Ancistrocladus tectorius Extract Inhibits Obesity by Promoting Thermogenesis and Mitochondrial Dynamics in High-Fat Diet-Fed Mice
Abstract
1. Introduction
2. Results
2.1. HPLC Analyses of AT Extract and its Butanol (BuOH) Fraction
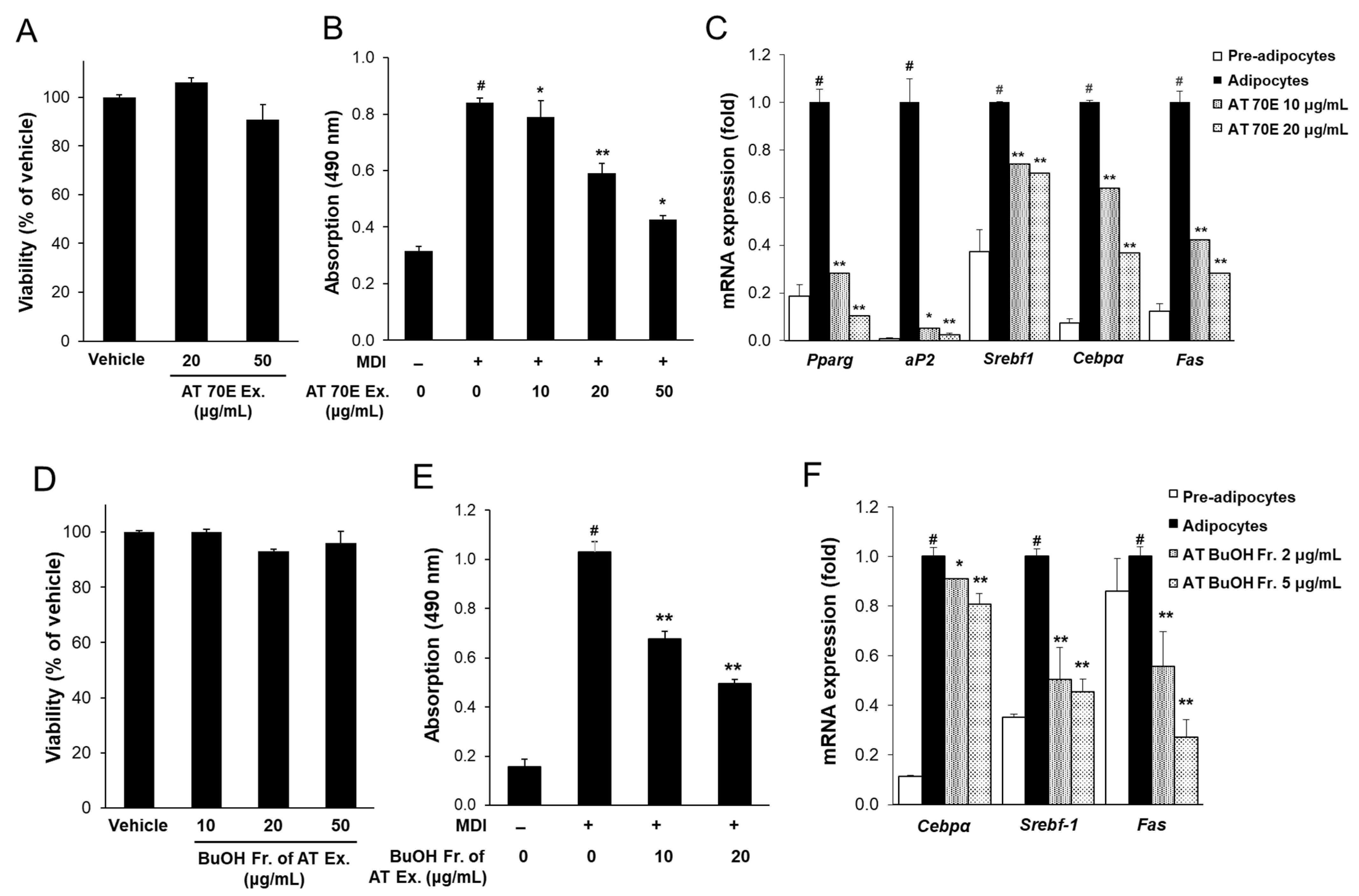
2.2. Effect of AT Extract and Its BuOH Fraction on Cytotoxicity and Adipogenesis in 3T3-L1 Cells
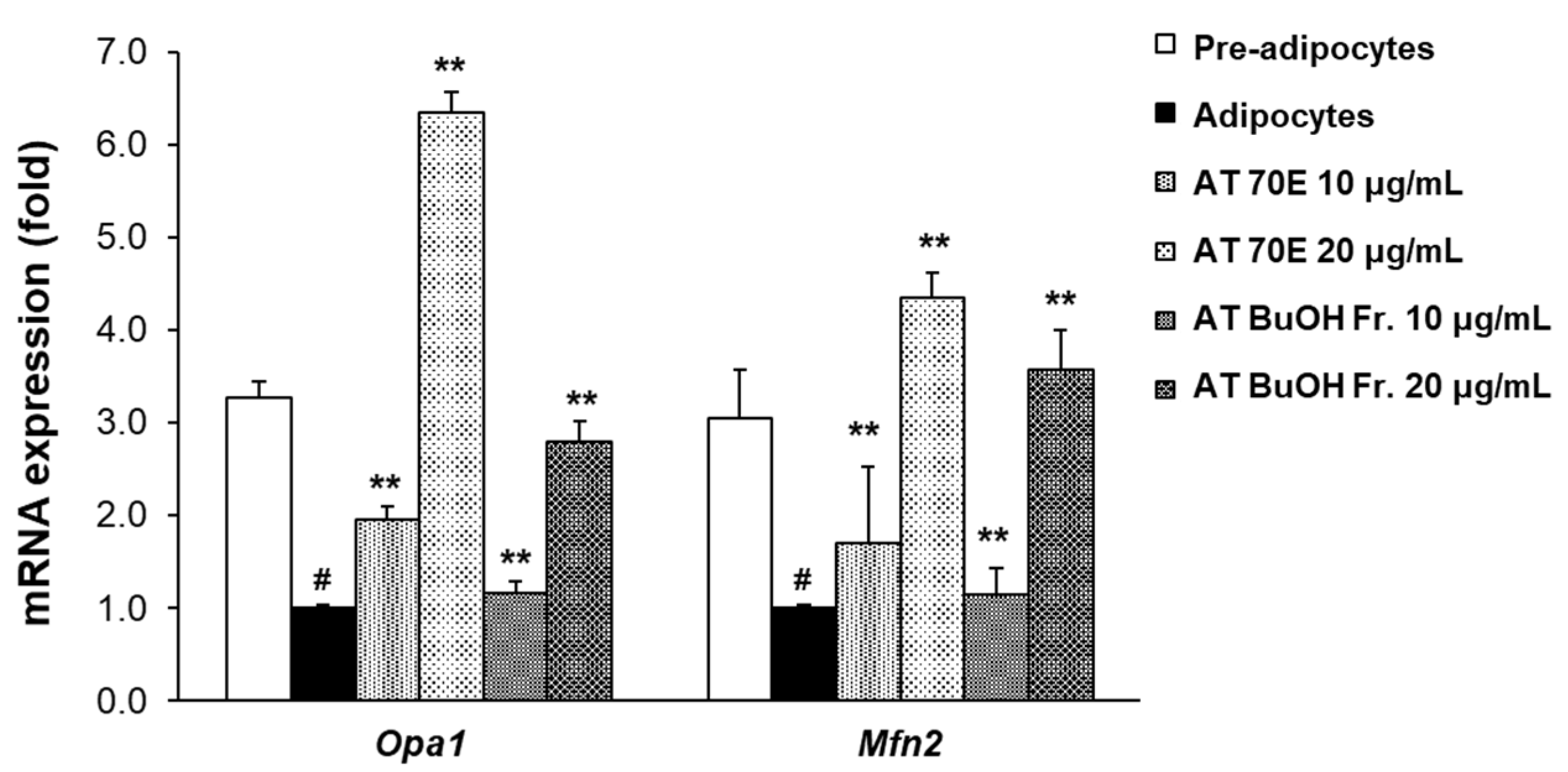
2.3. Effect of AT Extract and Its BuOH fraction on Mitochondrial Dynamics in Mature 3T3-L1 Adipocytes
2.4. Effect of AT 70% EtOH Extract on Body and Organ Weights
2.5. Effect of AT Extract on Glucose Homeostasis and Plasma Profiles

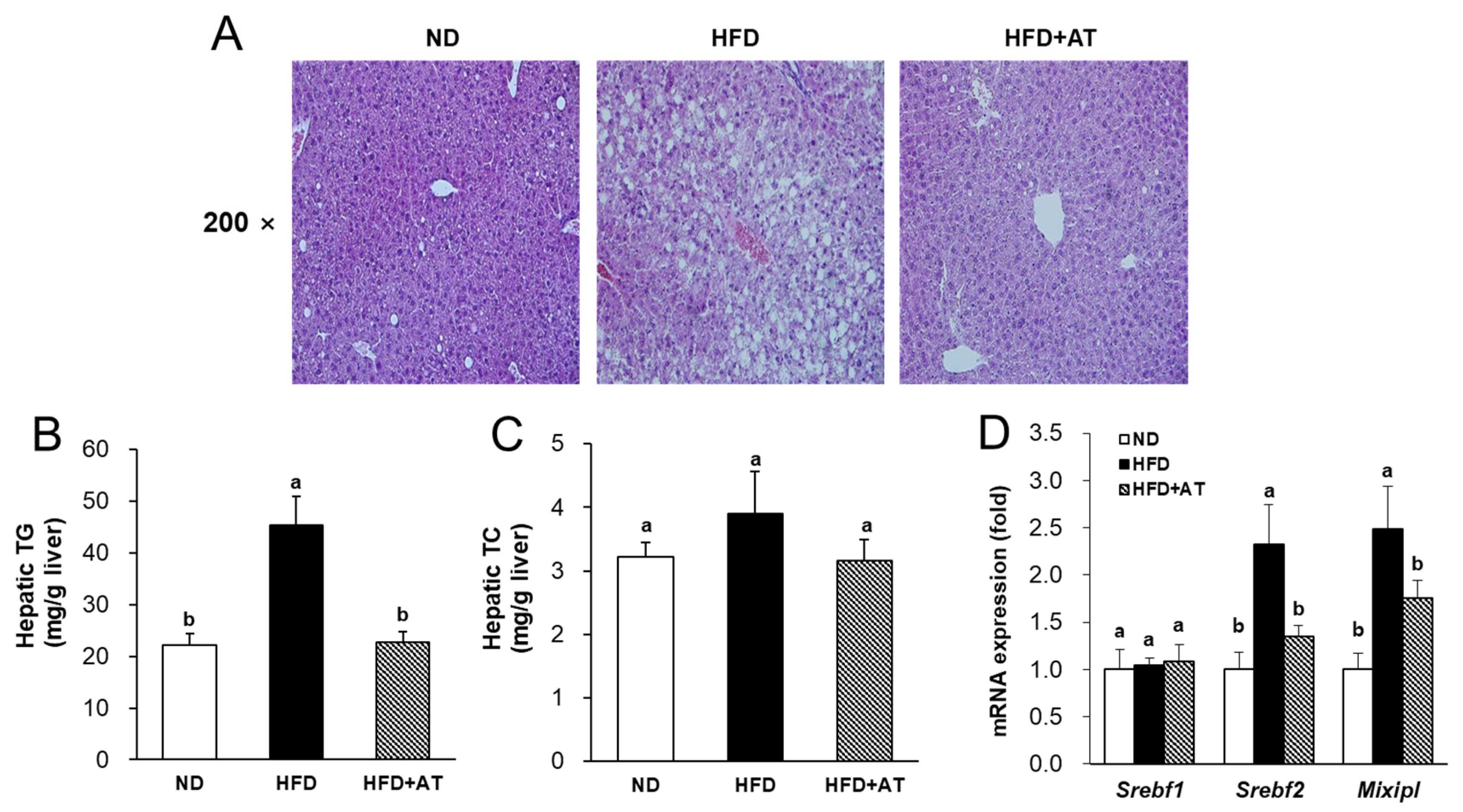
2.6. Effect of AT Extract on Regulation of Hepatic Steatosis
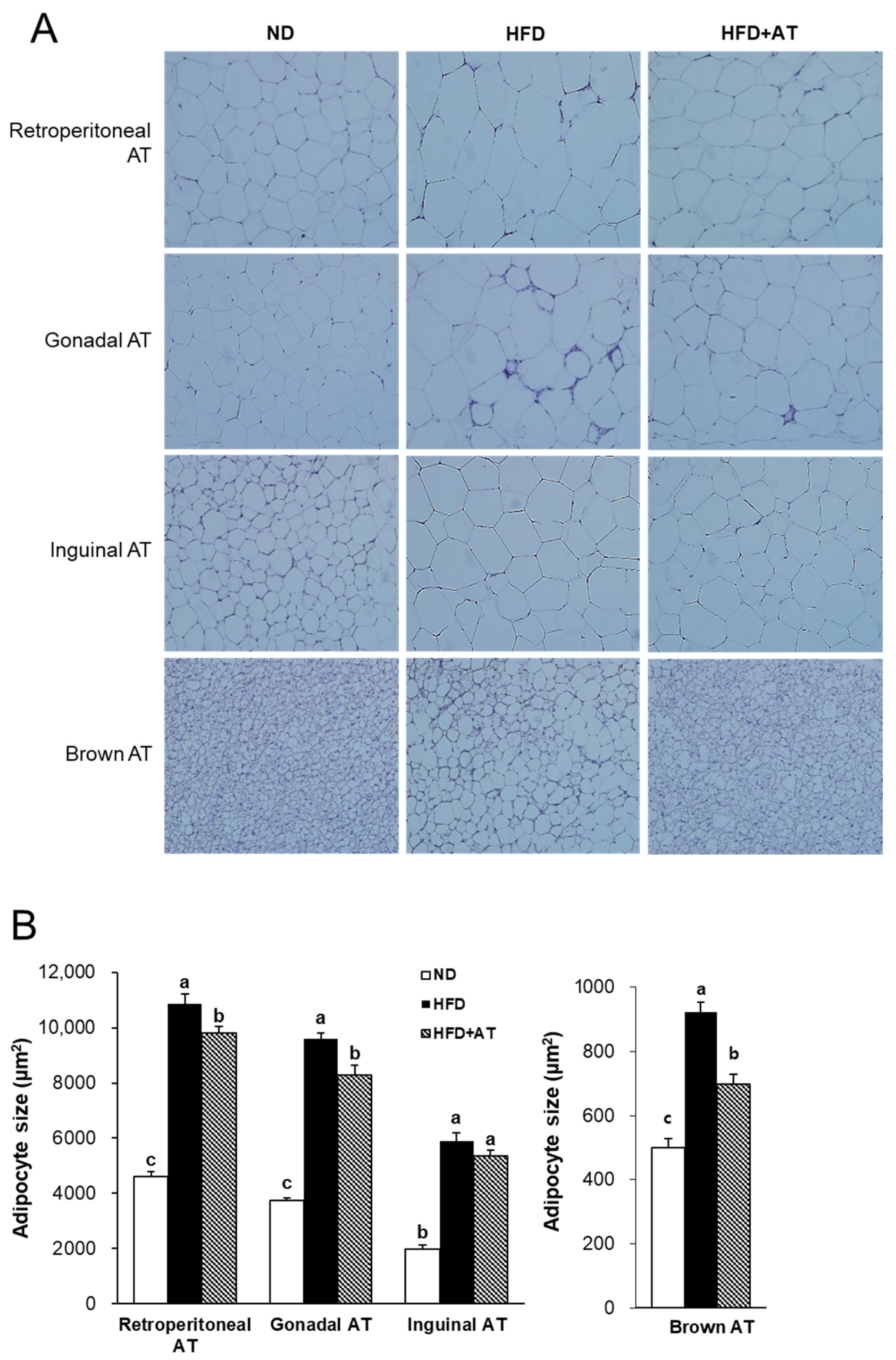
2.7. Effect of AT Extract on Adipocyte Hypertrophy in Adipose Tissues
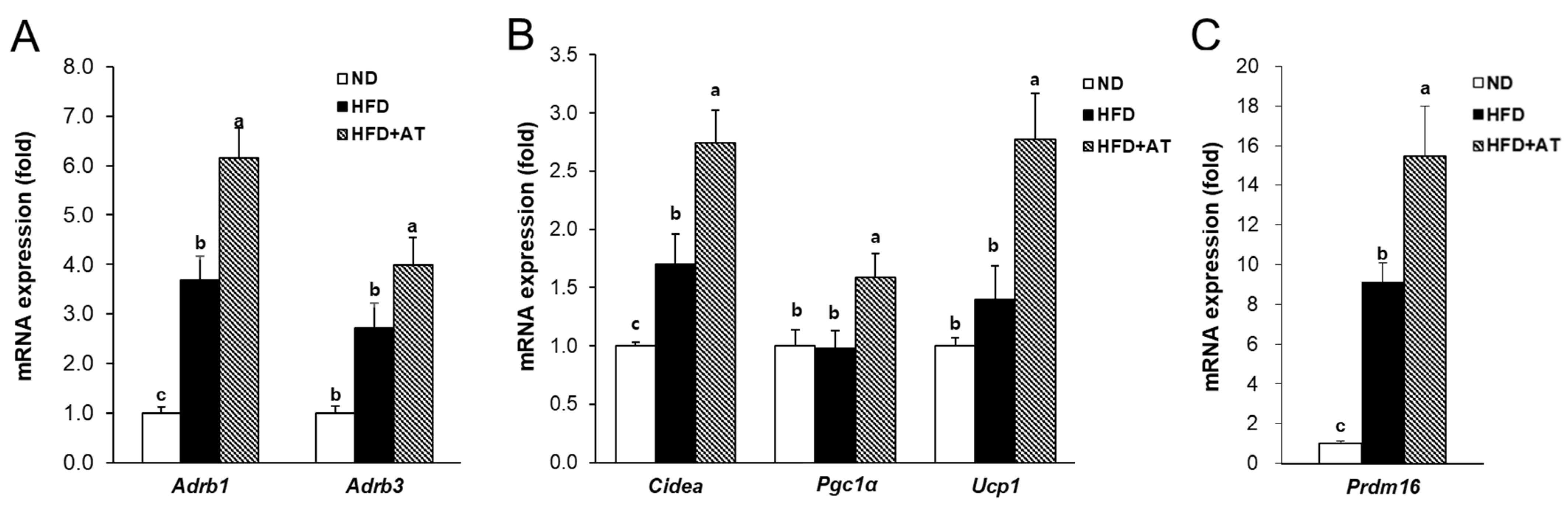
2.8. Effect of AT Extract on Thermogenesis of BAT in HFD-Induced Obese Mice
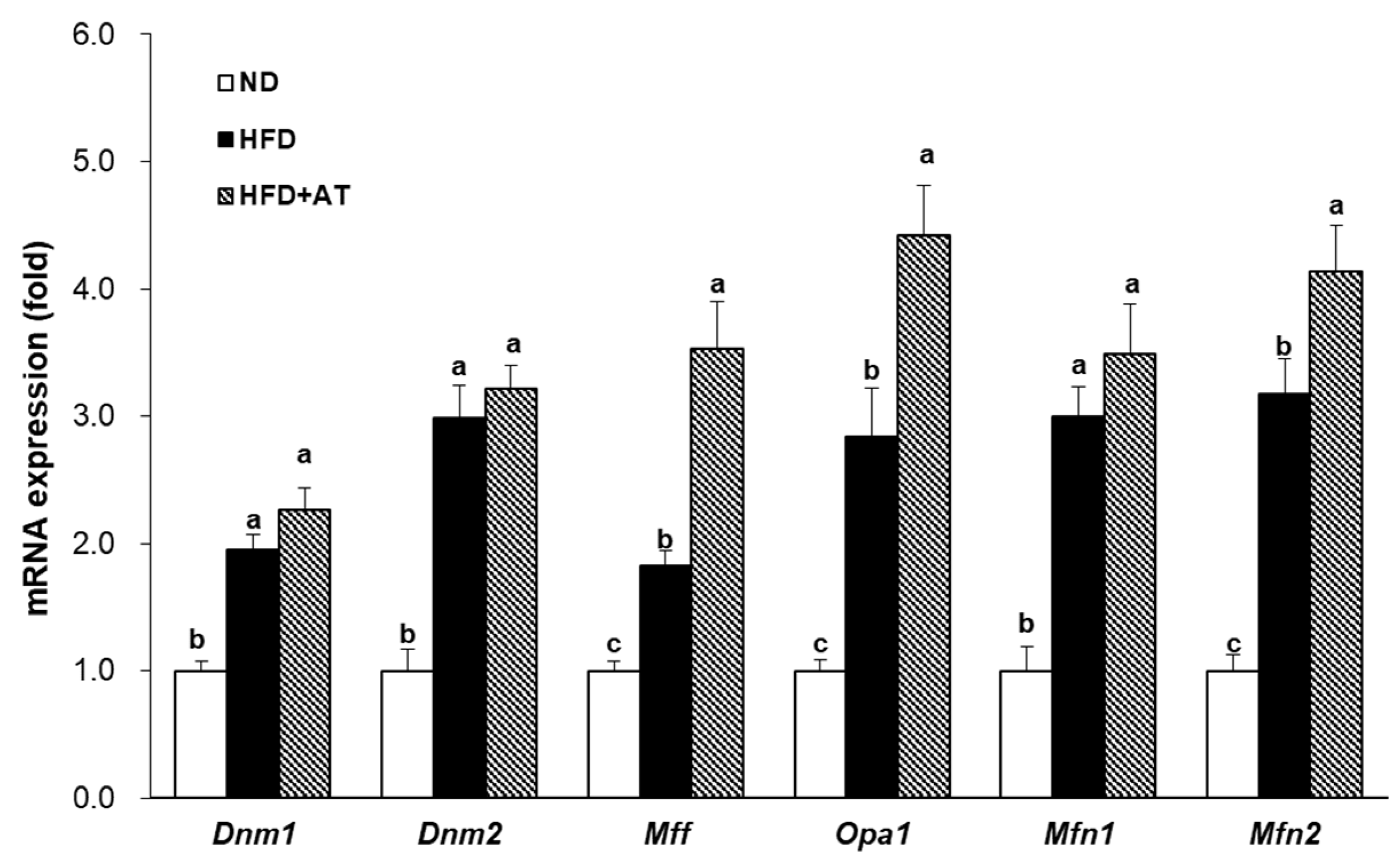
2.9. Effect of AT Extract on BAT Mitochondrial Dynamics in HFD-Fed Mice
3. Discussion
4. Materials and Methods
4.1. Preparation of AT Extract and Its BuOH fraction
4.2. HPLC Analyses of AT Extract and Its Butanol (BuOH) Fraction
4.3. Culture and Differentiation of 3T3-L1 Cells
4.4. Animals and Diets
4.5. Measurement of Body Weight and Plasma Profiles
4.6. Oral Glucose-Tolerance Test (OGTT)
4.7. Histological Analysis
4.8. Quantitative Real-Time RT-PCR (qRT-PCR)
4.9. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- White, U. Adipose tissue expansion in obesity, health, and disease. Front. Cell Dev. Biol. 2023, 11, 1188844. [Google Scholar] [CrossRef] [PubMed]
- Zou, Y.; Sheng, G.; Yu, M.; Xie, G. The association between triglycerides and ectopic fat obesity: An inverted U-shaped curve. PLoS ONE 2020, 15, e0243068. [Google Scholar] [CrossRef]
- Rhee, E.J. The influence of obesity and metabolic health on vascular health. Endocrinol. Metab. 2022, 37, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Liesa, M.; Shirihai, O.S. Mitochondrial dynamics in the regulation of nutrient utilization and energy expenditure. Cell Metab. 2013, 17, 491–506. [Google Scholar] [CrossRef] [PubMed]
- Luo, L.; Liu, M. Adipose tissue in control of metabolism. J. Endocrinol. 2016, 231, R77–R99. [Google Scholar] [CrossRef]
- Cannon, B.; Nedergaard, J. Brown adipose tissue: Function and physiological significance. Physiol. Rev. 2004, 84, 277–359. [Google Scholar] [CrossRef] [PubMed]
- Porter, C. Quantification of UCP1 function in human brown adipose tissue. Adipocyte 2017, 6, 167–174. [Google Scholar] [CrossRef] [PubMed]
- Auger, C.; Kajimura, S. Adipose tissue remodeling in pathophysiology. Annu. Rev. Pathol. 2023, 18, 71–93. [Google Scholar] [CrossRef]
- Adebayo, M.; Singh, S.; Singh, A.P.; Dasgupta, S. Mitochondrial fusion and fission: The fine-tune balance for cellular homeostasis. Faseb. J. 2021, 35, e21620. [Google Scholar] [CrossRef]
- Giacomello, M.; Pyakurel, A.; Glytsou, C.; Scorrano, L. The cell biology of mitochondrial membrane dynamics. Nat. Rev. Mol. Cell Biol. 2020, 21, 204–224. [Google Scholar] [CrossRef]
- Tilokani, L.; Nagashima, S.; Paupe, V.; Prudent, J. Mitochondrial dynamics: Overview of molecular mechanisms. Essays Biochem. 2018, 62, 341–360. [Google Scholar] [PubMed]
- Ruangrungsi, N.; Wongpanich, V.; Tantivatana, P.; Cowe, H.J.; Cox, P.J.; Funayama, S.; Cordell, G.A. Traditional medicinal plants of Thailand, V. Ancistrotectorine, a new naphthalene-isoquinoline alkaloid from Ancistrocladus tectorius. J. Nat. Prod. 1985, 48, 529–535. [Google Scholar] [CrossRef]
- Siti Munirah, M.Y. Ancistrocladus tectorius (Malaysia). Malaysia Biodiversity Information System (MyBIS). Available online: https://www.mybis.gov.my/art/149 (accessed on 25 March 2024).
- Jiang, C.; Li, Z.L.; Gong, P.; Kang, S.L.; Liu, M.S.; Pei, Y.H.; Jing, Y.K.; Hua, H.M. Five novel naphthylisoquinoline alkaloids with growth inhibitory activities against human leukemia cells HL-60, K562 and U937 from stems and leaves of Ancistrocladus tectorius. Fitoterapia 2013, 91, 305–312. [Google Scholar] [CrossRef] [PubMed]
- Said, I.M.; Ahmad, I.B.; Yahya, M.D.; Marini, A.M. Biological activity studies of Ancistrocladus tectorius. Pharmaceut. Biol. 2008, 39, 357–363. [Google Scholar] [CrossRef]
- Chen, Z.; Wang, B.; Qin, K.; Zhang, B.; Su, Q.; Lin, Q. Isolation and identification of alkaloids from Ancistrocladus tectorius. Acta Pharm. Sin. 1981, 16, 519–523. [Google Scholar]
- Bringmann, G.; Zhang, G.; Olschläger, T.; Stich, A.; Wu, J.; Chatterjee, M.; Brun, R. Highly selective antiplasmodial naphthylisoquinoline alkaloids from Ancistrocladus tectorius. Phytochemistry 2013, 91, 220–228. [Google Scholar] [CrossRef] [PubMed]
- François, G.; Timperman, G.; Haller, R.; Bär, S.; Isahakia, M.; Robertson, S.; Zhao, C.; De Souza, N.; Aké Assi, L.; Holenz, J. Growth inhibition of asexual erythrocytic forms of Plasmodium falciparum and P. berghei in vitro by naphthylisoquinoline alkaloid-containing extracts of Ancistrocladus and Triphyophyllum species. Int. J. Pharmacogn. 1997, 35, 55–59. [Google Scholar] [CrossRef]
- Wiart, C.; Mogana, S.; Khalifah, S.; Mahan, M.; Ismail, S.; Buckle, M.; Narayana, A.K.; Sulaiman, M. Antimicrobial screening of plants used for traditional medicine in the state of Perak, Peninsular Malaysia. Fitoterapia 2004, 75, 68–73. [Google Scholar]
- Bringmann, G.; Kajahn, I.; Reichert, M.; Pedersen, S.E.; Faber, J.H.; Gulder, T.; Brun, R.; Christensen, S.B.; Ponte-Sucre, A.; Moll, H.; et al. Ancistrocladinium A and B, the First N,C-Coupled Naphthyldihydroisoquinoline Alkaloids, from a Congolese Ancistrocladus Species. J. Org. Chem. 2006, 71, 9348–9356. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.H.; Park, A.; Oh, K.J.; Lee, S.C.; Kim, W.K.; Bae, K.H. The role of adipose tissue mitochondria: Regulation of mitochondrial function for the treatment of metabolic diseases. Int. J. Mol. Sci. 2019, 20, 4924. [Google Scholar] [CrossRef]
- Fernández-Verdejo, R.; Marlatt, K.L.; Ravussin, E.; Galgani, J.E. Contribution of brown adipose tissue to human energy metabolism. Mol. Aspects. Med. 2019, 68, 82–89. [Google Scholar] [CrossRef]
- Hill, J.O.; Wyatt, H.R.; Peters, J.C. Energy balance and obesity. Circulation 2012, 126, 126–132. [Google Scholar] [CrossRef] [PubMed]
- Lin, X.; Li, H. Obesity: Epidemiology, pathophysiology, and therapeutics. Front. Endocrinol. 2021, 12, 706978. [Google Scholar] [CrossRef] [PubMed]
- Kotzbeck, P.; Giordano, A.; Mondini, E.; Murano, I.; Severi, I.; Venema, W.; Cecchini, M.P.; Kershaw, E.E.; Barbatelli, G.; Haemmerle, G.; et al. Brown adipose tissue whitening leads to brown adipocyte death and adipose tissue inflammation. J. Lipid Res. 2018, 59, 784–794. [Google Scholar] [CrossRef] [PubMed]
- Villarroya, F.; Cereijo, R.; Gavaldà-Navarro, A.; Villarroya, J.; Giralt, M. Inflammation of brown/beige adipose tissues in obesity and metabolic disease. J. Intern. Med. 2018, 284, 492–504. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Zhang, Z.; Song, Y.; Xie, H.; Dong, M. An update on brown adipose tissue and obesity intervention: Function, regulation and therapeutic implications. Front. Endocrinol. 2022, 13, 1065263. [Google Scholar] [CrossRef] [PubMed]
- Townsend, K.; Tseng, Y.H. Brown adipose tissue: Recent insights into development, metabolic function and therapeutic potential. Adipocyte 2012, 1, 13–24. [Google Scholar] [CrossRef]
- Auger, C.; Kajimura, S. Detouring adrenergic stimulation to induce adipose thermogenesis. Nat. Rev. Endocrinol. 2021, 17, 579–580. [Google Scholar] [CrossRef] [PubMed]
- Cero, C.; Lea, H.J.; Zhu, K.Y.; Shamsi, F.; Tseng, Y.H.; Cypess, A.M. Cypess, β3-Adrenergic receptors regulate human brown/beige adipocyte lipolysis and thermogenesis. JCI Insight 2021, 6, e139160. [Google Scholar] [CrossRef]
- Stanford, K.I.; Middelbeek, R.J.; Townsend, K.L.; An, D.; Nygaard, E.B.; Hitchcox, K.M.; Markan, K.R.; Nakano, K.; Hirshman, M.F.; Tseng, Y.H.; et al. Brown adipose tissue regulates glucose homeostasis and insulin sensitivity. J. Clin. Investig. 2013, 123, 215–223. [Google Scholar] [CrossRef]
- Ohno, H.; Shinoda, K.; Spiegelman, B.M.; Kajimura, S. PPARγ agonists induce a white-to-brown fat conversion through stabilization of PRDM16 protein. Cell Metab. 2012, 15, 395–404. [Google Scholar] [CrossRef]
- Lin, S.C.; Li, P. CIDE-A, a novel link between brown adipose tissue and obesity. Trends Mol. Med. 2004, 10, 434–439. [Google Scholar] [CrossRef] [PubMed]
- Cinti, S. Transdifferentiation properties of adipocytes in the adipose organ. Am. J. Physiol. Endocrinol. Metab. 2009, 297, E977–E986. [Google Scholar] [CrossRef] [PubMed]
- Dai, W.; Jiang, L. Dysregulated mitochondrial dynamics and detabolism in obesity, diabetes, and cancer. Front. Endocrinol. 2019, 10, 570. [Google Scholar] [CrossRef] [PubMed]
- Luo, M.; Santulli, G. Editorial: The link between obesity, type 2 diabetes, and mitochondria. Front. Endocrinol. 2023, 14, 1229935. [Google Scholar] [CrossRef] [PubMed]
- Westermann, B. Mitochondrial fusion and fission in cell life and death. Nat. Rev. Mol. Cell Biol. 2010, 11, 872–884. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Detmer, S.A.; Ewald, A.J.; Griffin, E.E.; Fraser, S.E.; Chan, D.C. Mitofusins Mfn1 and Mfn2 coordinately regulate mitochondrial fusion and are essential for embryonic development. J. Cell Biol. 2003, 160, 189–200. [Google Scholar] [CrossRef] [PubMed]
- Ishihara, N. Mitofusin 1 and 2 play distinct roles in mitochondrial fusion reactions via GTPase activity. J. Cell Sci. 2004, 117, 6535–6546. [Google Scholar] [CrossRef] [PubMed]
- Eura, Y.; Ishihara, N.; Yokota, S.; Mihara, K. Two mitofusin proteins, mammalian homologues of FZO, with distinct functions are both required for mitochondrial fusion. J. Biochem. 2003, 134, 333–344. [Google Scholar] [CrossRef]
- Kita, T.; Nishida, H.; Shibata, H.; Niimi, S.; Higuti, T.; Arakaki, N. Possible role of mitochondrial remodelling on cellular triacylglycerol accumulation. J. Biochem. 2009, 146, 787–796. [Google Scholar] [CrossRef]

| ND | HFD | HFD + AT | |
|---|---|---|---|
| Initial body weight (g) | 22.9 ± 0.3 | 23.2 ± 0.2 | 22.7 ± 0.4 |
| Final body weight (g) | 31.1 ± 0.5 c | 45.9 ± 1.0 a | 42.1 ± 1.1 b |
| Body-weight gain (g) | 8.2 ± 0.5 c | 22.8 ± 0.8 a | 19.7 ± 1.4 b |
| Food intake (g/day) | 2.4 ± 0.0 | 2.4 ± 0.1 | 2.4 ± 0.0 |
| Liver (g) | 1.08 ± 0.07 c | 1.51 ± 0.07 a | 1.03 ± 0.04 b |
| Pancreas (g) | 0.15 ± 0.01 c | 0.24 ± 0.01 a | 0.19 ± 0.02 b |
| White adipose tissue (WAT) (g) | |||
| Retroperitoneal AT | 0.57 ± 0.05 c | 1.47 ± 0.06 a | 1.29 ± 0.05 b |
| Gonadal AT | 1.14 ± 0.05 b | 2.65 ± 0.09 a | 2.70 ± 0.10 a |
| Inguinal AT | 1.11 ± 0.06 c | 3.10 ± 0.08 a | 2.63 ± 0.25 b |
| Total WAT | 2.82 ± 0.14 c | 7.21 ± 0.15 a | 6.62 ± 0.35 b |
| Brown adipose tissue (g) | 0.22 ± 0.02 b | 0.34 ± 0.03 a | 0.28 ± 0.04 b |
| Parameters | ND | HFD | HFD + AT |
|---|---|---|---|
| Glucose (mg/dL) | 93 ± 10 c | 163 ± 8 a | 128 ± 10 b |
| Insulin (ng/mL) | 1.1 ± 0.1 b | 3.2 ± 0.6 a | 2.7 ± 0.4 a |
| HOMA-IR | 5.4 ± 0.3 c | 31.6± 0.6 a | 20.3 ± 0.3 b |
| TG (mg/dL) | 93 ± 6 b | 161 ± 14 a | 99 ± 6 b |
| TC (mg/dL) | 179 ± 9 b | 235 ± 8 a | 183 ± 9 b |
| AST (IU/L) | 56 ± 13 b | 88 ± 10 a | 65 ± 6 b |
| ALT (IU/L) | 16 ± 8 b | 46 ± 7 a | 20 ± 2 b |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, M.; Paik, J.H.; Lee, H.; Kim, M.J.; Eum, S.M.; Kim, S.Y.; Choi, S.; Park, H.-Y.; Jeong, H.G.; Jeong, T.-S. Ancistrocladus tectorius Extract Inhibits Obesity by Promoting Thermogenesis and Mitochondrial Dynamics in High-Fat Diet-Fed Mice. Int. J. Mol. Sci. 2024, 25, 3743. https://doi.org/10.3390/ijms25073743
Kim M, Paik JH, Lee H, Kim MJ, Eum SM, Kim SY, Choi S, Park H-Y, Jeong HG, Jeong T-S. Ancistrocladus tectorius Extract Inhibits Obesity by Promoting Thermogenesis and Mitochondrial Dynamics in High-Fat Diet-Fed Mice. International Journal of Molecular Sciences. 2024; 25(7):3743. https://doi.org/10.3390/ijms25073743
Chicago/Turabian StyleKim, Minju, Jin Hyub Paik, Hwa Lee, Min Ji Kim, Sang Mi Eum, Soo Yong Kim, Sangho Choi, Ho-Yong Park, Hye Gwang Jeong, and Tae-Sook Jeong. 2024. "Ancistrocladus tectorius Extract Inhibits Obesity by Promoting Thermogenesis and Mitochondrial Dynamics in High-Fat Diet-Fed Mice" International Journal of Molecular Sciences 25, no. 7: 3743. https://doi.org/10.3390/ijms25073743
APA StyleKim, M., Paik, J. H., Lee, H., Kim, M. J., Eum, S. M., Kim, S. Y., Choi, S., Park, H.-Y., Jeong, H. G., & Jeong, T.-S. (2024). Ancistrocladus tectorius Extract Inhibits Obesity by Promoting Thermogenesis and Mitochondrial Dynamics in High-Fat Diet-Fed Mice. International Journal of Molecular Sciences, 25(7), 3743. https://doi.org/10.3390/ijms25073743






