SARS-CoV-2 Antibodies in Breastmilk Three and Six Months Postpartum in Relation to the Trimester of Maternal SARS-CoV-2 Infection—An Exploratory Study
Abstract
:1. Introduction
2. Results
2.1. Demographics
2.2. IgA and IgG Frequency in Breastmilk and Serum Three and Six Months Post-Partum
2.2.1. Breastmilk
2.2.2. Serum
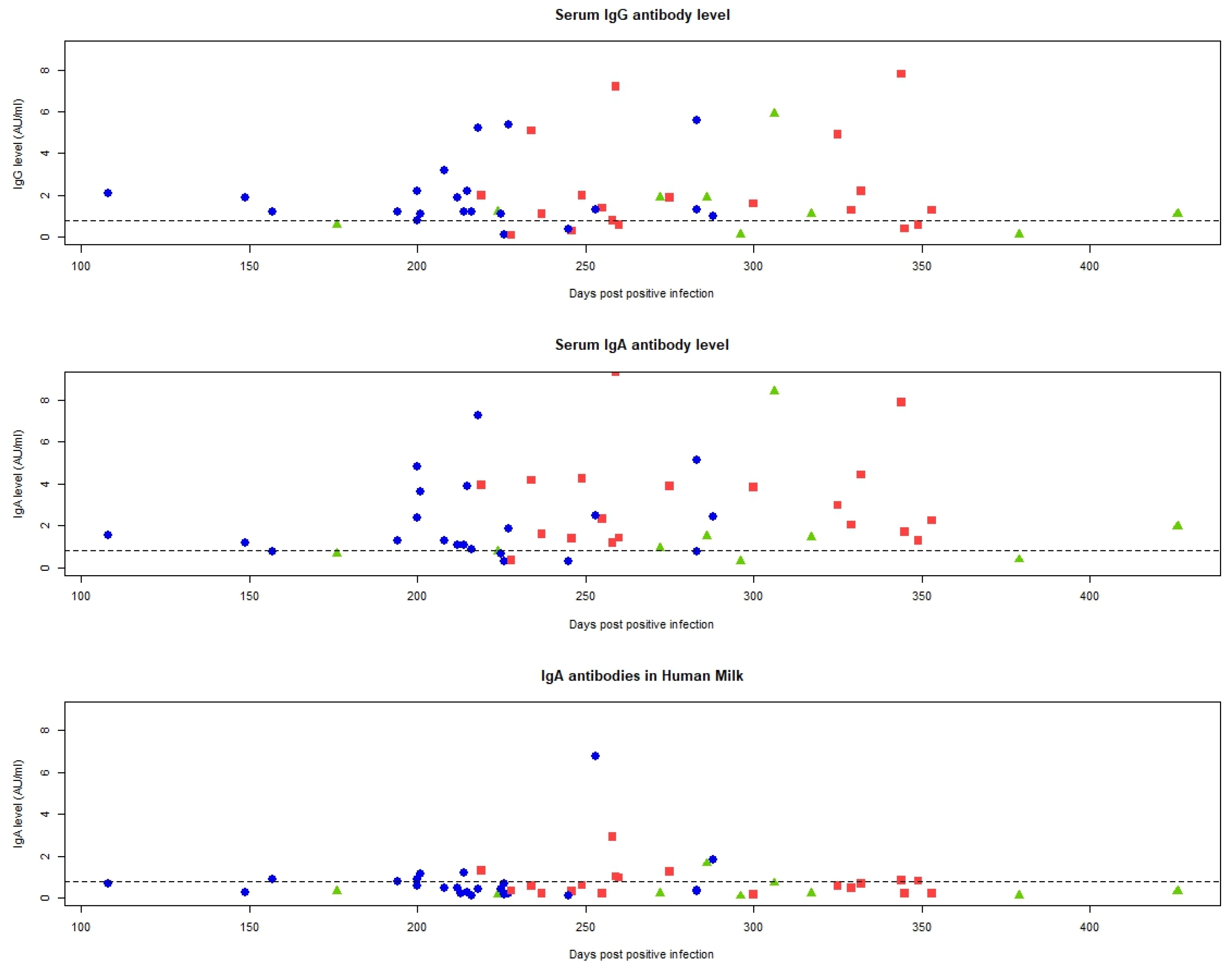
2.3. IgA and IgG Duration and Concentration in Breastmilk and Serum
2.3.1. Breastmilk
2.3.2. Serum
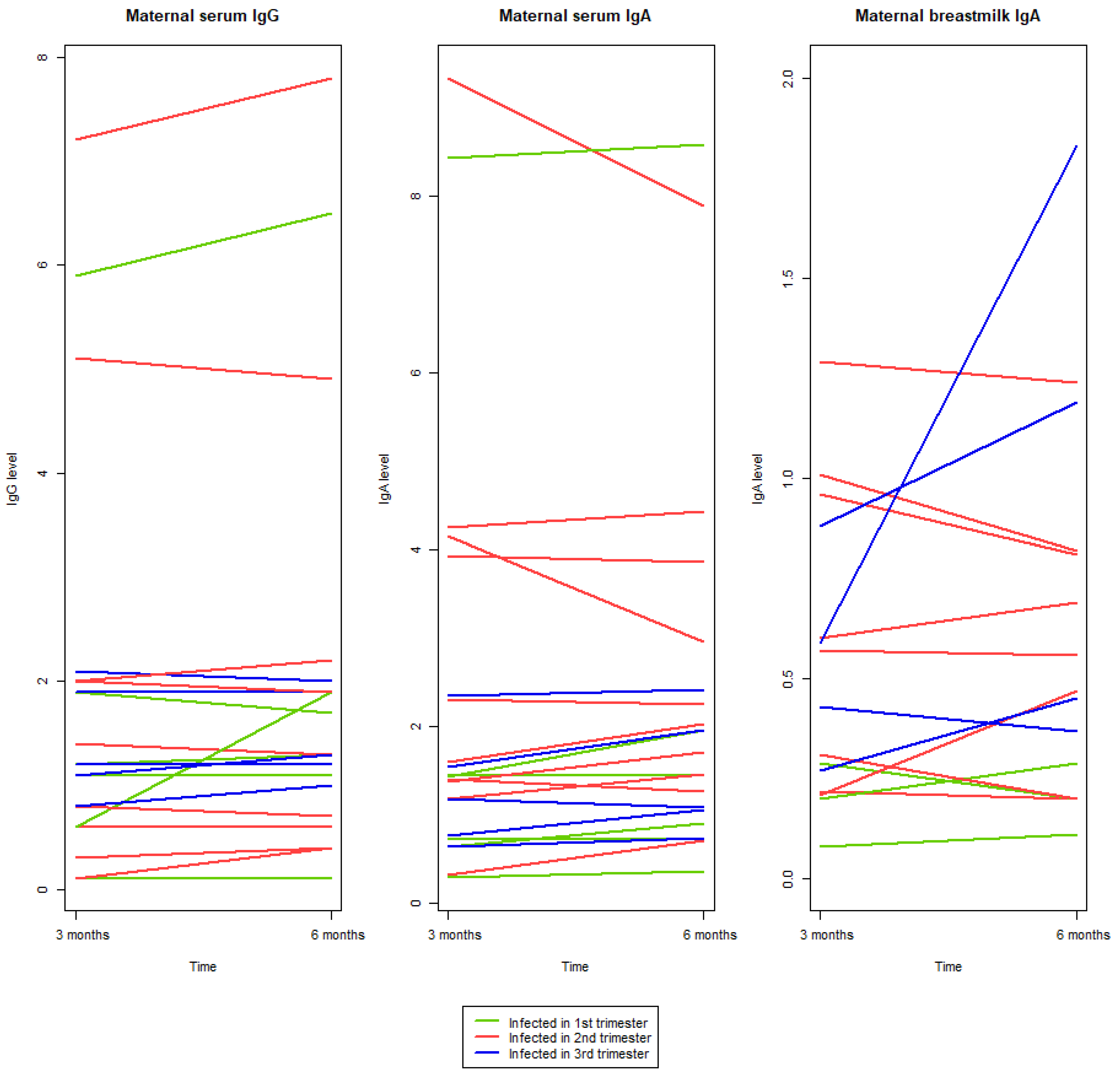
2.4. Antibody Development from Three to Six Months pp
2.5. Ratio of Breastmilk IgA and Serum IgG/IgA
2.6. Admission to Pediatric Emergency Ward
3. Discussion
4. Materials and Methods
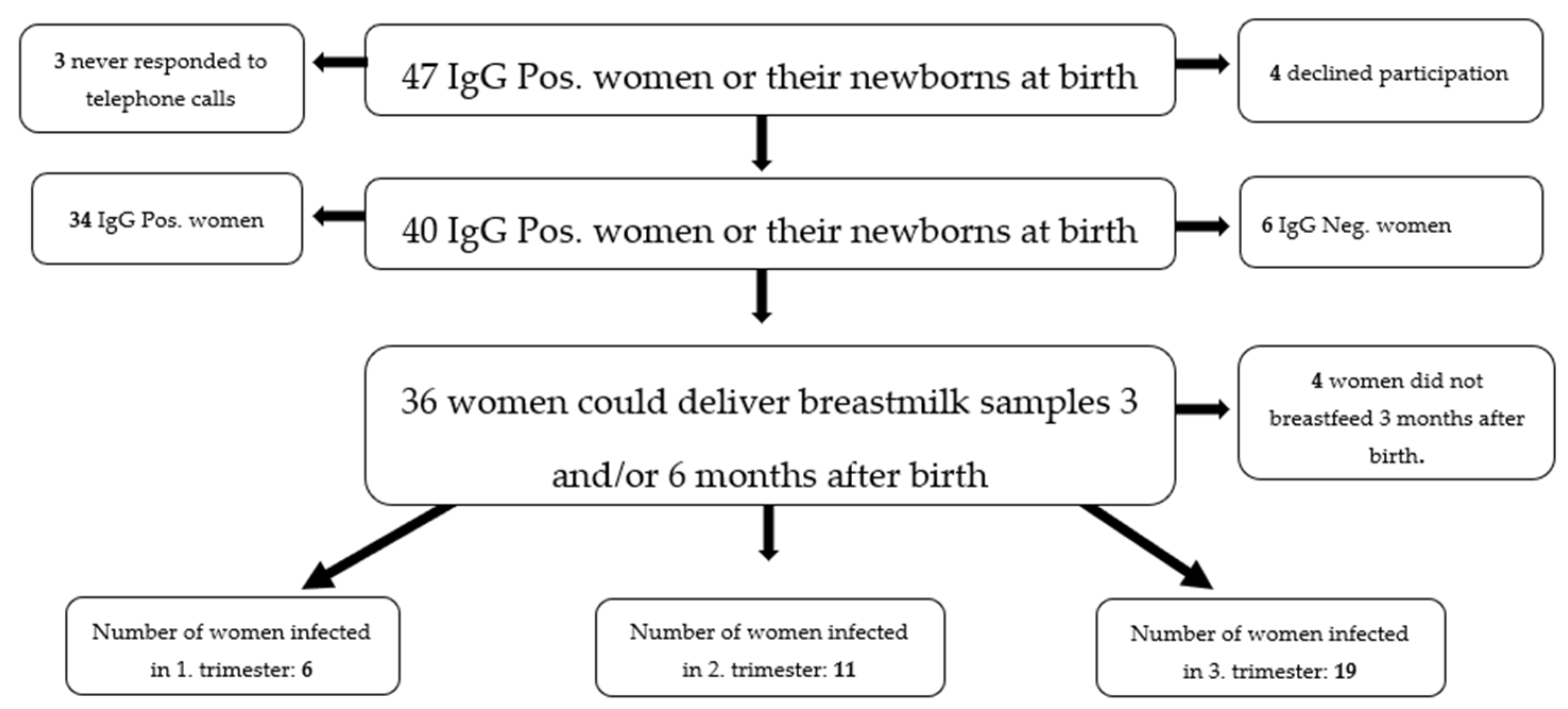
4.1. Study Population
4.2. Sample Collection
4.3. Pediatric Emergency Admission
4.4. Antibody Analysis
4.5. Statistical Analysis
4.6. Ethics
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- World Health Organization. Coronavirus Disease (COVID-19): Breastfeeding. Available online: https://www.who.int/emergencies/diseases/novel-coronavirus-2019/question-and-answers-hub/q-a-detail/coronavirus-disease-covid-19-breastfeeding (accessed on 12 April 2023).
- Centers for Disease Control and Prevention. 6 Things to Know about COVID-19 Vaccination for Children. 2023. Available online: https://www.cdc.gov/vaccines/covid-19/planning/children/6-things-to-know.html (accessed on 23 March 2023).
- Zavala, E.; Kruber, C.B.; Jaffe, E.F.; Nicklin, A.; Gur-Arie, G.; Wonodi, C.; Faden, R.R.; Karron, R.A. Global disparities in public health guidance for the use of COVID-19 vaccines in pregnancy. BMJ Global Health 2022, 7, e007730. [Google Scholar] [CrossRef]
- Low, J.M.; Low, Y.W.; Zhong, Y.; Lee, C.Y.C.; Chan, M.; Ng, N.B.H.; Amin, Z.; Ng, Y.P.M. Titres and neutralising capacity of SARS-CoV-2-specific antibodies in human milk: A systematic review. Arch. Dis. Child.-Fetal Neonatal Ed. 2021, 107, 174–180. [Google Scholar] [CrossRef] [PubMed]
- Young, B.E.; Seppo, A.E.; Diaz, N.; Rosen-Carole, C.; Nowak-Wegrzyn, A.; Vasquez, J.M.C.; Ferri-Huerta, R.; Nguyen-Contant, P.; Fitzgerald, T.; Sangster, M.Y.; et al. Association of Human Milk Antibody Induction, persistence, and neutralizing capacity with SARS-CoV-2 infection vs. mrna vaccination. JAMA Pediatr. 2022, 176, 159. [Google Scholar] [CrossRef] [PubMed]
- Fich, L.; Christiansen, A.H.; Hviid, K.V.R.; Aabakke, A.J.M.; Hoffmann, E.; Ingham, A.; Ollé-Lopez, J.; Rodríguez, J.B.; Juul-Larsen, G.B.; Kelstrup, L.; et al. SARS-CoV-2 Infection and Antibody Levels in Relation to Trimester of Infection: A Study from 11 Danish Obstetric Departments. Department of Obstetrics and Gynecology, Copenhagen University Hospital Hvidovre, Hvidovre, Denmark. 2023; (Not submitted). [Google Scholar]
- Wachman, E.M.; Snyder-Cappione, J.; Devera, J.; Boateng, J.; Dhole, Y.; Clarke, K.; Yuen, R.R.; Parker, S.E.; Hunnewell, J.; Ferraro, R.; et al. Maternal, infant, and breast milk antibody response following COVID-19 infection in early versus late gestation. Pediatr. Infect. Dis. J. 2023, 42, e70–e76. [Google Scholar] [CrossRef] [PubMed]
- Szczygioł, P.; Łukianowski, B.; Kościelska-Kasprzak, K.; Jakuszko, K.; Bartoszek, D.; Krajewska, M.; Królak-Olejnik, B. Antibodies in the breastmilk of COVID-19 recovered women. BMC Pregnancy Childbirth 2022, 22, 635. [Google Scholar] [CrossRef] [PubMed]
- Bobik, T.V.; Kostin, N.N.; Skryabin, G.A.; Tsabai, P.N.; Simonova, M.A.; Knorre, V.D.; Mokrushina, Y.A.; Smirnov, I.V.; Kosolapova, J.A.; Vtorushina, V.V.; et al. Epitope-specific response of human milk immunoglobulins in COVID-19 recovered women. Pathogens 2021, 10, 705. [Google Scholar] [CrossRef] [PubMed]
- Palmeira, P.; Carneiro-Sampaio, M. Immunology of Breast Milk. Rev. Assoc. Médica Bras. 2016, 62, 584–593. [Google Scholar] [CrossRef] [PubMed]
- Malek, A.; Sager, R.; Kuhn, P.; Nicolaides, K.H.; Schneider, H. Evolution of maternofetal transport of Immunoglobulins during human pregnancy. Am. J. Reprod. Immunol. 1996, 36, 248–255. [Google Scholar] [CrossRef] [PubMed]
- Albrecht, M.; Arck, P.C. Vertically transferred immunity in neonates: Mothers, mechanisms and mediators. Front. Immunol. 2020, 11, 555. [Google Scholar] [CrossRef] [PubMed]
- Zhong, Z.; Haltalli, M.; Holder, B.; Rice, T.; Donaldson, B.; O’Driscoll, M.; Le-Doare, K.; Kampmann, B.; Tregoning, J.S. The impact of timing of maternal influenza immunization on infant antibody levels at birth. Clin. Exp. Immunol. 2019, 195, 139–152. [Google Scholar] [CrossRef] [PubMed]
- Zilver, S.J.M.; de Groot, C.J.M.; Grobben, M.; Remmelzwaal, S.; Burgers, E.; Velasco, D.N.; Juncker, H.G.; van Keulen, B.J.; van Goudoever, J.B.; de Leeuw, R.A.; et al. Vaccination from the early second trimester onwards gives a robust SARS-CoV-2 antibody response throughout pregnancy and provides antibodies for the neonate. Int. J. Infect. Dis. 2023, 130, 126–135. [Google Scholar] [CrossRef] [PubMed]
- Dehgani-Mobaraki, P.; Zaidi, A.K.; Yadav, N.; Floridi, A.; Floridi, E. Longitudinal observation of antibody responses for 14 months after SARS-CoV-2 infection. Clin. Immunol. 2021, 230, 108814. [Google Scholar] [CrossRef] [PubMed]
- Gudbjartsson, D.F.; Norddahl, G.L.; Melsted, P.; Gunnarsdottir, K.; Holm, H.; Eythorsson, E.; Arnthorsson, A.O.; Helgason, D.; Bjarnadottir, K.; Ingvarsson, R.F.; et al. Humoral immune response to SARS-CoV-2 in Iceland. N. Engl. J. Med. 2020, 383, 1724–1734. [Google Scholar] [CrossRef] [PubMed]
- Flaherman, V.J.; Afshar, Y.; Boscardin, W.J.; Keller, R.L.; Mardy, A.H.; Prahl, M.K.; Phillips, C.T.; Asiodu, I.V.; Berghella, V.; Chambers, B.D.; et al. Infant outcomes following maternal infection with severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2): First report from the Pregnancy Coronavirus Outcomes Registry (priority) study. Clin. Infect. Dis. 2021, 73, e2810–e2813. [Google Scholar] [CrossRef] [PubMed]
- COVID-19 i Danmark—SST. Available online: https://www.sst.dk/-/media/Nyheder/2020/COVID-19-i-Danmark_-Epidemiens-foerste-boelge_-Status-og-Strategi_-Version-23_-marts-2020.ashx?la=da&hash=263A3D8EAB851F406EAA6DA81D6EA91A64F1A087 (accessed on 30 May 2023).
- The Comprehensive R Archive Network. Implementation of Case-Control Optimal Matching, R Package ccoptimalmatch Version 0.1.0.; R Foundation for Statistical Computing: Vienna, Austria, 2021; Available online: https://cran.rproject.org/web/packages/ccoptimalmatch/index.html (accessed on 3 April 2023).
- The R Project for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria; Available online: https://www.r-project.org/ (accessed on 3 April 2023).

| Study Participants | No. of Samples 3 Months after Birth | No. of Samples 6 Months after Birth |
|---|---|---|
| First trimester of infection: | 6 | 3 |
| Second trimester of infection: | 11 | 8 |
| Third trimester of infection: | 5 | 18 |
| Cases (n = 36) | Controls (n = 180) | |||
|---|---|---|---|---|
| Variables | n | (%) | n | (%) |
| Age *, years; median(IQR) | 32 | (29;36) | 32 | (29;36) |
| Parity | ||||
| Primipara | 20 | (56) | 89 | (49) |
| Multipara | 16 | (44) | 91 | (51) |
| Body mass index, points; median(IQR) | 23 | (22;25) | 23 | (21;27) |
| Smoking, yes | ≤3 | 4 | (2) | |
| Obstetric complications, yes | ||||
| GestHT | 0 | (0) | 4 | (2) |
| GDM | ≤3 | (-) | 16 | (9) |
| Liver complications | ≤3 | (-) | ≤3 | (-) |
| PPROM | 0 | (0) | ≤3 | (-) |
| Post-partum bleeding | 7 | (19) | 50 | (28) |
| Acute sectio | 5 | (14) | 23 | (13) |
| Mode of delivery, yes | ||||
| Vaginal | 27 | (75) | 141 | (78) |
| Cesarian | 9 | (25) | 39 | (22) |
| Gestational age at birth *, days; median(IQR) | 281 | (277;289) | 281 | (277;289) |
| Apgar score | ||||
| At 5 min., points; median(IQR) | 10 | (10;10) | 10 | (10;10) |
| Less than 7 points, yes | 0 | (0) | ≤3 | (-) |
| Preterm birth, yes | ≤3 | (-) | 9 | (5) |
| IgG Serum | IgA Serum | IgA Breastmilk | ||||
|---|---|---|---|---|---|---|
| 3 Months | 6 Months | 3 Months | 6 Months | 3 Months | 6 Months | |
| Number | 22 | 33 | 22 | 33 | 22 | 29 |
| No. of samples per trimester | ||||||
| First trimester | 6 | 6 | 6 | 6 | 6 | 3 |
| Second trimester | 11 | 10 | 11 | 10 | 11 | 8 |
| Third trimester | 5 | 17 | 5 | 17 | 5 | 18 |
| No. (%) of positive samples per trimester | ||||||
| First trimester | 4 (67%) | 5 (83%) | 3 (50%) | 4 (67%) | <3 | 0 (0%) |
| Second trimester | 8 (73%) | 6 (60%) | 10 (90%) | 9 (91%) | 4 (36%) | 3 (38%) |
| Third trimester | 5 (100%) | 15 (88%) | 3 (60%) | 14 (82%) | <3 | 5 (28%) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fich, L.; Christiansen, A.-M.H.; Nilsson, A.C.; Lindman, J.; Juul-Larsen, H.G.; Hansen, C.B.; la Cour Freiesleben, N.; Khalil, M.R.; Nielsen, H.S. SARS-CoV-2 Antibodies in Breastmilk Three and Six Months Postpartum in Relation to the Trimester of Maternal SARS-CoV-2 Infection—An Exploratory Study. Int. J. Mol. Sci. 2024, 25, 3269. https://doi.org/10.3390/ijms25063269
Fich L, Christiansen A-MH, Nilsson AC, Lindman J, Juul-Larsen HG, Hansen CB, la Cour Freiesleben N, Khalil MR, Nielsen HS. SARS-CoV-2 Antibodies in Breastmilk Three and Six Months Postpartum in Relation to the Trimester of Maternal SARS-CoV-2 Infection—An Exploratory Study. International Journal of Molecular Sciences. 2024; 25(6):3269. https://doi.org/10.3390/ijms25063269
Chicago/Turabian StyleFich, Line, Ann-Marie Hellerung Christiansen, Anna Christine Nilsson, Johanna Lindman, Helle Gybel Juul-Larsen, Christine Bo Hansen, Nina la Cour Freiesleben, Mohammed Rohi Khalil, and Henriette Svarre Nielsen. 2024. "SARS-CoV-2 Antibodies in Breastmilk Three and Six Months Postpartum in Relation to the Trimester of Maternal SARS-CoV-2 Infection—An Exploratory Study" International Journal of Molecular Sciences 25, no. 6: 3269. https://doi.org/10.3390/ijms25063269






