The Interplay between Integrins and Immune Cells as a Regulator in Cancer Immunology
Abstract
1. Introduction
2. Potential Influence of Integrins on Immune Cells
2.1. B Cells
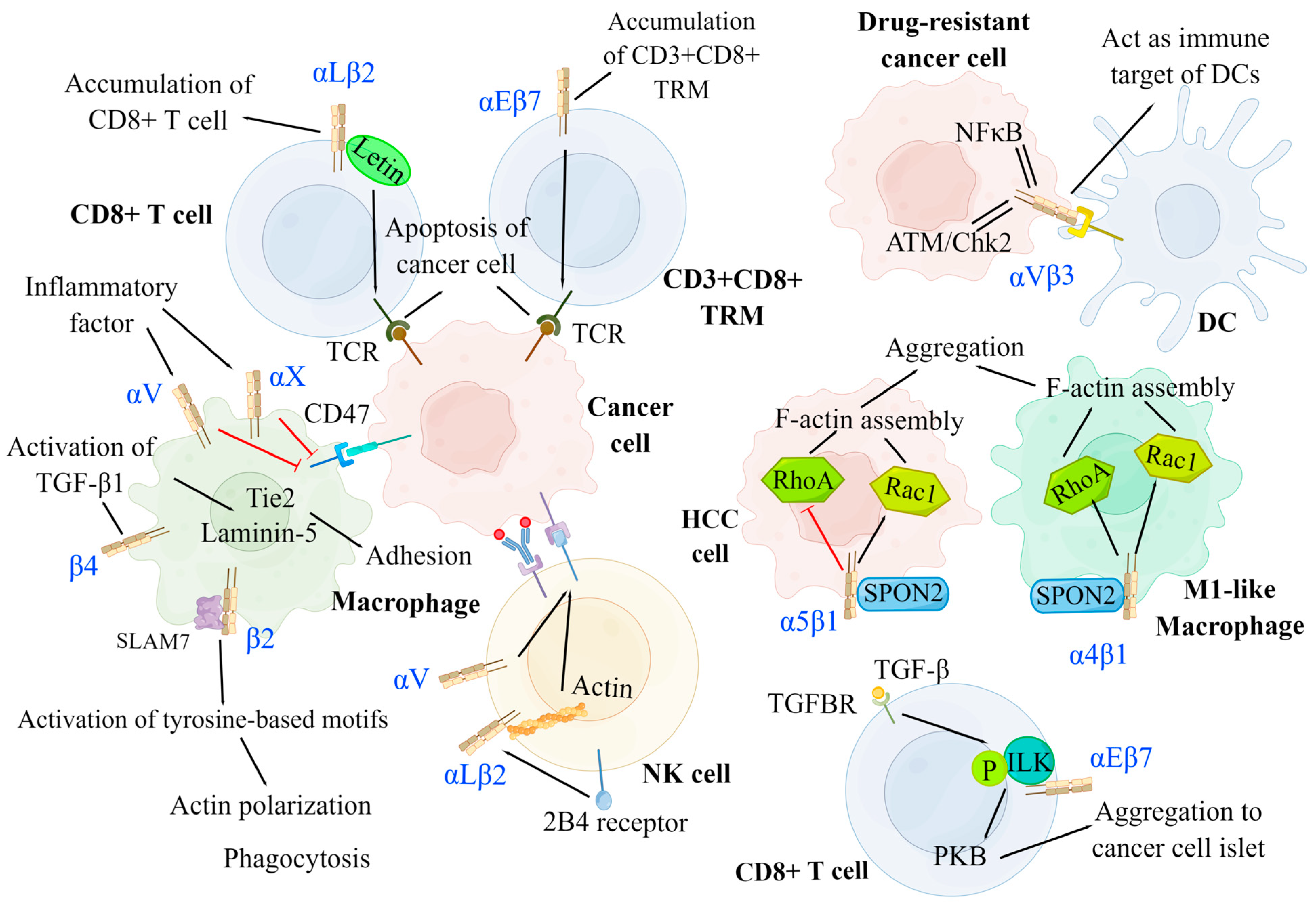
2.2. T Cells
2.3. Dendritic Cells
2.4. Natural Killer Cells
2.5. Neutrophils
2.6. Macrophages
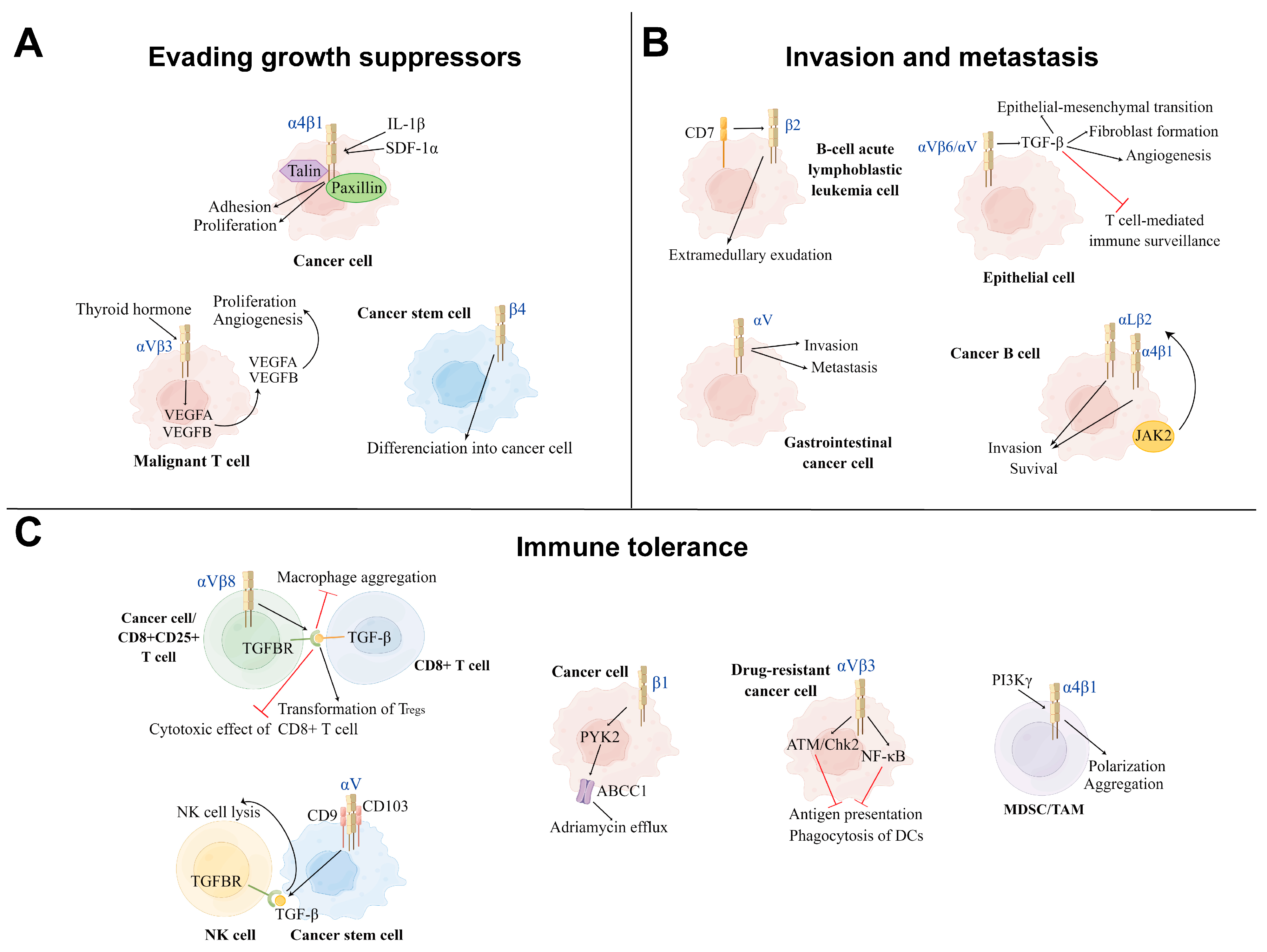
3. The Roles of Integrins in Cancer
3.1. Positive Modulation of Integrins in Cancer Development
3.1.1. Evading Growth Suppressors
3.1.2. Invasion and Metastasis
3.1.3. Immune Tolerance
3.2. Negative Modulation of Integrins on Cancer Development
3.3. Integrin-Targeted Immunotherapy
3.3.1. Chimeric Antigen Receptor T-Cell Immunotherapy
3.3.2. Oncolytic Virotherapy
3.3.3. Immune Checkpoint Inhibitor Therapy
3.3.4. Other Strategies
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Takada, Y.; Ye, X.; Simon, S. The integrins. Genome Biol. 2007, 8, 215. [Google Scholar] [CrossRef] [PubMed]
- Seetharaman, S.; Etienne-Manneville, S. Integrin diversity brings specificity in mechanotransduction. Biol. Cell 2018, 110, 49–64. [Google Scholar] [CrossRef] [PubMed]
- Lietha, D.; Izard, T. Roles of Membrane Domains in Integrin-Mediated Cell Adhesion. Int. J. Mol. Sci. 2020, 21, 5531. [Google Scholar] [CrossRef] [PubMed]
- Barczyk, M.; Carracedo, S.; Gullberg, D. Integrins. Cell Tissue Res. 2010, 339, 269–280. [Google Scholar] [CrossRef]
- Overstreet, M.G.; Gaylo, A.; Angermann, B.R.; Hughson, A.; Hyun, Y.M.; Lambert, K.; Acharya, M.; Billroth-Maclurg, A.C.; Rosenberg, A.F.; Topham, D.J.; et al. Inflammation-induced interstitial migration of effector CD4⁺ T cells is dependent on integrin αV. Nat. Immunol. 2013, 14, 949–958. [Google Scholar] [CrossRef]
- Pan, L.; Zhao, Y.; Yuan, Z.; Qin, G. Research advances on structure and biological functions of integrins. SpringerPlus 2016, 5, 1094. [Google Scholar] [CrossRef]
- Saez de Guinoa, J.; Barrio, L.; Carrasco, Y.R. Vinculin arrests motile B cells by stabilizing integrin clustering at the immune synapse. J. Immunol. 2013, 191, 2742–2751. [Google Scholar] [CrossRef]
- Campbell, I.D.; Humphries, M.J. Integrin structure, activation, and interactions. Cold Spring Harb. Perspect. Biol. 2011, 3, a004994. [Google Scholar] [CrossRef]
- Kanayama, M.; Morimoto, J.; Matsui, Y.; Ikesue, M.; Danzaki, K.; Kurotaki, D.; Ito, K.; Yoshida, T.; Uede, T. α9β1 integrin-mediated signaling serves as an intrinsic regulator of pathogenic Th17 cell generation. J. Immunol. 2011, 187, 5851–5864. [Google Scholar] [CrossRef]
- Yu, Y.; Ou-Yang, W.; Zhang, H.; Jiang, T.; Cho, W.C.; Zhu, H.; Xiao, Z.; Li, S. High-mobility Group Box 1 Facilitates CD4 T Cell Self-aggregation Via Integrin and STAT3 Activation Before Homing. Inflamm. Bowel Dis. 2020, 26, 1188–1198. [Google Scholar] [CrossRef]
- Taleb, S. Inflammation in atherosclerosis. Arch. Cardiovasc. Dis. 2016, 109, 708–715. [Google Scholar] [CrossRef] [PubMed]
- Glass, C.K.; Saijo, K.; Winner, B.; Marchetto, M.C.; Gage, F.H. Mechanisms underlying inflammation in neurodegeneration. Cell 2010, 140, 918–934. [Google Scholar] [CrossRef] [PubMed]
- Lee, I.T.; Yang, C.M. Inflammatory signalings involved in airway and pulmonary diseases. Mediat. Inflamm. 2013, 2013, 791231. [Google Scholar] [CrossRef] [PubMed]
- Cayrol, F.; Díaz Flaqué, M.C.; Fernando, T.; Yang, S.N.; Sterle, H.A.; Bolontrade, M.; Amorós, M.; Isse, B.; Farías, R.N.; Ahn, H.; et al. Integrin αvβ3 acting as membrane receptor for thyroid hormones mediates angiogenesis in malignant T cells. Blood 2015, 125, 841–851. [Google Scholar] [CrossRef]
- Jinushi, M.; Chiba, S.; Baghdadi, M.; Kinoshita, I.; Dosaka-Akita, H.; Ito, K.; Yoshiyama, H.; Yagita, H.; Uede, T.; Takaoka, A. ATM-mediated DNA damage signals mediate immune escape through integrin-αvβ3-dependent mechanisms. Cancer Res. 2012, 72, 56–65. [Google Scholar] [CrossRef]
- Kondoh, T.; Kuribayashi, K.; Tanaka, M.; Kobayashi, D.; Yanagihara, N.; Watanabe, N. CD7 promotes extramedullary involvement of the B-cell acute lymphoblastic leukemia line Tanoue by enhancing integrin β2-dependent cell adhesiveness. Int. J. Oncol. 2014, 45, 1073–1081. [Google Scholar] [CrossRef]
- Hoffmann, S.C.; Cohnen, A.; Ludwig, T.; Watzl, C. 2B4 engagement mediates rapid LFA-1 and actin-dependent NK cell adhesion to tumor cells as measured by single cell force spectroscopy. J. Immunol. 2011, 186, 2757–2764. [Google Scholar] [CrossRef] [PubMed]
- Takeichi, T.; Mocevicius, P.; Deduchovas, O.; Salnikova, O.; Castro-Santa, E.; Büchler, M.W.; Schmidt, J.; Ryschich, E. αL β2 integrin is indispensable for CD8+ T-cell recruitment in experimental pancreatic and hepatocellular cancer. Int. J. Cancer 2012, 130, 2067–2076. [Google Scholar] [CrossRef]
- Han, Z.; Ma, Y.; Cao, G.; Ma, Z.; Chen, R.; Cvijic, M.E.; Cheng, D. Integrin αVβ1 regulates procollagen I production through a non-canonical transforming growth factor β signaling pathway in human hepatic stellate cells. Biochem. J. 2021, 478, 1689–1703. [Google Scholar] [CrossRef]
- Hendesi, H.; Barbe, M.F.; Safadi, F.F.; Monroy, M.A.; Popoff, S.N. Integrin mediated adhesion of osteoblasts to connective tissue growth factor (CTGF/CCN2) induces cytoskeleton reorganization and cell differentiation. PLoS ONE 2015, 10, e0115325. [Google Scholar] [CrossRef]
- Pang, X.; He, X.; Qiu, Z.; Zhang, H.; Xie, R.; Liu, Z.; Gu, Y.; Zhao, N.; Xiang, Q.; Cui, Y. Targeting integrin pathways: Mechanisms and advances in therapy. Signal Transduct. Target. Ther. 2023, 8, 1. [Google Scholar] [CrossRef] [PubMed]
- Reed, N.I.; Jo, H.; Chen, C.; Tsujino, K.; Arnold, T.D.; DeGrado, W.F.; Sheppard, D. The αvβ1 integrin plays a critical in vivo role in tissue fibrosis. Sci. Transl. Med. 2015, 7, 288ra279. [Google Scholar] [CrossRef] [PubMed]
- Yamashiro, Y.; Thang, B.Q.; Ramirez, K.; Shin, S.J.; Kohata, T.; Ohata, S.; Nguyen, T.A.V.; Ohtsuki, S.; Nagayama, K.; Yanagisawa, H. Matrix mechanotransduction mediated by thrombospondin-1/integrin/YAP in the vascular remodeling. Proc. Natl. Acad. Sci. USA 2020, 117, 9896–9905. [Google Scholar] [CrossRef]
- Bishop, G.G.; McPherson, J.A.; Sanders, J.M.; Hesselbacher, S.E.; Feldman, M.J.; McNamara, C.A.; Gimple, L.W.; Powers, E.R.; Mousa, S.A.; Sarembock, I.J. Selective α(v)β(3)-receptor blockade reduces macrophage infiltration and restenosis after balloon angioplasty in the atherosclerotic rabbit. Circulation 2001, 103, 1906–1911. [Google Scholar] [CrossRef]
- Guermonprez, P.; Valladeau, J.; Zitvogel, L.; Théry, C.; Amigorena, S. Antigen presentation and T cell stimulation by dendritic cells. Annu. Rev. Immunol. 2002, 20, 621–667. [Google Scholar] [CrossRef] [PubMed]
- Kokubo, T.; Uchida, H.; Choi, E.T. Integrin α(v)β(3) as a target in the prevention of neointimal hyperplasia. J. Vasc. Surg. 2007, 45 (Suppl. A), A33–A38. [Google Scholar] [CrossRef]
- Asano, Y.; Ihn, H.; Jinnin, M.; Mimura, Y.; Tamaki, K. Involvement of αvβ5 integrin in the establishment of autocrine TGF-β signaling in dermal fibroblasts derived from localized scleroderma. J. Investig. Dermatol. 2006, 126, 1761–1769. [Google Scholar] [CrossRef]
- Kumawat, A.K.; Yu, C.; Mann, E.A.; Schridde, A.; Finnemann, S.C.; Mowat, A.M. Expression and characterization of αvβ5 integrin on intestinal macrophages. Eur. J. Immunol. 2018, 48, 1181–1187. [Google Scholar] [CrossRef]
- Oishi, Y.; Hashimoto, K.; Abe, A.; Kuroda, M.; Fujii, A.; Miyamoto, Y. Vitronectin regulates the axon specification of mouse cerebellar granule cell precursors via αvβ5 integrin in the differentiation stage. Neurosci. Lett. 2021, 746, 135648. [Google Scholar] [CrossRef]
- Porte, J.; Jenkins, G.; Tatler, A.L. Myofibroblast TGF-β Activation Measurement In Vitro. Methods Mol. Biol. 2021, 2299, 99–108. [Google Scholar] [CrossRef]
- Schiesser, J.V.; Loudovaris, T.; Thomas, H.E.; Elefanty, A.G.; Stanley, E.G. Integrin αvβ5 heterodimer is a specific marker of human pancreatic β cells. Sci. Rep. 2021, 11, 8315. [Google Scholar] [CrossRef] [PubMed]
- Tatler, A.L.; John, A.E.; Jolly, L.; Habgood, A.; Porte, J.; Brightling, C.; Knox, A.J.; Pang, L.; Sheppard, D.; Huang, X.; et al. Integrin αvβ5-mediated TGF-β activation by airway smooth muscle cells in asthma. J. Immunol. 2011, 187, 6094–6107. [Google Scholar] [CrossRef]
- Ansar, M.; Jan, A.; Santos-Cortez, R.L.; Wang, X.; Suliman, M.; Acharya, A.; Habib, R.; Abbe, I.; Ali, G.; Lee, K.; et al. Expansion of the spectrum of ITGB6-related disorders to adolescent alopecia, dentogingival abnormalities and intellectual disability. Eur. J. Hum. Genet. EJHG 2016, 24, 1223–1227. [Google Scholar] [CrossRef] [PubMed]
- Koivisto, L.; Bi, J.; Häkkinen, L.; Larjava, H. Integrin αvβ6: Structure, function and role in health and disease. Int. J. Biochem. Cell Biol. 2018, 99, 186–196. [Google Scholar] [CrossRef] [PubMed]
- Madala, S.K.; Korfhagen, T.R.; Schmidt, S.; Davidson, C.; Edukulla, R.; Ikegami, M.; Violette, S.M.; Weinreb, P.H.; Sheppard, D.; Hardie, W.D. Inhibition of the αvβ6 integrin leads to limited alteration of TGF-α-induced pulmonary fibrosis. Am. J. Physiol. Lung Cell. Mol. Physiol. 2014, 306, L726–L735. [Google Scholar] [CrossRef]
- White, J.B.; Hu, L.Y.; Boucher, D.L.; Sutcliffe, J.L. ImmunoPET Imaging of α(v)β(6) Expression Using an Engineered Anti-α(v)β(6) Cys-diabody Site-Specifically Radiolabeled with Cu-64: Considerations for Optimal Imaging with Antibody Fragments. Mol. Imaging Biol. 2018, 20, 103–113. [Google Scholar] [CrossRef]
- McCarty, J.H. αvβ8 integrin adhesion and signaling pathways in development, physiology and disease. J. Cell Sci. 2020, 133, jcs239434. [Google Scholar] [CrossRef]
- Zhou, M.; Niu, J.; Wang, J.; Gao, H.; Shahbaz, M.; Niu, Z.; Li, Z.; Zou, X.; Liang, B. Integrin αvβ8 serves as a Novel Marker of Poor Prognosis in Colon Carcinoma and Regulates Cell Invasiveness through the Activation of TGF-β1. J. Cancer 2020, 11, 3803–3815. [Google Scholar] [CrossRef]
- Hou, J.; Yan, D.; Liu, Y.; Huang, P.; Cui, H. The Roles of Integrin α5β1 in Human Cancer. OncoTargets Ther. 2020, 13, 13329–13344. [Google Scholar] [CrossRef]
- López-Luppo, M.; Catita, J.; Ramos, D.; Navarro, M.; Carretero, A.; Mendes-Jorge, L.; Muñoz-Cánoves, P.; Rodriguez-Baeza, A.; Nacher, V.; Ruberte, J. Cellular Senescence Is Associated With Human Retinal Microaneurysm Formation During Aging. Investig. Ophthalmol. Vis. Sci. 2017, 58, 2832–2842. [Google Scholar] [CrossRef]
- Lv, X.; Li, Z.; Guan, J.; Hu, S.; Zhang, J.; Lan, Y.; Zhao, K.; Lu, H.; Song, D.; He, H.; et al. Porcine Hemagglutinating Encephalomyelitis Virus Activation of the Integrin α5β1-FAK-Cofilin Pathway Causes Cytoskeletal Rearrangement To Promote Its Invasion of N2a Cells. J. Virol. 2019, 93, e01736-18. [Google Scholar] [CrossRef] [PubMed]
- Oh, S.H.; Kim, J.W.; Kim, Y.; Lee, M.N.; Kook, M.S.; Choi, E.Y.; Im, S.Y.; Koh, J.T. The extracellular matrix protein Edil3 stimulates osteoblast differentiation through the integrin α5β1/ERK/Runx2 pathway. PLoS ONE 2017, 12, e0188749. [Google Scholar] [CrossRef] [PubMed]
- Renner, G.; Noulet, F.; Mercier, M.C.; Choulier, L.; Etienne-Selloum, N.; Gies, J.P.; Lehmann, M.; Lelong-Rebel, I.; Martin, S.; Dontenwill, M. Expression/activation of α5β1 integrin is linked to the β-catenin signaling pathway to drive migration in glioma cells. Oncotarget 2016, 7, 62194–62207. [Google Scholar] [CrossRef]
- Zargham, R. Tensegrin in context: Dual role of α8 integrin in the migration of different cell types. Cell Adhes. Migr. 2010, 4, 485–490. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Li, X.; Shi, X.; Zhu, M.; Wang, J.; Huang, S.; Huang, X.; Wang, H.; Li, L.; Deng, H.; et al. Platelet integrin αIIbβ3: Signal transduction, regulation, and its therapeutic targeting. J. Hematol. Oncol. 2019, 12, 26. [Google Scholar] [CrossRef]
- Guenther, C. β2-Integrins—Regulatory and Executive Bridges in the Signaling Network Controlling Leukocyte Trafficking and Migration. Front. Immunol. 2022, 13, 809590. [Google Scholar] [CrossRef] [PubMed]
- Schenkel, A.R.; Mamdouh, Z.; Muller, W.A. Locomotion of monocytes on endothelium is a critical step during extravasation. Nat. Immunol. 2004, 5, 393–400. [Google Scholar] [CrossRef]
- Yuki, K.; Hou, L. Role of β2 Integrins in Neutrophils and Sepsis. Infect. Immun. 2020, 88, e00031-20. [Google Scholar] [CrossRef]
- Gaither, T.A.; Vargas, I.; Inada, S.; Frank, M.M. The complement fragment C3d facilitates phagocytosis by monocytes. Immunology 1987, 62, 405–411. [Google Scholar]
- Torres-Gomez, A.; Cabanas, C.; Lafuente, E.M. Phagocytic Integrins: Activation and Signaling. Front. Immunol. 2020, 11, 738. [Google Scholar] [CrossRef]
- Le Cabec, V.; Cols, C.; Maridonneau-Parini, I. Nonopsonic phagocytosis of zymosan and Mycobacterium kansasii by CR3 (CD11b/CD18) involves distinct molecular determinants and is or is not coupled with NADPH oxidase activation. Infect. Immun. 2000, 68, 4736–4745. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Bajic, G.; Andersen, G.R.; Christiansen, S.H.; Vorup-Jensen, T. The cationic peptide LL-37 binds Mac-1 (CD11b/CD18) with a low dissociation rate and promotes phagocytosis. Biochim. Biophys. Acta 2016, 1864, 471–478. [Google Scholar] [CrossRef] [PubMed]
- Guenther, C.; Faisal, I.; Fusciello, M.; Sokolova, M.; Harjunpää, H.; Ilander, M.; Tallberg, R.; Vartiainen, M.K.; Alon, R.; Gonzalez-Granado, J.M.; et al. β2-Integrin Adhesion Regulates Dendritic Cell Epigenetic and Transcriptional Landscapes to Restrict Dendritic Cell Maturation and Tumor Rejection. Cancer Immunol. Res. 2021, 9, 1354–1369. [Google Scholar] [CrossRef] [PubMed]
- Jawhara, S.; Pluskota, E.; Cao, W.; Plow, E.F.; Soloviev, D.A. Distinct Effects of Integrins αXβ2 and αMβ2 on Leukocyte Subpopulations during Inflammation and Antimicrobial Responses. Infect. Immun. 2017, 85, e00644-16. [Google Scholar] [CrossRef] [PubMed]
- Schack, L.; Stapulionis, R.; Christensen, B.; Kofod-Olsen, E.; Skov Sorensen, U.B.; Vorup-Jensen, T.; Sorensen, E.S.; Hollsberg, P. Osteopontin enhances phagocytosis through a novel osteopontin receptor, the αXβ2 integrin. J. Immunol. 2009, 182, 6943–6950. [Google Scholar] [CrossRef]
- Miyazaki, Y.; Vieira-de-Abreu, A.; Harris, E.S.; Shah, A.M.; Weyrich, A.S.; Castro-Faria-Neto, H.C.; Zimmerman, G.A. Integrin αDβ2 (CD11d/CD18) is expressed by human circulating and tissue myeloid leukocytes and mediates inflammatory signaling. PLoS ONE 2014, 9, e112770. [Google Scholar] [CrossRef]
- Fukui, T.; Fukaya, T.; Uto, T.; Takagi, H.; Nasu, J.; Miyanaga, N.; Nishikawa, Y.; Koseki, H.; Choijookhuu, N.; Hishikawa, Y.; et al. Pivotal role of CD103 in the development of psoriasiform dermatitis. Sci. Rep. 2020, 10, 8371. [Google Scholar] [CrossRef]
- Schreiber, T.D.; Steinl, C.; Essl, M.; Abele, H.; Geiger, K.; Müller, C.A.; Aicher, W.K.; Klein, G. The integrin α9β1 on hematopoietic stem and progenitor cells: Involvement in cell adhesion, proliferation and differentiation. Haematologica 2009, 94, 1493–1501. [Google Scholar] [CrossRef]
- Xu, S.; Zhang, T.; Cao, Z.; Zhong, W.; Zhang, C.; Li, H.; Song, J. Integrin-α9β1 as a Novel Therapeutic Target for Refractory Diseases: Recent Progress and Insights. Front. Immunol. 2021, 12, 638400. [Google Scholar] [CrossRef]
- Arthos, J.; Cicala, C.; Nawaz, F.; Byrareddy, S.N.; Villinger, F.; Santangelo, P.J.; Ansari, A.A.; Fauci, A.S. The Role of Integrin α(4)β(7) in HIV Pathogenesis and Treatment. Curr. HIV/AIDS Rep. 2018, 15, 127–135. [Google Scholar] [CrossRef]
- Li, H.; Huang, S.Y.; Shi, F.H.; Gu, Z.C.; Zhang, S.G.; Wei, J.F. α(4)β(7) integrin inhibitors: A patent review. Expert Opin. Ther. Pat. 2018, 28, 903–917. [Google Scholar] [CrossRef] [PubMed]
- Dennis, J.; Meehan, D.T.; Delimont, D.; Zallocchi, M.; Perry, G.A.; O’Brien, S.; Tu, H.; Pihlajaniemi, T.; Cosgrove, D. Collagen XIII induced in vascular endothelium mediates α1β1 integrin-dependent transmigration of monocytes in renal fibrosis. Am. J. Pathol. 2010, 177, 2527–2540. [Google Scholar] [CrossRef] [PubMed]
- Gardner, H. Integrin α1β1. Adv. Exp. Med. Biol. 2014, 819, 21–39. [Google Scholar] [CrossRef]
- Hamaia, S.W.; Pugh, N.; Raynal, N.; Némoz, B.; Stone, R.; Gullberg, D.; Bihan, D.; Farndale, R.W. Mapping of potent and specific binding motifs, GLOGEN and GVOGEA, for integrin α1β1 using collagen toolkits II and III. J. Biol. Chem. 2012, 287, 26019–26028. [Google Scholar] [CrossRef] [PubMed]
- Krieglstein, C.F.; Cerwinka, W.H.; Sprague, A.G.; Laroux, F.S.; Grisham, M.B.; Koteliansky, V.E.; Senninger, N.; Granger, D.N.; de Fougerolles, A.R. Collagen-binding integrin α1β1 regulates intestinal inflammation in experimental colitis. J. Clin. Investig. 2002, 110, 1773–1782. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, K.; Okuno, T.; Yamamoto, M.; Pasterkamp, R.J.; Takegahara, N.; Takamatsu, H.; Kitao, T.; Takagi, J.; Rennert, P.D.; Kolodkin, A.L.; et al. Semaphorin 7A initiates T-cell-mediated inflammatory responses through α1β1 integrin. Nature 2007, 446, 680–684. [Google Scholar] [CrossRef]
- El Azreq, M.A.; Arseneault, C.; Boisvert, M.; Pagé, N.; Allaeys, I.; Poubelle, P.E.; Tessier, P.A.; Aoudjit, F. Cooperation between IL-7 Receptor and Integrin α2β1 (CD49b) Drives Th17-Mediated Bone Loss. J. Immunol. 2015, 195, 4198–4209. [Google Scholar] [CrossRef] [PubMed]
- Grenache, D.G.; Zhang, Z.; Wells, L.E.; Santoro, S.A.; Davidson, J.M.; Zutter, M.M. Wound healing in the α2β1 integrin-deficient mouse: Altered keratinocyte biology and dysregulated matrix metalloproteinase expression. J. Investig. Dermatol. 2007, 127, 455–466. [Google Scholar] [CrossRef]
- Madamanchi, A.; Santoro, S.A.; Zutter, M.M. α2β1 Integrin. Adv. Exp. Med. Biol. 2014, 819, 41–60. [Google Scholar] [CrossRef]
- Zweers, M.C.; Davidson, J.M.; Pozzi, A.; Hallinger, R.; Janz, K.; Quondamatteo, F.; Leutgeb, B.; Krieg, T.; Eckes, B. Integrin α2β1 is required for regulation of murine wound angiogenesis but is dispensable for reepithelialization. J. Investig. Dermatol. 2007, 127, 467–478. [Google Scholar] [CrossRef]
- Camper, L.; Holmvall, K.; Wängnerud, C.; Aszódi, A.; Lundgren-Akerlund, E. Distribution of the collagen-binding integrin α10β1 during mouse development. Cell Tissue Res. 2001, 306, 107–116. [Google Scholar] [CrossRef] [PubMed]
- Lundgren-Åkerlund, E.; Aszòdi, A. Integrin α10β1: A collagen receptor critical in skeletal development. Adv. Exp. Med. Biol. 2014, 819, 61–71. [Google Scholar] [CrossRef] [PubMed]
- Barczyk, M.M.; Olsen, L.H.; da Franca, P.; Loos, B.G.; Mustafa, K.; Gullberg, D.; Bolstad, A.I. A role for α11β1 integrin in the human periodontal ligament. J. Dent. Res. 2009, 88, 621–626. [Google Scholar] [CrossRef] [PubMed]
- Erusappan, P.; Alam, J.; Lu, N.; Zeltz, C.; Gullberg, D. Integrin α11 cytoplasmic tail is required for FAK activation to initiate 3D cell invasion and ERK-mediated cell proliferation. Sci. Rep. 2019, 9, 15283. [Google Scholar] [CrossRef]
- Ramovs, V.; Te Molder, L.; Sonnenberg, A. The opposing roles of laminin-binding integrins in cancer. Matrix Biol. J. Int. Soc. Matrix Biol. 2017, 57–58, 213–243. [Google Scholar] [CrossRef]
- Delwel, G.O.; de Melker, A.A.; Hogervorst, F.; Jaspars, L.H.; Fles, D.L.; Kuikman, I.; Lindblom, A.; Paulsson, M.; Timpl, R.; Sonnenberg, A. Distinct and overlapping ligand specificities of the α 3A β 1 and α 6A β 1 integrins: Recognition of laminin isoforms. Mol. Biol. Cell 1994, 5, 203–215. [Google Scholar] [CrossRef]
- Fujiwara, H.; Kataoka, N.; Honda, T.; Ueda, M.; Yamada, S.; Nakamura, K.; Suginami, H.; Mori, T.; Maeda, M. Physiological roles of integrin α 6 β 1 in ovarian functions. Horm. Res. 1998, 50 (Suppl. 2), 25–29. [Google Scholar] [CrossRef]
- Georges-Labouesse, E.; Mark, M.; Messaddeq, N.; Gansmüller, A. Essential role of α 6 integrins in cortical and retinal lamination. Curr. Biol. CB 1998, 8, 983–986. [Google Scholar] [CrossRef]
- Reynolds, L.E.; D’Amico, G.; Lechertier, T.; Papachristodoulou, A.; Muñoz-Félix, J.M.; De Arcangelis, A.; Baker, M.; Serrels, B.; Hodivala-Dilke, K.M. Dual role of pericyte α6β1-integrin in tumour blood vessels. J. Cell Sci. 2017, 130, 1583–1595. [Google Scholar] [CrossRef]
- Soung, Y.H.; Clifford, J.L.; Chung, J. Crosstalk between integrin and receptor tyrosine kinase signaling in breast carcinoma progression. BMB Rep. 2010, 43, 311–318. [Google Scholar] [CrossRef]
- Wang, L.; Dong, Z.; Zhang, Y.; Miao, J. The roles of integrin β4 in vascular endothelial cells. J. Cell. Physiol. 2012, 227, 474–478. [Google Scholar] [CrossRef] [PubMed]
- Flintoff-Dye, N.L.; Welser, J.; Rooney, J.; Scowen, P.; Tamowski, S.; Hatton, W.; Burkin, D.J. Role for the α7β1 integrin in vascular development and integrity. Dev. Dyn. Off. Publ. Am. Assoc. Anat. 2005, 234, 11–21. [Google Scholar] [CrossRef] [PubMed]
- Hayashi, Y.K.; Chou, F.L.; Engvall, E.; Ogawa, M.; Matsuda, C.; Hirabayashi, S.; Yokochi, K.; Ziober, B.L.; Kramer, R.H.; Kaufman, S.J.; et al. Mutations in the integrin α7 gene cause congenital myopathy. Nat. Genet. 1998, 19, 94–97. [Google Scholar] [CrossRef]
- Belkin, A.M.; Stepp, M.A. Integrins as receptors for laminins. Microsc. Res. Technol. 2000, 51, 280–301. [Google Scholar] [CrossRef]
- Munksgaard Thorén, M.; Chmielarska Masoumi, K.; Krona, C.; Huang, X.; Kundu, S.; Schmidt, L.; Forsberg-Nilsson, K.; Floyd Keep, M.; Englund, E.; Nelander, S.; et al. Integrin α10, a Novel Therapeutic Target in Glioblastoma, Regulates Cell Migration, Proliferation, and Survival. Cancers 2019, 11, 587. [Google Scholar] [CrossRef]
- Sasaki, T.; Timpl, R. Domain IVa of laminin α5 chain is cell-adhesive and binds β1 and αVβ3 integrins through Arg-Gly-Asp. FEBS Lett. 2001, 509, 181–185. [Google Scholar] [CrossRef]
- Nagy-Baló, Z.; Kiss, R.; Menge, A.; Bödör, C.; Bajtay, Z.; Erdei, A. Activated Human Memory B Lymphocytes Use CR4 (CD11c/CD18) for Adhesion, Migration, and Proliferation. Front. Immunol. 2020, 11, 565458. [Google Scholar] [CrossRef]
- Manevich-Mendelson, E.; Grabovsky, V.; Feigelson, S.W.; Cinamon, G.; Gore, Y.; Goverse, G.; Monkley, S.J.; Margalit, R.; Melamed, D.; Mebius, R.E.; et al. Talin1 is required for integrin-dependent B lymphocyte homing to lymph nodes and the bone marrow but not for follicular B-cell maturation in the spleen. Blood 2010, 116, 5907–5918. [Google Scholar] [CrossRef]
- Hart, R.; Stanley, P.; Chakravarty, P.; Hogg, N. The kindlin 3 pleckstrin homology domain has an essential role in lymphocyte function-associated antigen 1 (LFA-1) integrin-mediated B cell adhesion and migration. J. Biol. Chem. 2013, 288, 14852–14862. [Google Scholar] [CrossRef]
- Ballet, R.; Brennan, M.; Brandl, C.; Feng, N.; Berri, J.; Cheng, J.; Ocón, B.; Alborzian Deh Sheikh, A.; Marki, A.; Bi, Y.; et al. A CD22-Shp1 phosphatase axis controls integrin β(7) display and B cell function in mucosal immunity. Nat. Immunol. 2021, 22, 381–390. [Google Scholar] [CrossRef]
- Schippers, A.; Kochut, A.; Pabst, O.; Frischmann, U.; Clahsen, T.; Tenbrock, K.; Müller, W.; Wagner, N. β7 integrin controls immunogenic and tolerogenic mucosal B cell responses. Clin. Immunol. 2012, 144, 87–97. [Google Scholar] [CrossRef] [PubMed]
- Waffarn, E.E.; Hastey, C.J.; Dixit, N.; Soo Choi, Y.; Cherry, S.; Kalinke, U.; Simon, S.I.; Baumgarth, N. Infection-induced type I interferons activate CD11b on B-1 cells for subsequent lymph node accumulation. Nat. Commun. 2015, 6, 8991. [Google Scholar] [CrossRef] [PubMed]
- van Spriel, A.B.; de Keijzer, S.; van der Schaaf, A.; Gartlan, K.H.; Sofi, M.; Light, A.; Linssen, P.C.; Boezeman, J.B.; Zuidscherwoude, M.; Reinieren-Beeren, I.; et al. The tetraspanin CD37 orchestrates the α(4)β(1) integrin-Akt signaling axis and supports long-lived plasma cell survival. Sci. Signal 2012, 5, ra82. [Google Scholar] [CrossRef] [PubMed]
- Fresno Vara, J.A.; Casado, E.; de Castro, J.; Cejas, P.; Belda-Iniesta, C.; González-Barón, M. PI3K/Akt signalling pathway and cancer. Cancer Treat. Rev. 2004, 30, 193–204. [Google Scholar] [CrossRef] [PubMed]
- Guzman, M.; Lundborg, L.R.; Yeasmin, S.; Tyler, C.J.; Zgajnar, N.R.; Taupin, V.; Dobaczewska, K.; Mikulski, Z.; Bamias, G.; Rivera-Nieves, J. An integrin αEβ7-dependent mechanism of IgA transcytosis requires direct plasma cell contact with intestinal epithelium. Mucosal Immunol. 2021, 14, 1347–1357. [Google Scholar] [CrossRef] [PubMed]
- Raso, F.; Sagadiev, S.; Du, S.; Gage, E.; Arkatkar, T.; Metzler, G.; Stuart, L.M.; Orr, M.T.; Rawlings, D.J.; Jackson, S.W.; et al. αv Integrins regulate germinal center B cell responses through noncanonical autophagy. J. Clin. Investig. 2018, 128, 4163–4178. [Google Scholar] [CrossRef] [PubMed]
- Ding, C.; Ma, Y.; Chen, X.; Liu, M.; Cai, Y.; Hu, X.; Xiang, D.; Nath, S.; Zhang, H.G.; Ye, H.; et al. Integrin CD11b negatively regulates BCR signalling to maintain autoreactive B cell tolerance. Nat. Commun. 2013, 4, 2813. [Google Scholar] [CrossRef]
- Acharya, M.; Raso, F.; Sagadiev, S.; Gilbertson, E.; Kadavy, L.; Li, Q.Z.; Yan, M.; Stuart, L.M.; Hamerman, J.A.; Lacy-Hulbert, A. B Cell αv Integrins Regulate TLR-Driven Autoimmunity. J. Immunol. 2020, 205, 1810–1818. [Google Scholar] [CrossRef]
- Mraz, M.; Zent, C.S.; Church, A.K.; Jelinek, D.F.; Wu, X.; Pospisilova, S.; Ansell, S.M.; Novak, A.J.; Kay, N.E.; Witzig, T.E.; et al. Bone marrow stromal cells protect lymphoma B-cells from rituximab-induced apoptosis and targeting integrin α-4-β-1 (VLA-4) with natalizumab can overcome this resistance. Br. J. Haematol. 2011, 155, 53–64. [Google Scholar] [CrossRef]
- Dorner, M.; Zucol, F.; Alessi, D.; Haerle, S.K.; Bossart, W.; Weber, M.; Byland, R.; Bernasconi, M.; Berger, C.; Tugizov, S.; et al. β1 integrin expression increases susceptibility of memory B cells to Epstein-Barr virus infection. J. Virol. 2010, 84, 6667–6677. [Google Scholar] [CrossRef]
- Bromley, S.K.; Akbaba, H.; Mani, V.; Mora-Buch, R.; Chasse, A.Y.; Sama, A.; Luster, A.D. CD49a Regulates Cutaneous Resident Memory CD8(+) T Cell Persistence and Response. Cell Rep. 2020, 32, 108085. [Google Scholar] [CrossRef] [PubMed]
- Rocha-Perugini, V.; González-Granado, J.M.; Tejera, E.; López-Martín, S.; Yañez-Mó, M.; Sánchez-Madrid, F. Tetraspanins CD9 and CD151 at the immune synapse support T-cell integrin signaling. Eur. J. Immunol. 2014, 44, 1967–1975. [Google Scholar] [CrossRef] [PubMed]
- Bachsais, M.; Salti, S.; Zaoui, K.; Hassan, G.S.; Aoudjit, F.; Mourad, W. CD154 inhibits death of T cells via a Cis interaction with the α5β1 integrin. PLoS ONE 2020, 15, e0235753. [Google Scholar] [CrossRef]
- Gitlin, A.D.; Heger, K.; Schubert, A.F.; Reja, R.; Yan, D.; Pham, V.C.; Suto, E.; Zhang, J.; Kwon, Y.C.; Freund, E.C.; et al. Integration of innate immune signalling by caspase-8 cleavage of N4BP1. Nature 2020, 587, 275–280. [Google Scholar] [CrossRef] [PubMed]
- Jankowska, K.I.; Williamson, E.K.; Roy, N.H.; Blumenthal, D.; Chandra, V.; Baumgart, T.; Burkhardt, J.K. Integrins Modulate T Cell Receptor Signaling by Constraining Actin Flow at the Immunological Synapse. Front. Immunol. 2018, 9, 25. [Google Scholar] [CrossRef] [PubMed]
- Mansour, A.A.; Raucci, F.; Sevim, M.; Saviano, A.; Begum, J.; Zhi, Z.; Pezhman, L.; Tull, S.; Maione, F.; Iqbal, A.J. Galectin-9 supports primary T cell transendothelial migration in a glycan and integrin dependent manner. Biomed. Pharmacother. Biomed. Pharmacother. 2022, 151, 113171. [Google Scholar] [CrossRef]
- Pérez-Rivero, G.; Cascio, G.; Soriano, S.F.; Sanz, Á.G.; de Guinoa, J.S.; Rodríguez-Frade, J.M.; Gomariz, R.P.; Holgado, B.L.; Cabañas, C.; Carrasco, Y.R.; et al. Janus kinases 1 and 2 regulate chemokine-mediated integrin activation and naïve T-cell homing. Eur. J. Immunol. 2013, 43, 1745–1757. [Google Scholar] [CrossRef]
- Evans, R.; Lellouch, A.C.; Svensson, L.; McDowall, A.; Hogg, N. The integrin LFA-1 signals through ZAP-70 to regulate expression of high-affinity LFA-1 on T lymphocytes. Blood 2011, 117, 3331–3342. [Google Scholar] [CrossRef]
- Lin, Y.-P.; Cheng, Y.-J.; Huang, J.-Y.; Lin, H.-C.; Yang, B.-C. Zap70 controls the interaction of talin with integrin to regulate the chemotactic directionality of T-cell migration. Mol. Immunol. 2010, 47, 2022–2029. [Google Scholar] [CrossRef]
- DeNucci, C.C.; Shimizu, Y. β1 integrin is critical for the maintenance of antigen-specific CD4 T cells in the bone marrow but not long-term immunological memory. J. Immunol. 2011, 186, 4019–4026. [Google Scholar] [CrossRef]
- Martens, R.; Permanyer, M.; Werth, K.; Yu, K.; Braun, A.; Halle, O.; Halle, S.; Patzer, G.E.; Bošnjak, B.; Kiefer, F.; et al. Efficient homing of T cells via afferent lymphatics requires mechanical arrest and integrin-supported chemokine guidance. Nat. Commun. 2020, 11, 1114. [Google Scholar] [CrossRef] [PubMed]
- Morrison, V.L.; MacPherson, M.; Savinko, T.; Lek, H.S.; Prescott, A.; Fagerholm, S.C. The β2 integrin-kindlin-3 interaction is essential for T-cell homing but dispensable for T-cell activation in vivo. Blood 2013, 122, 1428–1436. [Google Scholar] [CrossRef]
- Nordenfelt, P.; Elliott, H.L.; Springer, T.A. Coordinated integrin activation by actin-dependent force during T-cell migration. Nat. Commun. 2016, 7, 13119. [Google Scholar] [CrossRef] [PubMed]
- Manhas, K.R.; Marshall, P.A.; Wagner, C.E.; Jurutka, P.W.; Mancenido, M.V.; Debray, H.Z.; Blattman, J.N. Rexinoids Modulate Effector T Cell Expression of Mucosal Homing Markers CCR9 and α4β7 Integrin and Direct Their Migration In Vitro. Front. Immunol. 2022, 13, 746484. [Google Scholar] [CrossRef] [PubMed]
- Garçon, F.; Okkenhaug, K. PI3Kδ promotes CD4(+) T-cell interactions with antigen-presenting cells by increasing LFA-1 binding to ICAM-1. Immunology and cell biology 2016, 94, 486–495. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Zheng, Y.; Ran, X.; Du, H.; Feng, H.; Yang, L.; Wen, Y.; Lin, C.; Wang, S.; Huang, M.; et al. Integrin αEβ7(+) T cells direct intestinal stem cell fate decisions via adhesion signaling. Cell Res. 2021, 31, 1291–1307. [Google Scholar] [CrossRef]
- Zondler, L.; Herich, S.; Kotte, P.; Körner, K.; Schneider-Hohendorf, T.; Wiendl, H.; Schwab, N.; Zarbock, A. MCAM/CD146 Signaling via PLCγ1 Leads to Activation of β(1)-Integrins in Memory T-Cells Resulting in Increased Brain Infiltration. Front. Immunol. 2020, 11, 599936. [Google Scholar] [CrossRef]
- Glatigny, S.; Duhen, R.; Arbelaez, C.; Kumari, S.; Bettelli, E. Integrin α L controls the homing of regulatory T cells during CNS autoimmunity in the absence of integrin α 4. Sci. Rep. 2015, 5, 7834. [Google Scholar] [CrossRef]
- Bachsais, M.; Naddaf, N.; Yacoub, D.; Salti, S.; Alaaeddine, N.; Aoudjit, F.; Hassan, G.S.; Mourad, W. The Interaction of CD154 with the α5β1 Integrin Inhibits Fas-Induced T Cell Death. PloS ONE 2016, 11, e0158987. [Google Scholar] [CrossRef]
- Klann, J.E.; Kim, S.H.; Remedios, K.A.; He, Z.; Metz, P.J.; Lopez, J.; Tysl, T.; Olvera, J.G.; Ablack, J.N.; Cantor, J.M.; et al. Integrin Activation Controls Regulatory T Cell-Mediated Peripheral Tolerance. J. Immunol. 2018, 200, 4012–4023. [Google Scholar] [CrossRef]
- Braun, A.; Dewert, N.; Brunnert, F.; Schnabel, V.; Hardenberg, J.H.; Richter, B.; Zachmann, K.; Cording, S.; Claßen, A.; Brans, R.; et al. Integrin αE(CD103) Is Involved in Regulatory T-Cell Function in Allergic Contact Hypersensitivity. J. Investig. Dermatol. 2015, 135, 2982–2991. [Google Scholar] [CrossRef] [PubMed]
- Mair, I.; Zandee, S.E.J.; Toor, I.S.; Saul, L.; McPherson, R.C.; Leech, M.D.; Smyth, D.J.; O’Connor, R.A.; Henderson, N.C.; Anderton, S.M. A Context-Dependent Role for αv Integrins in Regulatory T Cell Accumulation at Sites of Inflammation. Front. Immunol. 2018, 9, 264. [Google Scholar] [CrossRef]
- Worthington, J.J.; Kelly, A.; Smedley, C.; Bauché, D.; Campbell, S.; Marie, J.C.; Travis, M.A. Integrin αvβ8-Mediated TGF-β Activation by Effector Regulatory T Cells Is Essential for Suppression of T-Cell-Mediated Inflammation. Immunity 2015, 42, 903–915. [Google Scholar] [CrossRef]
- Zhang, A.; Paidassi, H.; Lacy-Hulbert, A.; Savill, J. Apoptotic cells induce CD103 expression and immunoregulatory function in myeloid dendritic cell precursors through integrin αv and TGF-β activation. PLoS ONE 2020, 15, e0232307. [Google Scholar] [CrossRef] [PubMed]
- Boucard-Jourdin, M.; Kugler, D.; Endale Ahanda, M.L.; This, S.; De Calisto, J.; Zhang, A.; Mora, J.R.; Stuart, L.M.; Savill, J.; Lacy-Hulbert, A.; et al. β8 Integrin Expression and Activation of TGF-β by Intestinal Dendritic Cells Are Determined by Both Tissue Microenvironment and Cell Lineage. J. Immunol. 2016, 197, 1968–1978. [Google Scholar] [CrossRef] [PubMed]
- Garnier, A.; Laffont, S.; Garnier, L.; Kaba, E.; Deutsch, U.; Engelhardt, B.; Guéry, J.C. CD49d/CD29-integrin controls the accumulation of plasmacytoid dendritic cells into the CNS during neuroinflammation. Eur. J. Immunol. 2019, 49, 2030–2043. [Google Scholar] [CrossRef] [PubMed]
- Sie, C.; Perez, L.G.; Kreutzfeldt, M.; Potthast, M.; Ohnmacht, C.; Merkler, D.; Huber, S.; Krug, A.; Korn, T. Dendritic Cell Accumulation in the Gut and Central Nervous System Is Differentially Dependent on α4 Integrins. J. Immunol. 2019, 203, 1417–1427. [Google Scholar] [CrossRef] [PubMed]
- Duhan, V.; Khairnar, V.; Kitanovski, S.; Hamdan, T.A.; Klein, A.D.; Lang, J.; Ali, M.; Adomati, T.; Bhat, H.; Friedrich, S.K.; et al. Integrin α E (CD103) Limits Virus-Induced IFN-I Production in Conventional Dendritic Cells. Front. Immunol. 2020, 11, 607889. [Google Scholar] [CrossRef]
- Ling, G.S.; Bennett, J.; Woollard, K.J.; Szajna, M.; Fossati-Jimack, L.; Taylor, P.R.; Scott, D.; Franzoso, G.; Cook, H.T.; Botto, M. Integrin CD11b positively regulates TLR4-induced signalling pathways in dendritic cells but not in macrophages. Nat. Commun. 2014, 5, 3039. [Google Scholar] [CrossRef]
- Morrison, V.L.; James, M.J.; Grzes, K.; Cook, P.; Glass, D.G.; Savinko, T.; Lek, H.S.; Gawden-Bone, C.; Watts, C.; Millington, O.R.; et al. Loss of β2-integrin-mediated cytoskeletal linkage reprogrammes dendritic cells to a mature migratory phenotype. Nat. Commun. 2014, 5, 5359. [Google Scholar] [CrossRef]
- Gawden-Bone, C.; West, M.A.; Morrison, V.L.; Edgar, A.J.; McMillan, S.J.; Dill, B.D.; Trost, M.; Prescott, A.; Fagerholm, S.C.; Watts, C. A crucial role for β2 integrins in podosome formation, dynamics and Toll-like-receptor-signaled disassembly in dendritic cells. J. Cell. Sci. 2014, 127, 4213–4224. [Google Scholar] [CrossRef] [PubMed]
- Guenther, C.; Faisal, I.; Uotila, L.M.; Asens, M.L.; Harjunpaa, H.; Savinko, T.; Ohman, T.; Yao, S.; Moser, M.; Morris, S.W.; et al. A β2-Integrin/MRTF-A/SRF Pathway Regulates Dendritic Cell Gene Expression, Adhesion, and Traction Force Generation. Front. Immunol. 2019, 10, 1138. [Google Scholar] [CrossRef] [PubMed]
- Yokota-Nakatsuma, A.; Ohoka, Y.; Takeuchi, H.; Song, S.-Y.; Iwata, M. β 1-integrin ligation and TLR ligation enhance GM-CSF–induced ALDH1A2 expression in dendritic cells, but differentially regulate their anti-inflammatory properties. Sci. Rep. 2016, 6, 37914. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Wu, H.; An, J.; Ballantyne, C.M.; Cyster, J.G. Critical role of integrin CD11c in splenic dendritic cell capture of missing-self CD47 cells to induce adaptive immunity. Proc. Natl. Acad. Sci. USA 2018, 115, 6786–6791. [Google Scholar] [CrossRef] [PubMed]
- Worthington, J.J.; Klementowicz, J.E.; Rahman, S.; Czajkowska, B.I.; Smedley, C.; Waldmann, H.; Sparwasser, T.; Grencis, R.K.; Travis, M.A. Loss of the TGFβ-activating integrin αvβ8 on dendritic cells protects mice from chronic intestinal parasitic infection via control of type 2 immunity. PLoS Pathog. 2013, 9, e1003675. [Google Scholar] [CrossRef]
- Kitamura, H.; Cambier, S.; Somanath, S.; Barker, T.; Minagawa, S.; Markovics, J.; Goodsell, A.; Publicover, J.; Reichardt, L.; Jablons, D.; et al. Mouse and human lung fibroblasts regulate dendritic cell trafficking, airway inflammation, and fibrosis through integrin αvβ8-mediated activation of TGF-β. J. Clin. Investig. 2011, 121, 2863–2875. [Google Scholar] [CrossRef]
- Altorki, T.; Muller, W.; Brass, A.; Cruickshank, S. The role of β2 integrin in dendritic cell migration during infection. BMC Immunol. 2021, 22, 2. [Google Scholar] [CrossRef]
- Nakawesi, J.; This, S.; Hutter, J.; Boucard-Jourdin, M.; Barateau, V.; Muleta, K.G.; Gooday, L.J.; Thomsen, K.F.; Lopez, A.G.; Ulmert, I.; et al. αvβ8 integrin-expression by BATF3-dependent dendritic cells facilitates early IgA responses to Rotavirus. Mucosal Immunol. 2021, 14, 53–67. [Google Scholar] [CrossRef]
- Roe, M.M.; Swain, S.; Sebrell, T.A.; Sewell, M.A.; Collins, M.M.; Perrino, B.A.; Smith, P.D.; Smythies, L.E.; Bimczok, D. Differential regulation of CD103 (αE integrin) expression in human dendritic cells by retinoic acid and Toll-like receptor ligands. J. Leukoc. Biol. 2017, 101, 1169–1180. [Google Scholar] [CrossRef]
- Lukacsi, S.; Gerecsei, T.; Balazs, K.; Francz, B.; Szabo, B.; Erdei, A.; Bajtay, Z. The differential role of CR3 (CD11b/CD18) and CR4 (CD11c/CD18) in the adherence, migration and podosome formation of human macrophages and dendritic cells under inflammatory conditions. PLoS ONE 2020, 15, e0232432. [Google Scholar] [CrossRef]
- March, M.E.; Long, E.O. β2 integrin induces TCRzeta-Syk-phospholipase C-gamma phosphorylation and paxillin-dependent granule polarization in human NK cells. J. Immunol. 2011, 186, 2998–3005. [Google Scholar] [CrossRef] [PubMed]
- Gross, C.C.; Brzostowski, J.A.; Liu, D.; Long, E.O. Tethering of intercellular adhesion molecule on target cells is required for LFA-1-dependent NK cell adhesion and granule polarization. J. Immunol. 2010, 185, 2918–2926. [Google Scholar] [CrossRef] [PubMed]
- Urlaub, D.; Hofer, K.; Muller, M.L.; Watzl, C. LFA-1 Activation in NK Cells and Their Subsets: Influence of Receptors, Maturation, and Cytokine Stimulation. J. Immunol. 2017, 198, 1944–1951. [Google Scholar] [CrossRef] [PubMed]
- Mace, E.M.; Zhang, J.; Siminovitch, K.A.; Takei, F. Elucidation of the integrin LFA-1-mediated signaling pathway of actin polarization in natural killer cells. Blood 2010, 116, 1272–1279. [Google Scholar] [CrossRef]
- Todros-Dawda, I.; Kveberg, L.; Vaage, J.T.; Inngjerdingen, M. The tetraspanin CD53 modulates responses from activating NK cell receptors, promoting LFA-1 activation and dampening NK cell effector functions. PLoS ONE 2014, 9, e97844. [Google Scholar] [CrossRef]
- Xiang, R.F.; Li, S.; Ogbomo, H.; Stack, D.; Mody, C.H. β1 Integrins Are Required To Mediate NK Cell Killing of Cryptococcus neoformans. J. Immunol. 2018, 201, 2369–2376. [Google Scholar] [CrossRef]
- Nielsen, N.; Odum, N.; Urso, B.; Lanier, L.L.; Spee, P. Cytotoxicity of CD56(bright) NK cells towards autologous activated CD4+ T cells is mediated through NKG2D, LFA-1 and TRAIL and dampened via CD94/NKG2A. PLoS ONE 2012, 7, e31959. [Google Scholar] [CrossRef]
- Wang, S.; Zhou, X.; Yang, J. Integrin αvβ3 Is Essential for Maintenance of Decidua Tissue Homeostasis and of Natural Killer Cell Immune Tolerance During Pregnancy. Reprod Sci. 2018, 25, 1424–1430. [Google Scholar] [CrossRef]
- Stotesbury, C.; Alves-Peixoto, P.; Montoya, B.; Ferez, M.; Nair, S.; Snyder, C.M.; Zhang, S.; Knudson, C.J.; Sigal, L.J. α2β1 Integrin Is Required for Optimal NK Cell Proliferation during Viral Infection but Not for Acquisition of Effector Functions or NK Cell-Mediated Virus Control. J. Immunol. 2020, 204, 1582–1591. [Google Scholar] [CrossRef]
- Bouchentouf, M.; Forner, K.A.; Cuerquis, J.; Michaud, V.; Zheng, J.; Paradis, P.; Schiffrin, E.L.; Galipeau, J. Induction of cardiac angiogenesis requires killer cell lectin-like receptor 1 and α4β7 integrin expression by NK cells. J. Immunol. 2010, 185, 7014–7025. [Google Scholar] [CrossRef]
- Wen, L.; Marki, A.; Wang, Z.; Orecchioni, M.; Makings, J.; Billitti, M.; Wang, E.; Suthahar, S.S.A.; Kim, K.; Kiosses, W.B.; et al. A humanized β(2) integrin knockin mouse reveals localized intra- and extravascular neutrophil integrin activation in vivo. Cell Rep. 2022, 39, 110876. [Google Scholar] [CrossRef] [PubMed]
- McCormick, B.; Craig, H.E.; Chu, J.Y.; Carlin, L.M.; Canel, M.; Wollweber, F.; Toivakka, M.; Michael, M.; Astier, A.L.; Norton, L.; et al. A Negative Feedback Loop Regulates Integrin Inactivation and Promotes Neutrophil Recruitment to Inflammatory Sites. J. Immunol. 2019, 203, 1579–1588. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Xu, J.; Kumar, R.S.; Lakshmikanthan, S.; Kapur, R.; Kofron, M.; Chrzanowska-Wodnicka, M.; Filippi, M.D. The small GTPase Rap1b negatively regulates neutrophil chemotaxis and transcellular diapedesis by inhibiting Akt activation. J. Exp. Med. 2014, 211, 1741–1758. [Google Scholar] [CrossRef] [PubMed]
- Bromberger, T.; Klapproth, S.; Rohwedder, I.; Zhu, L.; Mittmann, L.; Reichel, C.A.; Sperandio, M.; Qin, J.; Moser, M. Direct Rap1/Talin1 interaction regulates platelet and neutrophil integrin activity in mice. Blood 2018, 132, 2754–2762. [Google Scholar] [CrossRef]
- Liu, L.; Aerbajinai, W.; Ahmed, S.M.; Rodgers, G.P.; Angers, S.; Parent, C.A. Radil controls neutrophil adhesion and motility through β2-integrin activation. Mol. Biol. Cell 2012, 23, 4751–4765. [Google Scholar] [CrossRef]
- Boras, M.; Volmering, S.; Bokemeyer, A.; Rossaint, J.; Block, H.; Bardel, B.; Van Marck, V.; Heitplatz, B.; Kliche, S.; Reinhold, A.; et al. Skap2 is required for β(2) integrin-mediated neutrophil recruitment and functions. J. Exp. Med. 2017, 214, 851–874. [Google Scholar] [CrossRef]
- Margraf, A.; Germena, G.; Drexler, H.C.A.; Rossaint, J.; Ludwig, N.; Prystaj, B.; Mersmann, S.; Thomas, K.; Block, H.; Gottschlich, W.; et al. The integrin-linked kinase is required for chemokine-triggered high-affinity conformation of the neutrophil β2-integrin LFA-1. Blood 2020, 136, 2200–2205. [Google Scholar] [CrossRef]
- Fan, Z.; McArdle, S.; Marki, A.; Mikulski, Z.; Gutierrez, E.; Engelhardt, B.; Deutsch, U.; Ginsberg, M.; Groisman, A.; Ley, K. Neutrophil recruitment limited by high-affinity bent β2 integrin binding ligand in cis. Nat. Commun. 2016, 7, 12658. [Google Scholar] [CrossRef]
- Gorina, R.; Lyck, R.; Vestweber, D.; Engelhardt, B. β2 integrin-mediated crawling on endothelial ICAM-1 and ICAM-2 is a prerequisite for transcellular neutrophil diapedesis across the inflamed blood-brain barrier. J. Immunol. 2014, 192, 324–337. [Google Scholar] [CrossRef]
- Hahm, E.; Li, J.; Kim, K.; Huh, S.; Rogelj, S.; Cho, J. Extracellular protein disulfide isomerase regulates ligand-binding activity of αMβ2 integrin and neutrophil recruitment during vascular inflammation. Blood 2013, 121, 3789–3800. [Google Scholar] [CrossRef]
- Silva, L.M.; Doyle, A.D.; Greenwell-Wild, T.; Dutzan, N.; Tran, C.L.; Abusleme, L.; Juang, L.J.; Leung, J.; Chun, E.M.; Lum, A.G.; et al. Fibrin is a critical regulator of neutrophil effector function at the oral mucosal barrier. Science 2021, 374, eabl5450. [Google Scholar] [CrossRef] [PubMed]
- El azreq, M.A.; Bourgoin, S.G. Cytohesin-1 regulates human blood neutrophil adhesion to endothelial cells through β2 integrin activation. Mol. Immunol. 2011, 48, 1408–1416. [Google Scholar] [CrossRef] [PubMed]
- Iqbal, A.J.; Krautter, F.; Blacksell, I.A.; Wright, R.D.; Austin-Williams, S.N.; Voisin, M.B.; Hussain, M.T.; Law, H.L.; Niki, T.; Hirashima, M.; et al. Galectin-9 mediates neutrophil capture and adhesion in a CD44 and β2 integrin-dependent manner. FASEB J. Off. Publ. Fed. Am. Soc. Exp. Biol. 2022, 36, e22065. [Google Scholar] [CrossRef]
- Pruenster, M.; Kurz, A.R.; Chung, K.J.; Cao-Ehlker, X.; Bieber, S.; Nussbaum, C.F.; Bierschenk, S.; Eggersmann, T.K.; Rohwedder, I.; Heinig, K.; et al. Extracellular MRP8/14 is a regulator of β2 integrin-dependent neutrophil slow rolling and adhesion. Nat. Commun. 2015, 6, 6915. [Google Scholar] [CrossRef] [PubMed]
- Wee, J.L.; Schulze, K.E.; Jones, E.L.; Yeung, L.; Cheng, Q.; Pereira, C.F.; Costin, A.; Ramm, G.; van Spriel, A.B.; Hickey, M.J.; et al. Tetraspanin CD37 Regulates β2 Integrin-Mediated Adhesion and Migration in Neutrophils. J. Immunol. 2015, 195, 5770–5779. [Google Scholar] [CrossRef] [PubMed]
- Subramanian, B.C.; Melis, N.; Chen, D.; Wang, W.; Gallardo, D.; Weigert, R.; Parent, C.A. The LTB4-BLT1 axis regulates actomyosin and β2-integrin dynamics during neutrophil extravasation. J. Cell Biol. 2020, 219, e201910215. [Google Scholar] [CrossRef] [PubMed]
- Bae, H.B.; Zmijewski, J.W.; Deshane, J.S.; Zhi, D.; Thompson, L.C.; Peterson, C.B.; Chaplin, D.D.; Abraham, E. Vitronectin inhibits neutrophil apoptosis through activation of integrin-associated signaling pathways. Am. J. Respir. Cell Mol. Biol. 2012, 46, 790–796. [Google Scholar] [CrossRef]
- Saldanha-Gama, R.F.; Moraes, J.A.; Mariano-Oliveira, A.; Coelho, A.L.; Walsh, E.M.; Marcinkiewicz, C.; Barja-Fidalgo, C. α(9)β(1) integrin engagement inhibits neutrophil spontaneous apoptosis: Involvement of Bcl-2 family members. Biochim. Et Biophys. Acta 2010, 1803, 848–857. [Google Scholar] [CrossRef]
- Zenaro, E.; Pietronigro, E.; Della Bianca, V.; Piacentino, G.; Marongiu, L.; Budui, S.; Turano, E.; Rossi, B.; Angiari, S.; Dusi, S.; et al. Neutrophils promote Alzheimer’s disease-like pathology and cognitive decline via LFA-1 integrin. Nat. Med. 2015, 21, 880–886. [Google Scholar] [CrossRef]
- Kumar, S.; Xu, J.; Perkins, C.; Guo, F.; Snapper, S.; Finkelman, F.D.; Zheng, Y.; Filippi, M.D. Cdc42 regulates neutrophil migration via crosstalk between WASp, CD11b, and microtubules. Blood 2012, 120, 3563–3574. [Google Scholar] [CrossRef]
- Herbert, J.A.; Deng, Y.; Hardelid, P.; Robinson, E.; Ren, L.; Moulding, D.; Smyth, R.L.; Smith, C.M. β(2)-integrin LFA1 mediates airway damage following neutrophil transepithelial migration during respiratory syncytial virus infection. Eur. Respir. J. 2020, 56, 1902216. [Google Scholar] [CrossRef] [PubMed]
- Rossaint, J.; Herter, J.M.; Van Aken, H.; Napirei, M.; Döring, Y.; Weber, C.; Soehnlein, O.; Zarbock, A. Synchronized integrin engagement and chemokine activation is crucial in neutrophil extracellular trap-mediated sterile inflammation. Blood 2014, 123, 2573–2584. [Google Scholar] [CrossRef] [PubMed]
- McMillan, S.J.; Sharma, R.S.; McKenzie, E.J.; Richards, H.E.; Zhang, J.; Prescott, A.; Crocker, P.R. Siglec-E is a negative regulator of acute pulmonary neutrophil inflammation and suppresses CD11b β2-integrin-dependent signaling. Blood 2013, 121, 2084–2094. [Google Scholar] [CrossRef]
- McMillan, S.J.; Sharma, R.S.; Richards, H.E.; Hegde, V.; Crocker, P.R. Siglec-E promotes β2-integrin-dependent NADPH oxidase activation to suppress neutrophil recruitment to the lung. J. Biol. Chem. 2014, 289, 20370–20376. [Google Scholar] [CrossRef]
- Patel, S.K.; Janjic, J.M. Macrophage targeted theranostics as personalized nanomedicine strategies for inflammatory diseases. Theranostics 2015, 5, 150–172. [Google Scholar] [CrossRef] [PubMed]
- Ho, T.C.; Yeh, S.I.; Chen, S.L.; Tsao, Y.P. Integrin αv and Vitronectin Prime Macrophage-Related Inflammation and Contribute the Development of Dry Eye Disease. Int. J. Mol. Sci. 2021, 22, 8410. [Google Scholar] [CrossRef]
- Cha, B.H.; Shin, S.R.; Leijten, J.; Li, Y.C.; Singh, S.; Liu, J.C.; Annabi, N.; Abdi, R.; Dokmeci, M.R.; Vrana, N.E.; et al. Integrin-Mediated Interactions Control Macrophage Polarization in 3D Hydrogels. Adv. Health Mater. 2017, 6, 1700289. [Google Scholar] [CrossRef]
- Paterson, N.; Lammermann, T. Macrophage network dynamics depend on haptokinesis for optimal local surveillance. Elife 2022, 11, e75354. [Google Scholar] [CrossRef]
- Cui, K.; Ardell, C.L.; Podolnikova, N.P.; Yakubenko, V.P. Distinct Migratory Properties of M1, M2, and Resident Macrophages Are Regulated by α(D)β(2) and α(M)β(2) Integrin-Mediated Adhesion. Front. Immunol. 2018, 9, 2650. [Google Scholar] [CrossRef]
- Becker, H.M.; Rullo, J.; Chen, M.; Ghazarian, M.; Bak, S.; Xiao, H.; Hay, J.B.; Cybulsky, M.I. α1β1 integrin-mediated adhesion inhibits macrophage exit from a peripheral inflammatory lesion. J. Immunol. 2013, 190, 4305–4314. [Google Scholar] [CrossRef]
- Guo, Q.; Furuta, K.; Lucien, F.; Gutierrez Sanchez, L.H.; Hirsova, P.; Krishnan, A.; Kabashima, A.; Pavelko, K.D.; Madden, B.; Alhuwaish, H.; et al. Integrin β(1)-enriched extracellular vesicles mediate monocyte adhesion and promote liver inflammation in murine NASH. J. Hepatol. 2019, 71, 1193–1205. [Google Scholar] [CrossRef] [PubMed]
- Miao, L.; Xin, X.; Xin, H.; Shen, X.; Zhu, Y.Z. Hydrogen Sulfide Recruits Macrophage Migration by Integrin β1-Src-FAK/Pyk2-Rac Pathway in Myocardial Infarction. Sci. Rep. 2016, 6, 22363. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Che, J.; Chang, L.; Guo, M.; Bao, X.; Mu, D.; Sun, X.; Zhang, X.; Lu, W.; Xie, J. CD47- and Integrin α4/β1-Comodified-Macrophage-Membrane-Coated Nanoparticles Enable Delivery of Colchicine to Atherosclerotic Plaque. Adv. Health Mater. 2022, 11, e2101788. [Google Scholar] [CrossRef] [PubMed]
- Furundzija, V.; Fritzsche, J.; Kaufmann, J.; Meyborg, H.; Fleck, E.; Kappert, K.; Stawowy, P. IGF-1 increases macrophage motility via PKC/p38-dependent αvβ3-integrin inside-out signaling. Biochem. Biophys. Res. Commun. 2010, 394, 786–791. [Google Scholar] [CrossRef]
- Wei, J.; Marisetty, A.; Schrand, B.; Gabrusiewicz, K.; Hashimoto, Y.; Ott, M.; Grami, Z.; Kong, L.Y.; Ling, X.; Caruso, H.; et al. Osteopontin mediates glioblastoma-associated macrophage infiltration and is a potential therapeutic target. J. Clin. Investig. 2019, 129, 137–149. [Google Scholar] [CrossRef]
- Lund, S.A.; Wilson, C.L.; Raines, E.W.; Tang, J.; Giachelli, C.M.; Scatena, M. Osteopontin mediates macrophage chemotaxis via α4 and α9 integrins and survival via the α4 integrin. J. Cell. Biochem. 2013, 114, 1194–1202. [Google Scholar] [CrossRef]
- Cui, K.; Podolnikova, N.P.; Bailey, W.; Szmuc, E.; Podrez, E.A.; Byzova, T.V.; Yakubenko, V.P. Inhibition of integrin α(D)β(2)-mediated macrophage adhesion to end product of docosahexaenoic acid (DHA) oxidation prevents macrophage accumulation during inflammation. J. Biol. Chem. 2019, 294, 14370–14382. [Google Scholar] [CrossRef]
- Aziz, M.H.; Cui, K.; Das, M.; Brown, K.E.; Ardell, C.L.; Febbraio, M.; Pluskota, E.; Han, J.; Wu, H.; Ballantyne, C.M.; et al. The Upregulation of Integrin α(D)β(2) (CD11d/CD18) on Inflammatory Macrophages Promotes Macrophage Retention in Vascular Lesions and Development of Atherosclerosis. J. Immunol. 2017, 198, 4855–4867. [Google Scholar] [CrossRef]
- Liu, L.; Zhao, Q.; Kong, M.; Mao, L.; Yang, Y.; Xu, Y. Myocardin-related transcription factor A regulates integrin β 2 transcription to promote macrophage infiltration and cardiac hypertrophy in mice. Cardiovasc. Res. 2022, 118, 844–858. [Google Scholar] [CrossRef]
- Guillot, A.; Guerri, L.; Feng, D.; Kim, S.J.; Ahmed, Y.A.; Paloczi, J.; He, Y.; Schuebel, K.; Dai, S.; Liu, F.; et al. Bile acid-activated macrophages promote biliary epithelial cell proliferation through integrin αvβ6 upregulation following liver injury. J. Clin. Investig. 2021, 131, e132305. [Google Scholar] [CrossRef]
- Wang, B.; Lim, J.H.; Kajikawa, T.; Li, X.; Vallance, B.A.; Moutsopoulos, N.M.; Chavakis, T.; Hajishengallis, G. Macrophage β2-Integrins Regulate IL-22 by ILC3s and Protect from Lethal Citrobacter rodentium-Induced Colitis. Cell. Rep. 2019, 26, 156–161; discussion 162–163. [Google Scholar] [CrossRef] [PubMed]
- Plosa, E.J.; Benjamin, J.T.; Sucre, J.M.; Gulleman, P.M.; Gleaves, L.A.; Han, W.; Kook, S.; Polosukhin, V.V.; Haake, S.M.; Guttentag, S.H.; et al. β1 Integrin regulates adult lung alveolar epithelial cell inflammation. JCI Insight 2020, 5, e129259. [Google Scholar] [CrossRef]
- Zhang, Y.; Shi, X.; Han, J.; Peng, W.; Fang, Z.; Zhou, Y.; Xu, X.; Lin, J.; Xiao, F.; Zhao, L.; et al. Convallatoxin Promotes M2 Macrophage Polarization to Attenuate Atherosclerosis Through PPARgamma-Integrin α(v)β(5) Signaling Pathway. Drug Des. Devel. Ther. 2021, 15, 803–812. [Google Scholar] [CrossRef] [PubMed]
- Antonov, A.S.; Antonova, G.N.; Munn, D.H.; Mivechi, N.; Lucas, R.; Catravas, J.D.; Verin, A.D. αVβ3 integrin regulates macrophage inflammatory responses via PI3 kinase/Akt-dependent NF-kappaB activation. J. Cell. Physiol. 2011, 226, 469–476. [Google Scholar] [CrossRef]
- Schmid, M.C.; Avraamides, C.J.; Foubert, P.; Shaked, Y.; Kang, S.W.; Kerbel, R.S.; Varner, J.A. Combined blockade of integrin-α4β1 plus cytokines SDF-1α or IL-1β potently inhibits tumor inflammation and growth. Cancer Res. 2011, 71, 6965–6975. [Google Scholar] [CrossRef] [PubMed]
- Ruan, S.; Lin, M.; Zhu, Y.; Lum, L.; Thakur, A.; Jin, R.; Shao, W.; Zhang, Y.; Hu, Y.; Huang, S.; et al. Integrin β4-Targeted Cancer Immunotherapies Inhibit Tumor Growth and Decrease Metastasis. Cancer Res. 2020, 80, 771–783. [Google Scholar] [CrossRef] [PubMed]
- Peng, Z.W.; Ikenaga, N.; Liu, S.B.; Sverdlov, D.Y.; Vaid, K.A.; Dixit, R.; Weinreb, P.H.; Violette, S.; Sheppard, D.; Schuppan, D.; et al. Integrin αvβ6 critically regulates hepatic progenitor cell function and promotes ductular reaction, fibrosis, and tumorigenesis. Hepatology 2016, 63, 217–232. [Google Scholar] [CrossRef]
- Gao, L.; Zhang, C.; Gao, D.; Liu, H.; Yu, X.; Lai, J.; Wang, F.; Lin, J.; Liu, Z. Enhanced Anti-Tumor Efficacy through a Combination of Integrin αvβ6-Targeted Photodynamic Therapy and Immune Checkpoint Inhibition. Theranostics 2016, 6, 627–637. [Google Scholar] [CrossRef]
- Zhang, B.; Ye, H.; Ren, X.; Zheng, S.; Zhou, Q.; Chen, C.; Lin, Q.; Li, G.; Wei, L.; Fu, Z.; et al. Macrophage-expressed CD51 promotes cancer stem cell properties via the TGF-β1/smad2/3 axis in pancreatic cancer. Cancer Lett. 2019, 459, 204–215. [Google Scholar] [CrossRef]
- Peng, P.; Zhu, H.; Liu, D.; Chen, Z.; Zhang, X.; Guo, Z.; Dong, M.; Wan, L.; Zhang, P.; Liu, G.; et al. TGFBI secreted by tumor-associated macrophages promotes glioblastoma stem cell-driven tumor growth via integrin αvβ5-Src-Stat3 signaling. Theranostics 2022, 12, 4221–4236. [Google Scholar] [CrossRef]
- Zhu, H.; Wang, G.; Zhu, H.; Xu, A. ITGA5 is a prognostic biomarker and correlated with immune infiltration in gastrointestinal tumors. BMC Cancer 2021, 21, 269. [Google Scholar] [CrossRef] [PubMed]
- Malenica, I.; Adam, J.; Corgnac, S.; Mezquita, L.; Auclin, E.; Damei, I.; Grynszpan, L.; Gros, G.; de Montpréville, V.; Planchard, D.; et al. Integrin-α(V)-mediated activation of TGF-β regulates anti-tumour CD8 T cell immunity and response to PD-1 blockade. Nat Commun 2021, 12, 5209. [Google Scholar] [CrossRef] [PubMed]
- Myint, P.K.; Park, E.J.; Gaowa, A.; Kawamoto, E.; Shimaoka, M. Targeted remodeling of breast cancer and immune cell homing niches by exosomal integrins. Diagn Pathol. 2020, 15, 38. [Google Scholar] [CrossRef] [PubMed]
- Montresor, A.; Toffali, L.; Mirenda, M.; Rigo, A.; Vinante, F.; Laudanna, C. JAK2 tyrosine kinase mediates integrin activation induced by CXCL12 in B-cell chronic lymphocytic leukemia. Oncotarget 2015, 6, 34245–34257. [Google Scholar] [CrossRef]
- Stockis, J.; Lienart, S.; Colau, D.; Collignon, A.; Nishimura, S.L.; Sheppard, D.; Coulie, P.G.; Lucas, S. Blocking immunosuppression by human Tregs in vivo with antibodies targeting integrin αVβ8. Proc. Natl. Acad. Sci. USA 2017, 114, E10161–E10168. [Google Scholar] [CrossRef]
- Gandoglia, I.; Ivaldi, F.; Carrega, P.; Armentani, E.; Ferlazzo, G.; Mancardi, G.; Kerlero de Rosbo, N.; Uccelli, A.; Laroni, A. In vitro VLA-4 blockade results in an impaired NK cell-mediated immune surveillance against melanoma. Immunol. Lett. 2017, 181, 109–115. [Google Scholar] [CrossRef]
- Busenhart, P.; Montalban-Arques, A.; Katkeviciute, E.; Morsy, Y.; Van Passen, C.; Hering, L.; Atrott, K.; Lang, S.; Garzon, J.F.G.; Naschberger, E.; et al. Inhibition of integrin αvβ6 sparks T-cell antitumor response and enhances immune checkpoint blockade therapy in colorectal cancer. J. Immunother. Cancer 2022, 10, e003465. [Google Scholar] [CrossRef]
- Laine, A.; Labiad, O.; Hernandez-Vargas, H.; This, S.; Sanlaville, A.; Leon, S.; Dalle, S.; Sheppard, D.; Travis, M.A.; Paidassi, H.; et al. Regulatory T cells promote cancer immune-escape through integrin αvβ8-mediated TGF-β activation. Nat. Commun. 2021, 12, 6228. [Google Scholar] [CrossRef]
- Foubert, P.; Kaneda, M.M.; Varner, J.A. PI3Kgamma Activates Integrin α4 and Promotes Immune Suppressive Myeloid Cell Polarization during Tumor Progression. Cancer Immunol. Res. 2017, 5, 957–968. [Google Scholar] [CrossRef]
- Berrazouane, S.; Boisvert, M.; Salti, S.; Mourad, W.; Al-Daccak, R.; Barabe, F.; Aoudjit, F. Beta1 integrin blockade overcomes doxorubicin resistance in human T-cell acute lymphoblastic leukemia. Cell Death Dis 2019, 10, 357. [Google Scholar] [CrossRef]
- Corgnac, S.; Boutet, M.; Kfoury, M.; Naltet, C.; Mami-Chouaib, F. The Emerging Role of CD8(+) Tissue Resident Memory T (T(RM)) Cells in Antitumor Immunity: A Unique Functional Contribution of the CD103 Integrin. Front. Immunol. 2018, 9, 1904. [Google Scholar] [CrossRef] [PubMed]
- Gauthier, L.; Corgnac, S.; Boutet, M.; Gros, G.; Validire, P.; Bismuth, G.; Mami-Chouaib, F. Paxillin Binding to the Cytoplasmic Domain of CD103 Promotes Cell Adhesion and Effector Functions for CD8(+) Resident Memory T Cells in Tumors. Cancer Res. 2017, 77, 7072–7082. [Google Scholar] [CrossRef] [PubMed]
- Hickman, A.; Koetsier, J.; Kurtanich, T.; Nielsen, M.C.; Winn, G.; Wang, Y.; Bentebibel, S.E.; Shi, L.; Punt, S.; Williams, L.; et al. LFA-1 activation enriches tumor-specific T cells in a cold tumor model and synergizes with CTLA-4 blockade. J. Clin. Investig. 2022, 132, e154152. [Google Scholar] [CrossRef] [PubMed]
- Mahuron, K.M.; Moreau, J.M.; Glasgow, J.E.; Boda, D.P.; Pauli, M.L.; Gouirand, V.; Panjabi, L.; Grewal, R.; Luber, J.M.; Mathur, A.N.; et al. Layilin augments integrin activation to promote antitumor immunity. J. Exp. Med. 2020, 217, e20192080. [Google Scholar] [CrossRef]
- Boutet, M.; Gauthier, L.; Leclerc, M.; Gros, G.; de Montpreville, V.; Théret, N.; Donnadieu, E.; Mami-Chouaib, F. TGFβ Signaling Intersects with CD103 Integrin Signaling to Promote T-Lymphocyte Accumulation and Antitumor Activity in the Lung Tumor Microenvironment. Cancer Res. 2016, 76, 1757–1769. [Google Scholar] [CrossRef]
- Franciszkiewicz, K.; Le Floc’h, A.; Boutet, M.; Vergnon, I.; Schmitt, A.; Mami-Chouaib, F. CD103 or LFA-1 engagement at the immune synapse between cytotoxic T cells and tumor cells promotes maturation and regulates T-cell effector functions. Cancer Res. 2013, 73, 617–628. [Google Scholar] [CrossRef]
- Anikeeva, N.; Steblyanko, M.; Fayngerts, S.; Kopylova, N.; Marshall, D.J.; Powers, G.D.; Sato, T.; Campbell, K.S.; Sykulev, Y. Integrin receptors on tumor cells facilitate NK cell-mediated antibody-dependent cytotoxicity. Eur. J. Immunol. 2014, 44, 2331–2339. [Google Scholar] [CrossRef]
- Tang, Z.; Davidson, D.; Li, R.; Zhong, M.C.; Qian, J.; Chen, J.; Veillette, A. Inflammatory macrophages exploit unconventional pro-phagocytic integrins for phagocytosis and anti-tumor immunity. Cell Rep. 2021, 37, 110111. [Google Scholar] [CrossRef]
- Evans, R.; Flores-Borja, F.; Nassiri, S.; Miranda, E.; Lawler, K.; Grigoriadis, A.; Monypenny, J.; Gillet, C.; Owen, J.; Gordon, P.; et al. Integrin-Mediated Macrophage Adhesion Promotes Lymphovascular Dissemination in Breast Cancer. Cell. Rep. 2019, 27, 1967–1978.e1964. [Google Scholar] [CrossRef]
- Guha, P.; Cunetta, M.; Somasundar, P.; Espat, N.J.; Junghans, R.P.; Katz, S.C. Frontline Science: Functionally impaired geriatric CAR-T cells rescued by increased α5β1 integrin expression. J. Leukoc. Biol. 2017, 102, 201–208. [Google Scholar] [CrossRef]
- Zhao, Z.; Chen, Y.; Francisco, N.M.; Zhang, Y.; Wu, M. The application of CAR-T cell therapy in hematological malignancies: Advantages and challenges. Acta Pharm. Sin. B 2018, 8, 539–551. [Google Scholar] [CrossRef] [PubMed]
- Phanthaphol, N.; Somboonpatarakun, C.; Suwanchiwasiri, K.; Chieochansin, T.; Sujjitjoon, J.; Wongkham, S.; Maher, J.; Junking, M.; Yenchitsomanus, P.T. Chimeric Antigen Receptor T Cells Targeting Integrin αvβ6 Expressed on Cholangiocarcinoma Cells. Front. Oncol. 2021, 11, 657868. [Google Scholar] [CrossRef] [PubMed]
- Whilding, L.M.; Parente-Pereira, A.C.; Zabinski, T.; Davies, D.M.; Petrovic, R.M.G.; Kao, Y.V.; Saxena, S.A.; Romain, A.; Costa-Guerra, J.A.; Violette, S.; et al. Targeting of Aberrant αvβ6 Integrin Expression in Solid Tumors Using Chimeric Antigen Receptor-Engineered T Cells. Mol. Ther. J. Am. Soc. Gene Ther. 2017, 25, 259–273. [Google Scholar] [CrossRef] [PubMed]
- Hosen, N.; Matsunaga, Y.; Hasegawa, K.; Matsuno, H.; Nakamura, Y.; Makita, M.; Watanabe, K.; Yoshida, M.; Satoh, K.; Morimoto, S.; et al. The activated conformation of integrin β(7) is a novel multiple myeloma-specific target for CAR T cell therapy. Nature medicine 2017, 23, 1436–1443. [Google Scholar] [CrossRef]
- Sterner, R.C.; Sterner, R.M. CAR-T cell therapy: Current limitations and potential strategies. Blood Cancer J. 2021, 11, 69. [Google Scholar] [CrossRef]
- Fujii, K.; Kurozumi, K.; Ichikawa, T.; Onishi, M.; Shimazu, Y.; Ishida, J.; Chiocca, E.A.; Kaur, B.; Date, I. The integrin inhibitor cilengitide enhances the anti-glioma efficacy of vasculostatin-expressing oncolytic virus. Cancer Gene Ther. 2013, 20, 437–444. [Google Scholar] [CrossRef]
- Lee, T.J.; Nair, M.; Banasavadi-Siddegowda, Y.; Liu, J.; Nallanagulagari, T.; Jaime-Ramirez, A.C.; Guo, J.Y.; Quadri, H.; Zhang, J.; Bockhorst, K.H.; et al. Enhancing Therapeutic Efficacy of Oncolytic Herpes Simplex Virus-1 with Integrin β1 Blocking Antibody OS2966. Mol. Cancer Ther. 2019, 18, 1127–1136. [Google Scholar] [CrossRef]
- Davies, J.A.; Marlow, G.; Uusi-Kerttula, H.K.; Seaton, G.; Piggott, L.; Badder, L.M.; Clarkson, R.W.E.; Chester, J.D.; Parker, A.L. Efficient Intravenous Tumor Targeting Using the αvβ6 Integrin-Selective Precision Virotherapy Ad5(NULL)-A20. Viruses 2021, 13, 864. [Google Scholar] [CrossRef]
- Uusi-Kerttula, H.; Davies, J.A.; Thompson, J.M.; Wongthida, P.; Evgin, L.; Shim, K.G.; Bradshaw, A.; Baker, A.T.; Rizkallah, P.J.; Jones, R.; et al. Ad5(NULL)-A20: A Tropism-Modified, αvβ6 Integrin-Selective Oncolytic Adenovirus for Epithelial Ovarian Cancer Therapies. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2018, 24, 4215–4224. [Google Scholar] [CrossRef]
- Pesonen, S.; Diaconu, I.; Cerullo, V.; Escutenaire, S.; Raki, M.; Kangasniemi, L.; Nokisalmi, P.; Dotti, G.; Guse, K.; Laasonen, L.; et al. Integrin targeted oncolytic adenoviruses Ad5-D24-RGD and Ad5-RGD-D24-GMCSF for treatment of patients with advanced chemotherapy refractory solid tumors. Int. J. Cancer 2012, 130, 1937–1947. [Google Scholar] [CrossRef]
- Watanabe, N.; McKenna, M.K.; Rosewell Shaw, A.; Suzuki, M. Clinical CAR-T Cell and Oncolytic Virotherapy for Cancer Treatment. Mol. Ther. J. Am. Soc. Gene Ther. 2021, 29, 505–520. [Google Scholar] [CrossRef] [PubMed]
- Gambhir, S.S.; National Cancer Institute; Stanford University. 18F-FPPRGD2 PET/CT or PET/MRI in Predicting Early Response in Patients With Cancer Receiving Anti-Angiogenesis Therapy. 2013. Available online: https://ClinicalTrials.gov/show/NCT01806675 (accessed on 21 March 2023).
- Healthcare, G. A Proof-of-concept Study to Assess the Ability of 18F AH-111585 PET Imaging to Detect Tumours and Angiogenesis. 2007. Available online: https://ClinicalTrials.gov/show/NCT00565721 (accessed on 21 March 2023).
- Rigshospitalet, D. PET/CT Imaging of Angiogenesis in Patients With Neuroendocrine Tumors Using 68Ga-NODAGA-E c(RGDyK) 2. 2017. Available online: https://ClinicalTrials.gov/show/NCT03271281 (accessed on 21 March 2023).
- Ma, P.; Jin, X.; Fan, Z.; Wang, Z.; Yue, S.; Wu, C.; Chen, S.; Wu, Y.; Chen, M.; Gu, D.; et al. Super-enhancer receives signals from the extracellular matrix to induce PD-L1-mediated immune evasion via integrin/BRAF/TAK1/ERK/ETV4 signaling. Cancer Biol. Med. 2021, 19, 669–684. [Google Scholar] [CrossRef] [PubMed]
- Vannini, A.; Leoni, V.; Barboni, C.; Sanapo, M.; Zaghini, A.; Malatesta, P.; Campadelli-Fiume, G.; Gianni, T. αvβ3-integrin regulates PD-L1 expression and is involved in cancer immune evasion. Proc. Natl. Acad. Sci. USA 2019, 116, 20141–20150. [Google Scholar] [CrossRef] [PubMed]
- Sofias, A.M.; Toner, Y.C.; Meerwaldt, A.E.; van Leent, M.M.T.; Soultanidis, G.; Elschot, M.; Gonai, H.; Grendstad, K.; Flobak, Å.; Neckmann, U.; et al. Tumor Targeting by α(v)β(3)-Integrin-Specific Lipid Nanoparticles Occurs via Phagocyte Hitchhiking. ACS Nano 2020, 14, 7832–7846. [Google Scholar] [CrossRef]
- Biogen. Natalizumab (BG00002, Tysabri) Study in Japanese Participants With Relapsing-Remitting Multiple Sclerosis (RRMS). 2010. Available online: https://ClinicalTrials.gov/show/NCT01440101 (accessed on 21 March 2023).
- Biogen; Pharmaceuticals, E. Safety Study of Natalizumab to Treat Multiple Sclerosis (MS). 2007. Available online: https://ClinicalTrials.gov/show/NCT00559702 (accessed on 21 March 2023).
- Biogen; Pharmaceuticals, E. A Pharmacokinetic (PK) Study of Natalizumab (Tysabri) at Steady State. 2008. Available online: https://ClinicalTrials.gov/show/NCT00744679 (accessed on 21 March 2023).
- Buffalo, U.A. Natalizumab Temporary Discontinuation Study. 2012. Available online: https://ClinicalTrials.gov/show/NCT02775110 (accessed on 21 March 2023).
- Pan, X.; Yi, M.; Liu, C.; Jin, Y.; Liu, B.; Hu, G.; Yuan, X. Cilengitide, an αvβ3-integrin inhibitor, enhances the efficacy of anti-programmed cell death-1 therapy in a murine melanoma model. Bioengineered 2022, 13, 4557–4572. [Google Scholar] [CrossRef]
- de Miguel, M.; Calvo, E. Clinical Challenges of Immune Checkpoint Inhibitors. Cancer Cell. 2020, 38, 326–333. [Google Scholar] [CrossRef]
- Szaryńska, M.; Olejniczak, A.; Kobiela, J.; Łaski, D.; Śledziński, Z.; Kmieć, Z. Cancer stem cells as targets for DC-based immunotherapy of colorectal cancer. Sci. Rep. 2018, 8, 12042. [Google Scholar] [CrossRef]
- van Montfoort, N.; Borst, L.; Korrer, M.J.; Sluijter, M.; Marijt, K.A.; Santegoets, S.J.; van Ham, V.J.; Ehsan, I.; Charoentong, P.; André, P.; et al. NKG2A Blockade Potentiates CD8 T Cell Immunity Induced by Cancer Vaccines. Cell 2018, 175, 1744–1755.e15. [Google Scholar] [CrossRef]
- Maeda, N.; Ohashi, T.; Chagan-Yasutan, H.; Hattori, T.; Takahashi, Y.; Harigae, H.; Hasegawa, H.; Yamada, Y.; Fujii, M.; Maenaka, K.; et al. Osteopontin-integrin interaction as a novel molecular target for antibody-mediated immunotherapy in adult T-cell leukemia. Retrovirology 2015, 12, 99. [Google Scholar] [CrossRef]
- Kelly, K.A.; Nahrendorf, M.; Yu, A.M.; Reynolds, F.; Weissleder, R. In vivo phage display selection yields atherosclerotic plaque targeted peptides for imaging. Mol. Imaging Biol. 2006, 8, 201–207. [Google Scholar] [CrossRef]
- Ailuno, G.; Baldassari, S.; Zuccari, G.; Schlich, M.; Caviglioli, G. Peptide-based nanosystems for vascular cell adhesion molecule-1 targeting: A real opportunity for therapeutic and diagnostic agents in inflammation associated disorders. J. Drug Deliv. Sci. Technol. 2020, 55, 101461. [Google Scholar] [CrossRef]
- Koudrina, A.; DeRosa, M.C. Advances in Medical Imaging: Aptamer- and Peptide-Targeted MRI and CT Contrast Agents. ACS Omega 2020, 5, 22691–22701. [Google Scholar] [CrossRef] [PubMed]
- Pastorino, S.; Baldassari, S.; Ailuno, G.; Zuccari, G.; Drava, G.; Petretto, A.; Cossu, V.; Marini, C.; Alfei, S.; Florio, T.; et al. Two Novel PET Radiopharmaceuticals for Endothelial Vascular Cell Adhesion Molecule-1 (VCAM-1) Targeting. Pharmaceutics 2021, 13, 1025. [Google Scholar] [CrossRef] [PubMed]
- Desgrosellier, J.S.; Cheresh, D.A. Integrins in cancer: Biological implications and therapeutic opportunities. Nat. Rev. Cancer 2010, 10, 9–22. [Google Scholar] [CrossRef]
- Haugh, A.M.; Probasco, J.C.; Johnson, D.B. Neurologic complications of immune checkpoint inhibitors. Expert Opin. Drug Safety 2020, 19, 479–488. [Google Scholar] [CrossRef]
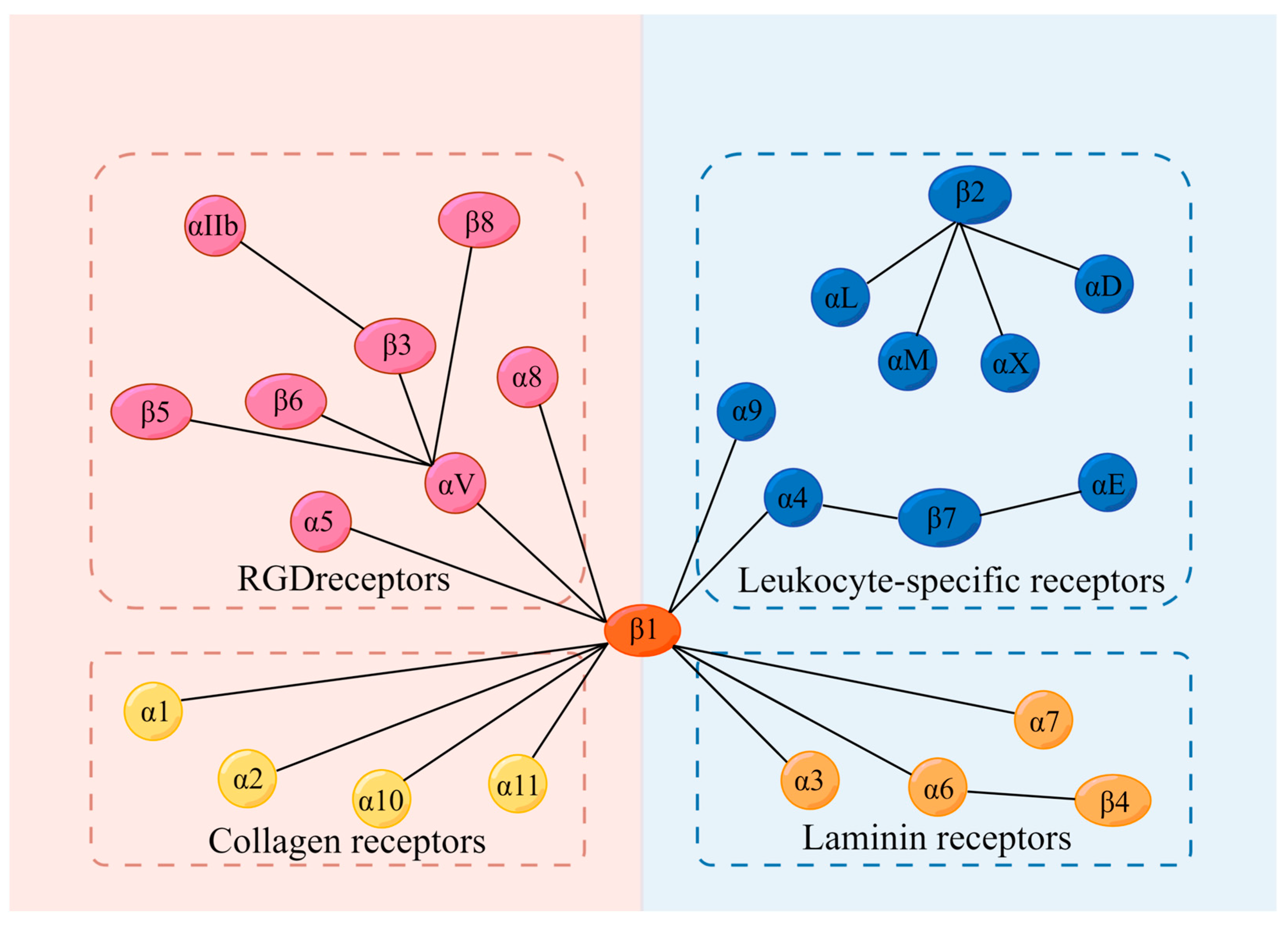

| Category | Integrin | Ligand | Major Function | Reference |
|---|---|---|---|---|
| RGD-binding integrins | αVβ1 | TGF-β; fibronectin; osteopontin; neural cell-adhesion molecule L1 | 1. Inducing the accumulation of fibroblasts in ECM; 2. Inducing cytoskeleton reorganization; 3. Inducing cell differentiation; 4. Inducing vascular response to mechanical stimulation | [1,19,20,21,22,23] |
| αVβ3 | Vitronectin; TGF-β; fibronectin; osteopontin; neural cell adhesion molecule L1; fibrinogen; von Willebrand factor; thrombospondin; fibrillin; tenascin | 1. Participating in angiogenesis, ECM regulation, vascular smooth muscle cell migration, and osteoclast adhesion to the bone matrix; 2. Regulating the migration and phagocytosis of immune cells | [1,24,25,26] | |
| αVβ5 | TGF-β; osteopontin; vitronectin; bone sialic protein; thrombospondin; NOV | 1. Participating in wound healing, matrix molecule synthesis, and type I procollagen expression; 2. Participating in cerebellar granule cell precursor differentiation | [1,27,28,29,30,31,32] | |
| αVβ6 | TGF-β; fibronectin; osteopontin; ADAM | 1. Regulating innate immunity and anti-inflammatory surveillance; 2. Participating in the process of tooth enamel formation | [1,33,34,35,36] | |
| αVβ8 | TGF-β | 1. Regulating neurovascular development; 2. Regulating immune cell recruitment and activation; 3. Regulating stem cell migration or differentiation | [37,38] | |
| α5β1 | Fibronectin; fibrinogen; fibrillin; osteopontin; thrombospondin | 1. Promoting cell migration, invasion, proliferation, and aging; 2. Maintaining the normal function of T cells | [39,40,41,42,43] | |
| α8β1 | TGF-β; tenascin; fibronectin; osteopontin; vitronectin; nephronectin | 1. A cell migration regulator | [1,44] | |
| αIIbβ3 | Fibrinogen; fibronectin; thrombospondin; vitronectin; von Willebrand factor | 1. Inducing platelet activation and arterial thrombosis | [1,45] | |
| Leukocyte cell-adhesion integrins | αLβ2 | ICAM-1 | 1. Regulating inflammation; 2. Inducing leukocyte spreading and crawling toward infection; 3. Enhancing the phagocytosis of neutrophils | [46,47,48] |
| αMβ2 | ICAM-2; C3d-opsonized particles; Zymosan; LL-37 | [46,48,49,50,51,52] | ||
| α4β1 | VCAM-1 | 1. Slowing the rolling of leukocytes | [46] | |
| αXβ2 | Osteopontin | 1. Regulating the anti-inflammatory function of macrophages | [53,54,55] | |
| αDβ2 | ICAM-1; ICAM-3; VCAM-1 | 1. Regulating inflammation | [48,56] | |
| αEβ7 | E-cadherin | 1. Mediating lymphocyte attachment to intestinal and skin epithelial cells | [57] | |
| α9β1 | 1. Regulating hematopoietic processes | [58,59] | ||
| α4β7 | VCAM-1; MAdCAM-1 | 1. Mediating the homing of lymphocytes to gut tissues | [60,61] | |
| Collagen (GFOGER)-binding integrins | α1β1 | Collagens I, III, IV, IX, XIII, XVI; Collagen IV chain-derived peptide | 1. A promoter of T cells in inflammatory responses; 2. Mediating monocyte transmigration; 3. Participating in damage repair processes | [62,63,64,65,66] |
| α2β1 | Collagens I, III, IV, V, XI, XVI, XXIII; lumican; decorin | 1. Stabilizing thrombi; 2. Mediating bone loss | [67,68,69,70] | |
| α10β1 | Collagens II | 1. Participating in cartilage production and skeletal development; 2. A biomarker of chondrogenic stem cells | [71,72] | |
| α11β1 | 1. Participating in tooth eruption; 2. Inducing osteogenic differentiation of mesenchymal stem cells; 3. Promoting myofibroblast differentiation; 4. Wound healing | [70,73,74] | ||
| Laminin-binding integrins | α3β1 | Laminin-332; laminin-511 | 1. Mediating cell adhesion to the basement membrane and cell-to-cell communication | [75] |
| α6β1 | Laminin-111; laminin-511; laminin-332 | 1. Regulating luteal formation and follicle growth; 2. A regulator of angiogenesis | [76,77,78,79] | |
| α6β4 | Laminins; epidermal integral ligand proteins | 1. Maintaining the integrity and stability of epithelial cells; 2. Participating in the regulation of cell death, autophagy, angiogenesis, aging, and differentiation | [75,80,81] | |
| α7β1 | Laminin-211; laminin-221 | 1. Participating in vascular development and integrity | [82,83] | |
| α1β1, α2β1, α10β1, αvβ3 | [84,85,86] |
| Name | Targeting Integrins | Indication | Mechanism | Clinical Trial | Reference |
|---|---|---|---|---|---|
| CAR-T therapy | β7 | Relapsed and/or refractory MS | Binding of CAR-T cells with integrins to enhance specificity | [224] | |
| αVβ6 | Solid tumor | [222,223] | |||
| αVβ3 | Solid tumor | [1] | |||
| Oncolytic virotherapy | αVβ6 | Pancreatic cancer and breast cancer | Modify specific virus to mediate immune killing | [228] | |
| Epithelial ovarian cancer | [229] | ||||
| β1 | Solid tumor | [227] | |||
| ICI therapy | αVβ3 | Solid tumor | Inhibition of the immune checkpoint on T cells to enhance T-cell immunotolerance | [202,232,233,234,235,236] | |
| αVβ6 | Colorectal cancer | [211] | |||
| Other strategies | β4 | B-CLL | [196] | ||
| αVβ3 | Solid tumor | NCT01806675 NCT00565721 NCT02775110 | [232,233,234,237] | ||
| α4β1 | MS | NCT01440101 NCT00559702 NCT00744679 NCT02775110 | [206,238,239,240,241] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, Q.; Zhang, S.; Chen, J.; Xie, Z. The Interplay between Integrins and Immune Cells as a Regulator in Cancer Immunology. Int. J. Mol. Sci. 2023, 24, 6170. https://doi.org/10.3390/ijms24076170
Zhang Q, Zhang S, Chen J, Xie Z. The Interplay between Integrins and Immune Cells as a Regulator in Cancer Immunology. International Journal of Molecular Sciences. 2023; 24(7):6170. https://doi.org/10.3390/ijms24076170
Chicago/Turabian StyleZhang, Qingfang, Shuo Zhang, Jianrui Chen, and Zhenzhen Xie. 2023. "The Interplay between Integrins and Immune Cells as a Regulator in Cancer Immunology" International Journal of Molecular Sciences 24, no. 7: 6170. https://doi.org/10.3390/ijms24076170
APA StyleZhang, Q., Zhang, S., Chen, J., & Xie, Z. (2023). The Interplay between Integrins and Immune Cells as a Regulator in Cancer Immunology. International Journal of Molecular Sciences, 24(7), 6170. https://doi.org/10.3390/ijms24076170





