Unlocking Precision Medicine: Liquid Biopsy Advancements in Renal Cancer Detection and Monitoring
Abstract
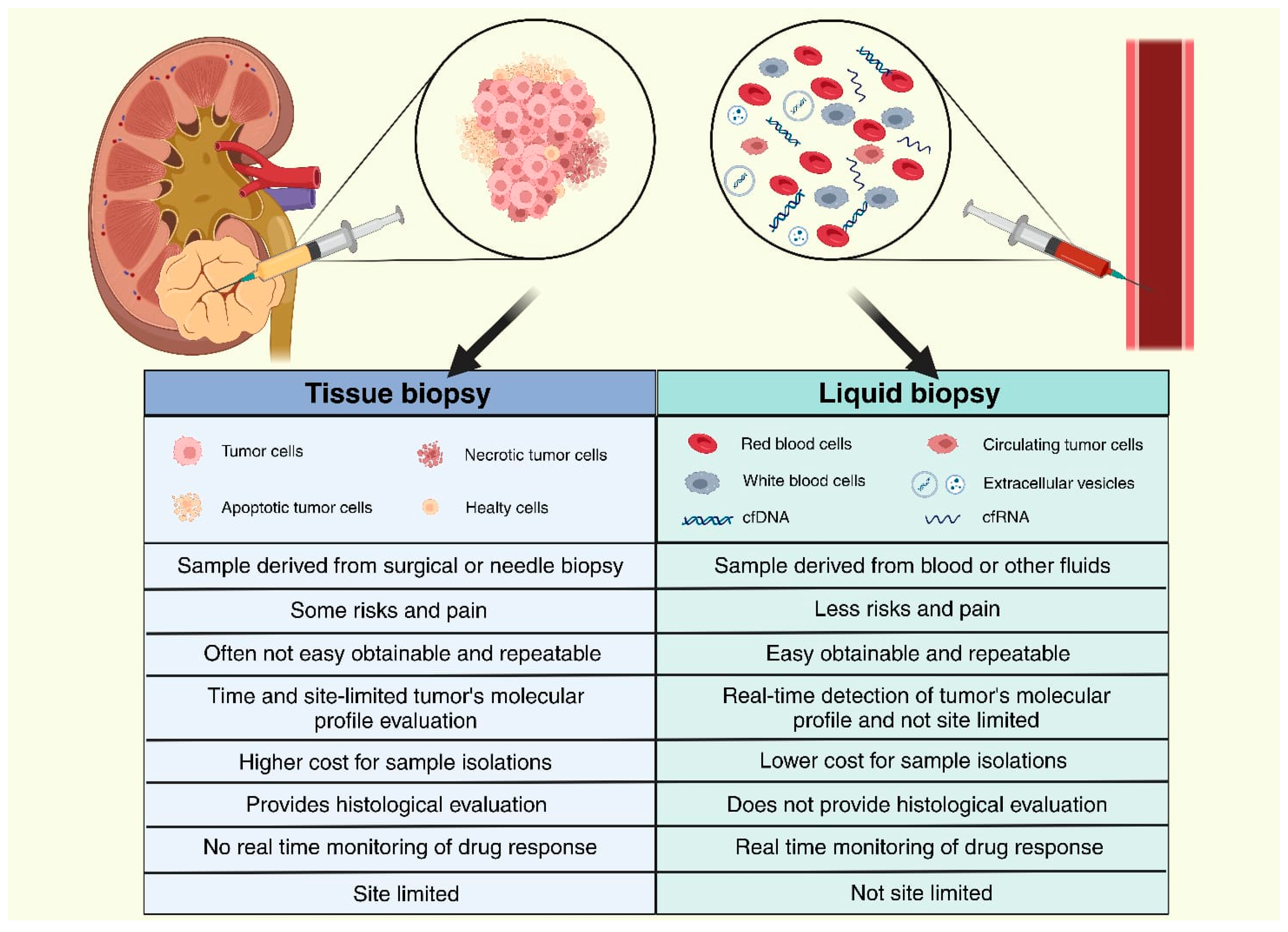
:1. Introduction
2. miRNAs
3. Exosomes
4. Circulating Tumor Cells
5. Circulating Tumor DNA
6. Conclusions
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Bahadoram, S.; Davoodi, M.; Hassanzadeh, S.; Bahadoram, M.; Barahman, M.; Mafakher, L. Renal Cell Carcinoma: An Overview of the Epidemiology, Diagnosis, and Treatment. G. Ital. Nefrol. 2022, 39, 1. [Google Scholar]
- Zieren, R.C.; Zondervan, P.J.; Pienta, K.J.; Bex, A.; de Reijke, T.M.; Bins, A.D. Diagnostic Liquid Biopsy Biomarkers in Renal Cell Cancer. Nat. Rev. Urol. 2023, 21, 133–157. [Google Scholar] [CrossRef]
- Ljungberg, B.; Albiges, L.; Abu-Ghanem, Y.; Bedke, J.; Capitanio, U.; Dabestani, S.; Fernández-Pello, S.; Giles, R.H.; Hofmann, F.; Hora, M.; et al. European Association of Urology Guidelines on Renal Cell Carcinoma: The 2022 Update. Eur. Urol. 2022, 82, 399–410. [Google Scholar] [CrossRef] [PubMed]
- Ferro, M.; Crocetto, F.; Barone, B.; Del Giudice, F.; Maggi, M.; Lucarelli, G.; Busetto, G.M.; Autorino, R.; Marchioni, M.; Cantiello, F.; et al. Artificial Intelligence and Radiomics in Evaluation of Kidney Lesions: A Comprehensive Literature Review. Ther. Adv. Urol. 2023, 15, 175628722311648. [Google Scholar] [CrossRef] [PubMed]
- Tataru, O.S.; Marchioni, M.; Crocetto, F.; Barone, B.; Lucarelli, G.; Del Giudice, F.; Busetto, G.M.; Veccia, A.; Lo Giudice, A.; Russo, G.I.; et al. Molecular Imaging Diagnosis of Renal Cancer Using 99mTc-Sestamibi SPECT/CT and Girentuximab PET-CT-Current Evidence and Future Development of Novel Techniques. Diagnostics 2023, 13, 593. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, J.J.; Purdue, M.P.; Signoretti, S.; Swanton, C.; Albiges, L.; Schmidinger, M.; Heng, D.Y.; Larkin, J.; Ficarra, V. Renal Cell Carcinoma. Nat. Rev. Dis. Primer 2017, 3, 17009. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Li, L.; Zheng, J.; Li, Z.; Li, S.; Wang, K.; Chen, X. Liquid Biopsy at the Frontier in Renal Cell Carcinoma: Recent Analysis of Techniques and Clinical Application. Mol. Cancer 2023, 22, 37. [Google Scholar] [CrossRef] [PubMed]
- Crocetto, F.; Barone, B.; Ferro, M.; Busetto, G.M.; La Civita, E.; Buonerba, C.; Di Lorenzo, G.; Terracciano, D.; Schalken, J.A. Liquid Biopsy in Bladder Cancer: State of the Art and Future Perspectives. Crit. Rev. Oncol. Hematol. 2022, 170, 103577. [Google Scholar] [CrossRef] [PubMed]
- Muljo, S.A.; Kanellopoulou, C.; Aravind, L. MicroRNA Targeting in Mammalian Genomes: Genes and Mechanisms. Wiley Interdiscip. Rev. Syst. Biol. Med. 2009, 2, 148–161. [Google Scholar] [CrossRef]
- Stavast, C.J.; Erkeland, S.J. The Non-Canonical Aspects of MicroRNAs: Many Roads to Gene Regulation. Cells 2019, 8, 1465. [Google Scholar] [CrossRef]
- Fabian, M.R.; Sonenberg, N.; Filipowicz, W. Regulation of mRNA Translation and Stability by microRNAs. Annu. Rev. Biochem. 2010, 79, 351–379. [Google Scholar] [CrossRef] [PubMed]
- Cardinali, B.; Tasso, R.; Piccioli, P.; Ciferri, M.C.; Quarto, R.; Del Mastro, L. Circulating miRNAs in Breast Cancer Diagnosis and Prognosis. Cancers 2022, 14, 2317. [Google Scholar] [CrossRef] [PubMed]
- Wani, J.A.; Majid, S.; Imtiyaz, Z.; Rehman, M.U.; Alsaffar, R.M.; Shah, N.N.; Alshehri, S.; Ghoneim, M.M.; Imam, S.S. MiRNAs in Lung Cancer: Diagnostic, Prognostic, and Therapeutic Potential. Diagnostics 2022, 12, 1610. [Google Scholar] [CrossRef] [PubMed]
- Mehrgou, A.; Ebadollahi, S.; Seidi, K.; Ayoubi-Joshaghani, M.H.; Yazdi, A.A.; Zare, P.; Jaymand, M.; Jahanban-Esfahlan, R. Roles of miRNAs in Colorectal Cancer: Therapeutic Implications and Clinical Opportunities. Adv. Pharm. Bull. 2021, 11, 233–247. [Google Scholar] [CrossRef] [PubMed]
- Gujrati, H.; Ha, S.; Wang, B.-D. Deregulated microRNAs Involved in Prostate Cancer Aggressiveness and Treatment Resistance Mechanisms. Cancers 2023, 15, 3140. [Google Scholar] [CrossRef] [PubMed]
- Dilsiz, N. Role of Exosomes and Exosomal microRNAs in Cancer. Future Sci. OA 2020, 6, FSO465. [Google Scholar] [CrossRef] [PubMed]
- Outeiro-Pinho, G.; Barros-Silva, D.; Aznar, E.; Sousa, A.-I.; Vieira-Coimbra, M.; Oliveira, J.; Gonçalves, C.S.; Costa, B.M.; Junker, K.; Henrique, R. MicroRNA-30a-5p Me: A Novel Diagnostic and Prognostic Biomarker for Clear Cell Renal Cell Carcinoma in Tissue and Urine Samples. J. Exp. Clin. Cancer Res. 2020, 39, 98. [Google Scholar] [CrossRef] [PubMed]
- Cochetti, G.; Cari, L.; Nocentini, G.; Maulà, V.; Suvieri, C.; Cagnani, R.; De Vermandois, J.A.R.; Mearini, E. Detection of Urinary miRNAs for Diagnosis of Clear Cell Renal Cell Carcinoma. Sci. Rep. 2020, 10, 21290. [Google Scholar] [CrossRef]
- Kalogirou, C.; Ellinger, J.; Kristiansen, G.; Hatzichristodoulou, G.; Kübler, H.; Kneitz, B.; Busch, J.; Fendler, A. Identification of miR-21-5p and miR-210-3p Serum Levels as Biomarkers for Patients with Papillary Renal Cell Carcinoma: A Multicenter Analysis. Transl. Androl. Urol. 2020, 9, 1314–1322. [Google Scholar] [CrossRef]
- Huang, G.; Li, X.; Chen, Z.; Wang, J.; Zhang, C.; Chen, X.; Peng, X.; Liu, K.; Zhao, L.; Lai, Y. A Three-microRNA Panel in Serum: Serving as a Potential Diagnostic Biomarker for Renal Cell Carcinoma. Pathol. Oncol. Res. 2020, 26, 2425–2434. [Google Scholar] [CrossRef]
- Luo, Y.; Chen, L.; Wang, G.; Xiao, Y.; Ju, L.; Wang, X. Identification of a three-miRNA signature as a novel potential prognostic biomarker in patients with clear cell renal cell carcinoma. J. Cell. Biochem. 2019, 120, 13751–13764. [Google Scholar] [CrossRef] [PubMed]
- Kowal, J.; Tkach, M.; Théry, C. Biogenesis and Secretion of Exosomes. Curr. Opin. Cell Biol. 2014, 29, 116–125. [Google Scholar] [CrossRef] [PubMed]
- Junker, K.; Heinzelmann, J.; Beckham, C.; Ochiya, T.; Jenster, G. Extracellular Vesicles and Their Role in Urologic Malignancies. Eur. Urol. 2016, 70, 323–331. [Google Scholar] [CrossRef] [PubMed]
- Merchant, M.L.; Rood, I.M.; Deegens, J.K.; Klein, J.B. Isolation and Characterization of Urinary Extracellular Vesicles: Implications for Biomarker Discovery. Nat. Rev. Nephrol. 2017, 13, 731–749. [Google Scholar] [CrossRef] [PubMed]
- Konoshenko, M.Y.; Lekchnov, E.A.; Vlassov, A.V.; Laktionov, P.P. Isolation of Extracellular Vesicles: General Methodologies and Latest Trends. BioMed Res. Int. 2018, 2018, 8545347. [Google Scholar] [CrossRef] [PubMed]
- Langevin, S.M.; Kuhnell, D.; Orr-Asman, M.A.; Biesiada, J.; Zhang, X.; Medvedovic, M.; Thomas, H.E. Balancing Yield, Purity and Practicality: A Modified Differential Ultracentrifugation Protocol for Efficient Isolation of Small Extracellular Vesicles from Human Serum. RNA Biol. 2019, 16, 5–12. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Wang, T.; Chen, C.; Wu, Z.; Bai, P.; Li, S.; Chen, B.; Liu, R.; Zhang, K.; Li, W.; et al. Serum Exosomal miR-210 as a Potential Biomarker for Clear Cell Renal Cell Carcinoma. J. Cell. Biochem. 2019, 120, 1492–1502. [Google Scholar] [CrossRef]
- Crentsil, V.C.; Liu, H.; Sellitti, D.F. Comparison of Exosomal microRNAs Secreted by 786-O Clear Cell Renal Carcinoma Cells and HK-2 Proximal Tubule-derived Cells in Culture Identifies microRNA-205 as a Potential Biomarker of Clear Cell Renal Carcinoma. Oncol. Lett. 2018, 16, 1285–1290. [Google Scholar] [CrossRef] [PubMed]
- Xiao, C.-T.; Lai, W.-J.; Zhu, W.-A.; Wang, H. MicroRNA Derived from Circulating Exosomes as Noninvasive Biomarkers for Diagnosing Renal Cell Carcinoma. OncoTargets Ther. 2020, 13, 10765–10774. [Google Scholar] [CrossRef]
- Ivanova, E.; Asadullina, D.; Gilyazova, G.; Rakhimov, R.; Izmailov, A.; Pavlov, V.; Khusnutdinova, E.; Gilyazova, I. Exosomal MicroRNA Levels Associated with Immune Checkpoint Inhibitor Therapy in Clear Cell Renal Cell Carcinoma. Biomedicines 2023, 11, 801. [Google Scholar] [CrossRef]
- Pereira-Veiga, T.; Schneegans, S.; Pantel, K.; Wikman, H. Circulating tumor cell-blood cell crosstalk: Biology and clinical relevance. Cell Rep. 2022, 40, 111298. [Google Scholar] [CrossRef] [PubMed]
- Deng, Z.; Wu, S.; Wang, Y.; Shi, D. Circulating tumor cell isolation for cancer diagnosis and prognosis. EBioMedicine 2022, 83, 104237. [Google Scholar] [CrossRef] [PubMed]
- Guan, Y.; Xu, F.; Tian, J.; Gao, K.; Wan, Z.; Wang, Y.; Gao, M.; Wang, Z.; Chong, T. The Prognostic Value of Circulating Tumour Cells (CTCs) and CTC White Blood Cell Clusters in Patients with Renal Cell Carcinoma. BMC Cancer 2021, 21, 826. [Google Scholar] [CrossRef] [PubMed]
- Vona, G.; Sabile, A.; Louha, M.; Sitruk, V.; Romana, S.P.; Schütze, K.; Capron, F.; Franco, D.; Pazzagli, M.; Vekemans, M.; et al. Isolation by Size of Epithelial Tumor Cells: A New Method for the Immunomorphological and Molecular Characterization of Circulating Tumor Cells. Am. J. Pathol. 2000, 156, 57–63. [Google Scholar] [CrossRef] [PubMed]
- van der Toom, E.E.; Verdone, J.E.; Gorin, M.A.; Pienta, K.J. Technical Challenges in the Isolation and Analysis of Circulating Tumor Cells. Oncotarget 2016, 7, 62754. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.-F.; Chen, Y.-C.; Fu, Y.-S.; Tsai, S.-W.; Wu, P.-K.; Chen, C.-M.; Chen, W.-M.; Wu, H.-T.H.; Lee, C.-H.; Chang, C.-L.; et al. Safety and Tolerability of Intra-Articular Injection of Adipose-Derived Mesenchymal Stem Cells GXCPC1 in 11 Subjects with Knee Osteoarthritis: A Nonrandomized Pilot Study without a Control Arm. Cell Transplant. 2024, 33, 09636897231221882. [Google Scholar] [CrossRef] [PubMed]
- Russo, G.I.; Musso, N.; Romano, A.; Caruso, G.; Petralia, S.; Lanzanò, L.; Broggi, G.; Camarda, M. The Role of Dielectrophoresis for Cancer Diagnosis and Prognosis. Cancers 2021, 14, 198. [Google Scholar] [CrossRef]
- Sperger, J.M.; Strotman, L.N.; Welsh, A.; Casavant, B.P.; Chalmers, Z.; Horn, S.; Heninger, E.; Thiede, S.M.; Tokar, J.; Gibbs, B.K.; et al. Integrated Analysis of Multiple Biomarkers from Circulating Tumor Cells Enabled by Exclusion-Based Analyte Isolation. Clin. Cancer Res. 2018, 23, 746–756. [Google Scholar] [CrossRef]
- Crocetto, F.; Cimmino, A.; Ferro, M.; Terracciano, D. Circulating Tumor Cells in Bladder Cancer: A New Horizon of Liquid Biopsy for Precision Medicine. J. Basic Clin. Physiol. Pharmacol. 2021, 33, 525–527. [Google Scholar] [CrossRef]
- Hu, F.; Mao, X.; Zhang, Y.; Zheng, X.; Gu, P.; Wang, H.; Zhang, X. Reliability of Using Circulating Tumor Cells for Detecting Epidermal Growth Factor Receptor Mutation Status in Advanced Non-Small-Cell Lung Cancer Patients: A Meta-Analysis and Systematic Review. OncoTargets Ther. 2018, 11, 1373–1384. [Google Scholar] [CrossRef]
- Tan, Y.; Wu, H. The Significant Prognostic Value of Circulating Tumor Cells in Colorectal Cancer: A Systematic Review and Meta-Analysis. Curr. Probl. Cancer 2018, 42, 95–106. [Google Scholar] [CrossRef] [PubMed]
- Crocetto, F.; Russo, G.; Di Zazzo, E.; Pisapia, P.; Mirto, B.F.; Palmieri, A.; Pepe, F.; Bellevicine, C.; Russo, A.; La Civita, E.; et al. Liquid Biopsy in Prostate Cancer Management—Current Challenges and Future Perspectives. Cancers 2022, 14, 3272. [Google Scholar] [CrossRef] [PubMed]
- Basso, U.; Facchinetti, A.; Rossi, E.; Maruzzo, M.; Conteduca, V.; Aieta, M.; Massari, F.; Fraccon, A.P.; Mucciarini, C.; Sava, T.; et al. Prognostic Role of Circulating Tumor Cells in Metastatic Renal Cell Carcinoma: A Large, Multicenter, Prospective Trial. Oncologist 2021, 26, 740–750. [Google Scholar] [CrossRef] [PubMed]
- Ferro, M.; Chiujdea, S.; Vartolomei, M.D.; Bove, P.; Porreca, A.; Busetto, G.M.; Del Giudice, F.; Antonelli, A.; Foschi, N.; Racioppi, M.; et al. Advanced Age Impacts Survival After Radical Nephroureterectomy for Upper Tract Urothelial Carcinoma. Clin. Genitourin. Cancer 2024, 22, 27–37. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.-L.; Zhang, P.; Li, H.-C.; Yang, X.-J.; Zhang, Y.-P.; Li, Z.-L.; Xue, L.; Xue, Y.-Q.; Li, H.-L.; Chen, Q.; et al. Dynamic Changes of Different Phenotypic and Genetic Circulating Tumor Cells as a Biomarker for Evaluating the Prognosis of RCC. Cancer Biol. Ther. 2019, 20, 505–512. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Liu, Y.; Xu, B.; Li, F.; Wang, Y.; Li, M.; Du, R.; Zhou, Y.; Salgia, M.; Yang, L.; et al. Circulating Tumor DNA Analysis of Metastatic Renal Cell Carcinoma. Mol. Clin. Oncol. 2021, 14, 16. [Google Scholar] [CrossRef] [PubMed]
- Sumiyoshi, T.; Yamasaki, T.; Takeda, M.; Mizuno, K.; Utsunomiya, N.; Sakamoto, H.; Nakamura, E.; Ogawa, O.; Akamatsu, S. Detection of von Hippel-Lindau Gene Mutation in Circulating Cell-Free DNA for Clear Cell Renal Cell Carcinoma. Cancer Sci. 2021, 112, 3363–3374. [Google Scholar] [CrossRef]
- Hoque, M.O.; Begum, S.; Topaloglu, O.; Jeronimo, C.; Mambo, E.; Westra, W.H.; Califano, J.A.; Sidransky, D. Quantitative Detection of Promoter Hypermethylation of Multiple Genes in the Tumor, Urine, and Serum DNA of Patients with Renal Cancer. Cancer Res. 2004, 64, 5511–5517. [Google Scholar] [CrossRef] [PubMed]
- Basu, A.; Kollipara, R.; Sudhaman, S.; Mahmood, T.; Pajak, N.; Carson, C.; Dutta, P.; Calhoun, M.; ElNaggar, A.; Liu, M.C.; et al. Longitudinal Detection of Circulating Tumor DNA in Patients with Advanced Renal Cell Carcinoma. J. Clin. Oncol. 2023, 41, 715. [Google Scholar] [CrossRef]
- Bettegowda, C.; Sausen, M.; Leary, R.J.; Kinde, I.; Wang, Y.; Agrawal, N.; Bartlett, B.R.; Wang, H.; Luber, B.; Alani, R.M.; et al. Detection of Circulating Tumor DNA in Early- and Late-Stage Human Malignancies. Sci. Transl. Med. 2014, 6, 224ra24. [Google Scholar] [CrossRef]
- Corrò, C.; Hejhal, T.; Poyet, C.; Sulser, T.; Hermanns, T.; Winder, T.; Prager, G.; Wild, P.J.; Frew, I.; Moch, H.; et al. Detecting Circulating Tumor DNA in Renal Cancer: An Open Challenge. Exp. Mol. Pathol. 2017, 102, 255–261. [Google Scholar] [CrossRef] [PubMed]
- Smith, C.G.; Moser, T.; Mouliere, F.; Field-Rayner, J.; Eldridge, M.; Riediger, A.L.; Chandrananda, D.; Heider, K.; Wan, J.C.M.; Warren, A.Y.; et al. Comprehensive Characterization of Cell-Free Tumor DNA in Plasma and Urine of Patients with Renal Tumors. Genome Med. 2020, 12, 23. [Google Scholar] [CrossRef] [PubMed]
- Wan, J.C.M.; Heider, K.; Gale, D.; Murphy, S.; Fisher, E.; Mouliere, F.; Ruiz-Valdepenas, A.; Santonja, A.; Morris, J.; Chandrananda, D.; et al. ctDNA Monitoring Using Patient-Specific Sequencing and Integration of Variant Reads. Sci. Transl. Med. 2020, 12, eaaz8084. [Google Scholar] [CrossRef] [PubMed]
- Lasseter, K.; Nassar, A.H.; Hamieh, L.; Berchuck, J.E.; Nuzzo, P.V.; Korthauer, K.; Shinagare, A.B.; Ogorek, B.; McKay, R.; Thorner, A.R.; et al. Plasma Cell-Free DNA Variant Analysis Compared with Methylated DNA Analysis in Renal Cell Carcinoma. Genet. Med. 2020, 22, 1366–1373. [Google Scholar] [CrossRef] [PubMed]
- Hahn, A.W.; Gill, D.M.; Maughan, B.; Agarwal, A.; Arjyal, L.; Gupta, S.; Streeter, J.; Bailey, E.; Pal, S.K.; Agarwal, N. Correlation of Genomic Alterations Assessed by Next-Generation Sequencing (NGS) of Tumor Tissue DNA and Circulating Tumor DNA (ctDNA) in Metastatic Renal Cell Carcinoma (mRCC): Potential Clinical Implications. Oncotarget 2017, 8, 33614–33620. [Google Scholar] [CrossRef] [PubMed]
- Pal, S.K.; Sonpavde, G.; Agarwal, N.; Vogelzang, N.J.; Srinivas, S.; Haas, N.B.; Signoretti, S.; McGregor, B.A.; Jones, J.; Lanman, R.B.; et al. Evolution of Circulating Tumor DNA Profile from First-Line to Subsequent Therapy in Metastatic Renal Cell Carcinoma. Eur. Urol. 2017, 72, 557–564. [Google Scholar] [CrossRef] [PubMed]
- Maia, M.C.; Bergerot, P.G.; Dizman, N.; Hsu, J.; Jones, J.; Lanman, R.B.; Banks, K.C.; Pal, S.K. Association of Circulating Tumor DNA (ctDNA) Detection in Metastatic Renal Cell Carcinoma (mRCC) with Tumor Burden. Kidney Cancer 2017, 1, 65–70. [Google Scholar] [CrossRef] [PubMed]
- Bacon, J.V.W.; Annala, M.; Soleimani, M.; Lavoie, J.-M.; So, A.; Gleave, M.E.; Fazli, L.; Wang, G.; Chi, K.N.; Kollmannsberger, C.K.; et al. Plasma Circulating Tumor DNA and Clonal Hematopoiesis in Metastatic Renal Cell Carcinoma. Clin. Genitourin. Cancer 2020, 18, 322–331.e2. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, Y.; Uemura, M.; Fujita, M.; Maejima, K.; Koh, Y.; Matsushita, M.; Nakano, K.; Hayashi, Y.; Wang, C.; Ishizuya, Y.; et al. Clinical Significance of the Mutational Landscape and Fragmentation of Circulating Tumor DNA in Renal Cell Carcinoma. Cancer Sci. 2019, 110, 617–628. [Google Scholar] [CrossRef]
- Jung, M.; Ellinger, J.; Gevensleben, H.; Syring, I.; Lüders, C.; de Vos, L.; Pützer, S.; Bootz, F.; Landsberg, J.; Kristiansen, G.; et al. Cell-Free SHOX2 DNA Methylation in Blood as a Molecular Staging Parameter for Risk Stratification in Renal Cell Carcinoma Patients: A Prospective Observational Cohort Study. Clin. Chem. 2019, 65, 559–568. [Google Scholar] [CrossRef]
- Lin, Y.-L.; Wang, Y.-P.; Li, H.-Z.; Zhan, X. Aberrant Promoter Methylation of PCDH17 (Protocadherin 17) in Serum and Its Clinical Significance in Renal Cell Carcinoma. Med. Sci. Monit. Int. Med. J. Exp. Clin. Res. 2017, 23, 3318–3323. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.J.; Kang, Y.; Kim, J.S.; Sung, H.H.; Jeon, H.G.; Jeong, B.C.; Seo, S.I.; Jeon, S.S.; Lee, H.M.; Park, D.; et al. Potential of Circulating Tumor DNA as a Predictor of Therapeutic Responses to Immune Checkpoint Blockades in Metastatic Renal Cell Carcinoma. Sci. Rep. 2021, 11, 5600. [Google Scholar] [CrossRef] [PubMed]
- Park, J.S.; Kim, H.; Jang, W.S.; Kim, J.; Ham, W.S.; Lee, S.-T. ctDNA Predicts Clinical T1a to Pathological T3a Upstaging after Partial Nephrectomy. Cancer Sci. 2024. [Google Scholar] [CrossRef] [PubMed]
- Iisager, L.; Ahrenfeldt, J.; Keller, A.K.; Nielsen, T.K.; Fristrup, N.; Lyskjær, I. KIDNEY-PAGER: Analysis of Circulating Tumor DNA as a Biomarker in Renal Cancer—An Observational Trial. Study Protocol. Acta Oncol. Stockh. Swed. 2024, 63, 51–55. [Google Scholar] [CrossRef] [PubMed]
| Biomarker | Region | Year | Detected Abnormality | Cohorts | Practice in Clinical | Results | Ref. |
|---|---|---|---|---|---|---|---|
| Urinary miRNAs | Italy | 2020 | miR-122, miR-1271, and miR-15b overexpression | 13 ccRCC-diagnosed patients and 14 healthy controls | Diagnosis | miR-122, miR-1271, and miR-15b candidate as potential biomarkers for ccRCC diagnosis | [18] |
| Serum miRNAs | Germany | 2020 | miR-21-5p and miR-210-3p overexpression | 34 pRCC type 1, 33 pRCC type 2 and 33 control subjects | Diagnosis | miR-21-5p and miR-210-3p candidates as potential biomarkers for pRCC diagnosis. Impossibility to make a differential diagnosis between pRCC type 1 and 2 based on miRNA expression | [19] |
| Serum miRNAs | China | 2020 | miR-224-5p, miR-34b-3p and miR-182-5p overexpression | 146 RCC patients and 150 healthy controls | Diagnosis | A panel formed by miR-224-5p, miR-34b-3p and miR-182-5p candidate as the most reliable biomarker for RCC non-invasive diagnosis | [20] |
| miRNAs expressed by ccRCC and paracancerous tissue | China | 2019 | miR130b, miR-18a, and miR-223 overexpression | 544 ccRCC tumor specimens and 71 adjacent nontumor renal specimens | Prognosis prediction | Multivariate Cox regression analysis and subgroup analysis showed that the three-miRNA signature was an independent prognostic factor that could be used to predict the prognosis of ccRCC patients by dosing the three miRNAs in biofluid | [21] |
| Exosomal miRNAs | China | 2018 | miR-210, miR-224, miR-452, miR-155, miR-21, and miR-34a expression | 45 patients diagnosed with ccRCC | Diagnosis, prognosis and recurrence prediction | Serum exosomal miR-210 may be a potential biomarker for the diagnosis, prognosis, and prediction of the recurrence of ccRCC, especially for metastatic ccRCC | [27] |
| Exosomal miRNAs | USA | 2018 | miR-210, MiR-34a, miR-155-5p and miR-150-5p overexpression | In vitro (786-O cell line (derived from ccRCC) as an in vitro ccRCC tumor model and the human renal proximal tubule cell line HK-2 as a normal renal tissue control) | Diagnosis | Exosomal content of miR-205 and possibly miR-150 were proportional to their respective contents in the cells that secreted them, serving as ccRCC biomarkers | [28] |
| Exosomal miRNAs | China | 2020 | hsa-mir-92a-1-5p, hsa-mir-149-3p and hsa-mir-424-3p abnormal expression | 22 RCC patients and 16 controls | Diagnosis | hsa-mir-92a-1-5p, hsa-mir-149-3p and hsa-mir-424-3p can be potentially used as plasmatic biomarkers for RCC diagnosis | [29] |
| Exosomal miRNAs | Russia | 2023 | miRNAs-144, -146a, -149, -126, and -155 expression | 35 RCC patients treated with immune checkpoint inhibitors | Prognosis prediction | miRNA-146a and miRNA-126 combined expression showed a potential role as biomarkers, even in assessing therapy effectiveness, in RCC | [30] |
| Exosomal miRNAs | Italy | 2021 | correlation between CTC counts and progression-free survival (PFS) in patients with metastatic RCC treated with an antiangiogenic tyrosine kinase inhibitor as a first-line regimen | 195 patients treated at the baseline with pazopanib or sunitinib | Prognosis prediction | the baseline number of isolated CTCs could serve as a predictive factor for prognosis in patients diagnosed with metastatic RCC | [43] |
| Exosomal miRNAs | China | 2018 | the relationship of dynamic changes of CTCs and Beclin-1 expression of CTCs with renal cell carcinoma (RCC) prognosis | 69 patients diagnosed with RCC and treated with surgery [metastasis-free group (n = 58) and metastatic group (n = 11)] | Prognosis prediction | recurrence or metastasis of RCC was related to the variation trend of CTCs, especially mesenchymal CTCs and Beclin1-positive CTCs | [45] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Crocetto, F.; Falcone, A.; Mirto, B.F.; Sicignano, E.; Pagano, G.; Dinacci, F.; Varriale, D.; Machiella, F.; Giampaglia, G.; Calogero, A.; et al. Unlocking Precision Medicine: Liquid Biopsy Advancements in Renal Cancer Detection and Monitoring. Int. J. Mol. Sci. 2024, 25, 3867. https://doi.org/10.3390/ijms25073867
Crocetto F, Falcone A, Mirto BF, Sicignano E, Pagano G, Dinacci F, Varriale D, Machiella F, Giampaglia G, Calogero A, et al. Unlocking Precision Medicine: Liquid Biopsy Advancements in Renal Cancer Detection and Monitoring. International Journal of Molecular Sciences. 2024; 25(7):3867. https://doi.org/10.3390/ijms25073867
Chicago/Turabian StyleCrocetto, Felice, Alfonso Falcone, Benito Fabio Mirto, Enrico Sicignano, Giovanni Pagano, Fabrizio Dinacci, Domenico Varriale, Fabio Machiella, Gaetano Giampaglia, Armando Calogero, and et al. 2024. "Unlocking Precision Medicine: Liquid Biopsy Advancements in Renal Cancer Detection and Monitoring" International Journal of Molecular Sciences 25, no. 7: 3867. https://doi.org/10.3390/ijms25073867












