Extracellular Vesicles Secreted by Pre-Hatching Bovine Embryos Produced In Vitro and In Vivo Alter the Expression of IFNtau-Stimulated Genes in Bovine Endometrial Cells
Abstract
:1. Introduction
2. Results
2.1. Characterization of Nanoparticles Collected from Embryo Culture Media
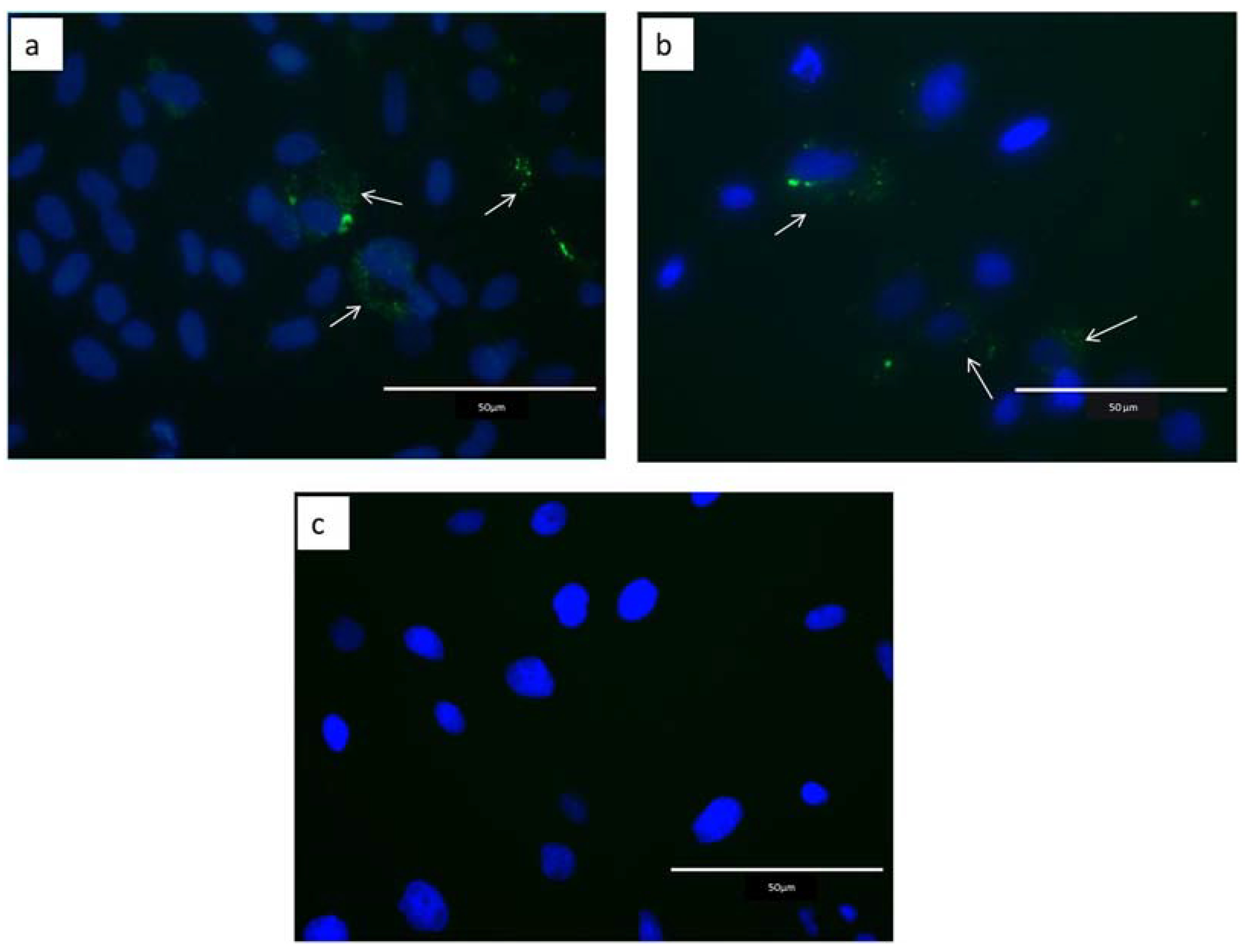
2.2. Internalization of Extracellular Vesicles
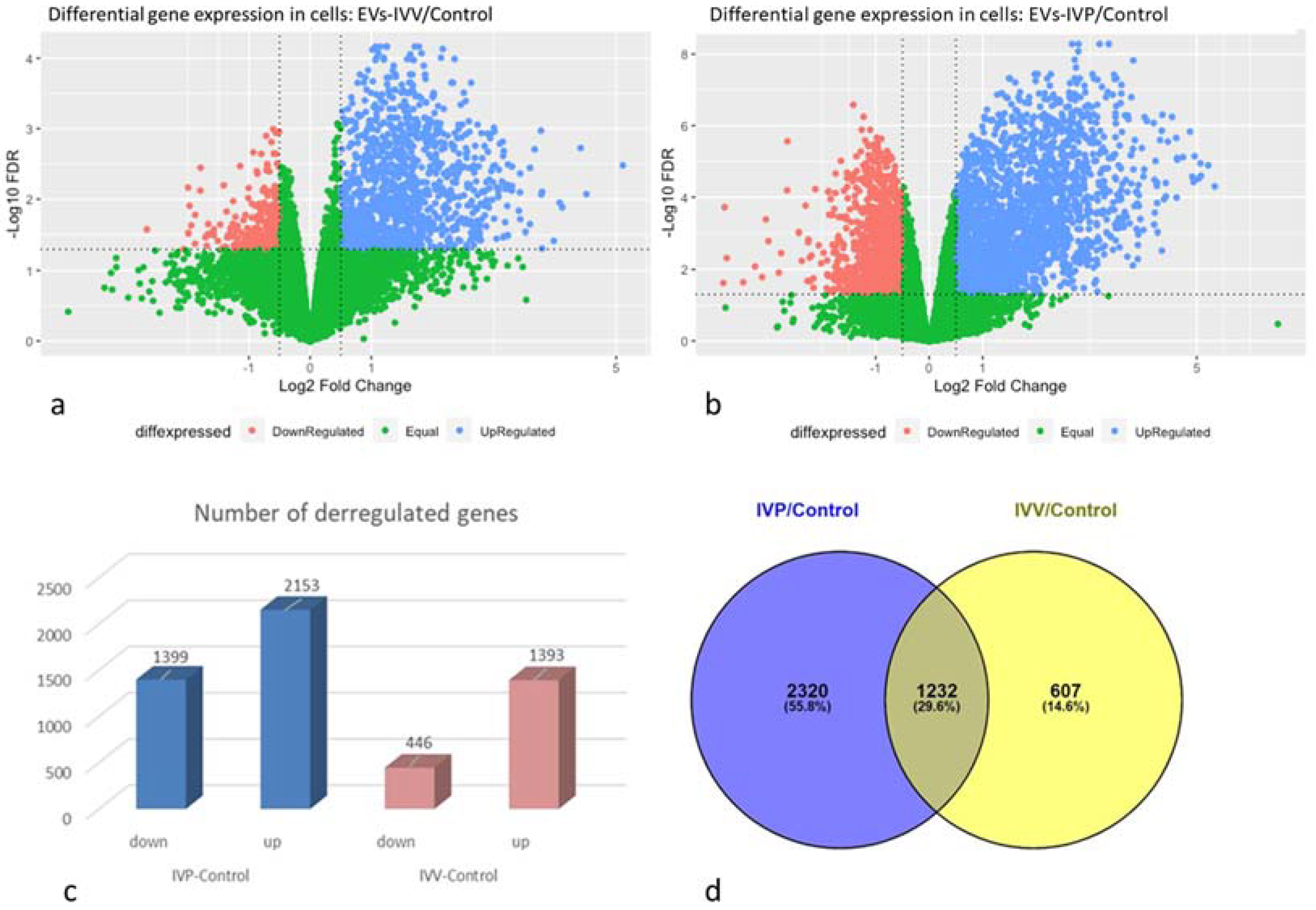
2.3. Transcriptomic Analysis in Endometrial Epithelial Cells
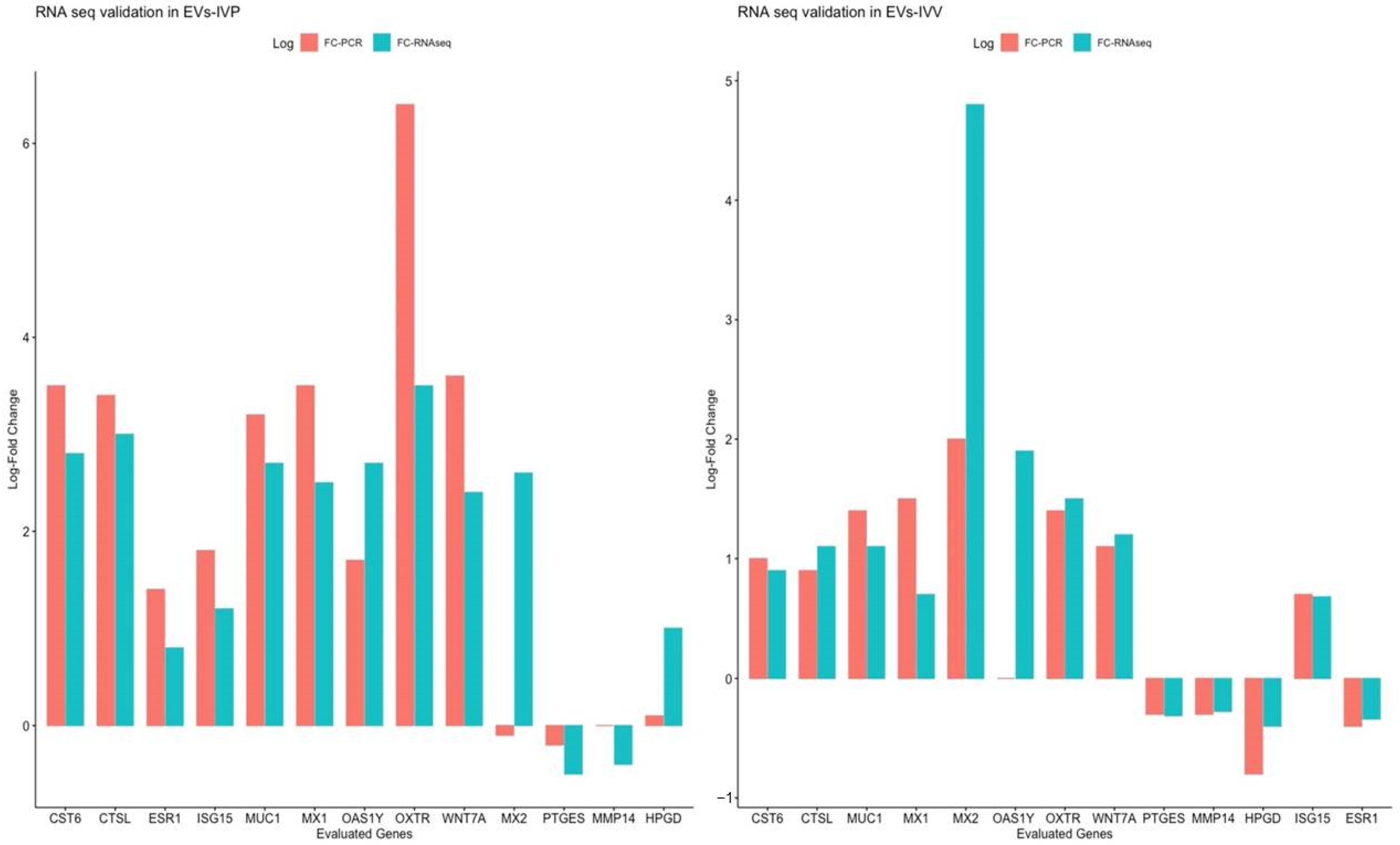
2.4. Validation of RNA Seq through Quantitative Reverse Transcription PCR Analysis
2.5. Gene Ontology Analysis
3. Discussion
4. Materials and Methods
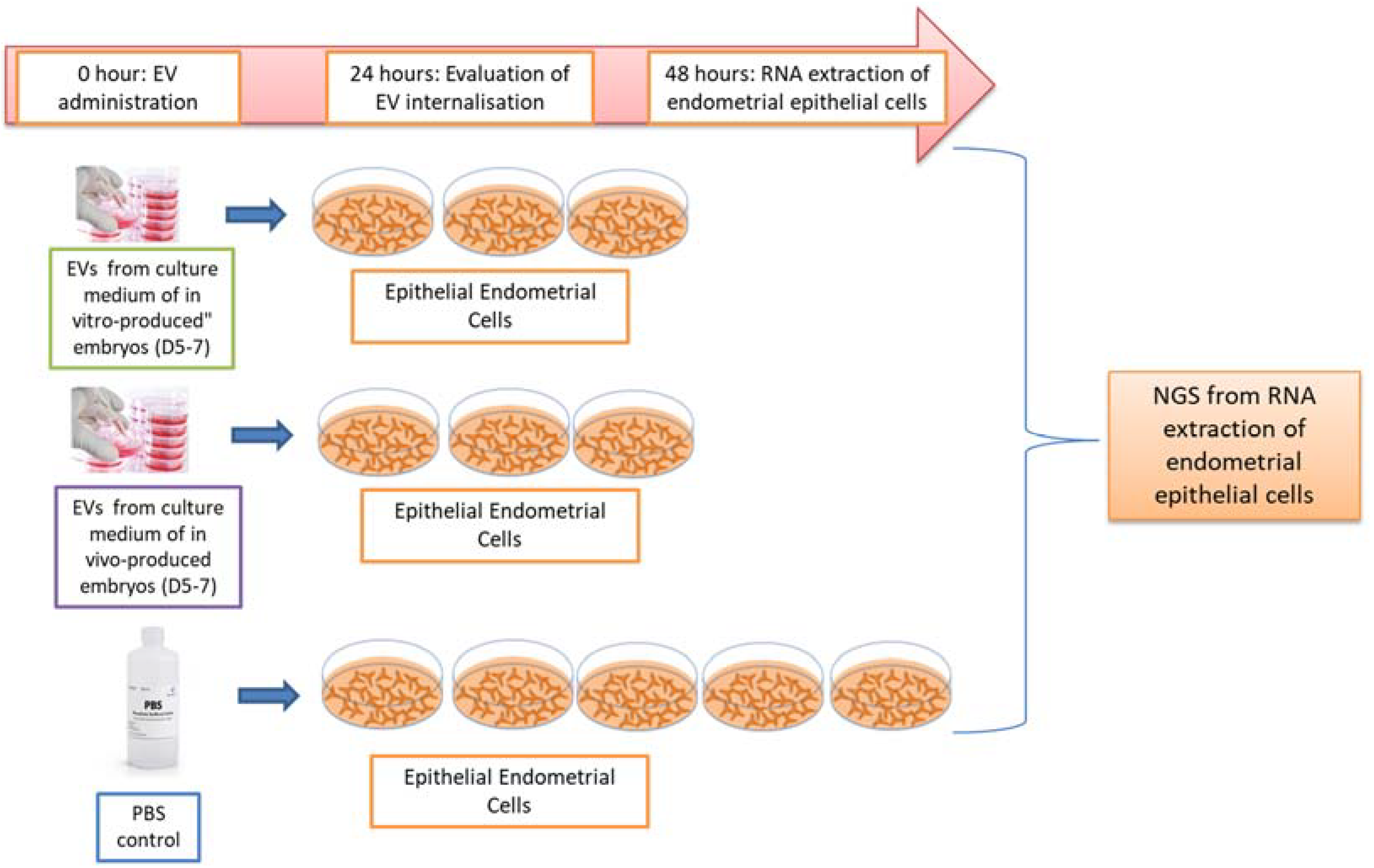
4.1. Experimental Design
4.2. In Vitro Embryo Production
4.3. In Vivo Embryo Production
4.4. EV Isolation from Embryo Culture Medium
4.5. EVs Characterization
4.5.1. Nanoparticle Tracking Analysis
4.5.2. Analysis of Surface Markers by Flow Cytometry
4.5.3. Transmission Electron Microscopy Analysis
4.6. Endometrial Bovine Cells Isolation and Culture
4.7. EVs Internalization by Epithelial Endometrial Cells
4.8. Endometrial Epithelial Cell mRNA Sequencing
4.9. Bioinformatic Analysis
4.10. Quantitative Reverse Transcription PCR Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Forde, N.; Lonergan, P. Interferon-tau and fertility in ruminants. Reproduction 2017, 154, F33–F43. [Google Scholar] [CrossRef]
- Lopera-Vásquez, R.; Hamdi, M.; Fernandez-Fuertes, B.; Maillo, V.; Beltrán-Breña, P.; Calle, A.; Redruello, A.; López-Martín, S.; Gutierrez-Adán, A.; Yañez-Mó, M.; et al. Extracellular Vesicles from BOEC in In Vitro Embryo Development and Quality. PLoS ONE 2016, 11, e0148083. [Google Scholar] [CrossRef]
- Maillo, V.; Lopera-Vasquez, R.; Hamdi, M.; Gutierrez-Adan, A.; Lonergan, P.; Rizos, D. Maternal-embryo interaction in the bovine oviduct: Evidence from in vivo and in vitro studies. Theriogenology 2016, 86, 443–450. [Google Scholar] [CrossRef] [PubMed]
- Hernandez-Ledezma, J.J.; Sikes, J.D.; Murphy, C.N.; Watson, A.J.; Schultz, G.A.; Roberts, R.M. Expression of bovine trophoblast interferon in conceptuses derived by in vitro techniques. Biol. Reprod. 1992, 47, 374–380. [Google Scholar] [CrossRef] [PubMed]
- Hernandez-Ledezma, J.J.; Mathialagan, N.; Villanueva, C.; Sikes, J.D.; Roberts, R.M. Expression of bovine trophoblast interferons by in vitro-derived blastocysts is correlated with their morphological quality and stage of development. Mol. Reprod. Dev. 1993, 36, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Bazer, F.W.; Spencer, T.E.; Ott, T.L. Interferon tau: A novel pregnancy recognition signal. Am. J. Reprod. Immunol. 1997, 37, 412–420. [Google Scholar] [CrossRef]
- Johnson, K.M.; Alvarez, X.; Borkhsenious, O.N.; Kubisch, H.M. Nuclear and cytoplasmic localization of interferon-tau in in vitro-produced bovine blastocysts. Reprod. Nutr. Dev. 2006, 46, 97–104. [Google Scholar] [CrossRef]
- Sponchiado, M.; Gomes, N.S.; Fontes, P.K.; Martins, T.; del Collado, M.; Pastore, A.D.A.; Pugliesi, G.; Nogueira, M.F.G.; Binelli, M. Pre-hatching embryo-dependent and -independent programming of endometrial function in cattle. PLoS ONE 2017, 12, e0175954. [Google Scholar] [CrossRef]
- Talukder, A.K.; Yousef, M.S.; Rashid, M.B.; Awai, K.; Acosta, T.J.; Shimizu, T.; Okuda, K.; Shimada, M.; Imakawa, K.; Miyamoto, A. Bovine embryo induces an anti-inflammatory response in uterine epithelial cells and immune cells in vitro: Possible involvement of interferon tau as an intermediator. J. Reprod. Dev. 2017, 63, 425–434. [Google Scholar] [CrossRef]
- Passaro, C.; Tutt, D.; Mathew, D.J.; Sanchez, J.M.; Browne, J.A.; Boe-Hansen, G.B.; Fair, T.; Lonergan, P. Blastocyst-induced changes in the bovine endometrial transcriptome. Reproduction 2018, 156, 219–229. [Google Scholar] [CrossRef]
- Sponchiado, M.; Gonella-Diaza, A.; Rocha, C.; Turco, E.; Pugliesi, G.; Leroy, j.; Binelli, M. The pre-hatching bovine embryo transforms the uterine luminal metabolite composition in vivo. Sci. Rep. 2019, 9, 8354. [Google Scholar] [CrossRef]
- Sponchiado, M.; Marei, W.F.A.; Beemster, G.T.S.; Bols, P.E.J.; Binelli, M.; Leroy, J. Molecular interactions at the bovine embryo-endometrial epithelium interface. Reproduction 2020, 160, 887–903. [Google Scholar] [CrossRef]
- Egashira, M.; Hirota, Y. Uterine receptivity and embryo-uterine interactions in embryo implantation: Lessons from mice. Reprod. Med. Biol. 2013, 12, 127–132. [Google Scholar] [CrossRef]
- Georgiou, A.S.; Sostaric, E.; Wong, C.H.; Snijders, A.P.; Wright, P.C.; Moore, H.D.; Fazeli, A. Gametes alter the oviductal secretory proteome. Mol. Cell. Proteom. 2005, 4, 1785–1796. [Google Scholar] [CrossRef]
- Almiñana, C.; Caballero, I.; Heath, P.; Maleki, S.; Parrilla, I.; Cuello, C.; Gil, M.; Vazquez, J.; Vazquez, J.; Roca, J.; et al. The battle of the sexes starts in the oviduct: Modulation of oviductal transcriptome by X and Y-bearing spermatozoa. BMC Genom. 2014, 15, 293. [Google Scholar] [CrossRef]
- Maillo, V.; Gaora, P.; Forde, N.; Besenfelder, U.; Havlicek, V.; Burns, G.W.; Spencer, T.E.; Gutierrez-Adan, A.; Lonergan, P.; Rizos, D. Oviduct-Embryo Interactions in Cattle: Two-Way Traffic or a One-Way Street? Biol. Reprod. 2015, 92, 144. [Google Scholar] [CrossRef]
- de Ávila, A.; da Silveira, J.C. Role of extracellular vesicles during oocyte maturation and early embryo development. Reprod. Fertil. Dev. 2019, 32, 56–64. [Google Scholar] [CrossRef]
- Bauersachs, S.; Mermillod, P.; Almiñana, C. The Oviductal Extracellular Vesicles’ RNA Cargo Regulates the Bovine Embryonic Transcriptome. Int. J. Mol. Sci. 2020, 21, 1303. [Google Scholar] [CrossRef]
- Banliat, C.; Le Bourhis, D.; Bernardi, O.; Tomas, D.; Labas, V.; Salvetti, P.; Guyonnet, B.; Mermillod, P.; Saint-Dizier, M. Oviduct Fluid Extracellular Vesicles Change the Phospholipid Composition of Bovine Embryos Developed In Vitro. Int. J. Mol. Sci. 2020, 21, 5326. [Google Scholar] [CrossRef] [PubMed]
- Taylor, D.D.; Gercel-Taylor, C. MicroRNA signatures of tumor-derived exosomes as diagnostic biomarkers of ovarian cancer. Gynecol. Oncol. 2008, 110, 13–21. [Google Scholar] [CrossRef] [PubMed]
- Ng, Y.H.; Rome, S.; Jalabert, A.; Forterre, A.; Singh, H.; Hincks, C.L.; Salamonsen, L.A. Endometrial exosomes/microvesicles in the uterine microenvironment: A new paradigm for embryo-endometrial cross talk at implantation. PLoS ONE 2013, 8, e58502. [Google Scholar] [CrossRef]
- Lee, H.; Zhang, D.; Zhu, Z.; Dela Cruz, C.S.; Jin, Y. Epithelial cell-derived microvesicles activate macrophages and promote inflammation via microvesicle-containing microRNAs. Sci. Rep. 2016, 6, 35250. [Google Scholar] [CrossRef] [PubMed]
- Mittelbrunn, M.; Gutiérrez-Vázquez, C.; Villarroya-Beltri, C.; González, S.; Sánchez-Cabo, F.; González, M.; Bernad, A.; Sánchez-Madrid, F. Unidirectional transfer of microRNA-loaded exosomes from T cells to antigen-presenting cells. Nat. Commun. 2011, 2, 282. [Google Scholar] [CrossRef] [PubMed]
- Valadi, H.; Ekström, K.; Bossios, A.; Sjöstrand, M.; Lee, J.J.; Lötvall, J.O. Exosome-mediated transfer of mRNAs and microRNAs is a novel mechanism of genetic exchange between cells. Nat. Cell Biol. 2007, 9, 654–659. [Google Scholar] [CrossRef] [PubMed]
- Bellingham, S.A.; Guo, B.B.; Coleman, B.M.; Hill, A.F. Exosomes: Vehicles for the transfer of toxic proteins associated with neurodegenerative diseases? Front. Physiol. 2012, 3, 124. [Google Scholar] [CrossRef]
- Mazzarella, R.; Bastos, N.M.; Bridi, A.; Del Collado, M.; Andrade, G.M.; Pinzon, J.; Prado, C.M.; Silva, L.A.; Meirelles, F.V.; Pugliesi, G.; et al. Changes in Oviductal Cells and Small Extracellular Vesicles miRNAs in Pregnant Cows. Front. Vet. Sci. 2021, 8, 639752. [Google Scholar] [CrossRef]
- Almiñana, C.; Corbin, E.; Tsikis, G.; Alcântara-Neto, A.S.; Labas, V.; Reynaud, K.; Galio, L.; Uzbekov, R.; Garanina, A.S.; Druart, X.; et al. Oviduct extracellular vesicles protein content and their role during oviduct-embryo cross-talk. Reproduction 2017, 154, 153–168. [Google Scholar] [CrossRef]
- Qu, P.; Zhao, Y.; Wang, R.; Zhang, Y.; Li, L.; Fan, J.; Liu, E. Extracellular vesicles derived from donor oviduct fluid improved birth rates after embryo transfer in mice. Reprod. Fertil. Dev. 2019, 31, 324–332. [Google Scholar] [CrossRef]
- Godakumara, K.; Ord, J.; Lättekivi, F.; Dissanayake, K.; Viil, J.; Boggavarapu, N.R.; Faridani, O.R.; Jääger, K.; Velthut-Meikas, A.; Jaakma, Ü.; et al. Trophoblast derived extracellular vesicles specifically alter the transcriptome of endometrial cells and may constitute a critical component of embryo-maternal communication. Reprod. Biol. Endocrinol. 2021, 19, 115. [Google Scholar] [CrossRef]
- Burns, G.W.; Brooks, K.E.; Spencer, T.E. Extracellular Vesicles Originate from the Conceptus and Uterus during Early Pregnancy in Sheep. Biol. Reprod. 2016, 94, 56. [Google Scholar] [CrossRef]
- Nakamura, K.; Kusama, K.; Bai, R.; Sakurai, T.; Isuzugawa, K.; Godkin, J.D.; Suda, Y.; Imakawa, K. Induction of IFNT-Stimulated Genes by Conceptus-Derived Exosomes during the Attachment Period. PLoS ONE 2016, 11, e0158278. [Google Scholar] [CrossRef]
- Bridi, A. Extracellular Vesicles Secreted by Bovine Embryos Produced In Vivo and In Vitro: miRNAs Content and Molecular Effects in Endometrial and Luteal Tissues; Universidade de São Paulo (USP): São Paulo, Brasil, 2021. [Google Scholar]
- Jorge Alberto, N.; Daniel, T.; Rene, L.H.; Jacques, M. Relationship Between IFN-t Production by Bovines Embryos Derived Ex Vivo and Completely Produced In Vitro. In Artificial Insemination in Farm Animals; Milad, M., Ed.; IntechOpen: Rijeka, Croatia, 2011; p. 15. [Google Scholar]
- Stojkovic, M.; Büttner, M.; Zakhartchenko, V.; Riedl, J.; Reichenbach, H.D.; Wenigerkind, H.; Brem, G.; Wolf, E. Secretion of interferon-tau by bovine embryos in long-term culture: Comparison of in vivo derived, in vitro produced, nuclear transfer and demi-embryos. Anim. Reprod. Sci. 1999, 55, 151–162. [Google Scholar] [CrossRef]
- Yao, N.; Wan, P.C.; Hao, Z.D.; Gao, F.F.; Yang, L.; Cui, M.S.; Wu, Y.; Liu, J.H.; Liu, S.; Chen, H.; et al. Expression of interferon-tau mRNA in bovine embryos derived from different procedures. Reprod. Domest. Anim. 2009, 44, 132–139. [Google Scholar] [CrossRef]
- Bauersachs, S.; Ulbrich, S.E.; Zakhartchenko, V.; Minten, M.; Reichenbach, M.; Reichenbach, H.D.; Blum, H.; Spencer, T.E.; Wolf, E. The endometrium responds differently to cloned versus fertilized embryos. Proc. Natl. Acad. Sci. USA 2009, 106, 5681–5686. [Google Scholar] [CrossRef]
- Mansouri-Attia, N.; Sandra, O.; Aubert, J.; Degrelle, S.; Everts, R.E.; Giraud-Delville, C.; Heyman, Y.; Galio, L.; Hue, I.; Yang, X.; et al. Endometrium as an early sensor of in vitro embryo manipulation technologies. Proc. Natl. Acad. Sci. USA 2009, 106, 5687–5692. [Google Scholar] [CrossRef]
- Mathew, D.J.; Sánchez, J.M.; Passaro, C.; Charpigny, G.; Behura, S.K.; Spencer, T.E.; Lonergan, P. Interferon tau-dependent and independent effects of the bovine conceptus on the endometrial transcriptome. Biol. Reprod. 2019, 100, 365–380. [Google Scholar] [CrossRef]
- Yang, Q.; Fu, W.; Wang, Y.; Miao, K.; Zhao, H.; Wang, R.; Guo, M.; Wang, Z.; Tian, J.; An, L. The proteome of IVF-induced aberrant embryo-maternal crosstalk by implantation stage in ewes. J. Anim. Sci. Biotechnol. 2020, 11, 7. [Google Scholar] [CrossRef]
- Théry, C.; Witwer, K.W.; Aikawa, E.; Alcaraz, M.J.; Anderson, J.D.; Andriantsitohaina, R.; Antoniou, A.; Arab, T.; Archer, F.; Atkin-Smith, G.K. Minimal information for studies of extracellular vesicles 2018 (MISEV2018): A position statement of the International Society for Extracellular Vesicles and update of the MISEV2014 guidelines. J. Extracell. Vesicles 2018, 7, 1535750. [Google Scholar] [CrossRef]
- Chaney, H.L.; Grose, L.F.; Charpigny, G.; Behura, S.K.; Sheldon, I.M.; Cronin, J.G.; Lonergan, P.; Spencer, T.E.; Mathew, D.J. Conceptus-induced, interferon tau-dependent gene expression in bovine endometrial epithelial and stromal cells†. Biol. Reprod. 2020, 104, 669–683. [Google Scholar] [CrossRef]
- Dorniak, P.; Bazer, F.W.; Wu, G.; Spencer, T.E. Conceptus-derived prostaglandins regulate endometrial function in sheep. Biol. Reprod. 2012, 87, 1–7. [Google Scholar] [CrossRef]
- Dorniak, P.; Welsh, T.H., Jr.; Bazer, F.W.; Spencer, T.E. Endometrial HSD11B1 and cortisol regeneration in the ovine uterus: Effects of pregnancy, interferon tau, and prostaglandins. Biol. Reprod. 2012, 86, 124. [Google Scholar] [CrossRef] [PubMed]
- Song, G.; Satterfield, M.C.; Kim, J.; Bazer, F.W.; Spencer, T.E. Gastrin-releasing peptide (GRP) in the ovine uterus: Regulation by interferon tau and progesterone. Biol. Reprod. 2008, 79, 376–386. [Google Scholar] [CrossRef] [PubMed]
- Schmaltz-Panneau, B.; Cordova, A.; Dhorne-Pollet, S.; Hennequet-Antier, C.; Uzbekova, S.; Martinot, E.; Doret, S.; Martin, P.; Mermillod, P.; Locatelli, Y. Early bovine embryos regulate oviduct epithelial cell gene expression during in vitro co-culture. Anim. Reprod. Sci. 2014, 149, 103–116. [Google Scholar] [CrossRef]
- Fleming, J.-A.G.; Song, G.; Choi, Y.; Spencer, T.E.; Bazer, F.W. Interferon regulatory factor 6 (IRF6) is expressed in the ovine uterus and functions as a transcriptional activator. Mol. Cell. Endocrinol. 2009, 299, 252–260. [Google Scholar] [CrossRef] [PubMed]
- Forde, N.; Spencer, T.E.; Bazer, F.W.; Song, G.; Roche, J.F.; Lonergan, P. Effect of pregnancy and progesterone concentration on expression of genes encoding for transporters or secreted proteins in the bovine endometrium. Physiol. Genom. 2010, 41, 53–62. [Google Scholar] [CrossRef] [PubMed]
- Kowalik, M.K.; Dobrzyn, K.; Rekawiecki, R.; Kotwica, J. Expression of membrane progestin receptors (mPRs) α, β and γ in the bovine uterus during the oestrous cycle and pregnancy. Theriogenology 2019, 140, 171–179. [Google Scholar] [CrossRef]
- Forde, N.; Mehta, J.P.; McGettigan, P.A.; Mamo, S.; Bazer, F.W.; Spencer, T.E.; Lonergan, P. Alterations in expression of endometrial genes coding for proteins secreted into the uterine lumen during conceptus elongation in cattle. BMC Genom. 2013, 14, 321. [Google Scholar] [CrossRef]
- Arosh, J.A.; Banu, S.K.; McCracken, J.A. Novel concepts on the role of prostaglandins on luteal maintenance and maternal recognition and establishment of pregnancy in ruminants. J. Dairy Sci. 2016, 99, 5926–5940. [Google Scholar] [CrossRef]
- Choi, Y.; Johnson, G.A.; Burghardt, R.C.; Berghman, L.R.; Joyce, M.M.; Taylor, K.M.; David Stewart, M.; Bazer, F.W.; Spencer, T.E. Interferon regulatory factor-two restricts expression of interferon-stimulated genes to the endometrial stroma and glandular epithelium of the ovine uterus. Biol. Reprod. 2001, 65, 1038–1049. [Google Scholar] [CrossRef]
- Song, G.; Bazer, F.W.; Spencer, T.E. Pregnancy and interferon tau regulate RSAD2 and IFIH1 expression in the ovine uterus. Reproduction 2007, 133, 285–295. [Google Scholar] [CrossRef]
- Dorniak, P.; Bazer, F.W.; Spencer, T.E. Prostaglandins regulate conceptus elongation and mediate effects of interferon tau on the ovine uterine endometrium. Biol. Reprod. 2011, 84, 1119–1127. [Google Scholar] [CrossRef]
- Cooke, P.S.; Spencer, T.E.; Bartol, F.F.; Hayashi, K. Uterine glands: Development, function and experimental model systems. Mol. Hum. Reprod. 2013, 19, 547–558. [Google Scholar] [CrossRef]
- Kemp, C.; Willems, E.; Abdo, S.; Lambiv, L.; Leyns, L. Expression of all Wnt genes and their secreted antagonists during mouse blastocyst and postimplantation development. Dev. Dyn. 2005, 233, 1064–1075. [Google Scholar] [CrossRef]
- Atli, M.O.; Guzeloglu, A.; Dinc, D.A. Expression of wingless type (WNT) genes and their antagonists at mRNA levels in equine endometrium during the estrous cycle and early pregnancy. Anim. Reprod. Sci. 2011, 125, 94–102. [Google Scholar] [CrossRef]
- Ashworth, M.D.; Ross, J.W.; Hu, J.; White, F.J.; Stein, D.R.; Desilva, U.; Johnson, G.A.; Spencer, T.E.; Geisert, R.D. Expression of porcine endometrial prostaglandin synthase during the estrous cycle and early pregnancy, and following endocrine disruption of pregnancy. Biol. Reprod. 2006, 74, 1007–1015. [Google Scholar] [CrossRef]
- Chankeaw, W.; Lignier, S.; Richard, C.; Ntallaris, T.; Raliou, M.; Guo, Y.; Plassard, D.; Bevilacqua, C.; Sandra, O.; Andersson, G.; et al. Analysis of the transcriptome of bovine endometrial cells isolated by laser micro-dissection (2): Impacts of post-partum negative energy balance on stromal, glandular and luminal epithelial cells. BMC Genom. 2021, 22, 450. [Google Scholar] [CrossRef]
- Deutsch, D.; Fröhlich, T.; Arnold, G.J. Proteomics of bovine endometrium, oocytes and early embryos. Biosci. Proc. 2019, 8, RDRRDR3. [Google Scholar] [CrossRef]
- Robinson, R.S.; Mann, G.E.; Gadd, T.S.; Lamming, G.E.; Wathes, D.C. The expression of the IGF system in the bovine uterus throughout the oestrous cycle and early pregnancy. J. Endocrinol. 2000, 165, 231–243. [Google Scholar] [CrossRef]
- Ulbrich, S.E.; Schulke, K.; Groebner, A.E.; Reichenbach, H.D.; Angioni, C.; Geisslinger, G.; Meyer, H.H. Quantitative characterization of prostaglandins in the uterus of early pregnant cattle. Reproduction 2009, 138, 371–382. [Google Scholar] [CrossRef]
- Giacomini, E.; Vago, R.; Sanchez, A.M.; Podini, P.; Zarovni, N.; Murdica, V.; Rizzo, R.; Bortolotti, D.; Candiani, M.; Viganò, P. Secretome of in vitro cultured human embryos contains extracellular vesicles that are uptaken by the maternal side. Sci. Rep. 2017, 7, 5210. [Google Scholar] [CrossRef]
- Hansen, T.; Austin, K.; Perry, D.; Pru, J.; Teixeira, M.; Johnson, G. Mechanism of action of interferon-tau in the uterus during early pregnancy. J. Reprod. Fertil. Suppl. 1999, 54, 329–339. [Google Scholar] [CrossRef] [PubMed]
- Spencer, T.E.; Sandra, O.; Wolf, E. Genes involved in conceptus–endometrial interactions in ruminants: Insights from reductionism and thoughts on holistic approaches. Reproduction 2008, 135, 165–179. [Google Scholar] [CrossRef]
- Betteridge, K.; Eaglesome, M.; Randall, G.; Mitchell, D.; Sugden, E. Maternal progesterone levels as evidence of luteotrophic or antiluteolytic effects of embryos transferred to heifers 12–17 days after estrus. Theriogenology 1978, 9, 86. [Google Scholar] [CrossRef]
- Guillomot, M.; Michel, C.; Gaye, P.; Charlier, N.; Trojan, J.; Martal, J. Cellular localization of an embryonic interferon, ovine trophoblastin and its mRNA in sheep embryos during early pregnancy. Biol. Cell 1990, 68, 205–211. [Google Scholar] [CrossRef] [PubMed]
- Rashid, M.B.; Talukder, A.K.; Kusama, K.; Haneda, S.; Takedomi, T.; Yoshino, H.; Moriyasu, S.; Matsui, M.; Shimada, M.; Imakawa, K. Evidence that interferon-tau secreted from Day-7 embryo in vivo generates anti-inflammatory immune response in the bovine uterus. Biochem. Biophys. Res. Commun. 2018, 500, 879–884. [Google Scholar] [CrossRef]
- Banu, S.K.; Lee, J.; Stephen, S.D.; Nithy, T.K.; Arosh, J.A. Interferon tau regulates PGF2α release from the ovine endometrial epithelial cells via activation of novel JAK/EGFR/ERK/EGR-1 pathways. Mol. Endocrinol. 2010, 24, 2315–2330. [Google Scholar] [CrossRef]
- Charpigny, G.; Reinaud, P.; Tamby, J.-P.; Créminon, C.; Martal, J.; Maclouf, J.; Guillomot, M. Expression of cyclooxygenase-1 and-2 in ovine endometrium during the estrous cycle and early pregnancy. Endocrinology 1997, 138, 2163–2171. [Google Scholar] [CrossRef]
- Meseguer, M.; Pellicer, A.; Simon, C. MUC1 and endometrial receptivity. Mol. Hum. Reprod. 1998, 4, 1089–1098. [Google Scholar] [CrossRef]
- Zhou, C.; Dobrinsky, J.; Tsoi, S.; Foxcroft, G.R.; Dixon, W.T.; Stothard, P.; Verstegen, J.; Dyck, M.K. Characterization of the altered gene expression profile in early porcine embryos generated from parthenogenesis and somatic cell chromatin transfer. PLoS ONE 2014, 9, e91728. [Google Scholar] [CrossRef]
- Melo-Baez, B.; Wong, Y.S.; Aguilera, C.J.; Cabezas, J.; Mançanares, A.C.; Riadi, G.; Castro, F.O.; Rodriguez-Alvarez, L. MicroRNAs from extracellular vesicles secreted by bovine embryos as early biomarkers of developmental competence. Int. J. Mol. Sci. 2020, 21, 8888. [Google Scholar] [CrossRef]
- Meerson, A.; Traurig, M.; Ossowski, V.; Fleming, J.; Mullins, M.; Baier, L. Human adipose microRNA-221 is upregulated in obesity and affects fat metabolism downstream of leptin and TNF-α. Diabetologia 2013, 56, 1971–1979. [Google Scholar] [CrossRef]
- Hansen, T.R.; Sinedino, L.D.; Spencer, T.E. Paracrine and endocrine actions of interferon tau (IFNT). Reproduction 2017, 154, F45–F59. [Google Scholar] [CrossRef]
- Cato, A.C.; Nestl, A.; Mink, S. Rapid actions of steroid receptors in cellular signaling pathways. Sci. STKE 2002, 2002, re9. [Google Scholar] [CrossRef]
- Kasubuchi, M.; Watanabe, K.; Hirano, K.; Inoue, D.; Li, X.; Terasawa, K.; Konishi, M.; Itoh, N.; Kimura, I. Membrane progesterone receptor beta (mPRβ/Paqr8) promotes progesterone-dependent neurite outgrowth in PC12 neuronal cells via non-G protein-coupled receptor (GPCR) signaling. Sci. Rep. 2017, 7, 5168. [Google Scholar] [CrossRef]
- Vázquez-Martínez, E.R.; Bello-Alvarez, C.; Hermenegildo-Molina, A.L.; Solís-Paredes, M.; Parra-Hernández, S.; Cruz-Orozco, O.; Silvestri-Tomassoni, J.R.; Escobar-Ponce, L.F.; Hernández-López, L.A.; Reyes-Mayoral, C.; et al. Expression of Membrane Progesterone Receptors in Eutopic and Ectopic Endometrium of Women with Endometriosis. Biomed. Res. Int. 2020, 2020, 2196024. [Google Scholar] [CrossRef]
- Sánchez, J.M.; Passaro, C.; Forde, N.; Browne, J.A.; Behura, S.K.; Fernández-Fuertes, B.; Mathew, D.J.; Kelly, A.K.; Butler, S.T.; Spencer, T.E.; et al. Do differences in the endometrial transcriptome between uterine horns ipsilateral and contralateral to the corpus luteum influence conceptus growth to day 14 in cattle? Biol. Reprod. 2019, 100, 86–100. [Google Scholar] [CrossRef]
- Ramos-Ibeas, P.; Heras, S.; Gómez-Redondo, I.; Planells, B.; Fernández-González, R.; Pericuesta, E.; Laguna-Barraza, R.; Pérez-Cerezales, S.; Gutiérrez-Adán, A. Embryo responses to stress induced by assisted reproductive technologies. Mol. Reprod. Dev. 2019, 86, 1292–1306. [Google Scholar] [CrossRef]
- Kubisch, H.M.; Larson, M.A.; Ealy, A.D.; Murphy, C.N.; Roberts, R.M. Genetic and environmental determinants of interferon-tau secretion by in vivo- and in vitro-derived bovine blastocysts. Anim. Reprod. Sci. 2001, 66, 1–13. [Google Scholar] [CrossRef]
- Mellisho, E.A.; Briones, M.A.; Velásquez, A.E.; Cabezas, J.; Castro, F.O.; Rodríguez-Álvarez, L. Extracellular vesicles secreted during blastulation show viability of bovine embryos. Reproduction 2019, 158, 477–492. [Google Scholar] [CrossRef]
- Mellisho, E.A.; Velásquez, A.E.; Nuñez, M.J.; Cabezas, J.G.; Cueto, J.A.; Fader, C.; Castro, F.O.; Rodríguez-Álvarez, L. Identification and characteristics of extracellular vesicles from bovine blastocysts produced in vitro. PLoS ONE 2017, 12, e0178306. [Google Scholar] [CrossRef]
- Saadeldin, I.M.; Kim, S.J.; Choi, Y.B.; Lee, B.C. Improvement of cloned embryos development by co-culturing with parthenotes: A possible role of exosomes/microvesicles for embryos paracrine communication. Cell Reprogram 2014, 16, 223–234. [Google Scholar] [CrossRef] [PubMed]
- Taqi, M.O.; Saeed-Zidane, M.; Gebremedhn, S.; Salilew-Wondim, D.; Khdrawy, O.; Rings, F.; Neuhoff, C.; Hoelker, M.; Schellander, K.; Tesfaye, D. Sexual dimorphic expression and release of transcription factors in bovine embryos exposed to oxidative stress. Mol. Reprod. Dev. 2019, 86, 2005–2019. [Google Scholar] [CrossRef] [PubMed]
- Bazer, F.W.; Burghardt, R.C.; Johnson, G.A.; Spencer, T.E.; Wu, G. Interferons and progesterone for establishment and maintenance of pregnancy: Interactions among novel cell signaling pathways. Reprod. Biol. 2008, 8, 179–211. [Google Scholar] [CrossRef] [PubMed]
- Okumu, L.A.; Forde, N.; Fahey, A.G.; Fitzpatrick, E.; Roche, J.F.; Crowe, M.A.; Lonergan, P. The effect of elevated progesterone and pregnancy status on mRNA expression and localisation of progesterone and oestrogen receptors in the bovine uterus. Reproduction 2010, 140, 143–153. [Google Scholar] [CrossRef]
- Bazer, F.W. Pregnancy recognition signaling mechanisms in ruminants and pigs. J. Anim. Sci. Biotechnol. 2013, 4, 23. [Google Scholar] [CrossRef]
- Nothnick, W.B. MicroRNAs and Progesterone Receptor Signaling in Endometriosis Pathophysiology. Cells 2022, 11, 1096. [Google Scholar] [CrossRef]
- Velasquez, A.E.; Castro, F.O.; Veraguas, D.; Cox, J.F.; Lara, E.; Briones, M.; Rodriguez-Alvarez, L. Splitting of IVP bovine blastocyst affects morphology and gene expression of resulting demi-embryos during in vitro culture and in vivo elongation. Zygote 2016, 24, 18–30. [Google Scholar] [CrossRef]
- Bó, G.; Mapletoft, R. Evaluation and classification of bovine embryos. Anim. Reprod. AR 2018, 10, 344–348. [Google Scholar]
- Théry, C.; Amigorena, S.; Raposo, G.; Clayton, A. Isolation and characterization of exosomes from cell culture supernatants and biological fluids. Curr. Protoc. Cell Biol. 2006, 30, 3.22.21–23.22.29. [Google Scholar] [CrossRef]
- Masuda, A.; Katoh, N.; Nakabayashi, K.; Kato, K.; Sonoda, K.; Kitade, M.; Takeda, S.; Hata, K.; Tomikawa, J. An improved method for isolation of epithelial and stromal cells from the human endometrium. J. Reprod. Dev. 2016, 62, 213–218. [Google Scholar] [CrossRef]
- Melo-Báez, B.; Mellisho, E.A.; Cabezas, J.; Velásquez, A.E.; Veraguas, D.; Escobar, D.A.C.; Castro, F.O.; Rodríguez-Álvarez, L. Nanoparticles from culture media are internalized by in vitro-produced bovine embryos and its depletion affect expression of pluripotency genes. Anim. Reprod. 2021, 18, e20200028. [Google Scholar] [CrossRef]
- Koh, Y.Q.; Peiris, H.N.; Vaswani, K.; Almughlliq, F.B.; Meier, S.; Burke, C.R.; Roche, J.R.; Reed, C.B.; Mitchell, M.D. Exosomes from dairy cows of divergent fertility; Action on endometrial cells. J. Reprod. Immunol. 2020, 137, 102624. [Google Scholar] [CrossRef]
- Andrews, S. FastQC: A Quality Control Tool for High Throughput Sequence Data. 2010. Available online: http://www.bioinformatics.babraham.ac.uk/projects/fastqc/ (accessed on 20 June 2021).
- Schäffer, A.A.; Nawrocki, E.P.; Choi, Y.; Kitts, P.A.; Karsch-Mizrachi, I.; McVeigh, R. VecScreen_plus_taxonomy: Imposing a tax (onomy) increase on vector contamination screening. Bioinformatics 2018, 34, 755–759. [Google Scholar] [CrossRef]
- Zhou, Q.; Su, X.; Jing, G.; Chen, S.; Ning, K. RNA-QC-chain: Comprehensive and fast quality control for RNA-Seq data. BMC Genom. 2018, 19, 144. [Google Scholar] [CrossRef]
- Bolger, A.M.; Lohse, M.; Usadel, B. Trimmomatic: A flexible trimmer for Illumina sequence data. Bioinformatics 2014, 30, 2114–2120. [Google Scholar] [CrossRef]
- Kim, D.; Langmead, B. Salzberg SLHISAT. A fast spliced aligner with low memory requirements. Nat. Methods 2015, 12, 357–360. [Google Scholar] [CrossRef]
- Li, H.; Handsaker, B.; Wysoker, A.; Fennell, T.; Ruan, J.; Homer, N.; Marth, G.; Abecasis, G.; Durbin, R.; Subgroup, G.P.D.P. The sequence alignment/map format and SAMtools. Bioinformatics 2009, 25, 2078–2079. [Google Scholar] [CrossRef]
- Pertea, M.; Pertea, G.M.; Antonescu, C.M.; Chang, T.-C.; Mendell, J.T.; Salzberg, S.L. StringTie enables improved reconstruction of a transcriptome from RNA-seq reads. Nat. Biotechnol. 2015, 33, 290–295. [Google Scholar] [CrossRef]
- Robinson, M.D.; McCarthy, D.J.; Smyth, G.K. edgeR: A Bioconductor package for differential expression analysis of digital gene expression data. Bioinformatics 2009, 26, 139–140. [Google Scholar] [CrossRef]
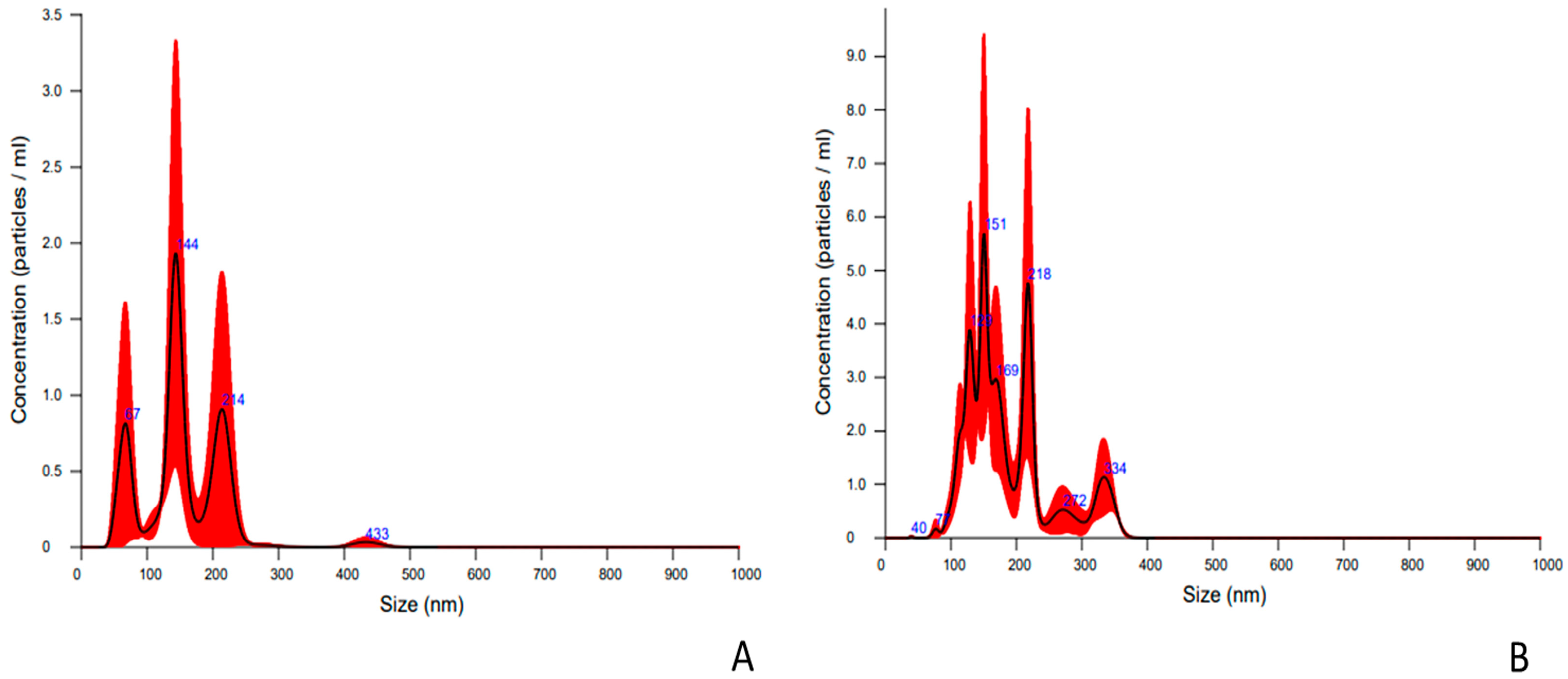



| EVs Parameter | EVs from In Vivo EMBRYOS | EVs from In Vitro Embryos |
|---|---|---|
| Media, nm (x +/− sd) | 186.4 +/− 62.9 nm | 168.9 +/− 80.4 nm |
| Mode, nm (x +/− sd) | 149.2 +/− 11.2 nm | 133.7 +/− 10.0 nm |
| Concentration | 4.24 × 109 +/− 4.36 × 108 particles/ml | 1.14 × 109 +/− 8.62 × 108 particles/mL 1 |
| Sample | CD63 | CD81 | CD40 | CD9 |
|---|---|---|---|---|
| 5–7 in vivo | 3% | 9.4% | 25.7% | 9.8% |
| 5–7 in vitro | 7.1% | 5.4% | 55% | 9.5% |
| Bovine Blood Serum (Positive control) | 5.6% | 8.1% | 14% | 15.4% |
| Beads (negative control) | 0% | 0% | 0% | 0% |
| Beads + antibody (negative control) | 0% | 0% | 0% | 0% |
| SOFdep (Oviductal synthetic fluid depleted) | 0.78% | 1.98% | 0.74% | 0.92% 1 |
| Gene ID | Category | Log2FC IVV/IVP | Reference |
|---|---|---|---|
| BATF2 | Bovine endometrium | 1.10 | [41] |
| CTSL | Stimulated by IFNT Ovine endometrium | −0.95 | [42,43] |
| GRP | Stimulated by IFNT in ovine uterus | 2.19 | [44] |
| HSD11B1 | Stimulated by IFNT Ovine endometrium | −1.39 | [42,43] |
| ICAM1 | Bovine endometrium | −1.5 | [8] |
| IFI6 | Induced by IFNT in bovine uterus | 0.87 | [45] |
| IRF1 | bovine endometrium | 1.07 | [41] |
| IRF6 | Induced by IFNT Ovine endometrium | −0.85 | [46] |
| LGALS9 | Bovine endometrium | −2.51 | [45] |
| LTF | Bovine endometrium | −2.2 | [47] |
| MCOLN3 | Bovine endometrium | −1.4 | [8] |
| MUC1 | Bovine endometrium | −1.09 | [8] |
| PAQR8 | Bovine endometrium | −0.68 | [48] |
| PIP | Bovine endometrium | −0.97 | [49] |
| PTGES | Corpus luteum and endometrium function | 0.95 | [50] |
| SLC2A1 | Stimulated by IFNT Ovine endometrium | −0.93 | [42,43] |
| SPP1 | Stimulated by IFNT Ovine endometrium | −1.33 | [42,43] |
| STAT2 | Ovine uterus | 0.52 | [51] 1 |
| Gene ID | Category | Log2FC EVs-IVV/Control | Log2FC EVs-IVP/Control | Reference |
|---|---|---|---|---|
| OAS1Y | INFT | 1.4 | 2.55 | [10] |
| MX1 | INFT | 0.70 | 2.48 | [10] |
| ISG15 | INFT | 0.68 | 1.23 | [10] |
| MX2 | INFT | 4.79 | 2.62 | [10] |
| IRF6 | INFT | 1.42 | 2.28 | [46] |
| IFIH1 | INFT | 1.02 | 1.53 | [52] |
| HSD11B1 | INFT | 1.12 | 2.51 | [53] |
| CTSL | INFT | 1.68 | 2.64 | [53] |
| CST6 | INFT | 1.87 | 2.12 | [53] |
| MUC1 | Cell adhesion | 1.37 | 2.47 | [8] |
| LGALS9 | Cell adhesion | 1.25 | 3.77 | [45] |
| WNT7B | Early embryonic development and endometrial function | 2.05 | 2.39 | [54] |
| WNT7A | Early embryonic development and endometrial function | 2.14 | 1.81 | [53] |
| WNT5A | Early embryonic development and endometrial function | −0.70 | −0.95 | [55,56] |
| WNT3A | Early embryonic development and endometrial function | 2.81 | 3.15 | [55] |
| PTGS2 | Endometrial function | 0.58 | 1.06 | [53] |
| PTGS1 | Endometrial function | 2.72 | 2.77 | [57] |
| MMP19 | Extracellular matrix remodeling | −0.61 | −0.5 | [8] |
| GRB7 | Growth factor signaling | 1.49 | 1.86 | [58] |
| IGF2BP3 | Growth factor signaling | 1.14 | 1.77 | [59] |
| IGFBP3 | Growth factor signaling | 1.08 | 1.15 | [60] |
| IFIT2 | Immune response | 1.43 | 2.47 | [45] |
| EPSTI1 | Immune response | 0.8 | 0.7 | [45] |
| SPP1 | Secretory activity | 0.58 | 1.91 | [42,43] |
| PIP | Secretory activity | 2.27 | 3.25 | [49] |
| LTF | Secretory activity | 0.95 | 3.16 | [47] |
| OXTR | Sex steroid signaling | 1.64 | 3.43 | [61] |
| PAQR8 | Sex steroid signaling | 0.89 | 1.78 | [48] |
| AKR1C4 | Sex steroid signaling | 0.59 | 4.14 | [8] |
| ESR1 | Sex steroid signaling | −0.34 (not significant) | 0.77 | [8] 1 |
| Gene | Primer Sequences (5′–3′) | Product Length (bp) | Annealing Temperature | Accession Number (NCBI) |
|---|---|---|---|---|
| MX2 | F: AAGTATGAGGAGAAGGTGCGGC | 112 | 57 °C | XM_015473641.2 |
| R: AGCTCTGGTCCCCGATAACG | ||||
| OXTR | F: ACAAGCACTCGCGCCTCTTCT | 107 | 56 °C | XR_003031707.1 |
| R: GCGGAACGTGATGTCCCACAGA | ||||
| MUC1 | F: ACATCCAGGCCCCTTTCCTC | 118 | 55 °C | NM_174115.3 |
| R: GTGGAAACTGGCGTAGTTCTGC | ||||
| OAS1Y | F: AGCACCGTGATGGAGCTCAG | 111 | 55 °C | NM_001040606.1 |
| R: GTCGATGGCTTCTTTGACCTGC | ||||
| CTSL | F: CCTCGCCACAGGTTTTTGAAC | 91 | 55 °C | NM_001083686.2 |
| R: TGTGGTCAAATTTTGGAGCAGC | ||||
| WNT7A | F: ATGGTCTACCTCCGGATCGGTG | 83 | 57 °C | NM_001192788.1 |
| R: GCCAGGCCTGGGATCTTGTTAC | ||||
| ISG15 | F: ACCTGACGGTGAAGATGCTAGG | 98 | 57 °C | NM_174366.1 |
| R: GATCTTCTGGGCGATGAACTGC | ||||
| ESR1 | R: CCAACAGGTGCCCTATTACCTG | 108 | 55 °C | NM_001001443.1 |
| R: CCACCTTGGCGTCGATTATCTG | ||||
| HPGD | F: AAGGTAGCGCTGGTCGATTGG | 120 | 54 °C | NM_001034419.2 |
| R: TTGTTCCTGATCGGCCACATCG | ||||
| PTGES | F: ATGTACGTGGTGGCCGTCATC | 108 | 56 °C | NM_174443.2 |
| R: GTCGTTCCGGCAATACTGGAGG | ||||
| MMP14 | F: AGTCCCTCTCAGCTGCCATTG | 89 | 56 °C | NM_174390.2 |
| R: CATGGCCTTCATGGTGTCTGC | ||||
| MX1 | F: AGGCAGGAGACCATCAACTTGG | 144 | 56 °C | XM_015473623.2 |
| R: ACCTTTGTCCACCAGATCGGG | ||||
| CST6 | F: GTGTCCCTGTAGAGACCCCT | 113 | 55 °C | NM_001012764.3 |
| R: TGCCGTAGATACGGTCCAAG | ||||
| ACTB | F: TGCCCTGAGGCTCTCTTCCA | 119 | 55 °C | NM_173979.3 |
| R: TTGGCGTAGAGGTCCTTGCG | ||||
| GAPDH | F: AGGTCGGAGTGAACGGATTC | 85 | 55 °C | NM_001034034.2 |
| R: ATGGCGACGATGTCCACTTT 1 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aguilera, C.; Velásquez, A.E.; Gutierrez-Reinoso, M.A.; Wong, Y.S.; Melo-Baez, B.; Cabezas, J.; Caamaño, D.; Navarrete, F.; Rojas, D.; Riadi, G.; et al. Extracellular Vesicles Secreted by Pre-Hatching Bovine Embryos Produced In Vitro and In Vivo Alter the Expression of IFNtau-Stimulated Genes in Bovine Endometrial Cells. Int. J. Mol. Sci. 2023, 24, 7438. https://doi.org/10.3390/ijms24087438
Aguilera C, Velásquez AE, Gutierrez-Reinoso MA, Wong YS, Melo-Baez B, Cabezas J, Caamaño D, Navarrete F, Rojas D, Riadi G, et al. Extracellular Vesicles Secreted by Pre-Hatching Bovine Embryos Produced In Vitro and In Vivo Alter the Expression of IFNtau-Stimulated Genes in Bovine Endometrial Cells. International Journal of Molecular Sciences. 2023; 24(8):7438. https://doi.org/10.3390/ijms24087438
Chicago/Turabian StyleAguilera, Constanza, Alejandra Estela Velásquez, Miguel Angel Gutierrez-Reinoso, Yat Sen Wong, Barbara Melo-Baez, Joel Cabezas, Diego Caamaño, Felipe Navarrete, Daniela Rojas, Gonzalo Riadi, and et al. 2023. "Extracellular Vesicles Secreted by Pre-Hatching Bovine Embryos Produced In Vitro and In Vivo Alter the Expression of IFNtau-Stimulated Genes in Bovine Endometrial Cells" International Journal of Molecular Sciences 24, no. 8: 7438. https://doi.org/10.3390/ijms24087438








