Promising Role of Alkaloids in the Prevention and Treatment of Thyroid Cancer and Autoimmune Thyroid Disease: A Comprehensive Review of the Current Evidence
Abstract
:1. Introduction
2. Results
2.1. Alkaloids and Thyroid Cancer
2.1.1. Alkaloids Effects on Cell Proliferation and Cell Death
2.1.2. Alkaloids Effects on Radioresistance and Chemosensitivity
2.2. Alkaloids and Autoimmune Thyroid Diseases
3. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Zaynab, M.; Fatima, M.; Abbas, S.; Sharif, Y.; Umair, M.; Zafar, M.H.; Bahadar, K. Role of secondary metabolites in plant defense against pathogens. Microb. Pathog. 2018, 124, 198–202. [Google Scholar] [CrossRef] [PubMed]
- Bhambhani, S.; Kondhare, K.R.; Giri, A.P. Diversity in Chemical Structures and Biological Properties of Plant Alkaloids. Molecules 2021, 26, 3374. [Google Scholar] [CrossRef] [PubMed]
- Dey, P.; Kundu, A.; Kumar, A.; Gupta, M.; Lee, B.M.; Bhakta, T.; Dash, S.; Kim, H.S. Analysis of alkaloids (indole alkaloids, isoquinoline alkaloids, tropane alkaloids). In Recent Advances in Natural Products Analysis; Elsevier: Amsterdam, The Netherlands, 2020; pp. 505–567. [Google Scholar]
- Thawabteh, A.; Juma, S.; Bader, M.; Karaman, D.; Scrano, L.; Bufo, S.A.; Karaman, R. The Biological Activity of Natural Alkaloids against Herbivores, Cancerous Cells and Pathogens. Toxins 2019, 11, 656. [Google Scholar] [CrossRef] [PubMed]
- Alasvand, M.; Assadollahi, V.; Ambra, R.; Hedayati, E.; Kooti, W.; Peluso, I. Antiangiogenic Effect of Alkaloids. Oxidative Med. Cell. Longev. 2019, 2019, 9475908. [Google Scholar] [CrossRef] [PubMed]
- Mondal, A.; Gandhi, A.; Fimognari, C.; Atanasov, A.G.; Bishayee, A. Alkaloids for cancer prevention and therapy: Current progress and future perspectives. Eur. J. Pharmacol. 2019, 858, 172472. [Google Scholar] [CrossRef] [PubMed]
- Olofinsan, K.; Abrahamse, H.; George, B.P. Therapeutic Role of Alkaloids and Alkaloid Derivatives in Cancer Management. Molecules 2023, 28, 5578. [Google Scholar] [CrossRef] [PubMed]
- Khan, H.; Ullah, H.; Khattak, S.; Aschner, M.; Aguilar, C.N.; Halimi, S.M.A.; Cauli, O.; Shah, S.M.M. Therapeutic potential of alkaloids in autoimmune diseases: Promising candidates for clinical trials. Phytother. Res. 2020, 35, 50–62. [Google Scholar] [CrossRef] [PubMed]
- Patel, A.; Vanecha, R.; Patel, J.; Patel, D.; Shah, U.; Bambharoliya, T. Development of Natural Bioactive Alkaloids: Anticancer Perspective. Mini-Rev. Med. Chem. 2022, 22, 200–212. [Google Scholar] [CrossRef] [PubMed]
- Efferth, T.; Oesch, F. Repurposing of plant alkaloids for cancer therapy: Pharmacology and toxicology. Semin. Cancer Biol. 2021, 68, 143–163. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Wu, Y.; Li, Y.; Li, S.; Liu, J.; Yang, X.; Xia, G.; Wang, G. Natural products and derivatives for breast cancer treatment: From drug discovery to molecular mechanism. Phytomedicine 2024, 129, 155600. [Google Scholar] [CrossRef]
- Khan, H.; Alam, W.; Alsharif, K.F.; Aschner, M.; Pervez, S.; Saso, L. Alkaloids and Colon Cancer: Molecular Mechanisms and Therapeutic Implications for Cell Cycle Arrest. Molecules 2022, 27, 920. [Google Scholar] [CrossRef] [PubMed]
- Di Dalmazi, G.; Giuliani, C. Plant constituents and thyroid: A revision of the main phytochemicals that interfere with thyroid function. Food Chem. Toxicol. 2021, 152, 112158. [Google Scholar] [CrossRef] [PubMed]
- Seib, C.D.; Sosa, J.A. Evolving Understanding of the Epidemiology of Thyroid Cancer. Endocrinol. Metab. Clin. N. Am. 2019, 48, 23–35. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Ngai, C.H.; Deng, Y.; Pun, C.N.; Lok, V.; Zhang, L.; Xu, Q.; Lucero-Prisno, D.E.; Xu, W.; Zheng, Z.-J.; et al. Incidence and mortality of thyroid cancer in 50 countries: A joinpoint regression analysis of global trends. Endocrine 2023, 80, 355–365. [Google Scholar] [CrossRef] [PubMed]
- Filetti, S.; Durante, C.; Hartl, D.; Leboulleux, S.; Locati, L.D.; Newbold, K.; Papotti, M.G.; Berruti, A. Thyroid cancer: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann. Oncol. 2019, 30, 1856–1883. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Harris, E.J.; Lorch, J.H. Treatment of Aggressive Thyroid Cancer. Surg. Pathol. Clin. 2019, 12, 943–950. [Google Scholar] [CrossRef]
- Haugen, B.R.; Alexander, E.K.; Bible, K.C.; Doherty, G.M.; Mandel, S.J.; Nikiforov, Y.E.; Pacini, F.; Randolph, G.W.; Sawka, A.M.; Schlumberger, M.; et al. 2015 American Thyroid Association Management Guidelines for Adult Patients with Thyroid Nodules and Differentiated Thyroid Cancer: The American Thyroid Association Guidelines Task Force on Thyroid Nodules and Differentiated Thyroid Cancer. Thyroid 2016, 26, 1–133. [Google Scholar] [CrossRef] [PubMed]
- Bulotta, S.; Celano, M.; Costante, G.; Russo, D. Emerging strategies for managing differentiated thyroid cancers refractory to radioiodine. Endocrine 2016, 52, 214–221. [Google Scholar] [CrossRef] [PubMed]
- Hollowell, J.G.; Staehling, N.W.; Flanders, W.D.; Hannon, W.H.; Gunter, E.W.; Spencer, C.A.; Braverman, L.E. Serum TSH, (T4), and Thyroid Antibodies in the United States Population (1988 to 1994): National Health and Nutrition Examination Survey (NHANES III). J. Clin. Endocrinol. Metab. 2002, 87, 489–499. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.-R.; Chen, S.-H.; Lin, C.-Y.; Chao, W.-Y.; Lim, Y.-P.; Yu, H.-I.; Lu, C.-H. In Vitro Antitumor Activity of Aloperine on Human Thyroid Cancer Cells through Caspase-Dependent Apoptosis. Int. J. Mol. Sci. 2018, 19, 312. [Google Scholar] [CrossRef] [PubMed]
- Yu, H.-I.; Shen, H.-C.; Chen, S.-H.; Lim, Y.-P.; Chuang, H.-H.; Tai, T.-S.; Kung, F.-P.; Lu, C.-H.; Hou, C.-Y.; Lee, Y.-R. Autophagy Modulation in Human Thyroid Cancer Cells following Aloperine Treatment. Int. J. Mol. Sci. 2019, 20, 5315. [Google Scholar] [CrossRef] [PubMed]
- Park, K.S.; Bin Kim, J.; Bae, J.; Park, S.-Y.; Jee, H.-G.; Lee, K.E.; Youn, Y.-K. Berberine inhibited the growth of thyroid cancer cell lines 8505C and TPC1. Yonsei Med J. 2012, 53, 346–351. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Wang, X.; Sharvan, R.; Gao, J.; Qu, S. Berberine could inhibit thyroid carcinoma cells by inducing mitochondrial apoptosis, G0/G1 cell cycle arrest and suppressing migration via PI3K-AKT and MAPK signaling pathways. Biomed. Pharmacother. 2017, 95, 1225–1231. [Google Scholar] [CrossRef]
- Shi, X.-Z.; Zhao, S.; Wang, Y.; Wang, M.-Y.; Su, S.-W.; Wu, Y.-Z.; Xiong, C. Antitumor Activity of Berberine by Activating Autophagy and Apoptosis in CAL-62 and BHT-101 Anaplastic Thyroid Carcinoma Cell Lines. Drug Des. Dev. Therapy 2023, 17, 1889–1906. [Google Scholar] [CrossRef] [PubMed]
- Kumarasamy, V.M.; Shin, Y.-J.; White, J.; Sun, D. Selective repression of RET proto-oncogene in medullary thyroid carcinoma by a natural alkaloid berberine. BMC Cancer 2015, 15, 599. [Google Scholar] [CrossRef] [PubMed]
- Gigliotti, C.L.; Ferrara, B.; Occhipinti, S.; Boggio, E.; Barrera, G.; Pizzimenti, S.; Giovarelli, M.; Fantozzi, R.; Chiocchetti, A.; Argenziano, M.; et al. Enhanced cytotoxic effect of camptothecin nanosponges in anaplastic thyroid cancer cells in vitro and in vivo on orthotopic xenograft tumors. Drug Deliv. 2017, 24, 670–680. [Google Scholar] [CrossRef] [PubMed]
- Xu, S.; Zhang, L.; Cheng, X.; Yu, H.; Bao, J.; Lu, R. Capsaicin inhibits the metastasis of human papillary thyroid carcinoma BCPAP cells through the modulation of the TRPV1 channel. Food Funct. 2018, 9, 344–354. [Google Scholar] [CrossRef] [PubMed]
- Xu, S.; Cheng, X.; Wu, L.; Zheng, J.; Wang, X.; Wu, J.; Yu, H.; Bao, J.; Zhang, L. Capsaicin induces mitochondrial dysfunction and apoptosis in anaplastic thyroid carcinoma cells via TRPV1-mediated mitochondrial calcium overload. Cell. Signal. 2020, 75, 109733. [Google Scholar] [CrossRef]
- Xu, S.; Cheng, X.; Wu, J.; Wang, Y.; Wang, X.; Wu, L.; Yu, H.; Bao, J.; Zhang, L. Capsaicin restores sodium iodine symporter-mediated radioiodine uptake through bypassing canonical TSH–TSHR pathway in anaplastic thyroid carcinoma cells. J. Mol. Cell Biol. 2022, 13, 791–807. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.; Xu, S.; Cheng, X.; Zhang, L.; Wang, Y.; Wu, J.; Bao, J.; Yu, H.; Lu, R. Capsaicin inhibits the stemness of anaplastic thyroid carcinoma cells by triggering autophagy-lysosome mediated OCT4A degradation. Phytother. Res. 2022, 36, 938–950. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Cui, Z.; Zhu, J.; Zhou, P.; Cao, X.; Li, X.; Ma, Y.; He, Q. Colchicine inhibits the proliferation and promotes the apoptosis of papillary thyroid carcinoma cells likely due to the inhibitory effect on HDAC1. Biochem. Biophys. Res. Commun. 2023, 679, 129–138. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Ding, H.; Rao, G.; Arora, S.; Saclarides, C.P.; Esparaz, J.; Gattuso, P.; Solorzano, C.C.; Prinz, R.A. Activation of the Sonic Hedgehog pathway in thyroid neoplasms and its potential role in tumor cell proliferation. Endocr.-Relat. Cancer 2012, 19, 167–179. [Google Scholar] [CrossRef] [PubMed]
- Williamson, A.J.; Doscas, M.E.; Ye, J.; Heiden, K.B.; Xing, M.; Li, Y.; Prinz, R.A.; Xu, X. The sonic hedgehog signaling pathway stimulates anaplastic thyroid cancer cell motility and invasiveness by activating Akt and c-Met. Oncotarget 2016, 7, 10472–10485. [Google Scholar] [CrossRef] [PubMed]
- Hinterseher, U.; Wunderlich, A.; Roth, S.; Ramaswamy, A.; Bartsch, D.K.; Hauptmann, S.; Greene, B.H.; Fendrich, V.; Hoffmann, S. Expression of hedgehog signalling pathway in anaplastic thyroid cancer. Endocrine 2014, 45, 439–447. [Google Scholar] [CrossRef]
- Kumarasamy, V.M.; Sun, D. Demonstration of a potent RET transcriptional inhibitor for the treatment of medullary thyroid carcinoma based on an ellipticine derivative. Int. J. Oncol. 2017, 51, 145–157. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.C.; Yu, C.H.; Wang, S.W.; Pu, H.F.; Kan, S.F.; Lin, L.C.; Chi, C.W.; Ho, L.L.; Lee, C.H.; Wang, P.S. anti-proliferative effects of evodiamine on human thyroid cancer cell line ARO. J. Cell. Biochem. 2010, 110, 1495–1503. [Google Scholar] [CrossRef] [PubMed]
- Follo, C.; Vidoni, C.; Morani, F.; Ferraresi, A.; Seca, C.; Isidoro, C. Amino acid response by Halofuginone in Cancer cells triggers autophagy through proteasome degradation of mTOR. Cell Commun. Signal. 2019, 17, 39. [Google Scholar] [CrossRef] [PubMed]
- Ruan, S.; Jia, F.; Li, J. Potential Antitumor Effect of Harmine in the Treatment of Thyroid Cancer. Evid.-Based Complement. Altern. Med. 2017, 2017, 9402615. [Google Scholar] [CrossRef] [PubMed]
- Broecker-Preuss, M.; Becher-Boveleth, N.; Gall, S.; Rehmann, K.; Schenke, S.; Mann, K. Induction of atypical cell death in thyroid carcinoma cells by the indirubin derivative 7-bromoindirubin-3’-oxime (7BIO). Cancer Cell Int. 2015, 15, 97. [Google Scholar] [CrossRef] [PubMed]
- Broecker-Preuss, M.; Becher-Boveleth, N.; Mann, K. Cell Death Induction by the Indirubin Derivative 7BIO and the BH3 Mimetic Drugs ABT-737 and GX15-070 in Medullary Thyroid Carcinoma Cells. Exp. Clin. Endocrinol. Diabetes 2016, 124, 324–330. [Google Scholar] [CrossRef] [PubMed]
- Yu, T.; Tong, L.; Ao, Y.; Zhang, G.; Liu, Y.; Zhang, H. NIR triggered PLGA coated Au-TiO(2) core loaded CPT-11 nanoparticles for human papillary thyroid carcinoma therapy. Drug Deliv. 2020, 27, 855–863. [Google Scholar] [CrossRef] [PubMed]
- Strock, C.J.; Park, J.-I.; Rosen, D.M.; Ruggeri, B.; Denmeade, S.R.; Ball, D.W.; Nelkin, B.D. Activity of irinotecan and the tyrosine kinase inhibitor cep-751 in medullary thyroid cancer. J. Clin. Endocrinol. Metab. 2006, 91, 79–84. [Google Scholar] [CrossRef] [PubMed]
- Di Desidero, T.; Antonelli, A.; Orlandi, P.; Ferrari, S.M.; Fioravanti, A.; Alì, G.; Fontanini, G.; Basolo, F.; Francia, G.; Bocci, G. Synergistic efficacy of irinotecan and sunitinib combination in preclinical models of anaplastic thyroid cancer. Cancer Lett. 2017, 411, 35–43. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Zhang, X.; Cui, S. Matrine inhibits TPC-1 human thyroid cancer cells via the miR-21/PTEN/Akt pathway. Oncol. Lett. 2018, 16, 2965–2970. [Google Scholar] [CrossRef] [PubMed]
- Fu, S.; Zhao, N.; Jing, G.; Yang, X.; Liu, J.; Zhen, D.; Tang, X. Matrine induces papillary thyroid cancer cell apoptosis in vitro and suppresses tumor growth in vivo by downregulating miR-182-5p. Biomed. Pharmacother. 2020, 128, 110327. [Google Scholar] [CrossRef] [PubMed]
- Fu, S.; Ma, C.; Tang, X.; Ma, X.; Jing, G.; Zhao, N.; Ran, J. MiR-192-5p inhibits proliferation, migration, and invasion in papillary thyroid carcinoma cells by regulation of SH3RF3. Biosci. Rep. 2021, 41, BSR20210342. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Zhang, S.; Wang, M.; Dong, S.; Wang, Y.; Liu, S.; Lu, T.; Fu, Y.; Wang, X.; Chen, G. Downregulated miR-21 mediates matrine-induced apoptosis via the PTEN/Akt signaling pathway in FTC-133 human follicular thyroid cancer cells. Oncol. Lett. 2019, 18, 3553–3560. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Wang, X.; Wang, L.; Mei, Y.; Yun, Y.; Yao, X.; Chen, Q.; Zhou, J.; Kou, B. Oridonin represses epithelial-mesenchymal transition and angiogenesis of thyroid cancer via downregulating JAK2/STAT3 signaling. Int. J. Med Sci. 2022, 19, 965–974. [Google Scholar] [CrossRef] [PubMed]
- Esposito, T.; Lucariello, A.; Hay, E.; Contieri, M.; Tammaro, P.; Varriale, B.; Guerra, G.; De Luca, A.; Perna, A. Effects of curcumin and its adjuvant on TPC1 thyroid cell line. Chem.-Biol. Interact. 2019, 305, 112–118. [Google Scholar] [CrossRef] [PubMed]
- Kung, F.-P.; Lim, Y.-P.; Chao, W.-Y.; Zhang, Y.-S.; Yu, H.-I.; Tai, T.-S.; Lu, C.-H.; Chen, S.-H.; Li, Y.-Z.; Zhao, P.-W.; et al. Piperlongumine, a Potent Anticancer Phytotherapeutic, Induces Cell Cycle Arrest and Apoptosis In Vitro and In Vivo through the ROS/Akt Pathway in Human Thyroid Cancer Cells. Cancers 2021, 13, 4266. [Google Scholar] [CrossRef] [PubMed]
- Khan, A.Q.; Mohamed, E.A.N.; Hakeem, I.; Nazeer, A.; Kuttikrishnan, S.; Prabhu, K.S.; Siveen, K.S.; Nawaz, Z.; Ahmad, A.; Zayed, H.; et al. Sanguinarine Induces Apoptosis in Papillary Thyroid Cancer Cells via Generation of Reactive Oxygen Species. Molecules 2020, 25, 1229. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Zhao, A.; Jia, X.; Li, X.; Liang, Y.; Liu, Y.; Xie, X.; Qu, X.; Wang, Q.; Zhang, Y.; et al. Sinomenine Hydrochloride Promotes TSHR-Dependent Redifferentiation in Papillary Thyroid Cancer. Int. J. Mol. Sci. 2022, 23, 10709. [Google Scholar] [CrossRef] [PubMed]
- Zhao, A.; Zhang, J.; Liu, Y.; Jia, X.; Lu, X.; Wang, Q.; Ji, T.; Yang, L.; Xue, J.; Gao, R.; et al. Synergic radiosensitization of sinomenine hydrochloride and radioiodine on human papillary thyroid carcinoma cells. Transl. Oncol. 2021, 14, 101172. [Google Scholar] [CrossRef] [PubMed]
- Fujieda, S.; Noda, I.; Saito, H.; Hoshino, T.; Yagita, M. Swainsonine augments the cytotoxicity of human lymphokine-activated killer cells against autologous thyroid cancer cells. Arch. Otolaryngol. Neck Surg. 1994, 120, 389–394. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Wang, G.; Liu, J.; Ouyang, L. Quinolizidine alkaloids derivatives from Sophora alopecuroides Linn: Bioactivities, structure-activity relationships and preliminary molecular mechanisms. Eur. J. Med. Chem. 2020, 188, 111972. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Zheng, Y.; Deng, H.; Liang, L.; Peng, J. Aloperine induces G2/M phase cell cycle arrest and apoptosis in HCT116 human colon cancer cells. Int. J. Mol. Med. 2014, 33, 1613–1620. [Google Scholar] [CrossRef] [PubMed]
- Samadi, P.; Sarvarian, P.; Gholipour, E.; Asenjan, K.S.; Aghebati-Maleki, L.; Motavalli, R.; Hojjat-Farsangi, M.; Yousefi, M. Berberine: A novel therapeutic strategy for cancer. IUBMB Life 2020, 72, 2065–2079. [Google Scholar] [CrossRef] [PubMed]
- Rauf, A.; Abu-Izneid, T.; Khalil, A.A.; Imran, M.; Shah, Z.A.; Emran, T.B.; Mitra, S.; Khan, Z.; Alhumaydhi, F.A.; Aljohani, A.S.M.; et al. Berberine as a Potential Anticancer Agent: A Comprehensive Review. Molecules 2021, 26, 7368. [Google Scholar] [CrossRef] [PubMed]
- Mondal, A.; Banerjee, S.; Terang, W.; Bishayee, A.; Zhang, J.; Ren, L.; da Silva, M.N.; Bishayee, A. Capsaicin: A chili pepper bioactive phytocompound with a potential role in suppressing cancer development and progression. Phytother. Res. 2024, 38, 1191–1223. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Chen, C.; Chiang, C.; Xiao, T.; Chen, Y.; Zhao, Y.; Zheng, D. The Impact of TRPV1 on Cancer Pathogenesis and Therapy: A Systematic Review. Int. J. Biol. Sci. 2021, 17, 2034–2049. [Google Scholar] [CrossRef] [PubMed]
- Gulfidan, G.; Soylu, M.; Demirel, D.; Erdonmez, H.B.C.; Beklen, H.; Ozbek Sarica, P.; Arga, K.Y.; Turanli, B. Systems biomarkers for papillary thyroid cancer prognosis and treatment through multi-omics networks. Arch. Biochem. Biophys. 2021, 715, 109085. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Wang, C. Traditional uses, phytochemistry, pharmacology, pharmacokinetics and toxicology of the fruit of Tetradium ruticarpum: A review. J. Ethnopharmacol. 2020, 263, 113231. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Liu, Y.; Chen, J.-X.; Lu, X.-Y.; Zhu, W.-J.; Qin, L.; Xun, Z.-X.; Zheng, Q.-Y.; Li, E.-M.; Sun, N.; et al. Harmine is an effective therapeutic small molecule for the treatment of cardiac hypertrophy. Acta Pharmacol. Sin. 2022, 43, 50–63. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Zhong, Z.; Ko, C.N.; Tian, T.; Yang, C. From mundane to classic: Sinomenine as a multi-therapeutic agent. Br. J. Pharmacol. 2023. [Google Scholar] [CrossRef] [PubMed]
- Caturegli, P.; De Remigis, A.; Ferlito, M.; Landek-Salgado, M.A.; Iwama, S.; Tzou, S.-C.; Ladenson, P.W. Anatabine Ameliorates Experimental Autoimmune Thyroiditis. Endocrinology 2012, 153, 4580–4587. [Google Scholar] [CrossRef] [PubMed]
- Li, C.W.; Menconi, F.; Osman, R.; Mezei, M.; Jacobson, E.M.; Concepcion, E.; David, C.S.; Kastrinsky, D.B.; Ohlmeyer, M.; Tomer, Y. Identifying a Small Molecule Blocking Antigen Presentation in Autoimmune Thyroiditis. J. Biol. Chem. 2016, 291, 4079–4090. [Google Scholar] [CrossRef] [PubMed]
- Li, C.W.; Osman, R.; Menconi, F.; Concepcion, E.; Tomer, Y. Cepharanthine blocks TSH receptor peptide presentation by HLA-DR3: Therapeutic implications to Graves’ disease. J. Autoimmun. 2020, 108, 102402. [Google Scholar] [CrossRef] [PubMed]
- Hou, X.; Zhou, J.; Yang, R.; Liu, S.; Bi, M.; Liu, T.; Fan, C.; Guan, H.; Teng, W.; Shan, Z.; et al. Effect of Halofuginone on the Pathogenesis of Autoimmune Thyroid Disease in Different Mice Models. Endocr. Metab. Immune Disord. Drug Targets 2017, 17, 141–148. [Google Scholar] [CrossRef]
- Di Dalmazi, G.; Chalan, P.; Caturegli, P. MYMD-1, a Novel Immunometabolic Regulator, Ameliorates Autoimmune Thyroiditis via Suppression of Th1 Responses and TNF-α Release. J. Immunol. 2019, 202, 1350–1362. [Google Scholar] [CrossRef] [PubMed]
- Schmeltz, L.R.; Blevins, T.C.; Aronoff, S.L.; Ozer, K.; Leffert, J.D.; Goldberg, M.A.; Horowitz, B.S.; Bertenshaw, R.H.; Troya, P.; Cohen, A.E.; et al. Anatabine supplementation decreases thyroglobulin antibodies in patients with chronic lymphocytic autoimmune (Hashimoto’s) thyroiditis: A randomized controlled clinical trial. J. Clin. Endocrinol. Metab. 2014, 99, E137–E142. [Google Scholar] [CrossRef]
- Kutlu Kaya, C.; Gümrükçü, S.; Saraç, A.S.; Kök, F.N. A multifunctional long-term release system for treatment of hypothyroidism. J. Biomed. Mater. Res. Part A 2020, 108, 760–759. [Google Scholar] [CrossRef] [PubMed]
- Pines, M.; Spector, I. Halofuginone—The multifaceted molecule. Molecules 2015, 20, 573–594. [Google Scholar] [CrossRef] [PubMed]
- Akinboye, A.J.; Kim, K.; Choi, S.; Yang, I.; Lee, J.-G. Alkaloids in food: A review of toxicity, analytical methods, occurrence and risk assessments. Food Sci. Biotechnol. 2023, 32, 1133–1158. [Google Scholar] [CrossRef]
- Zheng, X.; Wu, F.; Lin, X.; Shen, L.; Feng, Y. Developments in drug delivery of bioactive alkaloids derived from traditional Chinese medicine. Drug Deliv. 2018, 25, 398–416. [Google Scholar] [CrossRef] [PubMed]
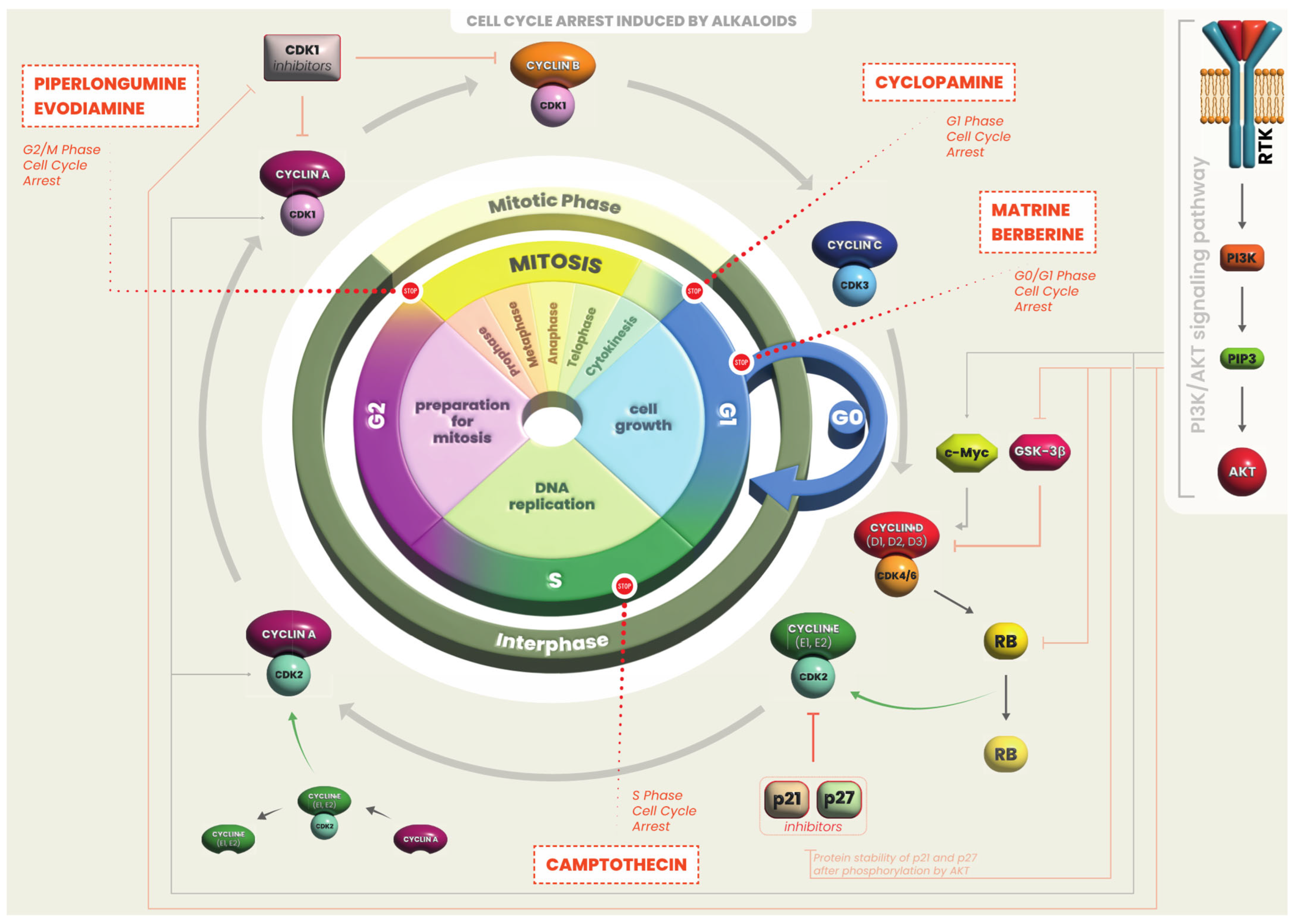
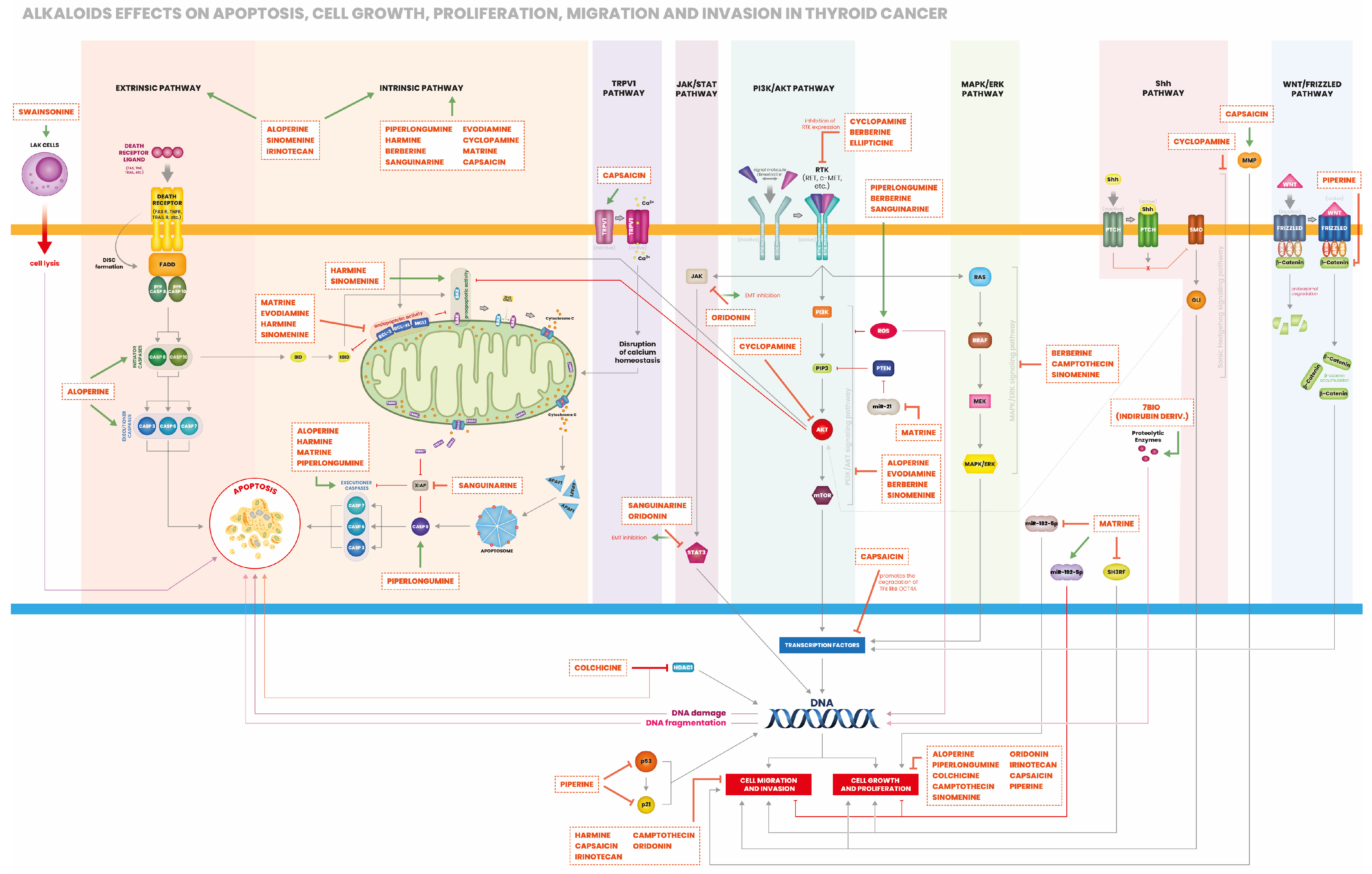

| Alkaloids | Class of Alkaloids | Type of TC | Experimental Model | Anti-Cancer Effects | Ref. |
|---|---|---|---|---|---|
| Aloperine | Quinolizidine | FTC | WRO cells | - Inhibited the tumor cell growth - Induced caspase-dependent apoptosis | [21] |
| PDTC | SW579 cells | - Inhibited the tumor cell growth - Induced caspase-dependent apoptosis | [21] | ||
| ATC | 8505C cells | - Inhibited the tumor cell growth - Induced caspase-dependent apoptosis | [21] | ||
| MDR PTC | IHH-4 cells | - Promoted autophagy through the Akt/mTOR pathway suppression | [22] | ||
| MDR ATC | KMH-2 cells | - Promoted autophagy through the Akt/mTOR pathway suppression | [22] | ||
| Berberine | Isoquinoline | PTC | TPC-1 cells | - Inhibited the tumor cell growth - Induced G0/G1cell cycle arrest - Upregulated p27 expression | [23] |
| ATC | 8505C, C643, OCUT1 cells | - Inhibited the tumor cell growth - Induced apoptosis - Induced apoptosis, G0/G1 cell cycle arrest and inhibited cell migration by modulating PI3K-AKT and MAPK pathways | [23] [24] | ||
| ATC | CAL-62, BHT-101 cells | - Inhibited the tumor cell growth - Induced apoptosis and autophagy through the PI3k/Akt/mTOR pathway suppression and ROS generation | [25] | ||
| MTC | TT cells | - Suppressed RET expression | [26] | ||
| Camptothecin | Quinoline | ATC | BHT-101, CAL-62 cells | - Inhibited cell proliferation, adhesion, and migration | [27] |
| Capsaicin | Capsacinoids | PTC | B-CPAP cells | - Inhibited cell migration and invasion via the activation of TRPV1 and downregulation of MMP expression | [28] |
| ATC | 8505C cells | - Induced mitochondrial dysfunction and apoptosis via the activation of TRPV1 | [29] | ||
| ATC | 8505C, FRO cells | - Restored thyroid-specific gene expression and enhanced radioiodine uptake | [30] | ||
| ATC | 8505C, FRO cells | - Induced autophagy via the activation of TRPV1 - Promoted the degradation of OCT4A | [31] | ||
| Colchicine | Tropolone | PTC | TPC-1, KTC-1 cells | - Inhibited the tumor cell growth - Induced apoptosis via HDA1C inhibition | [32] |
| Cyclopamine | Steroidal alkaloid | FTC | WRO82 cells | - Induced apoptosis and cell cycle arrest | [33] |
| ATC | KAT-18, SW1736 cells | - Induced apoptosis and cell cycle arrest - Decreased cell migration and invasion via AKT and c-Met activation | [33] [34] | ||
| ATC | C643, Hth74 cells | - Inhibited the tumor cell growth | [35] | ||
| Ellipticine | Indole | MTC | TT cells | - Reduced RET expression | [36] |
| Evodiamine | Indole | ATC | ARO cells | - Induced cell cycle arrest and caspase-dependent apoptosis | [37] |
| Febrifugine and Halofunginone * | Quinazolinone | FTC | WRO cells | - Induced autophagy | [38] |
| Harmine | Carboline | PTC | TPC-1 cells | - Induced apoptosis by regulating Bcl-2/Bax and elevating the activity of caspase-3 - Inhibited cell migration and invasion | [39] |
| Indirubin and 7BIO * | Indole | PTC | TPC-1, BHT101, B-CPAP cells | - Inhibited the tumor cell growth | [40] |
| FTC | ML1, TT2609, FTC1-33, FTC-236, FTC-238 cells | - Inhibited the tumor cell growth | [40] | ||
| ATC | SW1736, HTh7, C643, HTh74, 8305C, 8505C cells | - Inhibited the tumor cell growth - Induced a caspase-independent cell death and DNA fragmentation | [40] | ||
| MTC | TT cells | - Induced apoptosis | [41] | ||
| Irinotecan * | Quinoline | PTC | B-CPAP cells | - Induced cell death and reduced cell invasion | [42] |
| FTC | FTC-133 cells | - Induced cell death and reduced cell invasion | [42] | ||
| ATC | 8305C, FB3 cells | - Inhibited the tumor cell growth | [43] | ||
| MTC | TT cells | - Induced apoptosis | [44] | ||
| Matrine | Quinolizidine | PTC | TPC-1, B-CPAP, K1 cells | - Induced apoptosis and cell cycle arrest by up-regulating miR-21 and down-regulating phosphorylated Akt - Decreased the level of Bcl-2 and activated caspase-3 - Inhibited the tumor cell growth by downregulating the expression of miR-182-5p | [45,46] |
| PTC | TPC-1 cells | - Inhibited cell proliferation, migration, and invasion via modulating miR-192-5p/SH3RF3 pathway | [47] | ||
| FTC | FTC-133 cells | - Induced apoptosis and cell cycle arrest | [48] | ||
| Oridonin | Terpenoid | PTC | TPC-1, B-CPAP cells | - Inhibited cell proliferation, migration, and invasion - Inhibited angiogenesis and modulated EMT via the inactivation of JAK2/STAT3 signaling pathway | [49] |
| Piperine | Piperidine | PTC | TPC-1 cells | - Inhibited the tumor cell growth - Decreased the expression of p21, p53 and β-catenin | [50] |
| Piperlongumine (or piplartine) | Piperidine | PTC | IHH-4 cells | - Inhibited cell proliferation and colony formation - Promoted cell cycle arrest and cellular apoptosis through the intrinsic caspase-dependent pathway - Induced cellular apoptosis through the ROS-modulated Akt pathway | [51] |
| FTC | WRO cells | - Inhibited cell proliferation and colony formation - Promoted cell cycle arrest and cellular apoptosis through the intrinsic caspase-dependent pathway - Induced cellular apoptosis through the ROS-modulated Akt pathway | [51] | ||
| ATC | KMH-2, 8505c cells | - Inhibited cell proliferation and colony formation - Promoted cell cycle arrest and cellular apoptosis through the intrinsic caspase-dependent pathway - Induced cellular apoptosis through the ROS-modulated Akt pathway | [51] | ||
| Sanguinarine | Isoquinoline | PTC | TPC-1, B-CPAP cells | - Inhibited the tumor cell growth - Induced cell apoptosis via inactivation of STAT3 and reactive oxygen species generation - Sensitized PTC cells to chemotherapeutic drug cisplatin | [52] |
| Sinomenine | Isoquinoline | PTC | TPC-1, B-CPAP cells | - Inhibited cell proliferation - Induced cell apoptosis - Upregulated the membrane localization of NIS and enhanced radioiodine uptake | [53,54] |
| Swainsonine | Indolizidine | PTC | Autologous TC cells | - Modulated the activity of lymphokine-activated killer | [55] |
| Alkaloids | Class of Alkaloids | TC | Experimental Model | Effects | Ref. |
|---|---|---|---|---|---|
| Camptothecin | Quinoline | ATC | NSG mice with intrathyroidal injection of Cal-62 cells | - Decreased tumor growth, metastatization and tumor microvessel density | [27] |
| Ellipticine derivative | Pyridocarbazole | MTC | SCID mice injected s.c. with TT cells into the flank | - Decreased tumor growth - Reduced RET expression in tumor tissue | [36] |
| Harmine | β-carboline | PTC | Nude mice injected s.c. with TPC-1 cells in the left axillary space | - Decreased tumor growth | [39] |
| Irinotecan | Quinoline | MTC | Nude mice injected s.c. with TT cells into the flank | - Decreased tumor growth - Increased progression-free survival | [43] |
| ATC | NU/NU nude mice injected s.c. with 8305C cells | - Decreased tumor growth - Increased caspase-3 activity in tumor tissue | [44] | ||
| Matrine | Quinolizidine | PTC | BALB/c nude mice injected s.c. with TPC-1 cells | - Decreased tumor growth - Downregulated the expression of miR-182-5p | [46] |
| Oridonin | Terpenoid | PTC | BALB/C nude injected s.c. with TPC-1 cells into the flank | - Decreased tumor growth - Reduced JAK-2, N-cadherin and VEGFA expression in tumor tissue | [49] |
| Piperlongumine | Piperidine | PTC | BALB/c nude mice injected s.c. with IHH-4 cells into the flank | - Decreased tumor growth | [51] |
| Alkaloids | Class of Alkaloids | AITD | Experimental Model | Effects | Ref. |
|---|---|---|---|---|---|
| Anatabine | Nicotinic-acid derived alkaloids | AT | CBA/J mice Tg injected | - Decreased Tg-Ab levels - Suppressed the expression of IL-1 receptor type 2 and IL-18 | [66] |
| Cepharanthine | Biscoclaurine | AT | NOD mice Tg injected | - Blocked T-cell activation | [67] |
| GD | BALB/c-D3 mice injected with AdTSHR-289 | - Inhibited T-cell activation and cytokine response | [68] | ||
| Halofuginone * | Quinazolinone | AT | NOD.H-2h4 mice | - Decreased Tg-Ab levels - Decreased Th17 cells | [69] |
| MYMD-1 * | Nicotinic-acid derived alkaloids | AT | NOD.H-2h4 mice | - Decreased Tg-Ab levels - Decreased Th1 cells and TNF-α levels | [70] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Di Dalmazi, G.; Giuliani, C.; Bucci, I.; Mascitti, M.; Napolitano, G. Promising Role of Alkaloids in the Prevention and Treatment of Thyroid Cancer and Autoimmune Thyroid Disease: A Comprehensive Review of the Current Evidence. Int. J. Mol. Sci. 2024, 25, 5395. https://doi.org/10.3390/ijms25105395
Di Dalmazi G, Giuliani C, Bucci I, Mascitti M, Napolitano G. Promising Role of Alkaloids in the Prevention and Treatment of Thyroid Cancer and Autoimmune Thyroid Disease: A Comprehensive Review of the Current Evidence. International Journal of Molecular Sciences. 2024; 25(10):5395. https://doi.org/10.3390/ijms25105395
Chicago/Turabian StyleDi Dalmazi, Giulia, Cesidio Giuliani, Ines Bucci, Marco Mascitti, and Giorgio Napolitano. 2024. "Promising Role of Alkaloids in the Prevention and Treatment of Thyroid Cancer and Autoimmune Thyroid Disease: A Comprehensive Review of the Current Evidence" International Journal of Molecular Sciences 25, no. 10: 5395. https://doi.org/10.3390/ijms25105395






