Citric Acid Promotes Immune Function by Modulating the Intestinal Barrier
Abstract
:1. Introduction
2. Results
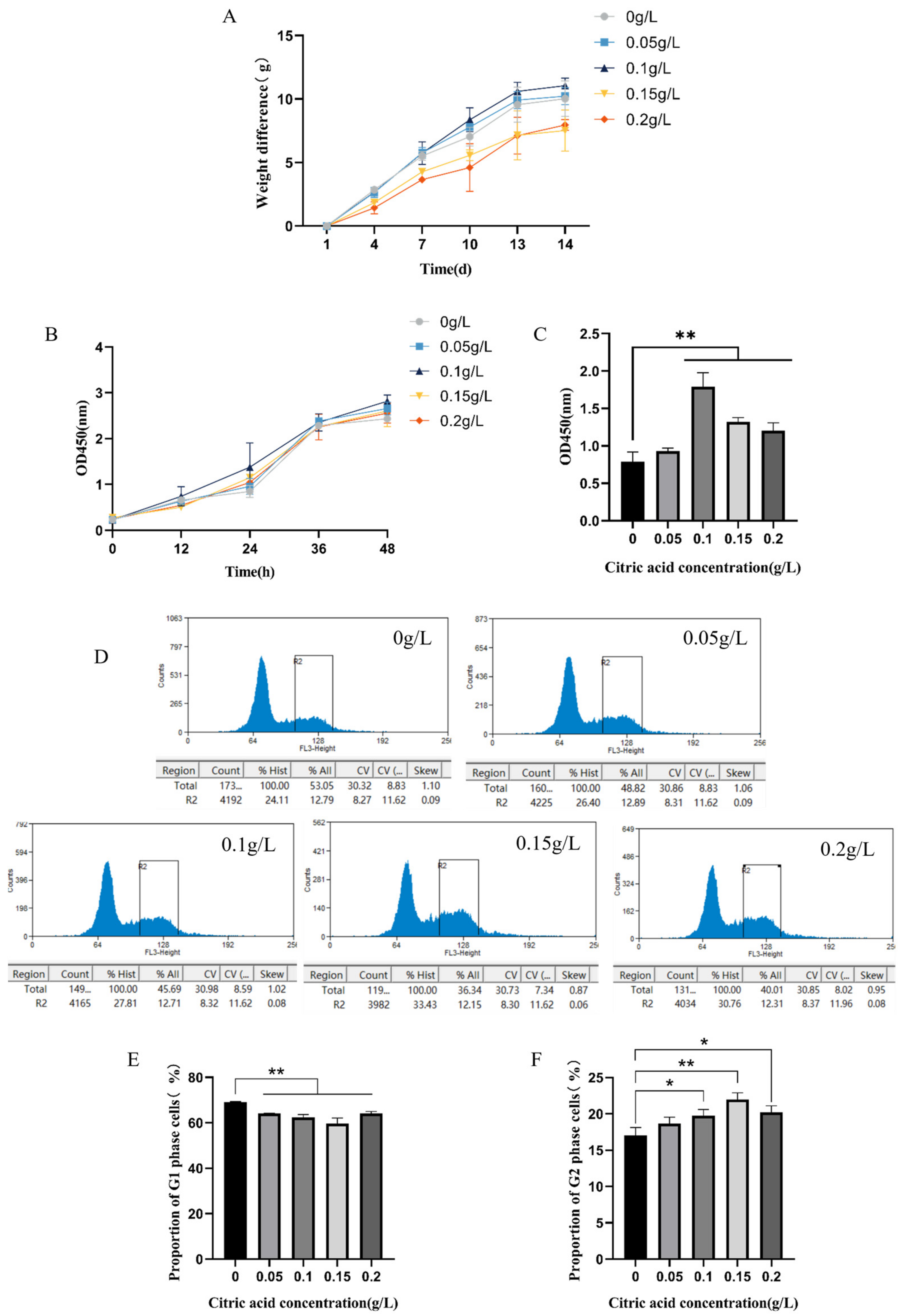
2.1. Citric Acid Increases Body Weight and Stimulates Cell Growth by Increasing the Number of G2 Phase Cells in IEC-6 Cells of Mice
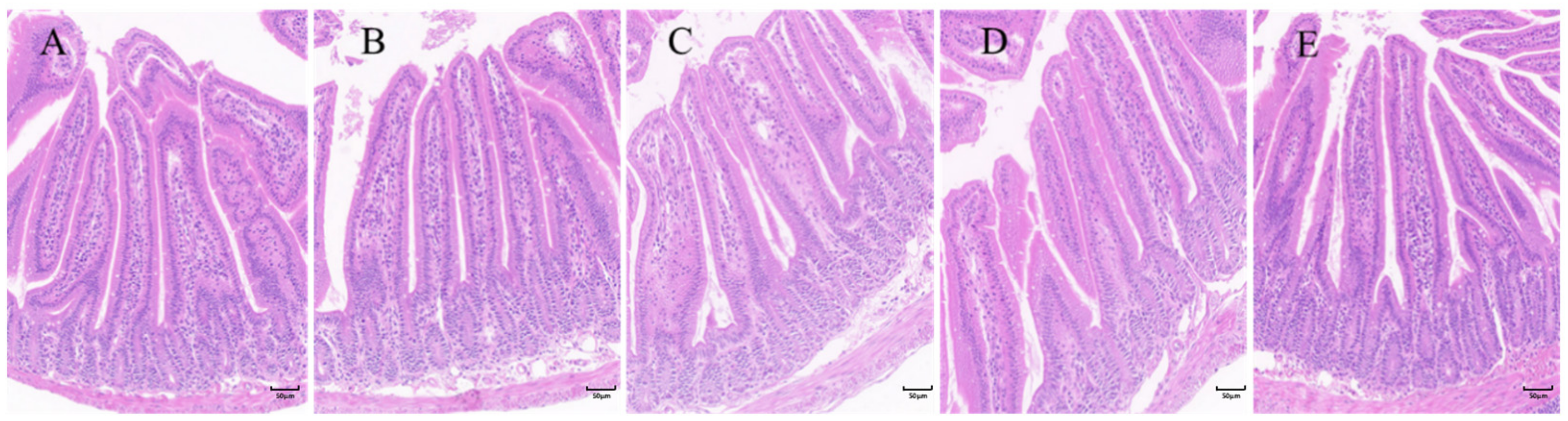
2.2. Improvement of Intestinal Structure by Citric Acid-Induced Increase in the Ratio of Villi to Crypts in Mouse Small Intestine
2.3. Citric Acid Enhances Growth of Common Probiotic Bacteria in the Cecum of Mice
2.4. Citric Acid Induced the Expression of Tight Junction Genes in Intestinal Tissue and IEC-6 Cells
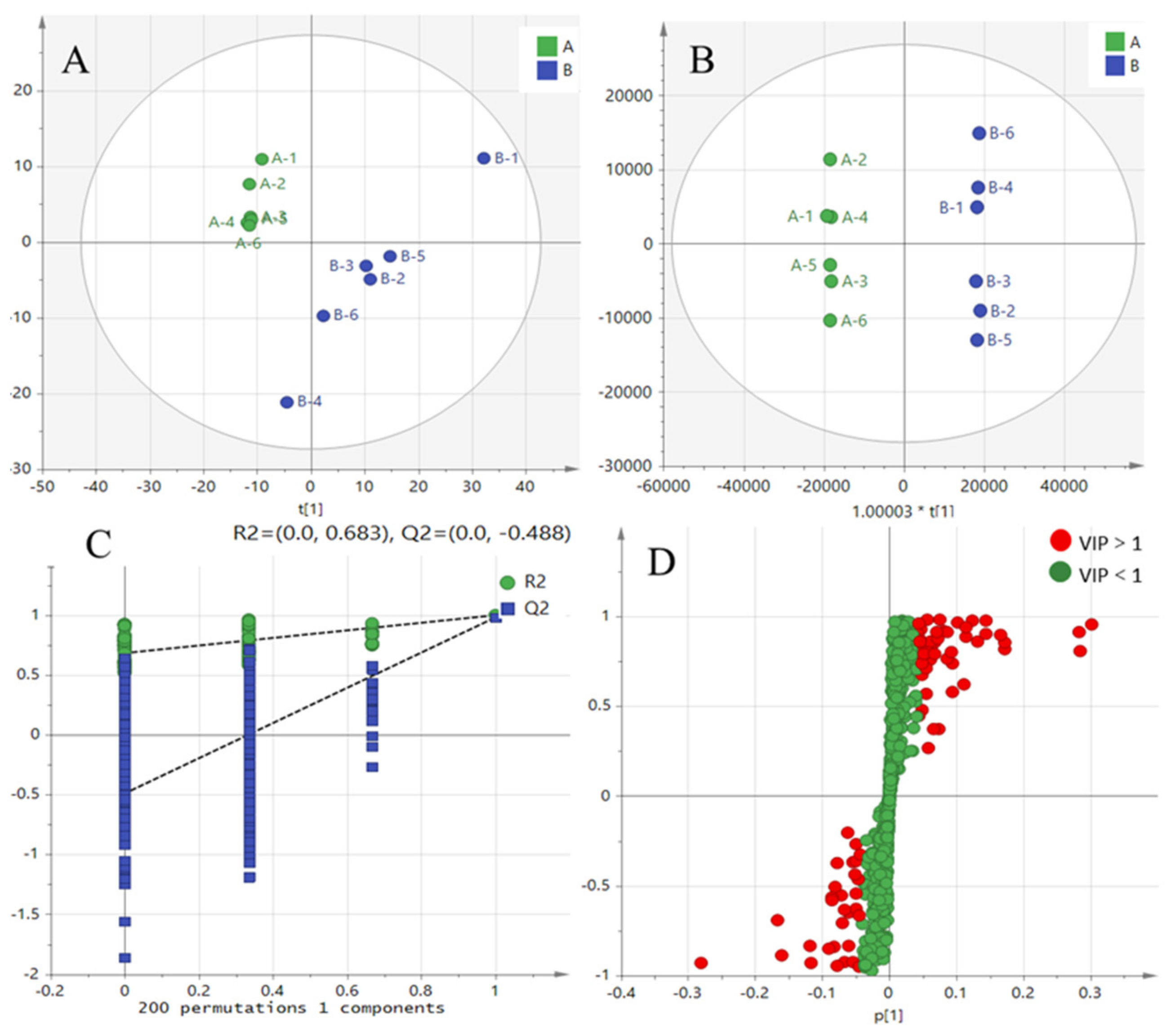
2.5. Transcriptome and Metabolic Pathway Analysis of Citric Acid-Induced Serum Metabolism in Mice
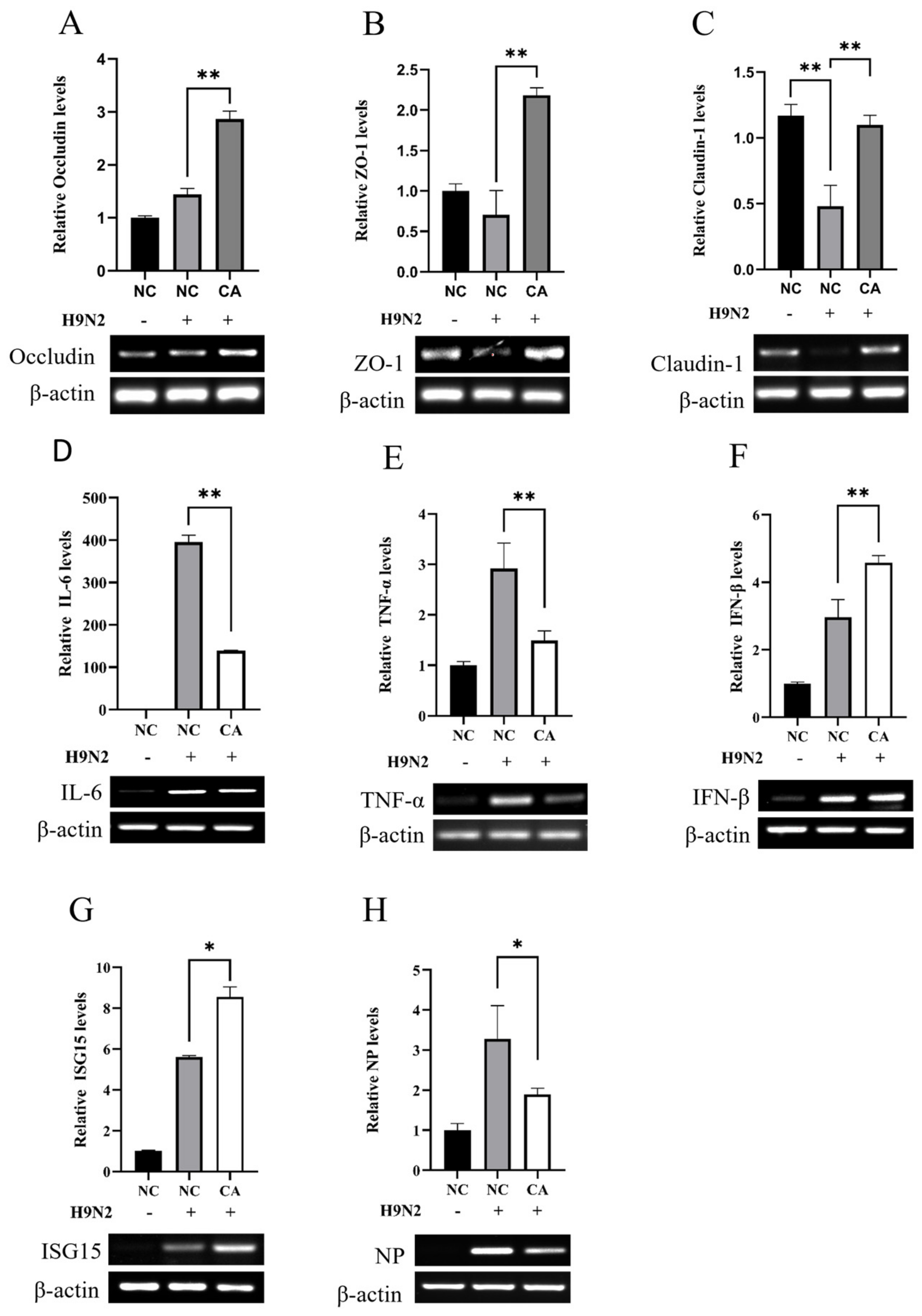
2.6. Citric Acid Maintains Tight Junction Gene Expression in IEC-6 Cells during H9N2 Infection and Inhibits Viral Replication
3. Discussion
4. Materials and Methods
4.1. Cells, Animals and Reagents
4.2. Grouping of Mice and Plotting of Body Weight Curves
4.3. Cell Assay
4.3.1. Cell Aggregation and Growth Curve Graphing
4.3.2. Cell Cycle Assay
4.4. Detection of Morphological Structure of Mouse Intestine
4.5. Detection of Bacterial Flora in the Contents of Mouse Cecum
Copy number (copies·μL−1) = (6.02 × 1023 × (ng·μL−1 × 10−9)/(DNA length × 660)).
4.6. Intestinal Tight Junction Assay
4.6.1. Detection of Mouse Intestinal Tight Junction Genes
4.6.2. Cellular Tight Junction Protein Gene Expression Assay
4.7. Detection of Serum Metabolites in Mice Using Untargeted Metabolomics
4.8. Detection of Inflammatory Factors and Type I Interferon in IAV-Infected IEC-6 Cells
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zhong, C.; Zhou, Y.; Zhao, J.; Fu, J.; Jiang, T.; Liu, B.; Chen, F.; Cao, G. High throughput sequencing reveals the abundance and diversity of antibiotic-resistant bacteria in aquaculture wastewaters, Shandong, China. 3 Biotech 2021, 11, 104. [Google Scholar] [CrossRef] [PubMed]
- Kuldeep, D.; Ruchi, T.; Ullah, K.R.; Sandip, C.; Marappan, G.; Kumaragurubaran, K.; Mani, S.; Arumugam, D.P.; Tulasi, S.L. Growth Promoters and Novel Feed Additives Improving Poultry Production and Health, Bioactive Principles and Beneficial Applications: The Trends and Advances—A Review. Int. J. Pharmacol. 2014, 10, 129–159. [Google Scholar]
- Yadav, A.S.; Kolluri, G.; Gopi, M.; Karthik, K.; Malik, Y.S.; Dhama, K. Exploring alternatives to antibiotics as health promoting agents in poultry—A review. J. Exp. Biol. Agric. Sci. 2016, 4, 368–383. [Google Scholar] [CrossRef]
- Killiny, N.; Nehela, Y.; Hijaz, F.; Vincent, C.I. A plant pathogenic bacterium exploits the tricarboxylic acid cycle metabolic pathway of its insect vector. Virulence 2018, 9, 99–109. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, C.; Hatayama, N.; Ogawa, T.; Nanizawa, E.; Otsuka, S.; Hata, K.; Seno, H.; Naito, M.; Hirai, S. Cardioprotection via Metabolism for Rat Heart Preservation Using the High-Pressure Gaseous Mixture of Carbon Monoxide and Oxygen. Int. J. Mol. Sci. 2020, 21, 8858. [Google Scholar] [CrossRef] [PubMed]
- Pileggi, V.J.; De Luca, H.F.; Cramer, J.W.; Steenbock, H. Citrate in the prevention of rickets in rats. Arch. Biochem. Biophys. 1956, 60, 52–57. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Xue, J.; Qi, Y.; Muhammad, I.; Wang, H.; Li, X.; Luo, Y.; Zhu, D.; Gao, Y.; Kong, L.; et al. Citric Acid Confers Broad Antibiotic Tolerance through Alteration of Bacterial Metabolism and Oxidative Stress. Int. J. Mol. Sci. 2023, 24, 9089. [Google Scholar] [CrossRef] [PubMed]
- Krauze, M.; Cendrowska-Pinkosz, M.; Matusevicius, P.; Stepniowska, A.; Jurczak, P.; Ognik, K. The Effect of Administration of a Phytobiotic Containing Cinnamon Oil and Citric Acid on the Metabolism, Immunity, and Growth Performance of Broiler Chickens. Animals 2021, 11, 399. [Google Scholar] [CrossRef]
- Kumar, A.; Toghyani, M.; Kheravii, S.K.; Pineda, L.; Han, Y.; Swick, R.A.; Wu, S.B. Organic acid blends improve intestinal integrity, modulate short-chain fatty acids profiles and alter microbiota of broilers under necrotic enteritis challenge. Anim. Nutr. 2022, 8, 82–90. [Google Scholar] [CrossRef]
- Jeong, H.Y.; Choi, Y.S.; Lee, J.K.; Lee, B.J.; Kim, W.K.; Kang, H. Anti-Inflammatory Activity of Citric Acid-Treated Wheat Germ Extract in Lipopolysaccharide-Stimulated Macrophages. Nutrients 2017, 9, 730. [Google Scholar] [CrossRef]
- Ren, Z.; Guo, C.; Yu, S.; Zhu, L.; Wang, Y.; Hu, H.; Deng, J. Progress in Mycotoxins Affecting Intestinal Mucosal Barrier Function. Int. J. Mol. Sci. 2019, 20, 2777. [Google Scholar] [CrossRef]
- Gonzalez-Mariscal, L.; Tapia, R.; Chamorro, D. Crosstalk of tight junction components with signaling pathways. Biochim. Biophys. Acta 2008, 1778, 729–756. [Google Scholar] [CrossRef] [PubMed]
- Yumine, N.; Matsumoto, Y.; Ohta, K.; Fukasawa, M.; Nishio, M. Claudin-1 inhibits human parainfluenza virus type 2 dissemination. Virology 2019, 531, 93–99. [Google Scholar] [CrossRef] [PubMed]
- Neish, A.S. Microbes in gastrointestinal health and disease. Gastroenterology 2009, 136, 65–80. [Google Scholar] [CrossRef] [PubMed]
- Neu, J. Gastrointestinal maturation and implications for infant feeding. Early Hum. Dev. 2007, 83, 767–775. [Google Scholar] [CrossRef]
- Dawood, F.S.; Jain, S.; Finelli, L.; Shaw, M.W.; Lindstrom, S.; Garten, R.J.; Gubareva, L.V.; Xu, X.; Bridges, C.B.; Uyeki, T.M. Emergence of a novel swine-origin influenza A (H1N1) virus in humans. N. Engl. J. Med. 2009, 360, 2605–2615. [Google Scholar] [CrossRef] [PubMed]
- Boone, S.A.; Gerba, C.P. The occurrence of influenza A virus on household and day care center fomites. J. Infect. 2005, 51, 103–109. [Google Scholar] [CrossRef] [PubMed]
- Post, J.; Burt, D.W.; Cornelissen, J.B.; Broks, V.; van Zoelen, D.; Peeters, B.; Rebel, J.M. Systemic virus distribution and host responses in brain and intestine of chickens infected with low pathogenic or high pathogenic avian influenza virus. Virol. J. 2012, 9, 61. [Google Scholar] [CrossRef]
- Yang, W.; Liu, X.; Wang, X. The immune system of chicken and its response to H9N2 avian influenza virus. Vet. Q. 2023, 43, 1–14. [Google Scholar] [CrossRef]
- Abd, E.M.; El-Saadony, M.T.; Alqhtani, A.H.; Swelum, A.A.; Salem, H.M.; Elbestawy, A.R.; Noreldin, A.E.; Babalghith, A.O.; Khafaga, A.F.; Hassan, M.I.; et al. The relationship among avian influenza, gut microbiota and chicken immunity: An updated overview. Poult. Sci. 2022, 101, 102021. [Google Scholar] [CrossRef]
- Wu, J.; Ye, Y.; Quan, J.; Ding, R.; Wang, X.; Zhuang, Z.; Zhou, S.; Geng, Q.; Xu, C.; Hong, L.; et al. Using nontargeted LC-MS metabolomics to identify the Association of Biomarkers in pig feces with feed efficiency. Porc. Health Manag. 2021, 7, 39. [Google Scholar] [CrossRef]
- Wahlstrom, A.; Sayin, S.I.; Marschall, H.U.; Backhed, F. Intestinal Crosstalk between Bile Acids and Microbiota and Its Impact on Host Metabolism. Cell Metab. 2016, 24, 41–50. [Google Scholar] [CrossRef]
- Winston, J.A.; Theriot, C.M. Diversification of host bile acids by members of the gut microbiota. Gut Microbes 2020, 11, 158–171. [Google Scholar] [CrossRef] [PubMed]
- Zhou, L.; Xu, J.D.; Zhou, S.S.; Zhu, H.; Kong, M.; Shen, H.; Zou, Y.T.; Cong, L.J.; Xu, J.; Li, S.L. Independent or integrative processing approach of metabolite datasets from different biospecimens potentially affects metabolic pathway recognition in metabolomics. J. Chromatogr. A 2019, 1587, 146–154. [Google Scholar] [CrossRef] [PubMed]
- May, J.D.; Lott, B.D.; Simmons, J.D. The effect of environmental temperature and body weight on growth rate and feed:gain of male broilers. Poult. Sci. 1998, 77, 499–501. [Google Scholar] [CrossRef] [PubMed]
- Lewis, R.M.; Emmans, G.C. The relationship between feed intake and liveweight in domestic animals. J. Anim. Sci. 2020, 98, skaa087. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Wang, A.; Li, R.; Zhu, H.; Hu, L.; Chen, W. Total ginsenosides promote the IEC-6 cell proliferation via affecting the regulatory mechanism mediated by polyamines. Saudi Pharm. J. 2021, 29, 1223–1232. [Google Scholar] [CrossRef]
- Di, W.; Lv, Y.; Xia, F.; Sheng, Y.; Liu, J.; Ding, G. Improvement of intestinal stem cells and barrier function via energy restriction in middle-aged C57BL/6 mice. Nutr. Res. 2020, 81, 47–57. [Google Scholar] [CrossRef]
- Wang, M.; Yang, C.; Wang, Q.; Li, J.; Huang, P.; Li, Y.; Ding, X.; Yang, H.; Yin, Y. The relationship between villous height and growth performance, small intestinal mucosal enzymes activities and nutrient transporters expression in weaned piglets. J. Anim. Physiol. Anim. Nutr. 2020, 104, 606–615. [Google Scholar] [CrossRef]
- Goh, T.W.; Hong, J.; You, D.H.; Han, Y.G.; Nam, S.O.; Kim, Y.Y. Effects of medium chain triglycerides with organic acids on growth performance, fecal score, blood profiles, intestinal morphology, and nutrient digestibility in weaning pigs. Anim. Biosci. 2022, 35, 916–926. [Google Scholar] [CrossRef]
- Zhang, H.; Guo, Y.; Wang, Z.; Wang, Y.; Chen, B.; Du, P.; Zhang, X.; Huang, Y.; Li, P.; Michiels, J.; et al. Acidification of drinking water improved tibia mass of broilers through the alterations of intestinal barrier and microbiota. Anim. Biosci. 2022, 35, 902–915. [Google Scholar] [CrossRef] [PubMed]
- Chwen, L.T.; Foo, H.L.; Thanh, N.T.; Choe, D.W. Growth performance, plasma Fatty acids, villous height and crypt depth of preweaning piglets fed with medium chain triacylglycerol. Asian Austral. J. Anim. 2013, 26, 700–704. [Google Scholar] [CrossRef]
- Qiao, J.; Shang, Z.; Liu, X.; Wang, K.; Wu, Z.; Wei, Q.; Li, H. Regulatory Effects of Combined Dietary Supplementation with Essential Oils and Organic Acids on Microbial Communities of Cobb Broilers. Front. Microbiol. 2021, 12, 814626. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.F.; Gao, K.; Wang, C.; Zhang, W.M.; Liu, J.X. Effects of feeding bamboo vinegar and acidifier as an antibiotic substitute on the growth performance and intestinal bacterial communities of weaned piglets. Acta Agric. Scand. Sect. A—Anim. Sci. 2013, 63, 143–150. [Google Scholar] [CrossRef]
- Oshiro, T.; Nagata, S.; Wang, C.; Takahashi, T.; Tsuji, H.; Asahara, T.; Nomoto, K.; Takei, H.; Nittono, H.; Yamashiro, Y. Bifidobacterium Supplementation of Colostrum and Breast Milk Enhances Weight Gain and Metabolic Responses Associated with Microbiota Establishment in Very-Preterm Infants. Biomed. Hub. 2019, 4, 1–10. [Google Scholar] [CrossRef]
- Gao, Q.; Wang, Y.; Li, J.; Bai, G.; Liu, L.; Zhong, R.; Ma, T.; Pan, H.; Zhang, H. Supplementation of multi-enzymes alone or combined with inactivated Lactobacillus benefits growth performance and gut microbiota in broilers fed wheat diets. Front. Microbiol. 2022, 13, 927932. [Google Scholar] [CrossRef]
- Grilli, E.; Tugnoli, B.; Passey, J.L.; Stahl, C.H.; Piva, A.; Moeser, A.J. Impact of dietary organic acids and botanicals on intestinal integrity and inflammation in weaned pigs. BMC Vet. Res. 2015, 11, 96. [Google Scholar] [CrossRef]
- Xu, Y.; Lahaye, L.; He, Z.; Zhang, J.; Yang, C.; Piao, X. Micro-encapsulated essential oils and organic acids combination improves intestinal barrier function, inflammatory responses and microbiota of weaned piglets challenged with enterotoxigenic Escherichia coli F4 (K88(+)). Anim. Nutr. 2020, 6, 269–277. [Google Scholar] [CrossRef]
- Ma, J.; Piao, X.; Shang, Q.; Long, S.; Liu, S.; Mahfuz, S. Mixed organic acids as an alternative to antibiotics improve serum biochemical parameters and intestinal health of weaned piglets. Anim. Nutr. 2021, 7, 737–749. [Google Scholar] [CrossRef]
- Wu, G. Important roles of dietary taurine, creatine, carnosine, anserine and 4-hydroxyproline in human nutrition and health. Amino Acids 2020, 52, 329–360. [Google Scholar] [CrossRef]
- Barua, M.; Liu, Y.; Quinn, M.R. Taurine chloramine inhibits inducible nitric oxide synthase and TNF-alpha gene expression in activated alveolar macrophages: Decreased NF-kappaB activation and IkappaB kinase activity. J. Immunol. 2001, 167, 2275–2281. [Google Scholar] [CrossRef] [PubMed]
- Kim, C.; Cha, Y.N. Taurine chloramine produced from taurine under inflammation provides anti-inflammatory and cytoprotective effects. Amino Acids 2014, 46, 89–100. [Google Scholar] [CrossRef] [PubMed]
- Gundala, N.; Naidu, V.; Das, U.N. Amelioration of streptozotocin-induced type 2 diabetes mellitus in Wistar rats by arachidonic acid. Biochem. Biophys. Res. Commun. 2018, 496, 105–113. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.S.; Moon, S.J.; Lee, S.E.; Hwang, G.W.; Yoo, H.J.; Song, J.W. The arachidonic acid metabolite 11,12-epoxyeicosatrienoic acid alleviates pulmonary fibrosis. Exp. Mol. Med. 2021, 53, 864–874. [Google Scholar] [CrossRef] [PubMed]
- Capdevila, J.; Wang, W. Role of cytochrome P450 epoxygenase in regulating renal membrane transport and hypertension. Curr. Opin. Nephrol. Hypertens. 2013, 22, 163–169. [Google Scholar] [CrossRef] [PubMed]
- Baranowska, I.; Gawrys, O.; Walkowska, A.; Olszynski, K.H.; Cervenka, L.; Falck, J.R.; Adebesin, A.M.; Imig, J.D.; Kompanowska-Jezierska, E. Epoxyeicosatrienoic Acid Analog and 20-HETE Antagonist Combination Prevent Hypertension Development in Spontaneously Hypertensive Rats. Front. Pharmacol. 2021, 12, 798642. [Google Scholar] [CrossRef] [PubMed]
- Kawaguchi, H.; Akazawa, Y.; Watanabe, Y.; Takakura, Y. Permeability modulation of human intestinal Caco-2 cell monolayers by interferons. Eur. J. Pharm. Biopharm. 2005, 59, 45–50. [Google Scholar] [CrossRef]
- Tang, X.; Liu, J.; Dong, W.; Li, P.; Li, L.; Lin, C.; Zheng, Y.; Hou, J.; Li, D. The cardioprotective effects of citric Acid and L-malic Acid on myocardial ischemia/reperfusion injury. Evid.-Based Complement. Altern. Med. 2013, 2013, 820695. [Google Scholar] [CrossRef]
- Abdel-Salam, O.M.; Youness, E.R.; Mohammed, N.A.; Morsy, S.M.; Omara, E.A.; Sleem, A.A. Citric acid effects on brain and liver oxidative stress in lipopolysaccharide-treated mice. J. Med. Food 2014, 17, 588–598. [Google Scholar] [CrossRef]
- Zhao, S.; Chen, Z.; Zheng, J.; Dai, J.; Ou, W.; Xu, W.; Ai, Q.; Zhang, W.; Niu, J.; Mai, K.; et al. Citric acid mitigates soybean meal induced inflammatory response and tight junction disruption by altering TLR signal transduction in the intestine of turbot, Scophthalmus maximus L. Fish Shellfish Immunol. 2019, 92, 181–187. [Google Scholar] [CrossRef]
- Ding, X.; Zhong, X.; Yang, Y.; Zhang, G.; Si, H. Citric Acid and Magnolol Ameliorate Clostridium perfringens Challenge in Broiler Chickens. Animals 2023, 13, 577. [Google Scholar] [CrossRef] [PubMed]
- Negoro, H.; Chatziantonio, C.; Razzaque, M.S. Therapeutic potential of 5-aminolevulinic acid and sodium-ferrous citrate for viral insults: Relevance to the COVID-19 crisis. Expert Rev. Anti-Infect. Ther. 2022, 20, 657–661. [Google Scholar] [CrossRef] [PubMed]
- Ma, L.L.; Zhang, P.; Wang, H.Q.; Li, Y.F.; Hu, J.; Jiang, J.D.; Li, Y.H. heme oxygenase-1 agonist CoPP suppresses influenza virus replication through IRF3-mediated generation of IFN-alpha/beta. Virology 2019, 528, 80–88. [Google Scholar] [CrossRef] [PubMed]
- Hossain, M.K.; Saha, S.K.; Abdal, D.A.; Kim, J.H.; Kim, K.; Yang, G.M.; Choi, H.Y.; Cho, S.G. Bax Inhibitor-1 Acts as an Anti-Influenza Factor by Inhibiting ROS Mediated Cell Death and Augmenting Heme-Oxygenase 1 Expression in Influenza Virus Infected Cells. Int. J. Mol. Sci. 2018, 19, 712. [Google Scholar] [CrossRef]



| Groups (g/L) | Villus Height to Crypt Depth Ratio |
|---|---|
| 0 | 3.944 ± 0.2297 |
| 0.05 | 4.373 ± 0.2492 |
| 0.1 | 5.011 ± 0.2797 AA |
| 0.15 | 4.720 ± 0.1643 AA |
| 0.2 | 4.095 ± 0.6054 |
| Bacterial Type | 0 g/L | 0.05 g/L | 0.1 g/L | 0.15 g/L | 0.2 g/L |
|---|---|---|---|---|---|
| Bifid. | 2.15 ± 0.03 | 2.09 ± 0.02 | 2.52 ± 0.01 AA | 2.34 ± 0.02 aa | 2.10 ± 0.07 |
| Lac. | 2.38 ± 0.03 | 2.37 ± 0.02 | 2.50 ± 0.08 | 2.45 ± 0.06 | 2.47 ± 0.15 |
| E. coli | 2.72 ± 0.01 | 2.72 ± 0.01 | 2.73 ± 0.01 | 2.71 ± 0.01 | 2.72 ± 0.01 |
| Name | Impact | Details |
|---|---|---|
| Taurine and hypotaurine metabolism | 0.42857 | KEGG |
| Arachidonic acid metabolism | 0.33292 | KEGG |
| Cysteine and methionine metabolism | 0.10446 | KEGG |
| Primary bile acid biosynthesis | 0.02239 | KEGG |
| Purine metabolism | 0.01945 | KEGG |
| Fatty acid biosynthesis | 0.01473 | KEGG |
| Glutathione metabolism | 0.00709 | KEGG |
| Steroid hormone biosynthesis | 0.00653 | KEGG |
| Biosynthesis of unsaturated fatty acids | 0 | KEGG |
| Phenylalanine metabolism | 0 | KEGG |
| Fatty acid elongation | 0 | KEGG |
| Aminoacyl-tRNA biosynthesis | 0 | KEGG |
| Gene | GenBank No. | Direction | Sense and Antisense Primer (5′–3′) |
|---|---|---|---|
| Occludin | NM_029865.2 | Forward | GTACCCACCAGTGACCAACA |
| Reverse | GTTGCTGGAGCTTAGCCTGT | ||
| ZO-1 | XM_036152895.1 | Forward | CGAGGCATCATCCCAAATAAGAAC |
| Reverse | TCCAGAAGTCTGCCCGATCAC | ||
| Claudin-1 | NM_016674.4 | Forward | TGGTAATTGGCATCCTGCTG |
| Reverse | CAGCCATCCACATCTTCTGC | ||
| Bifid. | AB064848.1 | Forward | GATTCTGGCTCAGGATGAACGC |
| Reverse | CTGATAGGACGAGACCCAT | ||
| Lac. | OM349561.1 | Forward | AGCAGTAGGGATCTTCCA |
| Reverse | ATTTCACCGCTACACATG | ||
| E.coli | OU548744.1 | Forward | GGGAGTAAAGTTAATACCTTTGCTC |
| Reverse | TTCCCGAAGGCACATTCT | ||
| TNF-α | XM_021218152.1 | Forward | GAGGCACTCCCCCAAAAGAT |
| Reverse | GAGGGAGGCCATTTGGGAAC | ||
| IL-6 | NM_031168.2 | Forward | GTCCTTCCTACCCCAATTTCCA |
| Reverse | TAACGCACTAGGTTTGCCGA | ||
| IFN-β | NM_010510.2 | Forward | AACTCCACCAGCAGACAGTG |
| Reverse | GGTACCTTTGCACCCTCCAG | ||
| ISG15 | NM_015783.3 | Forward | CGGGAACAAGTCCACGAAG |
| Reverse | CCCTCAGGCGCAAATGCT |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hu, P.; Yuan, M.; Guo, B.; Lin, J.; Yan, S.; Huang, H.; Chen, J.-L.; Wang, S.; Ma, Y. Citric Acid Promotes Immune Function by Modulating the Intestinal Barrier. Int. J. Mol. Sci. 2024, 25, 1239. https://doi.org/10.3390/ijms25021239
Hu P, Yuan M, Guo B, Lin J, Yan S, Huang H, Chen J-L, Wang S, Ma Y. Citric Acid Promotes Immune Function by Modulating the Intestinal Barrier. International Journal of Molecular Sciences. 2024; 25(2):1239. https://doi.org/10.3390/ijms25021239
Chicago/Turabian StyleHu, Pengcheng, Meng Yuan, Bolun Guo, Jiaqi Lin, Shihong Yan, Huiqing Huang, Ji-Long Chen, Song Wang, and Yanmei Ma. 2024. "Citric Acid Promotes Immune Function by Modulating the Intestinal Barrier" International Journal of Molecular Sciences 25, no. 2: 1239. https://doi.org/10.3390/ijms25021239





