Severe Acute Liver Dysfunction Induces Delayed Hepatocyte Swelling and Cytoplasmic Vacuolization, and Delayed Cortical Neuronal Cell Death
Abstract
:1. Introduction
2. Results
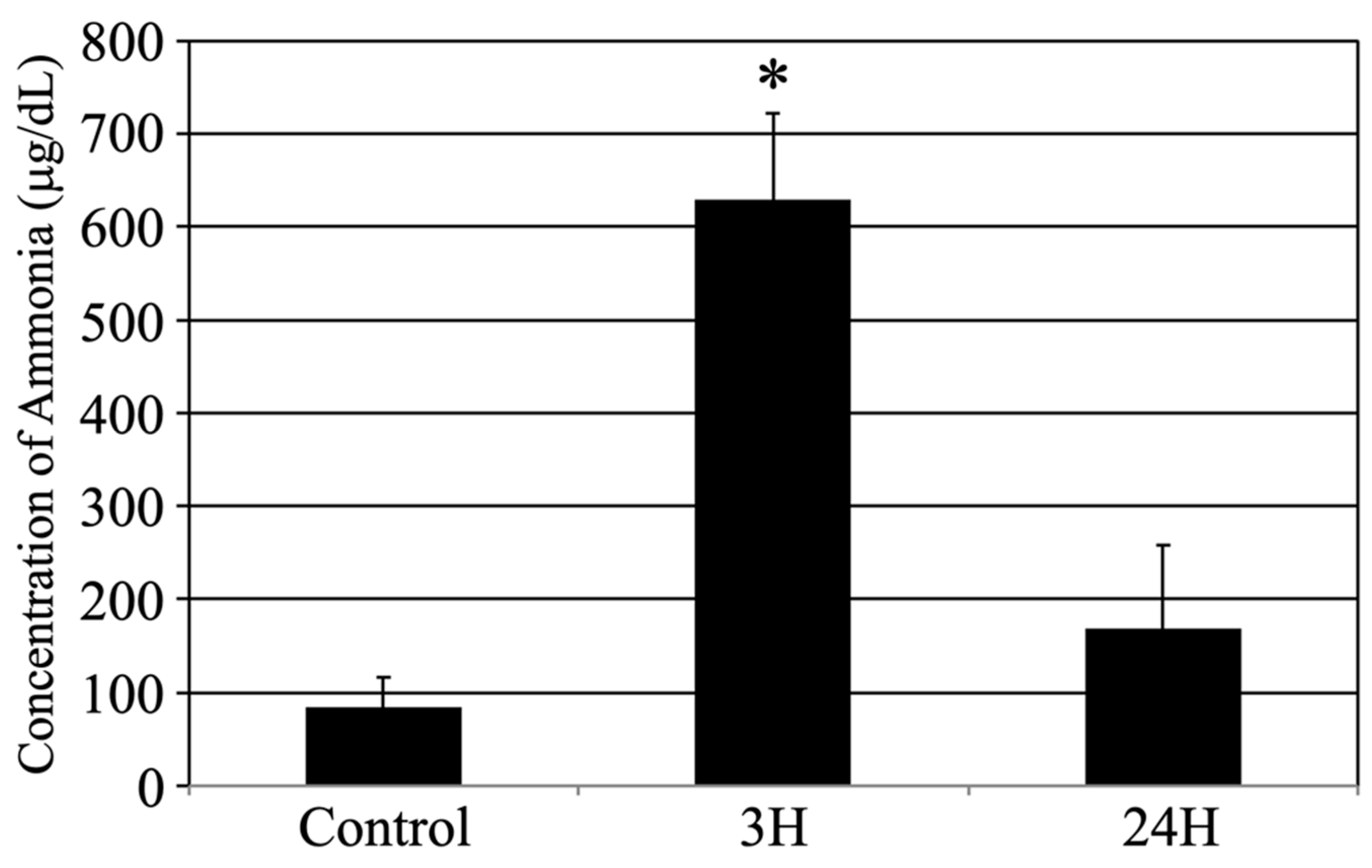
2.1. Induction of Hepatic Encephalopathy
2.2. Histopathological Evaluation of the Hepatic Structural Changes in Response to Hepatic Encephalopathy
2.3. Biochemical Analyses
2.4. Histopathological Evaluation of the Changes in the Cerebral Cortex in Response to Hepatic Encephalopathy
2.5. Brain Inflammatory Response after Hepatic Encephalopathy
3. Discussion
4. Materials and Methods
4.1. Study Design
4.2. Inducing Acute Liver Failure
4.3. Biochemical Analysis
4.4. Tissue Preparation
4.5. Brain Water Measurement
4.6. Histological Analysis
4.7. Electron Microscopical Analysis
4.8. Western Blot Analysis
4.9. Data Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Zhang, J.J.; Meng, X.; Li, Y.; Zhou, Y.; Xu, D.P.; Li, S.; Li, H.B. Effects of Melatonin on Liver Injuries and Diseases. Int. J. Mol. Sci. 2017, 18, 673. [Google Scholar] [CrossRef] [PubMed]
- Bemeur, C.; Cudalbu, C.; Dam, G.; Thrane, A.S.; Cooper, A.J.; Rose, C.F. Brain edema: A valid endpoint for measuring hepatic encephalopathy? Metab. Brain Dis. 2016, 31, 1249–1258. [Google Scholar] [CrossRef] [PubMed]
- Frontera, J.A.; Kalb, T. Neurological management of fulminant hepatic failure. Neurocrit Care 2011, 14, 318–327. [Google Scholar] [CrossRef] [PubMed]
- Munoz, S.J.; Robinson, M.; Northrup, B.; Bell, R.; Moritz, M.; Jarrell, B.; Martin, P.; Maddrey, W.C. Elevated intracranial pressure and computed tomography of the brain in fulminant hepatocellular failure. Hepatology 1991, 13, 209–212. [Google Scholar] [CrossRef] [PubMed]
- Stravitz, R.T.; Kramer, A.H.; Davern, T.; Shaikh, A.O.; Caldwell, S.H.; Mehta, R.L.; Blei, A.T.; Fontana, R.J.; McGuire, B.M.; Rossaro, L.; et al. Intensive care of patients with acute liver failure: Recommendations of the U.S. Acute Liver Failure Study Group. Crit. Care Med. 2007, 35, 2498–2508. [Google Scholar] [CrossRef] [PubMed]
- Bosoi, C.R.; Rose, C.F. Brain edema in acute liver failure and chronic liver disease: Similarities and differences. Neurochem. Int. 2013, 62, 446–457. [Google Scholar] [CrossRef] [PubMed]
- Unterberg, A.W.; Stover, J.; Kress, B.; Kiening, K.L. Edema and brain trauma. Neuroscience 2004, 129, 1021–1029. [Google Scholar] [CrossRef]
- Norenberg, M.D.; Rao, K.V.; Jayakumar, A.R. Mechanisms of ammonia-induced astrocyte swelling. Metab. Brain Dis. 2005, 20, 303–318. [Google Scholar] [CrossRef]
- Jayakumar, A.R.; Rao, K.V.; Murthy Ch, R.; Norenberg, M.D. Glutamine in the mechanism of ammonia-induced astrocyte swelling. Neurochem. Int. 2006, 48, 623–628. [Google Scholar] [CrossRef]
- Rama Rao, K.V.; Norenberg, M.D. Aquaporin-4 in hepatic encephalopathy. Metab. Brain Dis. 2007, 22, 265–275. [Google Scholar] [CrossRef]
- Thrane, A.S.; Rangroo Thrane, V.; Nedergaard, M. Drowning stars: Reassessing the role of astrocytes in brain edema. Trends Neurosci. 2014, 37, 620–628. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Wang, D.; Sun, J.; Wang, Y.; Wu, S.; Wang, J. Huperzine-A Improved Animal Behavior in Cuprizone-Induced Mouse Model by Alleviating Demyelination and Neuroinflammation. Int. J. Mol. Sci. 2022, 23, 16182. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.R.; Sun, M.Y.; Hang, W.; Xiao, Q.; Fan, H.J.; Jia, L.; Jin, X.M.; Zhang, B.; Xiao, B.G.; Ma, C.G.; et al. Wuzi Yanzong Pill relieves CPZ-induced demyelination by improving the microenvironment in the brain. Heliyon 2022, 8, e12277. [Google Scholar] [CrossRef]
- Bemeur, C.; Vaquero, J.; Desjardins, P.; Butterworth, R.F. N-acetylcysteine attenuates cerebral complications of non-acetaminophen-induced acute liver failure in mice: Antioxidant and anti-inflammatory mechanisms. Metab. Brain Dis. 2010, 25, 241–249. [Google Scholar] [CrossRef]
- Cheng, L.; Wang, X.; Ma, X.; Xu, H.; Yang, Y.; Zhang, D. Effect of dihydromyricetin on hepatic encephalopathy associated with acute hepatic failure in mice. Pharm. Biol. 2021, 59, 557–564. [Google Scholar] [CrossRef]
- Okano-Uchida, T.; Naruse, M.; Ikezawa, T.; Shibasaki, K.; Ishizaki, Y. Cerebellar neural stem cells differentiate into two distinct types of astrocytes in response to CNTF and BMP2. Neurosci. Lett. 2013, 552, 15–20. [Google Scholar] [CrossRef] [PubMed]
- Kadowaki, T.; Nakadate, K.; Sakakibara, S.; Hirata, K.; Ueda, S. Expression of Iba1 protein in microglial cells of zitter mutant rat. Neurosci. Lett. 2007, 411, 26–31. [Google Scholar] [CrossRef] [PubMed]
- Bosoi, C.R.; Rose, C.F. Identifying the direct effects of ammonia on the brain. Metab. Brain Dis. 2009, 24, 95–102. [Google Scholar] [CrossRef]
- Ge, P.S.; Runyon, B.A. Serum ammonia level for the evaluation of hepatic encephalopathy. JAMA 2014, 312, 643–644. [Google Scholar] [CrossRef]
- Ong, J.P.; Aggarwal, A.; Krieger, D.; Easley, K.A.; Karafa, M.T.; Van Lente, F.; Arroliga, A.C.; Mullen, K.D. Correlation between ammonia levels and the severity of hepatic encephalopathy. Am. J. Med. 2003, 114, 188–193. [Google Scholar] [CrossRef]
- Watanabe, M.; Ozaki, T.; Hirata, Y.; Yoshikuni, Y.; Kimura, K. Effect of lactitol (NS-4) on the increase in blood and brain ammonia concentration and on coma in newly developed rat models of hepatic coma. Nihon Yakurigaku Zasshi 1995, 105, 97–109. [Google Scholar] [CrossRef] [PubMed]
- Saheki, T.; Ohkubo, T.; Katsunuma, T. Regulation of urea synthesis in rat liver. Increase in the concentrations of ornithine and acetylglutamate in rat liver in response to urea synthesis stimulated by the injection of an ammonium salt. J. Biochem. 1978, 84, 1423–1430. [Google Scholar] [CrossRef] [PubMed]
- Azorin, I.; Minana, M.D.; Felipo, V.; Grisolia, S. A simple animal model of hyperammonemia. Hepatology 1989, 10, 311–314. [Google Scholar] [CrossRef] [PubMed]
- Bahrami, T.; Yaghmaei, P.; Yousofvand, N. The effects of Ibuprofen and 1, 8-cineol on anxiety and spatial memory in hyperammonemic rats. Metab. Brain Dis. 2022, 38, 613–620. [Google Scholar] [CrossRef]
- Kosenko, E.; Tikhonova, L.; Alilova, G.; Montoliu, C. A Look into Liver Mitochondrial Dysfunction as a Hallmark in Progression of Brain Energy Crisis and Development of Neurologic Symptoms in Hepatic Encephalopathy. J. Clin. Med. 2020, 9, 2259. [Google Scholar] [CrossRef]
- Witt, A.M.; Larsen, F.S.; Bjerring, P.N. Accumulation of lactate in the rat brain during hyperammonaemia is not associated with impaired mitochondrial respiratory capacity. Metab. Brain Dis. 2017, 32, 461–470. [Google Scholar] [CrossRef]
- Garcia-Lezana, T.; Oria, M.; Romero-Gimenez, J.; Bove, J.; Vila, M.; Genesca, J.; Chavarria, L.; Cordoba, J. Cerebellar neurodegeneration in a new rat model of episodic hepatic encephalopathy. J. Cereb. Blood Flow. Metab. 2017, 37, 927–937. [Google Scholar] [CrossRef]
- Cittolin-Santos, G.F.; de Assis, A.M.; Guazzelli, P.A.; Paniz, L.G.; da Silva, J.S.; Calcagnotto, M.E.; Hansel, G.; Zenki, K.C.; Kalinine, E.; Duarte, M.M.; et al. Guanosine Exerts Neuroprotective Effect in an Experimental Model of Acute Ammonia Intoxication. Mol. Neurobiol. 2017, 54, 3137–3148. [Google Scholar] [CrossRef]
- Nakadate, K.; Kamata, S. Severe Acute Hepatic Dysfunction Induced by Ammonium Acetate Treatment Results in Choroid Plexus Swelling and Ventricle Enlargement in the Brain. Int. J. Mol. Sci. 2022, 23, 2010. [Google Scholar] [CrossRef]
- Wright, G.; Soper, R.; Brooks, H.F.; Stadlbauer, V.; Vairappan, B.; Davies, N.A.; Andreola, F.; Hodges, S.; Moss, R.F.; Davies, D.C.; et al. Role of aquaporin-4 in the development of brain oedema in liver failure. J. Hepatol. 2010, 53, 91–97. [Google Scholar] [CrossRef]
- Rama Rao, K.V.; Verkman, A.S.; Curtis, K.M.; Norenberg, M.D. Aquaporin-4 deletion in mice reduces encephalopathy and brain edema in experimental acute liver failure. Neurobiol. Dis. 2014, 63, 222–228. [Google Scholar] [CrossRef] [PubMed]
- Rama Rao, K.V.; Jayakumar, A.R.; Tong, X.; Curtis, K.M.; Norenberg, M.D. Brain aquaporin-4 in experimental acute liver failure. J. Neuropathol. Exp. Neurol. 2010, 69, 869–879. [Google Scholar] [CrossRef] [PubMed]
- Eefsen, M.; Jelnes, P.; Schmidt, L.E.; Vainer, B.; Bisgaard, H.C.; Larsen, F.S. Brain expression of the water channels aquaporin-1 and -4 in mice with acute liver injury, hyperammonemia and brain edema. Metab. Brain Dis. 2010, 25, 315–323. [Google Scholar] [CrossRef] [PubMed]
- Badaut, J.; Lasbennes, F.; Magistretti, P.J.; Regli, L. Aquaporins in brain: Distribution, physiology, and pathophysiology. J. Cereb. Blood Flow. Metab. 2002, 22, 367–378. [Google Scholar] [CrossRef] [PubMed]
- Pan, C.F.; Zhu, S.M.; Zheng, Y.Y. Ammonia induces upregulation of aquaporin-4 in neocortical astrocytes of rats through the p38 mitogen-activated protein kinase pathway. Chin. Med. J. 2010, 123, 1888–1892. [Google Scholar] [PubMed]
- Bonomini, F.; Rezzani, R. Aquaporin and blood brain barrier. Curr. Neuropharmacol. 2010, 8, 92–96. [Google Scholar] [CrossRef] [PubMed]
- Nagelhus, E.A.; Horio, Y.; Inanobe, A.; Fujita, A.; Haug, F.M.; Nielsen, S.; Kurachi, Y.; Ottersen, O.P. Immunogold evidence suggests that coupling of K+ siphoning and water transport in rat retinal Muller cells is mediated by a coenrichment of Kir4.1 and AQP4 in specific membrane domains. Glia 1999, 26, 47–54. [Google Scholar] [CrossRef]
- Saadoun, S.; Papadopoulos, M.C.; Davies, D.C.; Krishna, S.; Bell, B.A. Aquaporin-4 expression is increased in oedematous human brain tumours. J. Neurol. Neurosurg. Psychiatry 2002, 72, 262–265. [Google Scholar] [CrossRef]
- Assentoft, M.; Kaptan, S.; Schneider, H.P.; Deitmer, J.W.; de Groot, B.L.; MacAulay, N. Aquaporin 4 as a NH3 Channel. J. Biol. Chem. 2016, 291, 19184–19195. [Google Scholar] [CrossRef]
- Molina-Berenguer, M.; Vila-Julia, F.; Perez-Ramos, S.; Salcedo-Allende, M.T.; Camara, Y.; Torres-Torronteras, J.; Marti, R. Dysfunctional mitochondrial translation and combined oxidative phosphorylation deficiency in a mouse model of hepatoencephalopathy due to Gfm1 mutations. FASEB J. 2022, 36, e22091. [Google Scholar] [CrossRef]
- Kosenko, E.; Tikhonova, L.; Alilova, G.; Montoliu, C. Is NMDA-Receptor-Mediated Oxidative Stress in Mitochondria of Peripheral Tissues the Essential Factor in the Pathogenesis of Hepatic Encephalopathy? J. Clin. Med. 2022, 11, 827. [Google Scholar] [CrossRef] [PubMed]
- Zoratti, M.; Szabo, I. The mitochondrial permeability transition. Biochim. Biophys. Acta 1995, 1241, 139–176. [Google Scholar] [CrossRef] [PubMed]
- Zorov, D.B.; Filburn, C.R.; Klotz, L.O.; Zweier, J.L.; Sollott, S.J. Reactive oxygen species (ROS)-induced ROS release: A new phenomenon accompanying induction of the mitochondrial permeability transition in cardiac myocytes. J. Exp. Med. 2000, 192, 1001–1014. [Google Scholar] [CrossRef] [PubMed]
- Votyakova, T.V.; Reynolds, I.J. Ca2+-induced permeabilization promotes free radical release from rat brain mitochondria with partially inhibited complex I. J. Neurochem. 2005, 93, 526–537. [Google Scholar] [CrossRef] [PubMed]
- Bai, G.; Rama Rao, K.V.; Murthy, C.R.; Panickar, K.S.; Jayakumar, A.R.; Norenberg, M.D. Ammonia induces the mitochondrial permeability transition in primary cultures of rat astrocytes. J. Neurosci. Res. 2001, 66, 981–991. [Google Scholar] [CrossRef]
- Rama Rao, K.V.; Reddy, P.V.; Tong, X.; Norenberg, M.D. Brain edema in acute liver failure: Inhibition by L-histidine. Am. J. Pathol. 2010, 176, 1400–1408. [Google Scholar] [CrossRef]
- Niknahad, H.; Jamshidzadeh, A.; Heidari, R.; Zarei, M.; Ommati, M.M. Ammonia-induced mitochondrial dysfunction and energy metabolism disturbances in isolated brain and liver mitochondria, and the effect of taurine administration: Relevance to hepatic encephalopathy treatment. Clin. Exp. Hepatol. 2017, 3, 141–151. [Google Scholar] [CrossRef]
- Rackayova, V.; Braissant, O.; McLin, V.A.; Berset, C.; Lanz, B.; Cudalbu, C. 1H and 31P magnetic resonance spectroscopy in a rat model of chronic hepatic encephalopathy: In vivo longitudinal measurements of brain energy metabolism. Metab. Brain Dis. 2016, 31, 1303–1314. [Google Scholar] [CrossRef]
- Rama Rao, K.V.; Norenberg, M.D. Brain energy metabolism and mitochondrial dysfunction in acute and chronic hepatic encephalopathy. Neurochem. Int. 2012, 60, 697–706. [Google Scholar] [CrossRef]
- Tabata, H. Diverse subtypes of astrocytes and their development during corticogenesis. Front. Neurosci. 2015, 9, 114. [Google Scholar] [CrossRef]
- Liu, H.T.; Tashmukhamedov, B.A.; Inoue, H.; Okada, Y.; Sabirov, R.Z. Roles of two types of anion channels in glutamate release from mouse astrocytes under ischemic or osmotic stress. Glia 2006, 54, 343–357. [Google Scholar] [CrossRef] [PubMed]
- Larionova, N.P.; Reutov, V.P.; Samosudova, N.V.; Chailakhian, L.M. Two types of reactions of glial cells to the stimulation of parallel fibers in the presence of an NO-generating compound as a morphological expression of the physiological activity of two types of astrocytes in the frog cerebellum. Dokl. Biol. Sci. 2005, 401, 95–99. [Google Scholar] [CrossRef] [PubMed]
- Raff, M.C.; Abney, E.R.; Cohen, J.; Lindsay, R.; Noble, M. Two types of astrocytes in cultures of developing rat white matter: Differences in morphology, surface gangliosides, and growth characteristics. J. Neurosci. 1983, 3, 1289–1300. [Google Scholar] [CrossRef]
- Belanger, M.; Desjardins, P.; Chatauret, N.; Butterworth, R.F. Loss of expression of glial fibrillary acidic protein in acute hyperammonemia. Neurochem. Int. 2002, 41, 155–160. [Google Scholar] [CrossRef] [PubMed]
- Park, C.H.; Joo, Y.E.; Kim, H.S.; Choi, S.K.; Rew, J.S.; Kim, S.J. Neostigmine for the treatment of acute hepatic encephalopathy with acute intestinal pseudo-obstruction in a cirrhotic patient. J. Korean Med. Sci. 2005, 20, 150–152. [Google Scholar] [CrossRef] [PubMed]
- Plauth, M.; Merli, M.; Kondrup, J.; Weimann, A.; Ferenci, P.; Muller, M.J.; Group, E.C. ESPEN guidelines for nutrition in liver disease and transplantation. Clin. Nutr. 1997, 16, 43–55. [Google Scholar] [CrossRef] [PubMed]
- Trovato, G.M.; Catalano, D.; Carpinteri, G.; Runcio, N.; Mazzone, O. Effects of lactitol [correction of lactilol] on hepatic encephalopathy and plasma amino-acid imbalance. Recenti Prog. Med. 1995, 86, 299–303. [Google Scholar]
- Calvey, H.; Davis, M.; Williams, R. Controlled trial of nutritional supplementation, with and without branched chain amino acid enrichment, in treatment of acute alcoholic hepatitis. J. Hepatol. 1985, 1, 141–151. [Google Scholar] [CrossRef]
- Tranah, T.H.; Ballester, M.P.; Carbonell-Asins, J.A.; Ampuero, J.; Alexandrino, G.; Caracostea, A.; Sanchez-Torrijos, Y.; Thomsen, K.L.; Kerbert, A.J.C.; Capilla-Lozano, M.; et al. Plasma ammonia levels predict hospitalisation with liver-related complications and mortality in clinically stable outpatients with cirrhosis. J. Hepatol. 2022, 77, 1554–1563. [Google Scholar] [CrossRef]
- Lipe, D.N.; Hoxha, B.; Sahai, S.K. Sunitinib-associated hyperammonemic encephalopathy. Am. J. Emerg. Med. 2021, 46, 282–283. [Google Scholar] [CrossRef]
- Kim, G.N.; Ho, S.; Saulino, D.; Liu, X. Severe Protein-Calorie Malnutrition-Associated Hepatic Steatosis in a Woman Who Had Roux-en-Y Gastric Bypass for Morbid Obesity Thirteen Years Ago. Gastroenterol. Res. 2021, 14, 129–137. [Google Scholar] [CrossRef] [PubMed]
- Hey, P.; Gow, P.; Testro, A.G.; Apostolov, R.; Chapman, B.; Sinclair, M. Nutraceuticals for the treatment of sarcopenia in chronic liver disease. Clin. Nutr. ESPEN 2021, 41, 13–22. [Google Scholar] [CrossRef] [PubMed]
- Hashiguchi, M.; Tamai, T.; Kiyama, K.; Ooi, T.; Yamauchi, T.; Kamikihara, Y.; Kodama, T.; Kawahira, M.; Nakamura, Y.; Yano, H.; et al. Two cases of liver cirrhosis with hyperammonemic encephalopathy caused by urease-producing bacteria in the urinary tract. Clin. J. Gastroenterol. 2021, 14, 650–655. [Google Scholar] [CrossRef] [PubMed]
- Di Federico, A.; Nuvola, G.; Sisi, M.; Lenzi, B.; Nobili, E.; Campana, D. Hyperammonemic encephalopathy during XELOX regimen. Is it capecitabine or oxaliplatin responsible? Anticancer. Drugs 2020, 31, 1103–1105. [Google Scholar] [CrossRef]
- Nakadate, K.; Imamura, K.; Watanabe, Y. Cellular and subcellular localization of alpha-1 adrenoceptors in the rat visual cortex. Neuroscience 2006, 141, 1783–1792. [Google Scholar] [CrossRef]
- Satoh, R.; Kawakami, K.; Nakadate, K. Effects of Smart Drugs on Cholinergic System and Non-Neuronal Acetylcholine in the Mouse Hippocampus: Histopathological Approach. J. Clin. Med. 2022, 11, 3310. [Google Scholar] [CrossRef]
- Nakadate, K.; Kawakami, K. Distribution and Localization of Mahogunin Ring Finger 1 in the Mouse Central Nervous System. Int. J. Mol. Sci. 2022, 23, 8956. [Google Scholar] [CrossRef]
- Nakadate, K.; Motojima, K.; Tanaka-Nakadate, S. Dilatation of sinusoidal capillary and swelling of sinusoidal fenestration in obesity: An ultrastructural study. Ultrastruct. Pathol. 2015, 39, 30–37. [Google Scholar] [CrossRef]
- Marmarou, A.; Poll, W.; Shulman, K.; Bhagavan, H. A simple gravimetric technique for measurement of cerebral edema. J. Neurosurg. 1978, 49, 530–537. [Google Scholar] [CrossRef]
- Nakadate, K. Developmental changes in the flotillin-1 expression pattern of the rat visual cortex. Neuroscience 2015, 292, 101–111. [Google Scholar] [CrossRef]







| TP (g/dL) | ALB (g/dL) | AST (IU/L) | ALT (IU/L) | ALP (IU/L) | |
|---|---|---|---|---|---|
| Control | 3.24 ± 0.29 | 1.49 ± 0.28 | 214.31 ± 3.94 | 10.83 ± 4.38 | 141.38 ± 18.15 |
| 3H | 2.47 ± 0.21 * | 1.69 ± 0.36 | 321.12 ± 28.47 * | 18.68 ± 3.12 | 172.09 ± 10.29 |
| 24H | 3.39 ± 0.28 | 1.74 ± 0.31 | 331.29 ± 29.11 * | 53.22 ± 3.19 * | 166.21 ± 14.34 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nakadate, K.; Sono, C.; Mita, H.; Itakura, Y.; Kawakami, K. Severe Acute Liver Dysfunction Induces Delayed Hepatocyte Swelling and Cytoplasmic Vacuolization, and Delayed Cortical Neuronal Cell Death. Int. J. Mol. Sci. 2023, 24, 7351. https://doi.org/10.3390/ijms24087351
Nakadate K, Sono C, Mita H, Itakura Y, Kawakami K. Severe Acute Liver Dysfunction Induces Delayed Hepatocyte Swelling and Cytoplasmic Vacuolization, and Delayed Cortical Neuronal Cell Death. International Journal of Molecular Sciences. 2023; 24(8):7351. https://doi.org/10.3390/ijms24087351
Chicago/Turabian StyleNakadate, Kazuhiko, Chiaki Sono, Homura Mita, Yuki Itakura, and Kiyoharu Kawakami. 2023. "Severe Acute Liver Dysfunction Induces Delayed Hepatocyte Swelling and Cytoplasmic Vacuolization, and Delayed Cortical Neuronal Cell Death" International Journal of Molecular Sciences 24, no. 8: 7351. https://doi.org/10.3390/ijms24087351






