Deletion of the Mitochondrial Membrane Protein Fam210b Is Associated with the Development of Systemic Lupus Erythematosus
Abstract
:1. Introduction
2. Results
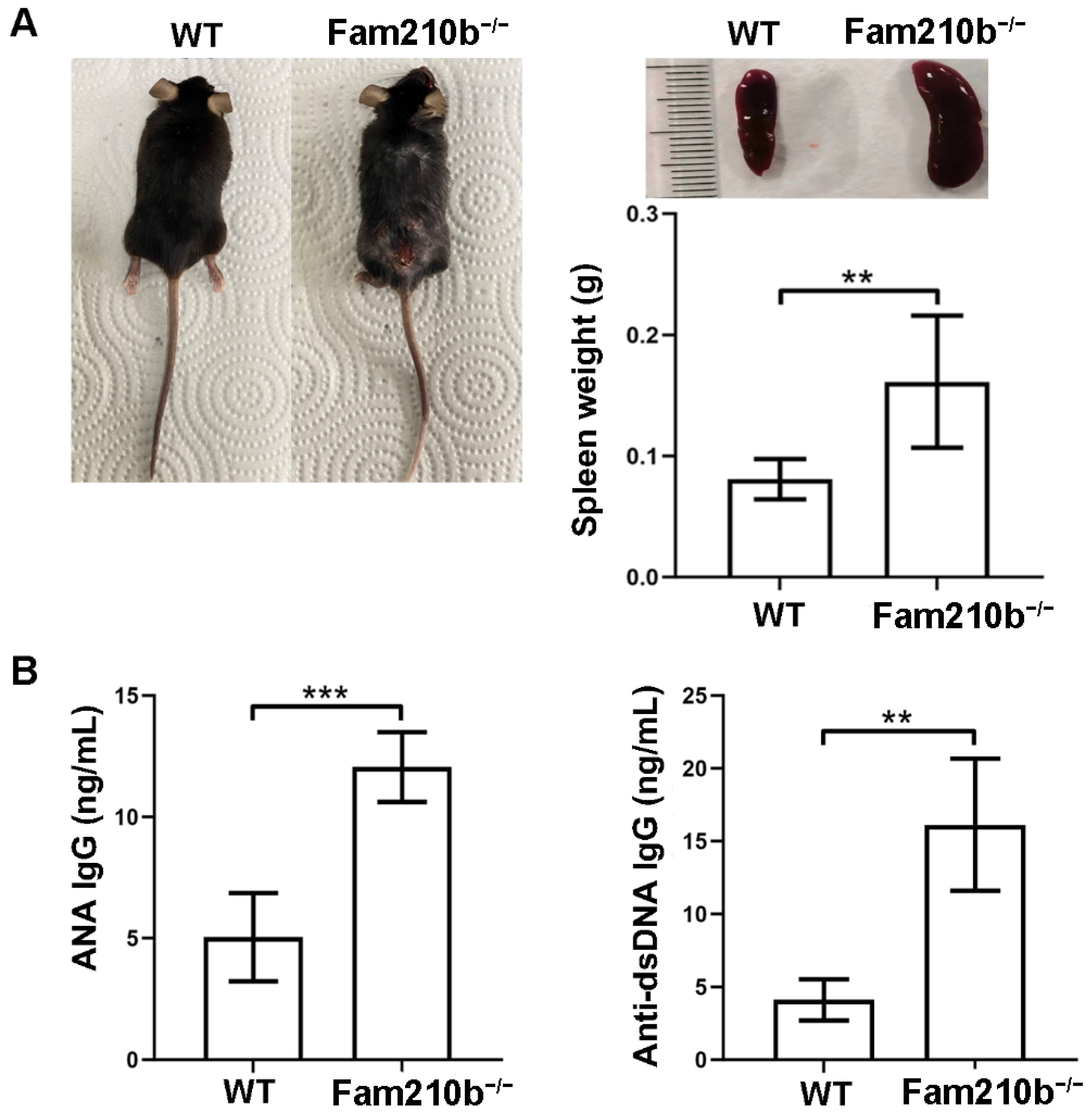
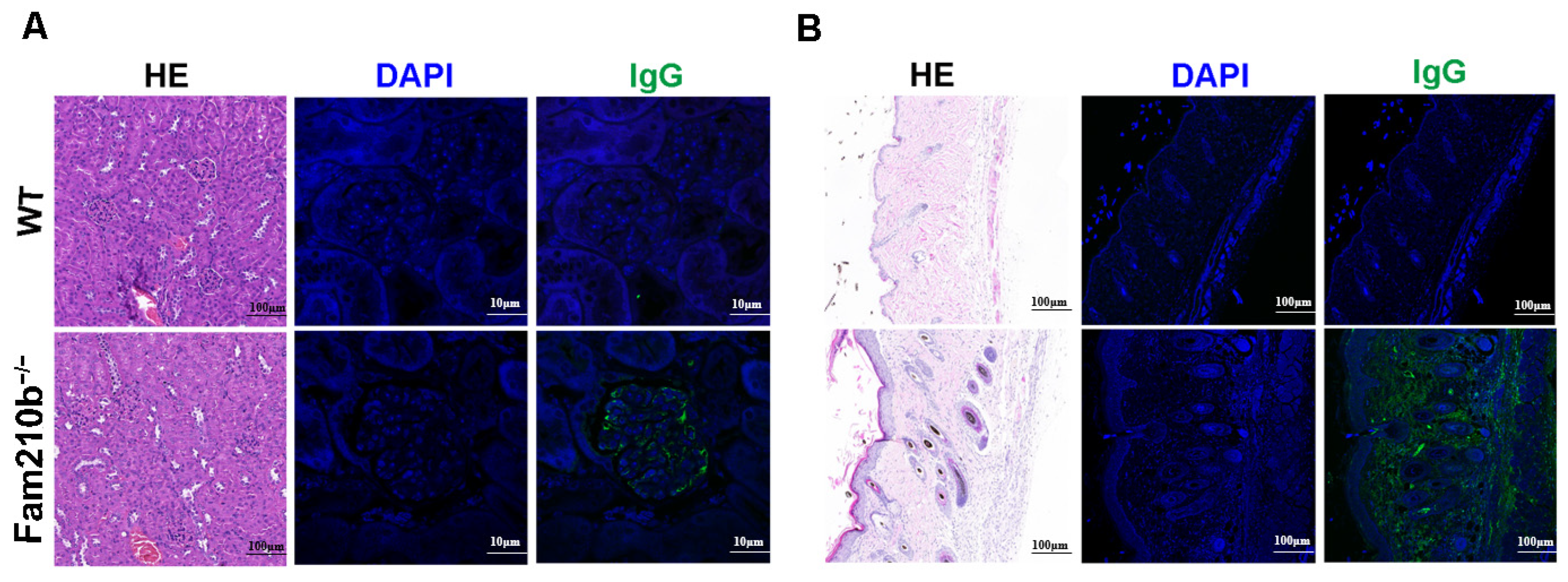
2.1. Fam210b Knockout Induced Lupus-Like Symptoms in Mice
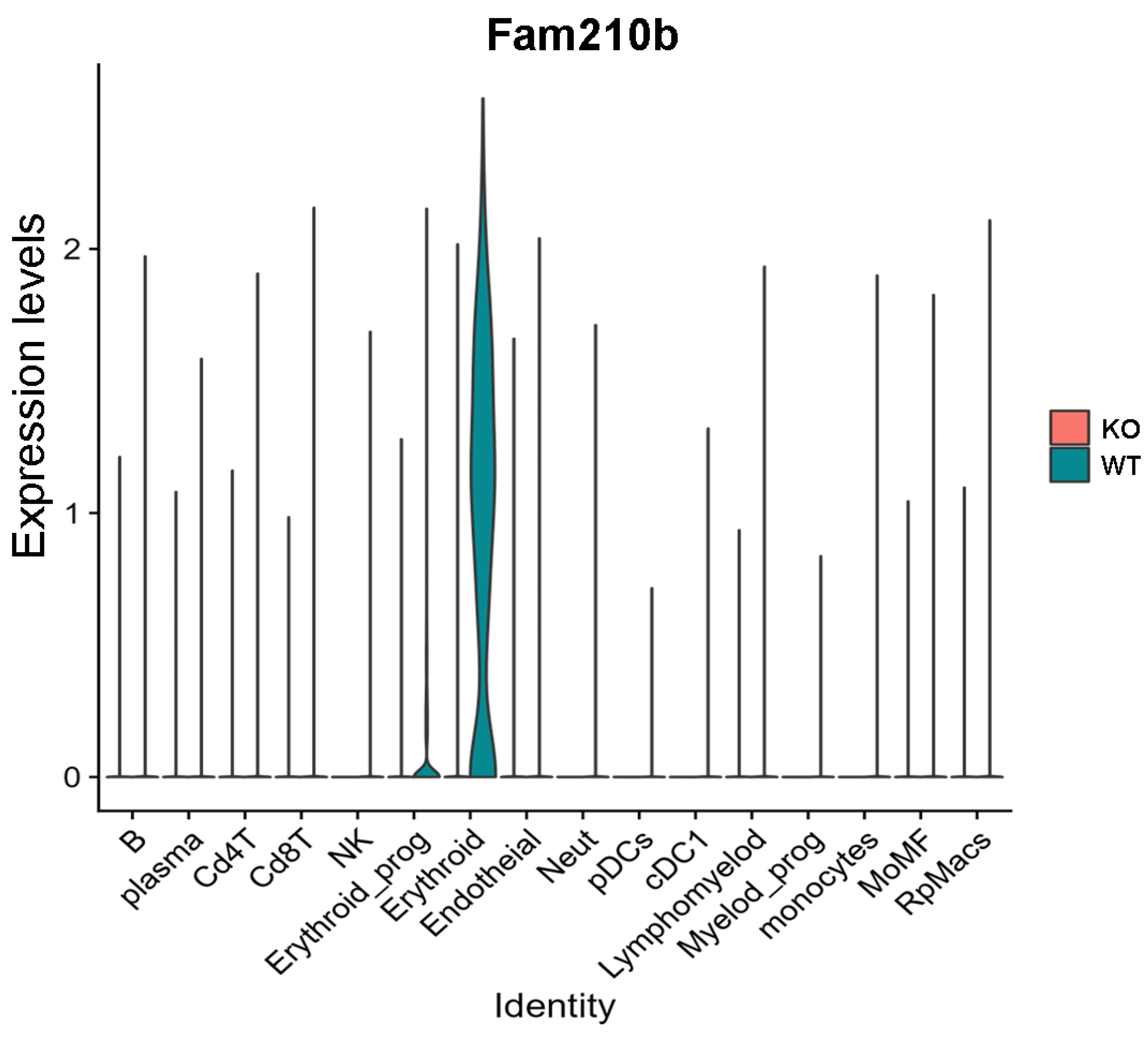
2.2. Fam210b Is Mainly Expressed in Erythroid Cells
2.3. Fam210b Knockout Inhibited Splenic Erythrocyte Differentiation and Development
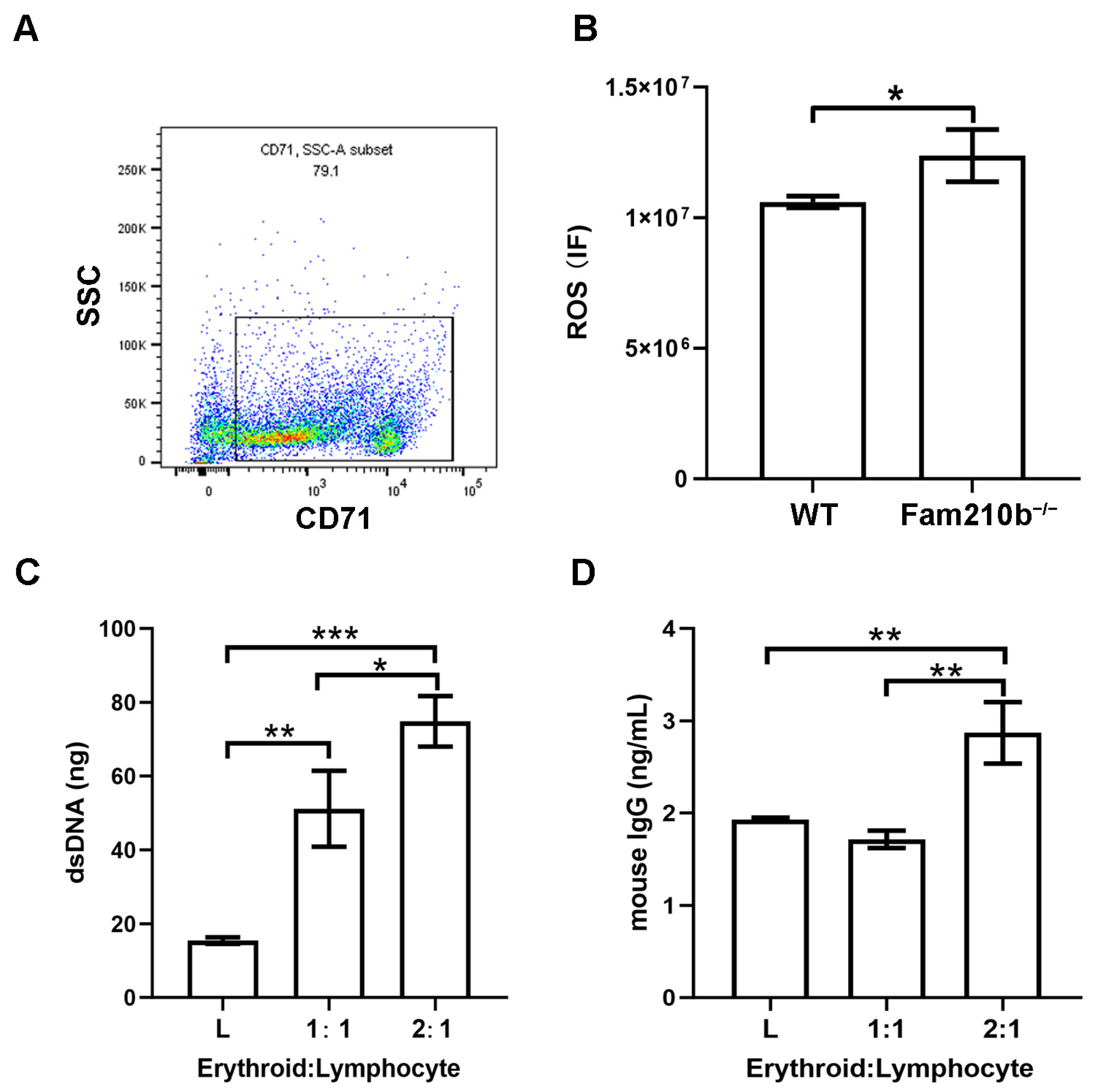
2.4. Fam210b−/− Erythroid Cells Stimulate Lymphocyte Activation
3. Discussion
4. Materials and Methods
4.1. Mice
4.2. Extraction of Peripheral Blood and Spleens from Mice
4.3. Single-Cell Sequencing of Spleens
4.4. Flow Cytometry
4.5. ELISA Assay
4.6. H&E Staining and Immunofluorescence
4.7. CD71+ Erythroid Cell Sorting
4.8. Co-Culture and Stimulation
4.9. dsDNA Content Detection
4.10. ROS Content Detection
4.11. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
List of Abbreviations
| SLE | systemic lupus erythematosus |
| mtDNA | mitochondrial DNA |
| anti-dsDNA | anti-double-stranded DNA |
| LPS | lipopolysaccharide |
| ANA | anti-nuclear antibody |
| TLRs | toll-like receptors |
| IFNs | type I interferons |
| NETs | neutrophil extracellular traps |
| pDCs | plasmacytoid dendritic cells |
| ROS | reactive oxygen species |
| GWAS | genome-wide association studies |
| ATP | adenosine triphosphate |
| IRGM | immune-related guanosine triphosphatase M protein |
| ND | NADH dehydrogenase |
| Fam210b−/− | Fam210b knockout |
| ERK | extracellular signal-regulated kinase |
| ELISA | enzyme-linked immunosorbent assay |
References
- Barber, M.R.W.; Drenkard, C.; Falasinnu, T.; Hoi, A.; Mak, A.; Kow, N.Y.; Svenungsson, E.; Peterson, J.; Clarke, A.E.; Goldman, R.R. Global epidemiology of systemic lupus erythematosus. Nat. Rev. Rheumatol. 2021, 17, 515–532. [Google Scholar] [CrossRef] [PubMed]
- Zucchi, D.; Silvagni, E.; Elefante, E.; Signorini, V.; Cardelli, C.; Trentin, F.; Schilirò, D.; Cascarano, G.; Valevich, A.; Bortoluzzi, A.; et al. Systemic lupus erythematosus: One year in review 2023. Clin. Exp. Rheumatol. 2023, 41, 997–1008. [Google Scholar] [CrossRef] [PubMed]
- Pisetsky, D.S. Pathogenesis of autoimmune disease. Nat. Rev. Nephrol. 2023, 19, 509–524. [Google Scholar] [CrossRef] [PubMed]
- Kwon, Y.C.; Chun, S.; Kwangwoo, K.; Mak, A. Update on the Genetics of Systemic Lupus Erythematosus: Genome-Wide Association Studies and Beyond. Cells 2019, 8, 1180. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Hu, X.; Xiao, F.; Zhang, X.; Zhao, L.; Wang, M. Mitochondrial impairment and repair in the pathogenesis of systemic lupus erythematosus. Front. Immunol. 2022, 13, 929520. [Google Scholar] [CrossRef]
- Miao, N.; Wang, Z.; Wang, Q.; Xie, H.; Yang, Y.; Wang, Y.; Wang, J.; Kang, H.; Bai, W.; Wang, Y.; et al. Oxidized mitochondrial DNA induces gasdermin D oligomerization in systemic lupus erythematosus. Nat. Commun. 2023, 14, 872. [Google Scholar] [CrossRef]
- Pazmandi, K.; Magyarics, Z.; Boldogh, I.; Csillag, A.; Rajnavolgyi, E.; Bacsi, A. Modulatory effects of low-dose hydrogen peroxide on the function of human plasmacytoid dendritic cells. Free Radic. Biol. Med. 2012, 52, 635–645. [Google Scholar] [CrossRef] [PubMed]
- Wójcik, P.; Gęgotek, A.; Žarković, N.; Skrzydlewska, E. Oxidative Stress and Lipid Mediators Modulate Immune Cell Functions in Autoimmune Diseases. Int. J. Mol. Sci. 2021, 22, 723. [Google Scholar] [CrossRef] [PubMed]
- Rai, P.; Janardhan, K.S.; Meacham, J.; Madenspacher, J.H.; Lin, W.C.; Karmaus, P.W.F.; Martinez, J.; Li, K.Z.; Yan, M.; Zeng, J.; et al. IRGM1 links mitochondrial quality control to autoimmunity. Nat. Immunol. 2021, 22, 312–321. [Google Scholar] [CrossRef]
- Kabi, A.; Nickerson, K.P.; Homer, C.R.; McDonald, C. Digesting the genetics of inflammatory bowel disease: Insights from studies of autophagy risk genes. Inflamm. Bowel Dis. 2012, 18, 782–792. [Google Scholar] [CrossRef]
- Becker, Y.L.C.; Duvvuri, B.; Fortin, P.R.; Lood, C.; Boilard, E. The role of mitochondria in rheumatic diseases. Nat. Rev. Rheumatol. 2022, 18, 621–640. [Google Scholar] [CrossRef] [PubMed]
- Kondo, A.; Fujiwara, T.; Okitsu, Y.; Fukuhara, N.; Onishi, Y.; Nakamura, Y.; Sawada, K.; Harigae, H. Identification of a novel putative mitochondrial protein FAM210B associated with erythroid differentiation. Int. J. Hematol. 2016, 103, 387–395. [Google Scholar] [CrossRef] [PubMed]
- Yien, Y.Y.; Shi, J.; Chen, C.; Cheung, J.T.M.; Grillo, A.S.; Shrestha, R.; Li, L.; Zhang, X.; Kafina, M.D.; Kingsley, P.D.; et al. FAM210B is an erythropoietin target and regulates erythroid heme synthesis by controlling mitochondrial iron import and ferrochelatase activity. J. Biol. Chem. 2018, 293, 19797–19811. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, C.; Fujiwara, T.; Shima, H.; Ono, K.; Saito, K.; Kato, H.; Onodera, K.; Ichikawa, S.; Fukuhara, N.; Onishi, Y.; et al. Elucidation of the Role of FAM210B in Mitochondrial Metabolism and Erythropoiesis. Mol. Cell Biol. 2022, 42, e0014322. [Google Scholar] [CrossRef]
- Sun, S.; Liu, J.; Zhao, M.; Han, Y.; Chen, P.; Mo, Q.; Wang, B.; Chen, G.; Fang, Y.; Tian, Y.; et al. Loss of the novel mitochondrial protein FAM210B promotes metastasis via PDK4-dependent metabolic reprogramming. Cell Death Dis. 2017, 8, e2870. [Google Scholar] [CrossRef]
- Li, H.; Zhou, L.; Zhou, W.; Zhang, X.; Shang, J.; Feng, X.; Yu, L.; Fan, J.; Ren, J.; Zhang, R.; et al. Decoding the mitochondrial connection: Development and validation of biomarkers for classifying and treating systemic lupus erythematosus through bioinformatics and machine learning. BMC Rheumatol. 2023, 7, 44. [Google Scholar] [CrossRef]
- Xu, Y.; Gao, R.; Zhang, M.; Zeng, Q.; Zhou, S.; Zhu, G.; Su, W.; Wang, R. Mendelian randomization study on causal association of FAM210B with drug-induced lupus. Clin. Rheumatol. 2024, 43, 1513–1520. [Google Scholar] [CrossRef]
- Aringer, M.; Johnson, S.R. Classifying and diagnosing systemic lupus erythematosus in the 21st century. Rheumatology 2020, 59, 4–11. [Google Scholar] [CrossRef] [PubMed]
- Accapezzato, D.; Caccavale, R.; Paroli, M.P.; Gioia, C.; Nguyen, B.L.; Spadea, L.; Paroli, M. Advances in the Pathogenesis and Treatment of Systemic Lupus Erythematosus. Int. J. Mol. Sci. 2023, 24, 6578. [Google Scholar] [CrossRef]
- Takagishi, T.; Takehara, M.; Seike, S.; Miyamoto, K.; Kobayashi, K.; Nagahama, M. Clostridium perfringens alpha-toxin impairs erythropoiesis by inhibition of erythroid differentiation. Sci. Rep. 2017, 7, 5217. [Google Scholar] [CrossRef]
- Georgakis, S.; Gkirtzimanaki, K.; Papadaki, G.; Gakiopoulou, H.; Drakos, E.; Eloranta, M.L.; Makridakis, M.; Kontostathi, G.; Zoidakis, J.; Baira, E.; et al. NETs decorated with bioactive IL-33 infiltrate inflamed tissues and induce IFN-alpha production in patients with SLE. JCI Insight 2021, 6, e147671. [Google Scholar] [CrossRef]
- Lou, H.; Ling, G.S.; Cao, X. Autoantibodies in systemic lupus erythematosus: From immunopathology to therapeutic target. J. Autoimmun. 2022, 132, 102861. [Google Scholar] [CrossRef]
- Brown, G.J.; Cañete, P.F.; Wang, H.; Medhavy, A.; Bones, J.; Roco, J.A.; He, Y.; Qin, Y.; Cappello, J.; Ellyard, J.; et al. TLR7 gain-of-function genetic variation causes human lupus. Nature 2022, 605, 349–356. [Google Scholar] [CrossRef]
- Liu, D.; Xu, H.; Shih, C.; Wan, Z.; Ma, X.; Ma, W.; Luo, D.; Qi, H. T-B-cell entanglement and ICOSL-driven feed-forward regulation of germinal centre reaction. Nature 2015, 517, 214–218. [Google Scholar] [CrossRef]
- Selvananda, S.; Kan, S.L. Performance of the 2019 European League against Rheumatism/American College of Rheumatology classification criteria for systemic lupus erythematosus in a multiethnic Malaysian cohort. Int. J. Rheum. Dis 2022, 25, 131–139. [Google Scholar] [CrossRef]
- Yee, C.S.; Gordon, C.; Isenberg, D.A.; Griffiths, B.; Teh, L.S.; Bruce, I.N.; Ahmad, Y.; Rahman, A.; Prabu, A.; Akil, M.; et al. Comparison of Responsiveness of British Isles Lupus Assessment Group 2004 Index, Systemic Lupus Erythematosus Disease Activity Index 2000, and British Isles Lupus Assessment Group 2004 Systems Tally. Arthritis Care Res. 2022, 74, 1623–1630. [Google Scholar] [CrossRef]
- Hattangadi, S.M.; Wong, P.; Zhang, L.; Flygare, J.; Lodish, H.F. From stem cell to red cell: Regulation of erythropoiesis at multiple levels by multiple proteins, RNAs, and chromatin modifications. Blood 2011, 118, 6258–6268. [Google Scholar] [CrossRef]
- Li, W.; Guo, R.; Song, Y.; Jiang, Z. Erythroblastic Island Macrophages Shape Normal Erythropoiesis and Drive Associated Disorders in Erythroid Hematopoietic Diseases. Front. Cell Dev. Biol. 2021, 8, 613885. [Google Scholar] [CrossRef]
- Moras, M.; Lefevre, S.D.; Ostuni, M.A. From Erythroblasts to Mature Red Blood Cells: Organelle Clearance in Mammals. Front. Physiol. 2017, 8, 1076. [Google Scholar] [CrossRef]
- Tusi, B.K.; Wolock, S.L.; Weinreb, C.; Hwang, Y.; Hidalgo, D.; Zilionis, R.; Waisman, A.; Huh, J.R.; Klein, A.M.; Socolovsky, M. Population snapshots predict early haematopoietic and erythroid hierarchies. Nature 2018, 555, 54–60. [Google Scholar] [CrossRef]
- Chami, N.; Chen, M.H.; Slater, A.J.; Eicher, J.D.; Evangelou, E.; Tajuddin, S.M.; Love-Gregory, L.; Kacprowski, T.; Schick, U.M.; Nomura, A.; et al. Exome Genotyping Identifies Pleiotropic Variants Associated with Red Blood Cell Traits. Am. J. Hum. Genet. 2016, 99, 8–21. [Google Scholar] [CrossRef]
- Ulirsch, J.C.; Nandakumar, S.K.; Wang, L.; Giani, F.C.; Zhang, X.; Rogov, P.; Melnikov, A.; McDonel, P.; Do, R.; Mikkelsen, T.S.; et al. Systematic Functional Dissection of Common Genetic Variation Affecting Red Blood Cell Traits. Cell 2016, 165, 1530–1545. [Google Scholar] [CrossRef]
- Grzywa, T.M.; Nowis, D.; Golab, J. The role of CD71+ erythroid cells in the regulation of the immune response. Pharmacol. Ther. 2021, 228, 107927. [Google Scholar] [CrossRef] [PubMed]
- Ghaffari, S. Oxidative stress in the regulation of normal and neoplastic hematopoiesis. Antioxid. Redox Signal. 2008, 10, 1923–1940. [Google Scholar] [CrossRef]
- Zhao, B.; Mei, Y.; Yang, J.; Ji, P. Erythropoietin-regulated oxidative stress negatively affects enucleation during terminal erythropoiesis. Exp. Hematol. 2016, 44, 975–981. [Google Scholar] [CrossRef] [PubMed]
- Elahi, S.; Ertelt, J.M.; Kinder, J.M.; Jiang, T.T.; Zhang, X.; Xin, L.; Chaturvedi, V.; Strong, B.S.; Qualls, J.E.; Steinbrecher, K.A.; et al. Immunosuppressive CD71+ erythroid cells compromise neonatal host defence against infection. Nature 2013, 504, 158–162. [Google Scholar] [CrossRef]
- Gomez-Lopez, N.; Romero, R.; Xu, Y.; Miller, D.; Unkel, R.; CMacKenzie, T.; Frascoli, M.; Hassan, S.S. Umbilical cord CD71+ erythroid cells are reduced in neonates born to women in spontaneous preterm labor. Am. J. Reprod. Immunol. 2016, 76, 280–284. [Google Scholar] [CrossRef]
- Chen, X.; Song, M.; Zhang, B.; Zhang, Y. Reactive Oxygen Species Regulate T Cell Immune Response in the Tumor Microenvironment. Oxid. Med. Cell Longev. 2016, 2016, 1580967. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Treviño, P.; Velásquez, M.; García, N. Mechanisms of mitochondrial DNA escape and its relationship with different metabolic diseases. Biochim. Biophys. Acta Mol. Basis Dis. 2020, 1866, 165761. [Google Scholar] [CrossRef]
- Rongvaux, A. Innate immunity and tolerance toward mitochondria. Mitochondrion 2018, 41, 14–20. [Google Scholar] [CrossRef]
- Schwarzer, A.; Oliveira, M.; Kleppa, M.J.; Slattery, S.D.; Anantha, A.; Cooper, A.; Hannink, M.; Schambach, A.; Dörrie, A.; Kotlyarov, A.; et al. Targeting Aggressive B-cell Lymphomas through Pharmacological Activation of the Mitochondrial Protease OMA1. Mol. Cancer Ther. 2023, 22, 1290–1303. [Google Scholar] [CrossRef] [PubMed]
- Reth, M. Hydrogen peroxide as second messenger in lymphocyte activation. Nat. Immunol. 2002, 3, 1129–1134. [Google Scholar] [CrossRef] [PubMed]
- Rhee, S.G. Cell signaling. H2O2, a necessary evil for cell signaling. Science 2006, 312, 1882–1883. [Google Scholar] [CrossRef] [PubMed]
- Dickinson, B.C.; Chang, C.J. Chemistry and biology of reactive oxygen species in signaling or stress responses. Nat. Chem. Biol. 2011, 7, 504–511. [Google Scholar] [CrossRef]
- Pivkin, I.V.; Peng, Z.; Karniadakis, G.E.; Buffet, P.A.; Dao, M.; Suresh, S. Biomechanics of red blood cells in human spleen and consequences for physiology and disease. Proc. Natl. Acad. Sci. USA 2016, 113, 7804–7809. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xu, Y.; Gao, R.; Zhang, M.; Zeng, Q.; Zhu, G.; Qiu, J.; Su, W.; Wang, R. Deletion of the Mitochondrial Membrane Protein Fam210b Is Associated with the Development of Systemic Lupus Erythematosus. Int. J. Mol. Sci. 2024, 25, 7253. https://doi.org/10.3390/ijms25137253
Xu Y, Gao R, Zhang M, Zeng Q, Zhu G, Qiu J, Su W, Wang R. Deletion of the Mitochondrial Membrane Protein Fam210b Is Associated with the Development of Systemic Lupus Erythematosus. International Journal of Molecular Sciences. 2024; 25(13):7253. https://doi.org/10.3390/ijms25137253
Chicago/Turabian StyleXu, Yaqi, Ran Gao, Min Zhang, Qi Zeng, Gaizhi Zhu, Jinming Qiu, Wenting Su, and Renxi Wang. 2024. "Deletion of the Mitochondrial Membrane Protein Fam210b Is Associated with the Development of Systemic Lupus Erythematosus" International Journal of Molecular Sciences 25, no. 13: 7253. https://doi.org/10.3390/ijms25137253




