The Mechanism of Action of L-Tyrosine Derivatives against Chikungunya Virus Infection In Vitro Depends on Structural Changes
Abstract
1. Introduction
2. Results
2.1. Prediction of the Pharmacokinetic Parameters of the Evaluated Compounds
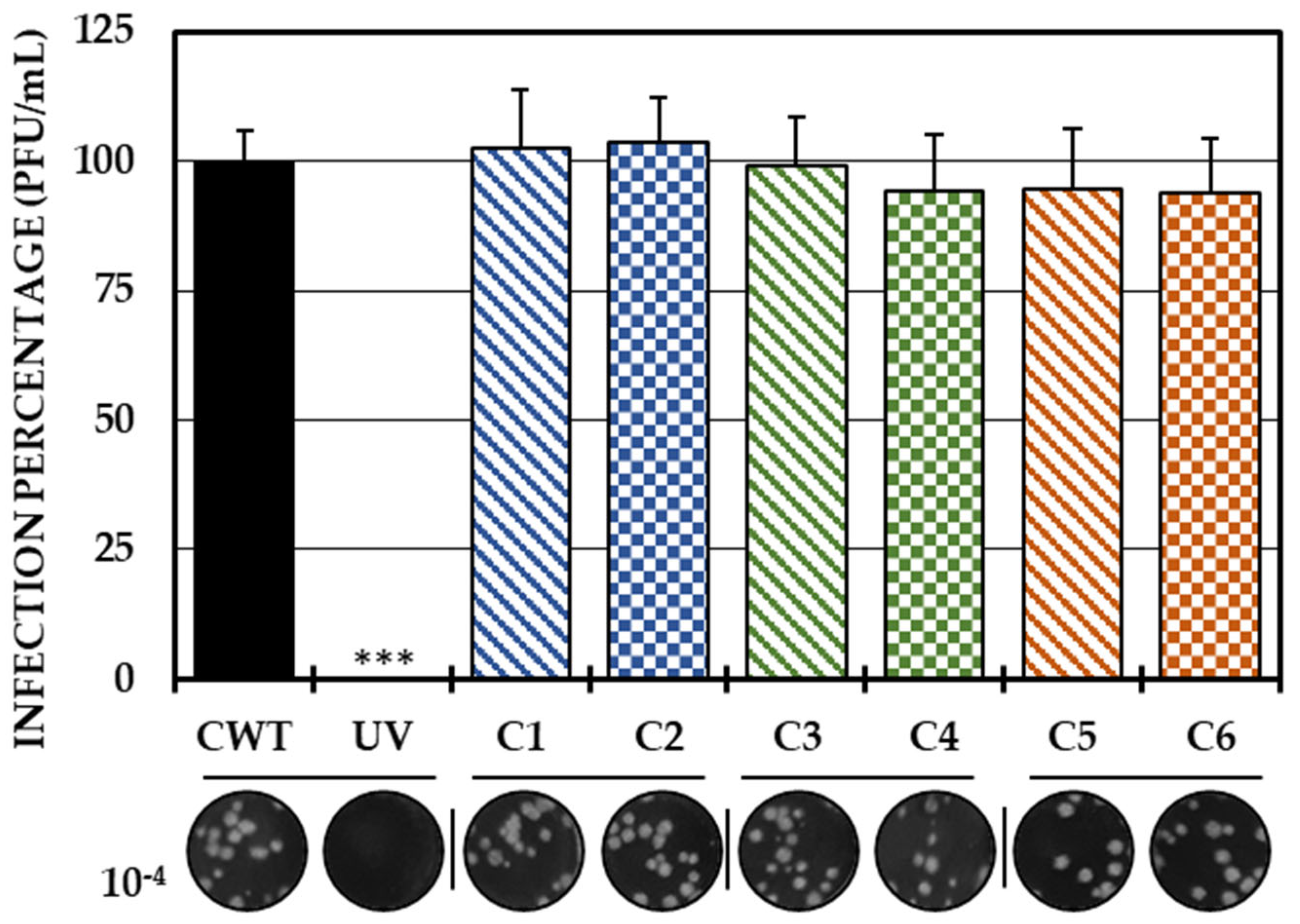
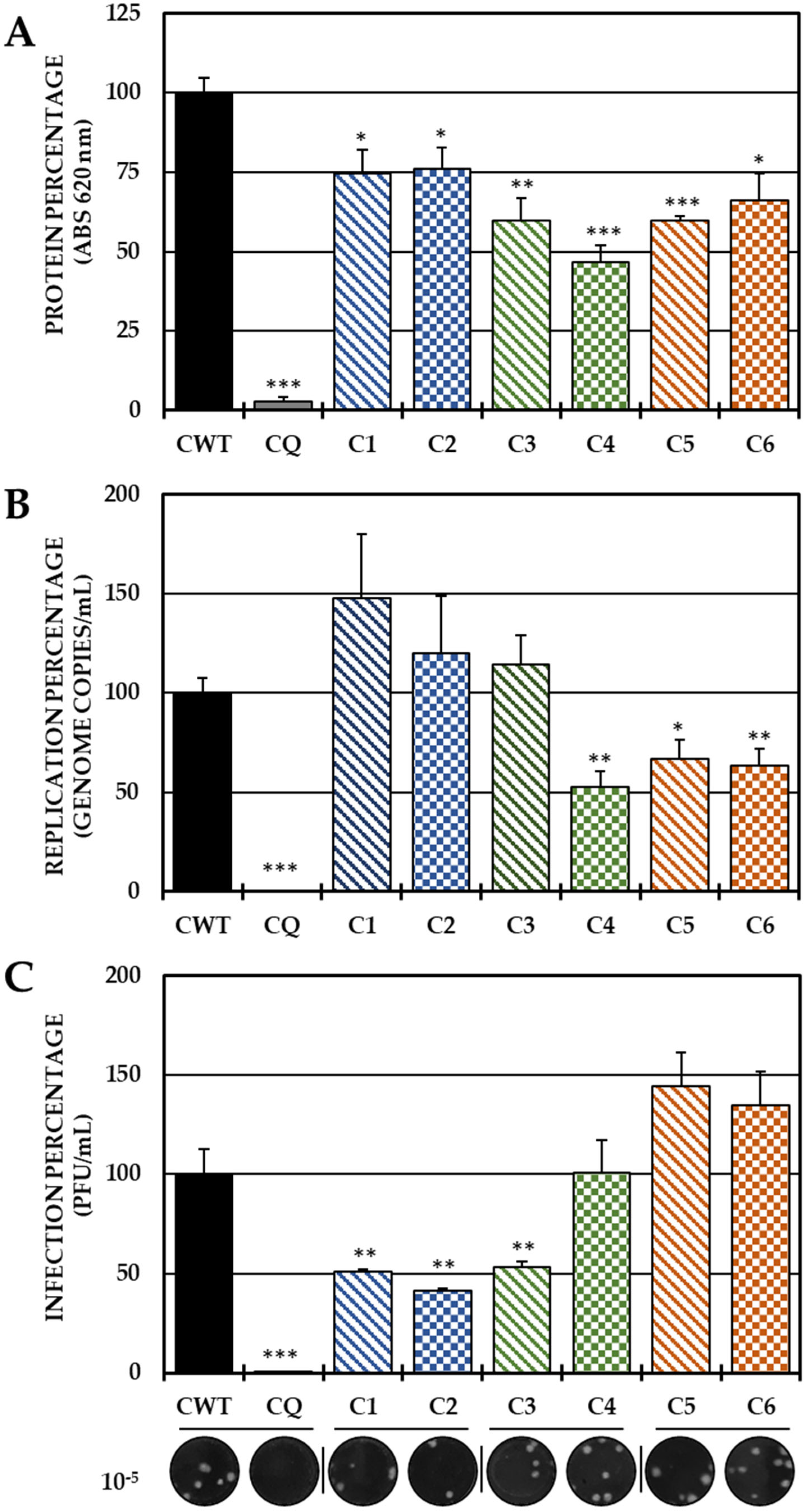
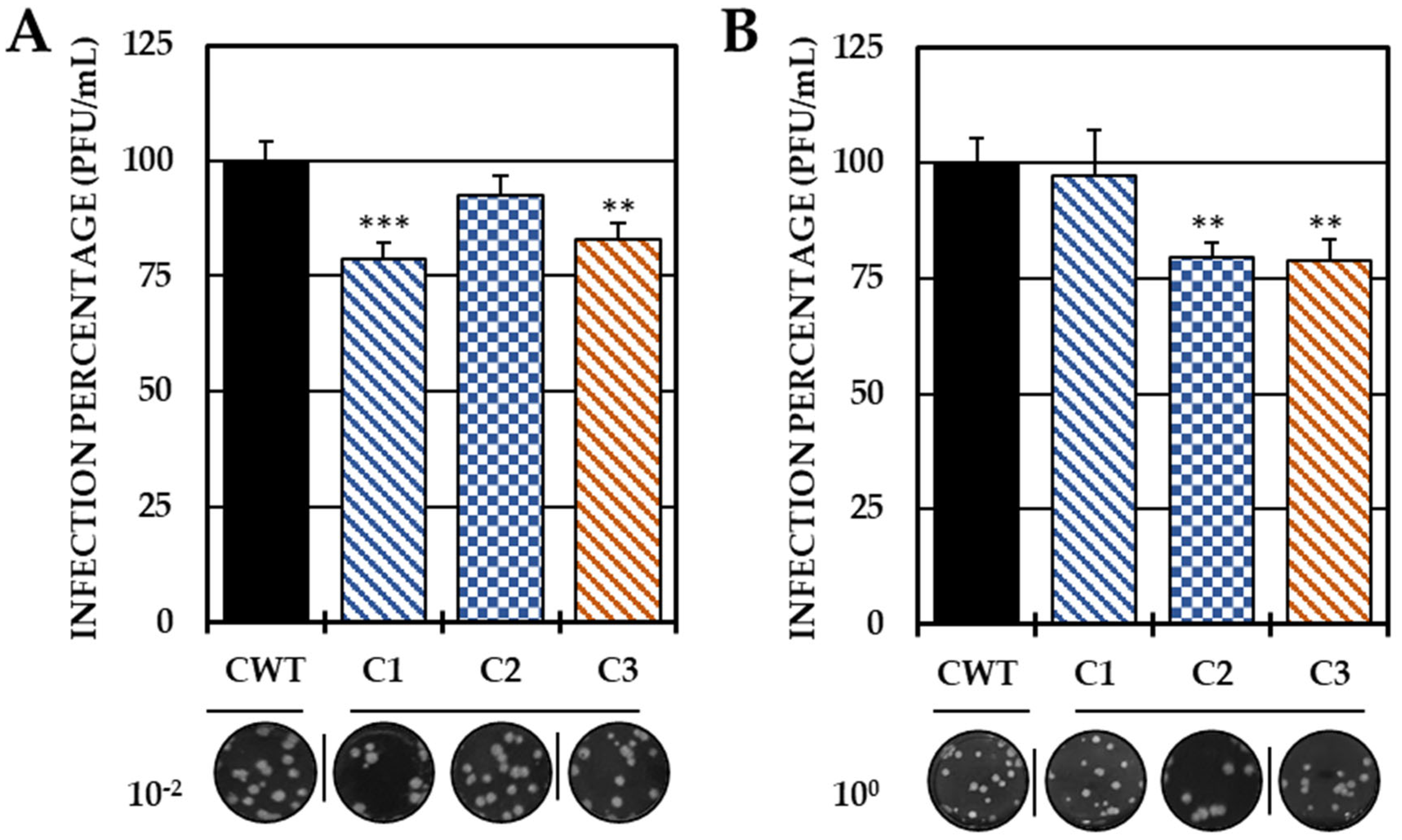
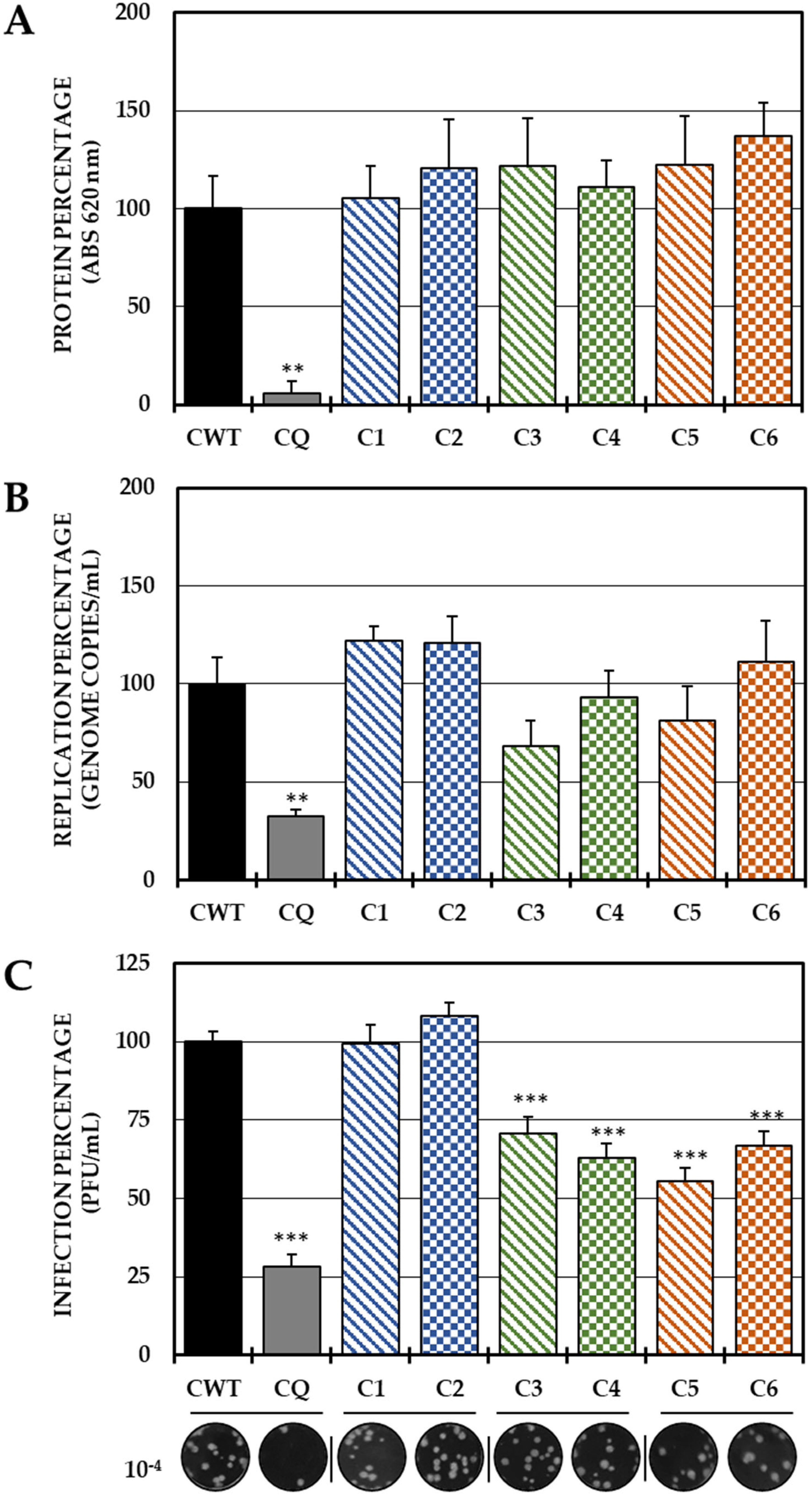
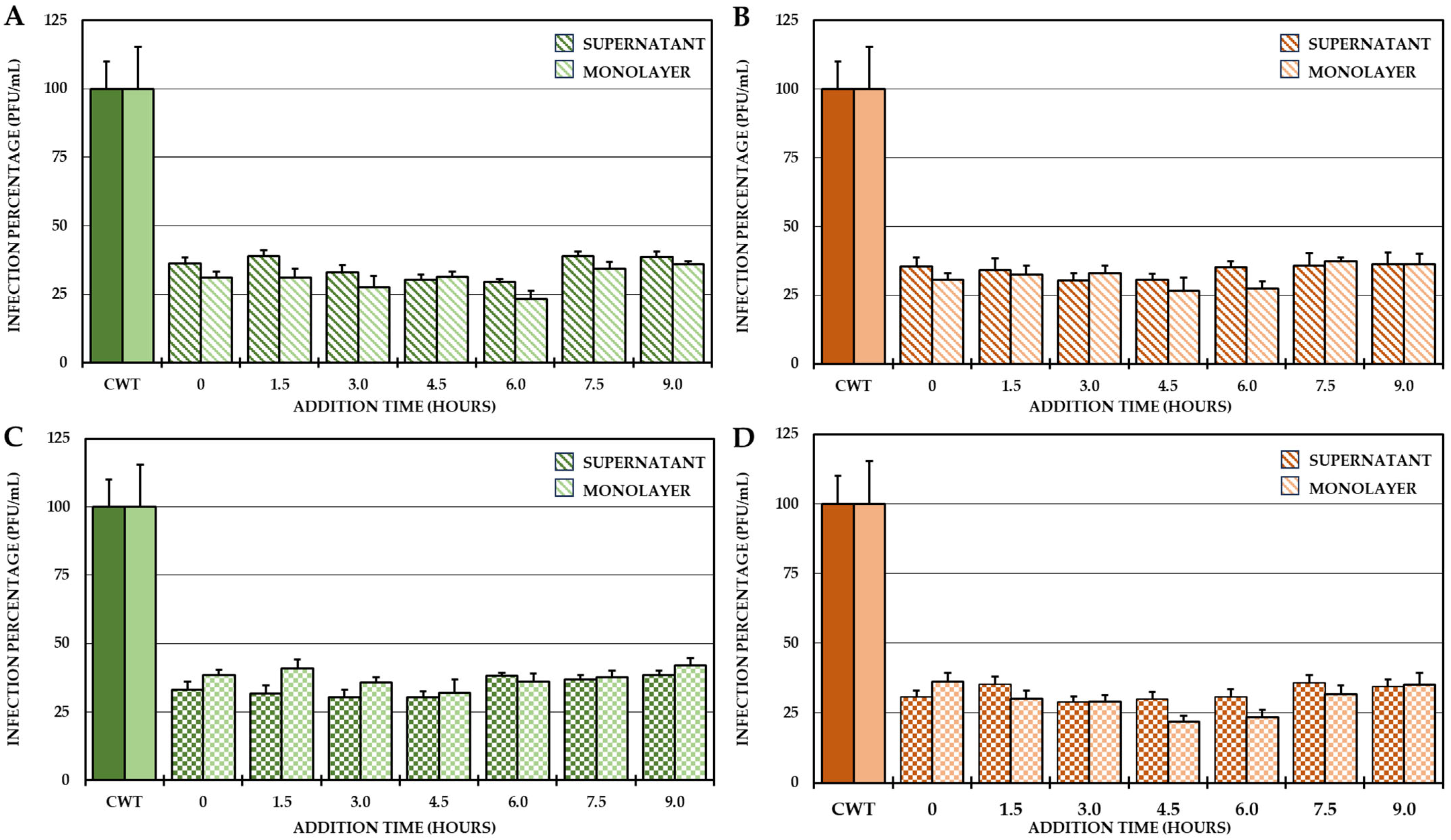
2.2. Evaluation of the Antiviral Mechanism by In Vitro Strategies
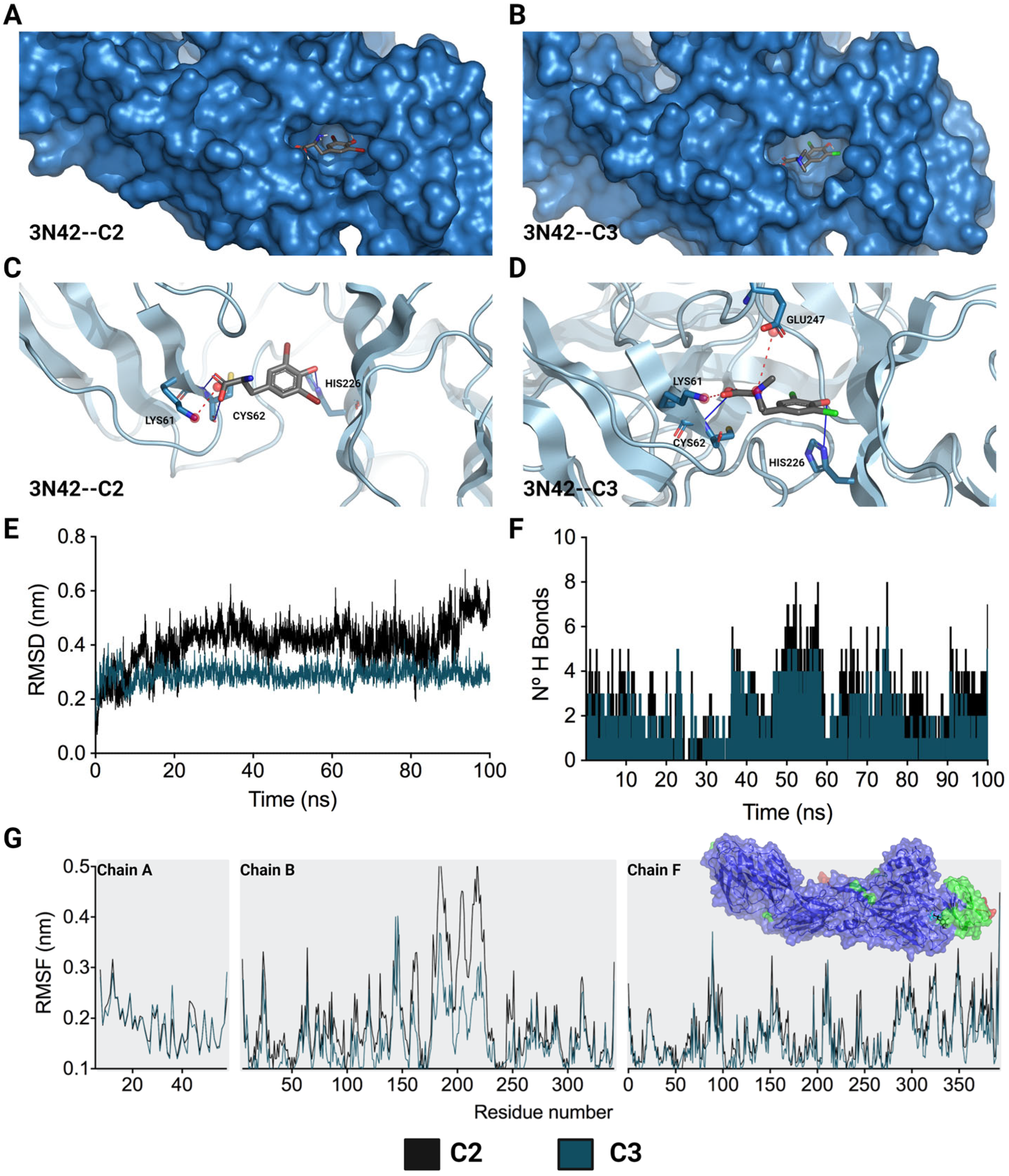
2.3. Evaluation of the Antiviral Mechanism by In Silico Strategies
3. Discussion
4. Materials and Methods
4.1. Cells and Viruses
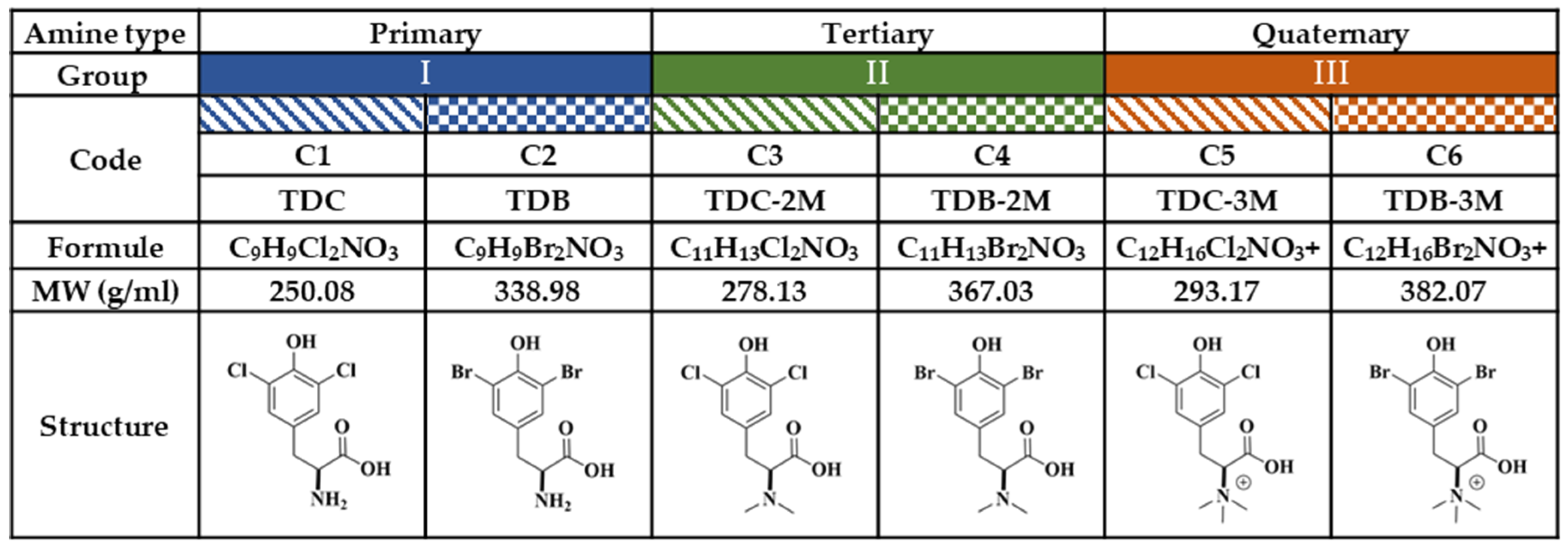
4.2. Compounds
4.3. Physicochemical Properties of the Compounds
4.4. In Vitro Treatment with the Compounds
4.4.1. Adhesion Inhibition Test
4.4.2. Internalization Inhibition Test
4.4.3. Addition Time Test
4.4.4. Quantification of CHIKV Infection
4.4.5. Statistical Analysis
4.5. In Silico Evaluation
4.5.1. Molecular Docking
4.5.2. Molecular Dynamics Simulations
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Morrison, T.E. Reemergence of chikungunya virus. J. Virol. 2014, 88, 11644–11647. [Google Scholar] [CrossRef]
- Ramirez, R.M.G.; Bohers, C.; Mousson, L.; Madec, Y.; Vazeille, M.; Piorkowski, G.; Moutailler, S.; Diaz, F.J.; Rua-Uribe, G.; Villar, L.A.; et al. Increased threat of urban arboviral diseases from Aedes aegypti mosquitoes in Colombia. IJID Reg. 2024, 11, 100360. [Google Scholar] [CrossRef]
- de Souza, W.M.; Fumagalli, M.J.; de Lima, S.T.S.; Parise, P.L.; Carvalho, D.C.M.; Hernandez, C.; de Jesus, R.; Delafiori, J.; Candido, D.S.; Carregari, V.C.; et al. Pathophysiology of chikungunya virus infection associated with fatal outcomes. Cell Host Microbe 2024, 32, 606–622.e8. [Google Scholar] [CrossRef] [PubMed]
- Jones, R.; Kulkarni, M.A.; Davidson, T.M.; Team, R.-L.R.; Talbot, B. Arbovirus vectors of epidemiological concern in the Americas: A scoping review of entomological studies on Zika, dengue and chikungunya virus vectors. PLoS ONE 2020, 15, e0220753. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-López, F.; González-Mazo, A.; Vélez-Mira, A.; Gómez, G.F.; Zuleta, L.; Uribe, S.; Vélez-Bernal, I.D. Presencia de Aedes (Stegomyia) aegypti (Linnaeus, 1762) y su infección natural con el virus del dengue en alturas no registradas para Colombia. Biomédica 2016, 36, 303–308. [Google Scholar] [CrossRef]
- Petersen, L.R.; Powers, A.M. Chikungunya: Epidemiology. F1000Research 2016, 5, 82. [Google Scholar] [CrossRef] [PubMed]
- Schuffenecker, I.; Iteman, I.; Michault, A.; Murri, S.; Frangeul, L.; Vaney, M.-C.; Lavenir, R.; Pardigon, N.; Reynes, J.-M.; Pettinelli, F.J.; et al. Genome microevolution of chikungunya viruses causing the Indian Ocean outbreak. PLoS Med. 2006, 3, e263. [Google Scholar] [CrossRef]
- Powers, A.M.; Logue, C.H. Changing patterns of chikungunya virus: Re-emergence of a zoonotic arbovirus. J. Gen. Virol. 2007, 88, 2363–2377. [Google Scholar] [CrossRef] [PubMed]
- Nunes, M.R.T.; Faria, N.R.; de Vasconcelos, J.M.; Golding, N.; Kraemer, M.U.; de Oliveira, L.F.; Azevedo, R.d.S.d.S.; da Silva, D.E.A.; da Silva, E.V.P.; da Silva, S.P.; et al. Emergence and potential for spread of Chikungunya virus in Brazil. BMC Med. 2015, 13, 102. [Google Scholar] [CrossRef]
- Solignat, M.; Gay, B.; Higgs, S.; Briant, L.; Devaux, C. Replication cycle of chikungunya: A re-emerging arbovirus. Virology 2009, 393, 183–197. [Google Scholar] [CrossRef]
- Ganesan, V.K.; Duan, B.; Reid, S.P. Chikungunya Virus: Pathophysiology, Mechanism, and Modeling. Viruses 2017, 9, 368. [Google Scholar] [CrossRef]
- Schwartz, O.; Albert, M.L. Biology and pathogenesis of chikungunya virus. Nat. Rev. Microbiol. 2010, 8, 491–500. [Google Scholar] [CrossRef] [PubMed]
- de Lima Cavalcanti, T.Y.V.; Pereira, M.R.; de Paula, S.O.; Franca, R.F.d.O. A review on chikungunya virus epidemiology, pathogenesis and current vaccine development. Viruses 2022, 14, 969. [Google Scholar] [CrossRef]
- Pan American Health Organization; Health Information Platform for The Americas (PLISA). Chikungunya Data. Available online: https://www3.paho.org/data/index.php/es/temas/chikv-es.html (accessed on 29 February 2024).
- Zara, A.L.d.S.A.; Santos, S.M.d.; Fernandes-Oliveira, E.S.; Carvalho, R.G.; Coelho, G.E. Aedes aegypti control strategies: A review. Epidemiol. Serviços Saúde 2016, 25, 391–404. [Google Scholar]
- National Library of Medicine; ClinicalTrials.gov. Available online: https://clinicaltrials.gov/ (accessed on 23 February 2024).
- Schneider, M.; Narciso-Abraham, M.; Hadl, S.; McMahon, R.; Toepfer, S.; Fuchs, U.; Hochreiter, R.; Bitzer, A.; Kosulin, K.; Larcher-Senn, J.; et al. Safety and immunogenicity of a single-shot live-attenuated chikungunya vaccine: A double-blind, multicentre, randomised, placebo-controlled, phase 3 trial. Lancet 2023, 401, 2138–2147. [Google Scholar] [CrossRef]
- Newman, D.J.; Cragg, G.M. Natural products as sources of new drugs over the nearly four decades from 01/1981 to 09/2019. J. Nat. Prod. 2020, 83, 770–803. [Google Scholar] [CrossRef] [PubMed]
- Henderson Sousa, F.; Ghaisani Komarudin, A.; Findlay-Greene, F.; Bowolaksono, A.; Sasmono, R.T.; Stevens, C.; Barlow, P.G. Evolution and immunopathology of chikungunya virus informs therapeutic development. Dis. Models Mech. 2023, 16, dmm049804. [Google Scholar] [CrossRef] [PubMed]
- Roques, P.; Thiberville, S.-D.; Dupuis-Maguiraga, L.; Lum, F.-M.; Labadie, K.; Martinon, F.; Gras, G.; Lebon, P.; Ng, L.F.; De Lamballerie, X. Paradoxical effect of chloroquine treatment in enhancing chikungunya virus infection. Viruses 2018, 10, 268. [Google Scholar] [CrossRef]
- National Library of Medicine (US). Oral Ivermectin for Chikungunya Viral Infection (NCT06259383). ClinicalTrials.gov. Available online: https://clinicaltrials.gov/study/NCT06259383 (accessed on 22 February 2024).
- El Sayed, K.A. Natural products as antiviral agents. Stud. Nat. Prod. Chem. 2000, 24, 473–572. [Google Scholar]
- Leyssen, P.; Smadja, J.; Rasoanaivo, P.; Gurib-Fakim, A.; Mahomoodally, M.F.; Canard, B.; Guillemot, J.C.; Litaudon, M.; Guéritte, F. Biodiversity as a source of potent and selective inhibitors of chikungunya virus replication. In Novel Plant Bioresources: Applications in Food, Medicine and Cosmetics; John Wiley & Sons, Ltd.: Chichester, UK, 2014; pp. 151–161. [Google Scholar]
- Monsalve-Escudero, L.M.; Loaiza-Cano, V.; Pájaro-González, Y.; Oliveros-Díaz, A.F.; Diaz-Castillo, F.; Quiñones, W.; Robledo, S.; Martinez-Gutierrez, M. Indole alkaloids inhibit zika and chikungunya virus infection in different cell lines. BMC Complement. Med. Ther. 2021, 21, 216. [Google Scholar] [CrossRef]
- Gómez-Calderón, C.; Mesa-Castro, C.; Robledo, S.; Gómez, S.; Bolivar-Avila, S.; Diaz-Castillo, F.; Martínez-Gutierrez, M. Antiviral effect of compounds derived from the seeds of Mammea americana and Tabernaemontana cymosa on Dengue and Chikungunya virus infections. BMC Complement. Altern. Med. 2017, 17, 57. [Google Scholar] [CrossRef] [PubMed]
- Galeano, E.; Martínez, A.; Thomas, O.P.; Robledo, S.; Munoz, D. Antiparasitic bromotyrosine derivatives from the Caribbean marine sponge Aiolochroia crassa. Quim. Nova 2012, 35, 1189–1193. [Google Scholar] [CrossRef]
- Rangel, S.Y.G.; Sánchez, K.; Lemus, A.; Chaustre, M.; Galeano, E.; Martínez-Gutiérrez, M. Estudio del efecto inhibitorio de halo-tirosinas sobre la replicación in vitro del Virus Dengue. Rev. Fac. Cienc. Salud UDES 2016, 3, 21. [Google Scholar] [CrossRef]
- Gómez Archila, L.G.; Zapata, W.; Galeano, E.; Martínez, A.; Díaz, F.J.; Rugeles, M.T. Bromotyrosine derivatives from marine sponges inhibit the HIV-1 replication in vitro. Vitae 2014, 21, 114–125. [Google Scholar] [CrossRef]
- Pastrana Restrepo, M.; Galeano Jaramillo, E.; Martínez Martínez, A.; Robledo Restrepo, S. Synthesis and trypanocide activity of chloro-l-tyrosine and bromo-l-tyrosine derivatives. Med. Chem. Res. 2018, 27, 2454–2465. [Google Scholar] [CrossRef]
- Chass, G.A.; Lovas, S.; Murphy, R.; Csizmadia, I. The role of enhanced aromatic-electron donating aptitude of the tyrosyl sidechain with respect to that of phenylalanyl in intramolecular interactions. Eur. Phys. J. D-At. Mol. Opt. Plasma Phys. 2002, 20, 481–497. [Google Scholar] [CrossRef]
- Serna-Arbeláez, M.S.; García-Cárcamo, V.; Rincón-Tabares, D.S.; Guerra, D.; Loaiza-Cano, V.; Martinez-Gutierrez, M.; Pereañez, J.A.; Pastrana-Restrepo, M.; Galeano, E.; Zapata, W. In Vitro and In Silico Antiviral Activity of Di-Halogenated Compounds Derived from L-Tyrosine against Human Immunodeficiency Virus 1 (HIV-1). Curr. Issues Mol. Biol. 2023, 45, 8173–8200. [Google Scholar] [CrossRef]
- Loaiza-Cano, V.; Monsalve-Escudero, L.M.; Restrepo, M.P.; Quintero-Gil, D.C.; Pulido Muñoz, S.A.; Galeano, E.; Zapata, W.; Martinez-Gutierrez, M. In vitro and in silico anti-Arboviral activities of Dihalogenated phenolic Derivates of L-tyrosine. Molecules 2021, 26, 3430. [Google Scholar] [CrossRef]
- Sobolev, O.V.; Afonine, P.V.; Moriarty, N.W.; Hekkelman, M.L.; Joosten, R.P.; Perrakis, A.; Adams, P.D. A global Ramachandran score identifies protein structures with unlikely stereochemistry. Structure 2020, 28, 1249–1258.e2. [Google Scholar] [CrossRef]
- Staples, J.E.; Breiman, R.F.; Powers, A.M. Chikungunya fever: An epidemiological review of a re-emerging infectious disease. Clin. Infect. Dis. 2009, 49, 942–948. [Google Scholar] [CrossRef]
- Campos, D.; Navarro, S.; Llamas-González, Y.Y.; Sugasti, M.; González-Santamaría, J. Broad antiviral activity of ginkgolic acid against chikungunya, Mayaro, una, and Zika viruses. Viruses 2020, 12, 449. [Google Scholar] [CrossRef]
- Ferraz, A.C.; Moraes, T.d.F.S.; da Cruz Nizer, W.S.; Dos Santos, M.; Tótola, A.H.; Ferreira, J.M.S.; Vieira-Filho, S.A.; Rodrigues, V.G.; Duarte, L.P.; de Brito Magalhaes, C.L. Virucidal activity of proanthocyanidin against Mayaro virus. Antivir. Res. 2019, 168, 76–81. [Google Scholar] [CrossRef] [PubMed]
- Lani, R.; Hassandarvish, P.; Shu, M.-H.; Phoon, W.H.; Chu, J.J.H.; Higgs, S.; Vanlandingham, D.; Bakar, S.A.; Zandi, K. Antiviral activity of selected flavonoids against Chikungunya virus. Antivir. Res. 2016, 133, 50–61. [Google Scholar] [CrossRef]
- Oo, A.; Rausalu, K.; Merits, A.; Higgs, S.; Vanlandingham, D.; Bakar, S.A.; Zandi, K. Deciphering the potential of baicalin as an antiviral agent for Chikungunya virus infection. Antivir. Res. 2018, 150, 101–111. [Google Scholar] [CrossRef]
- Loaiza-Cano, V.; Monsalve-Escudero, L.M.; Filho, C.d.S.M.B.; Martinez-Gutierrez, M.; Sousa, D.P.d. Antiviral role of phenolic compounds against dengue virus: A review. Biomolecules 2020, 11, 11. [Google Scholar] [CrossRef]
- Putri, G.N.; Gudla, C.S.; Singh, M.; Ng, C.H.; Idris, F.F.H.; Oo, Y.; Tan, J.H.Y.; Wong, J.F.J.; Chu, J.J.H.; Selvam, V. Expanding the anti-flaviviral arsenal: Discovery of a baicalein-derived Compound with potent activity against DENV and ZIKV. Antivir. Res. 2023, 220, 105739. [Google Scholar] [CrossRef]
- van Duijl-Richter, M.K.; Hoornweg, T.E.; Rodenhuis-Zybert, I.A.; Smit, J.M. Early events in chikungunya virus infection—From virus cell binding to membrane fusion. Viruses 2015, 7, 3647–3674. [Google Scholar] [CrossRef] [PubMed]
- Mendez, L.; Henriquez, G.; Sirimulla, S.; Narayan, M. Looking Back, Looking Forward at Halogen Bonding in Drug Discovery. Molecules 2017, 22, 1397. [Google Scholar] [CrossRef] [PubMed]
- Lim, J.Y.C.; Beer, P.D. Sigma-Hole Interactions in Anion Recognition. Chem 2018, 4, 731–783. [Google Scholar] [CrossRef]
- García-Ariza, L.L.; Rocha-Roa, C.; Padilla-Sanabria, L.; Castaño-Osorio, J.C. Virtual screening of drug-like compounds as potential inhibitors of the dengue virus NS5 protein. Front. Chem. 2022, 10, 637266. [Google Scholar] [CrossRef]
- Chang, M.W.; Lindstrom, W.; Olson, A.J.; Belew, R.K. Analysis of HIV wild-type and mutant structures via in silico docking against diverse ligand libraries. J. Chem. Inf. Model. 2007, 47, 1258–1262. [Google Scholar] [CrossRef] [PubMed]
- Monsalve-Escudero, L.M.; Loaiza-Cano, V.; Zapata-Cardona, M.I.; Quintero-Gil, D.C.; Hernández-Mira, E.; Pájaro-González, Y.; Oliveros-Díaz, A.F.; Diaz-Castillo, F.; Quiñones, W.; Robledo, S.; et al. The antiviral and virucidal activities of voacangine and structural analogs extracted from Tabernaemontana cymosa depend on the Dengue virus strain. Plants 2021, 10, 1280. [Google Scholar] [CrossRef] [PubMed]
- Valdés-López, J.F.; Fernandez, G.J.; Urcuqui-Inchima, S. Interleukin 27 as an inducer of antiviral response against chikungunya virus infection in human macrophages. Cell. Immunol. 2021, 367, 104411. [Google Scholar] [CrossRef]
- Chain, M.M.; Doane, F.W.; McLean, D. Morphological development of Chikungunya virus. Can. J. Microbiol. 1966, 12, 895–900. [Google Scholar] [CrossRef] [PubMed]
- Reis, E.V.S.; Damas, B.M.; Mendonca, D.C.; Abrahao, J.S.; Bonjardim, C.A. In-Depth Characterization of the Chikungunya Virus Replication Cycle. J. Virol. 2022, 96, e0173221. [Google Scholar] [CrossRef] [PubMed]
- Daina, A.; Michielin, O.; Zoete, V. SwissADME: A free web tool to evaluate pharmacokinetics, drug-likeness and medicinal chemistry friendliness of small molecules. Sci. Rep. 2017, 7, 42717. [Google Scholar] [CrossRef] [PubMed]
- Pardo-Rodriguez, D.; Cifuentes-López, A.; Bravo-Espejo, J.; Romero, I.; Robles, J.; Cuervo, C.; Mejía, S.M.; Tellez, J. Lupeol Acetate and α-Amyrin Terpenes Activity against Trypanosoma cruzi: Insights into Toxicity and Potential Mechanisms of Action. Trop. Med. Infect. Dis. 2023, 8, 263. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Gutierrez, M.; Castellanos, J.E.; Gallego-Gómez, J.C. Statins reduce dengue virus production via decreased virion assembly. Intervirology 2011, 54, 202–216. [Google Scholar] [CrossRef]
- Voss, J.E.; Vaney, M.-C.; Duquerroy, S.; Vonrhein, C.; Girard-Blanc, C.; Crublet, E.; Thompson, A.; Bricogne, G.; Rey, F.A. Glycoprotein organization of Chikungunya virus particles revealed by X-ray crystallography. Nature 2010, 468, 709–712. [Google Scholar] [CrossRef]
- Trott, O.; Olson, A.J. AutoDock Vina: Improving the speed and accuracy of docking with a new scoring function, efficient optimization, and multithreading. J. Comput. Chem. 2010, 31, 455–461. [Google Scholar] [CrossRef]
- Salentin, S.; Schreiber, S.; Haupt, V.J.; Adasme, M.F.; Schroeder, M. PLIP: Fully automated protein–ligand interaction profiler. Nucleic Acids Res. 2015, 43, W443–W447. [Google Scholar] [CrossRef] [PubMed]
- Zoete, V.; Cuendet, M.A.; Grosdidier, A.; Michielin, O. SwissParam: A fast force field generation tool for small organic molecules. J. Comput. Chem. 2011, 32, 2359–2368. [Google Scholar] [CrossRef] [PubMed]
- Van Der Spoel, D.; Lindahl, E.; Hess, B.; Groenhof, G.; Mark, A.E.; Berendsen, H.J. GROMACS: Fast, flexible, and free. J. Comput. Chem. 2005, 26, 1701–1718. [Google Scholar] [CrossRef] [PubMed]
- Jorgensen, W.L.; Chandrasekhar, J.; Madura, J.D.; Impey, R.W.; Klein, M.L. Comparison of simple potential functions for simulating liquid water. J. Chem. Phys. 1983, 79, 926–935. [Google Scholar] [CrossRef]
- Joung, I.S.; Cheatham, T.E., III. Molecular dynamics simulations of the dynamic and energetic properties of alkali and halide ions using water-model-specific ion parameters. J. Phys. Chem. B 2009, 113, 13279–13290. [Google Scholar] [CrossRef] [PubMed]
- Hess, B. P-LINCS: A parallel linear constraint solver for molecular simulation. J. Chem. Theory Comput. 2008, 4, 116–122. [Google Scholar] [CrossRef] [PubMed]
- Bussi, G.; Donadio, D.; Parrinello, M. Canonical sampling through velocity rescaling. J. Chem. Phys. 2007, 126, 014101. [Google Scholar] [CrossRef]
- Humphrey, W.; Dalke, A.; Schulten, K. VMD: Visual molecular dynamics. J. Mol. Graph. 1996, 14, 33–38. [Google Scholar] [CrossRef]
- Kumar, R.; Maurya, R.; Saran, S. Introducing a simple model system for binding studies of known and novel inhibitors of AMPK: A therapeutic target for prostate cancer. J. Biomol. Struct. Dyn. 2019, 37, 781–795. [Google Scholar] [CrossRef]

| TPSA | HBA | HBD | #Rot | Log P | ESOL | Medicinal Chemistry | Pharmacokinetics | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| (Å2) | Log S | Class | SA | LV | PAINS | Brenk | GIA | BBB | CYPi | P-gps | ||||||
| I | C1 | 83.55 | 4 | 3 | 3 | 0.74 | −1.17 | V-S | 1.96 | 0 | 0 | 0 | High | No | No | No |
| C2 | 83.55 | 4 | 3 | 3 | 0.91 | −1.80 | V-S | 2.13 | 0 | 0 | 0 | High | No | No | No | |
| II | C3 | 60.77 | 4 | 2 | 4 | 1.84 | −1.86 | V-S | 2.15 | 0 | 0 | 0 | High | Yes | No | No |
| C4 | 60.77 | 4 | 2 | 4 | 1.96 | −2.49 | S | 2.31 | 0 | 0 | 0 | High | Yes | No | No | |
| III | C5 | 57.53 | 3 | 2 | 4 | 0.37 | −3.39 | S | 2.29 | 0 | 0 | 1 | High | Yes | No | Yes |
| C6 | 57.53 | 3 | 2 | 4 | 0.58 | −4.01 | Md-S | 2.44 | 0 | 0 | 1 | High | Yes | No | Yes | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Loaiza-Cano, V.; Hernández-Mira, E.; Pastrana-Restrepo, M.; Galeano, E.; Pardo-Rodriguez, D.; Martinez-Gutierrez, M. The Mechanism of Action of L-Tyrosine Derivatives against Chikungunya Virus Infection In Vitro Depends on Structural Changes. Int. J. Mol. Sci. 2024, 25, 7972. https://doi.org/10.3390/ijms25147972
Loaiza-Cano V, Hernández-Mira E, Pastrana-Restrepo M, Galeano E, Pardo-Rodriguez D, Martinez-Gutierrez M. The Mechanism of Action of L-Tyrosine Derivatives against Chikungunya Virus Infection In Vitro Depends on Structural Changes. International Journal of Molecular Sciences. 2024; 25(14):7972. https://doi.org/10.3390/ijms25147972
Chicago/Turabian StyleLoaiza-Cano, Vanessa, Estiven Hernández-Mira, Manuel Pastrana-Restrepo, Elkin Galeano, Daniel Pardo-Rodriguez, and Marlen Martinez-Gutierrez. 2024. "The Mechanism of Action of L-Tyrosine Derivatives against Chikungunya Virus Infection In Vitro Depends on Structural Changes" International Journal of Molecular Sciences 25, no. 14: 7972. https://doi.org/10.3390/ijms25147972
APA StyleLoaiza-Cano, V., Hernández-Mira, E., Pastrana-Restrepo, M., Galeano, E., Pardo-Rodriguez, D., & Martinez-Gutierrez, M. (2024). The Mechanism of Action of L-Tyrosine Derivatives against Chikungunya Virus Infection In Vitro Depends on Structural Changes. International Journal of Molecular Sciences, 25(14), 7972. https://doi.org/10.3390/ijms25147972





