Perivascular Adipose Tissue Becomes Pro-Contractile and Remodels in an IL10−/− Colitis Model of Inflammatory Bowel Disease
Abstract
1. Introduction
2. Results
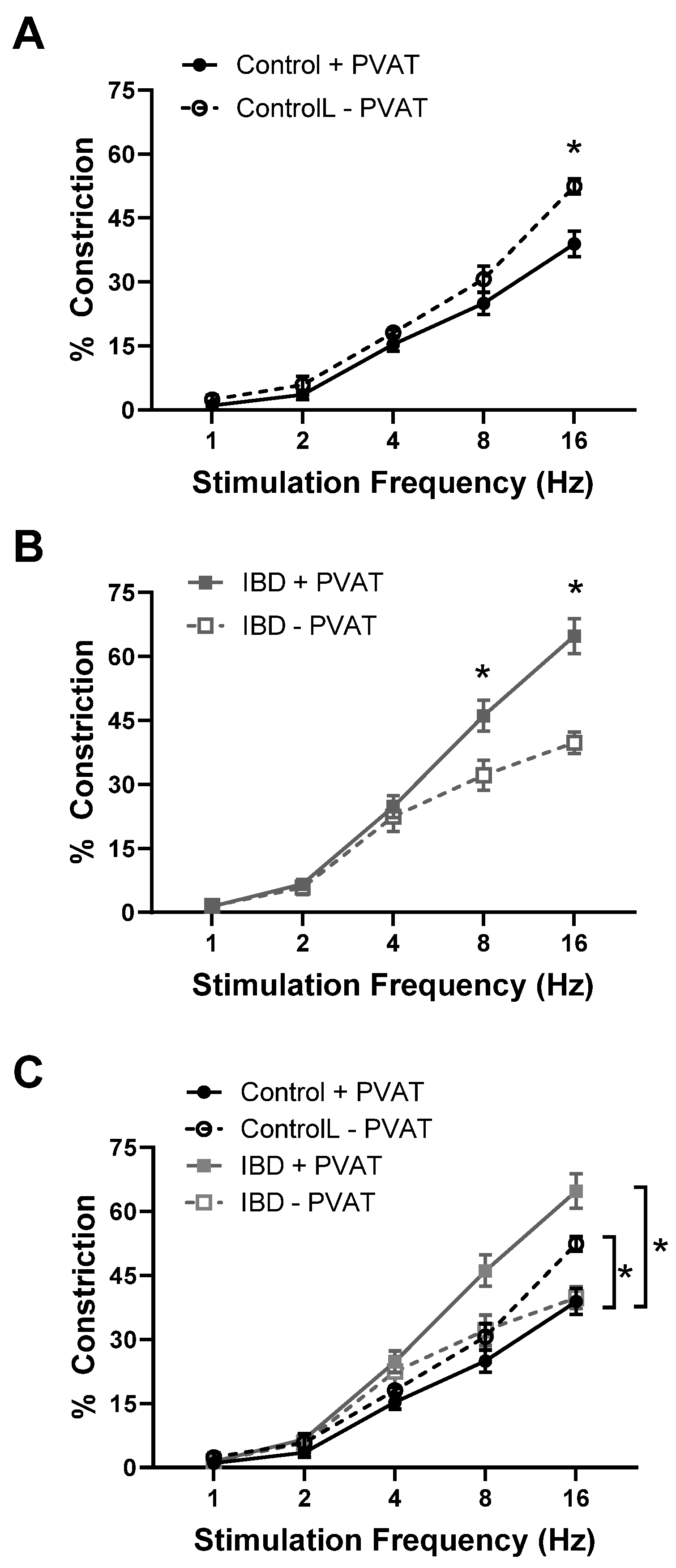
2.1. PVAT Becomes Pro-Contractile in Mouse Mesenteric Arteries with IBD
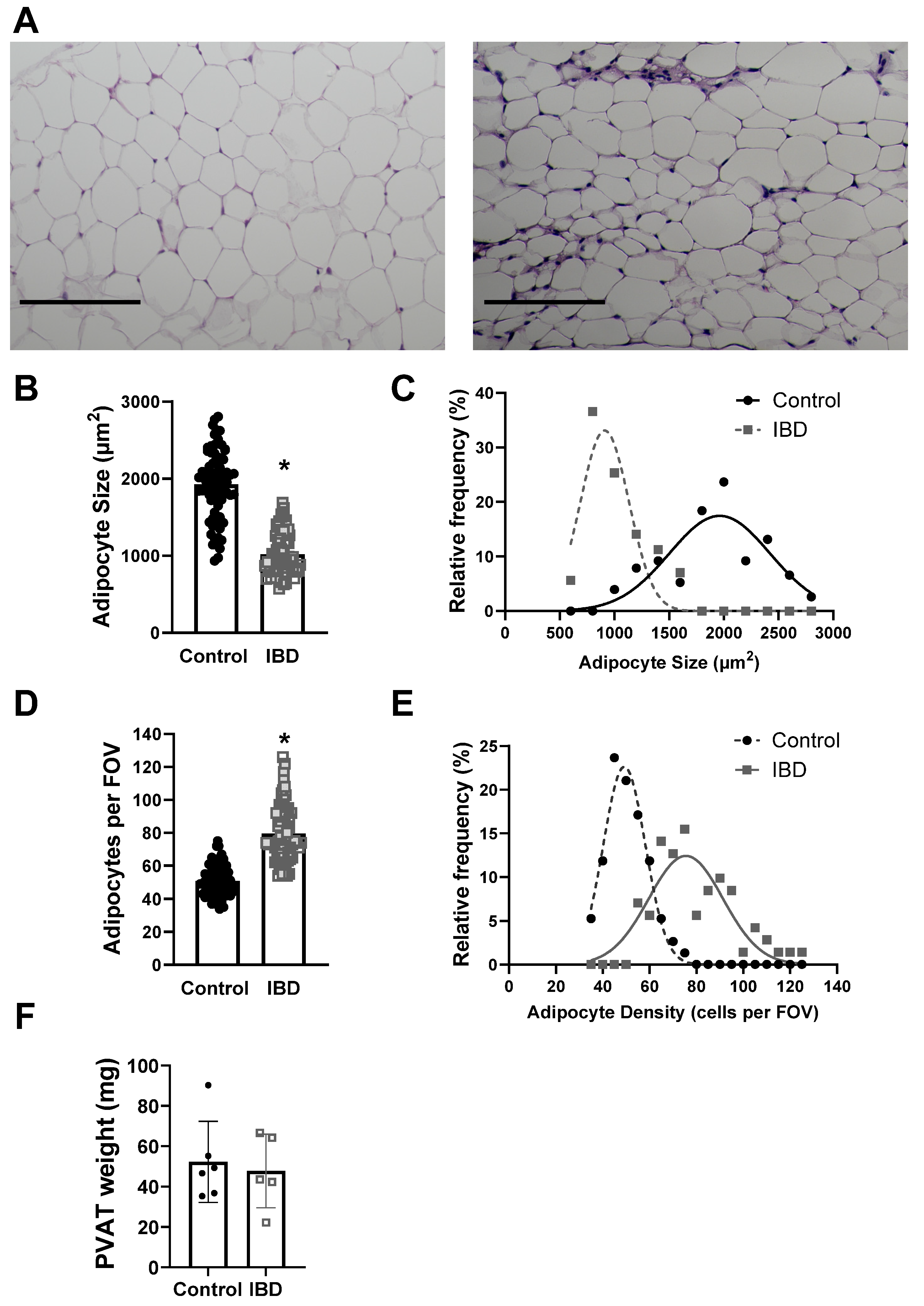
2.2. PVAT Remodels with IBD
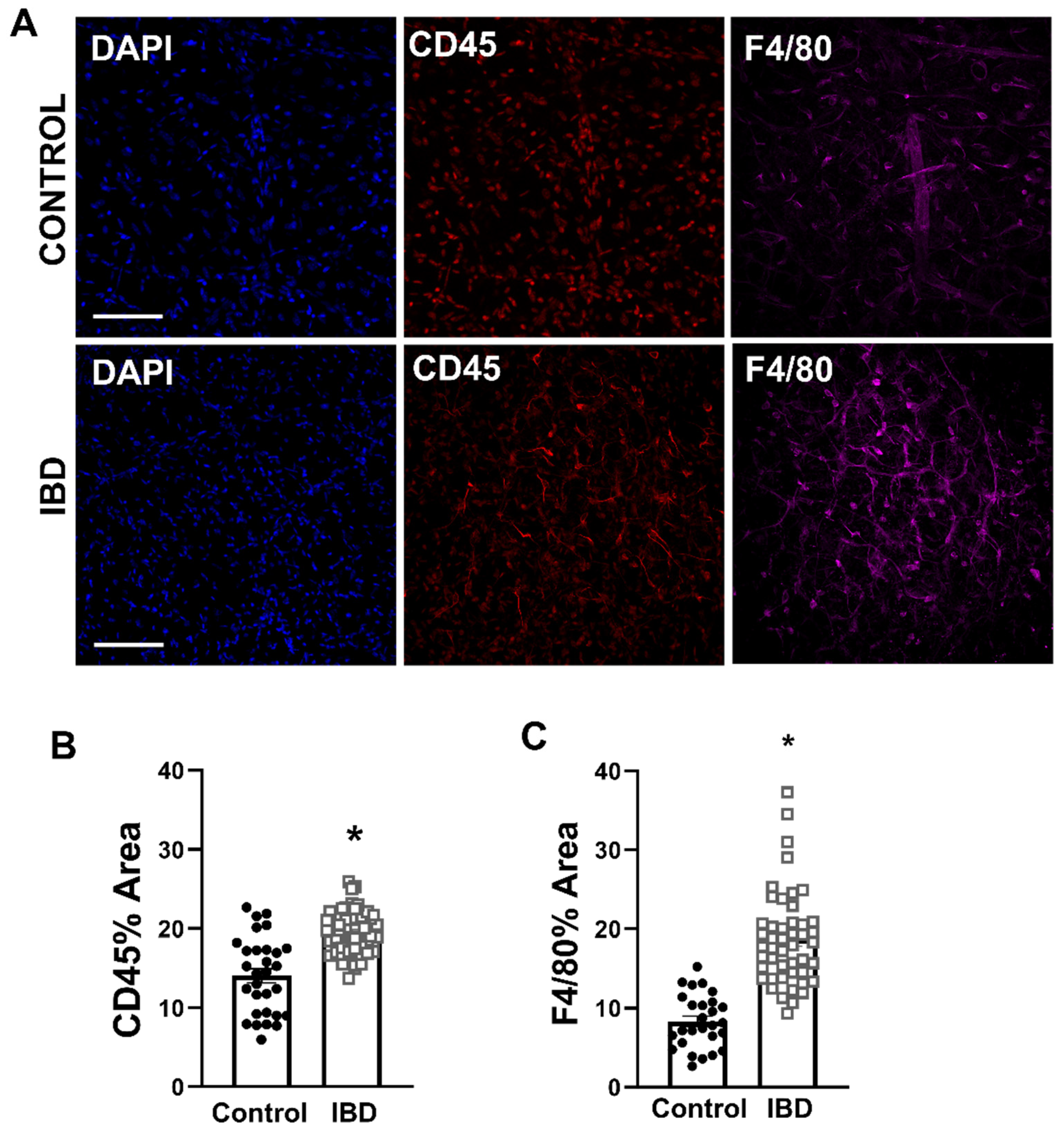
2.3. Macrophages Accumulate in PVAT with IBD
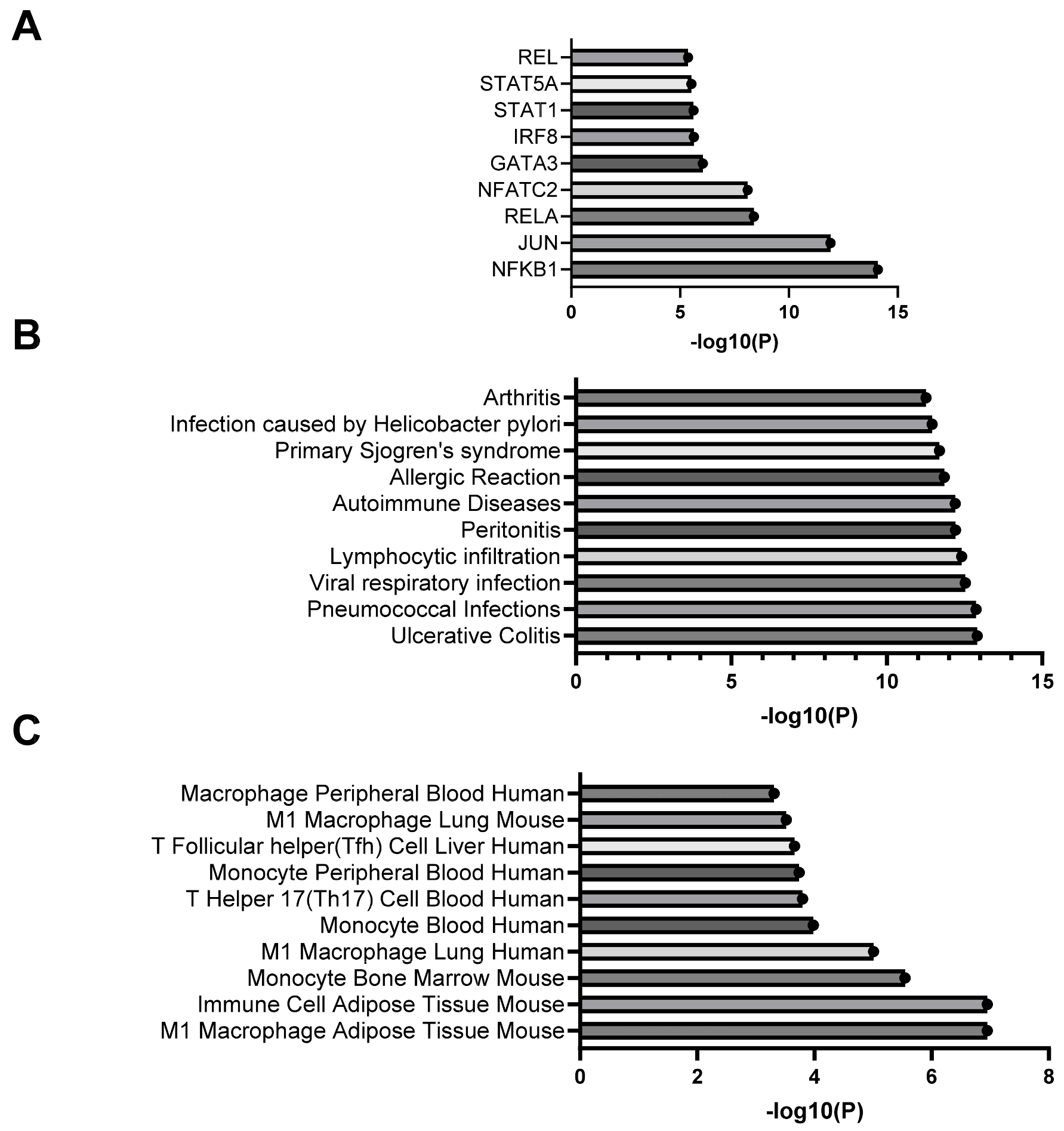
2.4. IBD Leads to Altered PVAT Expression of Cytokine and Chemokine Genes
3. Discussion
4. Materials and Methods
4.1. Animals and Tissue Collection
4.2. Isolated Artery Studies
4.3. PVAT Sectioning and Adipocyte Measurements
4.4. Immunofluorescence and Quantitation
4.5. PCR Arrays
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Wang, R.; Li, Z.; Liu, S.; Zhang, D. Global, regional and national burden of inflammatory bowel disease in 204 countries and territories from 1990 to 2019: A systematic analysis based on the Global Burden of Disease Study 2019. BMJ Open 2023, 13, e065186. [Google Scholar] [CrossRef] [PubMed]
- Weisman, M.H.; Oleg, S.; Seok Kim, H.; Hou, J.K.; Miller, F.W.; Dillon, C.F. Inflammatory Bowel Disease Prevalence: Surveillance data from the U.S. National Health and Nutrition Examination Survey. Prev. Med. Rep. 2023, 33, 102173. [Google Scholar] [CrossRef] [PubMed]
- Hultén, L.; Lindhagen, J.; Lundgren, O.; Fasth, S.; Ahrén, C. Regional intestinal blood flow in ulcerative colitis and Crohn’s disease. Gastroenterology 1977, 72, 388–396. [Google Scholar] [CrossRef]
- Harris, N.R.; Carter, P.R.; Lee, S.; Watts, M.N.; Zhang, S.; Grisham, M.B. Association between blood flow and inflammatory state in a T-cell transfer model of inflammatory bowel disease in mice. Inflamm. Bowel Dis. 2010, 16, 776–782. [Google Scholar] [CrossRef]
- Saevik, F.; Nylund, K.; Hausken, T.; Odegaard, S.; Gilja, O.H. Bowel perfusion measured with dynamic contrast-enhanced ultrasound predicts treatment outcome in patients with Crohn’s disease. Inflamm. Bowel Dis. 2014, 20, 2029–2037. [Google Scholar] [CrossRef]
- Ludwig, D.; Wiener, S.; Brüning, A.; Schwarting, K.; Jantschek, G.; Stange, E.F. Mesenteric blood flow is related to disease activity and risk of relapse in Crohn’s disease: A prospective follow-up study. Am. J. Gastroenterol. 1999, 94, 2942–2950. [Google Scholar] [CrossRef]
- Roifman, I.; Sun, Y.C.; Fedwick, J.P.; Panaccione, R.; Buret, A.G.; Liu, H.; Rostom, A.; Anderson, T.J.; Beck, P.L. Evidence of endothelial dysfunction in patients with inflammatory bowel disease. Clin. Gastroenterol. Hepatol. 2009, 7, 175–182. [Google Scholar] [CrossRef] [PubMed]
- Hatoum, O.A.; Binion, D.G.; Otterson, M.F.; Gutterman, D.D. Acquired microvascular dysfunction in inflammatory bowel disease: Loss of nitric oxide-mediated vasodilation. Gastroenterology 2003, 125, 58–69. [Google Scholar] [CrossRef]
- Zanoli, L.; Rastelli, S.; Inserra, G.; Castellino, P. Arterial structure and function in inflammatory bowel disease. World J. Gastroenterol. 2015, 21, 11304–11311. [Google Scholar] [CrossRef]
- Zanoli, L.; Cannavò, M.; Rastelli, S.; Di Pino, L.; Monte, I.; Di Gangi, M.; Boutouyrie, P.; Inserra, G.; Laurent, S.; Castellino, P. Arterial stiffness is increased in patients with inflammatory bowel disease. J. Hypertens. 2012, 30, 1775–1781. [Google Scholar] [CrossRef]
- Zanoli, L.; Rastelli, S.; Inserra, G.; Lentini, P.; Valvo, E.; Calcagno, E.; Boutouyrie, P.; Laurent, S.; Castellino, P. Increased arterial stiffness in inflammatory bowel diseases is dependent upon inflammation and reduced by immunomodulatory drugs. Atherosclerosis 2014, 234, 346–351. [Google Scholar] [CrossRef] [PubMed]
- Norton, C.E.; Grunz-Borgmann, E.A.; Hart, M.L.; Jones, B.W.; Franklin, C.L.; Boerman, E.M. Role of perivascular nerve and sensory neurotransmitter dysfunction in inflammatory bowel disease. Am. J. Physiol. Heart Circ. Physiol. 2021, 320, H1887–H1902. [Google Scholar] [CrossRef] [PubMed]
- Grunz, E.A.; Jones, B.W.; Lateef, O.M.; Sen, S.; Wilkinson, K.; Joshi, T.; Boerman, E.M. Adventitial macrophage accumulation impairs perivascular nerve function in mesenteric arteries with inflammatory bowel disease. Front. Physiol. 2023, 14, 1198066. [Google Scholar] [CrossRef]
- Ramirez, J.G.; O’Malley, E.J.; Ho, W.S.V. Pro-contractile effects of perivascular fat in health and disease. Br. J. Pharmacol. 2017, 174, 3482–3495. [Google Scholar] [CrossRef]
- Shi, H.; Wu, H.; Winkler, M.A.; Belin de Chantemele, E.J.; Lee, R.; Kim, H.W.; Weintraub, N.L. Perivascular adipose tissue in autoimmune rheumatic diseases. Pharmacol. Res. 2022, 182, 106354. [Google Scholar] [CrossRef]
- Shu, W.; Wang, Y.; Li, C.; Zhang, L.; Zhuoma, D.; Yang, P.; Yan, G.; Chen, C.; Ba, Y.; Du, P.; et al. Single-cell Expression Atlas Reveals Cell Heterogeneity in the Creeping Fat of Crohn’s Disease. Inflamm. Bowel Dis. 2023, 29, 850–865. [Google Scholar] [CrossRef]
- Althoff, P.; Schmiegel, W.; Lang, G.; Nicolas, V.; Brechmann, T. Creeping Fat Assessed by Small Bowel MRI Is Linked to Bowel Damage and Abdominal Surgery in Crohn’s Disease. Dig. Dis. Sci. 2019, 64, 204–212. [Google Scholar] [CrossRef] [PubMed]
- Mizoguchi, A. Animal models of inflammatory bowel disease. Prog. Mol. Biol. Transl. Sci. 2012, 105, 263–320. [Google Scholar] [CrossRef]
- Baydi, Z.; Limami, Y.; Khalki, L.; Zaid, N.; Naya, A.; Mtairag, E.M.; Oudghiri, M.; Zaid, Y. An Update of Research Animal Models of Inflammatory Bowel Disease. Sci. World J. 2021, 2021, 7479540. [Google Scholar] [CrossRef]
- Katsandegwaza, B.; Horsnell, W.; Smith, K. Inflammatory Bowel Disease: A Review of Pre-Clinical Murine Models of Human Disease. Int. J. Mol. Sci. 2022, 23I, 9344. [Google Scholar] [CrossRef]
- Norton, C.E.; Boerman, E.M.; Segal, S.S. Differential hyperpolarization to substance P and calcitonin gene-related peptide in smooth muscle versus endothelium of mouse mesenteric artery. Microcirculation 2021, 28, e12733. [Google Scholar] [CrossRef] [PubMed]
- Ketonen, J.; Shi, J.; Martonen, E.; Mervaala, E. Periadventitial adipose tissue promotes endothelial dysfunction via oxidative stress in diet-induced obese C57Bl/6 mice. Circ. J. 2010, 74, 1479–1487. [Google Scholar] [CrossRef] [PubMed]
- Marchesi, C.; Ebrahimian, T.; Angulo, O.; Paradis, P.; Schiffrin, E.L. Endothelial nitric oxide synthase uncoupling and perivascular adipose oxidative stress and inflammation contribute to vascular dysfunction in a rodent model of metabolic syndrome. Hypertension 2009, 54, 1384–1392. [Google Scholar] [CrossRef] [PubMed]
- Galvez-Prieto, B.; Somoza, B.; Gil-Ortega, M.; Garcia-Prieto, C.F.; de Las Heras, A.I.; Gonzalez, M.C.; Arribas, S.; Aranguez, I.; Bolbrinker, J.; Kreutz, R.; et al. Anticontractile Effect of Perivascular Adipose Tissue and Leptin are Reduced in Hypertension. Front. Pharmacol. 2012, 3, 103. [Google Scholar] [CrossRef]
- Agabiti-Rosei, C.; Favero, G.; De Ciuceis, C.; Rossini, C.; Porteri, E.; Rodella, L.F.; Franceschetti, L.; Maria Sarkar, A.; Agabiti-Rosei, E.; Rizzoni, D.; et al. Effect of long-term treatment with melatonin on vascular markers of oxidative stress/inflammation and on the anticontractile activity of perivascular fat in aging mice. Hypertens. Res. 2017, 40, 41–50. [Google Scholar] [CrossRef]
- Leitner, B.P.; Huang, S.; Brychta, R.J.; Duckworth, C.J.; Baskin, A.S.; McGehee, S.; Tal, I.; Dieckmann, W.; Gupta, G.; Kolodny, G.M.; et al. Mapping of human brown adipose tissue in lean and obese young men. Proc. Natl. Acad. Sci. USA 2017, 114, 8649–8654. [Google Scholar] [CrossRef]
- Sakers, A.; De Siqueira, M.K.; Seale, P.; Villanueva, C.J. Adipose-tissue plasticity in health and disease. Cell 2022, 185, 419–446. [Google Scholar] [CrossRef]
- Stanek, A.; Brozyna-Tkaczyk, K.; Myslinski, W. The Role of Obesity-Induced Perivascular Adipose Tissue (PVAT) Dysfunction in Vascular Homeostasis. Nutrients 2021, 13, 3843. [Google Scholar] [CrossRef]
- Karaskova, E.; Velganova-Veghova, M.; Geryk, M.; Foltenova, H.; Kucerova, V.; Karasek, D. Role of Adipose Tissue in Inflammatory Bowel Disease. Int. J. Mol. Sci. 2021, 22, 4226. [Google Scholar] [CrossRef]
- Hwang, N.; Kang, D.; Shin, S.J.; Yoon, B.K.; Chun, J.; Kim, J.W.; Fang, S. Creeping fat exhibits distinct Inflammation-specific adipogenic preadipocytes in Crohn’s disease. Front. Immunol. 2023, 14, 1198905. [Google Scholar] [CrossRef]
- Candela, J.; Wang, R.; White, C. Microvascular Endothelial Dysfunction in Obesity Is Driven by Macrophage-Dependent Hydrogen Sulfide Depletion. Arterioscler. Thromb. Vasc. Biol. 2017, 37, 889–899. [Google Scholar] [CrossRef] [PubMed]
- Zou, L.; Wang, W.; Liu, S.; Zhao, X.; Lyv, Y.; Du, C.; Su, X.; Geng, B.; Xu, G. Spontaneous hypertension occurs with adipose tissue dysfunction in perilipin-1 null mice. Biochim. Biophys. Acta 2016, 1862, 182–191. [Google Scholar] [CrossRef] [PubMed]
- Henrichot, E.; Juge-Aubry, C.E.; Pernin, A.; Pache, J.C.; Velebit, V.; Dayer, J.M.; Meda, P.; Chizzolini, C.; Meier, C.A. Production of chemokines by perivascular adipose tissue: A role in the pathogenesis of atherosclerosis? Arterioscler. Thromb. Vasc. Biol. 2005, 25, 2594–2599. [Google Scholar] [CrossRef] [PubMed]
- Nosalski, R.; Guzik, T.J. Perivascular adipose tissue inflammation in vascular disease. Br. J. Pharmacol. 2017, 174, 3496–3513. [Google Scholar] [CrossRef]
- Ajuebor, M.N.; Swain, M.G. Role of chemokines and chemokine receptors in the gastrointestinal tract. Immunology 2002, 105, 137–143. [Google Scholar] [CrossRef]
- Chan, C.T.; Moore, J.P.; Budzyn, K.; Guida, E.; Diep, H.; Vinh, A.; Jones, E.S.; Widdop, R.E.; Armitage, J.A.; Sakkal, S.; et al. Reversal of vascular macrophage accumulation and hypertension by a CCR2 antagonist in deoxycorticosterone/salt-treated mice. Hypertension 2012, 60, 1207–1212. [Google Scholar] [CrossRef]
- DeLeon-Pennell, K.Y.; Iyer, R.P.; Ero, O.K.; Cates, C.A.; Flynn, E.R.; Cannon, P.L.; Jung, M.; Shannon, D.; Garrett, M.R.; Buchanan, W.; et al. Periodontal-induced chronic inflammation triggers macrophage secretion of Ccl12 to inhibit fibroblast-mediated cardiac wound healing. JCI Insight 2017, 2. [Google Scholar] [CrossRef]
- Stronati, L.; Palone, F.; Negroni, A.; Colantoni, E.; Mancuso, A.B.; Cucchiara, S.; Cesi, V.; Isoldi, S.; Vitali, R. Dipotassium Glycyrrhizate Improves Intestinal Mucosal Healing by Modulating Extracellular Matrix Remodeling Genes and Restoring Epithelial Barrier Functions. Front. Immunol. 2019, 10, 939. [Google Scholar] [CrossRef]
- Lau, W.B.; Zhang, Y.; Zhao, J.; Liu, B.; Wang, X.; Yuan, Y.; Christopher, T.A.; Lopez, B.; Gao, E.; Koch, W.J.; et al. Lymphotoxin-alpha is a novel adiponectin expression suppressor following myocardial ischemia/reperfusion. Am. J. Physiol. Endocrinol. Metab. 2013, 304, E661–E667. [Google Scholar] [CrossRef][Green Version]
- Sowka, A.; Dobrzyn, P. Role of Perivascular Adipose Tissue-Derived Adiponectin in Vascular Homeostasis. Cells 2021, 10, 1485. [Google Scholar] [CrossRef]
- Cardilo-Reis, L.; Gruber, S.; Schreier, S.M.; Drechsler, M.; Papac-Milicevic, N.; Weber, C.; Wagner, O.; Stangl, H.; Soehnlein, O.; Binder, C.J. Interleukin-13 protects from atherosclerosis and modulates plaque composition by skewing the macrophage phenotype. EMBO Mol. Med. 2012, 4, 1072–1086. [Google Scholar] [CrossRef] [PubMed]
- Zhong, Y.; Zhang, X.; Chong, W. Interleukin-24 Immunobiology and Its Roles in Inflammatory Diseases. Int. J. Mol. Sci. 2022, 23, 627. [Google Scholar] [CrossRef] [PubMed]
- Kim, E.Y.; Noh, H.M.; Choi, B.; Park, J.E.; Kim, J.E.; Jang, Y.; Lee, H.K.; Chang, E.J. Interleukin-22 Induces the Infiltration of Visceral Fat Tissue by a Discrete Subset of Duffy Antigen Receptor for Chemokine-Positive M2-Like Macrophages in Response to a High Fat Diet. Cells 2019, 8, 1587. [Google Scholar] [CrossRef]
- Mukherjee, S.; Aseer, K.R.; Yun, J.W. Roles of Macrophage Colony Stimulating Factor in White and Brown Adipocytes. Biomed. Eng. 2020, 25, 29–38. [Google Scholar] [CrossRef]
- Ajuebor, M.N.; Kunkel, S.L.; Hogaboam, C.M. The role of CCL3/macrophage inflammatory protein-1alpha in experimental colitis. Eur. J. Pharmacol. 2004, 497, 343–349. [Google Scholar] [CrossRef]
- Tourniaire, F.; Romier-Crouzet, B.; Lee, J.H.; Marcotorchino, J.; Gouranton, E.; Salles, J.; Malezet, C.; Astier, J.; Darmon, P.; Blouin, E.; et al. Chemokine Expression in Inflamed Adipose Tissue Is Mainly Mediated by NF-kappaB. PLoS ONE 2013, 8, e66515. [Google Scholar] [CrossRef]
- Griffin, M.J. On the Immunometabolic Role of NF-kappaB in Adipocytes. Immunometabolism 2022, 4, e220003. [Google Scholar] [CrossRef] [PubMed]
- McDaniel, D.K.; Eden, K.; Ringel, V.M.; Allen, I.C. Emerging Roles for Noncanonical NF-kappaB Signaling in the Modulation of Inflammatory Bowel Disease Pathobiology. Inflamm. Bowel Dis. 2016, 22, 2265–2279. [Google Scholar] [CrossRef]
- Committee for the Update of the Guide for the Care and Use of Laboratory Animals, N.R.C.U.S. Guide for the Care and Use of Laboratory Animals, 8th ed.; National Academies Press: Washington, DC, USA, 2011; p. xxv. 220p. [Google Scholar]
- Boerman, E.M.; Segal, S.S. Depressed perivascular sensory innervation of mouse mesenteric arteries with advanced age. J. Physiol. 2016, 594, 2323–2338. [Google Scholar] [CrossRef]
- Schindelin, J.; Arganda-Carreras, I.; Frise, E.; Kaynig, V.; Longair, M.; Pietzsch, T.; Preibisch, S.; Rueden, C.; Saalfeld, S.; Schmid, B.; et al. Fiji: An open-source platform for biological-image analysis. Nat. Methods 2012, 9, 676–682. [Google Scholar] [CrossRef]
- Han, H.; Cho, J.W.; Lee, S.; Yun, A.; Kim, H.; Bae, D.; Yang, S.; Kim, C.Y.; Lee, M.; Kim, E.; et al. TRRUST v2: An expanded reference database of human and mouse transcriptional regulatory interactions. Nucleic Acids Res. 2018, 46, D380–D386. [Google Scholar] [CrossRef] [PubMed]
- Pinero, J.; Ramirez-Anguita, J.M.; Sauch-Pitarch, J.; Ronzano, F.; Centeno, E.; Sanz, F.; Furlong, L.I. The DisGeNET knowledge platform for disease genomics: 2019 update. Nucleic Acids Res. 2020, 48, D845–D855. [Google Scholar] [CrossRef] [PubMed]
- Hu, C.; Li, T.; Xu, Y.; Zhang, X.; Li, F.; Bai, J.; Chen, J.; Jiang, W.; Yang, K.; Ou, Q.; et al. CellMarker 2.0: An updated database of manually curated cell markers in human/mouse and web tools based on scRNA-seq data. Nucleic Acids Res. 2023, 51, D870–D876. [Google Scholar] [CrossRef]
- Xie, Z.; Bailey, A.; Kuleshov, M.V.; Clarke, D.J.B.; Evangelista, J.E.; Jenkins, S.L.; Lachmann, A.; Wojciechowicz, M.L.; Kropiwnicki, E.; Jagodnik, K.M.; et al. Gene Set Knowledge Discovery with Enrichr. Curr. Protoc. 2021, 1, e90. [Google Scholar] [CrossRef] [PubMed]
| Gene | Description | Control | IBD | Fold Change | p-Value | ||
|---|---|---|---|---|---|---|---|
| Ct | SD | Ct | SD | ||||
| Ltb | Lymphotoxin B | 26.44 | 0.58 | 23.84 | 0.36 | 3.93 | 0.05 |
| Ccl12 | Chemokine (C-C motif) ligand 12 | 28.4 | 1.12 | 26.07 | 0.52 | 3.27 | 0.05 |
| Lta | Lymphotoxin A | 30.9 | 0.23 | 28.67 | 0.25 | 3.04 | 0.14 |
| Cxcl13 | Chemokine (C-X-C motif) ligand 13 | 22.94 | 0.78 | 20.84 | 0.35 | 2.8 | 0.04 |
| Cxcl1 | Chemokine (C-X-C motif) ligand 1 | 31.82 | 1.34 | 29.79 | 0.32 | 2.66 | 0.13 |
| Ccl5 | Chemokine (C-C motif) ligand 5 | 25.52 | 0.54 | 23.55 | 0.45 | 2.56 | 0.07 |
| Cxcl16 | Chemokine (C-X-C motif) ligand 16 | 27.26 | 0.8 | 25.55 | 0.41 | 2.12 | 0.05 |
| Il16 | Interleukin 16 | 26.9 | 0.65 | 25.43 | 0.39 | 1.79 | 0.15 |
| Il6 | Interleukin 6 | 31.08 | 0.1 | 29.64 | 0.28 | 1.76 | 0.96 |
| B2m | Beta-2 microglobulin | 18.54 | 0.49 | 17.23 | 0.57 | 1.6 | 0.17 |
| Il17a | Interleukin 17A | 32.84 | 0.23 | 31.54 | 0.15 | 1.6 | 0.6 |
| Il1b | Interleukin 1 beta | 28.37 | 0.64 | 27.13 | 0.28 | 1.53 | 0.34 |
| Tnf | Tumor necrosis factor | 29.03 | 0.1 | 27.82 | 0.28 | 1.51 | 0.98 |
| Ccl1 | Chemokine (C-C motif) ligand 1 | 32.95 | 0.33 | 31.79 | 0.48 | 1.45 | 0.42 |
| Ppbp | Pro-platelet basic protein | 27.67 | 0.38 | 26.52 | 0.7 | 1.45 | 0.86 |
| Ifna2 | Interferon alpha 2 | 30.86 | 1.66 | 29.77 | 0.74 | 1.38 | 0.34 |
| Ccl7 | Chemokine (C-C motif) ligand 7 | 25.29 | 1.13 | 24.22 | 0.7 | 1.36 | 0.7 |
| Ccl22 | Chemokine (C-C motif) ligand 22 | 29.61 | 0.74 | 28.55 | 0.53 | 1.36 | 0.94 |
| Tgfb2 | Transforming growth factor, beta 2 | 27.5 | 0.95 | 26.45 | 0.49 | 1.35 | 0.32 |
| Il4 | Interleukin 4 | 29.48 | 0.36 | 28.43 | 0.47 | 1.35 | 0.62 |
| Bmp2 | Bone morphogenetic protein 2 | 28.69 | 0.82 | 27.67 | 0.71 | 1.32 | 0.36 |
| Fasl | Fas ligand (TNF superfamily, member 6) | 29.78 | 0.21 | 28.77 | 0.13 | 1.31 | 0.91 |
| Gpi1 | Glucose phosphate isomerase 1 | 21.27 | 0.74 | 20.27 | 0.33 | 1.3 | 0.25 |
| Cxcl5 | Chemokine (C-X-C motif) ligand 5 | 30.24 | 0.33 | 29.27 | 0.59 | 1.27 | 0.53 |
| Pf4 | Platelet factor 4 | 23.67 | 0.84 | 22.71 | 0.68 | 1.26 | 0.27 |
| Nodal | Nodal | 30.87 | 0.61 | 29.94 | 0.4 | 1.24 | 0.55 |
| Tnfsf13b | Tumor necrosis factor superfamily, 13b | 26.59 | 0.89 | 25.67 | 0.42 | 1.23 | 0.38 |
| Il21 | Interleukin 21 | 30.63 | 0.54 | 29.71 | 0.25 | 1.23 | 0.94 |
| Tnfsf10 | Tumor necrosis factor superfamily, member 10 | 25.92 | 0.33 | 25.02 | 0.13 | 1.22 | 0.95 |
| Il11 | Interleukin 11 | 30.51 | 0.86 | 29.61 | 0.1 | 1.2 | 0.71 |
| Spp1 | Secreted phosphoprotein 1 | 29.3 | 0.51 | 28.41 | 0.53 | 1.2 | 0.83 |
| Tnfsf11 | Tumor necrosis factor superfamily, member 11 | 28.54 | 0.25 | 27.67 | 0.45 | 1.19 | 0.82 |
| Gapdh | Glyceraldehyde-3-phosphate dehydrogenase | 19.74 | 0.78 | 18.88 | 0.57 | 1.18 | 0.28 |
| Cxcl10 | Chemokine (C-X-C motif) ligand 10 | 24.81 | 0.66 | 23.96 | 0.39 | 1.18 | 0.4 |
| Ccl2 | Chemokine (C-C motif) ligand 2 | 25.52 | 0.91 | 24.68 | 0.71 | 1.16 | 0.53 |
| Il18 | Interleukin 18 | 25.58 | 1 | 24.74 | 0.47 | 1.16 | 0.8 |
| Mif | Macrophage migration inhibitory factor | 21.88 | 0.69 | 21.06 | 0.44 | 1.15 | 0.29 |
| Ccl19 | Chemokine (C-C motif) ligand 19 | 24.28 | 0.47 | 23.47 | 0.25 | 1.14 | 0.73 |
| Ifng | Interferon gamma | 28.7 | 0.36 | 27.91 | 0.39 | 1.12 | 0.93 |
| Ccl24 | Chemokine (C-C motif) ligand 24 | 29.39 | 1.24 | 28.62 | 0.83 | 1.11 | 0.96 |
| Cd40lg | CD40 ligand | 28.78 | 0.24 | 28.02 | 0.39 | 1.1 | 0.66 |
| Adipoq | Adiponectin, C1Q and collagen domain containing | 19.16 | 0.7 | 18.42 | 0.7 | 1.09 | 0.62 |
| Tnfrsf11b | Tumor necrosis factor receptor superfamily, member 11b | 25.67 | 1.11 | 24.96 | 0.7 | 1.07 | 0.9 |
| Xcl1 | Chemokine (C motif) ligand 1 | 27.99 | 0.32 | 27.28 | 0.35 | 1.06 | 0.69 |
| Il1rn | Interleukin 1 receptor antagonist | 28.55 | 0.44 | 27.85 | 0.32 | 1.05 | 0.9 |
| Hsp90ab1 | Heat shock protein 90 alpha, class B member 1 | 19.29 | 0.76 | 18.62 | 0.41 | 1.03 | 0.66 |
| Ccl11 | Chemokine (C-C motif) ligand 11 | 24.31 | 0.69 | 23.67 | 0.68 | 1.01 | 0.77 |
| Gene | Description | Control | IBD | Fold Change | p-Value | ||
|---|---|---|---|---|---|---|---|
| Ct | SD | Ct | SD | ||||
| Il13 | Interleukin 13 | 28.75 | 0.24 | 29.39 | 0.47 | 0.42 | 0.17 |
| Il24 | Interleukin 24 | 28.87 | 0.11 | 29.38 | 0.32 | 0.46 | 0.16 |
| Csf3 | Colony stimulating factor 3 | 29.43 | 0.28 | 30.89 | 0.58 | 0.37 | 0.05 |
| Ccl3 | Chemokine (C-C motif) ligand 3 | 28.61 | 0.67 | 29 | 0.5 | 0.5 | 0.02 |
| Il22 | Interleukin 22 | 28.63 | 0.33 | 28.96 | 0.46 | 0.5 | 0.13 |
| Csf2 | Colony stimulating factor 2 | 30.07 | 0.45 | 30.39 | 0.37 | 0.5 | 0.16 |
| Cd70 | CD70 antigen | 29.76 | 0.44 | 30.08 | 0.87 | 0.5 | 0.22 |
| Il2 | Interleukin 2 | 29.88 | 0.15 | 30.13 | 0.41 | 0.55 | 0.64 |
| Hc | Hemolytic complement | 29.02 | 0.39 | 29.2 | 0.34 | 0.57 | 0.14 |
| Cxcl3 | Chemokine (C-X-C motif) ligand 3 | 29.98 | 0.25 | 30.14 | 0.3 | 0.58 | 0.28 |
| Il3 | Interleukin 3 | 29.56 | 0.2 | 29.67 | 0.32 | 0.6 | 0.29 |
| Mstn | Myostatin | 29.57 | 0.35 | 29.66 | 0.36 | 0.61 | 0.19 |
| Il23a | Interleukin 23, alpha subunit p19 | 28.97 | 0.36 | 28.97 | 0.51 | 0.65 | 0.22 |
| Cxcl11 | Chemokine (C-X-C motif) ligand 11 | 26.78 | 0.46 | 26.73 | 0.42 | 0.67 | 0.22 |
| Thpo | Thrombopoietin | 27.29 | 0.71 | 27.18 | 0.51 | 0.7 | 0.01 |
| Cntf | Ciliary neurotrophic factor | 27.84 | 0.58 | 27.72 | 0.41 | 0.71 | 0.39 |
| Il5 | Interleukin 5 | 28.99 | 0.33 | 28.84 | 0.44 | 0.73 | 0.29 |
| Cxcl12 | Chemokine (C-X-C motif) ligand 12 | 22.05 | 0.72 | 21.85 | 0.38 | 0.75 | 0.21 |
| Il1a | Interleukin 1 alpha | 28.76 | 0.41 | 28.56 | 0.29 | 0.75 | 0.54 |
| Bmp4 | Bone morphogenetic protein 4 | 25.29 | 0.93 | 25.07 | 0.58 | 0.76 | 0.36 |
| Il7 | Interleukin 7 | 26.21 | 0.77 | 25.96 | 0.46 | 0.77 | 0.14 |
| Il17f | Interleukin 17F | 29.58 | 0.37 | 29.35 | 0.45 | 0.77 | 0.25 |
| Ccl20 | Chemokine (C-C motif) ligand 20 | 31.2 | 0.15 | 30.94 | 0.38 | 0.78 | 0.41 |
| Bmp6 | Bone morphogenetic protein 6 | 24.81 | 0.63 | 24.53 | 0.26 | 0.79 | 0.5 |
| Il15 | Interleukin 15 | 25.48 | 0.64 | 25.17 | 0.34 | 0.8 | 0.31 |
| Vegfa | Vascular endothelial growth factor A | 20.95 | 0.71 | 20.63 | 0.37 | 0.81 | 0.36 |
| Csf1 | Colony stimulating factor 1 | 23.06 | 0.79 | 22.7 | 0.48 | 0.83 | 0.04 |
| Bmp7 | Bone morphogenetic protein 7 | 27.18 | 0.86 | 26.8 | 0.5 | 0.84 | 0.33 |
| Ctf1 | Cardiotrophin 1 | 26.71 | 0.75 | 26.32 | 0.49 | 0.85 | 0.25 |
| Il12a | Interleukin 12A | 28.66 | 0.38 | 28.28 | 0.31 | 0.85 | 0.48 |
| Il9 | Interleukin 9 | 29.66 | 0.35 | 29.25 | 0.27 | 0.86 | 0.61 |
| Ccl17 | Chemokine (C-C motif) ligand 17 | 28.44 | 0.29 | 28.02 | 0.32 | 0.87 | 0.46 |
| Cx3cl1 | Chemokine (C-X3-C motif) ligand 1 | 27.4 | 0.69 | 26.94 | 0.39 | 0.89 | 0.56 |
| Ccl4 | Chemokine (C-C motif) ligand 4 | 28.24 | 0.54 | 27.79 | 0.35 | 0.89 | 0.74 |
| Cxcl9 | Chemokine (C-X-C motif) ligand 9 | 27.7 | 0.12 | 27.21 | 0.54 | 0.92 | 0.43 |
| Osm | Oncostatin M | 28.93 | 0.51 | 28.43 | 0.46 | 0.92 | 0.52 |
| Lif | Leukemia inhibitory factor | 29.05 | 0.61 | 28.5 | 0.23 | 0.95 | 0.99 |
| Il12b | Interleukin 12b | 28.63 | 0.3 | 28.06 | 0.32 | 0.96 | 0.76 |
| Gusb | Glucuronidase, beta | 23.11 | 0.74 | 22.52 | 0.54 | 0.97 | 0.8 |
| Il27 | Interleukin 27 | 29.31 | 0.44 | 28.7 | 0.65 | 0.99 | 0.65 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jenkins, S.W., III; Grunz, E.A.; Ramos, K.R.; Boerman, E.M. Perivascular Adipose Tissue Becomes Pro-Contractile and Remodels in an IL10−/− Colitis Model of Inflammatory Bowel Disease. Int. J. Mol. Sci. 2024, 25, 10726. https://doi.org/10.3390/ijms251910726
Jenkins SW III, Grunz EA, Ramos KR, Boerman EM. Perivascular Adipose Tissue Becomes Pro-Contractile and Remodels in an IL10−/− Colitis Model of Inflammatory Bowel Disease. International Journal of Molecular Sciences. 2024; 25(19):10726. https://doi.org/10.3390/ijms251910726
Chicago/Turabian StyleJenkins, Samuel W., III, Elizabeth A. Grunz, Kassandra R. Ramos, and Erika M. Boerman. 2024. "Perivascular Adipose Tissue Becomes Pro-Contractile and Remodels in an IL10−/− Colitis Model of Inflammatory Bowel Disease" International Journal of Molecular Sciences 25, no. 19: 10726. https://doi.org/10.3390/ijms251910726
APA StyleJenkins, S. W., III, Grunz, E. A., Ramos, K. R., & Boerman, E. M. (2024). Perivascular Adipose Tissue Becomes Pro-Contractile and Remodels in an IL10−/− Colitis Model of Inflammatory Bowel Disease. International Journal of Molecular Sciences, 25(19), 10726. https://doi.org/10.3390/ijms251910726







