Therapeutic Efficacy of YM155 to Regulate an Epigenetic Enzyme in Major Subtypes of RCC
Abstract
:1. Introduction
2. Results
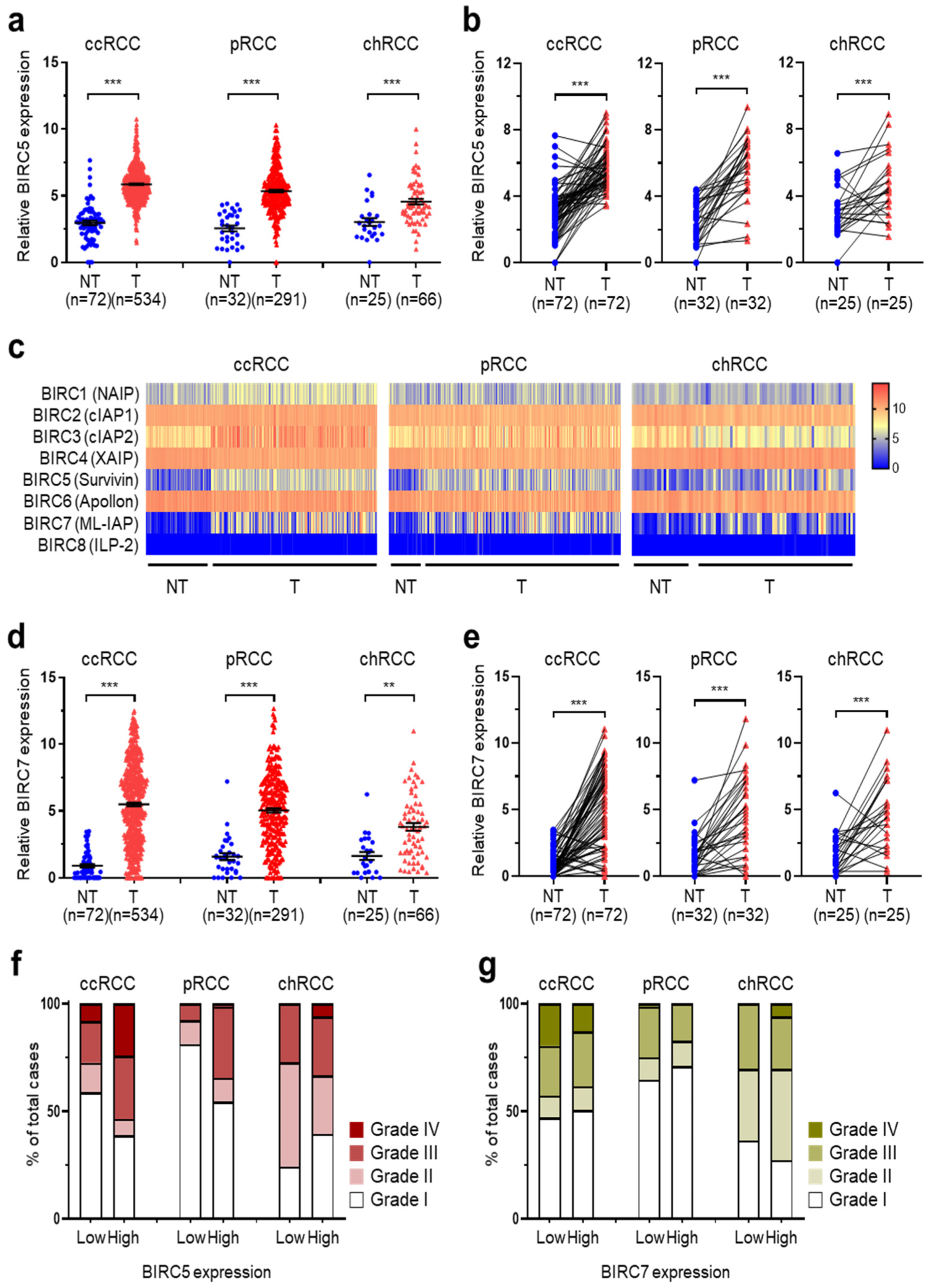
2.1. Correlation between Overexpressed BIRC5 and RCC Tumor Progression
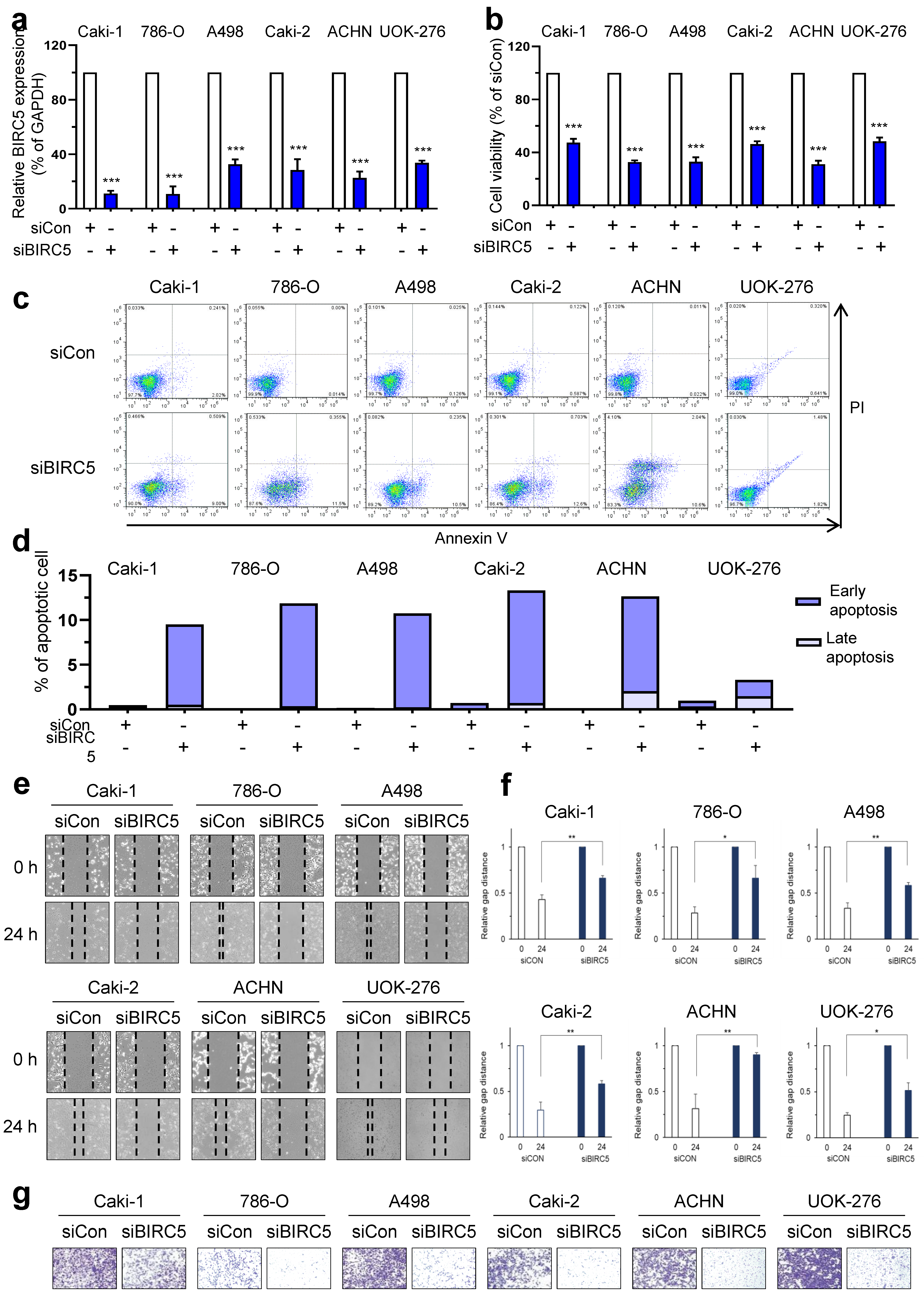
2.2. Evaluation of Function by BIRC5 Knockdown in RCC Cells
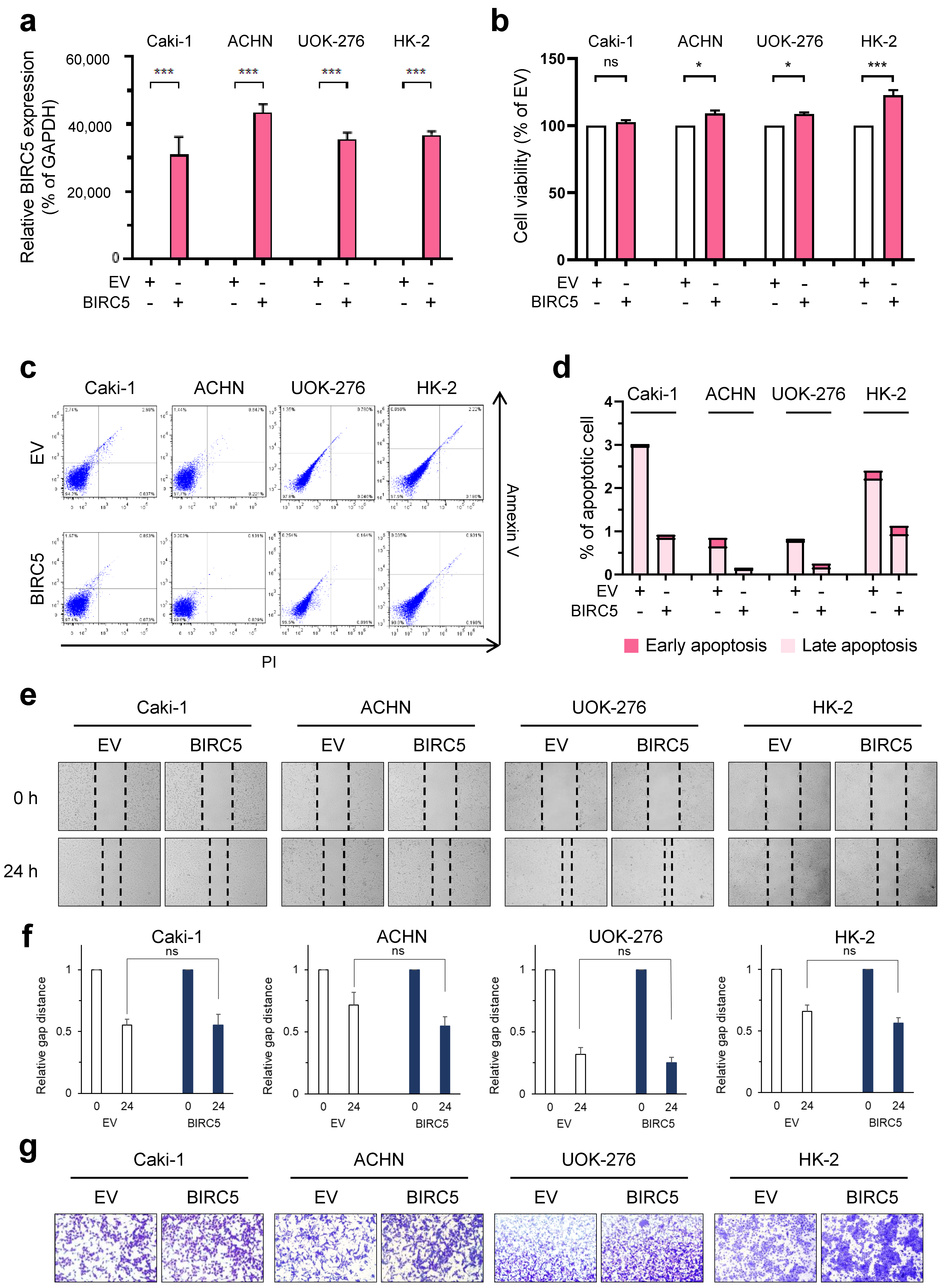
2.3. Evaluation of Function by BIRC5 Overexpression in RCC Cells
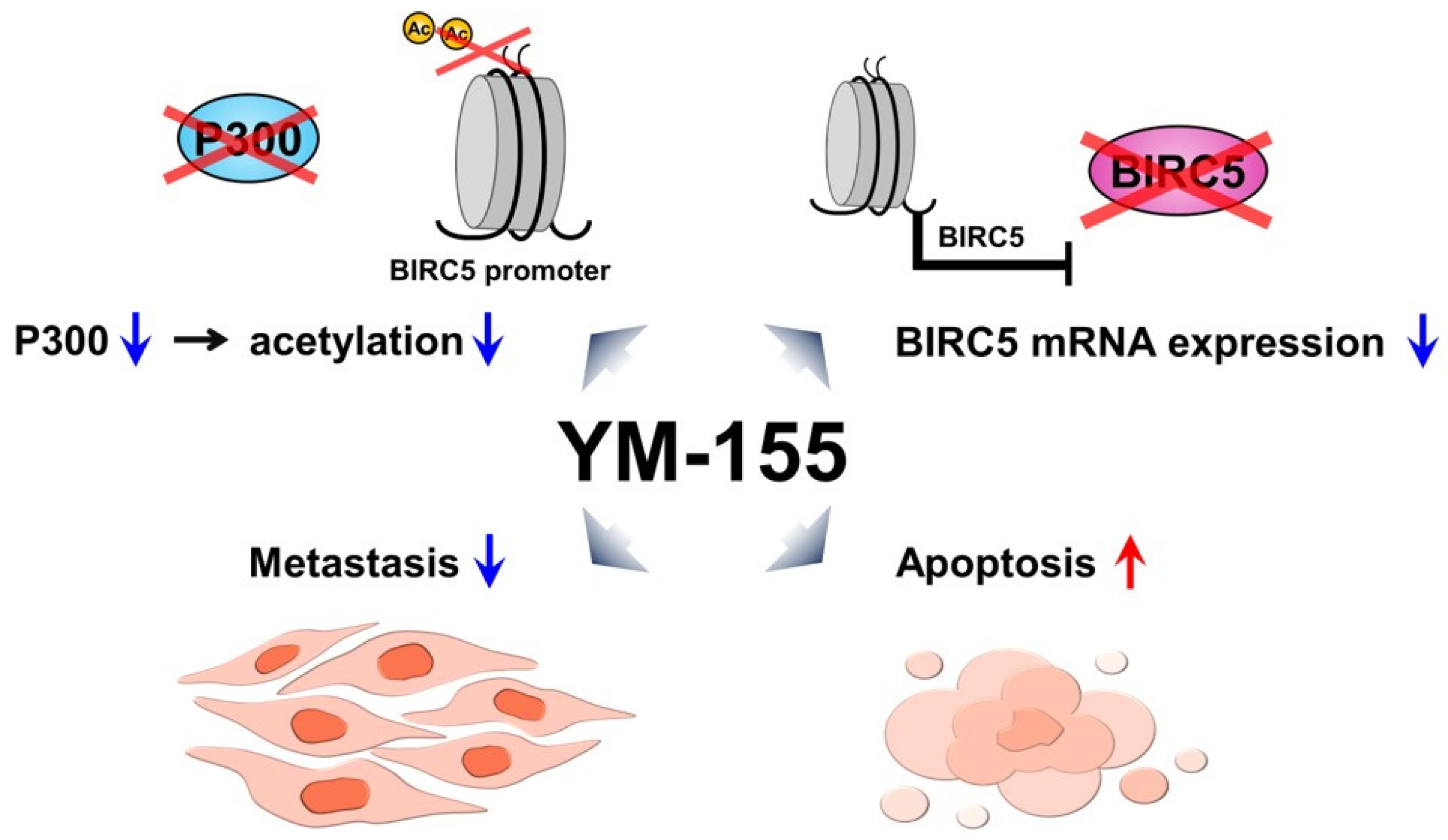
2.4. Confirmation of Anti-Cancer Effect by BIRC5 Inhibition Using YM155
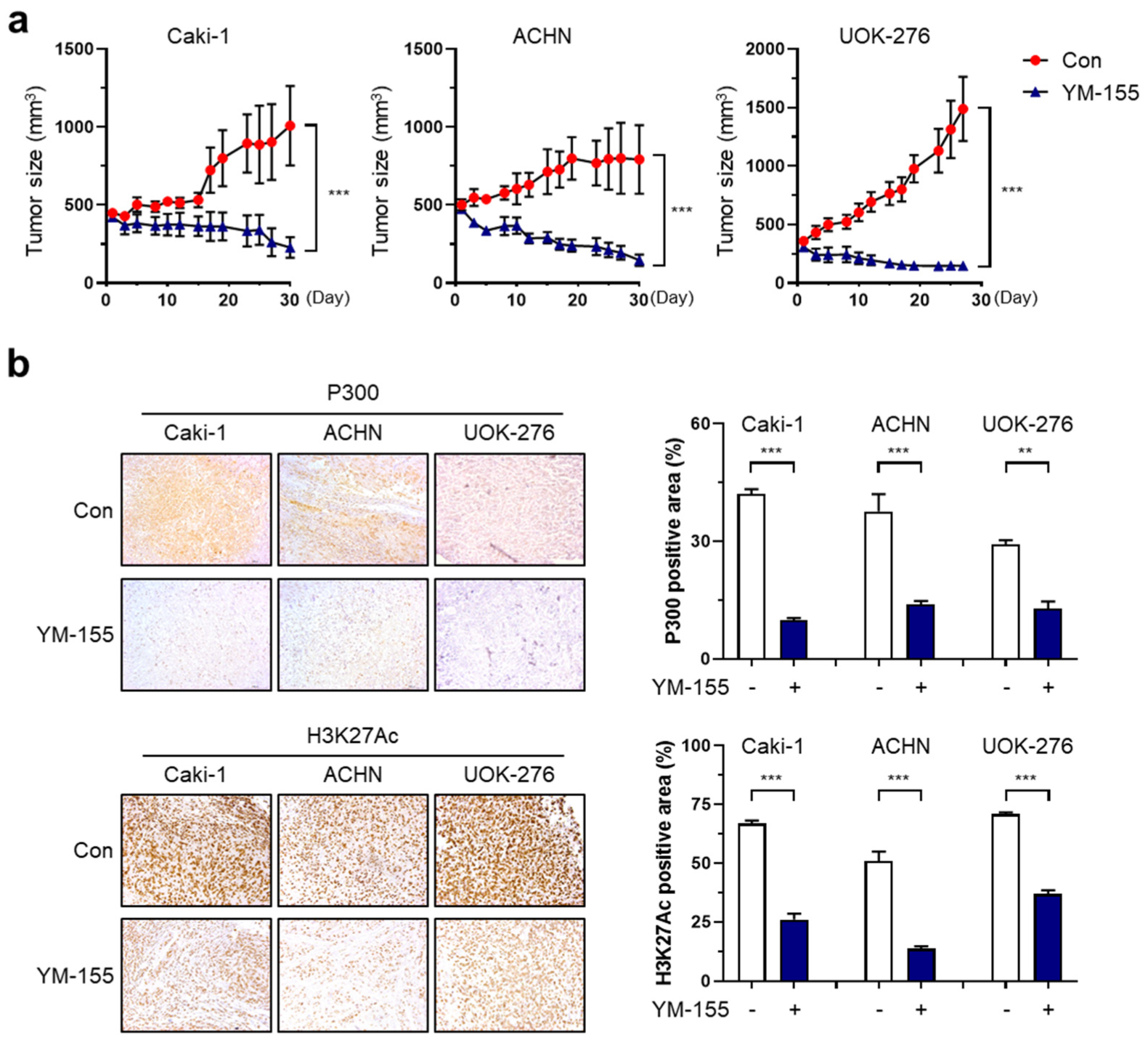
2.5. Identification of the Mechanism Underlying the Antitumor Effect of YM155
2.6. Confirmation of Antitumor Effect of YM155 on RCC Cells In Vivo
3. Discussion
4. Materials and Methods
4.1. Cell Culture
4.2. siRNA Transfection
4.3. Transfection of Plasmids into Cells
4.4. RNA Extraction and Reverse Transcription PCR
4.5. Chomatin Immunoprecipitation (ChIP)
4.6. Wound Healing Assay
4.7. Invasion Assay
4.8. Annexin V/Propidium Iodide Staining Assay
4.9. In Vivo Tumor Growth Experiment
4.10. Immunohistochemistry
4.11. The Cancer Genome Atlas (TCGA) Data Analysis
4.12. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Beksac, A.T.; Paulucci, D.J.; Blum, K.A.; Yadav, S.S.; Sfakianos, J.P.; Badani, K.K. Heterogeneity in renal cell carcinoma. Urol. Oncol. 2017, 35, 507–515. [Google Scholar] [CrossRef] [PubMed]
- Ferlay, J.; Soerjomataram, I.; Dikshit, R.; Eser, S.; Mathers, C.; Rebelo, M.; Parkin, D.M.; Forman, D.; Bray, F. Cancer incidence and mortality worldwide: Sources, methods and major patterns in GLOBOCAN 2012. Int. J. Cancer 2015, 136, E359–E386. [Google Scholar] [CrossRef]
- Moch, H. An overview of renal cell cancer: Pathology and genetics. Semin. Cancer Biol. 2013, 23, 3–9. [Google Scholar] [CrossRef] [PubMed]
- Ito, K. Recent advances in the systemic treatment of metastatic non-clear cell renal cell carcinomas. Int. J. Urol. 2019, 26, 868–877. [Google Scholar] [CrossRef] [PubMed]
- Padala, S.A.; Barsouk, A.; Thandra, K.C.; Saginala, K.; Mohammed, A.; Vakiti, A.; Rawla, P.; Barsouk, A. Epidemiology of Renal Cell Carcinoma. World J. Oncol. 2020, 11, 79–87. [Google Scholar] [CrossRef] [PubMed]
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer Statistics, 2017. CA Cancer J. Clin. 2017, 67, 7–30. [Google Scholar] [CrossRef] [PubMed]
- Rini, B.I. Metastatic renal cell carcinoma: Many treatment options, one patient. J. Clin. Oncol. 2009, 27, 3225–3234. [Google Scholar] [CrossRef]
- Rini, B.I. New strategies in kidney cancer: Therapeutic advances through understanding the molecular basis of response and resistance. Clin. Cancer Res. 2010, 16, 1348–1354. [Google Scholar] [CrossRef]
- Sorbellini, M.; Kattan, M.W.; Snyder, M.E.; Reuter, V.; Motzer, R.; Goetzl, M.; McKiernan, J.; Russo, P. A postoperative prognostic nomogram predicting recurrence for patients with conventional clear cell renal cell carcinoma. J. Urol. 2005, 173, 48–51. [Google Scholar] [CrossRef]
- Goyal, R.; Gersbach, E.; Yang, X.J.; Rohan, S.M. Differential diagnosis of renal tumors with clear cytoplasm: Clinical relevance of renal tumor subclassification in the era of targeted therapies and personalized medicine. Arch. Pathol. Lab. Med. 2013, 137, 467–480. [Google Scholar] [CrossRef]
- Yang, Y.L.; Li, X.M. The IAP family: Endogenous caspase inhibitors with multiple biological activities. Cell Res. 2000, 10, 169–177. [Google Scholar] [CrossRef] [PubMed]
- Molina, A.M.; Lin, X.; Korytowsky, B.; Matczak, E.; Lechuga, M.J.; Wiltshire, R.; Motzer, R.J. Sunitinib objective response in metastatic renal cell carcinoma: Analysis of 1059 patients treated on clinical trials. Eur. J. Cancer 2014, 50, 351–358. [Google Scholar] [CrossRef] [PubMed]
- Papanikolaou, D.; Ioannidou, P.; Koukourikis, P.; Moysidis, K.; Meditskou, S.; Koutsoumparis, D.; Hatzimouratidis, K.; Hatzivassiliou, E. Systemic therapy for chromophobe renal cell carcinoma: A systematic review. Urol. Oncol. 2020, 38, 137–149. [Google Scholar] [CrossRef] [PubMed]
- Deveraux, Q.L.; Reed, J.C. IAP family proteins--suppressors of apoptosis. Genes Dev. 1999, 13, 239–252. [Google Scholar] [CrossRef]
- Miller, L.K. An exegesis of IAPs: Salvation and surprises from BIR motifs. Trends Cell Biol. 1999, 9, 323–328. [Google Scholar] [CrossRef]
- Altieri, D.C. Targeting survivin in cancer. Cancer Lett. 2013, 332, 225–228. [Google Scholar] [CrossRef]
- Li, F.; Ambrosini, G.; Chu, E.Y.; Plescia, J.; Tognin, S.; Marchisio, P.C.; Altieri, D.C. Control of apoptosis and mitotic spindle checkpoint by survivin. Nature 1998, 396, 580–584. [Google Scholar] [CrossRef]
- Garg, H.; Suri, P.; Gupta, J.C.; Talwar, G.P.; Dubey, S. Survivin: A unique target for tumor therapy. Cancer Cell Int. 2016, 16, 49. [Google Scholar] [CrossRef]
- Salz, W.; Eisenberg, D.; Plescia, J.; Garlick, D.S.; Weiss, R.M.; Wu, X.R.; Sun, T.T.; Altieri, D.C. A survivin gene signature predicts aggressive tumor behavior. Cancer Res. 2005, 65, 3531–3534. [Google Scholar] [CrossRef]
- Ye, C.P.; Qiu, C.Z.; Huang, Z.X.; Su, Q.C.; Zhuang, W.; Wu, R.L.; Li, X.F. Relationship between survivin expression and recurrence, and prognosis in hepatocellular carcinoma. World J. Gastroenterol. 2007, 13, 6264–6268. [Google Scholar] [CrossRef]
- Park, E.; Gang, E.J.; Hsieh, Y.T.; Schaefer, P.; Chae, S.; Klemm, L.; Huantes, S.; Loh, M.; Conway, E.M.; Kang, E.S.; et al. Targeting survivin overcomes drug resistance in acute lymphoblastic leukemia. Blood 2011, 118, 2191–2199. [Google Scholar] [CrossRef] [PubMed]
- Dai, J.B.; Zhu, B.; Lin, W.J.; Gao, H.Y.; Dai, H.; Zheng, L.; Shi, W.H.; Chen, W.X. Identification of prognostic significance of BIRC5 in breast cancer using integrative bioinformatics analysis. Biosci. Rep. 2020, 40, BSR20193678. [Google Scholar] [CrossRef] [PubMed]
- Hennigs, J.K.; Minner, S.; Tennstedt, P.; Loser, R.; Huland, H.; Klose, H.; Graefen, M.; Schlomm, T.; Sauter, G.; Bokemeyer, C.; et al. Subcellular Compartmentalization of Survivin is Associated with Biological Aggressiveness and Prognosis in Prostate Cancer. Sci. Rep. 2020, 10, 3250. [Google Scholar] [CrossRef] [PubMed]
- Shang, X.; Liu, G.; Zhang, Y.; Tang, P.; Zhang, H.; Jiang, H.; Yu, Z. Downregulation of BIRC5 inhibits the migration and invasion of esophageal cancer cells by interacting with the PI3K/Akt signaling pathway. Oncol. Lett. 2018, 16, 3373–3379. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.B.; Wang, W.H. Survivin and pancreatic cancer. World J. Clin. Oncol. 2011, 2, 164–168. [Google Scholar] [CrossRef] [PubMed]
- Tong, X.; Yang, P.; Wang, K.; Liu, Y.; Liu, X.; Shan, X.; Huang, R.; Zhang, K.; Wang, J. Survivin is a prognostic indicator in glioblastoma and may be a target of microRNA-218. Oncol. Lett. 2019, 18, 359–367. [Google Scholar] [CrossRef]
- Cao, Y.; Zhu, W.; Chen, W.; Wu, J.; Hou, G.; Li, Y. Prognostic Value of BIRC5 in Lung Adenocarcinoma Lacking EGFR, KRAS, and ALK Mutations by Integrated Bioinformatics Analysis. Dis. Markers 2019, 2019, 5451290. [Google Scholar] [CrossRef]
- Kitamura, H.; Honma, I.; Torigoe, T.; Hariu, H.; Asanuma, H.; Hirohashi, Y.; Sato, E.; Sato, N.; Tsukamoto, T. Expression of livin in renal cell carcinoma and detection of anti-livin autoantibody in patients. Urology 2007, 70, 38–42. [Google Scholar] [CrossRef]
- Chen, S.; Ma, P.; Li, B.; Zhu, D.; Chen, X.; Xiang, Y.; Wang, T.; Ren, X.; Liu, C.; Jin, X. LncRNA CCAT1 inhibits cell apoptosis of renal cell carcinoma through up-regulation of Livin protein. Mol. Cell. Biochem. 2017, 434, 135–142. [Google Scholar] [CrossRef]
- Shi, Z.G.; Li, S.Q.; Li, Z.J.; Zhu, X.J.; Xu, P.; Liu, G. Expression of vimentin and survivin in clear cell renal cell carcinoma and correlation with p53. Clin. Transl. Oncol. 2015, 17, 65–73. [Google Scholar] [CrossRef]
- Oto, O.A.; Paydas, S.; Tanriverdi, K.; Seydaoglu, G.; Yavuz, S.; Disel, U. Survivin and EPR-1 expression in acute leukemias: Prognostic significance and review of the literature. Leuk. Res. 2007, 31, 1495–1501. [Google Scholar] [CrossRef] [PubMed]
- Berglund, A.; Amankwah, E.K.; Kim, Y.C.; Spiess, P.E.; Sexton, W.J.; Manley, B.; Park, H.Y.; Wang, L.; Chahoud, J.; Chakrabarti, R.; et al. Influence of gene expression on survival of clear cell renal cell carcinoma. Cancer Med. 2020, 9, 8662–8675. [Google Scholar] [CrossRef]
- Ma, C.; Lu, B.; Sun, E. Clinicopathological and prognostic significance of survivin expression in renal cancer patients: A meta-analysis. Postgrad. Med. J. 2017, 93, 186–192. [Google Scholar] [CrossRef] [PubMed]
- Petitprez, F.; Ayadi, M.; Reyniès, A.; Fridman, W.H.; Fridman, C.; Job, S. Review of Prognostic Expression Markers for Clear Cell Renal Cell Carcinoma. Front. Oncol. 2021, 11, 643065. [Google Scholar] [CrossRef]
- Pu, Z.; Wang, Q.; Xie, H.; Wang, G.; Hao, H. Clinicalpathological and prognostic significance of survivin expression in renal cell carcinoma: A meta-analysis. Oncotarget 2017, 8, 19825–19833. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Chen, M.; Dang, C.; Zhang, H.; Wang, X.; Yin, J.; Jia, R.; Zhang, Y. The Early Diagnostic and Prognostic Value of BIRC5 in Clear-Cell Renal Cell Carcinoma Based on the Cancer Genome Atlas Data. Urol. Int. 2022, 106, 344–351. [Google Scholar] [CrossRef] [PubMed]
- Dai, C.H.; Shu, Y.; Chen, P.; Wu, J.N.; Zhu, L.H.; Yuan, R.X.; Long, W.G.; Zhu, Y.M.; Li, J. YM155 sensitizes non-small cell lung cancer cells to EGFR-tyrosine kinase inhibitors through the mechanism of autophagy induction. Biochim. Biophys. Acta Mol. Basis Dis. 2018, 1864, 3786–3798. [Google Scholar] [CrossRef] [PubMed]
- Cheng, S.M.; Chang, Y.C.; Liu, C.Y.; Lee, J.Y.; Chan, H.H.; Kuo, C.W.; Lin, K.Y.; Tsai, S.L.; Chen, S.H.; Li, C.F.; et al. YM155 down-regulates survivin and XIAP, modulates autophagy and induces autophagy-dependent DNA damage in breast cancer cells. Br. J. Pharmacol. 2015, 172, 214–234. [Google Scholar] [CrossRef]
- Wang, Q.; Chen, Z.; Diao, X.; Huang, S. Induction of autophagy-dependent apoptosis by the survivin suppressant YM155 in prostate cancer cells. Cancer Lett. 2011, 302, 29–36. [Google Scholar] [CrossRef]
- Mita, A.C.; Mita, M.M.; Nawrocki, S.T.; Giles, F.J. Survivin: Key regulator of mitosis and apoptosis and novel target for cancer therapeutics. Clin. Cancer Res. 2008, 14, 5000–5005. [Google Scholar] [CrossRef]
- Hong, M.; Ren, M.Q.; Silva, J.; Paul, A.; Wilson, W.D.; Schroeder, C.; Weinberger, P.; Janik, J.; Hao, Z. YM155 inhibits topoisomerase function. Anticancer Drugs 2017, 28, 142–152. [Google Scholar] [CrossRef] [PubMed]
- Kuo, H.H.; Ahmad, R.; Lee, G.Q.; Gao, C.; Chen, H.R.; Ouyang, Z.; Szucs, M.J.; Kim, D.; Tsibris, A.; Chun, T.W.; et al. Anti-apoptotic Protein BIRC5 Maintains Survival of HIV-1-Infected CD4(+) T Cells. Immunity 2018, 48, 1183–1194.e5. [Google Scholar] [CrossRef]
- Jennifer, S.; Carew, J.S.; Claudia, M.; Espitia, C.M.; Weiguo Zhao, W.; Mita, M.M.; Mita, A.C.; Nawrocki, S.T. Targeting Survivin Inhibits Renal Cell Carcinoma Progression and Enhances the Activity of Temsirolimus. Mol. Cancer Ther. 2015, 14, 1404–1413. [Google Scholar] [CrossRef]
- Sim, M.Y.; Shyi, J.S.; Yuen, P.; Go, M.G. Anti-survivin effect of the small molecule inhibitor YM155 in RCC cells is mediated by time-dependent inhibition of the NF-κB pathway. Sci. Rep. 2018, 8, 10289. [Google Scholar] [CrossRef] [PubMed]
- Creyghton, M.P.; Cheng, A.W.; Welstead, G.G.; Kooistra, T.; Carey, B.W.; Steine, E.J.; Hanna, J.; Lodato, M.A.; Frampton, G.M.; Sharp, P.A.; et al. Histone H3K27ac separates active from poised enhancers and predicts developmental state. Proc. Natl. Acad. Sci. USA 2010, 107, 21931–21936. [Google Scholar] [CrossRef] [PubMed]
- Jabbari, K.; Bernardi, G. Cytosine methylation and CpG, TpG (CpA) and TpA frequencies. Gene 2004, 333, 143–149. [Google Scholar] [CrossRef] [PubMed]
- Saxonov, S.; Berg, P.; Brutlag, D.L. A genome-wide analysis of CpG dinucleotides in the human genome distinguishes two distinct classes of promoters. Proc. Natl. Acad. Sci. USA 2006, 103, 1412–1417. [Google Scholar] [CrossRef] [PubMed]
- Deaton, A.M.; Bird, A. CpG islands and the regulation of transcription. Genes Dev. 2011, 25, 1010–1022. [Google Scholar] [CrossRef]
- Pezaro, C.; Woo, H.H.; Davis, I.D. Prostate cancer: Measuring PSA. Intern. Med. J. 2014, 44, 433–440. [Google Scholar] [CrossRef]
- Yang, J.D.; Heimbach, J.K. New advances in the diagnosis and management of hepatocellular carcinoma. BMJ 2020, 371, m3544. [Google Scholar] [CrossRef]
- Swiderska, M.; Choromanska, B.; Dabrowska, E.; Konarzewska-Duchnowska, E.; Choromanska, K.; Szczurko, G.; Mysliwiec, P.; Dadan, J.; Ladny, J.R.; Zwierz, K. The diagnostics of colorectal cancer. Contemp. Oncol. (Pozn.) 2014, 18, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Cheng, S.; Jin, Y.; Zhao, Y.; Wang, Y. Roles of CA125 in diagnosis, prediction, and oncogenesis of ovarian cancer. Biochim. Biophys. Acta Rev. Cancer 2021, 1875, 188503. [Google Scholar] [CrossRef] [PubMed]
- Luo, G.; Jin, K.; Deng, S.; Cheng, H.; Fan, Z.; Gong, Y.; Qian, Y.; Huang, Q.; Ni, Q.; Liu, C.; et al. Roles of CA19-9 in pancreatic cancer: Biomarker, predictor and promoter. Biochim. Biophys. Acta Rev. Cancer 2021, 1875, 188409. [Google Scholar] [CrossRef] [PubMed]
- Haake, S.M.; Weyandt, J.D.; Rathmell, W.K. Insights into the Genetic Basis of the Renal Cell Carcinomas from The Cancer Genome Atlas. Mol. Cancer Res. 2016, 14, 589–598. [Google Scholar] [CrossRef]
- Batavia, A.A.; Schraml, P.; Moch, H. Clear cell renal cell carcinoma with wild-type von Hippel-Lindau gene: A non-existent or new tumour entity? Histopathology 2019, 74, 60–67. [Google Scholar] [CrossRef]
- Lindor, N.M.; Dechet, C.B.; Greene, M.H.; Jenkins, R.B.; Zincke, M.T.; Weaver, A.L.; Wilson, M.; Zincke, H.; Liu, W. Papillary renal cell carcinoma: Analysis of germline mutations in the MET proto-oncogene in a clinic-based population. Genet. Test. 2001, 5, 101–106. [Google Scholar] [CrossRef]
- Casuscelli, J.; Weinhold, N.; Gundem, G.; Wang, L.; Zabor, E.C.; Drill, E.; Wang, P.I.; Nanjangud, G.J.; Redzematovic, A.; Nargund, A.M.; et al. Genomic landscape and evolution of metastatic chromophobe renal cell carcinoma. JCI Insight 2017, 2, e92688. [Google Scholar] [CrossRef]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hong, S.H.; Lee, Y.J.; Jang, E.B.; Hwang, H.J.; Kim, E.S.; Son, D.H.; Park, S.Y.; Moon, H.S.; Yoon, Y.E. Therapeutic Efficacy of YM155 to Regulate an Epigenetic Enzyme in Major Subtypes of RCC. Int. J. Mol. Sci. 2024, 25, 216. https://doi.org/10.3390/ijms25010216
Hong SH, Lee YJ, Jang EB, Hwang HJ, Kim ES, Son DH, Park SY, Moon HS, Yoon YE. Therapeutic Efficacy of YM155 to Regulate an Epigenetic Enzyme in Major Subtypes of RCC. International Journal of Molecular Sciences. 2024; 25(1):216. https://doi.org/10.3390/ijms25010216
Chicago/Turabian StyleHong, Seong Hwi, Young Ju Lee, Eun Bi Jang, Hyun Ji Hwang, Eun Song Kim, Da Hyeon Son, Sung Yul Park, Hong Sang Moon, and Young Eun Yoon. 2024. "Therapeutic Efficacy of YM155 to Regulate an Epigenetic Enzyme in Major Subtypes of RCC" International Journal of Molecular Sciences 25, no. 1: 216. https://doi.org/10.3390/ijms25010216





