Descriptive Analysis of Common Fusion Mutations in Papillary Thyroid Carcinoma in Hungary
Abstract
:1. Introduction
2. Results
2.1. Study Population
2.2. Descriptive Analysis
2.3. Principal Component Analysis (PCA) Based on d-Correlation of Mixed Scale-Type Variables of Fusion Mutation Status and Clinicopathological Data
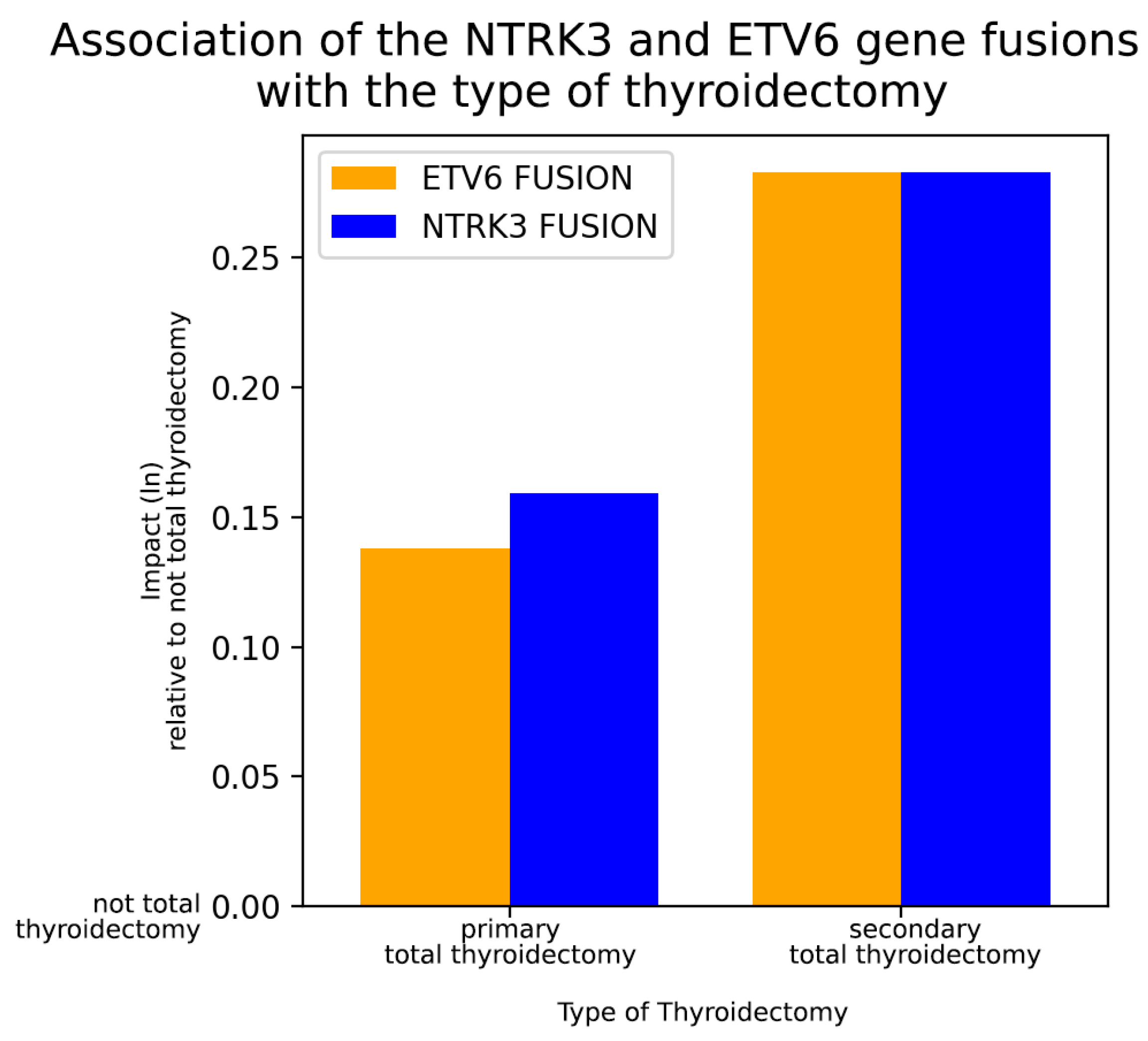
2.4. Deeper Analysis for Associations between NTRK3 and ETV6 Fusions and Clinicopathological Variables
2.5. Analysis for Associations between Ratio-Scale, Ordinal, and Clinicopathological Variables
2.6. Outlook toward Not Fusion-Related Clinicopathological Associations of PTC Patients
3. Discussion
4. Materials and Methods
4.1. Study Population, Sample Collection, and Histopathological Processing
4.2. Molecular Processing for Identification of Fusion Mutations (RNA Isolation, Quality Control (QC), RNA Quantification and Sequencing)
4.3. Data Analysis via Bioinformatics and Statistical Evaluation
4.4. Literature Search
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| Abbreviation | Meaning |
| ABL1 | tyrosine-protein kinase ABL1 gene |
| AJCC | American Joint Committee on Cancer |
| AKT3 | AKT serine/threonine kinase 3 gene |
| ALK | anaplastic lymphoma kinase gene |
| ATA | American Thyroid Association |
| AXL | AXL receptor tyrosine kinase gene |
| BCR | breakpoint cluster region protein gene |
| BRAF | proto-oncogene B-Raf |
| CA | California |
| CNV | copy number variation |
| DNA | deoxyribonucleic acid |
| DTC | differentiated thyroid cancer |
| EBRT | external beam radiation therapy |
| EGFR | epidermal growth factor receptor gene |
| ERBB2 | erb-b2 receptor tyrosine kinase 2 gene |
| ERG | ETS-related gene |
| ETV | ETS variant transcription factor gene |
| FFPE | formalin-fixed paraffin-embedded |
| FGFR | fibroblast growth factor receptor gene |
| FISH | fluorescence in situ hybridization |
| FNAB | fine-needle aspiration biopsy |
| FTC | follicular thyroid carcinoma |
| FuPa | fragmentase universal primer assay |
| I-131 | iodine-131 isotope |
| IHC | immunohistochemistry |
| IL | Illinois |
| ISP | Ion Sphere Particle |
| MAPK | mitogen-activated protein kinase |
| MET | c-Met gene |
| miRNA | micro-ribonucleic acid |
| NCBI | National Center for Biotechnology Information |
| NGS | next-generation sequencing |
| NTRK | neurotrophic tyrosine receptor kinase gene |
| PCA | principal component analysis |
| PCR | polymerase chain reaction |
| PDGFRA | platelet-derived growth factor receptor A gene |
| PI3K | phosphatidylinositol 3-kinase |
| PLCγ | phosphatidylinositol phospholipase C γ |
| PPARG | peroxisome proliferator-activated receptor γ gene |
| PTC | papillary thyroid carcinoma |
| QC | quality control |
| qPCR | quantitative polymerase chain reaction |
| R stage | residual tumor classification |
| RAF1 | Raf-1 proto-oncogene |
| RAI | radioiodine therapy |
| RAS | rat sarcoma virus gene |
| RET | ret proto-oncogene |
| RNA | ribonucleic acid |
| ROS1 | ROS proto-oncogene 1 |
| SD | standard deviation |
| TNM | TNM Classification of Malignant Tumors |
| USA | United States of America |
References
- World Cancer Research Fund International. Available online: https://www.wcrf.org/cancer-trends/worldwide-cancer-data (accessed on 31 August 2024).
- Fathimabeebi, P.; Noor Al, B.; Hilal Al, M. Epidemiology of Thyroid Cancer in Oman. Ann. Endocrinol. Metab. 2017, 1, 11–17. [Google Scholar] [CrossRef]
- National Cancer Institute. Available online: https://seer.cancer.gov/statfacts/html/thyro.html (accessed on 31 August 2024).
- Olson, E.; Wintheiser, G.; Wolfe, K.M.; Droessler, J.; Silberstein, P.T. Epidemiology of Thyroid Cancer: A Review of the National Cancer Database, 2000–2013. Cureus 2019, 11, e4127. [Google Scholar] [CrossRef] [PubMed]
- Prete, A.; Borges de Souza, P.; Censi, S.; Muzza, M.; Nucci, N.; Sponziello, M. Update on Fundamental Mechanisms of Thyroid Cancer. Front. Endocrinol. 2020, 11, 102. [Google Scholar] [CrossRef] [PubMed]
- Chiba, T. Molecular Pathology of Thyroid Tumors: Essential Points to Comprehend Regarding the Latest WHO Classification. Biomedicines 2024, 12, 712. [Google Scholar] [CrossRef] [PubMed]
- Haugen, B.R.; Alexander, E.K.; Bible, K.C.; Doherty, G.M.; Mandel, S.J.; Nikiforov, Y.E.; Pacini, F.; Randolph, G.W.; Sawka, A.M.; Schlumberger, M.; et al. 2015 American Thyroid Association Management Guidelines for Adult Patients with Thyroid Nodules and Differentiated Thyroid Cancer: The American Thyroid Association Guidelines Task Force on Thyroid Nodules and Differentiated Thyroid Cancer. Thyroid 2016, 26, 1–133. [Google Scholar] [CrossRef]
- Wu, J.; Hu, X.Y.; Ghaznavi, S.; Kinnear, S.; Symonds, C.J.; Grundy, P.; Parkins, V.M.; Sharma, P.; Lamb, D.; Khalil, M.; et al. The Prospective Implementation of the 2015 ATA Guidelines and Modified ATA Recurrence Risk Stratification System for Treatment of Differentiated Thyroid Cancer in a Canadian Tertiary Care Referral Setting. Thyroid 2022, 32, 1509–1518. [Google Scholar] [CrossRef]
- Ghosh, R.; Auh, S.; Gubbi, S.; Veeraraghavan, P.; Cochran, C.; Shobab, L.; Burman, K.D.; Wartofsky, L.; Klubo-Gwiezdzinska, J. Thyroid SAT519 Association of Free Thyroxine Level with Survival Outcome in Patients with Intermediate and High-Risk Differentiated Thyroid Cancer. J. Endocrine Soc. 2023, 7, bvad114-1991. [Google Scholar] [CrossRef]
- Borowczyk, M.; Kaczmarek-Ryś, M.; Hryhorowicz, S.; Sypniewski, M.; Filipowicz, D.; Dobosz, P.; Oszywa, M.; Ruchała, M.; Ziemnicka, K. Germline polymorphisms of the NOD2 pathway may predict the effectiveness of radioiodine in differentiated thyroid cancer treatment. J. Endocrinol. Investig. 2024. [Google Scholar] [CrossRef]
- Ulisse, S.; Baldini, E.; Lauro, A.; Pironi, D.; Tripodi, D.; Lori, E.; Ferent, I.C.; Amabile, M.I.; Catania, A.; Di Matteo, F.M.; et al. Papillary Thyroid Cancer Prognosis: An Evolving Field. Cancers 2021, 13, 5567. [Google Scholar] [CrossRef]
- Harrison’s Principles of Internal Medicine, 20th ed.; McGraw-Hill: New York, NY, USA, 2022.
- Lorusso, L.; Cappagli, V.; Valerio, L.; Giani, C.; Viola, D.; Puleo, L.; Gambale, C.; Minaldi, E.; Campopiano, M.C.; Matrone, A.; et al. Thyroid cancers: From surgery to current and future systemic therapies through their molecular identities. Int. J. Mol. Sci. 2021, 22, 3117. [Google Scholar] [CrossRef]
- Schlumberger, M.; Tahara, M.; Wirth, L.J.; Robinson, B.; Brose, M.S.; Elisei, R.; Habra, M.A.; Newbold, K.; Shah, M.H.; Hoff, A.O.; et al. Lenvatinib versus Placebo in Radioiodine-Refractory Thyroid Cancer. N. Engl. J. Med. 2015, 372, 621–630. [Google Scholar] [CrossRef] [PubMed]
- Cortas, C.; Charalambous, H. Tyrosine Kinase Inhibitors for Radioactive Iodine Refractory Differentiated Thyroid Cancer. Life 2024, 14, 22. [Google Scholar] [CrossRef] [PubMed]
- Silaghi, H.; Lozovanu, V.; Georgescu, C.E.; Pop, C.; Nasui, B.A.; Cătoi, A.F.; Silaghi, C.A. State of the Art in the Current Management and Future Directions of Targeted Therapy for Differentiated Thyroid Cancer. Int. J. Mol. Sci. 2022, 23, 3470. [Google Scholar] [CrossRef] [PubMed]
- Xing, M. Molecular pathogenesis and mechanisms of thyroid cancer. Nat. Rev. Cancer 2013, 13, 184–199. [Google Scholar] [CrossRef]
- Elisei, R.; Grande, E.; Kreissl, M.C.; Leboulleux, S.; Puri, T.; Fasnacht, N.; Capdevila, J. Current perspectives on the management of patients with advanced RET-driven thyroid cancer in Europe. Front. Oncol. 2023, 13, 1141314. [Google Scholar] [CrossRef]
- Agrawal, N.; Akbani, R.; Aksoy, B.A.; Ally, A.; Arachchi, H.; Asa, S.L.; Auman, J.T.; Balasundaram, M.; Balu, S.; Baylin, S.B.; et al. Integrated Genomic Characterization of Papillary Thyroid Carcinoma. Cell 2014, 159, 676–690. [Google Scholar] [CrossRef]
- Armos, R.; Bojtor, B.; Papp, M.; Illyes, I.; Lengyel, B.; Kiss, A.; Szili, B.; Tobias, B.; Balla, B.; Piko, H.; et al. MicroRNA Profiling in Papillary Thyroid Cancer. Int. J. Mol. Sci. 2024, 25, 9362. [Google Scholar] [CrossRef]
- Taniue, K.; Akimitsu, N. Fusion genes and RNAs in cancer development. Non-Coding RNA 2021, 7, 10. [Google Scholar] [CrossRef]
- Ju, G.; Sun, Y.; Wang, H.; Zhang, X.; Mu, Z.; Sun, D.; Huang, L.; Lin, R.; Xing, T.; Cheng, W.; et al. Fusion Oncogenes in Patients With Locally Advanced or Distant Metastatic Differentiated Thyroid Cancer. J. Clin. Endocrinol. Metab. 2024, 109, 505–515. [Google Scholar] [CrossRef]
- Yakushina, V.D.; Lerner, L.V.; Lavrov, A.V. Gene fusions in thyroid cancer. Thyroid 2018, 28, 158–167. [Google Scholar] [CrossRef]
- Lu, Z.; Zhang, Y.; Feng, D.; Sheng, J.; Yang, W.; Liu, B. Targeted Next Generation Sequencing Identifies Somatic Mutations and Gene Fusions in Papillary Thyroid Carcinoma. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5542227/ (accessed on 31 August 2024).
- ClinicalTrials.gov. Cabozantinib-S-Malate in Treating Patients with Refractory Thyroid Cancer. Available online: https://clinicaltrials.gov/study/NCT01811212 (accessed on 31 August 2024).
- ClinicalTrials.gov. Larotrectinib to Enhance RAI Avidity in Differentiated Thyroid Cancer. Available online: https://clinicaltrials.gov/study/NCT05783323 (accessed on 31 August 2024).
- European Medicines Agency. Selpercatinib. Available online: https://www.ema.europa.eu/en/medicines/human/EPAR/retsevmo (accessed on 31 August 2024).
- Toda, S.; Iwasaki, H.; Okubo, Y.; Hayashi, H.; Kadoya, M.; Takahashi, H.; Yokose, T.; Hiroshima, Y.; Masudo, K. The frequency of mutations in advanced thyroid cancer in Japan: A single-center study. Endocr. J. 2024, 71, 31–37. [Google Scholar] [CrossRef] [PubMed]
- Pekova, B.B.; Sykorova, V.; Mastnikova, K.; Vaclavikova, E.; Moravcova, J.; Vlcek, P.; Lancova, L.; Lastuvka, P.; Katra, R.; Bavor, P.; et al. RET fusion genes in pediatric and adult thyroid carcinomas: Cohort characteristics and prognosis. Endocr. Relat. Cancer 2023, 30, e230117. [Google Scholar]
- Ji, J.H.; Oh, Y.L.; Hong, M.; Yun, J.W.; Lee, H.-W.; Kim, D.; Ji, Y.; Kim, D.-H.; Park, W.-Y.; Shin, H.-T.; et al. Identification of Driving ALK Fusion Genes and Genomic Landscape of Medullary Thyroid Cancer. PLoS Genet. 2015, 11, e1005467. [Google Scholar] [CrossRef] [PubMed]
- Nikiforova, M.N.; ENikiforov, Y. Molecular genetics of thyroid cancer: Implications for diagnosis, treatment and prognosis. Expert Rev. Mol. Diagn. 2008, 8, 83–95. [Google Scholar] [CrossRef] [PubMed]
- Glenfield, C.; Innan, H. Gene duplication and gene fusion are important drivers of tumourigenesis during cancer evolution. Genes 2021, 12, 1376. [Google Scholar] [CrossRef]
- Skálová, A.; Ptáková, N.; Santana, T.; Agaimy, A.; Ihrler, S.; Uro-Coste, E.; Thompson, L.D.; Bishop, J.A.; Baněčkova, M.; Rupp, N.J.; et al. NCOA4-RET and TRIM27-RET Are Characteristic Gene Fusions in Salivary Intraductal Carcinoma, Including Invasive and Metastatic Tumors Is “Intraductal” Correct? Am. J. Surg. Pathol. 2019, 43, 1303–1313. [Google Scholar] [CrossRef]
- Mertens, F.; Johansson, B.; Fioretos, T.; Mitelman, F. The emerging complexity of gene fusions in cancer. Nat. Rev. Cancer 2015, 15, 371–381. [Google Scholar] [CrossRef]
- Awad, M.M.; Oxnard, G.R.; Jackman, D.M.; Savukoski, D.O.; Hall, D.; Shivdasani, P.; Heng, J.C.; Dahlberg, S.E.; Jänne, P.A.; Verma, S.; et al. MET exon 14 mutations in Non-small-cell lung cancer are associated with advanced age and stage-dependent MET genomic amplification and c-Met overexpression. J. Clin. Oncol. 2016, 34, 721–730. [Google Scholar] [CrossRef]
- Podani, J.; Schmera, D.; Bagella, S. Correlating variables with different scale types: A new framework based on matrix comparisons. Methods Ecol. Evol. 2023, 14, 1049–1060. [Google Scholar] [CrossRef]
- Amin, M.B.; Greene, F.L.; Edge, S.B.; Compton, C.C.; Gershenwald, J.E.; Brookland, R.K.; Meyer, L.; Gress, D.M.; Byrd, D.R.; Winchester, D.P. The Eighth Edition AJCC Cancer Staging Manual: Continuing to build a bridge from a population-based to a more “personalized” approach to cancer staging. CA Cancer J. Clin. 2017, 67, 93–99. [Google Scholar] [CrossRef]
- Lee, Y.A.; Lee, H.; Im, S.-W.; Song, Y.S.; Oh, D.-Y.; Kang, H.J.; Won, J.-K.; Jung, K.C.; Kwon, D.; Chung, E.-J.; et al. NTRK and RET fusion–directed therapy in pediatric thyroid cancer yields a tumor response and radioiodine uptake. J. Clin. Investig. 2021, 131, e144847. [Google Scholar] [CrossRef] [PubMed]
- Yan, K.L.; Li, S.; Tseng, C.-H.; Kim, J.; Nguyen, D.T.; Dawood, N.B.; Livhits, M.J.; Yeh, M.W.; Leung, A.M. Rising incidence and incidence-based mortality of thyroid cancer in California, 2000–2017. J. Clin. Endocrinol. Metab. 2020, 105, 1770–1777. [Google Scholar] [CrossRef] [PubMed]
- Vuong, H.G.; Le, H.T.; Le, T.T.B.; Le, T.; Hassell, L.; Kakudo, K. Clinicopathological significance of major fusion oncogenes in papillary thyroid carcinoma: An individual patient data meta-analysis. Pathol. Res. Pract. 2022, 240, 154180. [Google Scholar] [CrossRef] [PubMed]
- Colombo, C.; Minna, E.; Gargiuli, C.; Muzza, M.; Dugo, M.; De Cecco, L.; Pogliaghi, G.; Tosi, D.; Bulfamante, G.; Greco, A.; et al. The molecular and gene/miRNA expression profiles of radioiodine resistant papillary thyroid cancer. J. Exp. Clin. Cancer Res. 2020, 39, 245. [Google Scholar] [CrossRef]
- Ullmann, T.M.; Thiesmeyer, J.W.; Lee, Y.J.; Beg, S.; Mosquera, J.M.; Elemento, O.; Fahey, T.J., III; Scognamiglio, T.; Houvras, Y. RET Fusion-Positive Papillary Thyroid Cancers are Associated with a More Aggressive Phenotype. Ann. Surg. Oncol. 2022, 29, 4266–4273. [Google Scholar] [CrossRef]
- McLeod, D.S.A.; Zhang, L.; Durante, C.; Cooper, D.S. Contemporary debates in adult papillary thyroid cancer management. Endocr. Rev. 2019, 40, 1481–1499. [Google Scholar] [CrossRef]





| PTC Subtype | n = 100 |
|---|---|
| conventional | 69 |
| follicular variant | 17 |
| oncocytic | 6 |
| tall cell | 3 |
| columnar cell | 2 |
| encapsulated conventional | 1 |
| trabecular | 1 |
| Warthin-like | 1 |
| Fusion Gene | Cancer Type |
|---|---|
| RET | thyroid carcinoma, salivary intraductal carcinoma |
| NTRK3 | breast carcinoma, fibrosarcoma |
| CCDC6 | thyroid carcinoma |
| ETV6 | acute lymphoblastic leukemia, breast carcinoma, fibrosarcoma |
| MET | NSCLC |
| ALK | anaplastic large T-cell lymphoma |
| NCOA4 | prostate cancer, salivary intraductal carcinoma |
| EML4 | lung cancer |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Armos, R.; Bojtor, B.; Podani, J.; Illyes, I.; Balla, B.; Putz, Z.; Kiss, A.; Kohanka, A.; Toth, E.; Takacs, I.; et al. Descriptive Analysis of Common Fusion Mutations in Papillary Thyroid Carcinoma in Hungary. Int. J. Mol. Sci. 2024, 25, 10787. https://doi.org/10.3390/ijms251910787
Armos R, Bojtor B, Podani J, Illyes I, Balla B, Putz Z, Kiss A, Kohanka A, Toth E, Takacs I, et al. Descriptive Analysis of Common Fusion Mutations in Papillary Thyroid Carcinoma in Hungary. International Journal of Molecular Sciences. 2024; 25(19):10787. https://doi.org/10.3390/ijms251910787
Chicago/Turabian StyleArmos, Richard, Bence Bojtor, Janos Podani, Ildiko Illyes, Bernadett Balla, Zsuzsanna Putz, Andras Kiss, Andrea Kohanka, Erika Toth, Istvan Takacs, and et al. 2024. "Descriptive Analysis of Common Fusion Mutations in Papillary Thyroid Carcinoma in Hungary" International Journal of Molecular Sciences 25, no. 19: 10787. https://doi.org/10.3390/ijms251910787








