Lipids and Lipid-Mediated Signaling in Plant–Pathogen Interactions
Abstract
:1. Introduction
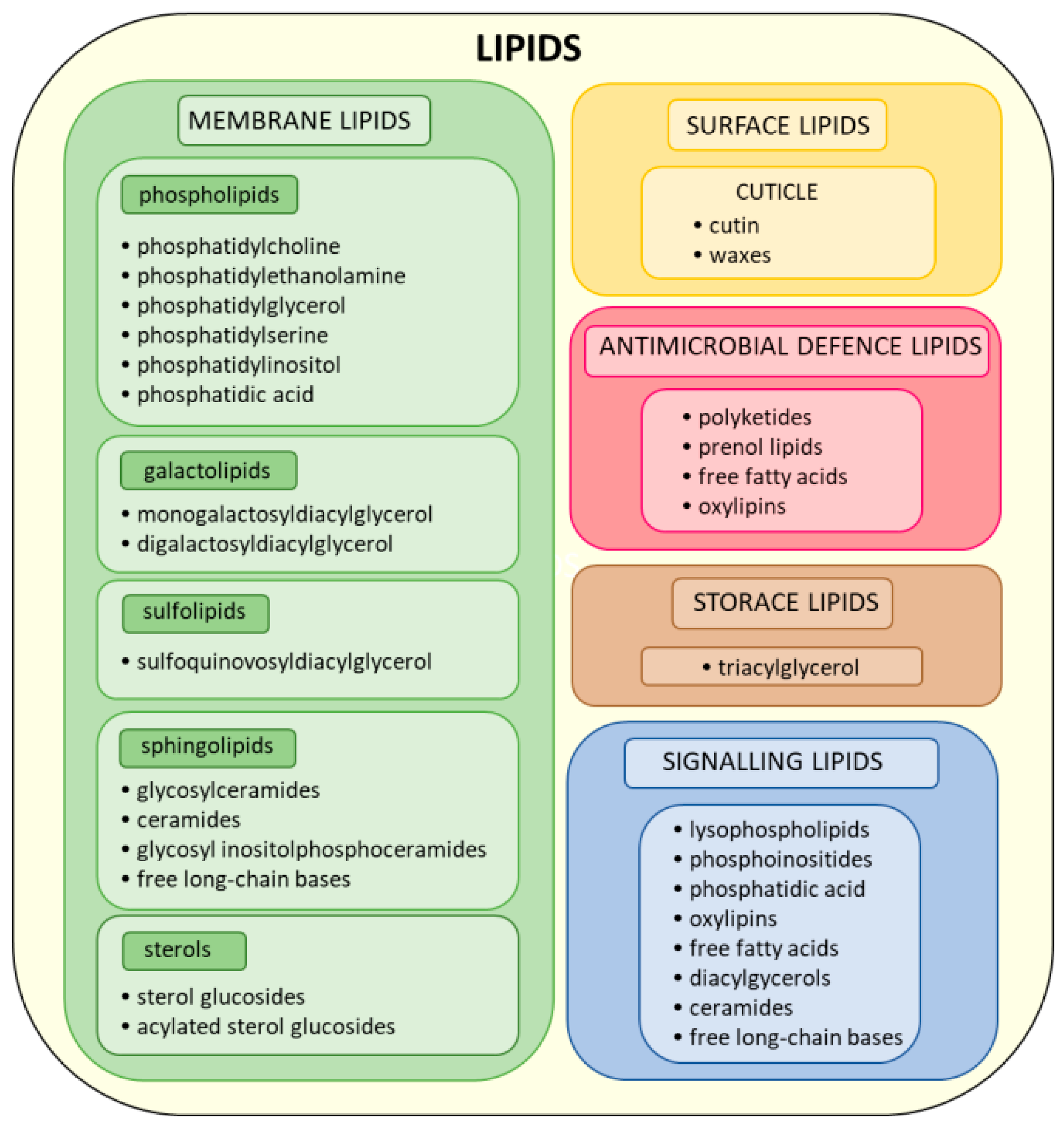
2. Classification, Composition, and Function of Plant Lipids
3. A Brief Overview of the Plant Defense System
4. Plant Surface Lipids in Plant–Pathogen Interactions
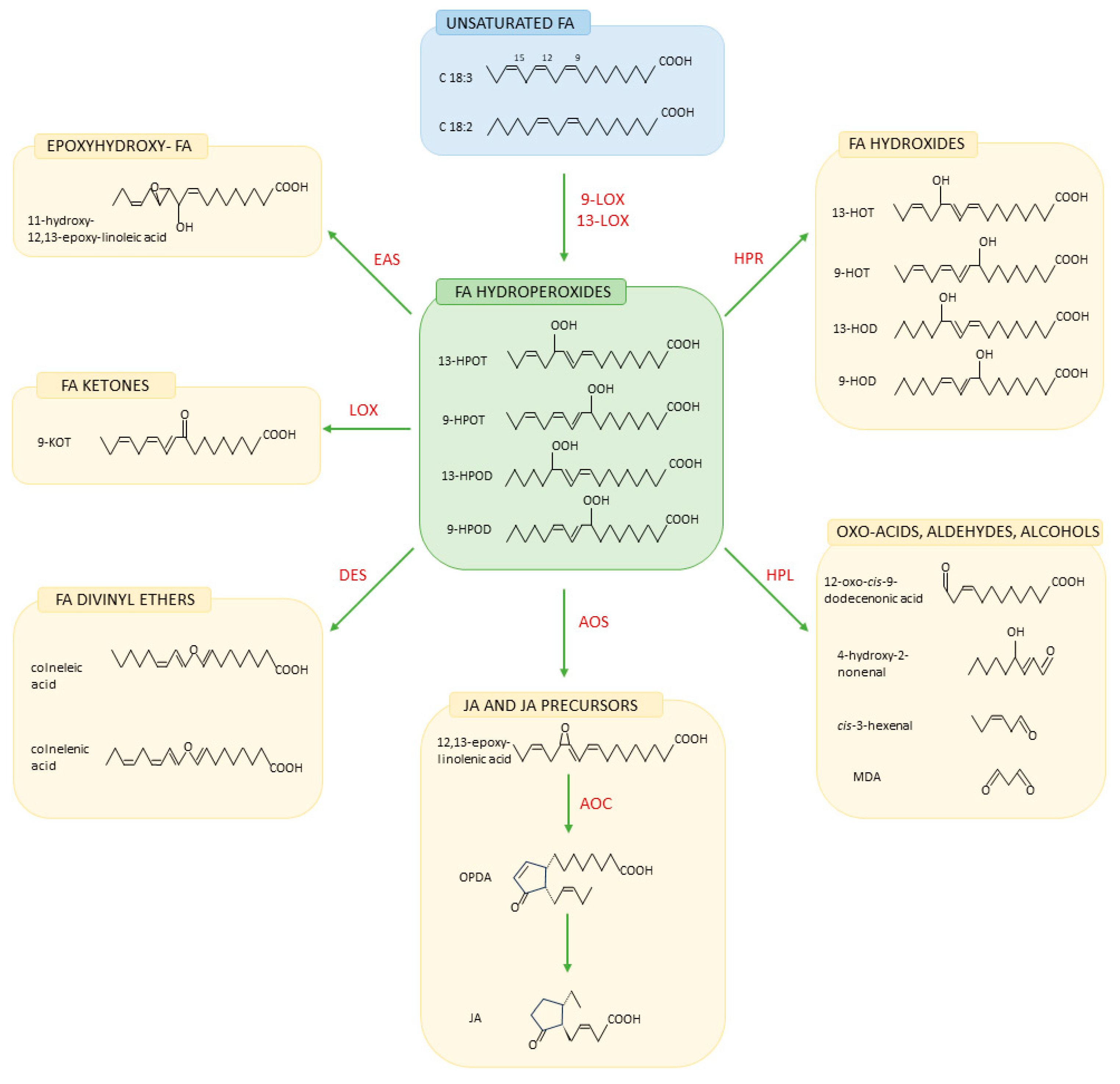
5. Free Fatty Acids and Phytooxylipins in Plant–Pathogen Interactions
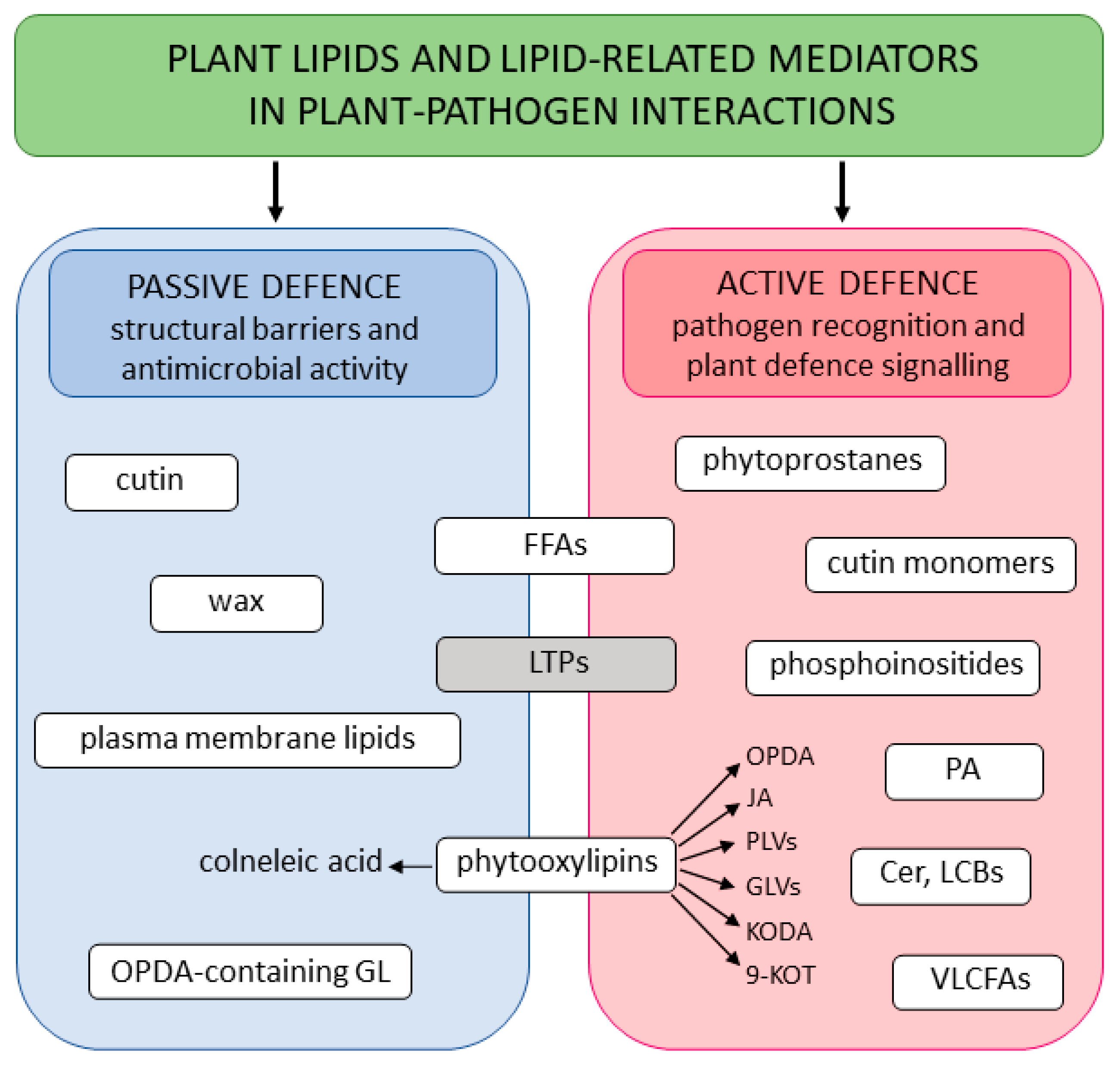
6. Other Lipids and Lipid-Related Mediators Involved in Plant–Pathogen Interactions
7. Concluding Remarks and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Shah, J. Lipids, Lipases, and Lipid-Modifying Enzymes in Plant Disease Resistance. Annu. Rev. Phytopathol. 2005, 43, 229–260. [Google Scholar] [CrossRef]
- Siebers, M.; Brands, M.; Wewer, V.; Duan, Y.; Hölzl, G.; Dörmann, P. Lipids in Plant–Microbe Interactions. Biochim. Biophys. Acta Mol. Cell Biol. Lipids 2016, 1861, 1379–1395. [Google Scholar] [CrossRef]
- Pretorius, C.J.; Zeiss, D.R.; Dubery, I.A. The Presence of Oxygenated Lipids in Plant Defense in Response to Biotic Stress: A Metabolomics Appraisal. Plant Signal. Behav. 2021, 16, 1989215. [Google Scholar] [CrossRef]
- Seth, T.; Asija, S.; Umar, S.; Gupta, R. The Intricate Role of Lipids in Orchestrating Plant Defense Responses. Plant Sci. 2024, 338, 111904. [Google Scholar] [CrossRef]
- Cavaco, A.R.; Matos, A.R.; Figueiredo, A. Speaking the Language of Lipids: The Cross-Talk between Plants and Pathogens in Defence and Disease. Cell. Mol. Life Sci. 2021, 78, 4399–4415. [Google Scholar] [CrossRef]
- Reszczyńska, E.; Hanaka, A. Lipids Composition in Plant Membranes. Cell Biochem. Biophys. 2020, 78, 401–414. [Google Scholar] [CrossRef]
- Kehelpannala, C.; Rupasinghe, T.; Hennessy, T.; Bradley, D.; Ebert, B.; Roessner, U. The State of the Art in Plant Lipidomics. Mol. Omics 2021, 17, 894–910. [Google Scholar] [CrossRef]
- Shimojima, M. Biosynthesis and Functions of the Plant Sulfolipid. Prog. Lipid Res. 2011, 50, 234–239. [Google Scholar] [CrossRef]
- Hölzl, G.; Dörmann, P. Chloroplast Lipids and Their Biosynthesis. Annu. Rev. Plant Biol. 2019, 70, 51–81. [Google Scholar] [CrossRef]
- Michaelson, L.V.; Napier, J.A.; Molino, D.; Faure, J. Plant Sphingolipids: Their Importance in Cellular Organization and Adaption. Biochim. Biophys. Acta 2016, 1861, 1329–1335. [Google Scholar] [CrossRef]
- Mamode Cassim, A.; Gouguet, P.; Gronnier, J.; Laurent, N.; Germain, V.; Grison, M.; Boutté, Y.; Gerbeau-Pissot, P.; Simon-Plas, F.; Mongrand, S. Plant Lipids: Key Players of Plasma Membrane Organization and Function. Prog. Lipid Res. 2019, 73, 1–27. [Google Scholar] [CrossRef]
- Furt, F.; Simon-Plas, F.; Mongrand, S. Lipids of the Plant Plasma Membrane. In The Plant Plasma Membrane. Plant Cell Monographs; Murphy, A., Schulz, B., Peer, W., Eds.; Springer: Berlin/Heidelberg, Germany, 2011; Volume 19, pp. 3–30. [Google Scholar]
- Macabuhay, A.; Arsova, B.; Walker, R.; Johnson, A.; Watt, M.; Roessner, U. Modulators or Facilitators? Roles of Lipids in Plant Root–Microbe Interactions. Trends Plant Sci. 2022, 27, 180–190. [Google Scholar] [CrossRef]
- Meï, C.; Michaud, M.; Cussac, M.; Albrieux, C.; Gros, V.; Maréchal, E.; Block, M.A.; Jouhet, J.; Rébeillé, F. Levels of Polyunsaturated Fatty Acids Correlate with Growth Rate in Plant Cell Cultures. Sci. Rep. 2015, 5, 15207. [Google Scholar] [CrossRef]
- Popko, J. Encyclopedia of Lipidomics. In Encyclopedia of Lipidomics; Wenk, M.R., Ed.; Springer: Dordrecht, The Netherlands, 2017; pp. 1–11. ISBN 9789400778641. [Google Scholar]
- Moreau, P.; Bessoule, J.J.; Mongrand, S.; Testet, E.; Vincent, P.; Cassagne, C. Lipid Trafficking in Plant Cells. Prog. Lipid Res. 1998, 37, 371–391. [Google Scholar] [CrossRef]
- Kunst, L.; Samuels, L. Plant Cuticles Shine: Advances in Wax Biosynthesis and Export. Curr. Opin. Plant Biol. 2009, 12, 721–727. [Google Scholar] [CrossRef]
- Lu, J.; Xu, Y.; Wang, J.; Singer, S.D.; Chen, G. The Role of Triacylglycerol in Plant Stress Response. Plants 2020, 9, 472. [Google Scholar] [CrossRef]
- Okazaki, Y.; Saito, K. Roles of Lipids as Signaling Molecules and Mitigators during Stress Response in Plants. Plant J. 2014, 79, 584–596. [Google Scholar] [CrossRef]
- Anjali; Kumar, S.; Korra, T.; Thakur, R.; Arytselvan, R.; Kashyap, A.S.; Nehela, Y.; Chaplygin, V.; Minkina, T.; Keswani, C. Role of Plant Secondary Metabolites in Defence and Transcriptional Regulation in Response to Biotic Stress. Plant Stress 2023, 8, 100154. [Google Scholar] [CrossRef]
- Cook, D.E.; Mesarich, C.H.; Thomma, B.P.H.J. Understanding Plant Immunity as a Surveillance System to Detect Invasion. Annu. Rev. Phytopathol. 2015, 53, 541–563. [Google Scholar] [CrossRef] [PubMed]
- Bentham, A.R.; de la Concepcion, J.C.; Mukhi, N.; Zdrzałek, R.; Draeger, M.; Gorenkin, D.; Hughes, R.K.; Banfield, M.J. A Molecular Roadmap to the Plant Immune System. J. Biol. Chem. 2020, 295, 14916–14935. [Google Scholar] [CrossRef] [PubMed]
- Arya, G.C.; Sarkar, S.; Manasherova, E.; Aharoni, A.; Cohen, H. The Plant Cuticle: An Ancient Guardian Barrier Set Against Long-Standing Rivals. Front. Plant Sci. 2021, 12, 663165. [Google Scholar] [CrossRef]
- Malinovsky, F.G.; Fangel, J.U.; Willats, W.G.T. The Role of the Cell Wall in Plant Immunity. Front. Plant Sci. 2014, 5, 1–12. [Google Scholar] [CrossRef]
- Bacete, L.; Mélida, H.; Miedes, E.; Molina, A. Plant Cell Wall-Mediated Immunity: Cell Wall Changes Trigger Disease Resistance Responses. Plant J. 2018, 93, 614–636. [Google Scholar] [CrossRef]
- Sela, D.; Buxdorf, K.; Shi, J.X.; Feldmesser, E.; Schreiber, L.; Aharoni, A.; Levy, M. Overexpression of AtSHN1/WIN1 Provokes Unique Defense Responses. PLoS ONE 2013, 8, e70146. [Google Scholar] [CrossRef]
- Wolf, S. Cell Wall Signaling in Plant Development and Defense. Annu. Rev. Plant Biol. 2022, 73, 323–353. [Google Scholar] [CrossRef]
- Gust, A.A.; Pruitt, R.; Nürnberger, T. Sensing Danger: Key to Activating Plant Immunity. Trends Plant Sci. 2017, 22, 779–791. [Google Scholar] [CrossRef]
- Zhou, J.M.; Zhang, Y. Plant Immunity: Danger Perception and Signaling. Cell 2020, 181, 978–989. [Google Scholar] [CrossRef]
- Hou, S.; Liu, Z.; Shen, H.; Wu, D. Damage-Associated Molecular Pattern-Triggered Immunity in Plants. Front. Plant Sci. 2019, 10, 646. [Google Scholar] [CrossRef]
- Balint-Kurti, P. The Plant Hypersensitive Response: Concepts, Control and Consequences. Mol. Plant Pathol. 2019, 20, 1163–1178. [Google Scholar] [CrossRef]
- Vlot, A.C.; Sales, J.H.; Lenk, M.; Bauer, K.; Brambilla, A.; Sommer, A.; Chen, Y.; Wenig, M.; Nayem, S. Systemic Propagation of Immunity in Plants. New Phytol. 2020, 229, 1234–1250. [Google Scholar] [CrossRef]
- Ádám, A.L.; Nagy, Z.; Kátay, G.; Mergenthaler, E.; Viczián, O. Signals of Systemic Immunity in Plants: Progress and Open Questions. Int. J. Mol. Sci. 2018, 19, 1146. [Google Scholar] [CrossRef]
- Luna, E.; Bruce, T.J.A.; Roberts, M.R.; Flors, V.; Ton, J. Next-Generation Systemic Acquired Resistance. Plant Physiol. 2012, 158, 844–853. [Google Scholar] [CrossRef]
- López Sánchez, A.; Pascual-Pardo, D.; Furci, L.; Roberts, M.R.; Ton, J. Costs and Benefits of Transgenerational Induced Resistance in Arabidopsis. Front. Plant Sci. 2021, 12, 644999. [Google Scholar] [CrossRef]
- Klessig, D.F.; Choi, H.W.; Dempsey, D.A. Systemic Acquired Resistance and Salicylic Acid: Past, Present, and Future. Mol. Plant-Microbe Interact. 2018, 31, 871–888. [Google Scholar] [CrossRef]
- Yuan, M.; Ngou, B.P.M.; Ding, P.; Xin, X.F. PTI-ETI Crosstalk: An Integrative View of Plant Immunity. Curr. Opin. Plant Biol. 2021, 62, 102030. [Google Scholar] [CrossRef]
- Naveed, Z.A.; Wei, X.; Chen, J.; Mubeen, H.; Ali, G.S. The PTI to ETI Continuum in Phytophthora-Plant Interactions. Front. Plant Sci. 2020, 11, 593905. [Google Scholar] [CrossRef]
- Ziv, C.; Zhao, Z.; Gao, Y.G.; Xia, Y. Multifunctional Roles of Plant Cuticle during Plant-Pathogen Interactions. Front. Plant Sci. 2018, 9, 1088. [Google Scholar] [CrossRef]
- Hansjakob, A.; Bischof, S.; Bringmann, G.; Riederer, M.; Hildebrandt, U. Very-Long-Chain Aldehydes Promote in Vitro Prepenetration Processes of Blumeria Graminis in a Dose- and Chain Length-Dependent Manner. New Phytol. 2010, 188, 1039–1054. [Google Scholar] [CrossRef]
- Hansjakob, A.; Riederer, M.; Hildebrandt, U. Wax Matters: Absence of Very-Long-Chain Aldehydes from the Leaf Cuticular Wax of the Glossy11 Mutant of Maize Compromises the Prepenetration Processes of Blumeria Graminis. Plant Pathol. 2011, 60, 1151–1161. [Google Scholar] [CrossRef]
- Beattie, G.A.; Marcell, L.M. Effect of Alterations in Cuticular Wax Biosynthesis on the Physicochemical Properties and Topography of Maize Leaf Surfaces. Plant Cell Environ. Environ. 2002, 25, 1–16. [Google Scholar] [CrossRef]
- Burch, A.Y.; Zeisler, V.; Yokota, K.; Schreiber, L.; Lindow, S.E. The Hygroscopic Biosurfactant Syringafactin Produced by Pseudomonas Syringae Enhances Fitness on Leaf Surfaces during Fluctuating Humidity. Environ. Microbiol. 2014, 16, 2086–2098. [Google Scholar] [CrossRef]
- Ni, Y.; Guo, Y.-J.; Wang, J.; Xia, R.-E.; Wang, X.-Q.; Ash, G.; Li, J.-N. Responses of Physiological Indexes and Leaf Epicuticular Waxes of Brassica Napus to Sclerotinia Sclerotiorum Infection. Plant Pathol. 2014, 63, 174–184. [Google Scholar] [CrossRef]
- Yeats, T.H.; Rose, J.K.C. The Formation and Function of Plant Cuticles. Plant Physiol. 2013, 163, 5–20. [Google Scholar] [CrossRef]
- Wang, F.; Zhang, P.; Qiang, S.; Zhu, Y.Z.; Xu, L.L. Effects of Epicuticular Wax from Digitaria Sanguinalis and Festuca Arundinacea on Infection by Curvularia Eragrostidis. Australas. Plant Pathol. 2008, 37, 43–52. [Google Scholar] [CrossRef]
- Gniwotta, F.; Vogg, G.; Gartmann, V.; Carver, T.L.W.; Riederer, M.; Jetter, R. What Do Microbes Encounter at the Plant Surface? Chemical Composition of Pea Leaf Cuticular Waxes 1. Plant Physiol. 2005, 139, 519–530. [Google Scholar] [CrossRef]
- Uppalapati, S.R.; Ishiga, Y.; Doraiswamy, V.; Bedair, M.; Mittal, S.; Chen, J.; Nakashima, J.; Tang, Y.; Tadege, M.; Ratet, P.; et al. Loss of Abaxial Leaf Epicuticular Wax in Medicago Truncatula Irg1/Palm1 Mutants Results in Reduced Spore Differentiation of Anthracnose and Nonhost Rust Pathogens. Plant Cell 2012, 24, 353–370. [Google Scholar] [CrossRef]
- Reisige, K.; Gorzelanny, C.; Daniels, U.; Moerschbacher, B.M. The C28 Aldehyde Octacosanal Is a Morphogenetically Active Component Involved in Host Plant Recognition and Infection Structure Differentiation in the Wheat Stem Rust Fungus. Physiol. Mol. Plant Pathol. 2006, 68, 33–40. [Google Scholar] [CrossRef]
- Wang, W.; Liu, X.; Gai, X.; Ren, J.; Liu, X.; Cai, Y.; Wang, Q.; Ren, H. Cucumis sativus L. WAX2 Plays a Pivotal Role in Wax Biosynthesis, Influencing Pollen Fertility and Plant Biotic and Abiotic Stress Responses. Plant Cell Physiol. 2015, 56, 1339–1354. [Google Scholar] [CrossRef]
- Podila, G.K.; Rogers, L.M.; Kolattukudy, P.E. Chemical Signals from Avocado Surface Wax Trigger Germination and Appressorium Formation in Colletotrichum Gloeosporioides. Plant Physiol. 1993, 103, 267–272. [Google Scholar] [CrossRef]
- Bourdenx, B.; Bernard, A.; Domergue, F.; Pascal, S.; Léger, A.; Roby, D.; Pervent, M.; Vile, D.; Haslam, R.P.; Napier, J.A.; et al. Overexpression of Arabidopsis ECERIFERUM1 Promotes Wax Very-Long-Chain Alkane Biosynthesis and Influences Plant Response to Biotic and Abiotic Stresses. Plant Physiol. 2011, 156, 29–45. [Google Scholar] [CrossRef]
- Lewandowska, M.; Keyl, A.; Feussner, I. Wax Biosynthesis in Response to Danger: Its Regulation upon Abiotic and Biotic Stress. New Phytol. 2020, 227, 698–713. [Google Scholar] [CrossRef]
- Raffaele, S.; Vailleau, F.; Léger, A.; Joubès, J.; Miersch, O.; Huard, C.; Blée, E.; Mongrand, S.; Domergue, F.; Roby, D. A MYB Transcription Factor Regulates Very-Long-Chain Fatty Acid Biosynthesis for Activation of the Hypersensitive Cell Death Response in Arabidopsis. Plant Cell 2008, 20, 752–767. [Google Scholar] [CrossRef]
- Zhang, Y.-L.; Zhang, C.-L.; Wang, G.-L.; Wang, Y.-X.; Qi, C.-H.; Zhao, Q.; You, C.-X.; Li, Y.-Y.; Hao, Y.-J. The R2R3 MYB Transcription Factor MdMYB30 Modulates Plant Resistance against Pathogens by Regulating Cuticular Wax Biosynthesis. BMC Plant Biol. 2019, 19, 362. [Google Scholar] [CrossRef]
- Serrano, M.; Coluccia, F.; Torres, M.; L’Haridon, F.; Métraux, J.P. The Cuticle and Plant Defense to Pathogens. Front. Plant Sci. 2014, 5, 274. [Google Scholar] [CrossRef]
- L’Haridon, F.; Besson-Bard, A.; Binda, M.; Serrano, M.; Abou-Mansour, E.; Balet, F.; Schoonbeek, H.J.; Hess, S.; Mir, R.; Léon, J.; et al. A Permeable Cuticle Is Associated with the Release of Reactive Oxygen Species and Induction of Innate Immunity. PLoS Pathog. 2011, 7, e1002148. [Google Scholar] [CrossRef]
- Xia, Y.; Gao, Q.M.; Yu, K.; Lapchyk, L.; Navarre, D.R.; Hildebrand, D.; Kachroo, A.; Kachroo, P. An Intact Cuticle in Distal Tissues Is Essential for the Induction of Systemic Acquired Resistance in Plants. Cell Host Microbe 2009, 5, 151–165. [Google Scholar] [CrossRef]
- Li, Y.; Beisson, F.; Koo, A.J.K.; Molina, I.; Pollard, M.; Ohlrogge, J. Identification of Acyltransferases Required for Cutin Biosynthesis and Production of Cutin with Suberin-like Monomers. Proc. Natl. Acad. Sci. USA 2007, 104, 18339–18344. [Google Scholar] [CrossRef]
- Arya, G.C.; Cohen, H. The Multifaceted Roles of Fungal Cutinases during Infection. J. Fungi 2022, 8, 199. [Google Scholar] [CrossRef]
- Wang, Y.; Chen, J.; Li, D.W.; Zheng, L.; Huang, J. CglCUT1 Gene Required for Cutinase Activity and Pathogenicity of Colletotrichum Gloeosporioides Causing Anthracnose of Camellia Oleifera. Eur. J. Plant Pathol. 2017, 147, 103–114. [Google Scholar] [CrossRef]
- Li, D.; Ashby, A.M.; Johnstone, K. Molecular Evidence That the Extracellular Cutinase Pbc1 Is Required for Pathogenicity of Pyrenopeziza Brassicae on Oilseed Rape. Mol. Plant-Microbe Interact. 2003, 16, 545–552. [Google Scholar] [CrossRef]
- Lu, L.; Rong, W.; Massart, S.; Zhang, Z. Genome-Wide Identification and Expression Analysis of Cutinase Gene Family in Rhizoctonia Cerealis and Functional Study of an Active Cutinase RcCUT1 in the Fungal-Wheat Interaction. Front. Microbiol. 2018, 9, 1813. [Google Scholar] [CrossRef]
- Liu, T.; Hou, J.; Wang, Y.; Jin, Y.; Borth, W.; Zhao, F.; Liu, Z.; Hu, J.; Zuo, Y. Genome-Wide Identification, Classification and Expression Analysis in Fungal–Plant Interactions of Cutinase Gene Family and Functional Analysis of a Putative ClCUT7 in Curvularia Lunata. Mol. Genet. Genom. 2016, 291, 1105–1115. [Google Scholar] [CrossRef]
- Leroch, M.; Kleber, A.; Silva, E.; Coenen, T.; Koppenhöfer, D.; Shmaryahu, A.; Valenzuela, P.D.T.; Hahn, M. Transcriptome Profiling of Botrytis Cinerea Conidial Germination Reveals Upregulation of Infection-Related Genes during the Prepenetration Stage. Eucaryotic Cell 2013, 12, 614–625. [Google Scholar] [CrossRef]
- Gui, Y.J.; Zhang, W.Q.; Zhang, D.D.; Zhou, L.; Short, D.P.G.; Wang, J.; Ma, X.F.; Li, T.G.; Kong, Z.Q.; Wang, B.L.; et al. A Verticillium Dahliae Extracellular Cutinase Modulates Plant Immune Responses. Mol. Plant-Microbe Interact. 2018, 31, 260–273. [Google Scholar] [CrossRef]
- Stahl, D.J.; Schäfer, W. Cutinase Is Not Required for Fungal Pathogenicity on Pea. Plant Cell 1992, 4, 621–629. [Google Scholar] [CrossRef]
- Sweigard, J.A.; Chumley, F.G.; Valent, B. Disruption of a Magnaporthe Grisea Cutinase Gene. Mol. Gen. Genet. 1992, 232, 183–190. [Google Scholar] [CrossRef]
- Li, D.; Sirakova, T.; Rogers, L.; Ettinger, W.F.; Kolattukudy, P.E. Regulation of Constitutively Expressed and Induced Cutinase Genes by Different Zinc Finger Transcription Factors in Fusarium solani f. Sp. Pisi (Nectria haematococca). J. Biol. Chem. 2002, 277, 7905–7912. [Google Scholar] [CrossRef]
- Martins, I.; Hartmann, D.O.; Alves, P.C.; Martins, C.; Garcia, H.; Leclercq, C.C.; Ferreira, R.; He, J.; Renaut, J.; Becker, J.D.; et al. Elucidating How the Saprophytic Fungus Aspergillus Nidulans Uses the Plant Polyester Suberin as Carbon Source. BMC Genomics 2014, 15, 1–19. [Google Scholar] [CrossRef]
- Xiao, F.; Goodwin, S.M.; Xiao, Y.; Sun, Z.; Baker, D.; Tang, X.; Jenks, M.A.; Zhou, J.M. Arabidopsis CYP86A2 Represses Pseudomonas Syringae Type III Genes and Is Required for Cuticle Development. EMBO J. 2004, 23, 2903–2913. [Google Scholar] [CrossRef]
- Fauth, M.; Schweizer, P.; Buchala, A.; Markstadter, C.; Riederer, M.; Kato, T.; Kauss, H. Cutin Monomers and Surface Wax Constituents Elicit H2O2 in Conditioned Cucumber Hypocotyl Segments and Enhance the Activity of Other H2O2 Elicitors. Plant Physiol. 1998, 117, 1373–1380. [Google Scholar] [CrossRef]
- Schweizer, P.; Jeanguenat, A.; Whitacre, D.; Métraux, J.P.; Mösinger, E. Induction of Resistance in Barley against Erysiphe Graminis f.Sp. Hordei by Free Cutin Monomers. Physiol. Mol. Plant Pathol. 1996, 49, 103–120. [Google Scholar] [CrossRef]
- Schweizer, P.; Jeanguenat, A.; Mösinger, E.; Métraux, J.P. Plant Protection by Free Cutin Monomers in Two Cereal Pathosystems. Adv. Mol. Genet. Plant-Microbe Interact. 1994, 3, 371–374. [Google Scholar]
- Tanaka, K.; Heil, M. Damage-Associated Molecular Patterns (DAMPs) in Plant Innate Immunity: Applying the Danger Model and Evolutionary Perspectives. Annu. Rev. Phytopathol. 2021, 59, 53–75. [Google Scholar] [CrossRef]
- Zhao, J. Phospholipase D and Phosphatidic Acid in Plant Defence Response: From Protein-Protein and Lipid-Protein Interactions to Hormone Signalling. J. Exp. Bot. 2015, 66, 1721–1736. [Google Scholar] [CrossRef]
- Casillas-Vargas, G.; Ocasio-Malavé, C.; Medina, S.; Morales-Guzmán, C.; Del Valle, R.G.; Carballeira, N.M.; Sanabria-Ríos, D.J. Antibacterial Fatty Acids: An Update of Possible Mechanisms of Action and Implications in the Development of the next-Generation of Antibacterial Agents. Prog. Lipid Res. 2021, 82, 101093. [Google Scholar] [CrossRef]
- McGaw, L.J.; Jäger, A.K.; Van Staden, J. Antibacterial Effects of Fatty Acids and Related Compounds from Plants. S. Afr. J. Bot. 2002, 68, 417–423. [Google Scholar] [CrossRef]
- Liu, S.; Ruan, W.; Li, J.; Xu, H.; Wang, J.; Gao, Y.; Wang, J. Biological Control of Phytopathogenic Fungi by Fatty Acids. Mycopathologia 2008, 166, 93–102. [Google Scholar] [CrossRef]
- Walters, D.; Raynor, L.; Mitchell, A.; Walker, R.; Walker, K. Antifungal Activities of Four Fatty Acids against Plant Pathogenic Fungi. Mycopathologia 2004, 157, 87–90. [Google Scholar] [CrossRef]
- Guimarães, A.; Venâncio, A. The Potential of Fatty Acids and Their Derivatives as Antifungal Agents: A Review. Toxins 2022, 14, 188. [Google Scholar] [CrossRef]
- Yaeno, T.; Matsuda, O.; Iba, K. Role of Chloroplast Trienoic Fatty Acids in Plant Disease Defense Responses. Plant J. 2004, 40, 931–941. [Google Scholar] [CrossRef]
- Xiao, R.; Zou, Y.; Guo, X.; Li, H.; Lu, H. Fatty Acid Desaturases (FADs) Modulate Multiple Lipid Metabolism Pathways to Improve Plant Resistance. Mol. Biol. Rep. 2022, 49, 9997–10011. [Google Scholar] [CrossRef]
- Nandi, A.; Moeder, W.; Kachroo, P.; Klessig, D.F.; Shah, J. Arabidopsis Ssi2-Conferred Susceptibility to Botrytis Cinerea Is Dependent on EDS5 and PAD4. Mol. Plant-Microbe Interact. 2005, 18, 363–370. [Google Scholar] [CrossRef]
- Chandra-Shekara, A.C.; Venugopal, S.C.; Barman, S.R.; Kachroo, A.; Kachroo, P. Plastidial Fatty Acid Levels Regulate Resistance Gene-Dependent Defense Signaling in Arabidopsis. Proc. Natl. Acad. Sci. USA 2007, 104, 7277–7282. [Google Scholar] [CrossRef] [PubMed]
- Upchurch, R.G. Fatty Acid Unsaturation, Mobilization, and Regulation in the Response of Plants to Stress. Biotechnol. Lett. 2008, 30, 967–977. [Google Scholar] [CrossRef]
- Ruan, J.; Zhou, Y.; Zhou, M.; Yan, J.; Khurshid, M.; Weng, W.; Cheng, J.; Zhang, K. Jasmonic Acid Signaling Pathway in Plants. Int. J. Mol. Sci. 2019, 20, 2479. [Google Scholar] [CrossRef]
- Blée, E. Impact of Phyto-Oxylipins in Plant Defense. Trends Plant Sci. 2002, 7, 315–322. [Google Scholar] [CrossRef] [PubMed]
- Mosblech, A.; Feussner, I.; Heilmann, I. Oxylipins: Structurally Diverse Metabolites from Fatty Acid Oxidation. Plant Physiol. Biochem. 2009, 47, 511–517. [Google Scholar] [CrossRef]
- Wang, Y.; Mostafa, S.; Zeng, W.; Jin, B. Function and Mechanism of Jasmonic Acid in Plant Responses to Abiotic and Biotic Stresses. Int. J. Mol. Sci. 2021, 22, 8568. [Google Scholar] [CrossRef] [PubMed]
- Robert-Seilaniantz, A.; Grant, M.; Jones, J.D.G. Hormone Crosstalk in Plant Disease and Defense: More Than Just JASMONATE-SALICYLATE Antagonism. Annu. Rev. Phytopathol. 2011, 49, 317–343. [Google Scholar] [CrossRef]
- Berens, M.L.; Berry, H.M.; Mine, A.; Argueso, C.T.; Tsuda, K. Evolution of Hormone Signaling Networks in Plant Defense. Annu. Rev. Phytopathol. 2017, 55, 401–425. [Google Scholar] [CrossRef]
- Macioszek, V.K.; Jęcz, T.; Ciereszko, I.; Kononowicz, A.K. Jasmonic Acid as a Mediator in Plant Response to Necrotrophic Fungi. Cells 2023, 12, 1027. [Google Scholar] [CrossRef] [PubMed]
- Deboever, E.; Deleu, M.; Mongrand, S.; Lins, L.; Fauconnier, M.-L. Plant-Pathogen Interactions: Underestimated Roles of Phyto-Oxylipins. Trends Plant Sci. 2020, 25, 22–34. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Yu, G.; Cao, C.; Liu, P. Metabolism, Signaling, and Transport of Jasmonates. Plant Commun. 2021, 2, 100231. [Google Scholar] [CrossRef] [PubMed]
- Ghorbel, M.; Brini, F.; Sharma, A.; Landi, M. Role of Jasmonic Acid in Plants: The Molecular Point of View. Plant Cell Rep. 2021, 40, 1471–1494. [Google Scholar] [CrossRef] [PubMed]
- Jimenez Aleman, G.H.; Thirumalaikumar, V.P.; Jander, G.; Fernie, A.R.; Skirycz, A. OPDA, More than Just a Jasmonate Precursor. Phytochemistry 2022, 204, 113432. [Google Scholar] [CrossRef]
- Scalschi, L.; Sanmartín, M.; Camañes, G.; Troncho, P.; Sánchez-Serrano, J.J.; García-Agustín, P.; Vicedo, B. Silencing of OPR3 in Tomato Reveals the Role of OPDA in Callose Deposition during the Activation of Defense Responses against Botrytis Cinerea. Plant J. 2015, 81, 304–315. [Google Scholar] [CrossRef] [PubMed]
- Shinya, T.; Miyamoto, K.; Uchida, K.; Hojo, Y.; Yumoto, E.; Okada, K.; Yamane, H.; Galis, I. Chitooligosaccharide Elicitor and Oxylipins Synergistically Elevate Phytoalexin Production in Rice. Plant Mol. Biol. 2022, 109, 595–609. [Google Scholar] [CrossRef] [PubMed]
- Taki, N.; Sasaki-Sekimoto, Y.; Obayashi, T.; Kikuta, A.; Kobayashi, K.; Ainai, T.; Yagi, K.; Sakurai, N.; Suzuki, H.; Masuda, T.; et al. 12-Oxo-Phytodienoic Acid Triggers Expression of a Distinct Set of Genes and Plays a Role in Wound-Induced Gene Expression in Arabidopsis. Plant Physiol. 2005, 139, 1268–1283. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.D.; Borrego, E.J.; Kenerley, C.M.; Kolomiets, M.V. Oxylipins Other Than Jasmonic Acid Are Xylem-Resident Signals Regulating Systemic Resistance Induced by Trichoderma Virens in Maize. Plant Cell 2020, 32, 166–185. [Google Scholar] [CrossRef]
- Knieper, M.; Viehhauser, A.; Dietz, K.J. Oxylipins and Reactive Carbonyls as Regulators of the Plant Redox and Reactive Oxygen Species Network under Stress. Antioxidants 2023, 12, 814. [Google Scholar] [CrossRef]
- Wasternack, C.; Hause, B. OPDA-Ile—A New JA-Ile-Independent Signal? Plant Signal. Behav. 2016, 11, e1253646. [Google Scholar] [CrossRef] [PubMed]
- Mueller, S.; Hilbert, B.; Dueckershoff, K.; Roitsch, T.; Krischke, M.; Mueller, M.J.; Berger, S.; Biowissenschaften, J.; Biologie, P.; Wuerzburg, U. General Detoxification and Stress Responses Are Mediated by Oxidized Lipids through TGA Transcription Factors in Arabidopsis. Plant Cell 2008, 20, 768–785. [Google Scholar] [CrossRef] [PubMed]
- Kourtchenko, O.; Andersson, M.X.; Hamberg, M.; Brunnström, Å.; Göbel, C.; McPhail, K.L.; Gerwick, W.H.; Feussner, I.; Ellerström, M. Oxo-Phytodienoic Acid-Containing Galactolipids in Arabidopsis: Jasmonate Signaling Dependence. Plant Physiol. 2007, 145, 1658–1669. [Google Scholar] [CrossRef] [PubMed]
- Nilsson, A.K.; Johansson, O.N.; Fahlberg, P.; Steinhart, F.; Gustavsson, M.B.; Ellerström, M.; Andersson, M.X. Formation of Oxidized Phosphatidylinositol and 12-Oxo-Phytodienoic Acid Containing Acylated Phosphatidylglycerol during the Hypersensitive Response in Arabidopsis. Phytochemistry 2014, 101, 65–75. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Park, S.-W. 12-Oxo-Phytodienoic Acid: A Fuse and/or Switch of Plant Growth and Defense Responses? Front. Plant Sci. 2021, 12, 724079. [Google Scholar] [CrossRef] [PubMed]
- Cheong, H.; Dos Santos, I.B.; Liu, W.; Gosse, H.N.; Park, S.-W.; Liu, W.; Gosse, H.N.; Park, W. Cyclophilin 20-3 Is Positioned as a Regulatory Hub between Light-Dependent Redox and 12-Oxo-Phytodienoic Acid Signaling. Plant Signal. Behav. 2017, 12, e1362520. [Google Scholar] [CrossRef] [PubMed]
- Gorman, Z.; Tolley, J.P.; Koiwa, H.; Kolomiets, M.V. The Synthesis of Pentyl Leaf Volatiles and Their Role in Resistance to Anthracnose Leaf Blight. Front. Plant Sci. 2021, 12, 719587. [Google Scholar] [CrossRef] [PubMed]
- Tolley, J.P.; Gorman, Z.; Lei, J.; Yeo, I.-C.; Nagashima, Y.; Jashi, V.; Zhu-Salzman, K.; Kolomiets, M.V.; Koiwa, H. Overexpression of Maize ZmLOX6 in Arabidopsis Thaliana Enhances Damage-Induced Pentyl Leaf Volatile Emissions That Affect Plant Growth and Interaction with Aphids. J. Exp. Bot. 2023, 74, 1990–2004. [Google Scholar] [CrossRef]
- Gorman, Z.; Christensen, S.A.; Yan, Y.; He, Y.; Borrego, E.; Kolomiets, M.V. Green Leaf Volatiles and Jasmonic Acid Enhance Susceptibility to Anthracnose Diseases Caused by Colletotrichum Graminicola in Maize. Mol. Plant Pathol. 2020, 21, 702–715. [Google Scholar] [CrossRef]
- Song, G.C.; Ryu, C.M. Two Volatile Organic Compounds Trigger Plant Self-Defense against a Bacterial Pathogen and a Sucking Insect in Cucumber under Open Field Conditions. Int. J. Mol. Sci. 2013, 14, 9803–9819. [Google Scholar] [CrossRef]
- Song, G.C.; Choi, H.K.; Ryu, C.-M. Gaseous 3-Pentanol Primes Plant Immunity against a Bacterial Speck Pathogen, Pseudomonas Syringae Pv. Tomato via Salicylic Acid and Jasmonic Acid-Dependent Signaling Pathways in Arabidopsis. Front. Plant Sci. 2015, 6, 821. [Google Scholar] [CrossRef]
- Shen, J.; Tieman, D.; Jones, J.B.; Taylor, M.G.; Schmelz, E.; Huffaker, A.; Bies, D.; Chen, K.; Klee, H.J. A 13-Lipoxygenase, TomloxC, Is Essential for Synthesis of C5 Flavour Volatiles in Tomato. J. Exp. Bot. 2014, 65, 419–428. [Google Scholar] [CrossRef] [PubMed]
- Vicente, J.; Cascón, T.; Vicedo, B.; García-Agustín, P.; Hamberg, M.; Castresana, C. Role of 9-Lipoxygenase and α-Dioxygenase Oxylipin Pathways as Modulators of Local and Systemic Defense. Mol. Plant 2012, 5, 914–928. [Google Scholar] [CrossRef]
- Göbel, C.; Feussner, I.; Schmidt, A.; Scheel, D.; Sanchez-Serrano, J.; Hamberg, M.; Rosahl, S. Oxylipin Profiling Reveals the Preferential Stimulation of the 9-Lipoxygenase Pathway in Elicitor-Treated Potato Cells. J. Biol. Chem. 2001, 276, 6267–6273. [Google Scholar] [CrossRef] [PubMed]
- Mène-Saffrané, L.; Esquerré-Tugayé, M.T.; Fournier, J. Constitutive Expression of an Inducible Lipoxygenase in Transgenic Tobacco Decreases Susceptibility to Phytophthora Parasitica Var. Nicotianae. Mol. Breed. 2003, 12, 271–282. [Google Scholar] [CrossRef]
- Fauconnier, M.-L.; Rojas-Beltran, J.; Dupuis, B.; Delaplace, P.; Frettinger, P.; Gosset, V.; du Jardin, P. Changes in Oxylipin Synthesis after Phytophthora Infestans Infection of Potato Leaves Do Not Correlate with Resistance. Plant Physiol. Biochem. 2008, 46, 823–831. [Google Scholar] [CrossRef]
- Zoeller, M.; Stingl, N.; Krischke, M.; Fekete, A.; Waller, F.; Berger, S.; Mueller, M.J. Lipid Profiling of the Arabidopsis Hypersensitive Response Reveals Specific Lipid Peroxidation and Fragmentation Processes: Biogenesis of Pimelic and Azelaic Acid. Plant Physiol. 2012, 160, 365–378. [Google Scholar] [CrossRef] [PubMed]
- Lim, G.H.; Singhal, R.; Kachroo, A.; Kachroo, P. Fatty Acid- and Lipid-Mediated Signaling in Plant Defense. Annu. Rev. Phytopathol. 2017, 55, 505–536. [Google Scholar] [CrossRef] [PubMed]
- Gao, Q.M.; Yu, K.; Xia, Y.; Shine, M.B.; Wang, C.; Navarre, D.R.; Kachroo, A.; Kachroo, P. Mono- and Digalactosyldiacylglycerol Lipids Function Nonredundantly to Regulate Systemic Acquired Resistance in Plants. Cell Rep. 2014, 9, 1681–1691. [Google Scholar] [CrossRef]
- Thoma, I.; Loeffler, C.; Sinha, A.K.; Gupta, M.; Krischke, M.; Steffan, B.; Roitsch, T.; Mueller, M.J. Cyclopentenone Isoprostanes Induced by Reactive Oxygen Species Trigger Defense Gene Activation and Phytoalexin Accumulation in Plants. Plant J. 2003, 34, 363–375. [Google Scholar] [CrossRef]
- Loeffler, C.; Berger, S.; Guy, A.; Durand, T.; Bringmann, G.; Mueller, M.J.; Dreyer, M.; Von Rad, U.; Durner, J.; Mueller, M.J. B1-Phytoprostanes Trigger Plant Defense and Detoxification Responses. Plant Physiol. 2005, 137, 328–340. [Google Scholar] [CrossRef] [PubMed]
- Christensen, S.A.; Kolomiets, M.V. The Lipid Language of Plant—Fungal Interactions. Fungal Genet. Biol. 2011, 48, 4–14. [Google Scholar] [CrossRef] [PubMed]
- Tsitsigiannis, D.I.; Kunze, S.; Willis, D.K.; Feussner, I.; Keller, N.P. Aspergillus Infection Inhibits the Expression of Peanut 13S-HPODE-Forming Seed Lipoxygenases. Mol. Plant-Microbe Interact. 2005, 18, 1081–1089. [Google Scholar] [CrossRef] [PubMed]
- Brodhagen, M.; Tsitsigiannis, D.I.; Hornung, E.; Goebel, C.; Feussner, I.; Keller, N.P. Reciprocal Oxylipin-Mediated Cross-Talk in the Aspergillus—Seed Pathosystem. Mol. Microbiol. 2008, 67, 378–391. [Google Scholar] [CrossRef] [PubMed]
- Beccaccioli, M.; Reverberi, M.; Scala, V. Fungal Lipids: Biosynthesis and Signalling during Plant-Pathogen Interaction. Front. Biosci. Landmark 2019, 24, 172–185. [Google Scholar] [CrossRef]
- Battilani, P.; Lanubile, A.; Scala, V.; Reverberi, M.; Gregori, R.; Falavigna, C.; Dall’Asta, C.; Park, Y.-S.; Bennett, J.; Borrego, E.J.; et al. Oxylipins from Both Pathogen and Host Antagonize Jasmonic Acid-Mediated Defence via the 9-Lipoxygenase Pathway in Fusarium Verticillioides Infection of Maize. Mol. Plant Pathol. 2018, 19, 2162–2176. [Google Scholar] [CrossRef] [PubMed]
- Eckardt, N.A. Oxylipin Signaling in Plant Stress Responses. Plant Cell 2008, 20, 495–497. [Google Scholar] [CrossRef] [PubMed]
- Andersson, M.X.; Hamberg, M.; Kourtchenko, O.; Brunnstro, Å.; McPhail, K.L.; Gerwick, W.H.; Göbel, C.; Feussner, I.; Ellerström, M. Oxylipin Profiling of the Hypersensitive Response in Arabidopsis Thaliana: Formation of a Novel Oxo-Phytodienoic Acid-Containing Galactolipid, Arabidopside E. J. Biol. Chem. 2006, 281, 31528–31537. [Google Scholar] [CrossRef]
- Han, Z.; Xiong, D.; Schneiter, R.; Tian, C. The Function of Plant PR1 and Other Members of the CAP Protein Superfamily in Plant–Pathogen Interactions. Mol. Plant Pathol. 2023, 24, 651–668. [Google Scholar] [CrossRef]
- Safi, H.; Saibi, W.; Alaoui, M.M.; Hmyene, A.; Masmoudi, K.; Hanin, M.; Brini, F. A Wheat Lipid Transfer Protein (TdLTP4) Promotes Tolerance to Abiotic and Biotic Stress in Arabidopsis Thaliana. Plant Physiol. Biochem. 2015, 89, 64–75. [Google Scholar] [CrossRef]
- Patkar, R.N.; Chattoo, B.B. Transgenic Indica Rice Expressing Ns-LTP-like Protein Shows Enhanced Resistance to Both Fungal and Bacterial Pathogens. Mol. Breed. 2006, 17, 159–171. [Google Scholar] [CrossRef]
- Gao, S.; Guo, W.; Feng, W.; Liu, L.; Song, X.; Chen, J.; Hou, W.; Zhu, H.; Tang, S.; Hu, J. LTP3 Contributes to Disease Susceptibility in Arabidopsis by Enhancing Abscisic Acid (ABA) Biosynthesis. Mol. Plant Pathol. 2016, 17, 412–426. [Google Scholar] [CrossRef] [PubMed]
- Buhot, N.; Gomès, E.; Milat, M.-L.; Ponchet, M.; Marion, D.; Lequeu, J.; Delrot, S.; Coutos-Thévenot, P.; Blein, J.-P. Modulation of the Biological Activity of a Tobacco LTP1 by Lipid Complexation. Mol. Biol. Cell 2004, 15, 5047–5052. [Google Scholar] [CrossRef] [PubMed]
- Girault, T.; François, J.; Rogniaux, H.; Pascal, S.; Delrot, S.; Coutos-Thévenot, P.; Gomès, E. Exogenous Application of a Lipid Transfer Protein-Jasmonic Acid Complex Induces Protection of Grapevine towards Infection by Botrytis Cinerea. Plant Physiol. Biochem. 2008, 46, 140–149. [Google Scholar] [CrossRef] [PubMed]
- Gao, H.; Ma, K.; Ji, G.; Pan, L.; Zhou, Q. Lipid Transfer Proteins Involved in Plant—Pathogen Interactions and Their Molecular Mechanisms. Mol. Plant Pathol. 2022, 23, 1815–1829. [Google Scholar] [CrossRef] [PubMed]
- Jung, H.W.; Tschaplinski, T.J.; Wang, L.; Glazebrook, J.; Greenberg, J.T. Priming in Systemic Resistance. Science (80-.) 2009, 324, 89–92. [Google Scholar] [CrossRef] [PubMed]
- Zeng, H.Y.; Bao, H.N.; Chen, Y.L.; Chen, D.K.; Zhang, K.; Liu, S.K.; Yang, L.; Li, Y.K.; Yao, N. The Two Classes of Ceramide Synthases Play Different Roles in Plant Immunity and Cell Death. Front. Plant Sci. 2022, 13, 824585. [Google Scholar] [CrossRef] [PubMed]
- Berkey, R.; Bendigeri, D.; Xiao, S. Sphingolipids and Plant Defense/Disease: The “Death” Connection and Beyond. Front. Plant Sci. 2012, 3, 68. [Google Scholar] [CrossRef] [PubMed]
- Kong, L.; Ma, X.; Zhang, C.; Kim, S.-I.; Li, B.; Xie, Y.; Yeo, I.-C.; Thapa, H.; Chen, S.; Devarenne, T.P.; et al. Dual Phosphorylation of DGK5-Mediated PA Burst Regulates ROS in Plant Immunity. Cell 2024, 187, 609–623. [Google Scholar] [CrossRef]
- De Jong, C.F.; Laxalt, A.M.; Bargmann, B.O.R.; De Wit, P.J.G.M.; Joosten, M.H.A.J.; Munnik, T. Phosphatidic Acid Accumulation Is an Early Response in the Cf-4/Avr4 Interaction. Plant J. 2004, 39, 1–12. [Google Scholar] [CrossRef]
- Li, J.; Wang, X. Phospholipase D and Phosphatidic Acid in Plant Immunity. Plant Sci. 2019, 279, 45–50. [Google Scholar] [CrossRef] [PubMed]
- Xing, J.; Zhang, L.; Duan, Z.; Lin, J. Coordination of Phospholipid-Based Signaling and Membrane Trafficking in Plant Immunity. Trends Plant Sci. 2021, 26, 407–420. [Google Scholar] [CrossRef] [PubMed]
- Pečenková, T.; Pleskot, R.; Žárský, V. Subcellular Localization of Arabidopsis Pathogenesis-Related 1 (PR1) Protein. Int. J. Mol. Sci. 2017, 18, 825. [Google Scholar] [CrossRef] [PubMed]
- Xing, J.; Li, X.; Wang, X.; Lv, X.; Wang, L.; Zhang, L.; Zhu, Y.; Shen, Q.; Baluška, F.; Šamaj, J.; et al. Secretion of Phospholipase Dδ Functions as a Regulatory Mechanism in Plant Innate Immunity. Plant Cell 2019, 31, 3015–3032. [Google Scholar] [CrossRef]
- Pinosa, F.; Buhot, N.; Kwaaitaal, M.; Fahlberg, P.; Thordal-Christensen, H.; Ellerström, M.; Andersson, M.X. Arabidopsis Phospholipase Dδ Is Involved in Basal Defense and Nonhost Resistance to Powdery Mildew Fungi. Plant Physiol. 2013, 163, 896–906. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kuźniak, E.; Gajewska, E. Lipids and Lipid-Mediated Signaling in Plant–Pathogen Interactions. Int. J. Mol. Sci. 2024, 25, 7255. https://doi.org/10.3390/ijms25137255
Kuźniak E, Gajewska E. Lipids and Lipid-Mediated Signaling in Plant–Pathogen Interactions. International Journal of Molecular Sciences. 2024; 25(13):7255. https://doi.org/10.3390/ijms25137255
Chicago/Turabian StyleKuźniak, Elżbieta, and Ewa Gajewska. 2024. "Lipids and Lipid-Mediated Signaling in Plant–Pathogen Interactions" International Journal of Molecular Sciences 25, no. 13: 7255. https://doi.org/10.3390/ijms25137255




