui Ai. 2023. "Effects of GRP78 on Endoplasmic Reticulum Stress and Inflammatory Response in Macrophages of Large Yellow Croaker (Larimichthys crocea)" International Journal of Molecular Sciences 24, no. 6: 5855. https://doi.org/10.3390/ijms24065855



