Puerarin Modulates Hepatic Farnesoid X Receptor and Gut Microbiota in High-Fat Diet-Induced Obese Mice
Abstract
:1. Introduction
2. Results
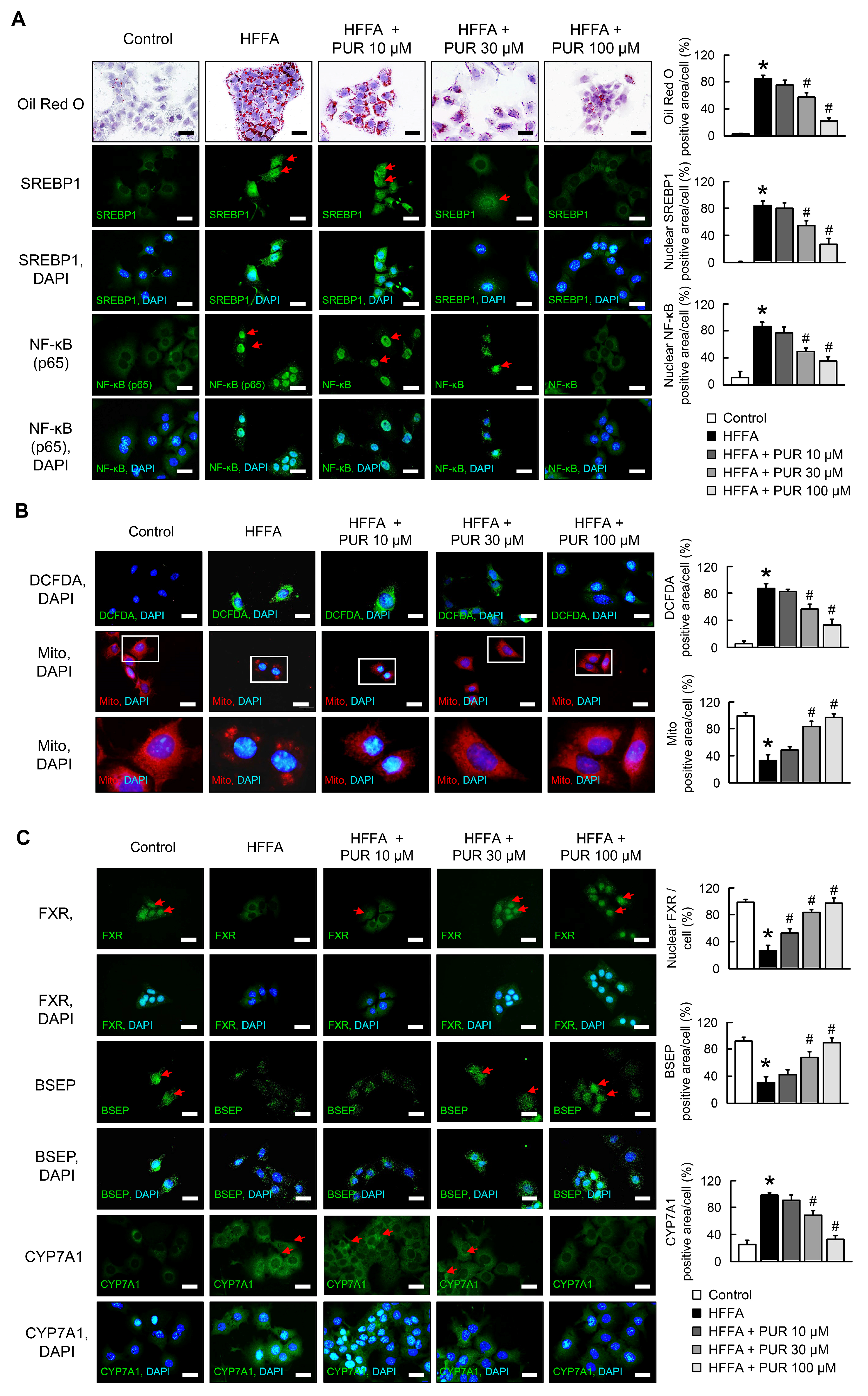
2.1. Puerarin Ameliorates HFFA-Induced Hepatic Lipid Accumulation, Mitochondrial Dysfunction, and FXR Signaing
2.2. Puerarin Modulation of HFFA-Mediated Hepatotoxicity in siFXR AML12 Cells
2.3. Puerarin Alleviates HFFA-Induced Inflammation and Mitochondrial Dysfunction in Hepatocytes
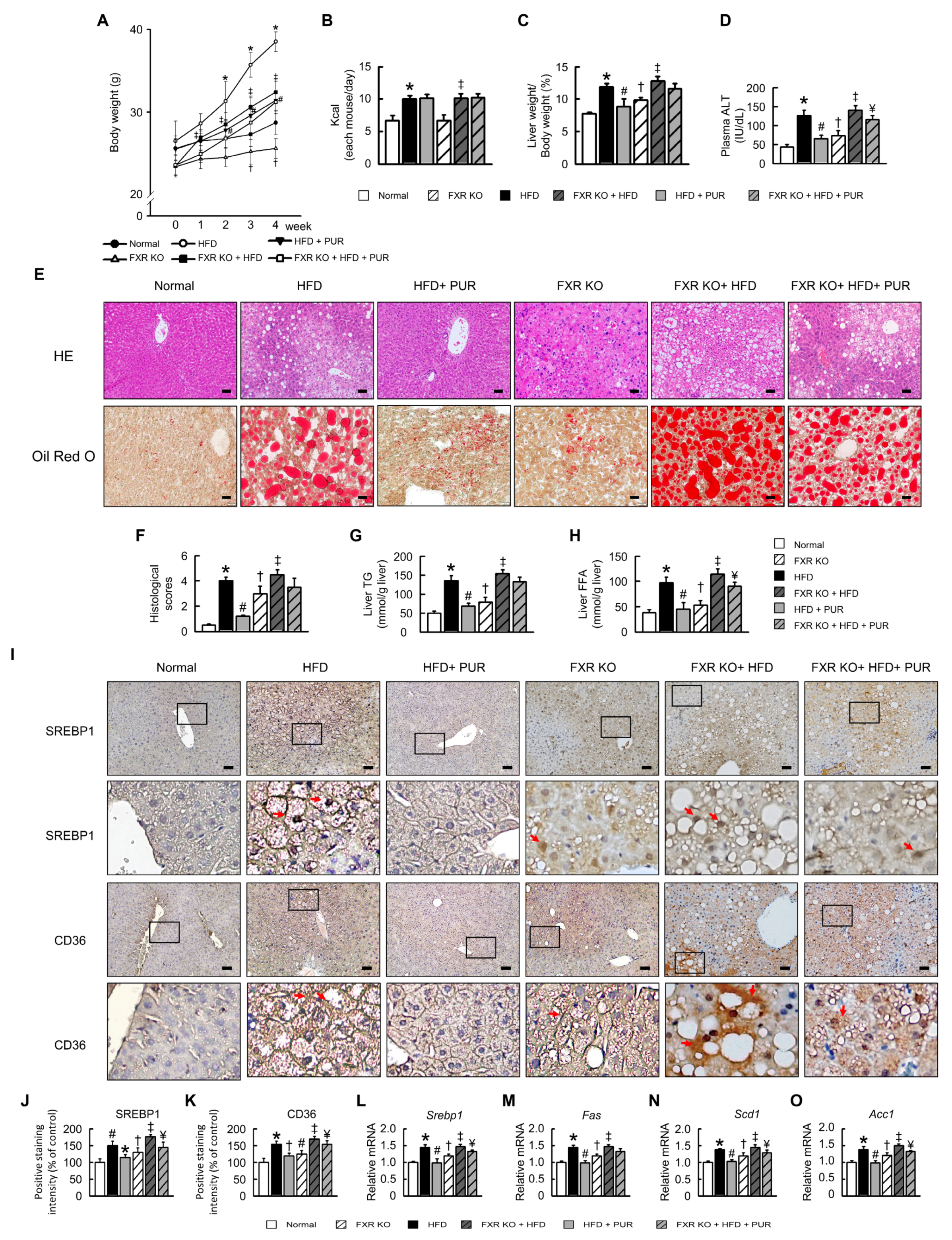
2.4. Puerarin Improves Liver Damage and Lipid Accumulation in HFD-Induced Obesity Mice
2.5. Puerarin Suppresses Inflammatory Response in Liver of HFD-Induced Obesity Mice
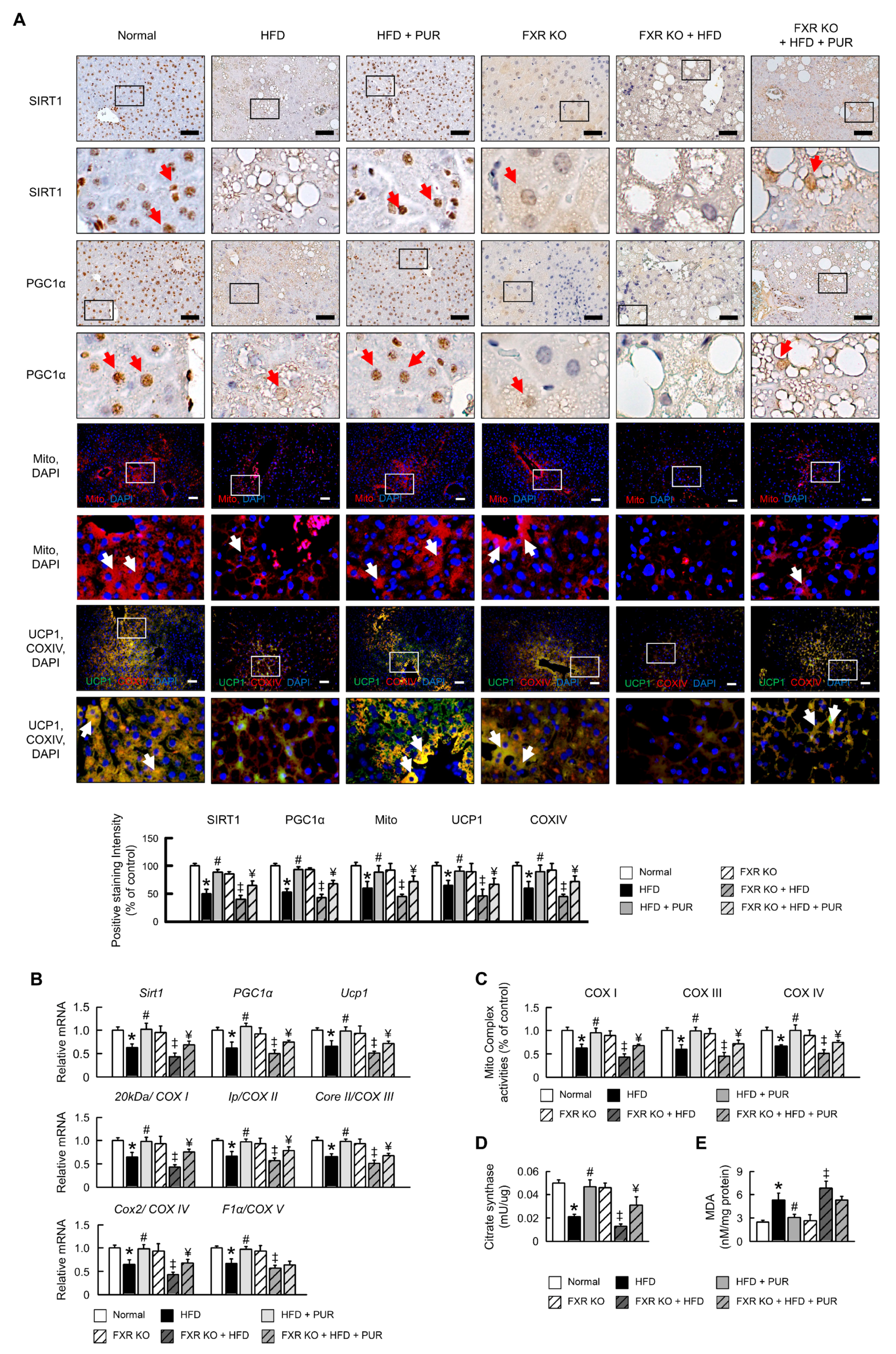
2.6. Regulation of Mitochondrial Biogenesis by Puerarin in HFD-Induced Obesity Mice
2.7. Regulation of Mitophagy and the Ubiquitin-Proteasome System by Puerarin in Obese Mice
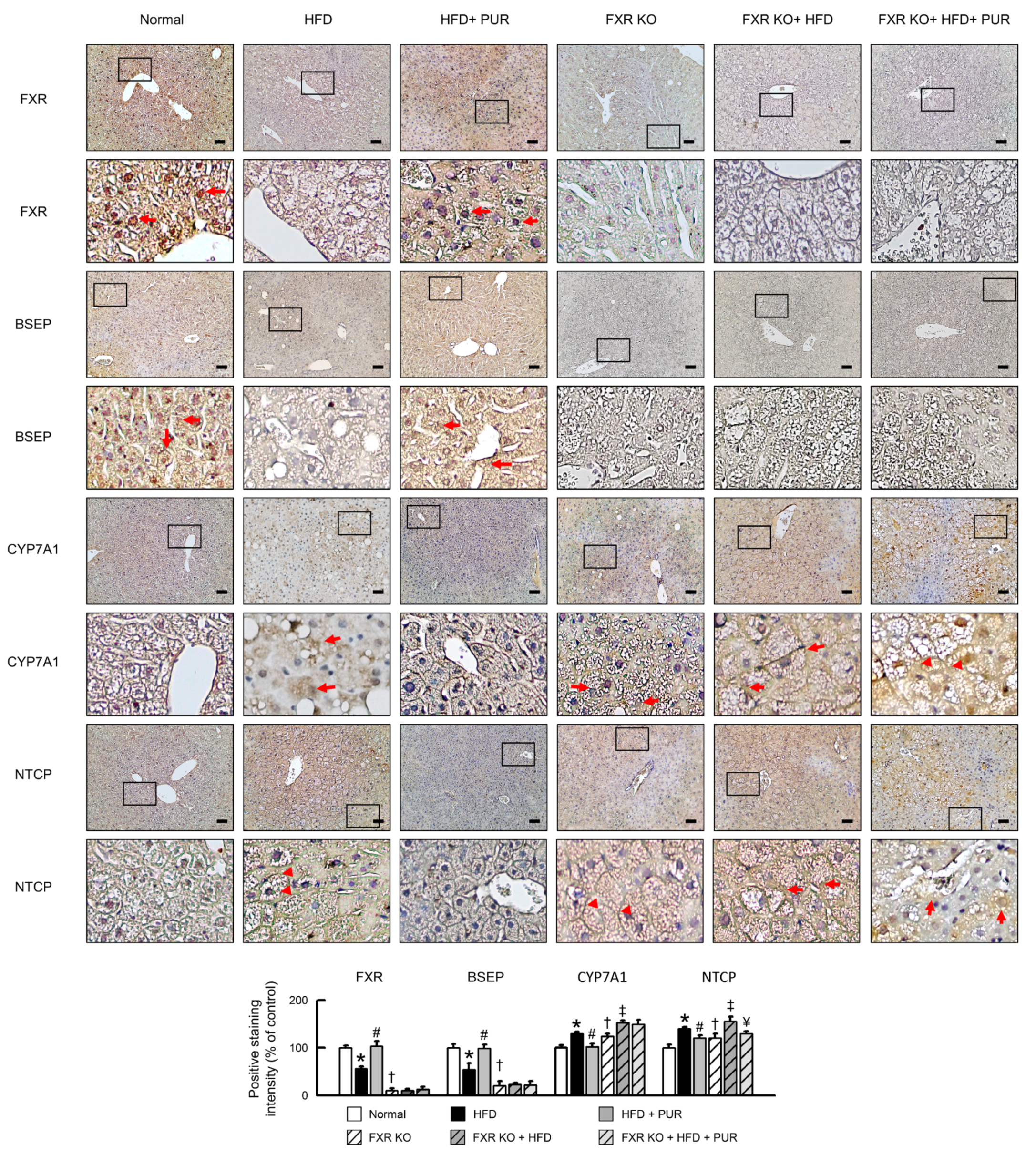
2.8. Impact of Puerarin on Bile Acid Transport Proteins in Obese Mice
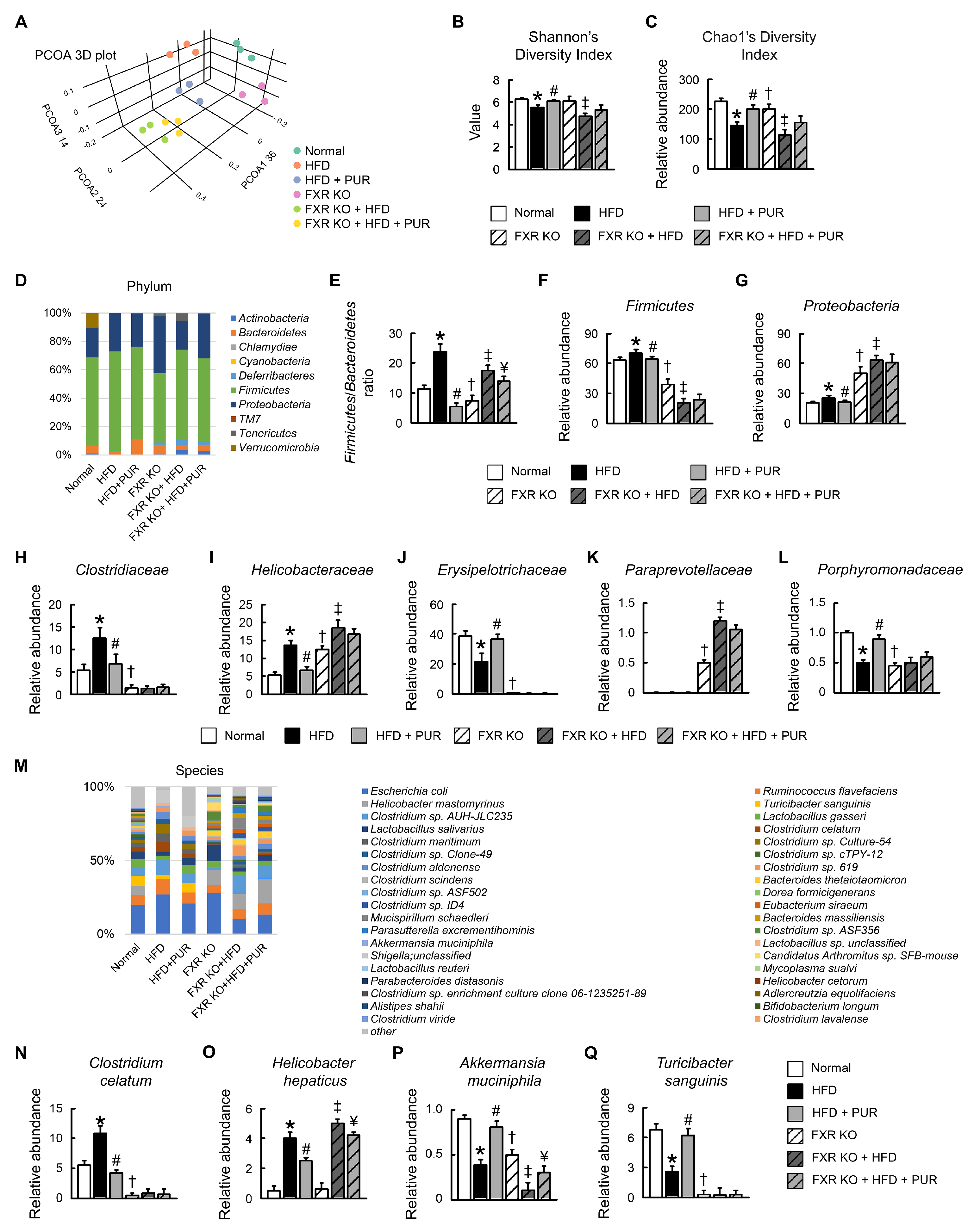
2.9. Puerarin Modulates Gut Microbiota Composition in HFD-Induced Obesity Mice
3. Discussion
4. Materials and Methods
4.1. Cell Culture
4.2. siRNA Transfection
4.3. Mitochondrial Analysis
4.4. Oil Red O Staining
4.5. Mitochondria Mass and DNA Assay and Mitochondrial Respiratory Complexes
4.6. Flow Cytometry for ROS and Mitochondrial Membrane Potential Assay
4.7. RNA Isolation and Real-Time PCR Analyses
4.8. Immunofluorescence Staining
4.9. Experimental Animals
4.10. Histopathology and Immunohistochemistry
4.11. Gut Microbiota Analysis via 16S rRNA and Intestinal Tissue DNA Extraction
4.12. 16S rRNA Analysis
4.13. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- Kahn, S.E.; Hull, R.L.; Utzschneider, K.M. Mechanisms linking obesity to insulin resistance and type 2 diabetes. Nature 2006, 444, 840–846. [Google Scholar] [CrossRef] [PubMed]
- Eckel, R.H.; Grundy, S.M.; Zimmet, P.Z. The metabolic syndrome. Lancet 2005, 365, 1415–1428. [Google Scholar] [CrossRef]
- Fabbrini, E.; Sullivan, S.; Klein, S. Obesity and nonalcoholic fatty liver disease: Biochemical, metabolic, and clinical implications. Hepatology 2010, 51, 679–689. [Google Scholar] [CrossRef]
- Segura, J.; Ruilope, L.M. Obesity, essential hypertension and renin-angiotensin system. Public Health Nutr. 2007, 10, 1151–1155. [Google Scholar] [CrossRef]
- Shimano, H. SREBPs: Physiology and pathophysiology of the SREBP family. FEBS J. 2009, 276, 616–621. [Google Scholar] [CrossRef]
- Kim, F.; Pham, M.; Luttrell, I.; Bannerman, D.D.; Tupper, J.; Thaler, J.; Hawn, T.R.; Raines, E.W.; Schwartz, M.W. Toll-like receptor-4 mediates vascular inflammation and insulin resistance in diet-induced obesity. Circ. Res. 2007, 100, 1589–1596. [Google Scholar] [CrossRef]
- Shi, H.; Kokoeva, M.V.; Inouye, K.; Tzameli, I.; Yin, H.; Flier, J.S. TLR4 links innate immunity and fatty acid-induced insulin resistance. J. Clin. Investig. 2006, 116, 3015–3025. [Google Scholar] [CrossRef] [PubMed]
- Tilg, H.; Moschen, A.R. Evolution of inflammation in nonalcoholic fatty liver disease: The multiple parallel hits hypothesis. Hepatology 2010, 52, 1836–1846. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Kamat, A.; Pergola, P.; Swamy, A.; Tio, F.; Cusi, K. Metabolic factors in the development of hepatic steatosis and altered mitochondrial gene expression in vivo. Metabolism 2011, 60, 1090–1099. [Google Scholar] [CrossRef]
- Jiao, N.; Baker, S.S.; Chapa-Rodriguez, A.; Liu, W.; Nugent, C.A.; Tsompana, M.; Mastrandrea, L.; Buck, M.J.; Baker, R.D.; Genco, R.J.; et al. Suppressed hepatic bile acid signalling despite elevated production of primary and secondary bile acids in NAFLD. Gut 2018, 67, 1881–1891. [Google Scholar] [CrossRef]
- Jiang, C.; Xie, C.; Lv, Y.; Li, J.; Krausz, K.W.; Shi, J.; Brocker, C.N.; Desai, D.; Amin, S.G.; Bisson, W.H.; et al. Intestine-selective farnesoid X receptor inhibition improves obesity-related metabolic dysfunction. Nat. Commun. 2015, 6, 10166. [Google Scholar] [CrossRef]
- Sayin, S.I.; Wahlström, A.; Felin, J.; Jäntti, S.; Marschall, H.U.; Bamberg, K.; Angelin, B.; Hyötyläinen, T.; Orešič, M.; Bäckhed, F. Gut microbiota regulates bile acid metabolism by reducing the levels of tauro-beta-muricholic acid, a naturally occurring FXR antagonist. Cell Metab. 2013, 17, 225–235. [Google Scholar] [CrossRef]
- Wahlström, A.; Sayin, S.I.; Marschall, H.U.; Bäckhed, F. Intestinal Crosstalk between Bile Acids and Microbiota and Its Impact on Host Metabolism. Cell Metab. 2016, 24, 41–50. [Google Scholar] [CrossRef]
- Meng, F.; Guo, B.; Ma, Y.Q.; Li, K.W.; Niu, F.J. Puerarin: A review of its mechanisms of action and clinical studies in ophthalmology. Phytomedicine 2022, 107, 154465. [Google Scholar] [CrossRef]
- Wang, S.; Zhang, S.; Wang, S.; Gao, P.; Dai, L. A comprehensive review on Pueraria: Insights on its chemistry and medicinal value. Biomed. Pharmacother. 2020, 131, 110734. [Google Scholar] [CrossRef]
- Li, W.; Zhao, W.; Wu, Q.; Lu, Y.; Shi, J.; Chen, X. Puerarin Improves Diabetic Aorta Injury by Inhibiting NADPH Oxidase-Derived Oxidative Stress in STZ-Induced Diabetic Rats. J. Diabetes. Res. 2016, 2016, 8541520. [Google Scholar] [CrossRef]
- Ling, C.; Liang, J.; Zhang, C.; Li, R.; Mou, Q.; Qin, J.; Li, X.; Wang, J. Synergistic Effects of Salvianolic Acid B and Puerarin on Cerebral Ischemia Reperfusion Injury. Molecules 2018, 23, 564. [Google Scholar] [CrossRef]
- Liu, S.H.; Cheng, Y.C. Old formula, new Rx: The journey of PHY906 as cancer adjuvant therapy. J. Ethnopharmacol. 2012, 140, 614–623. [Google Scholar] [CrossRef]
- Hwang, S.L.; Shih, P.H.; Yen, G.C. Neuroprotective effects of citrus flavonoids. J. Ethnopharmacol. 2012, 60, 877–885. [Google Scholar] [CrossRef] [PubMed]
- Veteläinen, R.L.; Bennink, R.J.; de Bruin, K.; van Vliet, A.; van Gulik, T.M. Hepatobiliary function assessed by 99mTc-mebrofenin cholescintigraphy in the evaluation of severity of steatosis in a rat model. Eur. J. Nucl. Med. Mol. Imaging 2006, 33, 1107–1114. [Google Scholar] [CrossRef] [PubMed]
- Ryan, K.K.; Tremaroli, V.; Clemmensen, C.; Kovatcheva-Datchary, P.; Myronovych, A.; Karns, R.; Wilson-Pérez, H.E.; Sandoval, D.A.; Kohli, R.; Bäckhed, F.; et al. FXR is a molecular target for the effects of vertical sleeve gastrectomy. Nature 2014, 509, 183–188. [Google Scholar] [CrossRef]
- Joyce, S.A.; MacSharry, J.; Casey, P.G.; Kinsella, M.; Murphy, E.F.; Shanahan, F.; Hill, C.; Gahan, C.G. Regulation of host weight gain and lipid metabolism by bacterial bile acid modification in the gut. Proc. Natl. Acad. Sci. USA 2014, 111, 7421–7426. [Google Scholar] [CrossRef]
- Mullish, B.H.; Pechlivanis, A.; Barker, G.F.; Thursz, M.R.; Marchesi, J.R.; McDonald, J.A.K. Functional microbiomics: Evaluation of gut microbiota-bile acid metabolism interactions in health and disease. Methods 2018, 149, 49–58. [Google Scholar] [CrossRef]
- Zhang, Y.; Lee, F.Y.; Barrera, G.; Lee, H.; Vales, C.; Gonzalez, F.J.; Willson, T.M.; Edwards, P.A. Activation of the nuclear receptor FXR improves hyperglycemia and hyperlipidemia in diabetic mice. Proc. Natl. Acad. Sci. USA 2006, 103, 1006–1011. [Google Scholar] [CrossRef]
- Yang, F.; Huang, X.; Yi, T.; Yen, Y.; Moore, D.D.; Huang, W. Spontaneous development of liver tumors in the absence of the bile acid receptor farnesoid X receptor. Cancer Res. 2007, 67, 863–867. [Google Scholar] [CrossRef]
- Zhao, Y.Z.; Zhang, L.; Gupta, P.K.; Tian, F.R.; Mao, K.L.; Qiu, K.Y.; Yang, W.; Lv, C.Z.; Lu, C.T. Using PG-Liposome-Based System to Enhance Puerarin Liver-Targeted Therapy for Alcohol-Induced Liver Disease. AAPS PharmSciTech 2016, 17, 1376–1382. [Google Scholar] [CrossRef]
- Wang, S.; Shi, X.L.; Feng, M.; Wang, X.; Zhang, Z.H.; Zhao, X.; Han, B.; Ma, H.C.; Dai, B.; Ding, Y.T. Puerarin protects against CCl4-induced liver fibrosis in mice: Possible role of PARP-1 inhibition. Int. Immunopharmacol. 2016, 38, 238–245. [Google Scholar] [CrossRef]
- Liu, T.; Xu, G.; Liang, L.; Xiao, X.; Zhao, Y.; Bai, Z. Pharmacological effects of Chinese medicine modulating NLRP3 inflammasomes in fatty liver treatment. Front. Pharmacol. 2022, 13, 967594. [Google Scholar] [CrossRef]
- Bindl, E.; Lang, W.; Rau, W. Light dependent carotenoid synthesis: VI. Time course of synthesis of various carotenoids in Fusarium aquaeductuum after various inductive treatments. Planta 1970, 94, 156–174. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Xue, H.; Wang, W.; Wang, Y. Parental Expectations and Child Screen and Academic Sedentary Behaviors in China. Am. J. Prev. Med. 2017, 52, 680–689. [Google Scholar] [CrossRef] [PubMed]
- Levy, F.; Bohbot, Y.; Sanhadji, K.; Rusinaru, D.; Ringle, A.; Delpierre, Q.; Smaali, S.; Gun, M.; Marechaux, S.; Tribouilloy, C. Impact of pulmonary hypertension on long-term outcome in patients with severe aortic stenosis. Eur. Heart. J. Cardiovasc. 913 Imaging 2018, 19, 553–561. [Google Scholar] [CrossRef]
- Gan, C.; Cai, Q.; Tang, C.; Gao, J. Inflammasomes and Pyroptosis of Liver Cells in Liver Fibrosis. Front. Immunol. 2022, 13, 896473. [Google Scholar] [CrossRef]
- Neil, J.A.; Laurent, D.; Das, K.; Mehta, H. Retrograde cysto-myelogram: Case Report. Surg. Neurol. Int. 2015, 6, S124–S126. [Google Scholar]
- Li, S.; Xu, Y.; Guo, W.; Chen, F.; Zhang, C.; Tan, H.Y.; Wang, N.; Feng, Y. The Impacts of Herbal Medicines and Natural Products on Regulating the Hepatic Lipid Metabolism. Front. Pharmacol. 2020, 11, 351. [Google Scholar] [CrossRef]
- Morant-Ferrando, B.; Jimenez-Blasco, D.; Alonso-Batan, P.; Agulla, J.; Lapresa, R.; Garcia-Rodriguez, D.; Yunta-Sanchez, S.; Lopez-Fabuel, I.; Fernandez, E.; Carmeliet, P.; et al. Fatty acid oxidation organizes mitochondrial supercomplexes to sustain astrocytic ROS and cognition. Nat. Metab. 2023, 5, 1290–1302. [Google Scholar] [CrossRef]
- Patergnani, S.; Bouhamida, E.; Leo, S.; Pinton, P.; Rimessi, A. Mitochondrial Oxidative Stress and “Mito-Inflammation”: Actors in the Diseases. Biomedicines 2021, 9, 216. [Google Scholar] [CrossRef]
- Anand, P.K. Lipids, inflammasomes, metabolism, and disease. Immunol. Rev. 2020, 297, 108–122. [Google Scholar] [CrossRef]
- Fuller, K.N.Z.; McCoin, C.S.; Von Schulze, A.T.; Houchen, C.J.; Choi, M.A.; Thyfault, J.P. Estradiol treatment or modest exercise improves hepatic health and mitochondrial outcomes in female mice following ovariectomy. Am. J. Physiol. Endocrinol. Metab. 2021, 320, E1020–E1031. [Google Scholar] [CrossRef]
- Fu, T.; Coulter, S.; Yoshihara, E.; Oh, T.G.; Fang, S.; Cayabyab, F.; Zhu, Q.; Zhang, T.; Leblanc, M.; Liu, S.; et al. FXR Regulates Intestinal Cancer Stem Cell Proliferation. Cell 2019, 176, 1098–1112. [Google Scholar] [CrossRef]
- Liu, H.M.; Chang, Z.Y.; Yang, C.W.; Chang, H.H.; Lee, T.Y. Farnesoid X Receptor Agonist GW4064 Protects Lipopolysaccharide-Induced Intestinal Epithelial Barrier Function and Colorectal Tumorigenesis Signaling through the αKlotho/βKlotho/FGFs Pathways in Mice. Int. J. Mol. Sci. 2023, 24, 16932. [Google Scholar] [CrossRef]
- Wei, S.; He, T.; Zhao, X.; Jing, M.; Li, H.; Chen, L.; Zheng, R.; Zhao, Y. Alterations in the gut microbiota and serum metabolomics of spontaneous cholestasis caused by loss of FXR signal in mice. Front. Pharmacol. 2023, 14, 1197847. [Google Scholar] [CrossRef] [PubMed]
- Jiang, C.; Xie, C.; Li, F.; Zhang, L.; Nichols, R.G.; Krausz, K.W.; Cai, J.; Qi, Y.; Fang, Z.Z.; Takahashi, S.; et al. Intestinal farnesoid X receptor signaling promotes nonalcoholic fatty liver disease. J. Clin. Investig. 2015, 125, 386–402. [Google Scholar] [CrossRef] [PubMed]
- Younossi, Z.M.; Stepanova, M.; Noureddin, M.; Kowdley, K.V.; Strasser, S.I.; Kohli, A.; Ruane, P.; Shiffman, M.L.; Sheikh, A.; Gunn, N.; et al. Improvements of Fibrosis and Disease Activity Are Associated With Improvement of Patient-Reported Outcomes in Patients With Advanced Fibrosis Due to Nonalcoholic Steatohepatitis. Hepatol. Commun. 2021, 5, 1201–1211. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Chen, W.D.; Wang, Y.D. Nuclear receptors: A bridge linking the gut microbiome and the host. Mol. Med. 2021, 27, 144. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Qin, P.; Wang, J. High-Fat Diet Alters the Intestinal Microbiota in Streptozotocin-Induced Type 2 Diabetic Mice. Microorganisms 2019, 7, 176. [Google Scholar] [CrossRef] [PubMed]
- Jena, P.K.; Sheng, L.; Liu, H.X.; Kalanetra, K.M.; Mirsoian, A.; Murphy, W.J.; French, S.W.; Krishnan, V.V.; Mills, D.A.; Wan, Y.Y. Western Diet-Induced Dysbiosis in Farnesoid X Receptor Knockout Mice Causes Persistent Hepatic Inflammation after Antibiotic Treatment. Am. J. Pathol. 2017, 187, 1800–1813. [Google Scholar] [CrossRef] [PubMed]
- Inagaki, T.; Moschetta, A.; Lee, Y.K.; Peng, L.; Zhao, G.; Downes, M.; Yu, R.T.; Shelton, J.M.; Richardson, J.A.; Repa, J.J.; et al. Regulation of antibacterial defense in the small intestine by the nuclear bile acid receptor. Proc. Natl. Acad. Sci. USA 2006, 103, 3920–3925. [Google Scholar] [CrossRef] [PubMed]
- Jia, W.; Xie, G.; Jia, W. Bile acid-microbiota crosstalk in gastrointestinal inflammation and carcinogenesis. Nat. Rev. Gastroenterol. Hepatol. 2018, 15, 111–128. [Google Scholar] [CrossRef] [PubMed]
- Jena, P.K.; Sheng, L.; Nguyen, M.; Di Lucente, J.; Hu, Y.; Li, Y.; Maezawa, I.; Jin, L.W.; Wan, Y.Y. Dysregulated bile acid receptor-mediated signaling and IL-17A induction are implicated in diet-associated hepatic health and cognitive function. Biomark. Res. 2020, 8, 59. [Google Scholar]
- Wang, L.; Wu, Y.; Zhuang, L.; Chen, X.; Min, H.; Song, S.; Liang, Q.; Li, A.D.; Gao, Q. Puerarin prevents high-fat diet-induced obesity by enriching Akkermansia muciniphila in the gut microbiota of mice. PLoS ONE 2019, 14, e0218490. [Google Scholar]
- Ma, M.H.; Gao, L.L.; Chen, C.B.; Gu, F.L.; Wu, S.Q.; Li, F.; Han, B.X. Dendrobium huoshanense Polysaccharide Improves High-Fat Diet Induced Liver Injury by Regulating the Gut-Liver Axis. Chem. Biodivers. 2023, 20, e202300980. [Google Scholar] [CrossRef]
- Thangamani, S.; Monasky, R.; Lee, J.K.; Antharam, V.; HogenEsch, H.; Hazbun, T.R.; Jin, Y.; Gu, H.; Guo, G.L. Bile Acid Regulates the Colonization and Dissemination of Candida albicans from the Gastrointestinal Tract by Controlling Host Defense System and Microbiota. J. Fungi 2021, 7, 1030. [Google Scholar] [CrossRef]
- Fox, J.G.; Dewhirst, F.E.; Tully, J.G.; Paster, B.J.; Yan, L.; Taylor, N.S.; Collins, M.J., Jr.; Gorelick, P.L.; Ward, J.M. Helicobacter hepaticus sp. nov., a microaerophilic bacterium isolated from livers and intestinal mucosal scrapings from mice. J. Clin. Microbiol. 1994, 32, 1238–1245. [Google Scholar] [CrossRef]
- Ward, J.M.; Anver, M.R.; Haines, D.C.; Benveniste, R.E. Chronic active hepatitis in mice caused by Helicobacter hepaticus. Am. J. Pathol. 1994, 145, 959–968. [Google Scholar]
- Alvarez, C.S.; Florio, A.A.; Butt, J.; Rivera-Andrade, A.; Kroker-Lobos, M.F.; Waterboer, T.; Camargo, M.C.; Freedman, N.D.; Graubard, B.I.; Lazo, M.; et al. Associations between Helicobacter pylori with nonalcoholic fatty liver disease and other metabolic conditions in Guatemala. Helicobacter 2020, 25, e127566. [Google Scholar] [CrossRef]
- Kohli, R.; Pan, X.; Malladi, P.; Wainwright, M.S.; Whitington, P.F. Mitochondrial reactive oxygen species signal hepatocyte steatosis by regulating the phosphatidylinositol 3-kinase cell survival pathway. J. Biol. Chem. 2007, 282, 21327–21336. [Google Scholar] [CrossRef]
- Kuo, J.J.; Chang, H.H.; Tsai, T.H.; Lee, T.Y. Curcumin ameliorates mitochondrial dysfunction associated with inhibition of gluconeogenesis in free fatty acid-mediated hepatic lipoapoptosis. Int. J. Mol. Med. 2012, 30, 643–649. [Google Scholar] [CrossRef]
- Wang, C.H.; Liu, H.M.; Chang, Z.Y.; Lee, M.C.; Hsu, C.H.; Lee, T.Y. Antioxidants Rich Herbal Formula Ger-Gen-Chyn-Lian-Tang Protects Lipotoxicity and Ameliorates Inflammation Signaling through Regulation of Mitochondrial Biogenesis and Mitophagy in Nonalcoholic Fatty Liver Disease Mice. Front. Biosci. 2022, 27, 242. [Google Scholar] [CrossRef]
- de Haan, L.R.; Verheij, J.; van Golen, R.F.; Horneffer-van der Sluis, V.; Lewis, M.R.; Beuers, U.H.W.; van Gulik, T.M.; Olde Damink, S.W.M.; Schaap, F.G.; Heger, M.; et al. Unaltered Liver Regeneration in Post-Cholestatic Rats Treated with the FXR Agonist Obeticholic Acid. Biomolecules 2021, 11, 260. [Google Scholar] [CrossRef]
- Edgar, R.C. UPARSE: Highly accurate OTU sequences from microbial amplicon reads. Nat. Methods 2013, 10, 996–998. [Google Scholar] [CrossRef] [PubMed]
- Langmead, B.; Salzberg, S.L. Fast gapped-read alignment with Bowtie 2. Nat. Methods 2012, 9, 357–359. [Google Scholar] [CrossRef]
- Metsalu, T.; Vilo, J. ClustVis: A web tool for visualizing clustering of multivariate data using Principal Component Analysis and heatmap. Nucleic Acids Res. 2015, 43, W566–W570. [Google Scholar] [CrossRef]




| Histological | Severity | Description | Score |
|---|---|---|---|
| Steatosis | Absent | <10% | 0 |
| Mild | 10–30% | 1 | |
| Marked | 31–60% | 2 | |
| Severe | >60% | 3 | |
| Inflammation | None | 0 | |
| Moderate | Scattered a | 1 | |
| Marked | Focia a | 2 | |
| Severe | Diffuse a | 3 | |
| Necrosis | Absent | 0% | 0 |
| Mild | <10% | 1 | |
| Marked | 10–30% | 2 | |
| Severe | >50% | 3 | |
| Fibrosis b | Absent | 0 | |
| Mild | 1 | ||
| Marked | 2 | ||
| Severe | 3 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, C.-W.; Liu, H.-M.; Chang, Z.-Y.; Liu, G.-H.; Chang, H.-H.; Huang, P.-Y.; Lee, T.-Y. Puerarin Modulates Hepatic Farnesoid X Receptor and Gut Microbiota in High-Fat Diet-Induced Obese Mice. Int. J. Mol. Sci. 2024, 25, 5274. https://doi.org/10.3390/ijms25105274
Yang C-W, Liu H-M, Chang Z-Y, Liu G-H, Chang H-H, Huang P-Y, Lee T-Y. Puerarin Modulates Hepatic Farnesoid X Receptor and Gut Microbiota in High-Fat Diet-Induced Obese Mice. International Journal of Molecular Sciences. 2024; 25(10):5274. https://doi.org/10.3390/ijms25105274
Chicago/Turabian StyleYang, Ching-Wei, Hsuan-Miao Liu, Zi-Yu Chang, Geng-Hao Liu, Hen-Hong Chang, Po-Yu Huang, and Tzung-Yan Lee. 2024. "Puerarin Modulates Hepatic Farnesoid X Receptor and Gut Microbiota in High-Fat Diet-Induced Obese Mice" International Journal of Molecular Sciences 25, no. 10: 5274. https://doi.org/10.3390/ijms25105274






