Advancements in Plant-Based Therapeutics for Hepatic Fibrosis: Molecular Mechanisms and Nanoparticulate Drug Delivery Systems
Abstract
1. Introduction
2. Polyphenols and Hepatic Fibrosis
2.1. Phenolic Acids
| Class of Phenolic Acids | Bioactive Compounds | Cell Lines/ Animal Model | Pharmacological Effects | Reference |
|---|---|---|---|---|
| Hydroxycinnamic acids | Chlorogenic acid | LX-2 cells Sprague-Dawley rats |
| [25] |
| Sprague-Dawley rats |
| [26] | ||
| Ferulic acid | MPHs, RAW 264.7 cells, and LX-2 cells C57BL/6J mice |
| [27] | |
| Isochlorogenic acid B | C57BL/6 mice |
| [28] | |
| p-Coumaric acid | LX-2 cells C57BL/6 mice |
| [29] | |
| Rosmarinic acid | Sprague-Dawley rats HSC-T6 |
| [30] | |
| Salvianolic acid A | Sprague-Dawley rats |
| [31] | |
| Salvianolic acid B | C57BL/6 mice LO2 cells |
| [32] | |
| C57BL/6 mice LX2 and WRL68 cells |
| [33] | ||
| HSC-LX-2 cells BALB/c mice |
| [34] | ||
| JS1 and LX2 cells |
| [35] | ||
| Sprague-Dawley rats |
| [36] | ||
| LX-2 and T6 cells BALB/c mice |
| [37] | ||
| LX-2 cells |
| [38] | ||
| HSC-T6 and LX-2 cells |
| [39] | ||
| Sinapic acid | Sprague-Dawley rats |
| [40] | |
| Hydroxybenzoic acids | Gallic acid and dodecyl gallate | Wistar albino rats |
| [41] |
| Protocatechuic acid | HSC-T6 cells C57BL/6 mice |
| [42] | |
| Vanillic acid | Sprague-Dawley rats HSC-T6 cells |
| [43] |
2.2. Flavonoids
2.2.1. Flavanols
2.2.2. Flavonols
2.2.3. Flavones
2.2.4. Flavanones
2.2.5. Isoflavones
2.2.6. Anthocyanidins
2.2.7. Chalcones
2.3. Stilbenes
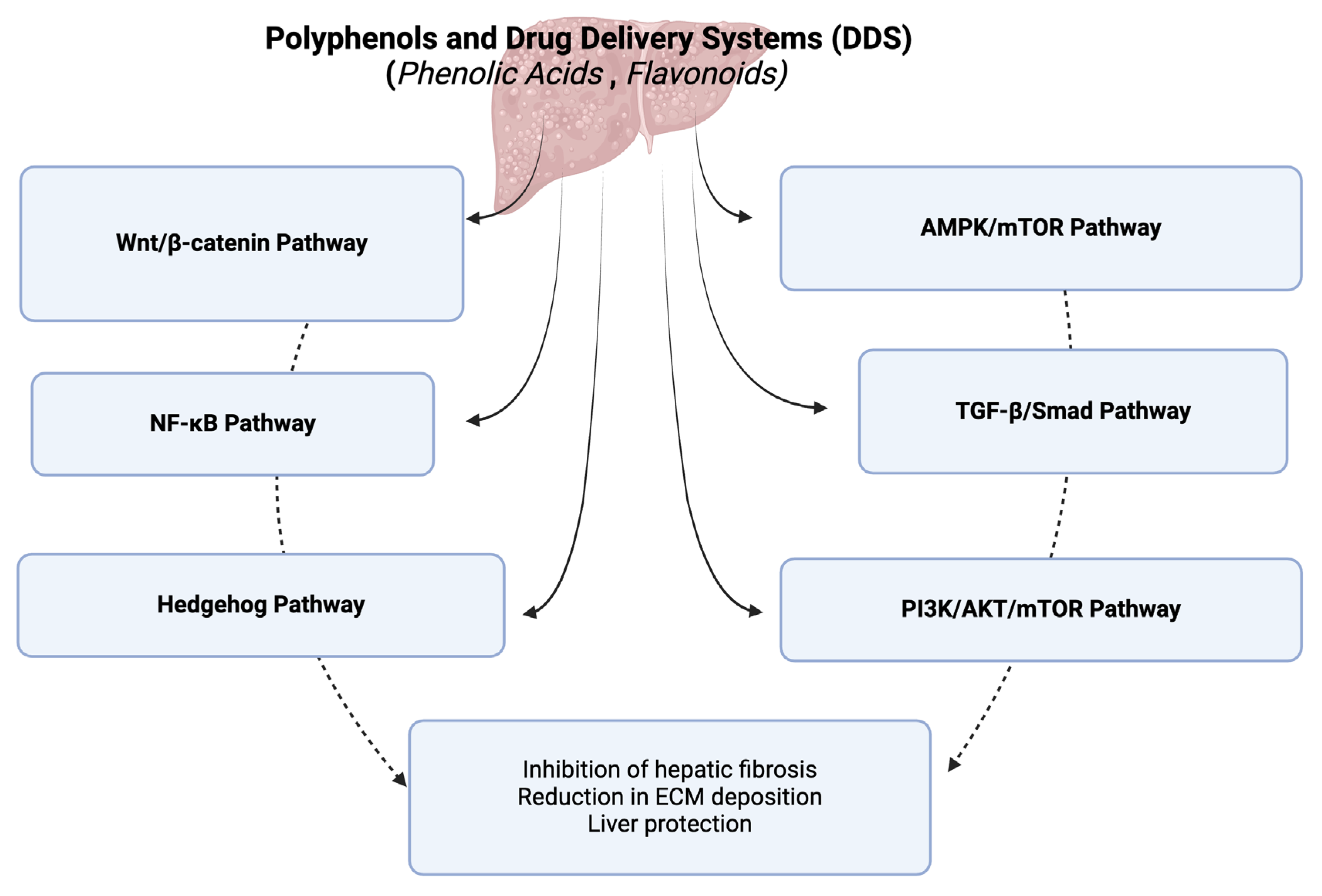
3. Polyphenol-Based Drug Delivery Systems and Hepatic Fibrosis
4. Conclusions and Perspectives
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Shan, L.; Wang, F.; Zhai, D.; Meng, X.; Liu, J.; Lv, X. New Drugs for Hepatic Fibrosis. Front. Pharmacol. 2022, 13, 874408. [Google Scholar] [CrossRef]
- Chang, J.; Huang, C.; Li, S.; Jiang, X.; Chang, H.; Li, M. Research Progress Regarding the Effect and Mechanism of Dietary Polyphenols in Liver Fibrosis. Molecules 2023, 29, 127. [Google Scholar] [CrossRef] [PubMed]
- Tan, Z.; Sun, H.; Xue, T.; Gan, C.; Liu, H.; Xie, Y.; Yao, Y.; Ye, T. Liver Fibrosis: Therapeutic Targets and Advances in Drug Therapy. Front. Cell Dev. Biol. 2021, 9, 730176. [Google Scholar] [CrossRef] [PubMed]
- Altamirano-Barrera, A.; Barranco-Fragoso, B.; Méndez-Sánchez, N. Management Strategies for Liver Fibrosis. Ann. Hepatol. 2017, 16, 48–56. [Google Scholar] [CrossRef] [PubMed]
- Duval, F.; Moreno-Cuevas, J.E.; González-Garza, M.T.; Rodríguez-Montalvo, C.; Cruz-Vega, D.E. Protective Mechanisms of Medicinal Plants Targeting Hepatic Stellate Cell Activation and Extracellular Matrix Deposition in Liver Fibrosis. Chin. Med. 2014, 9, 27. [Google Scholar] [CrossRef] [PubMed]
- Latief, U.; Ahmad, R. Herbal Remedies for Liver Fibrosis: A Review on the Mode of Action of Fifty Herbs. J. Tradit. Complement. Med. 2018, 8, 352–360. [Google Scholar] [CrossRef]
- Zhao, Q.; Luan, X.; Zheng, M.; Tian, X.-H.; Zhao, J.; Zhang, W.-D.; Ma, B.-L. Synergistic Mechanisms of Constituents in Herbal Extracts during Intestinal Absorption: Focus on Natural Occurring Nanoparticles. Pharmaceutics 2020, 12, 128. [Google Scholar] [CrossRef]
- Hermenean, A.; Smeu, C.; Gharbia, S.; Krizbai, I.A.; Ardelean, A. Plant-Derived Biomolecules and Drug Delivery Systems in the Treatment of Liver and Kidney Diseases. Curr. Pharm. Des. 2016, 22, 5415–5441. [Google Scholar] [CrossRef]
- Del Prete, A.; Scalera, A.; Iadevaia, M.D.; Miranda, A.; Zulli, C.; Gaeta, L.; Tuccillo, C.; Federico, A.; Loguercio, C. Herbal Products: Benefits, Limits, and Applications in Chronic Liver Disease. Evid.-Based Complement. Altern. Med. ECAM 2012, 2012, 837939. [Google Scholar] [CrossRef]
- Singh, S.; Sharma, N.; Shukla, S.; Behl, T.; Gupta, S.; Anwer, M.K.; Vargas-De-La-Cruz, C.; Bungau, S.G.; Brisc, C. Understanding the Potential Role of Nanotechnology in Liver Fibrosis: A Paradigm in Therapeutics. Molecules 2023, 28, 2811. [Google Scholar] [CrossRef]
- El-Tantawy, W.H.; Temraz, A. Anti-Fibrotic Activity of Natural Products, Herbal Extracts and Nutritional Components for Prevention of Liver Fibrosis: Review. Arch. Physiol. Biochem. 2022, 128, 382–393. [Google Scholar] [CrossRef]
- Rezaeiamiri, E.; Bahramsoltani, R.; Rahimi, R. Plant-Derived Natural Agents as Dietary Supplements for the Regulation of Glycosylated Hemoglobin: A Review of Clinical Trials. Clin. Nutr. 2020, 39, 331–342. [Google Scholar] [CrossRef]
- Kesarwani, K.; Gupta, R. Bioavailability Enhancers of Herbal Origin: An Overview. Asian Pac. J. Trop. Biomed. 2013, 3, 253–266. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.; Zhang, Y.; Sun, B. The Molecular Mechanisms of Liver Fibrosis and Its Potential Therapy in Application. Int. J. Mol. Sci. 2022, 23, 12572. [Google Scholar] [CrossRef]
- Laddomada, B.; Blanco, A.; Mita, G.; D’Amico, L.; Singh, R.P.; Ammar, K.; Crossa, J.; Guzmán, C. Drought and Heat Stress Impacts on Phenolic Acids Accumulation in Durum Wheat Cultivars. Foods 2021, 10, 2142. [Google Scholar] [CrossRef] [PubMed]
- Quideau, S.; Deffieux, D.; Douat-Casassus, C.; Pouységu, L. Plant Polyphenols: Chemical Properties, Biological Activities, and Synthesis. Angew. Chem. Int. Ed. Engl. 2011, 50, 586–621. [Google Scholar] [CrossRef]
- Scarano, A.; Chieppa, M.; Santino, A. Plant Polyphenols-Biofortified Foods as a Novel Tool for the Prevention of Human Gut Diseases. Antioxidants 2020, 9, 1225. [Google Scholar] [CrossRef]
- Scarano, A.; Chieppa, M.; Santino, A. Looking at Flavonoid Biodiversity in Horticultural Crops: A Colored Mine with Nutritional Benefits. Plants 2018, 7, 98. [Google Scholar] [CrossRef] [PubMed]
- Tsao, R. Chemistry and Biochemistry of Dietary Polyphenols. Nutrients 2010, 2, 1231–1246. [Google Scholar] [CrossRef]
- Cháirez-Ramírez, M.H.; de la Cruz-López, K.G.; García-Carrancá, A. Polyphenols as Antitumor Agents Targeting Key Players in Cancer-Driving Signaling Pathways. Front. Pharmacol. 2021, 12, 710304. [Google Scholar] [CrossRef]
- Al Mamari, H. Phenolic Compounds: Classification, Chemistry, and Updated Techniques of Analysis and Synthesis. In Phenolic Compounds—Chemistry, Synthesis, Diversity, Non-Conventional Industrial, Pharmaceutical and Therapeutic Applications; IntechOpen: London, UK, 2022; Volume 26. [Google Scholar] [CrossRef]
- El-Seedi, H.R.; El-Said, A.M.A.; Khalifa, S.A.M.; Göransson, U.; Bohlin, L.; Borg-Karlson, A.-K.; Verpoorte, R. Biosynthesis, Natural Sources, Dietary Intake, Pharmacokinetic Properties, and Biological Activities of Hydroxycinnamic Acids. J. Agric. Food Chem. 2012, 60, 10877–10895. [Google Scholar] [CrossRef] [PubMed]
- Manach, C.; Scalbert, A.; Morand, C.; Rémésy, C.; Jiménez, L. Polyphenols: Food Sources and Bioavailability. Am. J. Clin. Nutr. 2004, 79, 727–747. [Google Scholar] [CrossRef]
- Rocha, L.D.; Monteiro, M.C.; Teodoro, A.J. Anticancer Properties of Hydroxycinnamic Acids—A Review. Cancer Clin. Oncol. 2012, 1, 109. [Google Scholar] [CrossRef]
- Yang, F.; Luo, L.; Zhu, Z.-D.; Zhou, X.; Wang, Y.; Xue, J.; Zhang, J.; Cai, X.; Chen, Z.-L.; Ma, Q.; et al. Chlorogenic Acid Inhibits Liver Fibrosis by Blocking the miR-21-Regulated TGF-Β1/Smad7 Signaling Pathway in Vitro and in Vivo. Front. Pharmacol. 2017, 8, 929. [Google Scholar] [CrossRef]
- Shi, H.; Dong, L.; Jiang, J.; Zhao, J.; Zhao, G.; Dang, X.; Lu, X.; Jia, M. Chlorogenic Acid Reduces Liver Inflammation and Fibrosis through Inhibition of Toll-like Receptor 4 Signaling Pathway. Toxicology 2013, 303, 107–114. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Xue, X.; Fan, G.; Gu, Y.; Zhou, F.; Zheng, Q.; Liu, R.; Li, Y.; Ma, B.; Li, S.; et al. Ferulic Acid Ameliorates Hepatic Inflammation and Fibrotic Liver Injury by Inhibiting PTP1B Activity and Subsequent Promoting AMPK Phosphorylation. Front. Pharmacol. 2021, 12, 754976. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Huang, K.; Niu, Z.; Mei, D.; Zhang, B. Protective Effect of Isochlorogenic Acid B on Liver Fibrosis in Non-Alcoholic Steatohepatitis of Mice. Basic Clin. Pharmacol. Toxicol. 2019, 124, 144–153. [Google Scholar] [CrossRef] [PubMed]
- Truong, T.M.T.; Seo, S.H.; Chung, S.; Kang, I. Attenuation of Hepatic Fibrosis by P-Coumaric Acid via Modulation of NLRP3 Inflammasome Activation in C57BL/6 Mice. J. Nutr. Biochem. 2023, 112, 109204. [Google Scholar] [CrossRef]
- Li, G.-S.; Jiang, W.-L.; Tian, J.-W.; Qu, G.-W.; Zhu, H.-B.; Fu, F.-H. In Vitro and in Vivo Antifibrotic Effects of Rosmarinic Acid on Experimental Liver Fibrosis. Phytomedicine Int. J. Phytother. Phytopharm. 2010, 17, 282–288. [Google Scholar] [CrossRef]
- Wang, R.; Song, F.; Li, S.; Wu, B.; Gu, Y.; Yuan, Y. Salvianolic Acid A Attenuates CCl4-Induced Liver Fibrosis by Regulating the PI3K/AKT/mTOR, Bcl-2/Bax and Caspase-3/Cleaved Caspase-3 Signaling Pathways. Drug Des. Devel. Ther. 2019, 13, 1889–1900. [Google Scholar] [CrossRef]
- Fu, Y.; Zhou, X.; Wang, L.; Fan, W.; Gao, S.; Zhang, D.; Ling, Z.; Zhang, Y.; Ma, L.; Bai, F.; et al. Salvianolic Acid B Attenuates Liver Fibrosis by Targeting Ecm1 and Inhibiting Hepatocyte Ferroptosis. Redox Biol. 2024, 69, 103029. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.-B.; Jiang, L.; Ni, J.-D.; Xu, Y.-H.; Liu, F.; Liu, W.-M.; Wang, S.-G.; Liu, Z.-Q.; Wang, C.-Y. Salvianolic Acid B Suppresses Hepatic Fibrosis by Inhibiting Ceramide Glucosyltransferase in Hepatic Stellate Cells. Acta Pharmacol. Sin. 2023, 44, 1191–1205. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.; Li, S.; Chen, P.; Gu, Y.; Wang, S.; Wang, L.; Chen, C.; Wang, R.; Yuan, Y. Salvianolic Acid B Inhibits Hepatic Stellate Cell Activation and Liver Fibrosis by Targeting PDGFRβ. Int. Immunopharmacol. 2023, 122, 110550. [Google Scholar] [CrossRef] [PubMed]
- Jiang, N.; Zhang, J.; Ping, J.; Xu, L. Salvianolic Acid B Inhibits Autophagy and Activation of Hepatic Stellate Cells Induced by TGF-Β1 by Downregulating the MAPK Pathway. Front. Pharmacol. 2022, 13, 938856. [Google Scholar] [CrossRef]
- Tao, S.; Duan, R.; Xu, T.; Hong, J.; Gu, W.; Lin, A.; Lian, L.; Huang, H.; Lu, J.; Li, T. Salvianolic Acid B Inhibits the Progression of Liver Fibrosis in Rats via Modulation of the Hedgehog Signaling Pathway. Exp. Ther. Med. 2022, 23, 116. [Google Scholar] [CrossRef]
- Wang, R.; Li, S.; Chen, P.; Yue, X.; Wang, S.; Gu, Y.; Yuan, Y. Salvianolic Acid B Suppresses Hepatic Stellate Cell Activation and Liver Fibrosis by Inhibiting the NF-κB Signaling Pathway via miR-6499-3p/LncRNA-ROR. Phytomedicine Int. J. Phytother. Phytopharm. 2022, 107, 154435. [Google Scholar] [CrossRef]
- Tian, S.; Chen, M.; Wang, B.; Han, Y.; Shang, H.; Chen, J. Salvianolic Acid B Blocks Hepatic Stellate Cell Activation via FGF19/FGFR4 Signaling. Ann. Hepatol. 2021, 20, 100259. [Google Scholar] [CrossRef]
- Wu, C.; Chen, W.; Ding, H.; Li, D.; Wen, G.; Zhang, C.; Lu, W.; Chen, M.; Yang, Y. Salvianolic Acid B Exerts Anti-Liver Fibrosis Effects via Inhibition of MAPK-Mediated Phospho-Smad2/3 at Linker Regions in Vivo and in Vitro. Life Sci. 2019, 239, 116881. [Google Scholar] [CrossRef]
- Shin, D.-S.; Kim, K.W.; Chung, H.Y.; Yoon, S.; Moon, J.-O. Effect of Sinapic Acid against Dimethylnitrosamine-Induced Hepatic Fibrosis in Rats. Arch. Pharm. Res. 2013, 36, 608–618. [Google Scholar] [CrossRef]
- Perazzoli, M.R.A.; Perondi, C.K.; Baratto, C.M.; Winter, E.; Creczynski-Pasa, T.B.; Locatelli, C. Gallic Acid and Dodecyl Gallate Prevents Carbon Tetrachloride-Induced Acute and Chronic Hepatotoxicity by Enhancing Hepatic Antioxidant Status and Increasing P53 Expression. Biol. Pharm. Bull. 2017, 40, 425–434. [Google Scholar] [CrossRef]
- Cui, B.; Yang, Z.; Wang, S.; Guo, M.; Li, Q.; Zhang, Q.; Bi, X. The Protective Role of Protocatechuic Acid against Chemically Induced Liver Fibrosis in Vitro and in Vivo. Die Pharm. 2021, 76, 232–238. [Google Scholar] [CrossRef]
- Qin, L.; Tan, J.; Lv, X.; Zhang, J. Vanillic Acid Alleviates Liver Fibrosis through Inhibiting Autophagy in Hepatic Stellate Cells via the MIF/CD74 Signaling Pathway. Biomed. Pharmacother. Biomed. Pharmacother. 2023, 168, 115673. [Google Scholar] [CrossRef] [PubMed]
- Beecher, G.R. Overview of Dietary Flavonoids: Nomenclature, Occurrence and Intake. J. Nutr. 2003, 133, 3248S–3254S. [Google Scholar] [CrossRef] [PubMed]
- Cook, N.C.; Samman, S. Flavonoids—Chemistry, Metabolism, Cardioprotective Effects, and Dietary Sources. J. Nutr. Biochem. 1996, 7, 66–76. [Google Scholar] [CrossRef]
- Ratnam, D.V.; Ankola, D.D.; Bhardwaj, V.; Sahana, D.K.; Kumar, M.N.V.R. Role of Antioxidants in Prophylaxis and Therapy: A Pharmaceutical Perspective. J. Control. Release Off. J. Control. Release Soc. 2006, 113, 189–207. [Google Scholar] [CrossRef]
- Middleton, E.; Kandaswami, C.; Theoharides, T.C. The Effects of Plant Flavonoids on Mammalian Cells: Implications for Inflammation, Heart Disease, and Cancer. Pharmacol. Rev. 2000, 52, 673–751. [Google Scholar]
- Panche, A.N.; Diwan, A.D.; Chandra, S.R. Flavonoids: An Overview. J. Nutr. Sci. 2016, 5, e47. [Google Scholar] [CrossRef]
- Kubina, R.; Krzykawski, K.; Kabała-Dzik, A.; Wojtyczka, R.D.; Chodurek, E.; Dziedzic, A. Fisetin, a Potent Anticancer Flavonol Exhibiting Cytotoxic Activity against Neoplastic Malignant Cells and Cancerous Conditions: A Scoping, Comprehensive Review. Nutrients 2022, 14, 2604. [Google Scholar] [CrossRef]
- Forbes, A.M.; Meier, G.P.; Haendiges, S.; Taylor, L.P. Structure-Activity Relationship Studies of Flavonol Analogues on Pollen Germination. J. Agric. Food Chem. 2014, 62, 2175–2181. [Google Scholar] [CrossRef]
- George, J.; Tsuchishima, M.; Tsutsumi, M. Epigallocatechin-3-Gallate Inhibits Osteopontin Expression and Prevents Experimentally Induced Hepatic Fibrosis. Biomed. Pharmacother. Biomed. Pharmacother. 2022, 151, 113111. [Google Scholar] [CrossRef]
- Arffa, M.L.; Zapf, M.A.; Kothari, A.N.; Chang, V.; Gupta, G.N.; Ding, X.; Al-Gayyar, M.M.; Syn, W.; Elsherbiny, N.M.; Kuo, P.C.; et al. Epigallocatechin-3-Gallate Upregulates miR-221 to Inhibit Osteopontin-Dependent Hepatic Fibrosis. PLoS ONE 2016, 11, e0167435. [Google Scholar] [CrossRef]
- Yu, D.; Zhang, C.; Zhao, S.; Zhang, S.; Zhang, H.; Cai, S.; Shao, R.; He, H. The Anti-Fibrotic Effects of Epigallocatechin-3-Gallate in Bile Duct-Ligated Cholestatic Rats and Human Hepatic Stellate LX-2 Cells Are Mediated by the PI3K/Akt/Smad Pathway. Acta Pharmacol. Sin. 2015, 36, 473–482. [Google Scholar] [CrossRef] [PubMed]
- Tipoe, G.L.; Leung, T.M.; Liong, E.C.; Lau, T.Y.H.; Fung, M.L.; Nanji, A.A. Epigallocatechin-3-Gallate (EGCG) Reduces Liver Inflammation, Oxidative Stress and Fibrosis in Carbon Tetrachloride (CCl4)-Induced Liver Injury in Mice. Toxicology 2010, 273, 45–52. [Google Scholar] [CrossRef] [PubMed]
- Yasuda, Y.; Shimizu, M.; Sakai, H.; Iwasa, J.; Kubota, M.; Adachi, S.; Osawa, Y.; Tsurumi, H.; Hara, Y.; Moriwaki, H. (−)-Epigallocatechin Gallate Prevents Carbon Tetrachloride-Induced Rat Hepatic Fibrosis by Inhibiting the Expression of the PDGFRbeta and IGF-1R. Chem. Biol. Interact. 2009, 182, 159–164. [Google Scholar] [CrossRef] [PubMed]
- Zhen, M.-C.; Wang, Q.; Huang, X.-H.; Cao, L.-Q.; Chen, X.-L.; Sun, K.; Liu, Y.-J.; Li, W.; Zhang, L.-J. Green Tea Polyphenol Epigallocatechin-3-Gallate Inhibits Oxidative Damage and Preventive Effects on Carbon Tetrachloride-Induced Hepatic Fibrosis. J. Nutr. Biochem. 2007, 18, 795–805. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Liu, X.; Ding, C.; Gu, Y.; Liu, W. Dihydromyricetin Reverses Thioacetamide-Induced Liver Fibrosis Through Inhibiting NF-κB-Mediated Inflammation and TGF-Β1-Regulated of PI3K/Akt Signaling Pathway. Front. Pharmacol. 2021, 12, 783886. [Google Scholar] [CrossRef]
- Zhou, X.; Yu, L.; Zhou, M.; Hou, P.; Yi, L.; Mi, M. Dihydromyricetin Ameliorates Liver Fibrosis via Inhibition of Hepatic Stellate Cells by Inducing Autophagy and Natural Killer Cell-Mediated Killing Effect. Nutr. Metab. 2021, 18, 64. [Google Scholar] [CrossRef]
- Kong, R.; Wang, N.; Luo, H.; Lu, J. Hesperetin Mitigates Bile Duct Ligation-Induced Liver Fibrosis by Inhibiting Extracellular Matrix and Cell Apoptosis via the TGF-Β1/Smad Pathway. Curr. Mol. Med. 2018, 18, 15–24. [Google Scholar] [CrossRef]
- Chen, X.; Li, X.-F.; Chen, Y.; Zhu, S.; Li, H.-D.; Chen, S.-Y.; Wang, J.-N.; Pan, X.-Y.; Bu, F.-T.; Huang, C.; et al. Hesperetin Derivative Attenuates CCl4-Induced Hepatic Fibrosis and Inflammation by Gli-1-Dependent Mechanisms. Int. Immunopharmacol. 2019, 76, 105838. [Google Scholar] [CrossRef]
- Zhu, S.; Chen, X.; Chen, S.-Y.; Wang, A.; Wu, S.; Wu, Y.-Y.; Cheng, M.; Xu, J.-J.; Li, X.-F.; Huang, C.; et al. Hesperetin Derivative Decreases CCl4 -Induced Hepatic Fibrosis by Ptch1-Dependent Mechanisms. J. Biochem. Mol. Toxicol. 2022, 36, e23149. [Google Scholar] [CrossRef]
- Lin, X.; Kong, L.-N.; Huang, C.; Ma, T.-T.; Meng, X.-M.; He, Y.; Wang, Q.-Q.; Li, J. Hesperetin Derivative-7 Inhibits PDGF-BB-Induced Hepatic Stellate Cell Activation and Proliferation by Targeting Wnt/β-Catenin Pathway. Int. Immunopharmacol. 2015, 25, 311–320. [Google Scholar] [CrossRef] [PubMed]
- Li, W.-X.; Chen, X.; Yang, Y.; Huang, H.-M.; Li, H.; Huang, C.; Meng, X.-M.; Li, J. Hesperitin Derivative-11 Suppress Hepatic Stellate Cell Activation and Proliferation by Targeting PTEN/AKT Pathway. Toxicology 2017, 381, 75–86. [Google Scholar] [CrossRef] [PubMed]
- Li, J.-J.; Jiang, H.-C.; Wang, A.; Bu, F.-T.; Jia, P.-C.; Zhu, S.; Zhu, L.; Huang, C.; Li, J. Hesperetin Derivative-16 Attenuates CCl4-Induced Inflammation and Liver Fibrosis by Activating AMPK/SIRT3 Pathway. Eur. J. Pharmacol. 2022, 915, 174530. [Google Scholar] [CrossRef] [PubMed]
- Nasehi, Z.; Kheiripour, N.; Taheri, M.A.; Ardjmand, A.; Jozi, F.; Shahaboddin, M.E. Efficiency of Hesperidin against Liver Fibrosis Induced by Bile Duct Ligation in Rats. BioMed Res. Int. 2023, 2023, 5444301. [Google Scholar] [CrossRef]
- Pérez-Vargas, J.E.; Zarco, N.; Shibayama, M.; Segovia, J.; Tsutsumi, V.; Muriel, P. Hesperidin Prevents Liver Fibrosis in Rats by Decreasing the Expression of Nuclear Factor-κB, Transforming Growth Factor-β and Connective Tissue Growth Factor. Pharmacology 2014, 94, 80–89. [Google Scholar] [CrossRef] [PubMed]
- Geng, W.; Zhou, G.; Zhao, B.; Xiao, Q.; Li, C.; Fan, S.; Dong, P.; Zheng, J. Liquiritigenin Suppresses the Activation of Hepatic Stellate Cells via Targeting miR-181b/PTEN Axis. Phytomedicine Int. J. Phytother. Phytopharm. 2019, 66, 153108. [Google Scholar] [CrossRef]
- Lee, E.H.; Park, K.-I.; Kim, K.-Y.; Lee, J.-H.; Jang, E.J.; Ku, S.K.; Kim, S.C.; Suk, H.Y.; Park, J.Y.; Baek, S.Y.; et al. Liquiritigenin Inhibits Hepatic Fibrogenesis and TGF-Β1/Smad with Hippo/YAP Signal. Phytomed. Int. J. Phytother. Phytopharm. 2019, 62, 152780. [Google Scholar] [CrossRef]
- Chen, L.; Xia, S.; Wang, S.; Zhou, Y.; Wang, F.; Li, Z.; Li, Y.; Kong, D.; Zhang, Z.; Shao, J.; et al. Naringenin Is a Potential Immunomodulator for Inhibiting Liver Fibrosis by Inhibiting the cGAS-STING Pathway. J. Clin. Transl. Hepatol. 2023, 11, 26–37. [Google Scholar] [CrossRef]
- Hernández-Aquino, E.; Quezada-Ramírez, M.A.; Silva-Olivares, A.; Casas-Grajales, S.; Ramos-Tovar, E.; Flores-Beltrán, R.E.; Segovia, J.; Shibayama, M.; Muriel, P. Naringenin Attenuates the Progression of Liver Fibrosis via Inactivation of Hepatic Stellate Cells and Profibrogenic Pathways. Eur. J. Pharmacol. 2019, 865, 172730. [Google Scholar] [CrossRef]
- Hernández-Aquino, E.; Zarco, N.; Casas-Grajales, S.; Ramos-Tovar, E.; Flores-Beltrán, R.E.; Arauz, J.; Shibayama, M.; Favari, L.; Tsutsumi, V.; Segovia, J.; et al. Naringenin Prevents Experimental Liver Fibrosis by Blocking TGFβ-Smad3 and JNK-Smad3 Pathways. World J. Gastroenterol. 2017, 23, 4354–4368. [Google Scholar] [CrossRef]
- El-Mihi, K.A.; Kenawy, H.I.; El-Karef, A.; Elsherbiny, N.M.; Eissa, L.A. Naringin Attenuates Thioacetamide-Induced Liver Fibrosis in Rats through Modulation of the PI3K/Akt Pathway. Life Sci. 2017, 187, 50–57. [Google Scholar] [CrossRef]
- Gajender; Mazumder, A.; Sharma, A.; Azad, M.A.K. A Comprehensive Review of the Pharmacological Importance of Dietary Flavonoids as Hepatoprotective Agents. Evid.-Based Complement. Altern. Med. ECAM 2023, 2023, 4139117. [Google Scholar] [CrossRef]
- Spagnuolo, C.; Moccia, S.; Russo, G.L. Anti-Inflammatory Effects of Flavonoids in Neurodegenerative Disorders. Eur. J. Med. Chem. 2018, 153, 105–115. [Google Scholar] [CrossRef]
- Tahir, M.S.; Almezgagi, M.; Zhang, Y.; Bashir, A.; Abdullah, H.M.; Gamah, M.; Wang, X.; Zhu, Q.; Shen, X.; Ma, Q.; et al. Mechanistic New Insights of Flavonols on Neurodegenerative Diseases. Biomed. Pharmacother. 2021, 137, 111253. [Google Scholar] [CrossRef] [PubMed]
- Daryanavard, H.; Postiglione, A.E.; Mühlemann, J.K.; Muday, G.K. Flavonols Modulate Plant Development, Signaling, and Stress Responses. Curr. Opin. Plant Biol. 2023, 72, 102350. [Google Scholar] [CrossRef] [PubMed]
- Falcone Ferreyra, M.L.; Rius, S.P.; Casati, P. Flavonoids: Biosynthesis, Biological Functions, and Biotechnological Applications. Front. Plant Sci. 2012, 3, 222. [Google Scholar] [CrossRef]
- Popiolek-Kalisz, J.; Fornal, E. The Impact of Flavonols on Cardiovascular Risk. Nutrients 2022, 14, 1973. [Google Scholar] [CrossRef] [PubMed]
- Sharma, A.; Sharma, P.; Singh Tuli, H.; Sharma, A.K. Phytochemical and Pharmacological Properties of Flavonols. In eLS; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2018; pp. 1–12. ISBN 978-0-470-01590-2. [Google Scholar]
- El-Fadaly, A.A.; Afifi, N.A.; El-Eraky, W.; Salama, A.; Abdelhameed, M.F.; El-Rahman, S.S.A.; Ramadan, A. Fisetin Alleviates Thioacetamide-Induced Hepatic Fibrosis in Rats by Inhibiting Wnt/β-Catenin Signaling Pathway. Immunopharmacol. Immunotoxicol. 2022, 44, 355–366. [Google Scholar] [CrossRef] [PubMed]
- Xiong, Y.; Lu, H.; Xu, H. Galangin Reverses Hepatic Fibrosis by Inducing HSCs Apoptosis via the PI3K/Akt, Bax/Bcl-2, and Wnt/β-Catenin Pathway in LX-2 Cells. Biol. Pharm. Bull. 2020, 43, 1634–1642. [Google Scholar] [CrossRef]
- Wang, X.; Gong, G.; Yang, W.; Li, Y.; Jiang, M.; Li, L. Antifibrotic Activity of Galangin, a Novel Function Evaluated in Animal Liver Fibrosis Model. Environ. Toxicol. Pharmacol. 2013, 36, 288–295. [Google Scholar] [CrossRef]
- Rashidi, M.; Matour, E.; Beheshti Nasab, H.; Cheraghzadeh, M.; Shakerian, E. Isorhamnetin Exerts Antifibrotic Effects by Attenuating Platelet-Derived Growth Factor-BB-Induced HSC-T6 Cells Activation via Suppressing PI3K-AKT Signaling Pathway. Iran. Biomed. J. 2023, 27, 199–204. [Google Scholar] [CrossRef] [PubMed]
- Liu, N.; Feng, J.; Lu, X.; Yao, Z.; Liu, Q.; Lv, Y.; Han, Y.; Deng, J.; Zhou, Y. Isorhamnetin Inhibits Liver Fibrosis by Reducing Autophagy and Inhibiting Extracellular Matrix Formation via the TGF-Β1/Smad3 and TGF-Β1/P38 MAPK Pathways. Mediat. Inflamm. 2019, 2019, 6175091. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.H.; Kim, S.C.; Kim, K.M.; Jang, C.H.; Cho, S.S.; Kim, S.J.; Ku, S.K.; Cho, I.J.; Ki, S.H. Isorhamnetin Attenuates Liver Fibrosis by Inhibiting TGF-β/Smad Signaling and Relieving Oxidative Stress. Eur. J. Pharmacol. 2016, 783, 92–102. [Google Scholar] [CrossRef] [PubMed]
- Cao, R.; Cao, C.; Hu, X.; Du, K.; Zhang, J.; Li, M.; Li, B.; Lin, H.; Zhang, A.; Li, Y.; et al. Kaempferol Attenuates Carbon Tetrachloride (CCl4)-Induced Hepatic Fibrosis by Promoting ASIC1a Degradation and Suppression of the ASIC1a-Mediated ERS. Phytomed. Int. J. Phytother. Phytopharm. 2023, 121, 155125. [Google Scholar] [CrossRef]
- Zhou, G.; Li, C.; Zhang, R.; Zhan, Y.; Lin, L.; Lang, Z.; Tao, Q.; Zheng, J. Kaempferol Inhibits Hepatic Stellate Cell Activation by Regulating miR-26b-5p/Jag1 Axis and Notch Pathway. Front. Pharmacol. 2022, 13, 881855. [Google Scholar] [CrossRef]
- Xu, T.; Huang, S.; Huang, Q.; Ming, Z.; Wang, M.; Li, R.; Zhao, Y. Kaempferol Attenuates Liver Fibrosis by Inhibiting Activin Receptor-like Kinase 5. J. Cell. Mol. Med. 2019, 23, 6403–6410. [Google Scholar] [CrossRef]
- Huang, H.; Wei, S.; Wu, X.; Zhang, M.; Zhou, B.; Huang, D.; Dong, W. Dihydrokaempferol Attenuates CCl4-Induced Hepatic Fibrosis by Inhibiting PARP-1 to Affect Multiple Downstream Pathways and Cytokines. Toxicol. Appl. Pharmacol. 2023, 464, 116438. [Google Scholar] [CrossRef] [PubMed]
- Perumal, N.; Perumal, M.; Halagowder, D.; Sivasithamparam, N. Morin Attenuates Diethylnitrosamine-Induced Rat Liver Fibrosis and Hepatic Stellate Cell Activation by Co-Ordinated Regulation of Hippo/Yap and TGF-Β1/Smad Signaling. Biochimie 2017, 140, 10–19. [Google Scholar] [CrossRef]
- Sang, L.; Wang, X.-M.; Xu, D.-Y.; Sang, L.-X.; Han, Y.; Jiang, L.-Y. Morin Enhances Hepatic Nrf2 Expression in a Liver Fibrosis Rat Model. World J. Gastroenterol. 2017, 23, 8334–8344. [Google Scholar] [CrossRef]
- Yuan, W.; Ahmad, S.; Najar, A. Morin, a Plant Derived Flavonoid, Modulates the Expression of Peroxisome Proliferator-Activated Receptor-γ Coactivator-1α Mediated by AMPK Pathway in Hepatic Stellate Cells. Am. J. Transl. Res. 2017, 9, 5662–5670. [Google Scholar]
- MadanKumar, P.; NaveenKumar, P.; Devaraj, H.; NiranjaliDevaraj, S. Morin, a Dietary Flavonoid, Exhibits Anti-Fibrotic Effect and Induces Apoptosis of Activated Hepatic Stellate Cells by Suppressing Canonical NF-κB Signaling. Biochimie 2015, 110, 107–118. [Google Scholar] [CrossRef] [PubMed]
- Heeba, G.H.; Mahmoud, M.E. Therapeutic Potential of Morin against Liver Fibrosis in Rats: Modulation of Oxidative Stress, Cytokine Production and Nuclear Factor Kappa B. Environ. Toxicol. Pharmacol. 2014, 37, 662–671. [Google Scholar] [CrossRef] [PubMed]
- MadanKumar, P.; NaveenKumar, P.; Manikandan, S.; Devaraj, H.; NiranjaliDevaraj, S. Morin Ameliorates Chemically Induced Liver Fibrosis in Vivo and Inhibits Stellate Cell Proliferation in Vitro by Suppressing Wnt/β-Catenin Signaling. Toxicol. Appl. Pharmacol. 2014, 277, 210–220. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.-S.; Jung, K.H.; Park, I.-S.; Kwon, S.W.; Lee, D.-H.; Hong, S.-S. Protective Effect of Morin on Dimethylnitrosamine-Induced Hepatic Fibrosis in Rats. Dig. Dis. Sci. 2009, 54, 782–788. [Google Scholar] [CrossRef] [PubMed]
- Geng, Y.; Sun, Q.; Li, W.; Lu, Z.-M.; Xu, H.-Y.; Shi, J.-S.; Xu, Z.-H. The Common Dietary Flavonoid Myricetin Attenuates Liver Fibrosis in Carbon Tetrachloride Treated Mice. Mol. Nutr. Food Res. 2017, 61, 1600392. [Google Scholar] [CrossRef]
- Domitrović, R.; Rashed, K.; Cvijanović, O.; Vladimir-Knežević, S.; Škoda, M.; Višnić, A. Myricitrin Exhibits Antioxidant, Anti-Inflammatory and Antifibrotic Activity in Carbon Tetrachloride-Intoxicated Mice. Chem. Biol. Interact. 2015, 230, 21–29. [Google Scholar] [CrossRef]
- Salama, Y.A.; Hassan, H.M.; El-Gayar, A.M.; Abdel-Rahman, N. Combined Quercetin and Simvastatin Attenuate Hepatic Fibrosis in Rats by Modulating SphK1/NLRP3 Pathways. Life Sci. 2024, 337, 122349. [Google Scholar] [CrossRef]
- Aslam, A.; Sheikh, N.; Shahzad, M.; Saeed, G.; Fatima, N.; Akhtar, T. Quercetin Ameliorates Thioacetamide-Induced Hepatic Fibrosis and Oxidative Stress by Antagonizing the Hedgehog Signaling Pathway. J. Cell. Biochem. 2022, 123, 1356–1365. [Google Scholar] [CrossRef]
- Li, X.; Jin, Q.; Yao, Q.; Xu, B.; Li, L.; Zhang, S.; Tu, C. The Flavonoid Quercetin Ameliorates Liver Inflammation and Fibrosis by Regulating Hepatic Macrophages Activation and Polarization in Mice. Front. Pharmacol. 2018, 9, 72. [Google Scholar] [CrossRef]
- Wu, L.; Zhang, Q.; Mo, W.; Feng, J.; Li, S.; Li, J.; Liu, T.; Xu, S.; Wang, W.; Lu, X.; et al. Quercetin Prevents Hepatic Fibrosis by Inhibiting Hepatic Stellate Cell Activation and Reducing Autophagy via the TGF-Β1/Smads and PI3K/Akt Pathways. Sci. Rep. 2017, 7, 9289. [Google Scholar] [CrossRef]
- Li, X.; Jin, Q.; Yao, Q.; Xu, B.; Li, Z.; Tu, C. Quercetin Attenuates the Activation of Hepatic Stellate Cells and Liver Fibrosis in Mice through Modulation of HMGB1-TLR2/4-NF-κB Signaling Pathways. Toxicol. Lett. 2016, 261, 1–12. [Google Scholar] [CrossRef]
- Hernández-Ortega, L.D.; Alcántar-Díaz, B.E.; Ruiz-Corro, L.A.; Sandoval-Rodriguez, A.; Bueno-Topete, M.; Armendariz-Borunda, J.; Salazar-Montes, A.M. Quercetin Improves Hepatic Fibrosis Reducing Hepatic Stellate Cells and Regulating Pro-Fibrogenic/Anti-Fibrogenic Molecules Balance. J. Gastroenterol. Hepatol. 2012, 27, 1865–1872. [Google Scholar] [CrossRef] [PubMed]
- Gürler, S.; Kiraz, Y.; Baran, Y. Chapter 21. Flavonoids in Cancer Therapy: Current and Future Trends. In Biodiversity and Biomedicine; Ozturk, M., Egamberdieva, D., Pešić, M., Eds.; Academic Press: Cambridge, MA, USA, 2020; pp. 403–440. [Google Scholar]
- Hostetler, G.L.; Ralston, R.A.; Schwartz, S.J. Flavones: Food Sources, Bioavailability, Metabolism, and Bioactivity. Adv. Nutr. Bethesda Md 2017, 8, 423–435. [Google Scholar] [CrossRef] [PubMed]
- Hano, C.; Tungmunnithum, D. Plant Polyphenols, More than Just Simple Natural Antioxidants: Oxidative Stress, Aging and Age-Related Diseases. Medicines 2020, 7, 26. [Google Scholar] [CrossRef]
- Zhu, Z.; Hu, R.; Li, J.; Xing, X.; Chen, J.; Zhou, Q.; Sun, J. Alpinetin Exerts Anti-Inflammatory, Anti-Oxidative and Anti-Angiogenic Effects through Activating the Nrf2 Pathway and Inhibiting NLRP3 Pathway in Carbon Tetrachloride-Induced Liver Fibrosis. Int. Immunopharmacol. 2021, 96, 107660. [Google Scholar] [CrossRef] [PubMed]
- Melaibari, M.; Alkreathy, H.M.; Esmat, A.; Rajeh, N.A.; Shaik, R.A.; Alghamdi, A.A.; Ahmad, A. Anti-Fibrotic Efficacy of Apigenin in a Mice Model of Carbon Tetrachloride-Induced Hepatic Fibrosis by Modulation of Oxidative Stress, Inflammation, and Fibrogenesis: A Preclinical Study. Biomedicines 2023, 11, 1342. [Google Scholar] [CrossRef]
- Ji, J.; Yu, Q.; Dai, W.; Wu, L.; Feng, J.; Zheng, Y.; Li, Y.; Guo, C. Apigenin Alleviates Liver Fibrosis by Inhibiting Hepatic Stellate Cell Activation and Autophagy via TGF-Β1/Smad3 and P38/PPARα Pathways. PPAR Res. 2021, 2021, 6651839. [Google Scholar] [CrossRef]
- Qiao, M.; Yang, J.; Zhu, Y.; Zhao, Y.; Hu, J. Transcriptomics and Proteomics Analysis of System-Level Mechanisms in the Liver of Apigenin-Treated Fibrotic Rats. Life Sci. 2020, 248, 117475. [Google Scholar] [CrossRef]
- Wu, X.; Zhi, F.; Lun, W.; Deng, Q.; Zhang, W. Baicalin Inhibits PDGF-BB-Induced Hepatic Stellate Cell Proliferation, Apoptosis, Invasion, Migration and Activation via the miR-3595/ACSL4 Axis. Int. J. Mol. Med. 2018, 41, 1992–2002. [Google Scholar] [CrossRef]
- Qiao, H.; Han, H.; Hong, D.; Ren, Z.; Chen, Y.; Zhou, C. Protective Effects of Baicalin on Carbon Tetrachloride Induced Liver Injury by Activating PPARγ and Inhibiting TGFβ1. Pharm. Biol. 2011, 49, 38–45. [Google Scholar] [CrossRef]
- Sun, H.; Che, Q.-M.; Zhao, X.; Pu, X.-P. Antifibrotic Effects of Chronic Baicalein Administration in a CCl4 Liver Fibrosis Model in Rats. Eur. J. Pharmacol. 2010, 631, 53–60. [Google Scholar] [CrossRef] [PubMed]
- Balta, C.; Ciceu, A.; Herman, H.; Rosu, M.; Boldura, O.M.; Hermenean, A. Dose-Dependent Antifibrotic Effect of Chrysin on Regression of Liver Fibrosis: The Role in Extracellular Matrix Remodeling. Dose-Response Publ. Int. Hormesis Soc. 2018, 16, 1559325818789835. [Google Scholar] [CrossRef] [PubMed]
- Balta, C.; Herman, H.; Boldura, O.M.; Gasca, I.; Rosu, M.; Ardelean, A.; Hermenean, A. Chrysin Attenuates Liver Fibrosis and Hepatic Stellate Cell Activation through TGF-β/Smad Signaling Pathway. Chem. Biol. Interact. 2015, 240, 94–101. [Google Scholar] [CrossRef]
- Hasan, H.F.; Abdel-Rafei, M.K.; Galal, S.M. Diosmin Attenuates Radiation-Induced Hepatic Fibrosis by Boosting PPAR-γ Expression and Hampering miR-17-5p-Activated Canonical Wnt-β-Catenin Signaling. Biochem. Cell Biol. Biochim. Biol. Cell. 2017, 95, 400–414. [Google Scholar] [CrossRef]
- Hu, J.; Liu, Y.; Pan, Z.; Huang, X.; Wang, J.; Cao, W.; Chen, Z. Eupatilin Ameliorates Hepatic Fibrosis and Hepatic Stellate Cell Activation by Suppressing β-Catenin/PAI-1 Pathway. Int. J. Mol. Sci. 2023, 24, 5933. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.-X.; Huang, Q.-F.; Lin, X.; Wei, J.-B. [Protective effect of isoorientin on alcohol-induced hepatic fibrosis in rats]. Zhongguo Zhong Yao Za Zhi Zhongguo Zhongyao Zazhi China J. Chin. Mater. Medica 2013, 38, 3726–3730. [Google Scholar]
- Huang, Q.F.; Zhang, S.J.; Zheng, L.; Liao, M.; He, M.; Huang, R.; Zhuo, L.; Lin, X. Protective Effect of Isoorientin-2″-O-α-L-Arabinopyranosyl Isolated from Gypsophila Elegans on Alcohol Induced Hepatic Fibrosis in Rats. Food Chem. Toxicol. Int. J. Publ. Br. Ind. Biol. Res. Assoc. 2012, 50, 1992–2001. [Google Scholar] [CrossRef]
- Huang, Y.; Luo, W.; Chen, S.; Su, H.; Zhu, W.; Wei, Y.; Qiu, Y.; Long, Y.; Shi, Y.; Wei, J. Isovitexin Alleviates Hepatic Fibrosis by Regulating miR-21-Mediated PI3K/Akt Signaling and Glutathione Metabolic Pathway: Based on Transcriptomics and Metabolomics. Phytomed. Int. J. Phytother. Phytopharm. 2023, 121, 155117. [Google Scholar] [CrossRef]
- Kang, R.; Tian, W.; Cao, W.; Sun, Y.; Zhang, H.-N.; Feng, Y.-D.; Li, C.; Li, Z.-Z.; Li, X.-Q. Ligustroflavone Ameliorates CCl4-Induced Liver Fibrosis through down-Regulating the TGF-β/Smad Signaling Pathway. Chin. J. Nat. Med. 2021, 19, 170–180. [Google Scholar] [CrossRef]
- Batudeligen; Han, Z.; Chen, H.; Narisu; Xu, Y.; Anda; Han, G. Luteolin Alleviates Liver Fibrosis in Rat Hepatic Stellate Cell HSC-T6: A Proteomic Analysis. Drug Des. Devel. Ther. 2023, 17, 1819–1829. [Google Scholar] [CrossRef]
- Li, J.; Li, X.; Xu, W.; Wang, S.; Hu, Z.; Zhang, Q.; Deng, X.; Wang, J.; Zhang, J.; Guo, C. Antifibrotic Effects of Luteolin on Hepatic Stellate Cells and Liver Fibrosis by Targeting AKT/mTOR/p70S6K and TGFβ/Smad Signalling Pathways. Liver Int. Off. J. Int. Assoc. Study Liver 2014, 35, 1222–1233. [Google Scholar] [CrossRef] [PubMed]
- Domitrović, R.; Jakovac, H.; Tomac, J.; Sain, I. Liver Fibrosis in Mice Induced by Carbon Tetrachloride and Its Reversion by Luteolin. Toxicol. Appl. Pharmacol. 2009, 241, 311–321. [Google Scholar] [CrossRef]
- Xiu, M.; Zhao, Y.; Wang, X.; Yuan, S.; Qin, B.; Sun, J.; Cui, L.; Song, J. Regulation of SIRT1-TLR2/TLR4 Pathway in Cell Communication from Macrophages to Hepatic Stellate Cells Contribute to Alleviates Hepatic Fibrosis by Luteoloside. Acta Histochem. 2023, 125, 151989. [Google Scholar] [CrossRef]
- Hao, Y.; Feng, D.; Ye, H.; Liao, W. Nobiletin Alleviated Epithelial-Mesenchymal Transition of Hepatocytes in Liver Fibrosis Based on Autophagy-Hippo/YAP Pathway. Mol. Nutr. Food Res. 2024, 68, e2300529. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Weng, J.; Chen, X.; Ma, S.; Zhang, Y.; Zhang, F.; Zhang, Z.; Wang, F.; Shao, J.; Zheng, S. Oroxylin A Activates Ferritinophagy to Induce Hepatic Stellate Cell Senescence against Hepatic Fibrosis by Regulating cGAS-STING Pathway. Biomed. Pharmacother. Biomed. Pharmacother. 2023, 162, 114653. [Google Scholar] [CrossRef]
- Shen, M.; Guo, M.; Wang, Z.; Li, Y.; Kong, D.; Shao, J.; Tan, S.; Chen, A.; Zhang, F.; Zhang, Z.; et al. ROS-Dependent Inhibition of the PI3K/Akt/mTOR Signaling Is Required for Oroxylin A to Exert Anti-Inflammatory Activity in Liver Fibrosis. Int. Immunopharmacol. 2020, 85, 106637. [Google Scholar] [CrossRef]
- Bian, M.; He, J.; Jin, H.; Lian, N.; Shao, J.; Guo, Q.; Wang, S.; Zhang, F.; Zheng, S. Oroxylin A Induces Apoptosis of Activated Hepatic Stellate Cells through Endoplasmic Reticulum Stress. Apoptosis Int. J. Program. Cell Death 2019, 24, 905–920. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Jia, Y.; Li, M.; Wang, L.; Shao, J.; Guo, Q.; Tan, S.; Ding, H.; Chen, A.; Zhang, F.; et al. Blockade of Glycolysis-Dependent Contraction by Oroxylin a via Inhibition of Lactate Dehydrogenase-a in Hepatic Stellate Cells. Cell Commun. Signal. CCS 2019, 17, 11. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Zhang, Z.; Yao, Z.; Wang, L.; Zhang, F.; Shao, J.; Chen, A.; Zheng, S. Activation of Autophagy Is Required for Oroxylin A to Alleviate Carbon Tetrachloride-Induced Liver Fibrosis and Hepatic Stellate Cell Activation. Int. Immunopharmacol. 2018, 56, 148–155. [Google Scholar] [CrossRef]
- Seki, N.; Toh, U.; Kawaguchi, K.; Ninomiya, M.; Koketsu, M.; Watanabe, K.; Aoki, M.; Fujii, T.; Nakamura, A.; Akagi, Y.; et al. Tricin Inhibits Proliferation of Human Hepatic Stellate Cells in Vitro by Blocking Tyrosine Phosphorylation of PDGF Receptor and Its Signaling Pathways. J. Cell. Biochem. 2012, 113, 2346–2355. [Google Scholar] [CrossRef]
- Du, X.-S.; Li, H.-D.; Yang, X.-J.; Li, J.-J.; Xu, J.-J.; Chen, Y.; Xu, Q.-Q.; Yang, L.; He, C.-S.; Huang, C.; et al. Wogonin Attenuates Liver Fibrosis via Regulating Hepatic Stellate Cell Activation and Apoptosis. Int. Immunopharmacol. 2019, 75, 105671. [Google Scholar] [CrossRef]
- Ma, J.-Q.; Sun, Y.-Z.; Ming, Q.-L.; Tian, Z.-K.; Yang, H.-X.; Liu, C.-M. Ampelopsin Attenuates Carbon Tetrachloride-Induced Mouse Liver Fibrosis and Hepatic Stellate Cell Activation Associated with the SIRT1/TGF-Β1/Smad3 and Autophagy Pathway. Int. Immunopharmacol. 2019, 77, 105984. [Google Scholar] [CrossRef] [PubMed]
- Shi, H.; Shi, H.; Ren, F.; Chen, D.; Chen, Y.; Duan, Z. Naringin in Ganshuang Granule Suppresses Activation of Hepatic Stellate Cells for Anti-Fibrosis Effect by Inhibition of Mammalian Target of Rapamycin. J. Cell. Mol. Med. 2017, 21, 500–509. [Google Scholar] [CrossRef] [PubMed]
- Said, M.M.; Azab, S.S.; Saeed, N.M.; El-Demerdash, E. Antifibrotic Mechanism of Pinocembrin: Impact on Oxidative Stress, Inflammation and TGF-β /Smad Inhibition in Rats. Ann. Hepatol. 2018, 17, 307–317. [Google Scholar] [CrossRef] [PubMed]
- Zhou, F.; Wang, A.; Li, D.; Wang, Y.; Lin, L. Pinocembrin from Penthorum Chinense Pursh Suppresses Hepatic Stellate Cells Activation through a Unified SIRT3-TGF-β-Smad Signaling Pathway. Toxicol. Appl. Pharmacol. 2018, 341, 38–50. [Google Scholar] [CrossRef]
- Zhang, M.; Wang, Y.; Zhu, G.; Sun, C.; Wang, J. Hepatoprotective Effect and Possible Mechanism of Phytoestrogen Calycosin on Carbon Tetrachloride-Induced Liver Fibrosis in Mice. Naunyn. Schmiedebergs Arch. Pharmacol. 2020, 394, 189–204. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Zhang, D.; Yang, H.; Liu, Y.; Zhang, L.; Zhang, C.; Chen, G.; Hu, Y.; Chen, J.; Zhang, H.; et al. Hepatoprotective Effect of Genistein against Dimethylnitrosamine-Induced Liver Fibrosis in Rats by Regulating Macrophage Functional Properties and Inhibiting the JAK2/STAT3/SOCS3 Signaling Pathway. Front. Biosci. Landmark Ed. 2021, 26, 1572–1584. [Google Scholar] [CrossRef]
- Ganai, A.A.; Husain, M. Genistein Attenuates D-GalN Induced Liver Fibrosis/Chronic Liver Damage in Rats by Blocking the TGF-β/Smad Signaling Pathways. Chem. Biol. Interact. 2017, 261, 80–85. [Google Scholar] [CrossRef] [PubMed]
- Huang, Q.; Huang, R.; Zhang, S.; Lin, J.; Wei, L.; He, M.; Zhuo, L.; Lin, X. Protective Effect of Genistein Isolated from Hydrocotyle Sibthorpioides on Hepatic Injury and Fibrosis Induced by Chronic Alcohol in Rats. Toxicol. Lett. 2013, 217, 102–110. [Google Scholar] [CrossRef]
- Salas, A.L.; Montezuma, T.D.; Fariña, G.G.; Reyes-Esparza, J.; Rodríguez-Fragoso, L. Genistein Modifies Liver Fibrosis and Improves Liver Function by Inducing uPA Expression and Proteolytic Activity in CCl4-Treated Rats. Pharmacology 2008, 81, 41–49. [Google Scholar] [CrossRef]
- Zhang, L.; Zhang, H.; Gu, J.; Xu, W.; Yuan, N.; Sun, J.; Li, H. Glabridin Inhibits Liver Fibrosis and Hepatic Stellate Cells Activation through Suppression of Inflammation and Oxidative Stress by Activating PPARγ in Carbon Tetrachloride-Treated Mice. Int. Immunopharmacol. 2022, 113, 109433. [Google Scholar] [CrossRef]
- Li, X.; Zhang, H.; Pan, L.; Zou, H.; Miao, X.; Cheng, J.; Wu, Y. Puerarin Alleviates Liver Fibrosis via Inhibition of the ERK1/2 Signaling Pathway in Thioacetamide-Induced Hepatic Fibrosis in Rats. Exp. Ther. Med. 2019, 18, 133–138. [Google Scholar] [CrossRef] [PubMed]
- Huang, G.-R.; Wei, S.-J.; Huang, Y.-Q.; Xing, W.; Wang, L.-Y.; Liang, L.-L. Mechanism of Combined Use of Vitamin D and Puerarin in Anti-Hepatic Fibrosis by Regulating the Wnt/β-Catenin Signalling Pathway. World J. Gastroenterol. 2018, 24, 4178–4185. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Shi, X.-L.; Feng, M.; Wang, X.; Zhang, Z.-H.; Zhao, X.; Han, B.; Ma, H.-C.; Dai, B.; Ding, Y.-T. Puerarin Protects against CCl4-Induced Liver Fibrosis in Mice: Possible Role of PARP-1 Inhibition. Int. Immunopharmacol. 2016, 38, 238–245. [Google Scholar] [CrossRef] [PubMed]
- Guo, C.; Xu, L.; He, Q.; Liang, T.; Duan, X.; Li, R. Anti-Fibrotic Effects of Puerarin on CCl4-Induced Hepatic Fibrosis in Rats Possibly through the Regulation of PPAR-γ Expression and Inhibition of PI3K/Akt Pathway. Food Chem. Toxicol. Int. J. Publ. Br. Ind. Biol. Res. Assoc. 2013, 56, 436–442. [Google Scholar] [CrossRef] [PubMed]
- Li, R.; Xu, L.; Liang, T.; Li, Y.; Zhang, S.; Duan, X. Puerarin Mediates Hepatoprotection against CCl4-Induced Hepatic Fibrosis Rats through Attenuation of Inflammation Response and Amelioration of Metabolic Function. Food Chem. Toxicol. Int. J. Publ. Br. Ind. Biol. Res. Assoc. 2013, 52, 69–75. [Google Scholar] [CrossRef]
- Xu, L.; Zheng, N.; He, Q.; Li, R.; Zhang, K.; Liang, T. Puerarin, Isolated from Pueraria Lobata (Willd.), Protects against Hepatotoxicity via Specific Inhibition of the TGF-Β1/Smad Signaling Pathway, Thereby Leading to Anti-Fibrotic Effect. Phytomed. Int. J. Phytother. Phytopharm. 2013, 20, 1172–1179. [Google Scholar] [CrossRef]
- Zhang, S.; Ji, G.; Liu, J. Reversal of Chemical-Induced Liver Fibrosis in Wistar Rats by Puerarin. J. Nutr. Biochem. 2006, 17, 485–491. [Google Scholar] [CrossRef]
- Li, J.-F.; Chen, B.-C.; Lai, D.-D.; Jia, Z.-R.; Andersson, R.; Zhang, B.; Yao, J.-G.; Yu, Z. Soy Isoflavone Delays the Progression of Thioacetamide-Induced Liver Fibrosis in Rats. Scand. J. Gastroenterol. 2011, 46, 341–349. [Google Scholar] [CrossRef]
- Gao, X.-X.; Shi, D.-H.; Chen, Y.-X.; Cui, J.-T.; Wang, Y.-R.; Jiang, C.-P.; Wu, J.-H. The Therapeutic Effects of Tectorigenin on Chemically Induced Liver Fibrosis in Rats and an Associated Metabonomic Investigation. Arch. Pharm. Res. 2012, 35, 1479–1493. [Google Scholar] [CrossRef]
- Zhan, W.; Liao, X.; Xie, R.-J.; Tian, T.; Yu, L.; Liu, X.; Liu, J.; Li, P.; Han, B.; Yang, T.; et al. The Effects of Blueberry Anthocyanins on Histone Acetylation in Rat Liver Fibrosis. Oncotarget 2017, 8, 96761–96773. [Google Scholar] [CrossRef]
- Zhan, W.; Liao, X.; Tian, T.; Yu, L.; Liu, X.; Li, B.; Liu, J.; Han, B.; Xie, R.J.; Ji, Q.H.; et al. Study on the Effects of Blueberry Treatment on Histone Acetylation Modification of CCl4-Induced Liver Disease in Rats. Genet. Mol. Res. GMR 2017, 16, 16019188. [Google Scholar] [CrossRef]
- Zhan, W.; Liao, X.; Yu, L.; Tian, T.; Liu, X.; Liu, J.; Cai, L.-J.; Xiao, X.; Xie, R.-J.; Yang, Q. Effects of Blueberries on Migration, Invasion, Proliferation, the Cell Cycle and Apoptosis in Hepatocellular Carcinoma Cells. Biomed. Rep. 2016, 5, 579–584. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Du, J.; Liu, L.; Fan, H.; Yu, Y.; Luo, Y.; Gu, F.; Yu, H.; Liao, X. Anthocyanins Improve Liver Fibrosis in Mice by Regulating the Autophagic Flux Level of Hepatic Stellate Cells by Mmu_circ_0000623. Food Sci. Nutr. 2023, 11, 3002–3018. [Google Scholar] [CrossRef]
- Hao, R.; Gao, J.; Liu, H.; Zhang, C.; Huang, J.; Fan, J.; Wei, J. Preventive Effect and Mechanism of Anthocyanins from Aronia Melanocarpa Elliot on Hepatic Fibrosis Through TGF-β/Smad Signaling Pathway. Cell Biochem. Biophys. 2022, 80, 737–745. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Wu, Y.; Long, C.; He, P.; Gu, J.; Yang, L.; Liang, Y.; Wang, Y. Anthocyanins Isolated from Blueberry Ameliorates CCl4 Induced Liver Fibrosis by Modulation of Oxidative Stress, Inflammation and Stellate Cell Activation in Mice. Food Chem. Toxicol. Int. J. Publ. Br. Ind. Biol. Res. Assoc. 2018, 120, 491–499. [Google Scholar] [CrossRef] [PubMed]
- Jiang, X.; Guo, H.; Shen, T.; Tang, X.; Yang, Y.; Ling, W. Cyanidin-3-O-β-Glucoside Purified from Black Rice Protects Mice against Hepatic Fibrosis Induced by Carbon Tetrachloride via Inhibiting Hepatic Stellate Cell Activation. J. Agric. Food Chem. 2015, 63, 6221–6230. [Google Scholar] [CrossRef]
- Domitrović, R.; Jakovac, H. Antifibrotic Activity of Anthocyanidin Delphinidin in Carbon Tetrachloride-Induced Hepatotoxicity in Mice. Toxicology 2010, 272, 1–10. [Google Scholar] [CrossRef]
- Ma, Y.; Li, Y.; Zhang, H.; Wang, Y.; Wu, C.; Huang, W. Malvidin Induces Hepatic Stellate Cell Apoptosis via the Endoplasmic Reticulum Stress Pathway and Mitochondrial Pathway. Food Sci. Nutr. 2020, 8, 5095–5106. [Google Scholar] [CrossRef]
- Shi, Y.-S.; Li, X.-X.; Li, H.-T.; Zhang, Y. Pelargonidin Ameliorates CCl4-Induced Liver Fibrosis by Suppressing the ROS-NLRP3-IL-1β Axis via Activating the Nrf2 Pathway. Food Funct. 2020, 11, 5156–5165. [Google Scholar] [CrossRef]
- Lee, S.H.; Nan, J.-X.; Zhao, Y.Z.; Woo, S.W.; Park, E.-J.; Kang, T.-H.; Seo, G.S.; Kim, Y.-C.; Sohn, D.H. The Chalcone Butein from Rhus Verniciflua Shows Antifibrogenic Activity. Planta Med. 2003, 69, 990–994. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Tian, H.; Chen, J.; Huang, D.; Ding, F.; Ma, T.; Xi, J.; Wu, C.; Zhang, Y. Isobavachalcone Attenuates Liver Fibrosis via Activation of the Nrf2/HO-1 Pathway in Rats. Int. Immunopharmacol. 2024, 128, 111398. [Google Scholar] [CrossRef] [PubMed]
- Singh, H.; Sidhu, S.; Chopra, K.; Khan, M.U. Hepatoprotective Effect of Trans-Chalcone on Experimentally Induced Hepatic Injury in Rats: Inhibition of Hepatic Inflammation and Fibrosis. Can. J. Physiol. Pharmacol. 2016, 94, 879–887. [Google Scholar] [CrossRef] [PubMed]
- Dorn, C.; Heilmann, J.; Hellerbrand, C. Protective Effect of Xanthohumol on Toxin-Induced Liver Inflammation and Fibrosis. Int. J. Clin. Exp. Pathol. 2012, 5, 29–36. [Google Scholar] [CrossRef]
- Dorn, C.; Kraus, B.; Motyl, M.; Weiss, T.S.; Gehrig, M.; Schölmerich, J.; Heilmann, J.; Hellerbrand, C. Xanthohumol, a Chalcon Derived from Hops, Inhibits Hepatic Inflammation and Fibrosis. Mol. Nutr. Food Res. 2010, 54 (Suppl. S2), S205–S213. [Google Scholar] [CrossRef]
- Le, C.T.; Nguyen, G.; Park, S.Y.; Dong, H.N.; Cho, Y.K.; Lee, J.-H.; Im, S.-S.; Choi, D.-H.; Cho, E.-H. Phloretin Ameliorates Succinate-Induced Liver Fibrosis by Regulating Hepatic Stellate Cells. Endocrinol. Metab. 2023, 38, 395–405. [Google Scholar] [CrossRef]
- Ye, L.; Yu, Y.; Zhao, Y. Icariin-Induced miR-875-5p Attenuates Epithelial-Mesenchymal Transition by Targeting Hedgehog Signaling in Liver Fibrosis. J. Gastroenterol. Hepatol. 2020, 35, 482–491. [Google Scholar] [CrossRef]
- Algandaby, M.M.; Breikaa, R.M.; Eid, B.G.; Neamatallah, T.A.; Abdel-Naim, A.B.; Ashour, O.M. Icariin Protects against Thioacetamide-Induced Liver Fibrosis in Rats: Implication of Anti-Angiogenic and Anti-Autophagic Properties. Pharmacol. Rep. PR 2017, 69, 616–624. [Google Scholar] [CrossRef]
- Li, J.; Liu, P.; Zhang, R.; Cao, L.; Qian, H.; Liao, J.; Xu, W.; Wu, M.; Yin, Z. Icaritin Induces Cell Death in Activated Hepatic Stellate Cells through Mitochondrial Activated Apoptosis and Ameliorates the Development of Liver Fibrosis in Rats. J. Ethnopharmacol. 2011, 137, 714–723. [Google Scholar] [CrossRef]
- Izzo, C.; Annunziata, M.; Melara, G.; Sciorio, R.; Dallio, M.; Masarone, M.; Federico, A.; Persico, M. The Role of Resveratrol in Liver Disease: A Comprehensive Review from In Vitro to Clinical Trials. Nutrients 2021, 13, 933. [Google Scholar] [CrossRef]
- Di Pascoli, M.; Diví, M.; Rodríguez-Vilarrupla, A.; Rosado, E.; Gracia-Sancho, J.; Vilaseca, M.; Bosch, J.; García-Pagán, J.C. Resveratrol Improves Intrahepatic Endothelial Dysfunction and Reduces Hepatic Fibrosis and Portal Pressure in Cirrhotic Rats. J. Hepatol. 2013, 58, 904–910. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.-Q.; Sun, P.; Jin, Q.; Li, X.; Zhang, Y.; Zhang, Y.-J.; Wu, Y.-L.; Nan, J.-X.; Lian, L.-H. Resveratrol Regulates Activated Hepatic Stellate Cells by Modulating NF-κB and the PI3K/Akt Signaling Pathway. J. Food Sci. 2016, 81, H240–H245. [Google Scholar] [CrossRef] [PubMed]
- Zhu, L.; Mou, Q.; Wang, Y.; Zhu, Z.; Cheng, M. Resveratrol Contributes to the Inhibition of Liver Fibrosis by Inducing Autophagy via the microRNA-20a-mediated Activation of the PTEN/PI3K/AKT Signaling Pathway. Int. J. Mol. Med. 2020, 46, 2035–2046. [Google Scholar] [CrossRef] [PubMed]
- Abdu, S.B.; Al-Bogami, F.M. Influence of Resveratrol on Liver Fibrosis Induced by Dimethylnitrosamine in Male Rats. Saudi J. Biol. Sci. 2019, 26, 201–209. [Google Scholar] [CrossRef]
- Lee, E.-S.; Shin, M.-O.; Yoon, S.; Moon, J.-O. Resveratrol Inhibits Dimethylnitrosamine-Induced Hepatic Fibrosis in Rats. Arch. Pharm. Res. 2010, 33, 925–932. [Google Scholar] [CrossRef] [PubMed]
- Hong, S.-W.; Jung, K.H.; Zheng, H.-M.; Lee, H.-S.; Suh, J.-K.; Park, I.-S.; Lee, D.-H.; Hong, S.-S. The Protective Effect of Resveratrol on Dimethylnitrosamine-Induced Liver Fibrosis in Rats. Arch. Pharm. Res. 2010, 33, 601–609. [Google Scholar] [CrossRef]
- Li, S.; Han, B.; Li, J.; Lv, Z.; Jiang, H.; Liu, Y.; Yang, X.; Lu, J.; Zhang, Z. Resveratrol Alleviates Liver Fibrosis Induced by Long-Term Inorganic Mercury Exposure through Activating the Sirt1/PGC-1α Signaling Pathway. J. Agric. Food Chem. 2024, 72, 15985–15997. [Google Scholar] [CrossRef]
- Chai, Y.-L.; Zhang, H.-N.; Gao, Y.; Li, D.-H.; Zhang, H. A Study on the Protective Impact of Resveratrol on Liver Damage in Rats with Obstructive Jaundice. Comb. Chem. High Throughput Screen. 2024, 27, 1–9. [Google Scholar] [CrossRef]
- Hung, W.-L.; Hsiao, Y.-T.; Chiou, Y.-S.; Nagabhushanam, K.; Ho, C.-T.; Pan, M.-H. Hepatoprotective Effect of Piceatannol against Carbon Tetrachloride-Induced Liver Fibrosis in Mice. Food Funct. 2021, 12, 11229–11240. [Google Scholar] [CrossRef]
- Lee, M.-F.; Liu, M.-L.; Cheng, A.-C.; Tsai, M.-L.; Ho, C.-T.; Liou, W.-S.; Pan, M.-H. Pterostilbene Inhibits Dimethylnitrosamine-Induced Liver Fibrosis in Rats. Food Chem. 2013, 138, 802–807. [Google Scholar] [CrossRef]
- Shi, B.; Qian, J.; Miao, H.; Zhang, S.; Hu, Y.; Liu, P.; Xu, L. Mulberroside A Ameliorates CCl4-Induced Liver Fibrosis in Mice via Inhibiting pro-Inflammatory Response. Food Sci. Nutr. 2023, 11, 3433–3441. [Google Scholar] [CrossRef] [PubMed]
- Chauhan, N.; Dilbaghi, N.; Gopal, M.; Kumar, R.; Kim, K.-H.; Kumar, S. Development of Chitosan Nanocapsules for the Controlled Release of Hexaconazole. Int. J. Biol. Macromol. 2017, 97, 616–624. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Bhanjana, G.; Verma, R.K.; Dhingra, D.; Dilbaghi, N.; Kim, K.-H. Metformin-Loaded Alginate Nanoparticles as an Effective Antidiabetic Agent for Controlled Drug Release. J. Pharm. Pharmacol. 2017, 69, 143–150. [Google Scholar] [CrossRef]
- Kumar, S.; Bhanjana, G.; Kumar, A.; Taneja, K.; Dilbaghi, N.; Kim, K.-H. Synthesis and Optimization of Ceftriaxone-Loaded Solid Lipid Nanocarriers. Chem. Phys. Lipids 2016, 200, 126–132. [Google Scholar] [CrossRef]
- Kumar, S.; Dilbaghi, N.; Saharan, R.; Bhanjana, G. Nanotechnology as Emerging Tool for Enhancing Solubility of Poorly Water-Soluble Drugs. BioNanoScience 2012, 2, 227–250. [Google Scholar] [CrossRef]
- De Jong, W.H.; Borm, P.J.A. Drug Delivery and Nanoparticles:Applications and Hazards. Int. J. Nanomed. 2008, 3, 133–149. [Google Scholar] [CrossRef]
- Hashemi, M.; Ebrahimian, M. Recent Advances in Nanoformulations for Co-Delivery of Curcumin and Chemotherapeutic Drugs. Nanomedi. J. 2017, 4, 1–7. [Google Scholar] [CrossRef]
- Patel, T.; Zhou, J.; Piepmeier, J.M.; Saltzman, W.M. Polymeric Nanoparticles for Drug Delivery to the Central Nervous System. Adv. Drug Deliv. Rev. 2012, 64, 701–705. [Google Scholar] [CrossRef]
- Kang, L.; Gao, Z.; Huang, W.; Jin, M.; Wang, Q. Nanocarrier-Mediated Co-Delivery of Chemotherapeutic Drugs and Gene Agents for Cancer Treatment. Acta Pharm. Sin. B 2015, 5, 169–175. [Google Scholar] [CrossRef]
- Afsharzadeh, M.; Hashemi, M.; Mokhtarzadeh, A.; Abnous, K.; Ramezani, M. Recent Advances in Co-Delivery Systems Based on Polymeric Nanoparticle for Cancer Treatment. Artif. Cells Nanomed. Biotechnol. 2018, 46, 1095–1110. [Google Scholar] [CrossRef]
- Pawar, A.; Rajalakshmi, S.; Mehta, P.; Shaikh, K.; Bothiraja, C. Strategies for Formulation Development of Andrographolide. RSC Adv. 2016, 6, 69282–69300. [Google Scholar] [CrossRef]
- Rajalakshmi, S.; Vyawahare, N.; Pawar, A.; Mahaparale, P.; Chellampillai, B. Current Development in Novel Drug Delivery Systems of Bioactive Molecule Plumbagin. Artif. Cells Nanomed. Biotechnol. 2018, 46, 209–218. [Google Scholar] [CrossRef] [PubMed]
- Jain, A.K.; Thareja, S. In Vitro and in Vivo Characterization of Pharmaceutical Nanocarriers Used for Drug Delivery. Artif. Cells Nanomed. Biotechnol. 2019, 47, 524–539. [Google Scholar] [CrossRef] [PubMed]
- Oroojan, A.A.; Ahangarpour, A.; Paknejad, B.; Zareian, P.; Hami, Z.; Abtahi, S.R. Effects of Myricitrin and Solid Lipid Nanoparticle-Containing Myricitrin on Reproductive System Disorders Induced by Diabetes in Male Mouse. World J. Mens Health 2021, 39, 147–157. [Google Scholar] [CrossRef]
- Jain, N.; Jain, R.; Thakur, N.; Gupta, B.; Jain, D.; Banveer, J.; Jain, S. Nanotechnology: A Seif and Effective Drug Delivery System. Asian J. Pharm. Clin. Res. 2010, 3, 159–165. [Google Scholar]
- Bilia, A.R.; Isacchi, B.; Righeschi, C.; Guccione, C.; Bergonzi, M.C. Flavonoids Loaded in Nanocarriers: An Opportunity to Increase Oral Bioavailability and Bioefficacy. Food Nutr. Sci. 2014, 5, 1212–1327. [Google Scholar] [CrossRef]
- Leonarduzzi, G.; Testa, G.; Sottero, B.; Gamba, P.; Poli, G. Design and Development of Nanovehicle-Based Delivery Systems for Preventive or Therapeutic Supplementation with Flavonoids. Curr. Med. Chem. 2010, 17, 74–95. [Google Scholar] [CrossRef]
- Liechty, W.B.; Kryscio, D.R.; Slaughter, B.V.; Peppas, N.A. Polymers for Drug Delivery Systems. Annu. Rev. Chem. Biomol. Eng. 2010, 1, 149–173. [Google Scholar] [CrossRef]
- Danhier, F.; Ansorena, E.; Silva, J.M.; Coco, R.; Le Breton, A.; Préat, V. PLGA-Based Nanoparticles: An Overview of Biomedical Applications. J. Control. Release Off. J. Control. Release Soc. 2012, 161, 505–522. [Google Scholar] [CrossRef]
- Sanna, V.; Roggio, A.M.; Posadino, A.M.; Cossu, A.; Marceddu, S.; Mariani, A.; Alzari, V.; Uzzau, S.; Pintus, G.; Sechi, M. Novel Docetaxel-Loaded Nanoparticles Based on Poly(Lactide-Co-Caprolactone) and Poly(Lactide-Co-Glycolide-Co-Caprolactone) for Prostate Cancer Treatment: Formulation, Characterization, and Cytotoxicity Studies. Nanoscale Res. Lett. 2011, 6, 260. [Google Scholar] [CrossRef]
- Avgoustakis, K. Pegylated Poly(Lactide) and Poly(Lactide-Co-Glycolide) Nanoparticles: Preparation, Properties and Possible Applications in Drug Delivery. Curr. Drug Deliv. 2004, 1, 321–333. [Google Scholar] [CrossRef] [PubMed]
- Langroodi, F.A.; Hafezi Ghahestani, Z.; Alibolandi, M.; Ebrahimian, M.; Hashemi, M. Evaluation of the Effect of Crocetin on Antitumor Activity of Doxorubicin Encapsulated in PLGA Nanoparticles. Nanomed. J. 2016, 3, 23–34. [Google Scholar] [CrossRef]
- Cheng, J.; Teply, B.A.; Sherifi, I.; Sung, J.; Luther, G.; Gu, F.X.; Levy-Nissenbaum, E.; Radovic-Moreno, A.F.; Langer, R.; Farokhzad, O.C. Formulation of Functionalized PLGA-PEG Nanoparticles for in Vivo Targeted Drug Delivery. Biomaterials 2007, 28, 869–876. [Google Scholar] [CrossRef] [PubMed]
- Panyam, J.; Sahoo, S.K.; Prabha, S.; Bargar, T.; Labhasetwar, V. Fluorescence and Electron Microscopy Probes for Cellular and Tissue Uptake of Poly(D,L-Lactide-Co-Glycolide) Nanoparticles. Int. J. Pharm. 2003, 262, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Sah, H.; Thoma, L.A.; Desu, H.R.; Sah, E.; Wood, G.C. Concepts and Practices Used to Develop Functional PLGA-Based Nanoparticulate Systems. Int. J. Nanomed. 2013, 8, 747–765. [Google Scholar] [CrossRef] [PubMed]
- Gref, R.; Minamitake, Y.; Peracchia, M.T.; Trubetskoy, V.; Torchilin, V.; Langer, R. Biodegradable Long-Circulating Polymeric Nanospheres. Science 1994, 263, 1600–1603. [Google Scholar] [CrossRef]
- Panyam, J.; Zhou, W.-Z.; Prabha, S.; Sahoo, S.K.; Labhasetwar, V. Rapid Endo-Lysosomal Escape of Poly(DL-Lactide-Co-Glycolide) Nanoparticles: Implications for Drug and Gene Delivery. FASEB J. Off. Publ. Fed. Am. Soc. Exp. Biol. 2002, 16, 1217–1226. [Google Scholar] [CrossRef]
- Acharya, S.; Sahoo, S.K. PLGA Nanoparticles Containing Various Anticancer Agents and Tumour Delivery by EPR Effect. Adv. Drug Deliv. Rev. 2011, 63, 170–183. [Google Scholar] [CrossRef]
- Owens, D.E.; Peppas, N.A. Opsonization, Biodistribution, and Pharmacokinetics of Polymeric Nanoparticles. Int. J. Pharm. 2006, 307, 93–102. [Google Scholar] [CrossRef]
- Betancourt, T.; Byrne, J.D.; Sunaryo, N.; Crowder, S.W.; Kadapakkam, M.; Patel, S.; Casciato, S.; Brannon-Peppas, L. PEGylation Strategies for Active Targeting of PLA/PLGA Nanoparticles. J. Biomed. Mater. Res. A 2009, 91, 263–276. [Google Scholar] [CrossRef]
- Sinha, V.R.; Bansal, K.; Kaushik, R.; Kumria, R.; Trehan, A. Poly-Epsilon-Caprolactone Microspheres and Nanospheres: An Overview. Int. J. Pharm. 2004, 278, 1–23. [Google Scholar] [CrossRef]
- Dash, T.K.; Konkimalla, V.B. Polymeric Modification and Its Implication in Drug Delivery: Poly-ε-Caprolactone (PCL) as a Model Polymer. Mol. Pharm. 2012, 9, 2365–2379. [Google Scholar] [CrossRef]
- Xiong, Y.; Wu, B.; Guo, X.; Shi, D.; Xia, H.; Xu, H.; Liu, X. Galangin Delivered by Retinoic Acid-Modified Nanoparticles Targeted Hepatic Stellate Cells for the Treatment of Hepatic Fibrosis. RSC Adv. 2023, 13, 10987–11001. [Google Scholar] [CrossRef]
- Zhang, Q.; Xu, D.; Guo, Q.; Shan, W.; Yang, J.; Lin, T.; Ye, S.; Zhou, X.; Ge, Y.; Bi, S.; et al. Theranostic Quercetin Nanoparticle for Treatment of Hepatic Fibrosis. Bioconjug. Chem. 2019, 30, 2939–2946. [Google Scholar] [CrossRef] [PubMed]
- Ciceu, A.; Balta, C.; Herman, H.; Gharbia, S.; Ignat, S.-R.; Dinescu, S.; Váradi, J.; Fenyvesi, F.; Gyöngyösi, S.; Hermenean, A.; et al. Complexation with Random Methyl-β-Cyclodextrin and (2-Hidroxypropyl)-β-Cyclodextrin Enhances In Vivo Anti-Fibrotic and Anti-Inflammatory Effects of Chrysin via the Inhibition of NF-κB and TGF-Β1/Smad Signaling Pathways and Modulation of Hepatic Pro/Anti-Fibrotic miRNA. Int. J. Mol. Sci. 2021, 22, 1869. [Google Scholar] [CrossRef] [PubMed]
- Ignat, S.-R.; Dinescu, S.; Váradi, J.; Fenyvesi, F.; Nguyen, T.L.P.; Ciceu, A.; Hermenean, A.; Costache, M. Complexation with Random Methyl-β-Cyclodextrin and (2-Hydroxypropyl)-β-Cyclodextrin Promotes Chrysin Effect and Potential for Liver Fibrosis Therapy. Materials 2020, 13, 5003. [Google Scholar] [CrossRef] [PubMed]
- Ashour, A.A.; El-Kamel, A.H.; Mehanna, R.A.; Mourad, G.; Heikal, L.A. Luteolin-Loaded Exosomes Derived from Bone Marrow Mesenchymal Stem Cells: A Promising Therapy for Liver Fibrosis. Drug Deliv. 2022, 29, 3270–3280. [Google Scholar] [CrossRef]
- Morsy, M.A.; Nair, A.B. Prevention of Rat Liver Fibrosis by Selective Targeting of Hepatic Stellate Cells Using Hesperidin Carriers. Int. J. Pharm. 2018, 552, 241–250. [Google Scholar] [CrossRef]
- Yang, F.; Hu, S.; Sheng, X.; Liu, Y. Naringenin Loaded Multifunctional Nanoparticles to Enhance the Chemotherapeutic Efficacy in Hepatic Fibrosis. Biomed. Microdevices 2020, 22, 68. [Google Scholar] [CrossRef]
- Hermenean, A.; Ardelean, A.; Stan, M.; Hadaruga, N.; Mihali, C.-V.; Costache, M.; Dinischiotu, A. Antioxidant and Hepatoprotective Effects of Naringenin and Its β-Cyclodextrin Formulation in Mice Intoxicated with Carbon Tetrachloride: A Comparative Study. J. Med. Food 2014, 17, 670–677. [Google Scholar] [CrossRef]
- Luo, J.; Zhang, Z.; Zeng, Y.; Dong, Y.; Ma, L. Co-Encapsulation of Collagenase Type I and Silibinin in Chondroitin Sulfate Coated Multilayered Nanoparticles for Targeted Treatment of Liver Fibrosis. Carbohydr. Polym. 2021, 263, 117964. [Google Scholar] [CrossRef] [PubMed]
- Yang, Q.; Tan, T.; He, Q.; Guo, C.; Chen, D.; Tan, Y.; Feng, J.; Song, X.; Gong, T.; Li, J. Combined Amphiphilic Silybin Meglumine Nanosuspension Effective Against Hepatic Fibrosis in Mice Model. Int. J. Nanomed. 2023, 18, 5197–5211. [Google Scholar] [CrossRef]
- Abdullah, A.S.; Sayed, I.E.T.E.; El-Torgoman, A.M.A.; Kalam, A.; Wageh, S.; Kamel, M.A. Green Synthesis of Silymarin-Chitosan Nanoparticles as a New Nano Formulation with Enhanced Anti-Fibrotic Effects against Liver Fibrosis. Int. J. Mol. Sci. 2022, 23, 5420. [Google Scholar] [CrossRef]
- Gharbia, S.; Balta, C.; Herman, H.; Rosu, M.; Váradi, J.; Bácskay, I.; Vecsernyés, M.; Gyöngyösi, S.; Fenyvesi, F.; Voicu, S.N.; et al. Enhancement of Silymarin Anti-Fibrotic Effects by Complexation With Hydroxypropyl (HPBCD) and Randomly Methylated (RAMEB) β-Cyclodextrins in a Mouse Model of Liver Fibrosis. Front. Pharmacol. 2018, 9, 883. [Google Scholar] [CrossRef]
- Younis, N.; Shaheen, M.A.; Abdallah, M.H. Silymarin-Loaded Eudragit(®) RS100 Nanoparticles Improved the Ability of Silymarin to Resolve Hepatic Fibrosis in Bile Duct Ligated Rats. Biomed. Pharmacother. Biomed. Pharmacother. 2016, 81, 93–103. [Google Scholar] [CrossRef] [PubMed]
- Elzoheiry, A.; Ayad, E.; Omar, N.; Elbakry, K.; Hyder, A. Anti-Liver Fibrosis Activity of Curcumin/Chitosan-Coated Green Silver Nanoparticles. Sci. Rep. 2022, 12, 18403. [Google Scholar] [CrossRef] [PubMed]
- Negm, M.; Ahmed, N.; Barakat, L. The Role of Curcumin—Chitosan Nanoparticles in the Prevention and Treatment of Liver Fibrosis in Mice. Alfarama J. Basic Appl. Sci. 2022, 3, 8–28. [Google Scholar] [CrossRef]
- Zhang, L.; Pan, X.; Xu, L.; Zhang, L.; Huang, H. Mitochondria-Targeted Curcumin Loaded CTPP-PEG-PCL Self-Assembled Micelles for Improving Liver Fibrosis Therapy. RSC Adv. 2021, 11, 5348–5360. [Google Scholar] [CrossRef]
- Wang, J.; Pan, W.; Wang, Y.; Lei, W.; Feng, B.; Du, C.; Wang, X.-J. Enhanced Efficacy of Curcumin with Phosphatidylserine-Decorated Nanoparticles in the Treatment of Hepatic Fibrosis. Drug Deliv. 2018, 25, 1–11. [Google Scholar] [CrossRef]
- Algandaby, M.M.; Al-Sawahli, M.M.; Ahmed, O.A.A.; Fahmy, U.A.; Abdallah, H.M.; Hattori, M.; Ashour, O.M.; Abdel-Naim, A.B. Curcumin-Zein Nanospheres Improve Liver Targeting and Antifibrotic Activity of Curcumin in Carbon Tetrachloride-Induced Mice Liver Fibrosis. J. Biomed. Nanotechnol. 2016, 12, 1746–1757. [Google Scholar] [CrossRef]
- Singh, N.; Khullar, N.; Kakkar, V.; Kaur, I.P. Attenuation of Carbon Tetrachloride-Induced Hepatic Injury with Curcumin-Loaded Solid Lipid Nanoparticles. BioDrugs Clin. Immunother. Biopharm. Gene Ther. 2014, 28, 297–312. [Google Scholar] [CrossRef] [PubMed]

| Bioactive Compounds | Cell Lines/ Animal Model | Pharmacological Effects | Reference |
|---|---|---|---|
| Epigallocatechin-3-gallate (EGCG) | Wistar rats |
| [51] |
| HepG2 cells Sprague-Dawley rats |
| [52] | |
| Human hepatic stellate LX-2 cells Sprague-Dawley rats |
| [53] | |
| C57BL/b6 mice |
| [54] | |
| Wistar rats |
| [55] | |
| Sprague-Dawley rats Rat HSCs |
| [56] | |
| Dihydromyricetin | ICR mice |
| [57] |
| LX2 and NK92 cells C57BL/6 J mice |
| [58] | |
| Hesperetin | C57BL/6J mice HSC-T6 cells |
| [59] |
| Hesperetin derivative | LX-2 cells C57BL/6J mice |
| [60] |
| Hesperetin derivative | C57BL/6J mice HSC-T6 cells |
| [61] |
| Hesperetin derivative-7 | KunMing mice HSC-T6 cells |
| [62] |
| Hesperetin derivative (HD-11) | Sprague-Dawley rats HSC-T6 cells |
| [63] |
| Hesperetin derivative-16 (HD-16) | LX-2 cells C57BL/6J mice |
| [64] |
| Hesperidin | Wistar rats |
| [65] |
| Wistar rats |
| [66] | |
| Liquiritigenin | C57BL/6J mice Primary HSCs |
| [67] |
| C57BL/6 mice LX-2 cells |
| [68] | |
| Naringenin | C57BL/6J mice Human LX2 and L02 cells |
| [69] |
| Wistar rats |
| [70] | |
| Wistar rats |
| [71] | |
| Naringin | Sprague-Dawley rats |
| [72] |
| Bioactive Compounds | Cell Lines/ Animal Model | Pharmacological Effects | Reference |
|---|---|---|---|
| Fisetin | Albino Wistar rats |
| [80] |
| Galangin | LX-2 cells |
| [81] |
| Sprague-Dawley rats |
| [82] | |
| Isorhamnetin | HSC-T6 cells |
| [83] |
| C57 mice |
| [84] | |
| ICR mice LX-2 cells |
| [85] | |
| Kaempferol | Sprague-Dawley rats HSC-T6 cells |
| [86] |
| C57BL/6 J mice Primary HSCs |
| [87] | |
| C57BL/6 mice Primary HSCs |
| [88] | |
| Dihydrokaempferol | C57BL/6 mice HepG2 and LX-2 cells |
| [89] |
| Morin | LX-2 cells Wistar rats |
| [90] |
| Sprague-Dawley rats |
| [91] | |
| Sprague-Dawley rats Cultured HSCs |
| [92] | |
| LX-2 cells Wistar rats |
| [93] | |
| Albino rats |
| [94] | |
| LX-2 cells Albino rats |
| [95] | |
| Sprague-Dawley rats |
| [96] | |
| Myricetin | BALB/c mice CFSC-8B cells |
| [97] |
| Myricitrin | BALB/c mice |
| [98] |
| Quercetin | Sprague-Dawley rats |
| [99] |
| Wistar rats |
| [100] | |
| BALB/c mice Raw 264.7 cells |
| [101] | |
| C57 mice |
| [102] | |
| BALB/c mice RHSteC cells |
| [103] | |
| Wistar rats |
| [104] |
| Bioactive Compounds | Cell Lines/ Animal Model | Pharmacological Effects | Reference |
|---|---|---|---|
| Alpinetin | C57BL/6 mice |
| [108] |
| Apigenin | Swiss albino mice |
| [109] |
| LX-2 cells C57 mice |
| [110] | |
| Wistar rats |
| [111] | |
| Baicalin | HSC-T6 cells |
| [112] |
| Sprague-Dawley |
| [113] | |
| Baicalein | Sprague-Dawley rats |
| [114] |
| Chrysin | CD1 mice |
| [115] |
| CD1 mice |
| [116] | |
| Diosmin | Albino rats |
| [117] |
| Eupatilin | LX-2 cells c57BL/6J mice |
| [118] |
| Isoorientin | Wistar rats |
| [119] |
| Isoorientin-2″-O-α-L-arabinopyranosyl | Wistar rats |
| [120] |
| Isovitexin | C57BL/6 mice LX2 and JS-1 cells |
| [121] |
| Ligustroflavone | C57BL/6J mice LX-2 cells |
| [122] |
| Luteolin | HSC-T6 cells Wistar rats |
| [123] |
| HSC-T6 cells Sprague-Dawley rats |
| [124] | |
| Balb/c mice |
| [125] | |
| Luteoloside | HSC-T6 cells |
| [126] |
| Nobiletin | C57/BL6J mice |
| [127] |
| Oroxylin A | LX-2 cells ICR mice |
| [128] |
| C57BL/6 mice Primary HSCs |
| [129] | |
| HSC-T6 cells ICR mice |
| [130] | |
| LX2 and LO2 cells ICR mice |
| [131] | |
| ICR mice Primary mouse HSCs |
| [132] | |
| Tricin | LI90 cells |
| [133] |
| Wogonin | C57BL/6 mice T6, LX-2 cells |
| [134] |
| Bioactive Compounds | Cell Lines/ Animal Model | Pharmacological Effects | Reference |
|---|---|---|---|
| Ampelopsin | ICR mice Primary mouse HSC cells |
| [135] |
| Naringin | Balb/c mice Primary mouse HSCs |
| [136] |
| Pinocembrin | Wistar rats |
| [137] |
| Human immortalized HSC LX-2 cells Rat immortalized HSCs HSC-T6 cells |
| [138] |
| Bioactive Compounds | Cell Lines/ Animal Model | Pharmacological Effects | Reference |
|---|---|---|---|
| Calycosin | C57BL/6 mice |
| [139] |
| Genistein | LX2 cells Wistar rats |
| [140] |
| Wistar rats |
| [141] | |
| SPF-Wistar rats |
| [142] | |
| Wistar rats |
| [143] | |
| Glabridin | JS1 cells C57BL/6 mice |
| [144] |
| Puerarin | Sprague-Dawley rats |
| [145] |
| Wistar rats |
| [146] | |
| C57BL/6J mice |
| [147] | |
| Wistar rats |
| [148] | |
| Wistar rats |
| [149] | |
| Sprague-Dawley rats HSC-T6 cells |
| [150] | |
| Wistar rats |
| [151] | |
| Soy isoflavone | Sprague-Dawley rats |
| [152] |
| Tectorigenin | Sprague-Dawley rats |
| [153] |
| Bioactive Compounds | Cell Lines/ Animal Model | Pharmacological Effects | Reference |
|---|---|---|---|
| Anthocyanins | C57BL/6J mice Mice HSC cell line |
| [157] |
| Anthocyanins from Aronia Melanocarpa Elliot | HSC-T6 cells |
| [158] |
| Anthocyanins from blueberry | C57BL/6J mice |
| [159] |
| HSC-T6 cells SD rats |
| [154] | |
| Cyanidin-3-O-β-glucoside | C57BL mice |
| [160] |
| Delphinidin | Balb/C mice |
| [161] |
| Malvidin | HSC-T6 cells |
| [162] |
| Pelargonidin | C57BL/6J mice LX-2 cells |
| [163] |
| Class of Chalcones | Bioactive Compounds | Cell Lines/ Animal Model | Pharmacological Effects | Reference |
|---|---|---|---|---|
| Chalcones | Butein | Sprague-Dawley rats |
| [164] |
| Isobavachalcone | SPF-grade SD rats HSC-T6 cells |
| [165] | |
| Trans-chalcone | Wistar rats |
| [166] | |
| Prenylated chalcones | Xanthohumol | BALB/c mice |
| [167] |
| Primary human hepatocytes (PHHs) and HSCS BALB/c mice |
| [168] | ||
| Dihydrochalcones | Phloretin | LX-2 cells C57BJ6 mice |
| [169] |
| Icariin | Mouse primary HSCs C57BL/6 J mice |
| [170] | |
| Wistar rats |
| [171] | ||
| Icaritin | Wistar rats HSC-T6 and LX-2 |
| [172] |
| Polyphenolic Compounds | Active Ingredient | Formulations | Experimental Model | Pharmacological Effects | Reference |
|---|---|---|---|---|---|
| Flavonols | Galangin | Galangin delivered by retinoic acid-modified nanoparticles | C57BL/6 mice |
| [216] |
| Quercetin | Theranostic quercetin nanoparticle | BABL-c mice HSC cells |
| [217] | |
| Flavones | Chrysin | CHR-HPBCD, CHR-RAMEB nanocomplexes | CD1 mice |
| [218] |
| CHR-HPBCD, CHR-RAMEB complexes | Huh7 and LX2 cells |
| [219] | ||
| Luteolin | Luteolin-loaded exosomes derived from bone marrow MSCs | Sprague-Dawley rats |
| [220] | |
| Flavanones | Hesperidin | Hesperidin-loaded liposomes | Wistar rats |
| [221] |
| Naringenin | Naringenin-loaded multifunctional nanoparticles | Wistar albino rats |
| [222] | |
| Naringenin and its β-cyclodextrin formulation | Swiss mice |
| [223] | ||
| Flavonolignans | Silibinin | Collagenase type I and silibinin in chondroitin sulfate-coated multilayered nanoparticles | Kunming mice |
| [224] |
| Silybin | Combined amphiphilic silybin meglumine nanosuspension | HepG2, LO2, LX-2, and RAW264.7 cells Kunming mice |
| [225] | |
| Sylimarin | Silymarin-chitosan nanoparticles | Sprague-Dawley rats |
| [226] | |
| Sylimarin–HPBCD and Sylimarin–RAMEB complexes | CD1 mice |
| [227] | ||
| Silymarin-loaded Eudragit® RS100 nanoparticles | Wistar albino rats |
| [228] | ||
| Curcuminoids | Curcumin | Curcumin/chitosan-coated green-silver nanoparticles | Mice |
| [229] |
| Curcumin–chitosan nanoparticles | Albino mice |
| [230] | ||
| Curcumin-loaded CTPP-PEG-PCL self-assembled micelles | Sprague-Dawley rats and Balb/c mice |
| [231] | ||
| Curcumin with phosphatidylserine-decorated nanoparticles | Sprague-Dawley rats |
| [232] | ||
| Curcumin-zein nanospheres | SWR mice |
| [233] | ||
| Curcumin-loaded solid lipid nanoparticles | Wistar rats |
| [234] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ciceu, A.; Fenyvesi, F.; Hermenean, A.; Ardelean, S.; Dumitra, S.; Puticiu, M. Advancements in Plant-Based Therapeutics for Hepatic Fibrosis: Molecular Mechanisms and Nanoparticulate Drug Delivery Systems. Int. J. Mol. Sci. 2024, 25, 9346. https://doi.org/10.3390/ijms25179346
Ciceu A, Fenyvesi F, Hermenean A, Ardelean S, Dumitra S, Puticiu M. Advancements in Plant-Based Therapeutics for Hepatic Fibrosis: Molecular Mechanisms and Nanoparticulate Drug Delivery Systems. International Journal of Molecular Sciences. 2024; 25(17):9346. https://doi.org/10.3390/ijms25179346
Chicago/Turabian StyleCiceu, Alina, Ferenc Fenyvesi, Anca Hermenean, Simona Ardelean, Simona Dumitra, and Monica Puticiu. 2024. "Advancements in Plant-Based Therapeutics for Hepatic Fibrosis: Molecular Mechanisms and Nanoparticulate Drug Delivery Systems" International Journal of Molecular Sciences 25, no. 17: 9346. https://doi.org/10.3390/ijms25179346
APA StyleCiceu, A., Fenyvesi, F., Hermenean, A., Ardelean, S., Dumitra, S., & Puticiu, M. (2024). Advancements in Plant-Based Therapeutics for Hepatic Fibrosis: Molecular Mechanisms and Nanoparticulate Drug Delivery Systems. International Journal of Molecular Sciences, 25(17), 9346. https://doi.org/10.3390/ijms25179346








