Rapid and Sensitive Detection of Streptococcus iniae in Trachinotus ovatus Based on Multienzyme Isothermal Rapid Amplification
Abstract
1. Introduction
2. Results
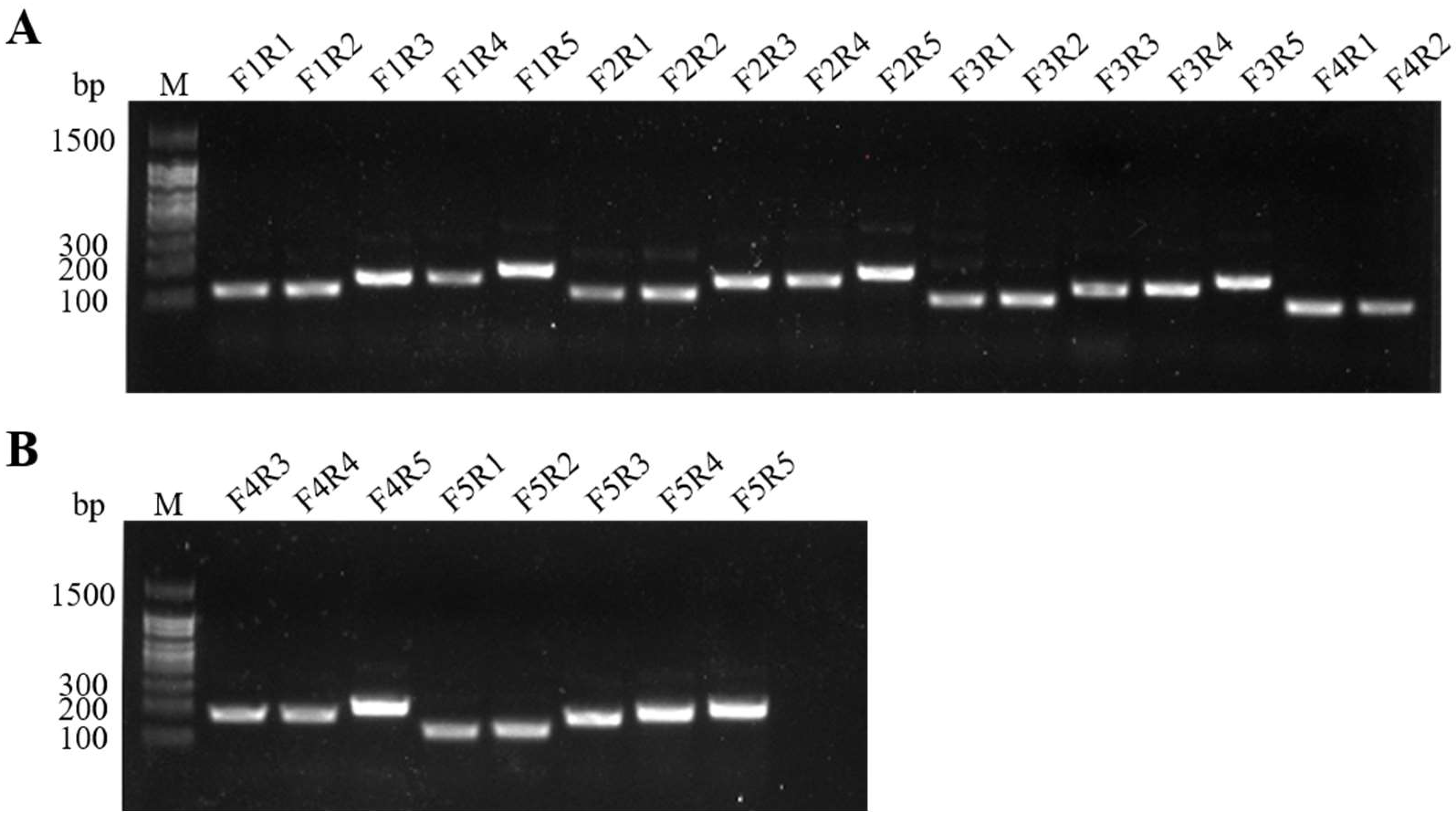
2.1. Evaluation of MIRA Primers
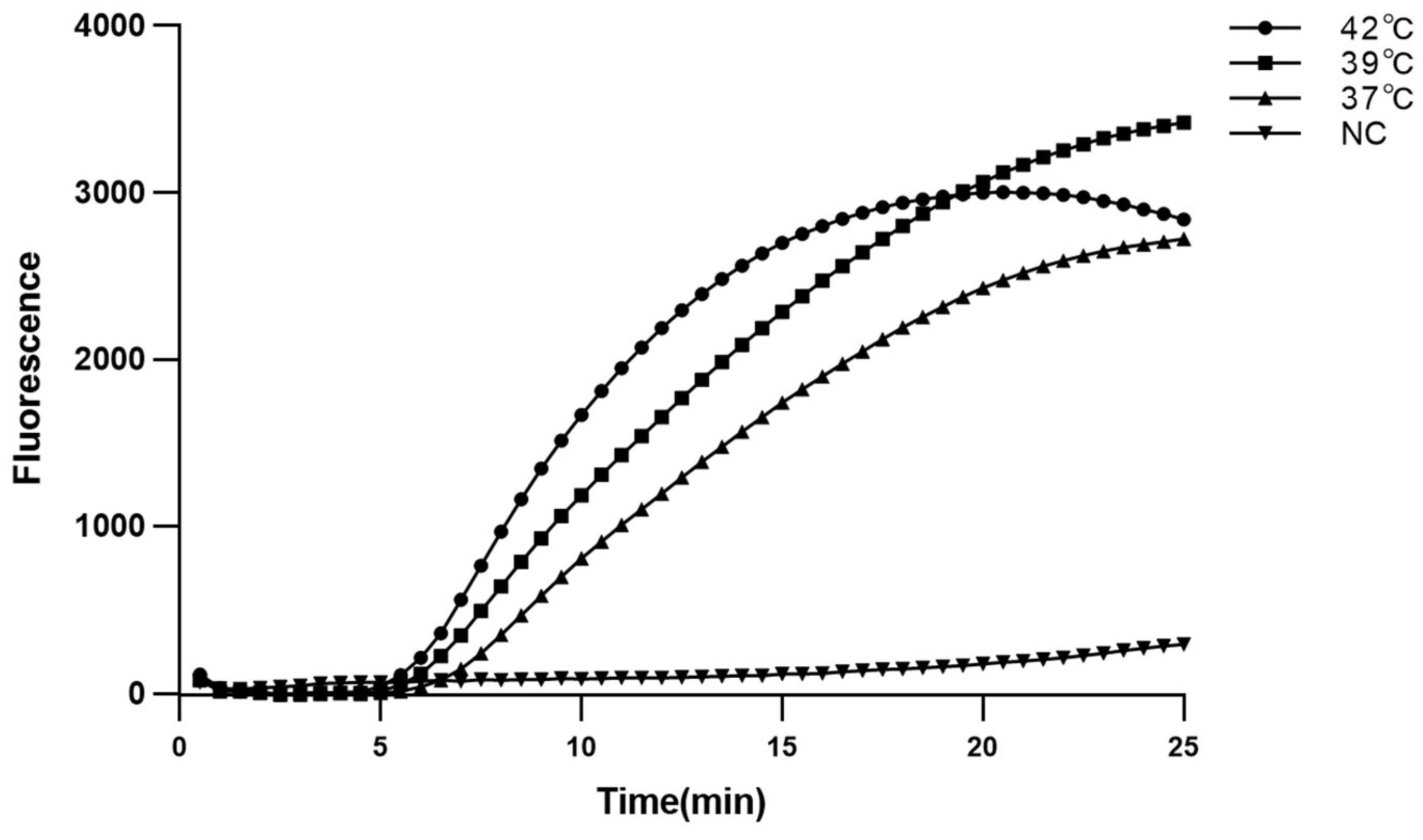
2.2. Optimal Reaction Conditions
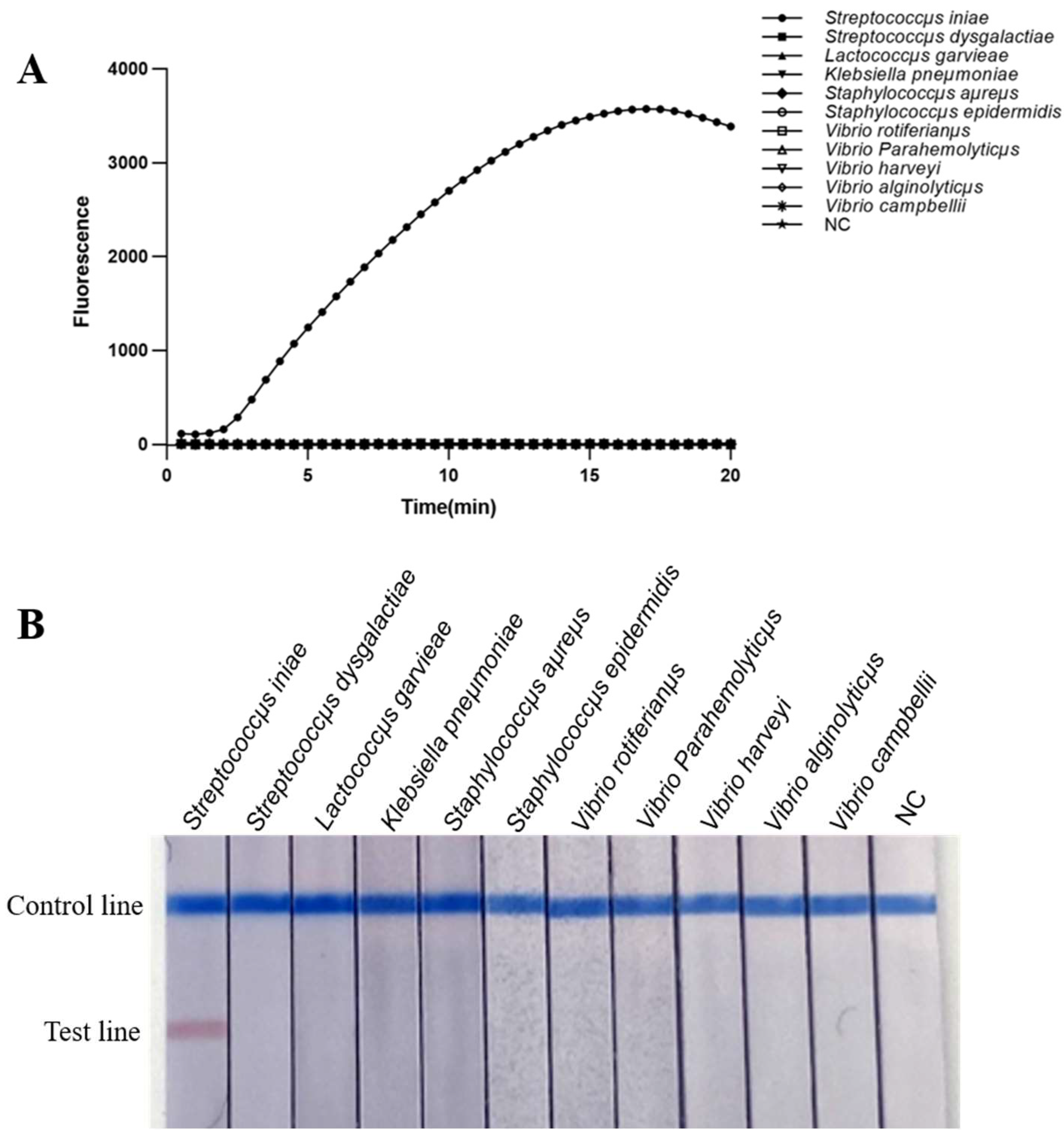
2.3. Specificity of Real-Time MIRA and MIRA-LFD Assays
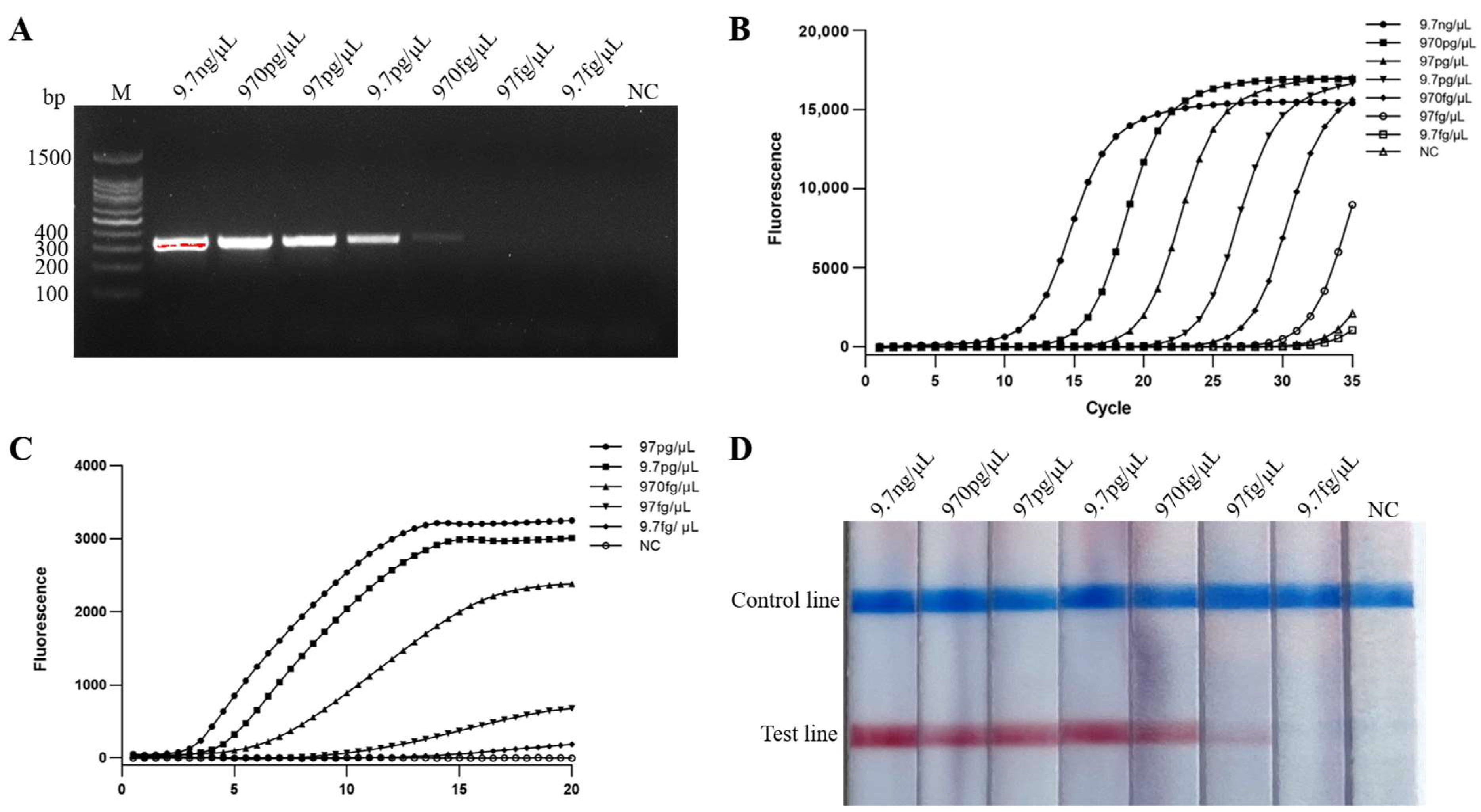
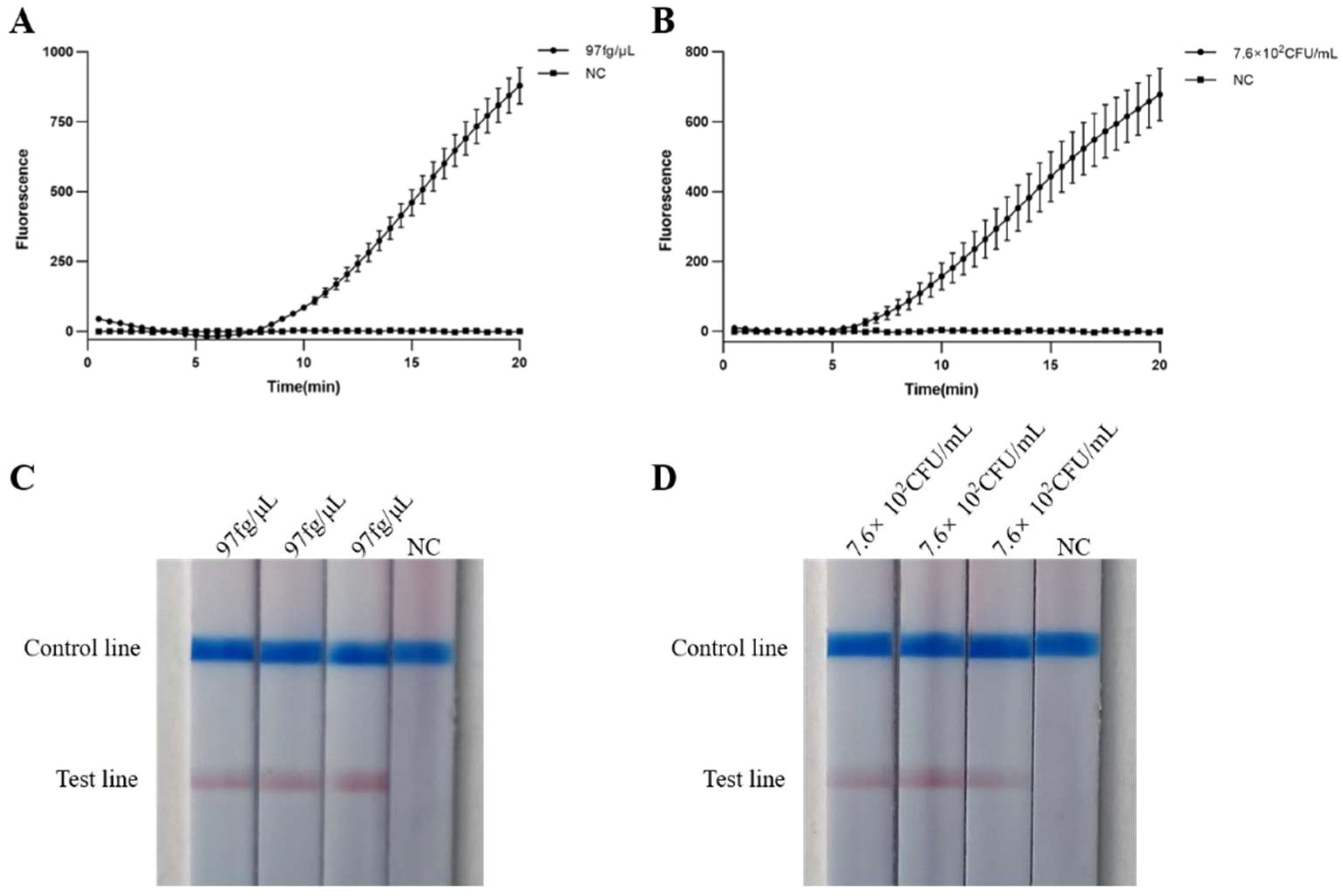
2.4. Sensitivity of Real-Time MIRA and MIRA-LFD Assays
2.5. Reproducibility of Real-Time MIRA and MIRA-LFD Assays
2.6. Practicability of Real-Time MIRA and MIRA-LFD Assays
3. Discussion
4. Materials and Methods
4.1. Fish and Bacterial Strains
4.2. Genomic DNA Extraction
4.3. Design of Primers and Probes
4.4. Establishment of MIRA, Real-Time MIRA, and MIRA-LFD Assays
4.5. Specificity of Real-Time MIRA and MIRA-LFD Assays
4.6. Sensitivity of Real-Time MIRA and MIRA-LFD Assays
4.7. Reproducibility of Real-Time MIRA and MIRA-LFD Assays
4.8. Practicability of Real-Time MIRA and MIRA-LFD Assays
5. Conclusions
6. Patents
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ma, Y.-M.; Yang, M.-J.; Wang, S.; Li, H.; Peng, X.-X. Liver functional metabolomics discloses an action of L-leucine against Streptococcus iniae infection in tilapias. Fish Shellfish Immunol. 2015, 45, 414–421. [Google Scholar] [CrossRef] [PubMed]
- Feng, Y.; Bai, M.; Geng, Y.; Chen, D.; Huang, X.; Ouyang, P.; Guo, H.; Zuo, Z.; Huang, C.; Lai, W. The potential risk of antibiotic resistance of Streptococcus iniae in sturgeon cultivation in Sichuan, China. Environ. Sci. Pollut. Res. Int. 2021, 28, 69171–69180. [Google Scholar] [CrossRef] [PubMed]
- Hayat, M.; Yusoff, M.; Samad, M.; Razak, I.A.; Yasin, I.; Thompson, K.; Hasni, K. Efficacy of Feed-Based Formalin-Killed Vaccine of Streptococcus iniae Stimulates the Gut-Associated Lymphoid Tissues and Immune Response of Red Hybrid Tilapia. Vaccines 2021, 9, 51. [Google Scholar] [CrossRef]
- Guo, S.; Mo, Z.; Wang, Z.; Xu, J.; Li, Y.; Dan, X.; Li, A. Isolation and pathogenicity of Streptococcus iniae in offshore cage-cultured Trachinotus ovatus in China. Aquaculture 2018, 492, 247–252. [Google Scholar] [CrossRef]
- Zhou, Y.; Xiao, J.; Ma, X.; Wang, Q.; Zhang, Y. An effective established biosensor of bifunctional probes-labeled AuNPs combined with LAMP for detection of fish pathogen Streptococcus iniae. Appl. Microbiol. Biotechnol. 2018, 102, 5299–5308. [Google Scholar] [CrossRef]
- Baiano, J.C.; A Tumbol, R.; Umapathy, A.; Barnes, A.C. Identification and molecular characterisation of a fibrinogen binding protein from Streptococcus iniae. BMC Microbiol. 2008, 8, 67. [Google Scholar] [CrossRef]
- Locke, J.B.; Aziz, R.K.; Vicknair, M.R.; Nizet, V.; Buchanan, J.T. Streptococcus iniae M-like protein contributes to virulence in fish and is a target for live attenuated vaccine development. PLoS ONE 2008, 3, e2824. [Google Scholar] [CrossRef]
- Irion, S.; Silayeva, O.; Sweet, M.; Chabanet, P.; Barnes, A.C.; Tortosa, P.; Séré, M.G. Molecular Investigation of Recurrent Streptococcus iniae Epizootics Affecting Coral Reef Fish on an Oceanic Island Suggests at Least Two Distinct Emergence Events. Front. Microbiol. 2021, 12, 749734. [Google Scholar] [CrossRef]
- Shelby, R.A.; Shoemaker, C.A.; Klesius, P.H. Detection of Humoral Response to Streptococcus iniae Infection of Nile Tilapia, Oreochromis niloticus, by a Monoclonal Antibody-Based ELISA. J. Appl. Aquac. 2002, 12, 23–31. [Google Scholar] [CrossRef]
- Klesius, P.; Evans, J.; Shoemaker, C.; Yeh, H.; Goodwin, A.; Adams, A.; Thompson, K. Rapid detection and identification of Streptococcus iniae using a monoclonal antibody-based indirect fluorescent antibody technique. Aquaculture 2006, 258, 180–186. [Google Scholar] [CrossRef]
- Mata, A.I.; Blanco, M.; Domıínguez, L.; Fernández-Garayzábal, J.F.; Gibello, A. Development of a PCR assay for Streptococcus iniae based on the lactate oxidase (lctO) gene with potential diagnostic value. Vet. Microbiol. 2004, 101, 109–116. [Google Scholar] [CrossRef]
- Torres-Corral, Y.; Santos, Y. Development of a real-time PCR assay for detection and quantification of Streptococcus iniae using the lactate permease gene. J. Fish Dis. 2021, 44, 53–61. [Google Scholar] [CrossRef]
- Wu, Y.-D.; Zhou, D.-H.; Zhang, L.-X.; Zheng, W.-B.; Ma, J.-G.; Wang, M.; Zhu, X.-Q.; Xu, M.-J. Recombinase polymerase amplification (RPA) combined with lateral flow (LF) strip for equipment-free detection of Cryptosporidium spp. oocysts in dairy cattle feces. Parasitol. Res. 2016, 115, 3551–3555. [Google Scholar] [CrossRef]
- Cheng, S.; Hu, Y.-H.; Jiao, X.-D.; Sun, L. Identification and immunoprotective analysis of a Streptococcus iniae subunit vaccine candidate. Vaccine 2010, 28, 2636–2641. [Google Scholar] [CrossRef]
- Geng, Y.; Liu, S.; Wang, J.; Nan, H.; Liu, L.; Sun, X.; Li, D.; Liu, M.; Wang, J.; Tan, K. Rapid Detection of Staphylococcus aureus in Food Using a Recombinase Polymerase Amplification-Based Assay. Food Anal. Methods 2018, 11, 2847–2856. [Google Scholar] [CrossRef]
- Cui, M.; Zhang, H.; Li, J.; Liu, R.; Wu, M.; Xu, D.; Zhang, Q. Differential PCR detection of Streptococcus agalactiae and Streptococcus iniae in tilapia (Oreochromis niloticus) by a single primer pair. J. Fish Dis. 2019, 42, 1211–1216. [Google Scholar] [CrossRef]
- Ding, S.-W.; Yang, S.-H.; Wu, W.-J.; Xie, H.; Xu, C.-L. Rapid diagnosis of Ditylenchus destructor by loop-mediated isothermal amplification assay based on 28S rRNA sequences. Eur. J. Plant Pathol. 2018, 153, 1165–1175. [Google Scholar] [CrossRef]
- Cai, S.-H.; Wang, B.; Lu, Y.-S.; Jian, J.-C.; Wu, Z.-H. Development of loop-mediated isothermal amplification method for rapid detection of Streptococcus iniae, the causative agent of streptococcicosis in fish. J. Basic Microbiol. 2012, 52, 116–122. [Google Scholar] [CrossRef]
- Zhang, Y.; Hu, J.; Li, Q.; Guo, J.; Zhang, G. Detection of microorganisms using recombinase polymerase amplification with lateral flow dipsticks. Methods Microbiol. 2020, 47, 319–349. [Google Scholar]
- Qu, Y.; Wang, Q.; Li, Y.; Wang, Y.; Yin, J.; Ren, Y.; Liu, C.; Liu, X.; Wang, Y.; Zeng, W. Development of a real-time recombinase polymerase amplification assay for rapid detection of Aeromonas hydrophila. J. Fish Dis. 2021, 44, 469–477. [Google Scholar] [CrossRef]
- Salazar, A.; Ochoa-Corona, F.M.; Talley, J.L.; Noden, B.H. Recombinase polymerase amplification (RPA) with lateral flow detection for three Anaplasma species of importance to livestock health. Sci. Rep. 2021, 11, 15962. [Google Scholar] [CrossRef]
- Jauset-Rubio, M.; Tomaso, H.; El-Shahawi, M.S.; Bashammakh, A.S.; Al-Youbi, A.O.; O’Sullivan, C.K. Duplex Lateral Flow Assay for the Simultaneous Detection of Yersinia pestis and Francisella tularensis. Anal. Chem. 2018, 90, 12745–12751. [Google Scholar] [CrossRef]
- Jarvi, S.I.; Atkinson, E.S.; Kaluna, L.M.; Snook, K.A.; Steel, A. Development of a recombinase polymerase amplification (RPA-EXO) and lateral flow assay (RPA-LFA) based on the ITS1 gene for the detection of Angiostrongylus cantonensis in gastropod intermediate hosts. Parasitology 2021, 148, 251–258. [Google Scholar] [CrossRef]
- Hu, W.-W.; He, J.-W.; Guo, S.-L.; Li, J. Development and evaluation of a rapid and sensitive multienzyme isothermal rapid amplification with a lateral flow dipstick assay for detection of Acinetobacter baumannii in spiked blood specimens. Front. Cell. Infect. Microbiol. 2022, 12, 1010201. [Google Scholar] [CrossRef]
- Heng, P.; Liu, J.; Song, Z.; Wu, C.; Yu, X.; He, Y. Rapid detection of Staphylococcus aureus using a novel multienzyme isothermal rapid amplification technique. Front. Microbiol. 2022, 13, 1027785. [Google Scholar] [CrossRef]
- Nie, Z.; Zhao, Y.; Shu, X.; Li, D.; Ao, Y.; Li, M.; Wang, S.; Cui, J.; An, X.; Zhan, X.; et al. Recombinase polymerase amplification with lateral flow strip for detecting Babesia microti infections. Parasitol. Int. 2021, 83, 102351. [Google Scholar] [CrossRef]
- Gao, B.; Ma, J.; Li, X.; Wang, R.; Chen, S. A real-time recombinase polymerase amplification assay for fast and accurate detection of Ditylenchus destructor. Mol. Cell. Probes 2022, 61, 101788. [Google Scholar] [CrossRef]
- James, A.; Macdonald, J. Recombinase polymerase amplification: Emergence as a critical molecular technology for rapid, low-resource diagnostics. Expert Rev. Mol. Diagn. 2015, 15, 1475–1489. [Google Scholar] [CrossRef]
- Ernst, C.; Bartel, A.; Elferink, J.W.; Huhn, J.; Eschbach, E.; Schönfeld, K.; Feßler, A.T.; Oberheitmann, B.; Schwarz, S. Improved DNA extraction and purification with magnetic nanoparticles for the detection of methicillin-resistant Staphylococcus aureus. Vet. Microbiol. 2019, 230, 45–48. [Google Scholar] [CrossRef]
- Vinayaka, A.C.; Ngo, T.A.; Kant, K.; Engelsmann, P.; Dave, V.P.; Shahbazi, M.-A.; Wolff, A.; Bang, D.D. Rapid detection of Salmonella enterica in food samples by a novel approach with combination of sample concentration and direct PCR. Biosens. Bioelectron. 2019, 129, 224–230. [Google Scholar] [CrossRef]
- Awasthi, S.P.; Chowdhury, N.; Neogi, S.B.; Hinenoya, A.; Hatanaka, N.; Chowdhury, G.; Ramamurthy, T.; Yamasaki, S. Development of a multiplex PCR assay for the detection of major virulence genes in Vibrio cholerae including non-O1 and non-O139 serogroups. J. Microbiol. Methods 2019, 157, 54–58. [Google Scholar] [CrossRef]
- Dai, T.; Yang, X.; Hu, T.; Jiao, B.; Xu, Y.; Zheng, X.; Shen, D. Comparative Evaluation of a Novel Recombinase Polymerase Amplification-Lateral Flow Dipstick (RPA-LFD) Assay, LAMP, Conventional PCR, and Leaf-Disc Baiting Methods for Detection of Phytophthora sojae. Front. Microbiol. 2019, 10, 1884. [Google Scholar] [CrossRef]
- Schwartz, T.; Volkmann, H.; Kirchen, S.; Kohnen, W.; Schön-Hölz, K.; Jansen, B.; Obst, U. Real-time PCR detection of Pseudomonas aeruginosa in clinical and municipal wastewater and genotyping of the ciprofloxacin-resistant isolates. FEMS Microbiol. Ecol. 2006, 57, 158–167. [Google Scholar] [CrossRef]
- Romano, J.W.; van Gemen, B.; Kievits, T. NASBA: A Novel, Isothermal Detection Technology for Qualitative and Quantitative HIV-1 RNA Measurements. Clin. Lab. Med. 1996, 16, 89–103. [Google Scholar] [CrossRef]
- Jenison, R.; Jaeckel, H.; Klonoski, J.; Latorra, D.; Wiens, J. Rapid amplification/detection of nucleic acid targets utilizing a HDA/thin film biosensor. Analyst 2014, 139, 3763–3769. [Google Scholar] [CrossRef]
- Han, H.-J.; Jung, S.-J.; Oh, M.-J.; Kim, D.-H. Rapid and sensitive detection of Streptococcus iniae by loop-mediated isothermal amplification (LAMP). J. Fish Dis. 2011, 34, 395–398. [Google Scholar] [CrossRef]


| Name | Sequence (5′–3′) | Location |
|---|---|---|
| simA-F1 | TAGAAGCGGCTAAGAAAGAAGCAGAAGAA | 923–951 |
| simA-F2 | TGACTAAAGCATTAGAAGCGGCTAAGAAA | 911–939 |
| simA-F3 | ACTAAAGCATTAGAAGCGGCTAAGAAAGA | 913–941 |
| simA-F4 | TAAAGCATTAGAAGCGGCTAAGAAAGAAG | 915–943 |
| simA-F5 | AAGCATTAGAAGCGGCTAAGAAAGAAGCAG | 917–946 |
| simA-R1 | GTGCTTTCTCAAGGTCTTTTTCAAGTTCT | 1057–1029 |
| simA-R2 | CTGCTTGTGCTTTCTCAAGGTCTTTTTCAA | 1063–1034 |
| simA-R3 | TCCAATTCAGCTTTTGTTTCTGCTAGTTTA | 1112–1083 |
| simA-R4 | TTTCCAATTCAGCTTTTGTTTCTGCTAGTT | 1114–1085 |
| simA-R5 | CAATAGTTGCTTCAAGTTCTGCTTTTTCA | 1153–1125 |
| Assay | Name | Sequence (5′–3′) |
|---|---|---|
| Real-time MIRA | exo-F | TAAAGCATTAGAAGCGGCTAAGAAAGAAG |
| exo-R | CAATAGTTGCTTCAAGTTCTGCTTTTTCA | |
| exo-P | TTTCCAATTCAGCTTTGTTTCTGCTAGT(i6FAMdT) (idSp)(iBHQ1dT)AGTTTCAAGGTCTTTAG | |
| MIRA-LFD | nfo-F | TAAAGCATTAGAAGCGGCTAAGAAAGAAG |
| nfo-R | (Biotin)CAATAGTTGCTTCAAGTTCTGCTTTTTCA | |
| nfo-P | (FAM)TTTCCAATTCAGCTTGTTTCTGCTAGTT (idSp)TAGTTTCAAGGTCTTTAG(C3 Spacer) |
| Name | Sequence (5′–3′) |
|---|---|
| PCR-F | TGAAGAGCTTGACAAACTAAATG |
| PCR-R | ACTTGCTGTGAAGAATGGGTTA |
| SilldP-F | ACACAGGTGAGCACGCTAAA |
| SilldP-R | CGTCACCATCGTCTTGGTCA |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, Y.; Niu, J.; Sun, M.; Li, Z.; Wang, X.; He, Y.; Qi, J. Rapid and Sensitive Detection of Streptococcus iniae in Trachinotus ovatus Based on Multienzyme Isothermal Rapid Amplification. Int. J. Mol. Sci. 2023, 24, 7733. https://doi.org/10.3390/ijms24097733
Wang Y, Niu J, Sun M, Li Z, Wang X, He Y, Qi J. Rapid and Sensitive Detection of Streptococcus iniae in Trachinotus ovatus Based on Multienzyme Isothermal Rapid Amplification. International Journal of Molecular Sciences. 2023; 24(9):7733. https://doi.org/10.3390/ijms24097733
Chicago/Turabian StyleWang, Yifen, Jingjing Niu, Minmin Sun, Ziyi Li, Xiangyuan Wang, Yan He, and Jie Qi. 2023. "Rapid and Sensitive Detection of Streptococcus iniae in Trachinotus ovatus Based on Multienzyme Isothermal Rapid Amplification" International Journal of Molecular Sciences 24, no. 9: 7733. https://doi.org/10.3390/ijms24097733
APA StyleWang, Y., Niu, J., Sun, M., Li, Z., Wang, X., He, Y., & Qi, J. (2023). Rapid and Sensitive Detection of Streptococcus iniae in Trachinotus ovatus Based on Multienzyme Isothermal Rapid Amplification. International Journal of Molecular Sciences, 24(9), 7733. https://doi.org/10.3390/ijms24097733





