Plant-Growth-Promoting Rhizobacteria Modulate Carbohydrate Metabolism in Connection with Host Plant Defense Mechanism
Abstract
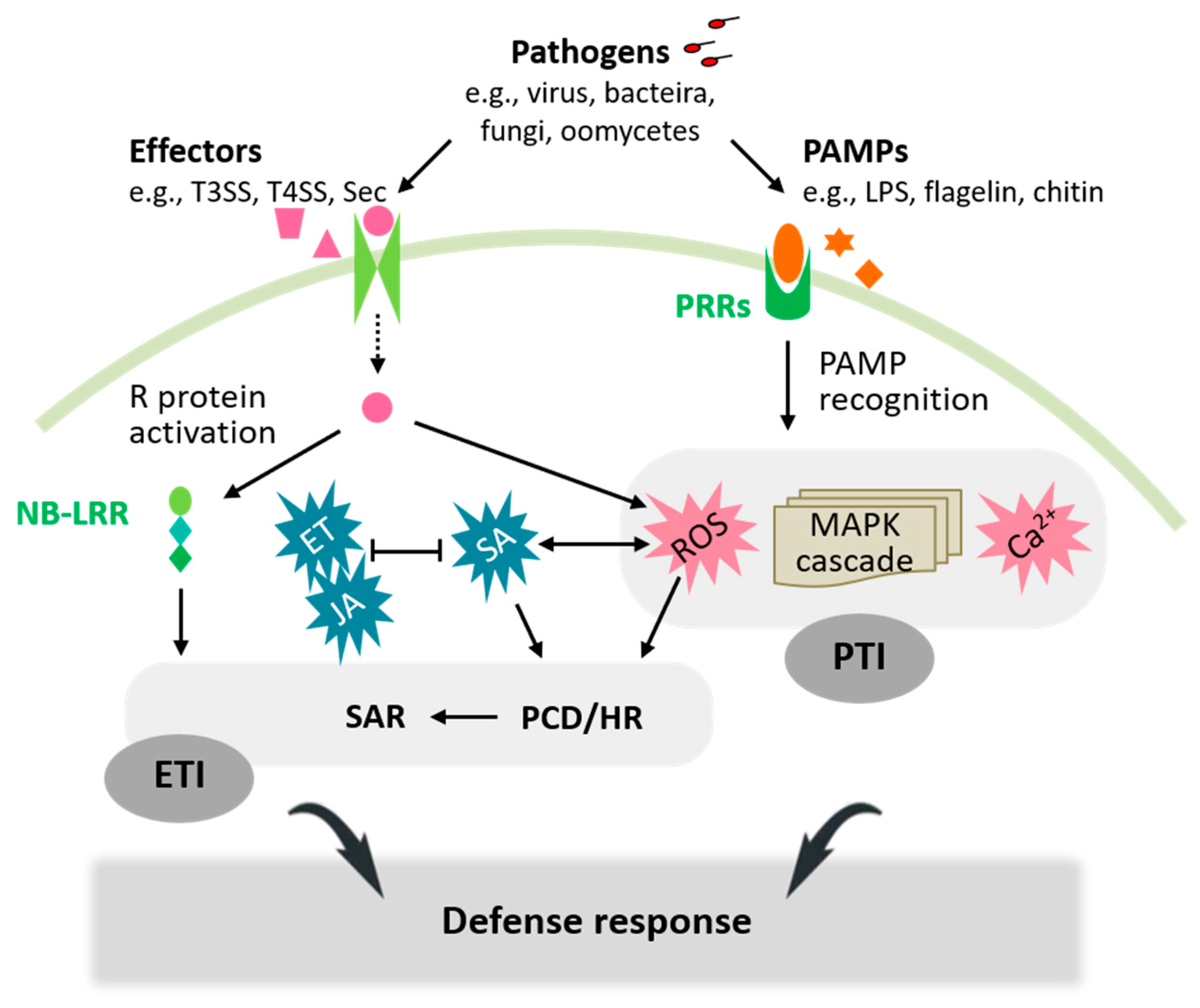
:1. Introduction
2. PGPR Inoculation Influences Carbohydrate Metabolism
2.1. Photosynthetic Pigments
| PGP Traits | PGPR Involved | Host Plant | Effect | Ref |
|---|---|---|---|---|
| Nitrogen fixation | Azospirillum sp. Sp7 Bacillus sphaericus UPMB10 | Banana plantlets | Chl accumulation | [11] |
| Aeromonas hydrophila P73 Serratia proteamaculans 1–102 S. liquefaciens 2–68 Pseudomonas putida G11-32 | Soybean | Pn enhancement | [35] | |
| Siderophore | Pseudomonas sp. RRLJ 008 | Eggplant, cabbage, tomato, bean, and kohlrabi | Chl accumulation | [12] |
| Siderophore and phosphorous solubilization | B. pumilus S4 | Runner bean | Chl accumulation, Pn enhancement, boosted POD and SOD activity during periods of intense photosynthesis | [13] |
| ACC deaminase | P. fluorescens YsS6 P. migulae 8R6 | Tomato | Chl accumulation | [28] |
| IAA | B. mycoides S7 | Runner bean | Chl accumulation, Pn enhancement, boosted POD and SOD activity during periods of intense photosynthesis | [13] |
| Paraburkholderia phytofirmans PsJN | Grapevine plantlets | Soluble sugar and starch accumulation | [36,37] | |
| Maize | Chl accumulation, PSII efficiency enhancement | [23] | ||
| ABA | A. brasilense Sp245 | Wheat, Grapevine Arabidopsis | Chl, arotenoid, and photoprotective pigment accumulation | [10,14,30] |
| Volatile compounds | B. subtilis GB03 | Arabidopsis | Chl accumulation, PSII efficiency enhancement, endogenous glucose modulation, overexpression of genes involved in photosynthesis | [15] |
| B. amyloliquefaciens FZB42 | Arabidopsis | Regulation of genes related to photosynthetic light harvesting | [21] | |
| B. subtilis JS | Tobacco | Regulation of genes involved in photosynthesis | [22] |
2.2. Photosystem Efficiencies
2.3. CO2 Fixation
2.4. Plant Host Carbohydrate Levels
3. PGPR-Induced Alterations in Photosynthesis and Carbon Fluxes Contribute to Plant Defense
3.1. Maintaining Chloroplast Structure and Function
3.2. Maintaining the Balance between ROS Production and Antioxidant Defense
3.3. Redistribution of Sugars
3.4. Fine-Tuning Trade-Offs between Defense and Carbohydrate Metabolism
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Wu, A.; Brider, J.; Busch, F.A.; Chen, M.; Chenu, K.; Clarke, V.C.; Collins, B.; Ermakova, M.; Evans, J.R.; Farquhar, G.D.; et al. A cross-scale analysis to understand and quantify the effects of photosynthetic enhancement on crop growth and yield across environments. Plant Cell Environ. 2022, 46, 23–44. [Google Scholar] [CrossRef] [PubMed]
- Gan, P.; Ikeda, K.; Irieda, H.; Narusaka, M.; O’Connell, R.J.; Narusaka, Y.; Takano, Y.; Kubo, Y.; Shirasu, K. Comparative genomic and transcriptomic analyses reveal the hemibiotrophic stage shift of Colletotrichum fungi. New Phytol. 2013, 197, 1236–1249. [Google Scholar] [CrossRef]
- Ghosh, S.; Kanwar, P.; Jha, G. Alterations in rice chloroplast integrity, photosynthesis and metabolome associated with pathogenesis of Rhizoctonia solani. Sci. Rep. 2017, 7, 41610. [Google Scholar] [CrossRef] [PubMed]
- Berger, S.; Papadopoulos, M.; Schreiber, U.; Kaiser, W.; Roitsch, T. Complex regulation of gene expression, photosynthesis and sugar levels by pathogen infection in tomato. Physiol. Plantarum 2004, 122, 419–428. [Google Scholar] [CrossRef]
- Hu, Y.; Zhong, S.; Zhang, M.; Liang, Y.; Gong, G.; Chang, X.; Tan, F.; Yang, H.; Qiu, X.; Luo, L. Potential role of photosynthesis in the regulation of reactive oxygen species and defence responses to Blumeria graminis f. sp. tritici in wheat. Int. J. Mol. Sci. 2020, 21, 5767. [Google Scholar] [CrossRef]
- Passari, A.K.; Upadhyaya, K.; Singh, G.; Abdel-Azeem, A.M.; Thankappan, S.; Uthandi, S.; Hashem, A.; Abd-Allah, E.F.; Malik, J.A.; As, A.; et al. Enhancement of disease resistance, growth potential, and photosynthesis in tomato (Solanum lycopersicum) by inoculation with an endophytic actinobacterium, Streptomyces thermocarboxydus strain BPSAC147. PLoS ONE 2019, 14, e0219014. [Google Scholar] [CrossRef] [PubMed]
- Shahzad, R.; Khan, A.L.; Bilal, S.; Asaf, S.; Lee, I.-J. Plant growth-promoting endophytic bacteria versus pathogenic infections: An example of Bacillus amyloliquefaciens RWL-1 and Fusarium oxysporum f. sp. lycopersici in tomato. PeerJ 2017, 5, e3107. [Google Scholar] [CrossRef]
- Herbers, K.; Takahata, Y.; Melzer, M.; Mock, H.P.; Hajirezaei, M.; Sonnewald, U. Regulation of carbohydrate partitioning during the interaction of potato virus Y with tobacco. Mol. Plant Pathol. 2000, 1, 51–59. [Google Scholar] [CrossRef]
- Chou, H.M.; Bundock, N.; Rolfe, S.A.; Scholes, J.D. Infection of Arabidopsis thaliana leaves with Albugo candida (white blister rust) causes a reprogramming of host metabolism. Mol. Plant Pathol. 2000, 1, 99–113. [Google Scholar] [CrossRef]
- Sabir, A.; Yazici, M.A.; Kara, Z.; Sahin, F. Growth and mineral acquisition response of grapevine rootstocks (Vitis spp.) to inoculation with different strains of plant growth-promoting rhizobacteria (PGPR). J. Sci. Food Agr. 2012, 92, 2148–2153. [Google Scholar] [CrossRef]
- Baset, M.; Shamsuddin, Z.; Wahab, Z.; Marziah, M. Effect of Plant Growth Promoting Rhizobacterial (PGPR) inoculation on growth and nitrogen incorporation of tissue-cultured ’Musa’ plantlets under nitrogen-free hydroponics condition. AJCS 2010, 4, 85–90. [Google Scholar]
- Deka Boruah, H.; Dileep Kumar, B. Plant disease suppression and growth promotion by a fluorescent Pseudomonas strain. Folia Microbiol. 2002, 47, 137–143. [Google Scholar] [CrossRef] [PubMed]
- Stefan, M.; Munteanu, N.; Stoleru, V.; Mihasan, M. Effects of inoculation with plant growth promoting rhizobacteria on photosynthesis, antioxidant status and yield of runner bean. Rom. Biotech. Lett. 2013, 18, 8132–8143. Available online: https://hero.epa.gov/hero/index.cfm/reference/details/reference_id/8085113 (accessed on 8 January 2024).
- Cohen, A.C.; Bottini, R.; Pontin, M.; Berli, F.J.; Moreno, D.; Boccanlandro, H.; Travaglia, C.N.; Piccoli, P.N. Azospirillum brasilense ameliorates the response of Arabidopsis thaliana to drought mainly via enhancement of ABA levels. Physiol. Plantarum 2015, 153, 79–90. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Xie, X.T.; Kim, M.S.; Kornyeyev, D.A.; Holaday, S.; Paré, P.W. Soil bacteria augment Arabidopsis photosynthesis by decreasing glucose sensing and abscisic acid levels in planta. Plant J. 2008, 56, 264–273. [Google Scholar] [CrossRef] [PubMed]
- Ding, F.; Wang, M.; Zhang, S. Overexpression of a Calvin cycle enzyme SBPase improves tolerance to chilling-induced oxidative stress in tomato plants. Sci. Hortic. 2017, 214, 27–33. [Google Scholar] [CrossRef]
- Simkin, A.J.; Lopez-Calcagno, P.E.; Davey, P.A.; Headland, L.R.; Lawson, T.; Timm, S.; Bauwe, H.; Raines, C.A. Simultaneous stimulation of sedoheptulose 1,7-bisphosphatase, fructose 1,6-bisphophate aldolase and the photorespiratory glycine decarboxylase-H protein increases CO2 assimilation, vegetative biomass and seed yield in Arabidopsis. Plant Biotechnol. J. 2017, 15, 805–816. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.; Feng, R.; Ding, Y.; Wang, R. Applying carbon dioxide, plant growth-promoting rhizobacterium and EDTA can enhance the phytoremediation efficiency of ryegrass in a soil polluted with zinc, arsenic, cadmium and lead. J. Environ. Manag. 2014, 141, 1–8. [Google Scholar] [CrossRef]
- Ainsworth, E.A.; Long, S.P. What have we learned from 15 years of free-air CO2 enrichment (FACE)? A meta-analytic review of the responses of photosynthesis, canopy properties and plant production to rising CO2. New Phytol. 2005, 165, 351–372. [Google Scholar] [CrossRef]
- Kwon, Y.S.; Lee, D.Y.; Rakwal, R.; Baek, S.B.; Lee, J.H.; Kwak, Y.S.; Seo, J.S.; Chung, W.S.; Bae, D.W.; Kim, S.G. Proteomic analyses of the interaction between the plant-growth promoting rhizobacterium Paenibacillus polymyxa E681 and Arabidopsis thaliana. Proteomics 2016, 16, 122–135. [Google Scholar] [CrossRef]
- Hao, H.T.; Zhao, X.; Shang, Q.H.; Wang, Y.; Guo, Z.H.; Zhang, Y.B.; Xie, Z.K.; Wang, R.Y. Comparative digital gene expression analysis of the Arabidopsis response to volatiles emitted by Bacillus amyloliquefaciens. PLoS ONE 2016, 11, e0158621. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.S.; Lee, J.; Seo, S.G.; Lee, C.; Su, Y.W.; Kim, S.H. Gene expression profile affected by volatiles of new plant growth promoting rhizobacteria, Bacillus subtilis strain JS, in tobacco. Genes Genom. 2015, 37, 387–397. [Google Scholar] [CrossRef]
- Naveed, M.; Mitter, B.; Reichenauer, T.G.; Wieczorek, K.; Sessitsch, A. Increased drought stress resilience of maize through endophytic colonization by Burkholderia phytofirmans PsJN and Enterobacter sp. FD17. Environ. Exp. Bot. 2014, 97, 30–39. [Google Scholar] [CrossRef]
- Li, J.; Wang, J.; Liu, H.; Macdonald, C.A.; Singh, B.K. Application of microbial inoculants significantly enhances crop productivity: A meta-analysis of studies from 2010 to 2020. J. Sustain. Agric. Environ. 2022, 1, 216–225. [Google Scholar] [CrossRef]
- Klee, H.J.; Hayford, M.B.; Kretzmer, K.A.; Barry, G.F.; Kishore, G.M. Control of ethylene synthesis by expression of a bacterial enzyme in transgenic tomato plants. Plant Cell 1991, 3, 1187–1193. [Google Scholar] [CrossRef]
- Binder, B.M. Ethylene signaling in plants. J. Biol. Chem. 2020, 295, 7710–7725. [Google Scholar] [CrossRef]
- Dong, J.; Wang, Y.; Xian, Q.; Chen, X.; Xu, J. Transcriptome analysis reveals ethylene-mediated defense responses to Fusarium oxysporum f. sp. cucumerinum infection in Cucumis sativus L. BMC Plant Biol. 2020, 20, 334. [Google Scholar] [CrossRef] [PubMed]
- Ali, S.; Charles, T.C.; Glick, B.R. Amelioration of high salinity stress damage by plant growth-promoting bacterial endophytes that contain ACC deaminase. Plant Physiol. Biochem. 2014, 80, 160–167. [Google Scholar] [CrossRef]
- Zhao, X.; Yuan, X.; Xing, Y.; Dao, J.; Zhao, D.; Li, Y.; Li, W.; Wang, Z. A meta-analysis on morphological, physiological and biochemical responses of plants with PGPR inoculation under drought stress. Plant Cell Environ. 2023, 46, 199–214. [Google Scholar] [CrossRef]
- Bashan, Y.; Bustillos, J.J.; Leyva, L.A.; Hernandez, J.P.; Bacilio, M. Increase in auxiliary photoprotective photosynthetic pigments in wheat seedlings induced by Azospirillum brasilense. Biol. Fertil. Soils. 2006, 42, 279–285. [Google Scholar] [CrossRef]
- Simkin, A.J.; Kapoor, L.; Doss, C.G.P.; Hofmann, T.A.; Lawson, T.; Ramamoorthy, S. The role of photosynthesis related pigments in light harvesting, photoprotection and enhancement of photosynthetic yield in planta. Photosynth. Res. 2022, 152, 23–42. [Google Scholar] [CrossRef]
- Berli, F.J.; Moreno, D.; Piccoli, P.; Hespanhol-Viana, L.; Silva, M.F.; Bressan-Smith, R.; Cavagnaro, J.B.; Bottini, R. Abscisic acid is involved in the response of grape (Vitis vinifera L.) cv. Malbec leaf tissues to ultraviolet-B radiation by enhancing ultraviolet-absorbing compounds, antioxidant enzymes and membrane sterols. Plant Cell Environ. 2010, 33, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Travaglia, C.; Reinoso, H.; Cohen, A.; Luna, C.; Tommasino, E.; Castillo, C.; Bottini, R. Exogenous ABA increases yield in field-grown wheat with moderate water restriction. J. Plant Growth Regul. 2010, 29, 366–374. [Google Scholar] [CrossRef]
- Travaglia, C.; Balboa, G.; Espósito, G.; Reinoso, H. ABA action on the production and redistribution of field-grown maize carbohydrates in semiarid regions. Plant Growth Regul. 2012, 67, 27–34. [Google Scholar] [CrossRef]
- Zhang, F.; Dashti, N.; Hynes, R.K.; Smith, D.L. Plant growth-promoting rhizobacteria and soybean [Glycine max (L.) Merr.] growth and physiology at suboptimal root zone temperatures. Ann. Bot. 1997, 79, 243–249. [Google Scholar] [CrossRef]
- Fernandez, O.; Theocharis, A.; Bordiec, S.; Feil, R.; Jacquens, L.; Clément, C.; Fontaine, F.; Aït Barka, E. Burkholderia phytofirmans PsJN acclimates grapevine to cold by modulating carbohydrate metabolism. APS 2012, 25, 496–504. [Google Scholar] [CrossRef]
- Su, F.; Gilard, F.; Guérard, F.; Citerne, S.; Clément, C.; Vaillant-Gaveau, N.; Dhondt-Cordelier, S. Spatio-temporal responses of Arabidopsis Leaves in photosynthetic performance and metabolite contents to Burkholderia phytofirmans PsJN. Front. Plant Sci. 2016, 7, 403. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Bueno, M.L.; Pineda, M.; Barón, M. Phenotyping plant responses to biotic stress by chlorophyll fluorescence imaging. Front. Plant Sci. 2019, 10, 1135. [Google Scholar] [CrossRef]
- Moustaka, J.; Moustakas, M. Early-stage detection of biotic and abiotic stress on plants by chlorophyll fluorescence imaging analysis. Biosensors 2023, 13, 796. [Google Scholar] [CrossRef]
- Ruíz-Sánchez, M.; Armada, E.; Munoz, Y.; de Salamone, I.E.G.; Aroca, R.; Ruíz-Lozano, J.M.; Azcón, R. Azospirillum and arbuscular mycorrhizal colonization enhance rice growth and physiological traits under well-watered and drought conditions. J. Plant Physiol. 2011, 168, 1031–1037. [Google Scholar] [CrossRef]
- Su, F.; Jacquard, C.; Villaume, S.; Michel, J.; Rabenoelina, F.; Clément, C.; Ait Barka, E.; Dhondt-Cordelier, S.; Vaillant-Gaveau, N. Burkholderia phytofirmans PsJN reduces impact to freezing temperatures on photosynthesis in Arabidopsis thaliana. Front. Plant Sci. 2015, 6, 810. [Google Scholar] [CrossRef] [PubMed]
- Rincón, A.; Valladares, F.; Gimeno, T.E.; Pueyo, J.J. Water stress responses of two Mediterranean tree species influenced by native soil microorganisms and inoculation with a plant growth promoting rhizobacterium. Tree physiol. 2008, 28, 1693–1701. [Google Scholar] [CrossRef] [PubMed]
- Ayyaz, A.; Miao, Y.; Hannan, F.; Islam, F.; Zhang, K.; Xu, J.; Farooq, M.A.; Zhou, W. Drought tolerance in Brassica napus is accompanied with enhanced antioxidative protection, photosynthetic and hormonal regulation at seedling stage. Physiol. Plantarum 2021, 172, 1133–1148. [Google Scholar] [CrossRef] [PubMed]
- Sommer, S.G.; Han, E.; Li, X.; Rosenqvist, E.; Liu, F. The chlorophyll fluorescence parameter Fv/Fm correlates with loss of grain yield after severe drought in three wheat genotypes grown at two CO2 concentrations. Plants 2023, 12, 436. [Google Scholar] [CrossRef]
- Lu, Y. Identification and roles of photosystem II assembly, stability, and repair factors in Arabidopsis. Front. Plant Sci. 2016, 7, 168. [Google Scholar] [CrossRef]
- Voisin, A.; Salon, C.; Jeudy, C.; Warembourg, F. Symbiotic N2 fixation activity in relation to C economy of Pisum sativum L. as a function of plant phenology. J. Exp. Bot. 2003, 54, 2733–2744. [Google Scholar] [CrossRef]
- Matiru, V.N.; Dakora, F.D. Xylem transport and shoot accumulation of lumichrome, a newly recognized rhizobial signal, alters root respiration, stomatal conductance, leaf transpiration and photosynthetic rates in legumes and cereals. New Phytol. 2005, 165, 847–855. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, Y.; Tang, Y.; Zhu, X.G. Stomata conductance as a goalkeeper for increased photosynthetic efficiency. Curr. Opin. Plant Biol. 2022, 70, 102310. [Google Scholar] [CrossRef]
- Sakurai, G.; Miklavcic, S.J. A whole leaf comparative study of stomatal conductance models. Front. Plant Sci. 2022, 13, 766975. [Google Scholar] [CrossRef]
- Schubert, K.R.; Coker, G.T. Carbon metabolism in Soybean roots and nodules: Role of dark CO2 fixation. In World Soybean Research Conference III; Shibles, R., Ed.; CRC Press: Boca Raton, FL, USA, 2022; pp. 815–823. [Google Scholar] [CrossRef]
- Dakora, F.D. Defining new roles for plant and rhizobial molecules in sole and mixed plant cultures involving symbiotic legumes. New Phytol. 2003, 158, 39–49. [Google Scholar] [CrossRef]
- Dakora, F.D. Lumichrome: A bacterial signal molecule influencing plant growth. In Biological Nitrogen Fixation; de Bruijn, F.J., Ed.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2015; Volume 2, pp. 389–396. [Google Scholar] [CrossRef]
- Stefan, M.; Munteanu, N.; Stoleru, V.; Mihasan, M.; Hritcu, L. Seed inoculation with plant growth promoting rhizobacteria enhances photosynthesis and yield of runner bean (Phaseolus coccineus L.). Sci. Hortic. 2013, 151, 22–29. [Google Scholar] [CrossRef]
- Han, H.; Lee, K. Plant growth promoting rhizobacteria effect on antioxidant status, photosynthesis, mineral uptake and growth of lettuce under soil salinity. Res. J. Agric. Biol. Sci. 2005, 1, 210–215. Available online: https://www.aensiweb.net/AENSIWEB/rjabs/rjabs/210-215.pdf (accessed on 8 January 2024).
- Levy, A.; Salas Gonzalez, I.; Mittelviefhaus, M.; Clingenpeel, S.; Herrera Paredes, S.; Miao, J.; Wang, K.; Devescovi, G.; Stillman, K.; Monteiro, F.; et al. Genomic features of bacterial adaptation to plants. Nat. Genet. 2018, 50, 138–150. [Google Scholar] [CrossRef]
- Zeng, Q.; Xie, J.; Li, Y.; Gao, T.; Xu, C.; Wang, Q. Comparative genomic and functional analyses of four sequenced Bacillus cereus genomes reveal conservation of genes relevant to plant-growth-promoting traits. Sci. Rep. 2018, 8, 17009. [Google Scholar] [CrossRef] [PubMed]
- Brusamarello-Santos, L.C.; Gilard, F.; Brule, L.; Quillere, I.; Gourion, B.; Ratet, P.; Maltempi de Souza, E.; Lea, P.J.; Hirel, B. Metabolic profiling of two maize (Zea mays L.) inbred lines inoculated with the nitrogen fixing plant-interacting bacteria Herbaspirillum seropedicae and Azospirillum brasilense. PLoS ONE 2017, 12, e0174576. [Google Scholar] [CrossRef]
- McAdam, E.L.; Meitzel, T.; Quittenden, L.J.; Davidson, S.E.; Dalmais, M.; Bendahmane, A.I.; Thompson, R.; Smith, J.J.; Nichols, D.S.; Urquhart, S.; et al. Evidence that auxin is required for normal seed size and starch synthesis in pea. New Phytol. 2017, 216, 193–204. [Google Scholar] [CrossRef] [PubMed]
- Geigenberger, P. Regulation of starch biosynthesis in response to a fluctuating environment. Plant Physiol. 2011, 155, 1566–1577. [Google Scholar] [CrossRef]
- Palacios, O.A.; Choix, F.J.; Bashan, Y.; de-Bashan, L.E. Influence of tryptophan and indole-3-acetic acid on starch accumulation in the synthetic mutualistic Chlorella sorokiniana-Azospirillum brasilense system under heterotrophic conditions. Res. Microbiol. 2016, 167, 367–379. [Google Scholar] [CrossRef]
- Pilar, M.H.; García, J.M.; Pozo, M.J. Induced systemic resistance against Botrytis cinerea by Micromonospora strains isolated from root nodules. Front. Microbiol. 2020, 6, 922. [Google Scholar] [CrossRef]
- Nguyen, N.H.; Trotel-Aziz, P.; Villaume, S.; Rabenoelina, F.; Schwarzenberg, A.; Nguema-Ona, E.; Clément, C.; Baillieul, F.; Aziz, A. Bacillus subtilis and Pseudomonas fluorescens trigger common and distinct systemic immune responses in Arabidopsis thaliana depending on the pathogen lifestyle. Vaccines. 2020, 8, 503. [Google Scholar] [CrossRef]
- Miotto-Vilanova, L.; Jacquard, C.; Courteaux, B.; Wortham, L.; Michel, J.; Clément, C.; Barka, E.A.; Sanchez, L. Burkholderia phytofirmans PsJN confers grapevine resistance against Botrytis cinerea via a direct antimicrobial effect combined with a better resource mobilization. Front. Plant Sci. 2016, 7, 1236. [Google Scholar] [CrossRef] [PubMed]
- Xie, S.; Jiang, H.; Ding, T.; Xu, Q.; Chai, W.; Cheng, B. Bacillus amyloliquefaciens FZB42 represses plant miR846 to induce systemic resistance via a jasmonic acid-dependent signalling pathway. Mol. Plant Pathol. 2018, 19, 1612–1623. [Google Scholar] [CrossRef] [PubMed]
- Syed Nabi, R.B.; Shahzad, R.; Tayade, R.; Shahid, M.; Hussain, A.; Ali, M.W.; Yun, B.W. Evaluation potential of PGPR to protect tomato against Fusarium wilt and promote plant growth. PeerJ 2021, 9, e11194. [Google Scholar] [CrossRef] [PubMed]
- Komal, K.; Kumar, A.; Khan, M.A.; Dev, A.; Kumar, A.; Prasad, S.; Ahamad, N. Effect of plant growth-promoting rhizobacterium (PGPR) on chlorophyll content of chickpea plant (Cicer arietinum L.). Pharma Innovation J. 2022, 11, 1021–1025. Available online: https://www.thepharmajournal.com/archives/2022/vol11issue4/PartO/11-4-172-441.pdf (accessed on 8 January 2024).
- Soliman, S.A.; Abdelhameed, R.E.; Metwally, R.A. In vivo and In vitro evaluation of the antifungal activity of the PGPR Bacillus amyloliquefaciens RaSh1 (MZ945930) against Alternaria alternata with growth promotion influences on Capsicum annuum L. plants. Microb. Cell Fact. 2023, 22, 70. [Google Scholar] [CrossRef] [PubMed]
- Lobato, A.K.S.; Gonçalves-Vidigal, M.C.; Vidigal Filho, P.S.; Andrade, C.A.B.; Kvitschal, M.V.; Bonato, C.M. Relationships between leaf pigments and photosynthesis in common bean plants infected by anthracnose. N. Z. J. Crop Hort. 2010, 38, 29–37. [Google Scholar] [CrossRef]
- Yang, H.; Luo, P. Changes in photosynthesis could provide important insight into the interaction between wheat and fungal pathogens. Int. J. Mol. Sci. 2021, 22, 8865. [Google Scholar] [CrossRef]
- Vega, C.; Rodríguez, M.; Llamas, I.; Béjar, V.; Sampedro, I. Silencing of phytopathogen communication by the halotolerant PGPR Staphylococcus Equorum Strain EN21. Microorganisms 2020, 8, 42. [Google Scholar] [CrossRef]
- Hashem, A.; Abd_Allah, E.F.; Alqarawi, A.A.; Radhakrishnan, R.; Kumar, A. Plant defense approach of Bacillus subtilis (BERA 71) against Macrophomina phaseolina (Tassi) Goid in mung bean. J. Plant Interact. 2017, 12, 390–401. [Google Scholar] [CrossRef]
- Dong, J.; Chen, W. The role of autophagy in chloroplast degradation and chlorophagy in immune defenses during Pst DC3000 (AvrRps4) infection. PLoS ONE 2013, 8, e73091. [Google Scholar] [CrossRef]
- Simon, U.K.; Bauer, R.; Rioux, D.; Simard, M.; Oberwinkler, F. The vegetative life-cycle of the clover pathogen Cymadothea trifolii as revealed by transmission electron microscopy. Mycol. Res. 2005, 109, 764–778. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Wang, M.; Li, Y.; Gu, Z.; Ling, N.; Shen, Q.; Guo, S. Wilted cucumber plants infected by Fusarium oxysporum f. sp. cucumerinum do not suffer from water shortage. Ann. Bot. 2017, 120, 427–436. [Google Scholar] [CrossRef] [PubMed]
- Rocha, A.G.; Vothknecht, U.C. The role of calcium in chloroplasts-an intriguing and unresolved puzzle. Protoplasma. 2012, 249, 957–966. [Google Scholar] [CrossRef] [PubMed]
- Dietz, K.-J.; Turkan, I.; Krieger-Liszkay, A. Redox-and reactive oxygen species-dependent signaling into and out of the photosynthesizing chloroplast. Plant Physiol. 2016, 171, 1541–1550. [Google Scholar] [CrossRef] [PubMed]
- de Torres Zabala, M.; Littlejohn, G.; Jayaraman, S.; Studholme, D.; Bailey, T.; Lawson, T.; Tillich, M.; Licht, D.; Bölter, B.; Delfino, L.; et al. Chloroplasts play a central role in plant defence and are targeted by pathogen effectors. Nat. Plants 2015, 1, 15074. [Google Scholar] [CrossRef]
- Jelenska, J.; Yao, N.; Vinatzer, B.A.; Wright, C.M.; Brodsky, J.L.; Greenberg, J.T. A J domain virulence effector of Pseudomonas syringae remodels host chloroplasts and suppresses defenses. Curr. Biol. 2007, 17, 499–508. [Google Scholar] [CrossRef]
- Rodriguez-Herva, J.J.; González-Melendi, P.; Cuartas-Lanza, R.; Antúnez-Lamas, M.; Río-Alvarez, I.; Li, Z.; López-Torrejón, G.; Díaz, I.; Del Pozo, J.C.; Chakravarthy, S. A bacterial cysteine protease effector protein interferes with photosynthesis to suppress plant innate immune responses. Cell. Microbiol. 2012, 14, 669–681. [Google Scholar] [CrossRef] [PubMed]
- Jelenska, J.; Van Hal, J.A.; Greenberg, J.T. Pseudomonas syringae hijacks plant stress chaperone machinery for virulence. Proc. Natl. Acad. Sci. USA 2010, 107, 13177–13182. [Google Scholar] [CrossRef]
- Zhou, C.; Guo, J.; Zhu, L.; Xiao, X.; Xie, Y.; Zhu, J.; Ma, Z.; Wang, J. Paenibacillus polymyxa BFKC01 enhances plant iron absorption via improved root systems and activated iron acquisition mechanisms. Plant Physiol. Biochem. 2016, 105, 162–173. [Google Scholar] [CrossRef]
- Sarkar, J.; Chakraborty, B.; Chakraborty, U. Plant growth promoting rhizobacteria protect wheat plants against temperature stress through antioxidant signalling and reducing chloroplast and membrane injury. J. Plant Growth Regul. 2018, 37, 1396–1412. [Google Scholar] [CrossRef]
- Jeun, Y.C.; Kim, K.W.; Kim, K.D.; Hyun, J.W. Comparative ultrastructure of cucumbers pretreated with plant growth-promoting rhizobacteria, DL-3-aminobutyric acid or amino salicylic acid after inoculation with Colletotrichum orbiculare. J. Phytopathol. 2007, 155, 416–425. [Google Scholar] [CrossRef]
- Wang, S.; Uddin, M.I.; Tanaka, K.; Yin, L.; Shi, Z.; Qi, Y.; Mano, J.i.; Matsui, K.; Shimomura, N.; Sakaki, T. Maintenance of chloroplast structure and function by overexpression of the rice MONOGALACTOSYLDIACYLGLYCEROL SYNTHASE gene leads to enhanced salt tolerance in tobacco. Plant Physiol. 2014, 165, 1144–1155. [Google Scholar] [CrossRef]
- Dumanović, J.; Nepovimova, E.; Natić, M.; Kuča, K.; Jaćević, V. The significance of reactive oxygen species and antioxidant defense system in plants: A concise overview. Front. Plant Sci. 2020, 11, 552969. [Google Scholar] [CrossRef] [PubMed]
- Takagi, D.; Takumi, S.; Hashiguchi, M.; Sejima, T.; Miyake, C. Superoxide and singlet oxygen produced within the thylakoid membranes both cause photosystem I photoinhibition. Plant Physiol. 2016, 171, 1626–1634. [Google Scholar] [CrossRef]
- Mubarakshina, M.M.; Ivanov, B.N. The production and scavenging of reactive oxygen species in the plastoquinone pool of chloroplast thylakoid membranes. Physiol. Plantarum 2010, 140, 103–110. [Google Scholar] [CrossRef]
- Bordiec, S.; Paquis, S.; Lacroix, H.; Dhondt, S.; Barka, E.A.; Kauffmann, S.; Jeandet, P.; Mazeyrat-Gourbeyre, F.; Clément, C.; Baillieul, F. Comparative analysis of defence responses induced by the endophytic plant growth-promoting rhizobacterium Burkholderia phytofirmans strain PsJN and the non-host bacterium Pseudomonas syringae pv. pisi in grapevine cell suspensions. J. Exp. Bot. 2011, 62, 595–603. [Google Scholar] [CrossRef] [PubMed]
- Trdá, L.; Fernandez, O.; Boutrot, F.; Héloir, M.C.; Kelloniemi, J.; Daire, X.; Adrian, M.; Clément, C.; Zipfel, C.; Dorey, S. The grapevine flagellin receptor VvFLS2 differentially recognizes flagellin-derived epitopes from the endophytic growth-promoting bacterium Burkholderia phytofirmans and plant pathogenic bacteria. New Phytol. 2014, 201, 1371–1384. [Google Scholar] [CrossRef] [PubMed]
- Cai, X.; Zhao, H.; Liang, C.; Li, M.; Liu, R. Effects and mechanisms of symbiotic microbial combination agents to control tomato Fusarium crown and root rot disease. Front. Microbiol. 2021, 12, 629793. [Google Scholar] [CrossRef]
- Pan, J.; Peng, F.; Xue, X.; You, Q.; Zhang, W.; Wang, T.; Huang, C. The growth promotion of two salt-tolerant plant groups with PGPR inoculation: A meta-analysis. Sustainability 2019, 11, 378. Available online: https://www.mdpi.com/2071-1050/11/2/378 (accessed on 8 January 2024). [CrossRef]
- Ali, B.; Wang, X.; Saleem, M.H.; Hafeez, A.; Afridi, M.S.; Khan, S.; Ullah, I.; Amaral Júnior, A.T.d.; Alatawi, A.; Ali, S. PGPR-mediated salt tolerance in maize by modulating plant physiology, antioxidant defense, compatible solutes accumulation and bio-surfactant producing genes. Plants 2022, 11, 345. [Google Scholar] [CrossRef]
- Ali, B.; Wang, X.; Saleem, M.H.; Azeem, M.A.; Afridi, M.S.; Nadeem, M.; Ghazal, M.; Batool, T.; Qayyum, A.; Alatawi, A. Bacillus mycoides PM35 reinforces photosynthetic efficiency, antioxidant defense, expression of stress-responsive genes, and ameliorates the effects of salinity stress in maize. Life 2022, 12, 219. [Google Scholar] [CrossRef] [PubMed]
- White, A.C.; Rogers, A.; Rees, M.; Osborne, C.P. How can we make plants grow faster? A source-sink perspective on growth rate. J. Exp. Bot. 2016, 67, 31–45. [Google Scholar] [CrossRef]
- van de Mortel, J.E.; de Vos, R.C.; Dekkers, E.; Pineda, A.; Guillod, L.; Bouwmeester, K.; van Loon, J.J.; Dicke, M.; Raaijmakers, J.M. Metabolic and transcriptomic changes induced in Arabidopsis by the rhizobacterium Pseudomonas fluorescens SS101. Plant Physiol. 2012, 160, 2173–2188. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.; Yang, D.l.; Kong, Y.; Chen, Y.; Li, X.Z.; Zeng, L.J.; Li, Q.; Wang, E.T.; He, Z.H. Sugar homeostasis mediated by cell wall invertase GRAIN INCOMPLETE FILLING 1 (GIF1) plays a role in pre-existing and induced defence in rice. Mol. Plant Pathol. 2014, 15, 161–173. [Google Scholar] [CrossRef] [PubMed]
- Cho, Y.-H.; Yoo, S.-D. Signaling role of fructose mediated by FINS1/FBP in Arabidopsis thaliana. PLoS Genet. 2011, 7, e1001263. [Google Scholar] [CrossRef]
- Riva, D.S.; Ribaudo, C.M. Inoculation with Pseudomonas pseudoalcaligenes Lead to changes in plant sugar metabolism and defense that enhance tolerance against the pathogenic fungus Sclerotium rolfsii. ASRJETS 2020, 69, 89–104. Available online: https://core.ac.uk/download/pdf/327183331.pdf (accessed on 8 January 2024).
- Lanka, S.K.; Elderd, B.D.; Davis, J.A.; Stout, M.J. Jasmonic acid-induced resistance to fall armyworm in soybeans: Variation among genotypes and tradeoffs with constitutive resistance. Basic Appl. Ecol. 2021, 56, 97–109. [Google Scholar] [CrossRef]
- Fariduddin, Q.; Hayat, S.; Ahmad, A. Salicylic acid influences net photosynthetic rate, carboxylation efficiency, nitrate reductase activity, and seed yield in Brassica juncea. Photosynthetica 2003, 41, 281–284. [Google Scholar] [CrossRef]
- Pancheva, T.V.; Popova, L.P. Effect of salicylic acid on the synthesis of ribulose-1, 5-bisphosphate carboxylase/oxygenase in barley leaves. J. Plant Physiol. 1998, 152, 381–386. [Google Scholar] [CrossRef]
- Major, I.T.; Yoshida, Y.; Campos, M.L.; Kapali, G.; Xin, X.-F.; Sugimoto, K.; de Oliveira Ferreira, D.; He, S.Y.; Howe, G.A. Regulation of growth–defense balance by the JASMONATE ZIM-DOMAIN (JAZ)-MYC transcriptional module. Nat. Commun. 2017, 215, 1533–1547. [Google Scholar] [CrossRef]
- Kalachova, T.; Janda, M.; Šašek, V.; Ortmannová, J.; Nováková, P.; Dobrev, I.P.; Kravets, V.; Guivarc’h, A.; Moura, D.; Burketová, L.; et al. Identification of salicylic acid-independent responses in an Arabidopsis phosphatidylinositol 4-kinase beta double mutant. Ann. Bot. 2020, 125, 775–784. [Google Scholar] [CrossRef] [PubMed]
- Ullmann-Zeunert, L.; Stanton, M.A.; Wielsch, N.; Bartram, S.; Hummert, C.; Svatoš, A.; Baldwin, I.T.; Groten, K. Quantification of growth–defense trade-offs in a common currency: Nitrogen required for phenolamide biosynthesis is not derived from ribulose-1,5-bisphosphate carboxylase/oxygenase turnover. Plant J. 2013, 75, 417–429. [Google Scholar] [CrossRef] [PubMed]
- Stanton, M.A.; Ullmann-Zeunert, L.; Wielsch, N.; Bartram, S.; Svatoš, A.; Baldwin, I.T.; Groten, K. Silencing ribulose-1, 5-bisphosphate carboxylase/oxygenase expression does not disrupt nitrogen allocation to defense after simulated herbivory in Nicotiana attenuata. Plant Signal. Behav. 2013, 8, e27570. [Google Scholar] [CrossRef] [PubMed]
- Gamez, R.M.; Rodríguez, F.; Vidal, N.M.; Ramirez, S.; Vera Alvarez, R.; Landsman, D.; Mariño-Ramírez, L. Banana (Musa acuminata) transcriptome profiling in response to rhizobacteria: Bacillus amyloliquefaciens Bs006 and Pseudomonas fluorescens Ps006. BMC Genom. 2019, 20, 378. [Google Scholar] [CrossRef]




Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Su, F.; Zhao, B.; Dhondt-Cordelier, S.; Vaillant-Gaveau, N. Plant-Growth-Promoting Rhizobacteria Modulate Carbohydrate Metabolism in Connection with Host Plant Defense Mechanism. Int. J. Mol. Sci. 2024, 25, 1465. https://doi.org/10.3390/ijms25031465
Su F, Zhao B, Dhondt-Cordelier S, Vaillant-Gaveau N. Plant-Growth-Promoting Rhizobacteria Modulate Carbohydrate Metabolism in Connection with Host Plant Defense Mechanism. International Journal of Molecular Sciences. 2024; 25(3):1465. https://doi.org/10.3390/ijms25031465
Chicago/Turabian StyleSu, Fan, Bin Zhao, Sandrine Dhondt-Cordelier, and Nathalie Vaillant-Gaveau. 2024. "Plant-Growth-Promoting Rhizobacteria Modulate Carbohydrate Metabolism in Connection with Host Plant Defense Mechanism" International Journal of Molecular Sciences 25, no. 3: 1465. https://doi.org/10.3390/ijms25031465






