Epigenetic Regulation of the Renin–Angiotensin–Aldosterone System in Hypertension
Abstract
:1. Introduction
2. Epigenetic Regulation of Gene Expression
2.1. DNA Methylation
2.2. Histone Modifications
2.3. Micro RNAs (miRNAs)
3. Epigenetic Regulation of the AGT Gene
3.1. Salt-Sensitive Hypertension (SSH)
3.2. Primary Aldosteronism
4. Epigenetic Regulation of the Angiotensin-Converting Enzyme (ACE)
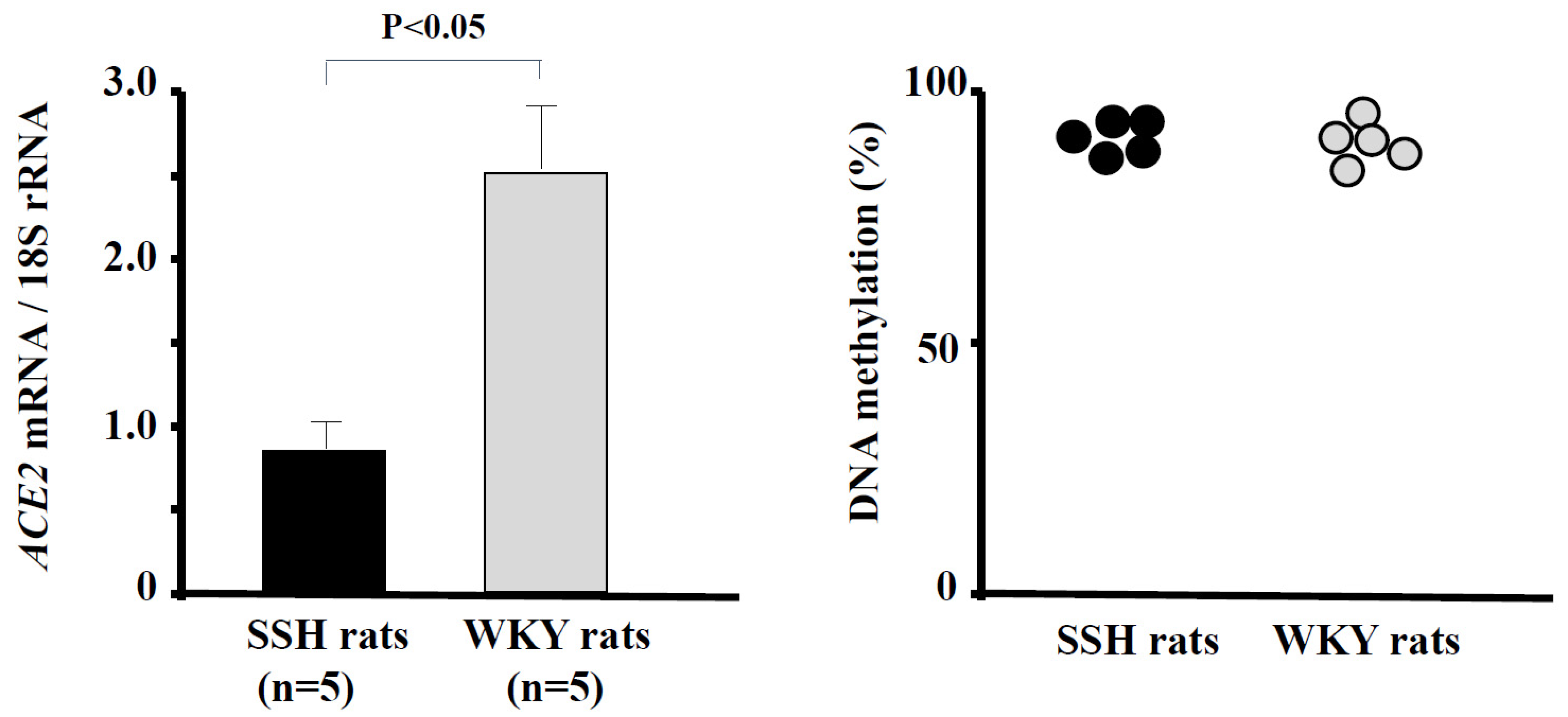
5. Epigenetic Regulation of ACE2
6. Epigenetic Regulation of AT1R
7. Epigenetic Regulation of CYP11B2
7.1. Epigenetics and Aldosterone-Producing Adenoma (APA)
7.2. Epigenetic Regulation of Mineralocorticoid-Related Genes in SSH
7.3. Epigenetic Control of Mineralocorticoid Receptors
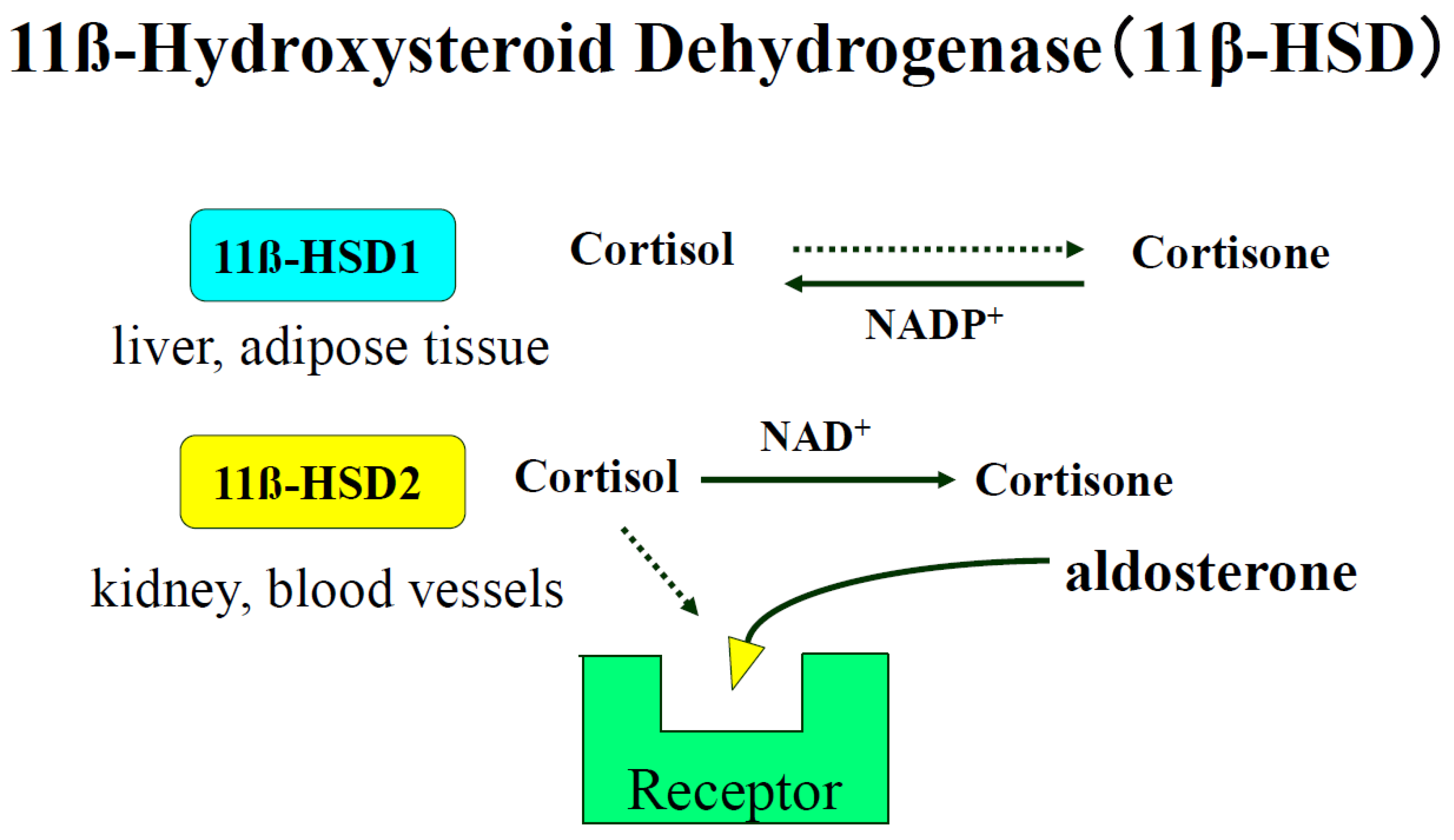
7.4. Epigenetic Control of 11ß-Hydroxysteroid Dehydrogenase Type 2
8. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| ACE | angiotensin-converting enzyme |
| AGT | angiotensinogen |
| APA | aldosterone-producing adenoma |
| AT1R | angiotensin II type 1 receptor |
| CEBP | CCAAT enhancer-binding protein |
| DNA | deoxyribonucleic acid |
| HATs | histone acetyl transferases |
| HCM | hypertrophic cardiomyopathy |
| HDACs | histone deacetylases |
| mRNA | messenger ribonucleic acid |
| miRNA | microRNA |
| MBD | methyl-CpG-binding domain |
| MR | mineralocorticoid receptor |
| MRA | mineralocorticoid receptor antagonist |
| PA | primary aldosteronism |
| RAAS | renin–angiotensin–aldosterone system |
| RNA | ribonucleic acid |
| SHR | spontaneously hypertensive rat |
| SSH | salt-sensitive hypertension |
| TFs | transcription factors |
| UTR | untranslated region |
References
- Paz Ocaranza, M.; Riquelme, J.A.; García, L.; Jalil, J.E.; Chiong, M.; Santos, R.A.S.; Lavandero, S. Counter-regulatory renin-angiotensin system in cardiovascular disease. Nat. Rev. Cardiol. 2020, 17, 116–129. [Google Scholar] [CrossRef] [PubMed]
- Azushima, K.; Morisawa, N.; Tamura, K.; Nishiyama, A. Recent research advances in renin-angiotensin-aldosterone system receptors. Curr. Hypertens. Rep. 2020, 22, 22. [Google Scholar] [CrossRef] [PubMed]
- Ames, M.K.; Atkins, C.E.; Pitt, B.J. The renin-angiotensin-aldosterone system and its suppression. Vet. Intern. Med. 2019, 33, 363–382. [Google Scholar] [CrossRef] [PubMed]
- Cruz-López, E.O.; Ye, D.; Wu, C.; Lu, H.S.; Uijl, E.; Mirabito Colafella, K.M.; Danser, A.H.J. Angiotensinogen suppression: A new tool to treat cardiovascular and renal disease. Hypertension 2022, 79, 2115–2126. [Google Scholar] [CrossRef] [PubMed]
- Yasue, S.; Masuzaki, H.; Okada, S.; Ishii, T.; Kozuka, C.; Tanaka, T.; Fujikura, J.; Ebihara, K.; Hosoda, K.; Katsurada, A.; et al. Adipose tissue-specific regulation of angiotensinogen in obese humans and mice: Impact of nutritional status and adipocyte hypertrophy. Am. J. Hypertens. 2010, 23, 425–431. [Google Scholar] [CrossRef] [PubMed]
- Cruz-López, E.O.; Uijl, E.; Danser, A.H.J. Perivascular adipose tissue in vascular function: Does locally synthesized angiotensinogen play a role? J. Cardiovasc. Pharmacol. 2021, 78 (Suppl. S6), S53–S62. [Google Scholar] [CrossRef]
- Khurana, V.; Goswami, B. Angiotensin converting enzyme (ACE). Clin. Chim. Acta 2022, 524, 113–122. [Google Scholar] [CrossRef]
- Le, D.; Brown, L.; Malik, K.; Murakami, S. Two opposing functions of angiotensin-converting enzyme (ACE) that links hypertension, dementia, and aging. Int. J. Mol. Sci. 2021, 22, 13178. [Google Scholar] [CrossRef] [PubMed]
- Clark, C.R.; Khalil, R.A. Regulation of vascular angiotensin II type 1 and type 2 receptor and angiotensin-(1–7)/MasR signaling in normal and hypertensive pregnancy. Biochem. Pharmacol. 2024, 220, 115963. [Google Scholar] [CrossRef]
- Bierstadt, S.; Casaro, E.B.; Range, É.B. COVID-19: Angiotensin-converting enzyme 2 (ACE2) expression and tissue susceptibility to SARS-CoV-2 infection. Eur. J. Clin. Microbiol. Infect. Dis. 2021, 40, 905–919. [Google Scholar] [CrossRef]
- Lawal, I.O.; Kgatle, M.M.; Mokoala, K.; Farate, A.; Sathekge, M.M. Cardiovascular disturbances in COVID-19: An updated review of the pathophysiology and clinical evidence of cardiovascular damage induced by SARS-CoV-2. BMC Cardiovasc. Disord. 2022, 22, 93. [Google Scholar] [CrossRef]
- Agarwal, R.; Kolkhof, P.; Bakris, G.; Bauersachs, J.; Haller, H.; Wada, T.; Zannad, F. Steroidal and non-steroidal mineralocorticoid receptor antagonists in cardiorenal medicine. Eur. Heart J. 2021, 42, 152–161. [Google Scholar] [CrossRef]
- Barrera-Chimal, J.; Lima-Posada, I.; Bakris, G.L.; Jaisser, F. Mineralocorticoid receptor antagonists in diabetic kidney diseasemechanistic and therapeutic effects. Nat. Rev. Nephrol. 2022, 18, 56–70. [Google Scholar] [CrossRef]
- Tsukamoto, S.; Uehara, T.; Azushima, K.; Wakui, H.; Tamura, K. Updates for cardio-kidney protective effects by angiotensin receptor-neprilysin inhibitor: Requirement for additional evidence of kidney protection. J. Am. Heart Assoc. 2023, 12, e029565. [Google Scholar] [CrossRef]
- Parksook, W.W.; Williamn, G.H. Aldosterone and cardiovascular diseases. Cardiovasc. Res. 2023, 119, 28–44. [Google Scholar] [CrossRef]
- Ohno, Y.; Sone, M.; Inagaki, N.; Yamasaki, T.; Ogawa, O.; Takeda, Y.; Kurihara, I.; Itoh, H.; Umakoshi, H.; Tsuiki, M.; et al. Prevalence of cardiovascular disease and its risk factors in primary aldosteronism: A multicenter study in Japan. Hypertension 2018, 71, 530–537. [Google Scholar] [CrossRef]
- Zennaro, M.C.; Boulkroun, S.; Fernandes-Rosa, F.L. Pathogenesis and treatment of primary aldosteronism. Nat. Rev. Endocrinol. 2020, 16, 578–589. [Google Scholar] [CrossRef]
- Takeda, Y.; Zhu, A.; Yoneda, T.; Usukura, M.; Takata, H.; Yamagishi, M. Effects of aldosterone and angiotensin II receptor blockade on cardiac angiotensinogen and angiotensin-converting enzyme 2 expression in Dahl salt-sensitive hypertensive rats. Am. J. Hypertens. 2007, 20, 1119–1124. [Google Scholar] [CrossRef]
- Ferreira, N.S.; Tostes, R.C.; Paradis, P.; Schiffrin, E.L. Aldosterone, inflammation, immune system, and hypertension. Am. J. Hypertens. 2021, 34, 15–27. [Google Scholar] [CrossRef]
- Dzau, V.J. Multiple pathways of angiotensin production in the blood vessel wall: Evidence, possibilities and hypotheses. J. Hypertens. 1989, 7, 933–936. [Google Scholar] [CrossRef]
- Briones, A.M.; Nguyen Dinh Cat, A.; Callera, G.E.; Yogi, A.; Burger, D.; He, Y.; Corrêa, J.W.; Gagnon, A.M.; Gomez-Sanchez, C.E.; Gomez-Sanchez, E.P.; et al. Adipocytes produce aldosterone through calcineurin-dependent signaling pathways: Implications in diabetes mellitus-associated obesity and vascular dysfunction. Hypertension 2012, 59, 1069–1078. [Google Scholar] [CrossRef]
- Takeda, Y. Role of cardiovascular aldosterone in hypertension. Curr. Med. Chem. Cardiovasc. Hematol. Agents 2005, 3, 261–266. [Google Scholar] [CrossRef]
- Xu, C. Extra-adrenal aldosterone: A mini review focusing on the physiology and pathophysiology of intrarenal aldosterone. Endocrine 2023, 83, 285–301. [Google Scholar] [CrossRef]
- Jeong, S.; Kim, J.Y.; Cho, Y.; Koh, S.B.; Kim, N.; Choi, J.R. Genetically, dietary sodium intake is causally associated with salt-sensitive hypertension risk in a community-based cohort study: A mendelian randomization approach. Curr. Hypertens. Rep. 2020, 22, 45. [Google Scholar] [CrossRef] [PubMed]
- Morimoto, A.; Uzu, T.; Fujii, T.; Nishimura, M.; Kuroda, S.; Nakamura, S.; Inenaga, T.; Kimura, G. Sodium sensitivity and cardiovascular events in patients with essential hypertension. Lancet 1997, 350, 1734–1737. [Google Scholar] [CrossRef]
- Weinberger, M.H.; Fineberg, N.S.; Fineberg, S.E.; Weinberger, M. Salt sensitivity, pulse pressure, and death in normal and hypertensive humans. Hypertension 2001, 37, 429–432. [Google Scholar] [CrossRef]
- Stoll, S.; Wang, C.; Qiu, H. DNA methylation and histone modification in hypertension. Int. J. Mol. Sci. 2018, 19, 1174. [Google Scholar] [CrossRef]
- Demura, M.; Demura, Y.; Takeda, Y.; Saijoh, K. Dynamic regulation of the angiotensinogen gene by DNA methylation, which is influenced by various stimuli experienced in daily life. Hypertens. Res. 2015, 38, 519–527. [Google Scholar] [CrossRef]
- Klimczak-Tomaniak, D.; Haponiuk-Skwarlińska, J.; Kuch, M.; Pączek, L. Crosstalk between microRNA and oxidative stress in heart failure: A systematic review. Int. J. Mol. Sci. 2022, 23, 15013. [Google Scholar] [CrossRef]
- Wang, F.; Demura, M.; Cheng, Y.; Zhu, A.; Karashima, S.; Yoneda, T.; Demura, Y.; Maeda, Y.; Namiki, M.; Ono, K.; et al. Dynamic CCAAT/enhancer binding protein-associated changes of DNA methylation in the angiotensinogen gene. Hypertension 2014, 63, 281–288. [Google Scholar] [CrossRef]
- Guarner-Lans, V.; Ramírez-Higuera, A.; Rubio-Ruiz, M.E.; Castrejón-Téllez, V.; Soto, M.E.; Pérez-Torres, I. Early programming of adult systemic essential hypertension. Int. J. Mol. Sci. 2020, 21, 1203. [Google Scholar] [CrossRef] [PubMed]
- Field, A.; Adelman, K. Evaluating enhancer function and transcription. Annu. Rev. Biochem. 2020, 89, 213–234. [Google Scholar] [CrossRef]
- Huang, Y.; Ting, P.Y.; Yao, T.M.; Homma, T.; Brooks, D.; Rangel, I.K.; Adler, G.K.; Romero, J.R.; Williams, J.S.; Pojoga, L.H.; et al. Histone demethylase LSD1 deficiency and biological sex: Impact on blood pressure and aldosterone production. J. Endocrinol. 2019, 240, 111–122. [Google Scholar] [CrossRef] [PubMed]
- Shah, A.M.; Giacca, M. Small non-coding RNA therapeutics for cardiovascular disease. Eur. Heart J. 2022, 43, 4548–4561. [Google Scholar] [CrossRef]
- Mahtal, N.; Lenoir, O.; Tinel, C.; Anglicheau, D.; Tharaux, P.L. MicroRNAs in kidney injury and disease. Nat. Rev. Nephrol. 2022, 18, 643–662. [Google Scholar] [CrossRef]
- Sharma, N.M.; Nandi, S.S.; Zheng, H.; Mishra, P.K.; Patel, K.P. A novel role for miR-133a in centrally mediated activation of the renin-angiotensin system in congestive heart failure. Am. J. Physiol. Heart Circ. Physiol. 2017, 312, H968–H979. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Han, X.; Zhao, T.; Zhang, X.; Qu, P.; Zhao, H. AGT, targeted by miR-149-5p, promotes IL-6-induced inflammatory responses of chondrocytes in osteoarthritis via activating JAK2/STAT3 pathway. Clin. Exp. Rheumatol. 2020, 38, 1088–1095. [Google Scholar]
- Liu, T.T.; Hao, Q.; Zhang, Y.; Li, Z.H.; Cui, Z.H.; Yang, W. Effects of microRNA-133b on retinal vascular endothelial cell proliferation and apoptosis through angiotensinogen-mediated angiotensin II- extracellular signal-regulated kinase 1/2 signalling pathway in rats with diabetic retinopathy. Acta Ophthalmol. 2018, 96, e626–e635. [Google Scholar] [CrossRef]
- Chen, X.K.; Ouyang, L.J.; Yin, Z.Q.; Xia, Y.Y.; Chen, X.R.; Shi, H.; Xiong, Y.; Pi, L.H. Effects of microRNA-29a on retinopathy of prematurity by targeting AGT in a mouse model. Am. J. Transl. Res. 2017, 9, 791–801. [Google Scholar]
- Novák, J.; Maceková, S.; Héžová, R.; Máchal, J.; Zlámal, F.; Hlinomaz, O.; Rezek, M.; Souček, M.; Vašků, A.; Slabý, O.; et al. Polymorphism rs7079 in miR-31/-584 binding site in angiotensinogen gene associates with earlier onset of coronary artery disease in Central European population. Genes 2022, 13, 1981. [Google Scholar] [CrossRef]
- Takeda, Y.; Demura, M.; Yoneda, T.; Takeda, Y. DNA methylation of the angiotensinogen gene, agt, and the aldosterone synthase gene, CYP11B2 in cardiovascular diseases. Int. J. Mol. Sci. 2021, 22, 4587. [Google Scholar] [CrossRef] [PubMed]
- Mopidevi, B.; Kaw, M.K.; Puri, N.; Ponnala, M.; Jain, S.; Rana, A.; Keetha, N.R.; Khuder, S.A.; Fiering, S.N.; Kumar, A. Variable transcriptional regulation of the human aldosterone synthase gene causes salt-dependent high blood pressure in transgenic mice. Circ. Cardiovasc. Genet. 2015, 8, 30–39. [Google Scholar] [CrossRef]
- Pellieux, C.; Montessuit, C.; Papageorgiou, I.; Pedrazzini, T.; Lerch, R. Differential effects of high-fat diet on myocardial lipid metabolism in failing and nonfailing hearts with angiotensin II-mediated cardiac remodeling in mice. Am. J. Physiol. Heart Circ. Physiol. 2012, 302, H1795–H1805. [Google Scholar] [CrossRef]
- Dogra, P.; Bancos, I.; Young, W.F., Jr. Primary aldosteronism: A pragmatic approach to diagnosis and management. Mayo Clin. Proc. 2023, 98, 1207–1215. [Google Scholar] [CrossRef]
- Hanslik, G.; Wallaschofski, H.; Dietz, A.; Riester, A.; Reincke, M.; Allolio, B.; Lang, K.; Quack, I.; Rump, L.C.; Willenberg, H.S.; et al. Increased prevalence of diabetes mellitus and the metabolic syndrome in patients with primary aldsoteronism of the German Conn’s registry. Eur. J. Endocrinol. 2015, 173, 665–675. [Google Scholar] [CrossRef] [PubMed]
- Akehi, Y.; Yanase, T.; Motonaga, R.; Umakoshi, H.; Tsuiki, M.; Takeda, Y.; Yoneda, T.; Kurihara, I.; Itoh, H.; Katabami, T.; et al. High prevalence of diabetes in patients with primary aldosteronism (PA) associated with subclinical hypercortisolism and prediabetes more prevalent in bilateral than unilateral PA: A large, multicenter cohort study in Japan. Diabetes Care. 2019, 42, 938–945. [Google Scholar] [CrossRef]
- Garg, R.; Adler, G.K. Role of mineralocorticoid receptor in insulin resistance. Curr. Opin. Endocrinol. Diabetes Obes. 2012, 19, 168–175. [Google Scholar] [CrossRef]
- Adler, G.K.; Murray, G.R.; Turcu, A.F.; Nian, H.; Yu, C.; Solorzano, C.C.; Manning, R.; Dungeng Peng, D.; Luther, J.M. Primary aldosteronism decreases insulin secretion and increases insulin clearance in humans. Hypertension 2020, 75, 1251–1259. [Google Scholar] [CrossRef]
- Wu, C.; Zhang, H.; Zhang, J.; Xie, C.; Fan, C.; Zhang, H.; Wu, P.; Wei, Q.; Tan, W.; Xu, L.; et al. Inflammation and fibrosis in peripheral adipose tissue of patients with aldosterone-producing adenoma. Endocrinology 2018, 159, 227–237. [Google Scholar] [CrossRef] [PubMed]
- Kalupahana, N.S.; Massiera, F.; Quignard-Boulange, A.; Ailhaud, G.; Voy, B.H.; Wasserman, D.H.; Moustaid-Moussa, N. Overproduction of angiotensinogen from adipose tissue induces adipose inflammation, glucose intolerance, and insulin resistance. Obesity 2012, 20, 48–56. [Google Scholar] [CrossRef]
- Bader, M.; Steckelings, U.M.; Alenina, N.; Santos, R.A.S.; Ferrario, C.M. Alternative renin-angiotensin system. Hypertension 2024, 81, 964–976. [Google Scholar] [CrossRef] [PubMed]
- Jarajapu, Y.P.R. Targeting angiotensin-converting enzyme-2/angiotensin-(1–7)/mas receptor axis in the vascular progenitor cells for cardiovascular diseases. Mol. Pharmacol. 2021, 99, 29–38. [Google Scholar] [CrossRef] [PubMed]
- Mudersbach, T.; Siuda, D.; Kohlstedt, K.; Fleming, I. Epigenetic control of the angiotensin-converting enzyme in endothelial cells during inflammation. PLoS ONE 2019, 14, e0216218. [Google Scholar] [CrossRef]
- Najafipour, R.; Mohammadi, D.; Momeni, A.; Moghbelinejad, S.J. Effect of B12 and folate deficiency in hypomethylation of Angiotensin I converting enzyme 2 gene and severity of disease among the acute respiratory distress syndrome patients. Clin. Lab. Anal. 2023, 37, e24846. [Google Scholar] [CrossRef]
- Ceconi, C.; Francolini, G.; Olivares, A.; Comini, L.; Bachetti, T.; Ferrari, R. Angiotensin-converting enzyme (ACE) inhibitors have different selectivity for bradykinin binding sites of human somatic ACE. Eur. J. Pharmacol. 2007, 577, 1–6. [Google Scholar] [CrossRef]
- Ryznar, R.J.; Phibbs, L.; Van Winkle, L.J. Epigenetic modifications at the center of the Barker hypothesis and their transgenerational implications. Int. J. Environ. Res. Public Health 2021, 18, 12728. [Google Scholar] [CrossRef] [PubMed]
- Goyal, R.; Goyal, D.; Leitzke, A.; Gheorghe, C.P.; Longo, L.D. Brain renin-angiotensin system: Fetal epigenetic programming by maternal protein restriction during pregnancy. Reprod. Sci. 2010, 17, 227–238. [Google Scholar] [CrossRef] [PubMed]
- Rivière, G.; Lienhard, D.; Andrieu, T.; Vieau, D.; Frey, B.M.; Frey, F.J. Epigenetic regulation of somatic angiotensin-converting enzyme by DNA methylation and histone acetylation. Epigenetics 2011, 6, 478–489. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.A.; Cho, H.M.; Lee, D.Y.; Kim, K.C.; Han, H.S.; Kim, I.K. Tissue-specific upregulation of angiotensin-converting enzyme 1 in spontaneously hypertensive rats through histone code modifications. Hypertension 2012, 59, 621–626. [Google Scholar] [CrossRef]
- Hu, B.; Song, J.T.; Qu, H.Y.; Bi, C.L.; Huang, X.Z.; Liu, X.X.; Zhang, M. Mechanical stretch suppresses microRNA-145 expression by activating extracellular signal-regulated kinase 1/2 and upregulating angiotensin-converting enzyme to alter vascular smooth muscle cell phenotype. PLoS ONE 2014, 9, e96338. [Google Scholar] [CrossRef]
- Kohlstedt, K.; Trouvain, C.; Boettger, T.; Shi, L.; Fisslthaler, B.; Fleming, I. AMP-activated protein kinase regulates endothelial cell angiotensin-converting enzyme expression via p53 and the post-transcriptional regulation of microRNA-143/145. Circ. Res. 2013, 112, 1150–1158. [Google Scholar] [CrossRef] [PubMed]
- Turner, A.J.; Nalivaeva, N.N. Angiotensin-converting enzyme 2 (ACE2): Two decades of revelations and re-evaluation. Peptides 2022, 151, 170766. [Google Scholar] [CrossRef] [PubMed]
- Mohammed, M.; Berdasco, C.; Lazartigues, E. Brain angiotensin converting enzyme-2 in central cardiovascular regulation. Clin. Sci. 2020, 134, 2535–2547. [Google Scholar] [CrossRef] [PubMed]
- Goyal, R.; Leitzke, A.; Goyal, D.; Gheorghe, C.P.; Longo, L.D. Antenatal maternal hypoxic stress: Adaptations in fetal lung renin-angiotensin system. Reprod. Sci. 2011, 18, 180–189. [Google Scholar] [CrossRef] [PubMed]
- Takeda, Y.; Demuea, M.; Yoneda, T.; Takeda, Y. Epigenesis of blood pressure regulating hormones. In Proceeding of the 45th Annual Scientific Meeting of Japanese Society of Hypertension, Osaka, Japan, 15–17 September 2023; p. 184. [Google Scholar]
- Sen, R.; Garbati, M.; Bryant, K.; Lu, Y. Epigenetic mechanisms influencing COVID-19. Genome 2021, 64, 372–385. [Google Scholar] [CrossRef]
- Lima, R.S.; Rocha, L.P.C.; Moreira, P.R. Genetic and epigenetic control of ACE2 expression and its possible role in COVID-19. Cell Biochem. Funct. 2021, 39, 713–726. [Google Scholar] [CrossRef] [PubMed]
- Chlamydas, S.; Papavassiliou, A.G.; Piperi, C. Epigenetic mechanisms regulating COVID-19 infection. Epigenetics 2021, 16, 263–270. [Google Scholar] [CrossRef] [PubMed]
- Pinto, B.G.G.; Oliveira, A.; Singh, Y.; Jimenez, L.; Gonçalves, A.N.A.; Ogava, R.L.T.; Creighton, R.; Schatzmann Peron, J.P.; Nakaya, H.I. ACE2 expression is increased in the lungs of patients with comorbidities associated with severe COVID-19. J. Infect. Dis. 2020, 222, 556–563. [Google Scholar] [CrossRef] [PubMed]
- Saponaro, F.; Rutigliano, G.; Sestito, S.; Bandini, L.; Storti, B.; Bizzarri, R.; Zucchi, R. ACE2 in the era of SARS-CoV-2: Controversies and novel perspectives. Front. Mol. Biosci. 2020, 7, 588618. [Google Scholar] [CrossRef]
- Elemam, N.M.; Hasswan, H.; Aljaibeji, H.; Sulaiman, N. Circulating soluble ace2 and upstream microRNA expressions in serum of type 2 diabetes mellitus patients. Int. J. Mol. Sci. 2021, 22, 5263. [Google Scholar] [CrossRef]
- Hejenkowska, E.D.; Mitash, N.; Donovan, J.E.; Chandra, A.; Bertrand, C.; De Santi, C.; Greene, C.M.; Mu, F.; Swiatecka-Urban, A. TGF-β1 inhibition of ace2 mediated by miRNA uncovers novel mechanism of SARS-CoV-2 pathogenesis. J. Innate Immun. 2023, 15, 629–646. [Google Scholar] [CrossRef] [PubMed]
- Gu, Q.; Wang, B.; Zhang, X.F.; Ma, Y.P.; Liu, J.D.; Wang, X.Z. Contribution of renin-angiotensin system to exercise-induced attenuation of aortic remodeling and improvement of endothelial function in spontaneously hypertensive rats. Cardiovasc. Pathol. 2014, 23, 298–305. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.J.; Huang, Y.L.; Shih, Y.Y.; Wu, H.Y.; Peng, C.T.; Lo, W.Y. MicroRNA-146a decreases high glucose/thrombin-induced endothelial inflammation by inhibiting NAPDH oxidase 4 expression. Mediators Inflamm. 2014, 2014, 379537. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.J.; Xu, R.; Yu, H.M.; Chang, Q.; Zhong, J.C. The ace2/apelin signaling, microRNAs, and hypertension. Int. J. Hypertens. 2015, 2015, 896861. [Google Scholar] [CrossRef] [PubMed]
- Xu, T.; Fan, X.; Zhao, M.; Wu, M.; Li, H.; Ji, B.; Zhu, X.; Li, L.; Ding, H.; Sun, M.; et al. DNA methylation-reprogrammed ang II (angiotensin II) type 1 receptor-early growth response gene 1-protein kinase C ε axis underlies vascular hypercontractility in antenatal hypoxic offspring. Hypertension 2021, 77, 491–506. [Google Scholar] [CrossRef] [PubMed]
- Papadopoulos, K.I.; Papadopoulou, A.; Aw, T.C. MicroRNA-155 mediates endogenous angiotensin II type 1 receptor regulation: Implications for innovative type 2 diabetes mellitus management. World J. Diabetes 2023, 14, 1334–1340. [Google Scholar] [CrossRef] [PubMed]
- Kawakami-Mori, F.; Nishimoto, M.; Reheman, L.; Kawarazaki, W.; Ayuzawa, N.; Ueda, K.; Hirohama, D.; Kohno, D.; Oba, S.; Shimosawa, T.; et al. DNA methylation of hypothalamic angiotensin receptor in prenatal programmed hypertension. JCI Insight 2018, 3, e95625. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, K.; Zhou, J.J.; Shao, J.Y.; Chen, S.R.; Pan, H.L. DNA demethylation in the hypothalamus promotes transcription of Agtr1a and Slc12a2 and hypertension development. J. Biol. Chem. 2023, 300, 105597. [Google Scholar] [CrossRef] [PubMed]
- Shan, M.; Li, S.; Zhang, Y.; Chen, Y.; Zhou, Y.; Shi, L. Maternal exercise upregulates the DNA methylation of Agtr1a to enhance vascular function in offspring of hypertensive rats. Hypertens. Res. 2023, 46, 654–666. [Google Scholar] [CrossRef]
- Zheng, L.; Xu, C.C.; Chen, W.D.; Shen, W.L.; Ruan, C.C.; Zhu, L.M.; Zhu, D.L.; Gao, P.J. MicroRNA-155 regulates angiotensin II type 1 receptor expression and phenotypic differentiation in vascular adventitial fibroblasts. Biochem. Biophys. Res. Commun. 2010, 400, 483–488. [Google Scholar] [CrossRef]
- Takeda, Y. Effects of eplerenone, a selective mineralocorticoid receptor antagonist, on clinical and experimental salt-sensitive hypertension. Hypertens. Res. 2009, 32, 321–324. [Google Scholar] [CrossRef] [PubMed]
- Forrester, S.J.; Booz, G.W.; Sigmund, C.D.; Coffman, T.M.; Kawai, T.; Rizzo, V.; Scalia, R.; Eguchi, S. Angiotensin II signal transduction: An update on mechanisms of physiology and pathophysiology. Physiol. Rev. 2018, 98, 1627–1738. [Google Scholar] [CrossRef] [PubMed]
- DuPont, J.J.; McCurley, A.; Davel, A.P.; McCarthy, J.; Bender, S.B.; Hong, K.; Yang, Y.; Yoo, J.K.; Aronovitz, M.; Baur, W.E.; et al. Vascular mineralocorticoid receptor regulates microRNA-155 to promote vasoconstriction and rising blood pressure with aging. JCI Insight 2016, 1, e88942. [Google Scholar] [CrossRef] [PubMed]
- Guo, D.F.; Sun, Y.L.; Hamet, P.; Inagami, T. The angiotensin II type 1 receptor and receptor-associated proteins. Cell Res. 2001, 11, 165–180. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Zhu, Q.; Sun, S.; Qiu, Y.; Li, J.; Liu, W.; Yuan, G.; Ma, H. Renal transplantation increases angiotensin II receptor-mediated vascular contractility associated with changes of epigenetic mechanisms. Int. J. Mol. Med. 2018, 41, 2375–2388. [Google Scholar] [CrossRef] [PubMed]
- Taguchi, S.; Azushima, K.; Yamaji, T.; Suzuki, T.; Abe, E.; Tanaka, S.; Hirota, K.; Tsukamoto, S.; Morita, R.; Kobayashi, R.; et al. Angiotensin II type 1 receptor-associated protein deletion combined with angiotensin II stimulation accelerates the development of diabetic kidney disease in mice on a C57BL/6 strain. Hypertens. Res. 2024, 47, 55–66. [Google Scholar] [CrossRef] [PubMed]
- Hirota, K.; Yamashita, A.; Abe, E.; Yamaji, T.; Azushima, K.; Tanaka, S.; Taguchi, S.; Tsukamoto, S.; Wakui, H.; Tamura, K. miR-125a-5p/miR-125b-5p contributes to pathological activation of angiotensin II-AT1R in mouse distal convoluted tubule cells by the suppression of Atrap. J. Biol. Chem. 2023, 299, 105478. [Google Scholar] [CrossRef]
- Takeda, Y.; Demura, M.; Wang, F.; Karashima, S.; Yoneda, T.; Kometani, M.; Hashimoto, A.; Aono, D.; Horike, S.I.; Meguro-Horike, M.; et al. Epigenetic regulation of aldosterone synthase gene by sodium and angiotensin II. J. Am. Heart Assoc. 2018, 7, e008281. [Google Scholar] [CrossRef] [PubMed]
- Robertson, S.; MacKenzie, S.M.; Alvarez-Madrazo, S.; Diver, L.A.; Lin, J.; Stewart, P.M.; Fraser, R.; Connell, J.M.; Davies, E. MicroRNA-24 is a novel regulator of aldosterone and cortisol production in the human adrenal cortex. Hypertension 2013, 62, 572–578. [Google Scholar] [CrossRef] [PubMed]
- Robertson, S.; Diver, L.A.; Alvarez-Madrazo, S.; Livie, C.; Ejaz, A.; Fraser, R.; Connell, J.M.; MacKenzie, S.M.; Davies, E. Regulation of corticosteroidogenic genes by microRNAs. Int. J. Endocrinol. 2017, 2017, 2021903. [Google Scholar] [CrossRef]
- Zhang, G.; Zou, X.; Liu, Q.; Xie, T.; Huang, R.; Kang, H.; Lai, C.; Zhu, J. MiR-193a-3p functions as a tumour suppressor in human aldosterone-producing adrenocortical adenoma by down-regulating CYP11B2. Int. J. Exp. Pathol. 2018, 99, 77–86. [Google Scholar] [CrossRef] [PubMed]
- Syed, M.; Ball, J.P.; Mathis, K.W.; Hall, M.E.; Ryan, M.J.; Rothenberg, M.E.; Yanes Cardozo, L.L.; Romero, D.G. MicroRNA-21 ablation exacerbates aldosterone-mediated cardiac injury, remodeling, and dysfunction. Am. J. Physiol. Endocrinol. Metab. 2018, 315, E1154–E1167. [Google Scholar] [CrossRef] [PubMed]
- Nishimoto, K.; Ogishima, T.; Sugiura, Y.; Suematsu, M.; Mukai, K. Pathology and gene mutations of aldosterone-producing lesions. Endocr. J. 2023, 70, 1113–1122. [Google Scholar] [CrossRef] [PubMed]
- Howard, B.; Wang, Y.; Xekouki, P.; Faucz, F.R.; Jain, M.; Zhang, L.; Meltzer, P.G.; Stratakis, C.A.; Kebebew, E. Integrated analysis of genome-wide methylation and gene expression shows epigenetic regulation of CYP11B2 in aldosteronomas. J. Clin. Endocrinol. Metab. 2014, 99, E536–E543. [Google Scholar] [CrossRef] [PubMed]
- Di Dalmazi, G.; Morandi, L.; Rubin, B.; Pilon, C.; Asioli, S.; Vicennati, V.; De Leo, A.; Ambrosi, F.; Santini, D.; Pagotto, U.; et al. DNA methylation of steroidogenic enzymes in benign adrenocortical tumors: New insights in aldosterone-producing adenomas. J. Clin. Endocrinol. Metab. 2020, 105, dgaa585. [Google Scholar] [CrossRef] [PubMed]
- Takeda, Y.; Demura, M.; Wang, F.; Karashima, S.; Yoneda, T.; Kometani, M.; Aomo, D.; Hashimoto, A.; Horike, S.; Meguro-Horike, M.; et al. Effect of potassium on DNA methylation of aldosterone synthase gene. J. Hypertens. 2021, 39, 1018–1024. [Google Scholar] [CrossRef] [PubMed]
- Kometani, M.; Yoneda, T.; Demura, M.; Aono, D.; Gondoh, Y.; Karashima, S.; Nishimoto, K.; Yasuda, M.; Horike, S.; Takeda, Y. Genetic and epigenetic analyses of aldosterone-producing adenoma with hypercortisolemia. Steroids 2019, 151, 108470. [Google Scholar] [CrossRef] [PubMed]
- Ayuzawa, N.; Fujita, T. The mineralocorticoid receptor in salt-sensitive hypertension and renal injury. J. Am. Soc. Nephrol. 2021, 32, 279–289. [Google Scholar] [CrossRef] [PubMed]
- Fujita, T. Recent advances in hypertension: Epigenetic mechanism involved in development of salt-sensitive hypertension. Hypertension 2023, 80, 711–718. [Google Scholar] [CrossRef]
- Cao, N.; Lan, C.; Chen, C.; Xu, Z.; Luo, H.; Zheng, S.; Gong, X.; Ren, H.; Li, Z.; Qu, S.; et al. Prenatal lipopolysaccharides exposure induces transgenerational inheritance of hypertension. Circulation 2022, 146, 1082–1095. [Google Scholar] [CrossRef]
- Takeda, Y.; Demura, M.; Kometani, M.; Karashima, S.; Yoneda, T.; Takeda, Y. Molecular and epigenetic control of aldosterone synthase, CYP11B2 and 11-hydroxylase, CYP11B1. Int. J. Mol. Sci. 2023, 24, 5782. [Google Scholar] [CrossRef] [PubMed]
- Takeda, Y.; Yoneda, T.; Demura, M.; Furukawa, K.; Miyamori, I.; Mabuchi, H. Effects of high sodium intake on cardiovascular aldosterone synthesis in stroke-prone spontaneously hypertensive rats. J. Hypertens. 2001, 19, 635–639. [Google Scholar] [CrossRef] [PubMed]
- Garnier, A.; Bendall, J.K.; Fuchs, S.; Escoubet, B.; Rochais, F.; Hoerter, J.; Nehme, J.; Ambroisine, M.L.; De Angelis, N.; Morineau, G.; et al. Cardiac specific increase in aldosterone production induces coronary dysfunction in aldosterone synthase-transgenic mice. Circulation 2004, 110, 1819–1825. [Google Scholar] [CrossRef] [PubMed]
- Alesutan, I.; Voelkl, J.; Feger, M.; Kratschmar, D.V.; Castor, T.; Mia, S.; Sacherer, M.; Viereck, R.; Borst, O.; Leibrock, C.; et al. Involvement of vascular aldosterone synthase in phosphate-induced osteogenic transformation of vascular smooth muscle cells. Sci. Rep. 2017, 7, 2059. [Google Scholar] [CrossRef] [PubMed]
- Takeda, Y.; Yoneda, T.; Demura, M.; Usukura, M.; Mabuchi, H. Calcineurin inhibition attenuates mineralocorticoid-induced cardiac hypertrophy. Circulation 2002, 105, 677–679. [Google Scholar] [CrossRef] [PubMed]
- Yoshimura, M.; Nakamura, S.; Ito, T.; Nakayama, M.; Harada, E.; Mizuno, Y.; Sakamoto, T.; Yamamuro, M.; Saito, Y.; Nakao, K.; et al. Expression of aldosterone synthase gene in failing human heart: Quantitative analysis using modified real-time polymerase chain reaction. J. Clin. Endocrinol. Metab. 2002, 87, 3936–3940. [Google Scholar] [CrossRef] [PubMed]
- Ibarrola, J.; Jaffe, I.Z. The mineralocorticoid receptor in the vasculature: Friend or foe? Annu. Rev. Physiol. 2024, 86, 49–70. [Google Scholar] [CrossRef]
- Brown, J.M. Adverse effects of aldosterone: Beyond blood pressure. J. Am. Heart Assoc. 2024, 13, e030142. [Google Scholar] [CrossRef]
- Hatakeyama, H.; Miyamori, I.; Fujita, T.; Takeda, Y.; Takeda, R.; Yamamoto, H. Vascular aldosterone. biosynthesis and a link to angiotensin II-induced hypertrophy of vascular smooth muscle cells. J. Biol. Chem. 1994, 269, 24316–24320. [Google Scholar] [CrossRef]
- Mesquita, T.R.; Auguste, G.; Falcón, D.; Ruiz-Hurtado, G.; Salazar-Enciso, R.; Sabourin, J.; Lefebvre, F.; Viengchareun, S.; Kobeissy, H.; Lechène, P.; et al. Specific activation of the alternative cardiac promoter of cacna1c by the mineralocorticoid receptor. Circ. Res. 2018, 122, e49–e61. [Google Scholar] [CrossRef]
- Zhang, X.; Zhou, X.; Huang, Z.; Fan, X.; Tan, X.; Lu, C.; Yang, J. Aldosterone is a possible new stimulating factor for promoting vascular calcification. Front. Biosci. 2021, 26, 1052–1063. [Google Scholar] [CrossRef]
- Lee, H.A.; Song, M.J.; Seok, Y.M.; Kang, S.H.; Kim, S.Y.; Kim, I. Histone Deacetylase 3 and 4 Complex Stimulates the Transcriptional Activity of the Mineralocorticoid Receptor. PLoS ONE 2015, 10, e0136801. [Google Scholar] [CrossRef]
- Zheng, Q.; Li, N.; Zhang, Y.; Li, J.; Zhang, E.; Xu, Z. Fat-diets in perinatal stages altered nr3c2-mediated Ca(2+) currents in mesenteric arteries of offspring rats. Mol. Nutr. Food Res. 2023, 67, e2200722. [Google Scholar] [CrossRef] [PubMed]
- Camarda, N.D.; Ibarrola, J.; Biwer, L.A.; Jaffe, I.Z. Mineralocorticoid receptors in vascular smooth muscle: Blood pressure and beyond. Hypertension 2024, 81, 1008–1020. [Google Scholar] [CrossRef] [PubMed]
- Sõber, S.; Laan, M.; Annilo, T. MicroRNAs miR-124 and miR-135a are potential regulators of the mineralocorticoid receptor gene (NR3C2) expression. Biochem. Biophys. Res. Commun. 2010, 391, 727–732. [Google Scholar] [CrossRef] [PubMed]
- Martinez, E.C.; Lilyanna, S.; Wang, P.; Vardy, L.A.; Jiang, X.; Armugam, A.; Jeyaseelan, K.; Richards, A.M. MicroRNA-31 promotes adverse cardiac remodeling and dysfunction in ischemic heart disease. J. Mol. Cell. Cardiol. 2017, 112, 27–39. [Google Scholar] [CrossRef] [PubMed]
- Garg, A.; Foinquinos, A.; Jung, M.; Janssen-Peters, H.; Biss, S.; Bauersachs, J.; Gupta, S.K.; Thum, T. MiRNA-181a is a novel regulator of aldosterone-mineralocorticoid receptor-mediated cardiac remodelling. Eur. J. Heart Fail. 2020, 22, 1366–1377. [Google Scholar] [CrossRef]
- Koyama, R.; Mannic, T.; Ito, J.; Amar, L.; Zennaro, M.C.; Rossier, M.F.; Maturana, A.D. MicroRNA-204 is necessary for aldosterone-stimulated T-type calcium channel expression in cardiomyocytes. Int. J. Mol. Sci. 2018, 19, 2941. [Google Scholar] [CrossRef] [PubMed]
- Hao, J.; Zhang, L.; Cong, G.; Ren, L.; Hao, L. MicroRNA-34b/c inhibits aldosterone-induced vascular smooth muscle cell calcification via a SATB2/Runx2 pathway. Cell Tissue Res. 2016, 366, 733–746. [Google Scholar] [CrossRef]
- Hayakawa, K.; Kawasaki, M.; Hirai, T.; Yoshida, Y.; Tsushima, H.; Fujishiro, M.; Ikeda, K.; Morimoto, S.; Takamori, K.; Sekigawa, I. MicroRNA-766-3p contributes to anti-inflammatory responses through the indirect inhibition of NF-kappaB signaling. Int. J. Mol. Sci. 2019, 20, 809. [Google Scholar] [CrossRef]
- Zhu, A.; Yoneda, T.; Demura, M.; Karashima, S.; Usukura, M.; Yamagishi, M.; Takeda, Y. Effect of mineralocorticoid receptor blockade on the renal renin-angiotensin system in Dahl salt-sensitive hypertensive rats. J. Hypertens. 2009, 27, 800–805. [Google Scholar] [CrossRef] [PubMed]
- Ozbaki-Yagan, N.; Liu, X.; Bodnar, A.J.; Ho, J.; Butterworth, M.B. Aldosterone-induced microRNAs act as feedback regulators of mineralocorticoid receptor signaling in kidney epithelia. FASEB J. 2020, 4, 11714–11728. [Google Scholar] [CrossRef]
- Sierra-Ramos, C.; Velazquez-Garcia, S.; Keskus, A.G.; Vastola-Mascolo, A.; Rodríguez-Rodríguez, A.E.; Luis-Lima, S.; Hernández, G.; Navarro-González, J.F.; Porrini, E.; Konu, O.; et al. Increased SGK1 activity potentiates mineralocorticoid/NaCl-induced kidney injury. Am. J. Physiol. Ren. Physiol. 2021, 320, F628–F643. [Google Scholar] [CrossRef]
- Park, E.-J.; Jung, H.J.; Choi, H.-J.; Cho, J.-I.; Park, H.-J.; Kwon, T.-H. miR-34c-5p and CaMKII are involved in aldosterone-induced fibrosis in kidney collecting duct cells. Am. J. Physiol. Ren. Physiol. 2018, 314, F329–F342. [Google Scholar] [CrossRef]
- Zhang, J.; Zhang, Y.; Gao, J.; Wang, M.; Li, X.; Cui, Z.; Fu, G. Long noncoding RNA Tug1 promotes angiotensin II-induced renal fibrosis by binding to mineralocorticoid receptor and negatively regulating microR-29b-3p. Hypertension 2021, 78, 693–705. [Google Scholar] [CrossRef] [PubMed]
- Li, X.-Z.; Xiong, Z.-C.; Zhang, S.-L.; Hao, Q.-Y.; Liu, Z.-Y.; Zhang, H.-F.; Wang, J.-F.; Gao, J.-W.; Liu, P.-M. Upregulated LncRNA H19 sponges miR-106a-5p and contributes to aldosterone-induced vascular calcification via activating the Runx2-dependent pathway. Arterioscler. Thromb. Vasc. Biol. 2023, 43, 1684–1699. [Google Scholar] [CrossRef]
- Elvira-Matelot, E.; Zhou, X.; Farman, N.; Beaurain, G.; Henrion-Caude, A.; Hadchouel, J.; Jeunemaitre, X. Regulation of WNK1 expression by miR-192 and aldosterone. J. Am. Soc. Nephrol. 2010, 21, 1724–1731. [Google Scholar] [CrossRef]
- Subramanya, A.R.; Yang, C.L.; McCormick, J.A.; Ellison, D.H. WNK kinases regulate sodium chloride and potassium transport by the aldosterone-sensitive distal nephron. Kidney Int. 2006, 70, 630–634. [Google Scholar] [CrossRef] [PubMed]
- Lin, D.H.; Yue, P.; Zhang, C.; Wang, W.H. MicroRNA-194 (miR-194) regulates ROMK channel activity by targeting intersectin 1. Am. J. Physiol. Ren. Physiol. 2014, 306, F53–F60. [Google Scholar] [CrossRef]
- Lin, D.H.; Yue, P.; Pan, C.; Sun, P.; Wang, W.H. MicroRNA 802 stimulates ROMK channels by suppressing caveolin-1. J. Am. Soc. Nephrol. 2011, 22, 1087–1098. [Google Scholar] [CrossRef]
- Edinger, R.S.; Coronnello, C.; Bodnar, A.J.; Labarca, M.; Bhalla, V.; LaFramboise, W.A.; Benos, P.V.; Ho, J.; Johnson, J.P.; Butterworth, M.B. Aldosterone regulates microRNAs in the cortical collecting duct to alter sodium transport. J. Am. Soc. Nephrol. 2014, 25, 2445–2457. [Google Scholar] [CrossRef] [PubMed]
- Funder, J.W. Apparent mineralocorticoid excess: Research as an art form. Endocrine 2020, 70, 439–440. [Google Scholar] [CrossRef] [PubMed]
- Ueda, K.; Nishimoto, M.; Hirohama, D.; Ayuzawa, N.; Kawarazaki, W.; Watanabe, A.; Shimosawa, T.; Loffing, J.; Zhang, M.-Z.; Marumo, T.; et al. Renal dysfunction induced by kidney-specific gene deletion of Hsd11b2 as a primary cause of salt-dependent hypertension. Hypertension 2017, 70, 111–118. [Google Scholar] [CrossRef]
- Takeda, Y. Pathophysiological roles of vascular 11beta-hydroxysteroid dehydrogenase and aldosterone. J. Steroid Biochem. Mol. Biol. 2003, 85, 443–447. [Google Scholar] [CrossRef] [PubMed]
- Alikhani-Koopaei, R.; Fouladkou, F.; Frey, F.J.; Frey, B.M. Epigenetic regulation of 11 beta-hydroxysteroid dehydrogenase type 2 expression. J. Clin. Investig. 2004, 114, 1146–1157. [Google Scholar] [CrossRef] [PubMed]
- White, P.C.; Mune, T.; Agarwal, A.K. 11 beta-Hydroxysteroid dehydrogenase and the syndrome of apparent mineralocorticoid excess. Endocr. Rev. 1997, 18, 135–156. [Google Scholar] [PubMed]
- Pizzolo, F.; Friso, S.; Morandini, F.; Antoniazzi, F.; Zaltron, C.; Udali, S.; Gandini, A.; Cavarzere, P.; Salvagno, G.; Giorgetti, A.; et al. Apparent mineralocorticoid excess by a novel mutation and epigenetic modulation by HSD11B2 promoter methylation. J. Clin. Endocrinol. Metab. 2015, 100, E1234–E1241. [Google Scholar] [CrossRef]
- Rezaei, M.; Andrieu, T.; Neuenschwander, S.; Bruggmann, R.; Mordasini, D.; Frey, F.J.; Vogt, B.; Frey, B.M. Regulation of 11beta-hydroxysteroid dehydrogenase type 2 by microRNA. Hypertension 2014, 64, 860–866. [Google Scholar] [CrossRef] [PubMed]
- Soleimani, M.; Barone, S.; Luo, H.; Zahedi, K. Pathogenesis of hypertension in metabolic syndrome: The role of fructose and salt. Int. J. Mol. Sci. 2023, 24, 4294. [Google Scholar] [CrossRef]
- Nouchi, Y.; Munetsuna, E.; Yamada, H.; Yamazaki, M.; Ando, Y.; Mizuno, G.; Ikeya, M.; Kageyama, I.; Wakasugi, T.; Teshigawara, A.; et al. Maternal high-fructose corn syrup intake impairs corticosterone clearance by reducing renal 11β-Hsd2 activity via miR-27a-mediated mechanism in rat offspring. Nutrients 2023, 15, 2122. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Takeda, Y.; Demura, M.; Yoneda, T.; Takeda, Y. Epigenetic Regulation of the Renin–Angiotensin–Aldosterone System in Hypertension. Int. J. Mol. Sci. 2024, 25, 8099. https://doi.org/10.3390/ijms25158099
Takeda Y, Demura M, Yoneda T, Takeda Y. Epigenetic Regulation of the Renin–Angiotensin–Aldosterone System in Hypertension. International Journal of Molecular Sciences. 2024; 25(15):8099. https://doi.org/10.3390/ijms25158099
Chicago/Turabian StyleTakeda, Yoshimichi, Masashi Demura, Takashi Yoneda, and Yoshiyu Takeda. 2024. "Epigenetic Regulation of the Renin–Angiotensin–Aldosterone System in Hypertension" International Journal of Molecular Sciences 25, no. 15: 8099. https://doi.org/10.3390/ijms25158099





