Functions of Hemp-Induced Exosomes against Periodontal Deterioration Caused by Fine Dust
Abstract
1. Introduction
2. Results
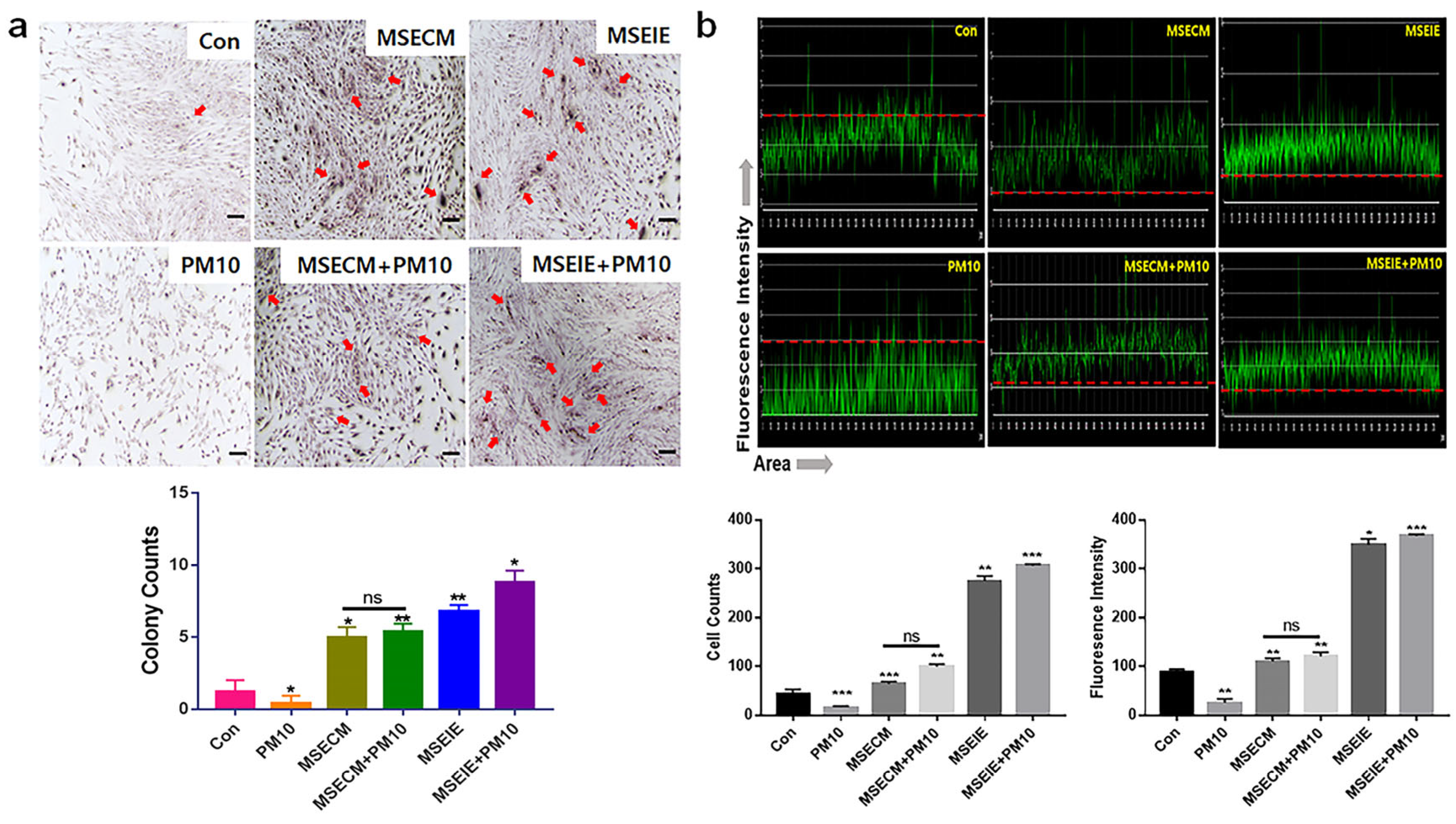
2.1. Protection of Gingival Cells against Fine Dust
2.2. Profiling of microRNAs in MSEIEs
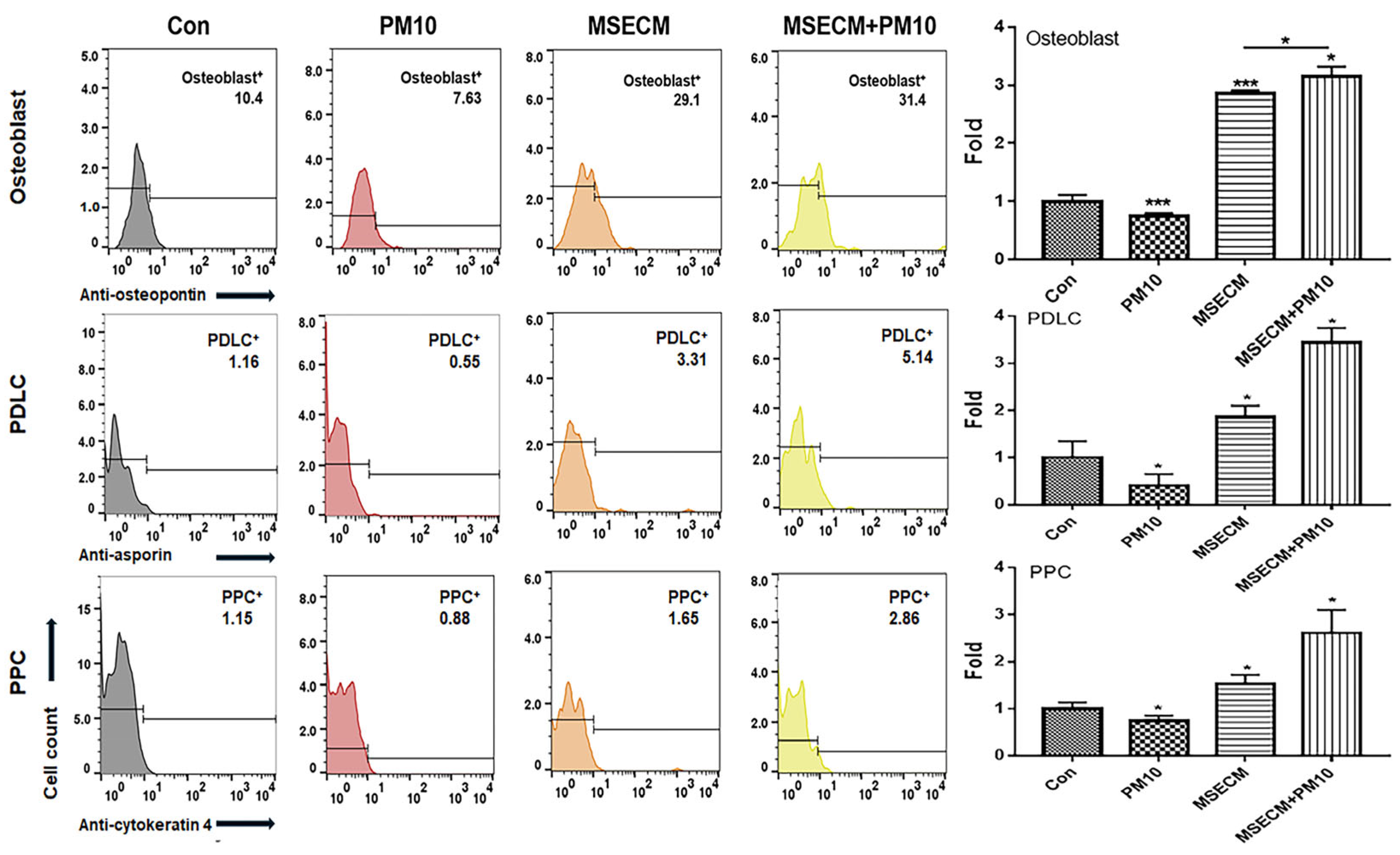
2.3. Activation of Osteogenic Differentiation by the MSEIEs
2.4. Activation of PDLC Differentiation by the MSEIEs
2.5. Activation of PPC Differentiation by the MSEIEs
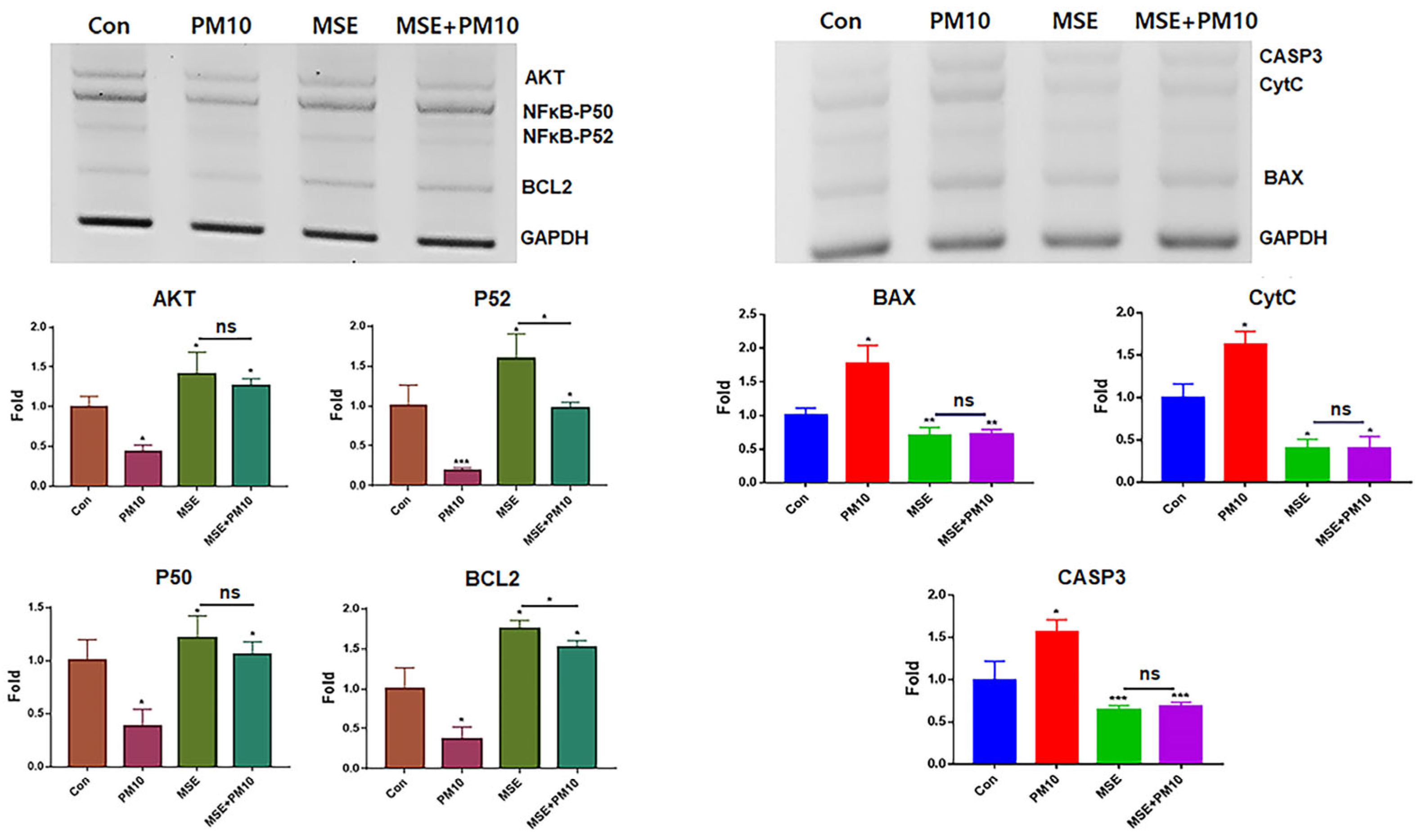
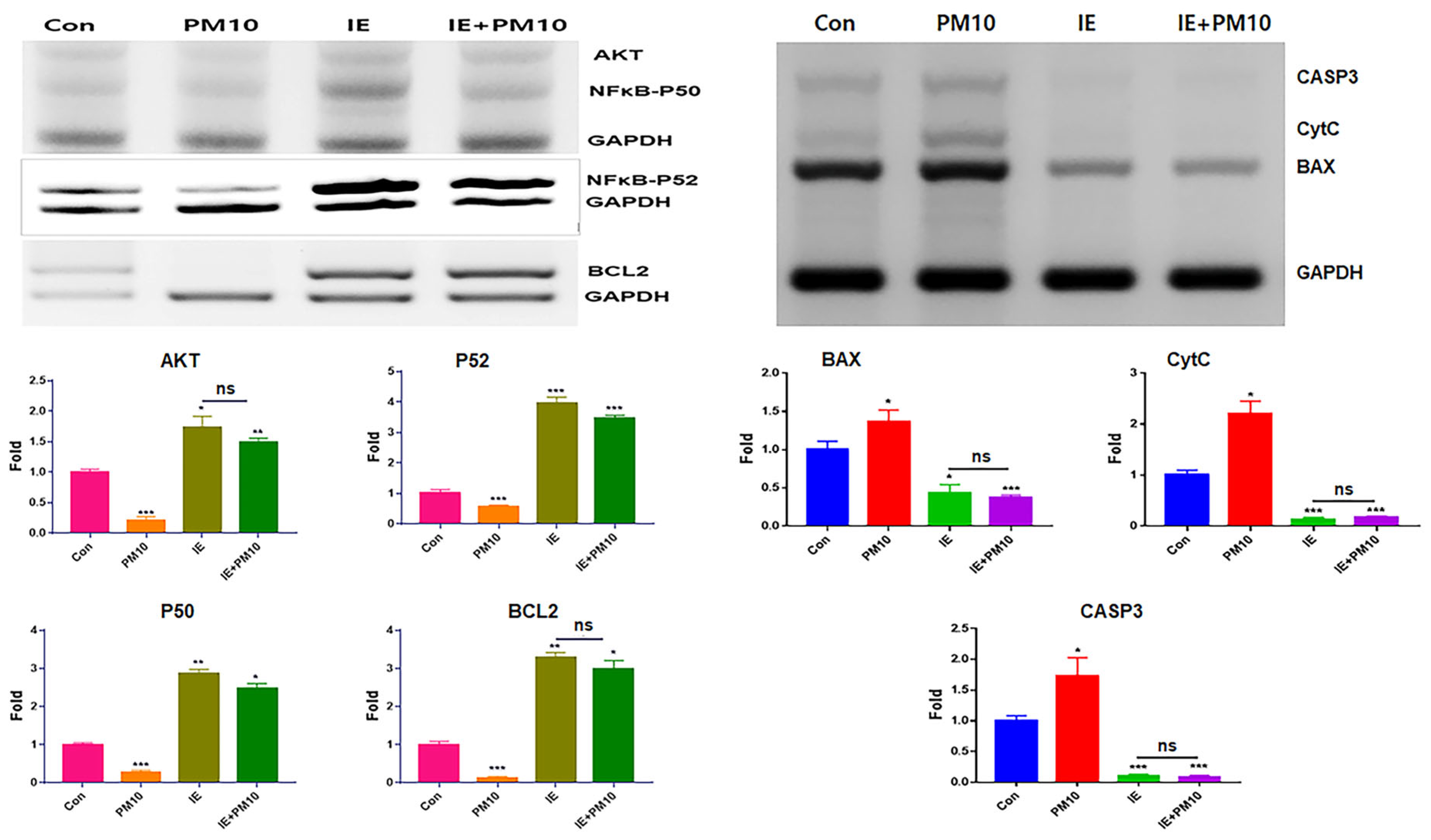
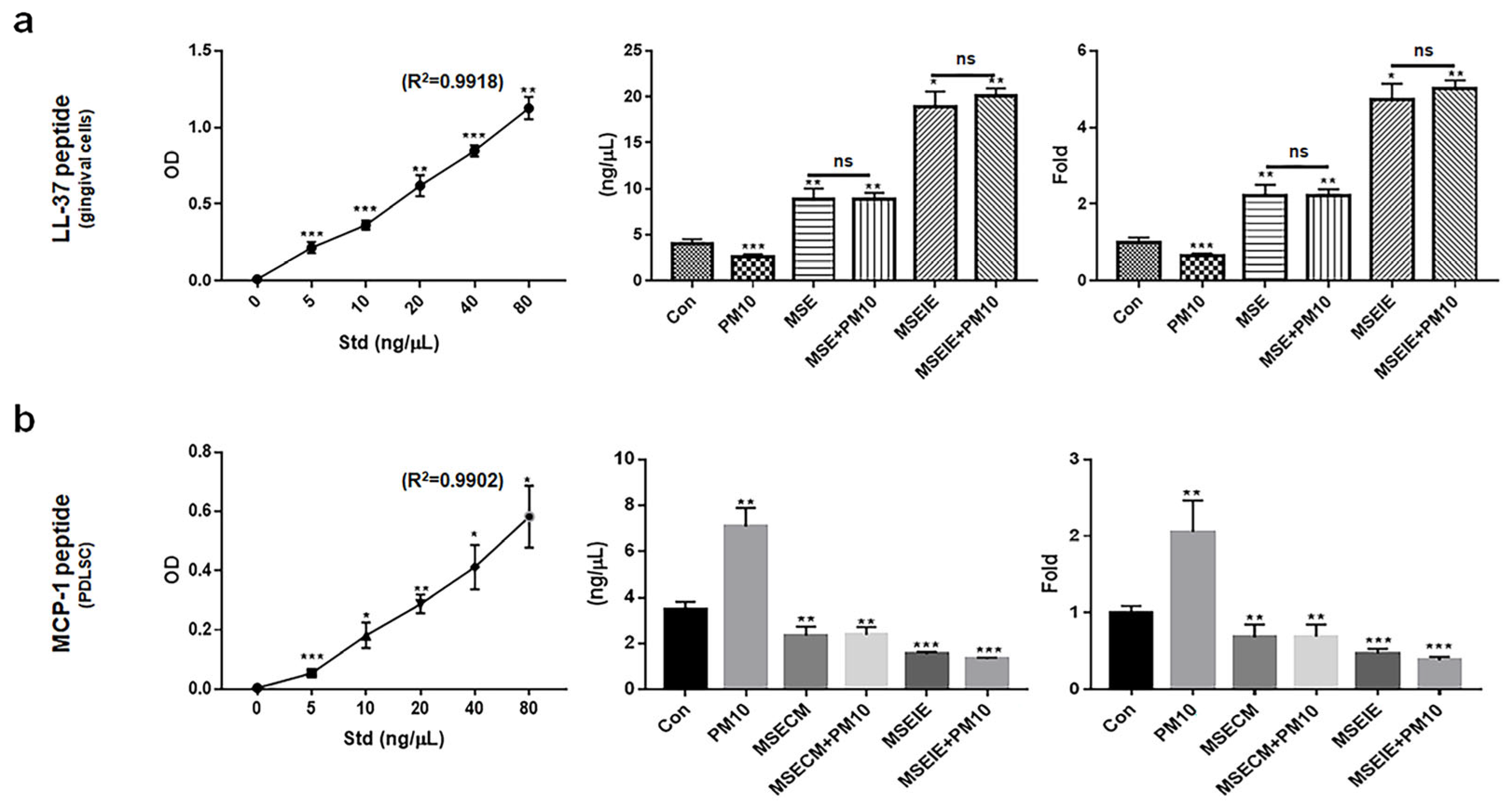
2.6. Modulation of the Expression of Homeostatic Proteins by MSEIEs
3. Discussion
4. Materials and Methods
4.1. Cell Culture and Cytotoxicity Test to Establish the Treatment Dosage
4.2. Anti-Apoptotic Activity of MSE
4.3. Profiling of microRNA in MSEIEs
4.4. PDLSC Differentiation Patterns under MSECM Treatment
4.5. Immunocytochemistry for Osteoblasts
4.6. Localization of PDLSC Markers Using Immunocytochemistry
4.7. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zhang, J.; Chen, Z.; Shan, D.; Wu, Y.; Zhao, Y.; Li, C.; Shu, Y.; Linghu, X.; Wang, B. Adverse effects of exposure to fine particles and ultrafine particles in the environment on different organs of organisms. J. Environ. Sci. 2024, 135, 449–473. [Google Scholar] [CrossRef]
- Yun, M.; Kim, B. Effects of Scutellaria baicalensis Extract-Induced Exosomes on the Periodontal Stem Cells and Immune Cells under Fine Dust. Nanomaterials 2024, 14, 1396. [Google Scholar] [CrossRef] [PubMed]
- Javadinejad, S.; Dara, R.; Jafary, F. Health impacts of extreme events. Saf. Extrem. Environ. 2020, 2, 171–181. [Google Scholar] [CrossRef]
- Xu, H.; Jia, Y.; Sun, Z.; Su, J.; Liu, Q.S.; Zhou, Q.; Jiang, G. Environmental pollution, a hidden culprit for health issues. Eco-Environ. Health 2022, 1, 31–45. [Google Scholar] [CrossRef] [PubMed]
- Mishra, R.; Krishnamoorthy, P.; Gangamma, S.; Raut, A.A.; Kumar, H. Particulate matter (PM10) enhances RNA virus infection through modulation of innate immune responses. Environ. Pollut. 2020, 266, 115148. [Google Scholar] [CrossRef]
- Chen, X.; Liu, J.; Zhou, J.; Wang, J.; Chen, C.; Song, Y.; Pan, J. Urban particulate matter (PM) suppresses airway antibacterial defence. Respir. Res. 2018, 19, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Franzetti, A.; Gandolfi, I.; Gaspari, E.; Ambrosini, R.; Bestetti, G. Seasonal variability of bacteria in fine and coarse urban air particulate matter. Appl. Microbiol. Biotechnol. 2011, 90, 745–753. [Google Scholar] [CrossRef]
- Davalos, D.; Akassoglou, K. Fibrinogen as a key regulator of inflammation in disease. Semin. Immunopathol. 2012, 34, 43–62. [Google Scholar] [CrossRef]
- Park, Y.; Shin, G.H.; Jin, G.S.; Jin, S.; Kim, B.; Lee, S.G. Effects of black jade on osteogenic differentiation of adipose derived stem cells under benzopyrene. Appl. Sci. 2021, 11, 1346. [Google Scholar] [CrossRef]
- Wada, Y.; Ikemoto, T.; Morine, Y.; Imura, S.; Saito, Y.; Yamada, S.; Shimada, M. The differences in the characteristics of insulin-producing cells using human adipose-tissue derived mesenchymal stem cells from subcutaneous and visceral tissues. Sci. Rep. 2019, 9, 13204. [Google Scholar] [CrossRef]
- Gardin, C.; Ricci, S.; Ferroni, L. Dental stem cells (DSCs): Classification and properties. In Dental Stem Cells: Regenerative Potential; Humana Press: Cham, Switzerland, 2016; pp. 1–25. [Google Scholar]
- Queiroz, A.; Albuquerque-Souza, E.; Gasparoni, L.M.; de França, B.N.; Pelissari, C.; Trierveiler, M.; Holzhausen, M. Therapeutic potential of periodontal ligament stem cells. World J. Stem Cells 2021, 13, 605. [Google Scholar] [CrossRef] [PubMed]
- Zheng, W.; Wang, S.; Wang, J.; Jin, F. Periodontitis promotes the proliferation and suppresses the differentiation potential of human periodontal ligament stem cells. Int. J. Mol. Med. 2015, 36, 915–922. [Google Scholar] [CrossRef]
- Ridyard, K.E.; Overhage, J. The potential of human peptide LL-37 as an antimicrobial and anti-biofilm agent. Antibiotics 2021, 10, 650. [Google Scholar] [CrossRef] [PubMed]
- Bechinger, B.; Gorr, S.-U. Antimicrobial peptides: Mechanisms of action and resistance. J. Dent. Res. 2017, 96, 254–260. [Google Scholar] [CrossRef]
- Aidoukovitch, A.; Anders, E.; Dahl, S.; Nebel, D.; Svensson, D.; Nilsson, B.O. The host defense peptide LL-37 is internalized by human periodontal ligament cells and prevents LPS-induced MCP-1 production. J. Periodontal Res. 2019, 54, 662–670. [Google Scholar] [CrossRef]
- Nilsson, B.O. Mechanisms involved in regulation of periodontal ligament cell production of pro-inflammatory cytokines: Implications in periodontitis. J. Periodontal Res. 2021, 56, 249–255. [Google Scholar] [CrossRef] [PubMed]
- Svensson, D.; Aidoukovitch, A.; Anders, E.; Jönsson, D.; Nebel, D.; Nilsson, B.-O. Secretory leukocyte protease inhibitor regulates human periodontal ligament cell production of pro-inflammatory cytokines. Inflamm. Res. 2017, 66, 823–831. [Google Scholar] [CrossRef]
- Tkach, M.; Théry, C. Communication by extracellular vesicles: Where we are and where we need to go. Cell 2016, 164, 1226–1232. [Google Scholar] [CrossRef]
- Shen, X.; Song, S.; Chen, N.; Liao, J.; Zeng, L. Stem cell-derived exosomes: A supernova in cosmetic dermatology. J. Cosmet. Dermatol. 2021, 20, 3812–3817. [Google Scholar] [CrossRef]
- Li, M.; Li, S.; Du, C.; Zhang, Y.; Li, Y.; Chu, L.; Han, X.; Galons, H.; Zhang, Y.; Sun, H. Exosomes from different cells: Characteristics, modifications, and therapeutic applications. Eur. J. Med. Chem. 2020, 207, 112784. [Google Scholar] [CrossRef]
- Park, Y.; Lee, K.; Kim, S.W.; Lee, M.W.; Kim, B.; Lee, S.G. Effects of induced exosomes from endometrial cancer cells on tumor activity in the presence of Aurea helianthus extract. Molecules 2021, 26, 2207. [Google Scholar] [CrossRef]
- Keller, N.M. The legalization of industrial hemp and what it could mean for Indiana’s biofuel industry. Indiana Int. Comp. Law Rev. 2013, 23, 555. [Google Scholar] [CrossRef]
- Johnson, R. Defining hemp: A fact sheet. Congr. Res. Serv. 2019, 44742, 1–12. [Google Scholar]
- Fordjour, E.; Manful, C.F.; Sey, A.A.; Javed, R.; Pham, T.H.; Thomas, R.; Cheema, M. Cannabis: A multifaceted plant with endless potentials. Front. Pharmacol. 2023, 14, 1200269. [Google Scholar] [CrossRef]
- Xu, J.; Bai, M.; Song, H.; Yang, L.; Zhu, D.; Liu, H. Hemp (Cannabis sativa subsp. sativa) Chemical composition and the application of hempseeds in food formulations. Plant Foods Hum. Nutr. 2022, 77, 504–513. [Google Scholar] [CrossRef]
- Aloo, S.O.; Kwame, F.O.; Oh, D.-H. Identification of possible bioactive compounds and a comparative study on in vitro biological properties of whole hemp seed and stem. Food Biosci. 2023, 51, 102329. [Google Scholar] [CrossRef]
- Adinew, G.M.; Taka, E.; Mendonca, P.; Messeha, S.S.; Soliman, K.F. The anticancer effects of flavonoids through miRNAs modulations in triple-negative breast cancer. Nutrients 2021, 13, 1212. [Google Scholar] [CrossRef]
- Pervez, M.A.; Khan, D.A.; Gilani, S.T.A.; Fatima, S.; Ijaz, A.; Nida, S. Hepato-protective effects of delta-tocotrienol and alpha-tocopherol in patients with non-alcoholic fatty liver disease: Regulation of circulating MicroRNA expression. Int. J. Mol. Sci. 2022, 24, 79. [Google Scholar] [CrossRef]
- Jopling, C. Liver-specific microRNA-122: Biogenesis and function. RNA Biol. 2012, 9, 137–142. [Google Scholar] [CrossRef]
- Barbu, M.G.; Condrat, C.E.; Thompson, D.C.; Bugnar, O.L.; Cretoiu, D.; Toader, O.D.; Suciu, N.; Voinea, S.C. MicroRNA involvement in signaling pathways during viral infection. Front. Cell Dev. Biol. 2020, 8, 143. [Google Scholar] [CrossRef]
- Bhuyan, R.; Bhuyan, S.K.; Mohanty, J.N.; Das, S.; Juliana, N.; Abu, I.F. Periodontitis and its inflammatory changes linked to various systemic diseases: A review of its underlying mechanisms. Biomedicines 2022, 10, 2659. [Google Scholar] [CrossRef]
- Marruganti, C.; Shin, H.-S.; Sim, S.-J.; Grandini, S.; Laforí, A.; Romandini, M. Air pollution as a risk indicator for periodontitis. Biomedicines 2023, 11, 443. [Google Scholar] [CrossRef]
- Rayad, S.; Dobrzyński, M.; Kuźniarski, A.; Styczyńska, M.; Diakowska, D.; Gedrange, T.; Klimas, S.; Gębarowski, T.; Dominiak, M. An In-Vitro Evaluation of Toxic Metals Concentration in the Third Molars from Residents of the Legnica-Głogów Copper Area and Risk Factors Determining the Accumulation of Those Metals: A Pilot Study. Appl. Sci. 2023, 13, 2904. [Google Scholar] [CrossRef]
- Cocârţă, D.M.; Prodana, M.; Demetrescu, I.; Lungu, P.E.M.; Didilescu, A.C. Indoor air pollution with fine particles and implications for workers’ health in dental offices: A brief review. Sustainability 2021, 13, 599. [Google Scholar] [CrossRef]
- Turner, M.C.; Andersen, Z.J.; Baccarelli, A.; Diver, W.R.; Gapstur, S.M.; Pope, C.A., III; Prada, D.; Samet, J.; Thurston, G.; Cohen, A. Outdoor air pollution and cancer: An overview of the current evidence and public health recommendations. CA Cancer J. Clin. 2020, 70, 460–479. [Google Scholar] [CrossRef]
- Luo, D.; Fan, H.; Ma, X.; Yang, C.; He, Y.; Ge, Y.; Jiang, M.; Xu, Z.; Yang, L. miR-1301-3p promotes cell proliferation and facilitates cell cycle progression via targeting SIRT1 in gastric cancer. Front. Oncol. 2021, 11, 664242. [Google Scholar] [CrossRef]
- Wang, C.; Liu, S.; Li, J.; Cheng, Y.; Wang, Z.; Feng, T.; Lu, G.; Wang, S.; Song, J.; Xia, P. Biological functions of let-7e-5p in promoting the differentiation of MC3T3-E1 cells. Front. Cell Dev. Biol. 2021, 9, 671170. [Google Scholar] [CrossRef]
- Li, C.; Qin, Y.; Ouyang, T.; Yao, M.; Zhang, A.; Luo, P.; Pan, X. miR-122-5p mediates fluoride-induced osteoblast activation by targeting CDK4. Biol. Trace Elem. Res. 2021, 199, 1215–1227. [Google Scholar] [CrossRef]
- Liu, W.; Ying, N.; Rao, X.; Chen, X. MiR-942-3p as a potential prognostic marker of gastric cancer associated with AR and MAPK/ERK signaling pathway. Curr. Issues Mol. Biol. 2022, 44, 3835–3848. [Google Scholar] [CrossRef]
- Yang, A.; Yang, L.; Xu, H.; Hu, J.; Cai, H.; Ning, Y.; Li, Z. Mechanism of Icariin regulation of the effect of miR-122-5p in osteoblast-derived exosomes on osteogenesis and migration of bone marrow mesenchymal stem cells. Res. Sq. 2022, 1–20. [Google Scholar] [CrossRef]
- Sartori, E.M.; Magro-Filho, O.; Silveira Mendonca, D.B.; Li, X.; Fu, J.; Mendonça, G. Modulation of Micro RNA Expression and Osteoblast Differentiation by Nanotopography. Int. J. Oral Maxillofac. Implant. 2018, 33, 269–280. [Google Scholar] [CrossRef]
- Chen, X.; Xie, W.; Zhang, M.; Shi, Y.; Xu, S.; Cheng, H.; Wu, L.; Pathak, J.L.; Zheng, Z. The Emerging Role of Non-Coding RNAs in Osteogenic Differentiation of Human Bone Marrow Mesenchymal Stem Cells. Front. Cell Dev. Biol. 2022, 10, 903278. [Google Scholar] [CrossRef]
- Wei, J.; Li, H.; Wang, S.; Li, T.; Fan, J.; Liang, X.; Li, J.; Han, Q.; Zhu, L.; Fan, L. let-7 enhances osteogenesis and bone formation while repressing adipogenesis of human stromal/mesenchymal stem cells by regulating HMGA2. Stem Cells Dev. 2014, 23, 1452–1463. [Google Scholar] [CrossRef]
- Alves, L.; Machado, V.; Botelho, J.; Mendes, J.J.; Cabral, J.M.; da Silva, C.L.; Carvalho, M.S. Enhanced proliferative and osteogenic potential of periodontal ligament stromal cells. Biomedicines 2023, 11, 1352. [Google Scholar] [CrossRef]
- Mohebichamkhorami, F.; Fattahi, R.; Niknam, Z.; Aliashrafi, M.; Khakpour Naeimi, S.; Gilanchi, S.; Zali, H. Periodontal ligament stem cells as a promising therapeutic target for neural damage. Stem Cell Res. Ther. 2022, 13, 273. [Google Scholar] [CrossRef]
- Marchesan, J.T.; Scanlon, C.S.; Soehren, S.; Matsuo, M.; Kapila, Y.L. Implications of cultured periodontal ligament cells for the clinical and experimental setting: A review. Arch. Oral Biol. 2011, 56, 933–943. [Google Scholar] [CrossRef]
- Heitz-Mayfield, L.J.; Mombelli, A. The therapy of peri-implantitis: A systematic review. Int. J. Oral Maxillofac. Implant. 2014, 29, 325–345. [Google Scholar] [CrossRef]
- Tuikampee, S.; Chaijareenont, P.; Rungsiyakull, P.; Yavirach, A. Titanium Surface Modification Techniques to Enhance Osteoblasts and Bone Formation for Dental Implants: A Narrative Review on Current Advances. Metals 2024, 14, 515. [Google Scholar] [CrossRef]
- Grawish, M.E.; Saeed, M.A.; Sultan, N.; Scheven, B.A. Therapeutic applications of dental pulp stem cells in regenerating dental, periodontal and oral-related structures. World J. Meta-Anal. 2021, 9, 176–192. [Google Scholar] [CrossRef]
- Yu, C.; Abbott, P.V. An overview of the dental pulp: Its functions and responses to injury. Aust. Dent. J. 2007, 52, S4–S6. [Google Scholar] [CrossRef]
- Tokajuk, J.; Deptuła, P.; Piktel, E.; Daniluk, T.; Chmielewska, S.; Wollny, T.; Wolak, P.; Fiedoruk, K.; Bucki, R. Cathelicidin LL-37 in health and diseases of the oral cavity. Biomedicines 2022, 10, 1086. [Google Scholar] [CrossRef] [PubMed]
- Jönsson, D.; Nilsson, B.O. The antimicrobial peptide LL-37 is anti-inflammatory and proapoptotic in human periodontal ligament cells. J. Periodontal Res. 2012, 47, 330–335. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.-J.; Chau, J.; Ebert, P.J.; Sylvester, G.; Min, H.; Liu, G.; Braich, R.; Manoharan, M.; Soutschek, J.; Skare, P. miR-181a is an intrinsic modulator of T cell sensitivity and selection. Cell 2007, 129, 147–161. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.; Anshita, D.; Ravichandiran, V. MCP-1: Function, regulation, and involvement in disease. Int. Immunopharmacol. 2021, 101, 107598. [Google Scholar] [CrossRef]
- Fernández-Rojas, B.; Gutiérrez-Venegas, G. Flavonoids exert multiple periodontic benefits including anti-inflammatory, periodontal ligament-supporting, and alveolar bone-preserving effects. Life Sci. 2018, 209, 435–454. [Google Scholar] [CrossRef]
- Boyerinas, B.; Park, S.-M.; Hau, A.; Murmann, A.E.; Peter, M.E. The role of let-7 in cell differentiation and cancer. Endocr. Relat. Cancer 2010, 17, F19–F36. [Google Scholar] [CrossRef]
- Park, S.-M.; Shell, S.; Radjabi, A.R.; Schickel, R.; Feig, C.; Boyerinas, B.; Dinulescu, D.M.; Lengyel, E.; Peter, M.E. Let-7 prevents early cancer progression by suppressing expression of the embryonic gene HMGA2. Cell Cycle 2007, 6, 2585–2590. [Google Scholar] [CrossRef]

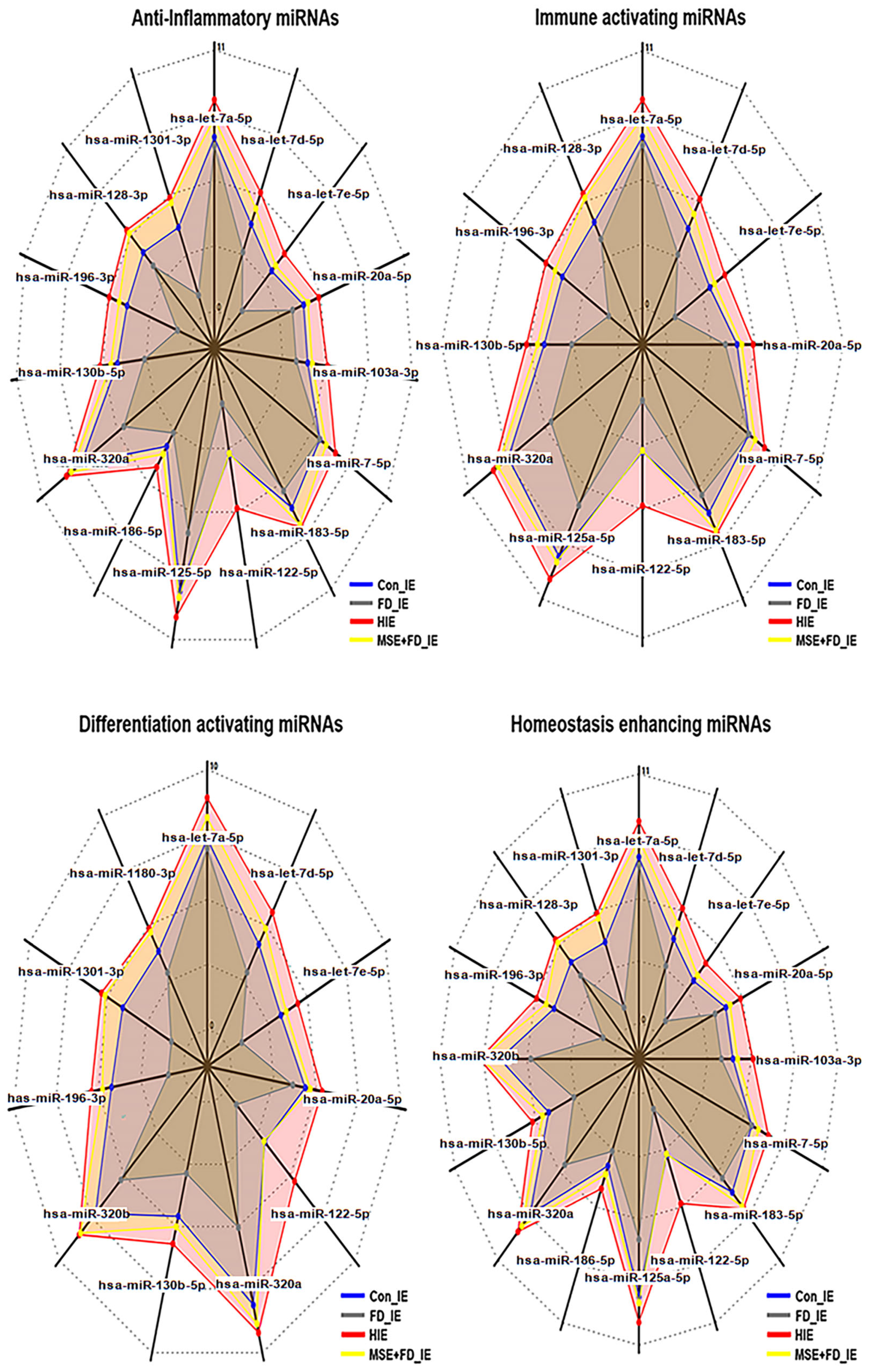

| Category | Anti-Inflammation | Immnue Activation | Differentiating Activation | Homeostatic Enhancement |
|---|---|---|---|---|
| Up-regulated genes | 16 | 11 | 11 | 15 |
| Dramatic Up-regulation | hsa-miR-1301-3p | hsa-miR-196-3p | hsa-miR-1301-3p | hsa-miR-1301-3p |
| hsa-miR-196-3p | hsa-let-7e-5p | hsa-miR-196-3p | hsa-miR-196-3p | |
| hsa-let-7e-5p | hsa-miR-122-5p | hsa-let-7e-5p | hsa-let-7e-5p | |
| hsa-miR-122-5p | hsa-miR-122-5p | hsa-miR-122-5p |
| Gene | Seq (5′ → 3′) |
|---|---|
| AKT | F: GGCTGCCAAGTGTCAAATCC R: AGTGCTCCCCCACTTACTTG |
| NFκB-P50 | F: CGGAGCCCTCTTTCACAGTT R: TTCAGCTTAGGAGCGAAGGC |
| NFκB-P52 | F: AGGTGCTGTAGCGGGATTTC R: AGAGGCACTGTATAGGGCAG |
| BCL2 | F: CTGCTGACATGCTTGGAAAA R: ATTGGGCTACCCCAGCAATG |
| BAX | F: AGCGCTCCCCCACTTACTTG R: GACAGGGACATCAGTCGCTT |
| CytC | F: ATGAATGACCACTCTAGCCA R: ATAGAAACAGCCAGGACCGC |
| CASP3 | F:TCCCTGGGAAGAAAGAGTTGTGG R:TGAACATGGCACCTCTGCAAC |
| GAPDH | F: GTGGTCTCCTCTGACTTCAACA R: CTCTTCCTCTTGTGCTCTTGCT |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, E.; Park, Y.; Yun, M.; Kim, B. Functions of Hemp-Induced Exosomes against Periodontal Deterioration Caused by Fine Dust. Int. J. Mol. Sci. 2024, 25, 10331. https://doi.org/10.3390/ijms251910331
Kim E, Park Y, Yun M, Kim B. Functions of Hemp-Induced Exosomes against Periodontal Deterioration Caused by Fine Dust. International Journal of Molecular Sciences. 2024; 25(19):10331. https://doi.org/10.3390/ijms251910331
Chicago/Turabian StyleKim, Eunhee, Yoonjin Park, Mihae Yun, and Boyong Kim. 2024. "Functions of Hemp-Induced Exosomes against Periodontal Deterioration Caused by Fine Dust" International Journal of Molecular Sciences 25, no. 19: 10331. https://doi.org/10.3390/ijms251910331
APA StyleKim, E., Park, Y., Yun, M., & Kim, B. (2024). Functions of Hemp-Induced Exosomes against Periodontal Deterioration Caused by Fine Dust. International Journal of Molecular Sciences, 25(19), 10331. https://doi.org/10.3390/ijms251910331





