Abstract
Allergies affect approximately 10–30% of people worldwide, with an increasing number of cases each year; however, the underlying mechanisms are still poorly understood. In recent years, extracellular vesicles (EVs) have been suggested to play a role in allergic sensitization and skew to a T helper type 2 (Th2) response. The aim of this review is to highlight the existing evidence of EV involvement in allergies. A total of 22 studies were reviewed; 12 studies showed EVs can influence a Th2 response, while 10 studies found EVs promoted a Th1 or Treg response. EVs can drive allergic sensitization through up-regulation of pro-Th2 cytokines, such as IL-4 and IL-13. In addition, EVs from MRSA can induce IgE hypersensitivity in mice towards MRSA. On the other hand, EVs can induce tolerance in the immune system; for example, pre-exposing OVA-loaded EVs prevented OVA sensitization in mice. The current literature thus suggests that EVs play an essential role in allergy. Further research utilizing human in vitro models and clinical studies is needed to give a reliable account of the role of EVs in allergy.
1. Introduction
Atopic allergies, also known as immediate or type I hypersensitivities, affect between 10% and 30% of the population [1]. Allergies affect people’s quality of life, impacting their work and personal lives; in addition, the treatments for allergies bear a heavy burden on healthcare systems across the globe. It has been reported that it costs the NHS over GBP one billion per annum [2]. However, the underlying mechanism of how an allergy develops is still poorly understood. The most frequent clinical manifestations of allergies are asthma and allergic rhinitis, but other atopic diseases include urticaria and anaphylactic shock.
Several factors can impact whether someone develops atopic allergy, such as environmental exposure, genetic predispositions or lifestyle factors, such as a Westernized diet, low vitamin D intake, antibiotic use, and the route of birth; all these form an intricate balance that determines the development of type 1 hypersensitivity [3].
Allergic sensitization, the first phase of atopic allergy development, is central to the initiation of atopic diseases. Typically, proteins within allergen sources initiate allergic sensitization through recognition by dendritic cells (DCs) and presentation to naïve T cells, which results in a cascade of reactions shifting the response in a Th2-dominant manner. Dendritic cells can be classified into four subsets—human plasmacytoid DC (pDC), conventional DC1 (cDC1), conventional DC2 (cDC2) and monocyte-derived DC (mo-DC) [4]. Subsequently, allergen-specific immunoglobulin E (IgE) is bound on mast cells through the FcεRI receptor. Conversely, naïve T cells can differentiate into Th1 or Treg cells, resulting in immune tolerance, which normally happens in healthy individuals. Recent research has suggested other components can drive allergic sensitization, such as lipids [5,6], carbohydrates [7], and most recently, EVs, which can, in fact, also carry proteins, lipids and carbohydrates [8,9].
EVs are small lipid-bilayer particles that are released naturally from cells within the body and by other sources, such as bacteria. They are key inter-cellular communicators, and their size can range from 20 nm to 10 µm [10]. EVs can transport proteins, nucleic acids (deoxyribonucleic acid (DNA), messenger ribonucleic acid (mRNA), and non-coding RNA), lipids, metabolites and specific cell surface markers depending on the cells from which they originate [11]. They influence recipient cells due to their capacity to transfer this ‘cargo’ [12]. When discriminating between EVs, their origin is considered; if they develop internally and are then released through the plasma membrane, they are called exosomes, where their size is usually smaller than 200 nm. If they bud off from the plasma membrane, they are called ectosomes, where their size is 100 nm to 10 µm. The recommendations for carrying out research into EVs are formulated in Minimal Information For Studies of Extracellular Vesicles (MISEV) 2023 [13].
Importantly, EVs and their contents represent the cell they come from, as well as the current condition of the cell [14]. Due to their ability to reflect the condition of their cellular origin, they are often used as biomarkers for diseases; however, studies have also examined their involvement in the pathogenesis of diseases [14]. Indeed, recent studies have been trying to understand the role of EVs in allergy. For example, in a study done by Fang et al. in 2020, it was found that by utilizing human mesenchymal stem cell (MSC) EVs, it was possible to alleviate inflammation in the lungs by restricting the infiltration of inflammatory cells and epithelial goblet cells by changing recruitment and polarization of the alveolar macrophages [15]. In asthma, EVs can be used as a diagnostic tool between healthy and asthmatic patients, and EVs can promote inflammation and airway remodeling, suggesting EVs also play a role in the pathogenesis of asthma, which has been systematically reviewed in a study done by Sangaphunchai et al., 2020 [16].
In this systematic review, the focus is on the role of EVs in allergic sensitization. The role of EVs generated from the host (termed Host-derived EVs) and from exogenous sources, such as bacteria (termed exogenous EVs), is discussed, and whether these EVs induce sensitization or tolerance.
2. Materials and Methods
2.1. Search Strategy
This review was conducted with the implementation of the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. The search strategy was constructed around two key terms: ‘extracellular vesicles’ and ‘allergy’. In this review, two databases were searched to find relevant papers: PubMed and Embase, from their inception to 31 December 2023, using the following search terms:
PubMed—(exosomes or exosome* or extracellular vesicle* or ectosome or microparticle or microvesicle or shedding vesicle*) AND (allergic sens* or allergic sensitization or type 1 hypersensitivity or allergy)
EMBASE—(exosomes or exosome* or extracellular vesicle* or ectosome or microparticle or microvesicle or shedding vesicle*) AND (allergic sens* or allergic sensitization or type 1 hypersensitivity or allergy).mp. [mp = title, abstract, heading word, drug trade name, original title, device manufacturer, drug manufacturer, device trade name, keyword, floating subheading word, candidate term word].
2.2. Selection of Studies
This systematic review aimed to examine the evidence on the role and function of EVs in allergic sensitization, with the hypothesis that EVs from various origins contribute to the development of allergy. For this review, other reviews, books, editorials, and conference proceedings were excluded, language was restricted to English, and there was no restriction on the date of publication. Publications were screened for their relevance against pre-defined inclusion and exclusion criteria (Table 1), and a random subset (10%) of these studies was reviewed independently by a second reviewer.

Table 1.
Selection criteria for papers included in the systematic review.
2.3. Study Quality Assessment
To assess the robustness of the studies included in this systematic review and to highlight the current research methodology in this field, a scoring system was developed, with specific criteria set in Table 2. Each study’s score was assessed by the criteria: model, robustness of model, sample size, EV isolation, EV characterization, and sensitization method. Scores from the studies were summed and divided by the highest possible total score to calculate the study bias score. Studies that received lower scores are not less reliable but are missing key methods in researching EVs, such as ensuring pure and intact EV populations through isolation.

Table 2.
Scoring system to assess the robustness of included papers.
3. Results
3.1. PRISMA Search and Selection
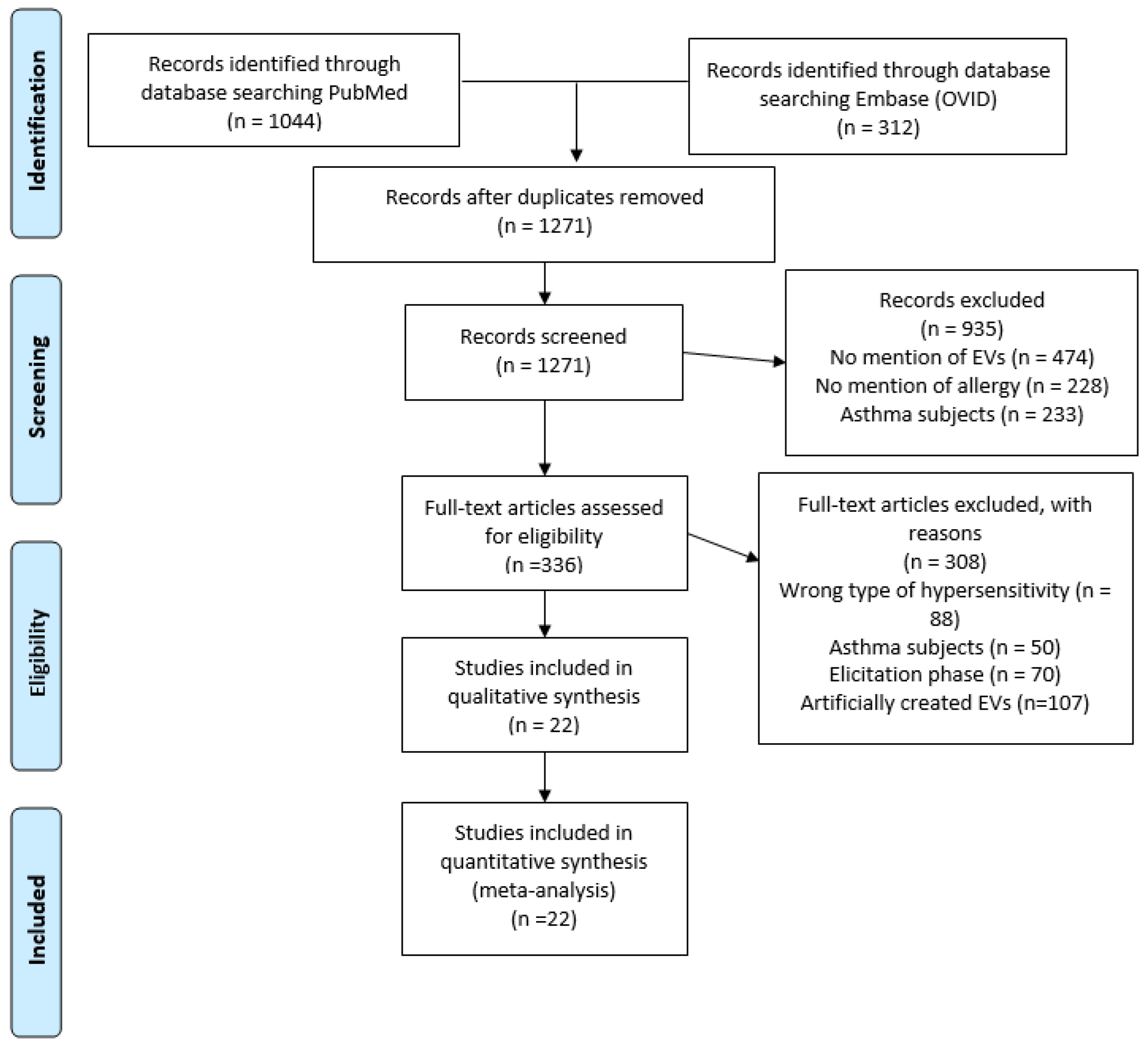
A PRISMA diagram was used to track the search details for this systematic review (Figure 1). The search resulted in 1356 papers: 1044 from PubMed and 312 from EMBASE. After the removal of duplicated publications, 1271 titles and abstracts of papers were examined for relevance. After the title and abstract review, 336 papers were kept as potential interest for the review. After selecting papers using the inclusion and exclusion criteria, 22 studies were included in the systematic review, which were relevant to understanding the role of EVs in allergic sensitization.
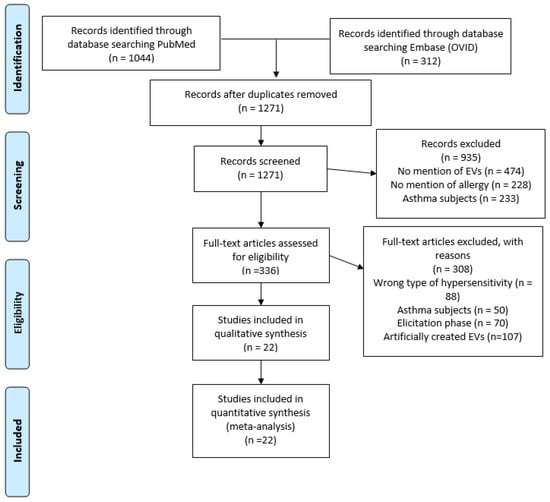
Figure 1.
PRISMA diagram of article search and selection. A PRISMA diagram detailing the process of searching and selecting EV studies in allergic sensitization, using PubMed and Embase (OVID) from 1974–2023.
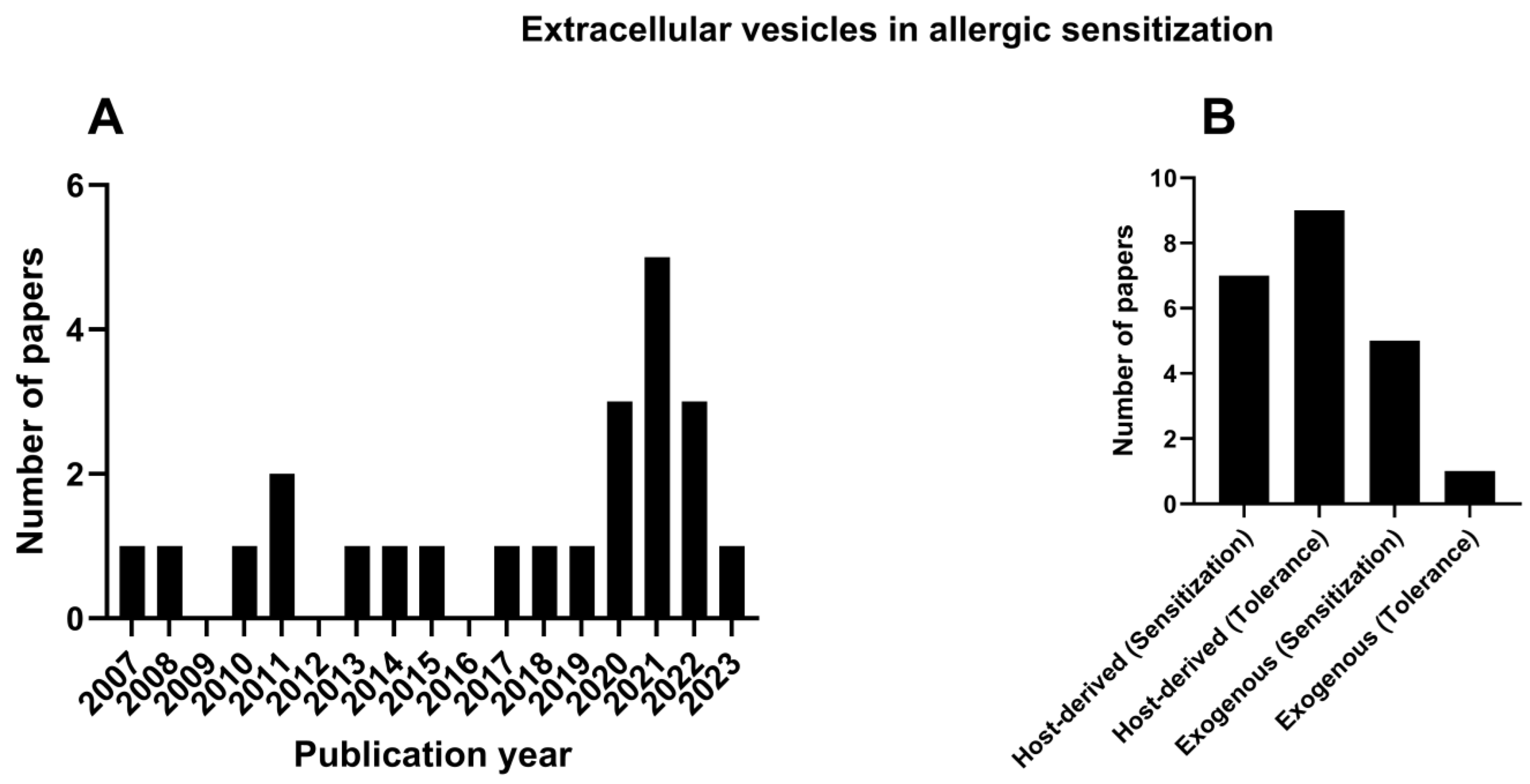
The first publication relevant to EVs in allergic sensitization was published in 2007; however, 60% of the papers were published in the last 8 years (Figure 2A).
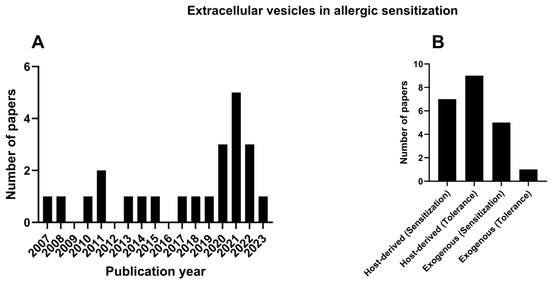
Figure 2.
Publication on EVs in allergic sensitization through the years (A). Publications concerning the host-derived EVs—sensitization and tolerance, and exogenous source EVs (B).
The results highlight two main types of EVs based on their source of origin: EVs that were produced from the host (host-derived) or from external sources (exogenous), such as bacteria (Figure 2B). Thus, the results will be discussed in terms of host-derived or exogenous EVs and their potential to either promote allergic sensitization or the tolerance of allergens.
3.2. Host-Derived Extracellular Vesicles
3.2.1. Promotion of Allergic Sensitization
In the allergic sensitization process, naïve T cells differentiate into Th2 cells, resulting in interleukin 4 (IL-4) production and IgE deposition on mast cells, but what drives this shift to Th2 is poorly understood. Publications suggesting that host-derived EVs can promote Th2 differentiation and, thus, the sensitizations were identified and summarized in Table 3.

Table 3.
Studies involving allergic sensitization and host-derived EVs.
Seven studies examined the role of EVs in promoting Th2-like cytokine production and inducing the differentiation to Th2 cells [17,18,19,20,21,22,23]. B-cell-derived EVs had allergen peptide-loaded major histocompatibility complex (MHC) molecules on their surface and were capable of stimulating T cells to proliferate and produce IL-5 and IL-13 [17]. A different study observed that plasma EVs have antigen-presenting capabilities and induced Th2 cell differentiation and increased levels of IL-13 [18]. One in vitro study found that thymic stromal lymphopoietin (TLSP)-activated DC EVs induced a higher number of Th2 cells by elevating IL-4 concentration and decreasing interferon-gamma (IFN-γ) concentration, thus reducing Th1 cells [19]. Investigating the effect of T follicular helper cell (Tfh) EVs found that these EVs could regulate DC maturation through miR-142-5p/CDK5/STAT3 pathway; however, most importantly, Tfh EVs promoted Th2 responses in naïve mice through elevated levels of IL-13 and IgE, while simultaneously decreasing IFN-γ and IL-17 levels [20]. Another study showed that DC-derived EVs could carry Fel d 1 and in co-culture with peripheral blood mononuclear cells (PBMCs) induced higher levels of IL-4 production [21]. By researching lncRNA Nuclear Paraspeckle Assembly Transcript 1 (NEAT1) it was found that it was responsible for the promotion of allergic rhinitis (AR) pathogenesis via EVs, which interacted with human nasal epithelial cells (HNECs) and induced IL-13 production and apoptosis, which can lead to damage to the epithelium and increase the likelihood of allergen exposure and development of allergic sensitization [22]. Epithelium-derived EVs carried long non-coding RNA growth arrest-specific 5 (GAS5), which can suppress Th1 differentiation by downregulating the enhancer of zeste 2 polycomb repressive complex 2 subunit (EZH2) and T-bet, where IFN-γ concentration and Th1 responses were decreased, while also stimulating IL-4 production and Th2 differentiation [23].
Of the seven papers describing host-derived EVs driving allergic sensitization, all studies used human samples for the isolation of EVs and applied them in an in vitro model [17,18,19,20,21,22,23], whereas one study also used mice and an in vivo model [20]. Regarding the EVs isolation methodology, six studies used serial ultracentrifugation [17,18,19,20,21,23], while one study used the ExoQuick precipitation kit, which utilized size exclusion chromatography (SEC) [22]. The identification process was different across the studies; however, six studies used TEM to detect the presence of EVs [18,19,20,21,22,23], and four studies used Western blotting to confirm the presence of tetraspanins [18,19,22,23]. Three papers used flow cytometry to detect tetraspanins on the EVs [17,18,21]. Two studies used NTA to measure the size of EVs [18,21]. The papers were scored based on their quality using specific criteria created for this systematic review (Table 2). Overall, the identification of EVs was sufficient in three papers [18,19,21] as two or more methods were used, while in the other papers [17,20,22,23] where only one identification method was used (TEM). Two papers received high scores of 63% [18,21] due to their thorough characterization of EVs, while three studies had the lowest scores of 52% [17,19,23] because of the limited work done in EV isolation and characterization. A full assessment of the papers is presented in Table 4.

Table 4.
Scoring assessment of papers involving host-derived EVs and allergic sensitization.
Overall, all seven studies show the potential of EVs influencing the immune system to shift towards a Th2 response by increasing the differentiation towards Th2 cells and Th2-like cytokine production, which could potentially reveal how EVs influence allergic sensitization. By understanding allergy development, it may be possible to prevent its occurrence.
3.2.2. Promotion of Immune Tolerance
Naïve CD4 T-cells can differentiate into Th1, Th2 or Treg cells; if it is a Th1- or Treg-dominant response, immune tolerance is promoted. Papers suggesting that EVs can drive immune tolerance are compiled in Table 5.

Table 5.
Studies in host-derived EVs and allergic tolerance.
Nine studies suggested that host-derived EVs were capable of suppressing Th2 responses, resulting in immune tolerance [24,25,26,27,28,29,30,31,32]. Four of these studies researched the influence of EVs on food allergy [24,25,26,27], one of which found intestinal epithelial-cell-derived integrin ɑβ6 and OVA-carrying EVs interacting with T cells could induce T cell differentiation to Treg cells, through the increased production of TGF-β [24]. Furthermore, EVs from IL-2 and OVA-stimulated DCs, with MHC class II molecules, interacting with OVA-specific CD4+ T cells induced differentiation into Tregs [25]. Another study relating to food allergy showed that EVs carry the allergen-MHC class 2 complexes and IL-10 and could induce Tr1 differentiation in healthy CD4+ T cells [26]. Another study found that MSC-derived EVs containing OVA pre-exposure in mice resulted in tolerance to OVA, with decreased levels of IgE and IL-4, along with elevated TGF-β levels [27].
Three studies examined the role of EVs in allergic rhinitis [28,29,30]. In these studies, it was found that when using EVs, it was possible to inhibit the Th2 cell responses and induce Treg differentiation by limiting the production of IgE and increased expression of TGF-β and IgG2a [29]. Another study showed that MSC-derived EVs also suppressed the Th2 response by reducing the production of IL-4, IL-9, and IL-13 via IL-10 production, thus increasing the levels of Tregs in the system [28]. The last allergic rhinitis study showed that human mesenchymal stem cells (HMSC)-EVs co-cultured with CD4+ T cells inhibited the Th2 differentiation by regulating the miR-146a-5p/SERPINB2 pathway [30].
Two studies looked at the role of allergens and EVs in immune tolerance; one of the studies looked at the differences between microvesicles and exosomes and their capacity to influence the immune responses, where it was found that exosomes induced a stronger response, which generated a Th1-like response by inducing the production of allergen-specific IgG abs and expansion of OVA-specific CD8+ T cell population [31]. The other study observed the potential of ‘bystander’ tolerance of allergens by EVs, where it was found that EVs inhibited the immune responses to the specific allergen in addition to another unrelated allergen by limiting the production of IL-5 and IL-13 and reducing the inflammatory responses in the lung [32].
Of the nine papers describing host-derived EVs in immune tolerance, seven studies used mice as their source of EVs [24,25,26,29,31,32], and two studies used human samples and human cell lines as their EV source [28,30]. EV isolation protocols were similar across the papers, where seven of the studies used some form of serial ultracentrifugation [24,25,26,29,30,31,32], one study used anion exchange chromatography [28], and one study utilized an Exospin isolation kit, which uses SEC [27]. Various methods were used to confirm the presence of isolated EVs; four studies used the Bradford assay [24,25,26,28], five studies used TEM [24,28,29,30,31], and one used SEM [27]. Two studies used Western blotting to detect the presence of tetraspanins [28,30], and one study used immunophenotyping [27]. Overall, the identification and characterization of EVs in these studies were lacking. The highest bias score was 63%, but this study lacked the use of multiple models [28], whilst the lowest score achieved was 44%, resulting from the limited use of models and inferior isolation technique [29]. Six out of nine studies scored over 50% [25,26,28,30,31,32]. The overall scores were compiled into a table below (Table 6).

Table 6.
Scoring assessment of papers in host-derived EVs and allergic tolerance.
Overall, these nine studies showed that EVs were capable of suppressing Th2 immune response and skewing towards Th1 responses, as well as the induction of Treg cells, which in turn reduce the inflammatory cytokine and IgE production in food allergy and allergic rhinitis. Thus, these results show the potential of using EVs as a vehicle for alleviating and/or preventing allergies.
3.3. Exogenous Extracellular Vesicles
3.3.1. Promotion of Allergic Sensitization via the Skin Epithelium
In allergic sensitization, the first ‘defense’ against allergens is epithelial cells, either as skin cells, lung cells or intestinal cells. EVs generated from exogenous sources, such as bacteria, must thus cross this first barrier in order to interact with the immune system and influence the allergic sensitization process. Publications suggesting that exogenous EVs can also promote sensitization resulting in allergies, were identified (Table 7).

Table 7.
Studies involving external EVs and allergic sensitization.
Five studies showed that EVs generated by bacteria could damage the skin barrier, leading to easier colonization of the skin, which in turn may influence allergic sensitization and AD [33,34,35,36,37]. Five studies were experimenting with S. aureus EVs and their influence on the induction of atopic dermatitis-like skin inflammation [33,34,35,36,37]; however, one paper showed that Lactobacillus plantarum-derived EVs could prevent damage to the skin from S. aureus EVs [36]. In one of these studies, the toxin α-Hemolysin produced by S. aureus was more potent in EV form and could induce IL-6 production associated with Th17 response, which caused skin barrier disruption and AD-like skin inflammation [33]. After investigating the potential effects of methicillin-resistant Staphylococcus aureus (MRSA) EVs in mice, it was found that the EVs act as immunostimulant that induces inflammatory responses, producing cytokines such as IL-4, IL-5 and IL-6 and triggering IgE-mediated hypersensitivity after MRSA infection [34].
The isolation protocols for the five studies were similar, where filtration and serial ultracentrifugation were used [33,34,35,36,37], which is not efficient enough to provide pure and intact EVs [38]. One study used filtration and an ExoQuickTC kit that utilized SEC [37]. The identification methods used in most studies were also not comprehensive, where 3 studies only used one method of identification for characterizing their EVs [35,36,37], while only one study utilized novel techniques on top of the standard methods [34]. Papers in external sources scored higher than the other sections in this systematic review. The highest scoring paper was 79% [34], due to the use of a novel identification method utilizing electrolytes in NP150 nanopore membranes on top of other conventional techniques such as Western blots, SDS-page gels and Bradford protein assays. The lowest scoring paper was also found in this section, with 38% [35], because of the poor isolation and characterization of their EVs, plus the use of only a murine in vivo model. The full assessment of papers can be found in Table 8.

Table 8.
Scoring assessment of studies in exogenous EVs and allergic sensitization.
Overall, in these five studies, bacterial EVs were shown to damage the epithelial cell barrier, which can lead to increased susceptibility to developing an allergic reaction.
3.3.2. Promotion of Immune Tolerance
It has been shown that immune cells can recognize and interact with these exogenous EVs to alter their responses to stimuli [39]. Only one study showed that exogenous EVs promote immune tolerance (Table 9). Observing that EVs produced by Gram-negative bacteria in indoor dust can induce neutrophilic pulmonary inflammation and increased production of IFN-γ and IL-17 associated with both Th1 and Th17 cell response. Furthermore, dust EVs from Gram-negative bacteria induced tumor necrosis factor alpha (TNF-α) and IL-6 production in macrophages [40].

Table 9.
Studies involving external EVs and allergic tolerance.
The one study showing that an exogenous source of EVs can tolerize the immune system used a mice model and a human in vitro model [40]. The isolation method utilized in this paper was ultracentrifugation [40]. The paper used NTA to size the EVs and used TEM [40] to confirm the morphology of the EVs. This paper scored well at a 58% bias score, as it utilized multiple model systems and fully described them [40]. A full breakdown of the scoring can be found in Table 10.

Table 10.
Assessment of studies involving exogenous EVs and allergic tolerance.
In summary, this paper shows that exogenous-sourced EVs were capable of shifting the immune response towards a Th1-like response.
4. Discussion
Allergies are a worldwide issue with an increasing number of cases in the population [41], yet the mechanisms behind the development of an allergy are still poorly understood. However, recently, there has been a focus on research on EVs and their role in allergies and their development. In this systematic review, we highlight the increasing evidence that EVs can promote allergic sensitization; conversely, there is also much evidence for their role in promoting tolerance to allergens.
Of the 22 studies relevant to EVs in allergic sensitization, 64% of studies [17,18,19,20,21,22,23,33,34,35,36,37,40] showed evidence of EVs enhancing the allergic sensitization process and shifting towards Th2-type responses, compared to 36%, which reported EVs could prevent allergic sensitization and shift the immune response towards a Th1 and Treg-like immune tolerance [24,25,26,28,29,30,31,32]. In terms of sensitization, this systematic review highlights that EVs promote the differentiation of naïve T cells into Th2 cells by interacting with immune cells to influence cytokine production, such as increasing IL-4, IL-13 and IL-5 concentration and decreasing IFN-γ levels [17,18,19,20,21,22,23]. In terms of tolerance, we highlight that EVs can induce immune tolerance by stimulating Th1 and Treg differentiation by increasing concentrations of TGF-β, IL-2 and IL-10 cytokines while suppressing the production of IL-4, IL-9, and IL-13 cytokines, leading to lower levels of Th2 differentiation [24,25,26,28,29,30,31,32]. In addition, this review highlights the origin of EV impacts, whether the EV promotes tolerance or sensitization, where EVs generated internally by the body influence both Th1 and Th2 responses, but exogenous EVs generated by bacteria mainly promote allergen exposure by increasing the permeability of the skin, thus resulting in increased chance of allergens interacting with immune cells [33,34,35,36,37,40]. Furthermore, bacteria-derived EVs can act as the ‘allergen’ and trigger allergic sensitization itself by stimulating IL-4, IL-5, and IL-6 production, and then, on re-exposure, trigger an IgE-mediated response [34].
One of the key mechanisms driving allergic sensitization suggested is EVs produced by DCs, B cells, Tfh, and epithelial cells that can affect the cytokine production in immune cells by promoting IL-4, IL-5, and IL-13 production while simultaneously decreasing the levels of IFN-γ and IL-17. For example, DC-derived EVs induced Th2 differentiation and suppressed Th1 responses, which can lead to allergic sensitization [17,18,19,20,21,23]. Currently, what induces naïve T cells to differentiate into Th2 cells during allergic sensitization is poorly understood [42]; however, the evidence presented here shows that EVs generated by immune cells can act as the signal to trigger the Th2 differentiation. Further research in this area could lead to developing new treatments to stop naïve—cell differentiation into Th2 cells, thus stopping allergic sensitization. Conversely, it is possible to reduce Th2 responses by inducing ‘desensitization’ by immunotherapy, where allergen-specific IgG, more specifically IgG4, antibodies are produced and compete with IgE and show inhibitory capacity for IgE-dependent events [43]. IgG4 can bind to the allergen specifically, thus blocking it from interacting with IgE, meaning decreased levels of IgE-allergen deposits on mast cells and, in turn, decreased histamine levels.
Furthermore, it has been shown that IgG4 competition can block IgE-allergen complexes binding to B cells, thereby inhibiting IgE-facilitated antigen presentation to T cells, which is a major driving force for generating allergen-specific Th2 responses [44]. Recent research has been exploring the potential of EVs used in therapies utilizing the ‘desensitization’ process [45]. Another mechanism by which EVs can promote allergic sensitization is by damaging the epithelial barrier by carrying lncRNA NEAT1, which interacts with HNECs to promote IL-13 and apoptosis, thus enhancing the likelihood of allergen exposure to immune cells [22].
In addition, a study by Gon et al. found that EVs released in the bronchoalveolar lavage fluid (BALF) were enriched with miRNAs once exposed to house-dust mites, and these EVs could upregulate Th2 cytokines [46], highlighting the importance of EVs as carriers of effector molecules. Furthermore, a study found key evidence where bronchial epithelial cells secreted by EVs carry a pivotal protein tissue factor (TF) in asthma and are involved in its pathogenesis [47]. This highlights the importance of studying the contents of EVs, as their cargo can be essential in their function.
As aforementioned, it is also possible for EVs to induce Th1 and Treg responses, thus inducing immune tolerance towards the allergen. One of the key mechanisms suggested is inducing Tregs in the immune system by increasing the production of TGF-β [24,25,26,28,29,31,32]. Furthermore, it is suggested that EVs carrying integrin αβ6 and OVA could interact with T cells and induce Treg differentiation, thus suppressing allergic sensitization [24].
Another mechanism of suppression of Th2 differentiation was by HMSC-derived EVs influencing the miR-146a-5p/SERPINB2 pathway in healthy CD4+ T cells [30], where it can be speculated that EVs can affect intrinsic pathways in the cells that they interact with either by activating the recipient cells receptors, thus initiating intracellular signaling pathways or cargo by fusion with the plasma membrane of target cells [48]. In addition, previously published literature has found how EVs can affect specific cells through intrinsic signaling pathways; for example, a study done by Salem et al. showed that EVs generated from bronchial fibroblasts could promote epithelial cell line remodeling through the TGF-β2 signaling pathway in severe asthma [49], suggesting that EVs are affecting the target cells’ intrinsic pathways and thus altering their function.
Another study found that EVs can also induce immune tolerance towards unrelated allergens, as well as specific allergens, by limiting the production of IL-5 and IL-13, thus inducing ‘bystander tolerance’ [32].
Recently, there has been more focus on the microbiome of humans and how it can affect the immune system [50]. It was found that EVs produced by bacteria, specifically S. aureus, could be the leading cause of damage to the skin and induction of AD-like symptoms, which are most likely caused by the EV cargo. For instance, the toxin α-Hemolysin was found to be more potent in EVs than in soluble form, causing further skin damage [33,35,36,37,40]. Damaged skin barrier can lead to increased susceptibility to allergic sensitization of allergens. This is due to the compromised barrier releasing defensins, which can predispose to allergic sensitization by subsequent interactions with T cells and can thus lead to atopic dermatitis [51]. In this systematic review, a major function of EVs was suggested by a study regarding MRSA-derived EVs, where EVs acted as immunostimulants, inducing IL-4, IL-5, and IL-6 cytokines in mice. Furthermore, following subsequent exposure to MRSA, an IgE-mediated hypersensitivity developed [34]. This suggests that EVs were able to sensitize the mice towards MRSA, directly suggesting that EVs can trigger allergic sensitization without additional signals from the mice.
It is worth noting that many of the studies examined had low-quality assessment scores, as many of the included studies had poor methods of EV isolation and characterization. The papers providing evidence for host-derived EVs and their influence on allergic sensitization scored a mean value of 57%, which was minimally higher by 5% than host-derived EVs inducing tolerance, showing the reliability of the key mechanisms was similar in these sections. However, the highest scoring section was exogenous EVs in promoting allergic sensitization with a 62% mean score, while exogenous EVs and tolerance had 58% scoring quality, indicating the robustness of these studies was of similar quality. As the sections do not have high variance between their scoring, there is no indication of higher reliability in the findings between the sections. The quality scoring highlights the need for further understanding of EVs and how to isolate them more efficiently to increase purity and maintain their original form without rupture. Only two studies [19,21] received the highest points for EV characterization. In addition, five different studies [20,28,30,33,37] had the highest score of EV isolation. Thus, future research must utilize new isolation techniques to ensure the highest purity of EVs and the exclusion of cytokines, as it has been shown that cytokine presence can affect the function of the EVs.
Furthermore, fully characterizing EVs would lead to a better understanding of the differences between the sub-populations of EVs and which receptors, and what cargo, drives their role in allergy. In addition, almost all studies lacked human ex vivo experiments, except one study [37], which used a foreskin puncture to show a more accurate representation of the immune and epithelial cell interactions. Interestingly, almost all studies had high scores regarding the robustness of the model, as they clearly described what cell type was used, and the duration, exposure method, and dosing of the allergen were fully explained. All the studies received the same score for material used for sensitization as the allergen was clearly described and measured; however, none of the studies performed endotoxin measurement to ensure the allergen was endotoxin-free.
This systematic review contained studies both using human and murine models, with 57% of studies utilizing murine models for the study of allergic sensitization. However, the use of mice in allergy studies is not preferred, as an allergy in mice is induced artificially and does not fully represent allergic sensitization in humans. In addition, mice have been shown to produce cytokines differently than humans; for example, IL-10 in mice is only produced by Th2 response, while in humans, it can be both Th1 and Th2 responses [52].
Future research in the field of EVs could open avenues for understanding the development of allergic sensitization, possibly leading to the development of new treatments and/or prevention of allergic sensitization. It is clear EVs play a role in allergies, either in their development or by induction of tolerance, and this is dependent on the cell of origin, health status, and environmental conditions in which the cells grow. In turn, this can influence the outcome of the EVs generated, resulting in different cargo encapsulations and differing cellular outcomes [53]. In the future, more research is required on EVs released by cells, investigating their cargo and proteins on the surface, which interact with immune cells to shift the response away from allergic sensitization and towards tolerance.
5. Conclusions
In summary, this systematic review provides evidence for the role of extracellular vesicles in allergies, where they have been shown to influence Th1, Th2 or Treg responses to allergens. This evidence suggests that EVs could be the driving force that decides the immune response to an allergen. However, with the limited number of papers that have been published, further research is crucial in fully understanding the role of EVs in allergies.
Author Contributions
D.T. wrote the manuscript and undertook the search for and reviewing of papers. G.H., D.O. and V.J. reviewed and edited the paper. W.B. created the scoring system. L.C.F. was involved in the conceptualization, supervision, review and editing of the paper. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Acknowledgments
The University of Nottingham provided access to online libraries and services.
Conflicts of Interest
There are no conflicts of interest.
References
- Pawankar, R. Allergic diseases and asthma: A global public health concern and a call to action. World Allergy Organ. J. 2014, 7, 12. [Google Scholar] [CrossRef] [PubMed]
- Gupta, R.; Sheikh, A.; Strachan, D.P.; Anderson, H.R. Burden of allergic disease in the UK: Secondary analyses of national databases. Clin. Exp. Allergy 2004, 34, 520–526. [Google Scholar] [CrossRef]
- Rennie, G.H.; Zhao, J.; Camus-Ela, M.; Shi, J.; Jiang, L.; Zhang, L.; Wang, J.; Raghavan, V. Influence of Lifestyle and Dietary Habits on the Prevalence of Food Allergies: A Scoping Review. Foods 2023, 12, 3290. [Google Scholar] [CrossRef]
- Collin, M.; Bigley, V. Human dendritic cell subsets: An update. Immunology 2018, 154, 3–20. [Google Scholar] [CrossRef]
- Hopkins, G.V.; Cochrane, S.; Onion, D.; Fairclough, L.C. The Role of Lipids in Allergic Sensitization: A Systematic Review. Front. Mol. Biosci. 2022, 9, 832330. [Google Scholar] [CrossRef] [PubMed]
- Hopkins, G.V.; Cochrane, S.; Onion, D.; Fairclough, L.C. Invariant NKT cells are more abundant in peanut-allergic adults and a subset of CD8(+) iNKT cells are depleted after peanut oil exposure. Front. Immunol. 2023, 14, 1293158. [Google Scholar] [CrossRef] [PubMed]
- Keumatio Doungstop, B.C.; van Vliet, S.J.; van Ree, R.; de Jong, E.C.; van Kooyk, Y. Carbohydrates in allergy: From disease to novel immunotherapies. Trends Immunol. 2021, 42, 635–648. [Google Scholar] [CrossRef]
- Demkow, U.; Stelmaszczyk-Emmel, A. Extracellular Vesicles in Allergic Rhinitis and Asthma and Laboratory Possibilities for Their Assessment. Int. J. Mol. Sci. 2021, 22, 2273. [Google Scholar] [CrossRef]
- Mathieu, M.; Martin-Jaular, L.; Lavieu, G.; Théry, C. Specificities of secretion and uptake of exosomes and other extracellular vesicles for cell-to-cell communication. Nat. Cell Biol. 2019, 21, 9–17. [Google Scholar] [CrossRef] [PubMed]
- Johnson, S.M.; Banyard, A.; Smith, C.; Mironov, A.; McCabe, M.G. Large Extracellular Vesicles Can be Characterised by Multiplex Labelling Using Imaging Flow Cytometry. Int. J. Mol. Sci. 2020, 21, 8723. [Google Scholar] [CrossRef]
- Alipoor, S.D.; Mortaz, E.; Garssen, J.; Movassaghi, M.; Mirsaeidi, M.; Adcock, I.M. Exosomes and Exosomal miRNA in Respiratory Diseases. Mediat. Inflamm. 2016, 2016, 5628404. [Google Scholar] [CrossRef] [PubMed]
- Schorey, J.S.; Cheng, Y.; Singh, P.P.; Smith, V.L. Exosomes and other extracellular vesicles in host-pathogen interactions. EMBO Rep. 2015, 16, 24–43. [Google Scholar] [CrossRef]
- Welsh, J.A.; Goberdhan, D.C.I.; O‘Driscoll, L.; Buzas, E.I.; Blenkiron, C.; Bussolati, B.; Cai, H.; Di Vizio, D.; Driedonks, T.A.P.; Erdbrügger, U.; et al. Minimal information for studies of extracellular vesicles (MISEV2023): From basic to advanced approaches. J. Extracell. Vesicles 2024, 13, e12404. [Google Scholar] [CrossRef]
- De Toro, J.; Herschlik, L.; Waldner, C.; Mongini, C. Emerging Roles of Exosomes in Normal and Pathological Conditions: New Insights for Diagnosis and Therapeutic Applications. Front. Immunol. 2015, 6, 203. [Google Scholar] [CrossRef] [PubMed]
- Fang, S.-B.; Zhang, H.-Y.; Meng, X.-C.; Wang, C.; He, B.-X.; Peng, Y.-Q.; Xu, Z.-B.; Fan, X.-L.; Wu, Z.-J.; Wu, Z.-C.; et al. Small extracellular vesicles derived from human MSCs prevent allergic airway inflammation via immunomodulation on pulmonary macrophages. Cell Death Dis. 2020, 11, 409. [Google Scholar] [CrossRef]
- Sangaphunchai, P.; Todd, I.; Fairclough, L.C. Extracellular vesicles and asthma: A review of the literature. Clin. Exp. Allergy 2020, 50, 291–307. [Google Scholar] [CrossRef] [PubMed]
- Admyre, C.; Bohle, B.; Johansson, S.M.; Focke-Tejkl, M.; Valenta, R.; Scheynius, A.; Gabrielsson, S. B cell-derived exosomes can present allergen peptides and activate allergen-specific T cells to proliferate and produce TH2-like cytokines. J. Allergy Clin. Immunol. 2007, 120, 1418–1424. [Google Scholar] [CrossRef]
- Fang, S.-B.; Zhou, Z.-R.; Peng, Y.-Q.; Liu, X.-Q.; He, B.-X.; Chen, D.-H.; Chen, D.; Fu, Q.-L. Plasma EVs Display Antigen-Presenting Characteristics in Patients With Allergic Rhinitis and Promote Differentiation of Th2 Cells. Front. Immunol. 2021, 12, 710372. [Google Scholar] [CrossRef]
- Huang, L.; Zhang, X.; Wang, M.; Chen, Z.; Yan, Y.; Gu, W.; Tan, J.; Jiang, W.; Ji, W. Exosomes from Thymic Stromal Lymphopoietin-Activated Dendritic Cells Promote Th2 Differentiation through the OX40 Ligand. Pathobiology 2018, 86, 111–117. [Google Scholar] [CrossRef]
- Teng, Z.X.; Zhou, X.C.; Xu, R.T.; Zhu, F.Y.; Bing, X.; Guo, N.; Shi, L.; Qi, W.W.; Liu, C.C.; Xia, M. Tfh Exosomes Derived from Allergic Rhinitis Promote DC Maturation Through miR-142-5p/CDK5/STAT3 Pathway. J. Inflamm. Res. 2022, 15, 3187–3205. [Google Scholar] [CrossRef]
- Vallhov, H.; Gutzeit, C.; Hultenby, K.; Valenta, R.; Grönlund, H.; Scheynius, A. Dendritic cell-derived exosomes carry the major cat allergen Fel d 1 and induce an allergic immune response. Allergy 2015, 70, 1651–1655. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Cai, W.; Wu, Q.; Chen, D.; Wang, P.; Xu, Z. Exosomal lncRNA Nuclear Paraspeckle Assembly Transcript 1 (NEAT1)contributes to the progression of allergic rhinitis via modulating microRNA-511/Nuclear Receptor Subfamily 4 Group A Member 2 (NR4A2) axis. Bioengineered 2021, 12, 8067–8079. [Google Scholar] [CrossRef] [PubMed]
- Zhu, X.; Wang, X.; Wang, Y.; Zhao, Y. Exosomal long non-coding RNA GAS5 suppresses Th1 differentiation and promotes Th2 differentiation via downregulating EZH2 and T-bet in allergic rhinitis. Mol. Immunol. 2020, 118, 30–39. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Song, C.H.; Feng, B.S.; Li, T.L.; Li, P.; Zheng, P.Y.; Chen, X.M.; Xing, Z.; Yang, P.C. Intestinal epithelial cell-derived integrin αβ6 plays an important role in the induction of regulatory T cells and inhibits an antigen-specific Th2 response. J. Leukoc. Biol. 2011, 90, 751–759. [Google Scholar] [CrossRef]
- Yu, D.; Liu, J.Q.; Mo, L.H.; Luo, X.Q.; Liu, Z.Q.; Wu, G.H.; Yang, L.T.; Liu, D.B.; Wang, S.; Liu, Z.G.; et al. Specific antigen-guiding exosomes inhibit food allergies by inducing regulatory T cells. Immunol. Cell Biol. 2020, 98, 639–649. [Google Scholar] [CrossRef]
- Zeng, H.T.; Liu, J.Q.; Zhao, M.; Yu, D.; Yang, G.; Mo, L.H.; Liu, Z.Q.; Wang, S.; Liu, Z.G.; Yang, P.C. Exosomes carry IL-10 and antigen/MHC II complexes to induce antigen-specific oral tolerance. Cytokine 2020, 133, 155176. [Google Scholar] [CrossRef] [PubMed]
- Asadirad, A.; Ghadiri, A.A.; Amari, A.; Ghasemi Dehcheshmeh, M.; Sadeghi, M.; Dehnavi, S. Sublingual prophylactic administration of OVA-loaded MSC-derived exosomes to prevent allergic sensitization. Int. Immunopharmacol. 2023, 120, 110405. [Google Scholar] [CrossRef] [PubMed]
- Peng, Y.Q.; Wu, Z.C.; Xu, Z.B.; Fang, S.B.; Chen, D.H.; Zhang, H.Y.; Liu, X.Q.; He, B.X.; Chen, D.; Akdis, C.A.; et al. Mesenchymal stromal cells-derived small extracellular vesicles modulate DC function to suppress Th2 responses via IL-10 in patients with allergic rhinitis. Eur. J. Immunol. 2022, 52, 1129–1140. [Google Scholar] [CrossRef] [PubMed]
- Prado, N.; Marazuela, E.G.; Segura, E.; Fernández-García, H.; Villalba, M.; Théry, C.; Rodríguez, R.; Batanero, E. Exosomes from bronchoalveolar fluid of tolerized mice prevent allergic reaction. J. Immunol. 2008, 181, 1519–1525. [Google Scholar] [CrossRef]
- Zhou, J.; Lu, Y.; Wu, W.; Feng, Y. HMSC-Derived Exosome Inhibited Th2 Cell Differentiation via Regulating miR-146a-5p/SERPINB2 Pathway. J. Immunol. Res. 2021, 2021, 6696525. [Google Scholar] [CrossRef]
- Wahlund, C.J.E.; Güclüler, G.; Hiltbrunner, S.; Veerman, R.E.; Näslund, T.I.; Gabrielsson, S. Exosomes from antigen-pulsed dendritic cells induce stronger antigen-specific immune responses than microvesicles in vivo. Sci. Rep. 2017, 7, 17095. [Google Scholar] [CrossRef] [PubMed]
- Prado, N.; Cañamero, M.; Villalba, M.; Rodríguez, R.; Batanero, E. Bystander suppression to unrelated allergen sensitization through intranasal administration of tolerogenic exosomes in mouse. Mol. Immunol. 2010, 47, 2148–2151. [Google Scholar] [CrossRef] [PubMed]
- Hong, S.W.; Choi, E.B.; Min, T.K.; Kim, J.H.; Kim, M.H.; Jeon, S.G.; Lee, B.J.; Gho, Y.S.; Jee, Y.K.; Pyun, B.Y.; et al. An important role of α-hemolysin in extracellular vesicles on the development of atopic dermatitis induced by Staphylococcus aureus. PLoS ONE 2014, 9, e100499. [Google Scholar] [CrossRef]
- Asano, K.; Hirose, S.; Narita, K.; Subsomwong, P.; Kawai, N.; Sukchawalit, R.; Nakane, A. Extracellular vesicles from methicillin resistant Staphylococcus aureus stimulate proinflammatory cytokine production and trigger IgE-mediated hypersensitivity. Emerg. Microbes Infect. 2021, 10, 2000–2009. [Google Scholar] [CrossRef]
- Hong, S.W.; Kim, M.R.; Lee, E.Y.; Kim, J.H.; Kim, Y.S.; Jeon, S.G.; Yang, J.M.; Lee, B.J.; Pyun, B.Y.; Gho, Y.S.; et al. Extracellular vesicles derived from Staphylococcus aureus induce atopic dermatitis-like skin inflammation. Allergy 2011, 66, 351–359. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.H.; Choi, S.J.; Choi, H.I.; Choi, J.P.; Park, H.K.; Kim, E.K.; Kim, M.J.; Moon, B.S.; Min, T.K.; Rho, M.; et al. Lactobacillus plantarum-derived Extracellular Vesicles Protect Atopic Dermatitis Induced by Staphylococcus aureus-derived Extracellular Vesicles. Allergy Asthma Immunol. Res. 2018, 10, 516–532. [Google Scholar] [CrossRef] [PubMed]
- Staudenmaier, L.; Focken, J.; Schlatterer, K.; Kretschmer, D.; Schittek, B. Bacterial membrane vesicles shape Staphylococcus aureus skin colonization and induction of innate immune responses. Exp. Dermatol. 2022, 31, 349–361. [Google Scholar] [CrossRef]
- Akbar, A.; Malekian, F.; Baghban, N.; Kodam, S.P.; Ullah, M. Methodologies to Isolate and Purify Clinical Grade Extracellular Vesicles for Medical Applications. Cells 2022, 11, 186. [Google Scholar] [CrossRef]
- Effah, C.Y.; Ding, X.; Drokow, E.K.; Li, X.; Tong, R.; Sun, T. Bacteria-derived extracellular vesicles: Endogenous roles, therapeutic potentials and their biomimetics for the treatment and prevention of sepsis. Front. Immunol. 2024, 15, 1296061. [Google Scholar] [CrossRef]
- Kim, Y.S.; Choi, E.J.; Lee, W.H.; Choi, S.J.; Roh, T.Y.; Park, J.; Jee, Y.K.; Zhu, Z.; Koh, Y.Y.; Gho, Y.S.; et al. Extracellular vesicles, especially derived from Gram-negative bacteria, in indoor dust induce neutrophilic pulmonary inflammation associated with both Th1 and Th17 cell responses. Clin. Exp. Allergy 2013, 43, 443–454. [Google Scholar] [CrossRef]
- Gutowska-Ślesik, J.; Samoliński, B.; Krzych-Fałta, E. The increase in allergic conditions based on a review of literature. Postep. Dermatol. Alergol. 2023, 40, 119009. [Google Scholar] [CrossRef] [PubMed]
- Lamiable, O.; Mayer, J.U.; Munoz-Erazo, L.; Ronchese, F. Dendritic cells in Th2 immune responses and allergic sensitization. Immunol. Cell Biol. 2020, 98, 807–818. [Google Scholar] [CrossRef] [PubMed]
- Della-Torre, E.; Germanò, T.; Ramirez, G.A.; Dagna, L.; Yacoub, M.R. IgG4-related disease and allergen-specific immunotherapy. Ann. Allergy Asthma Immunol. 2020, 124, 631–633. [Google Scholar] [CrossRef] [PubMed]
- Shamji, M.H.; Durham, S.R. Mechanisms of allergen immunotherapy for inhaled allergens and predictive biomarkers. J. Allergy Clin. Immunol. 2017, 140, 1485–1498. [Google Scholar] [CrossRef]
- Engeroff, P.; Vogel, M. The Potential of Exosomes in Allergy Immunotherapy. Vaccines 2022, 10, 133. [Google Scholar] [CrossRef] [PubMed]
- Gon, Y.; Maruoka, S.; Inoue, T.; Kuroda, K.; Yamagishi, K.; Kozu, Y.; Shikano, S.; Soda, K.; Lötvall, J.; Hashimoto, S. Selective release of miRNAs via extracellular vesicles is associated with house-dust mite allergen-induced airway inflammation. Clin. Exp. Allergy 2017, 47, 1586–1598. [Google Scholar] [CrossRef]
- Park, J.A.; Sharif, A.S.; Tschumperlin, D.J.; Lau, L.; Limbrey, R.; Howarth, P.; Drazen, J.M. Tissue factor-bearing exosome secretion from human mechanically stimulated bronchial epithelial cells in vitro and in vivo. J. Allergy Clin. Immunol. 2012, 130, 1375–1383. [Google Scholar] [CrossRef] [PubMed]
- Ginini, L.; Billan, S.; Fridman, E.; Gil, Z. Insight into Extracellular Vesicle-Cell Communication: From Cell Recognition to Intracellular Fate. Cells 2022, 11, 1375. [Google Scholar] [CrossRef] [PubMed]
- Haj-Salem, I.; Plante, S.; Gounni, A.S.; Rouabhia, M.; Chakir, J. Fibroblast-derived exosomes promote epithelial cell proliferation through TGF-β2 signalling pathway in severe asthma. Allergy 2018, 73, 178–186. [Google Scholar] [CrossRef]
- Lambring, C.B.; Siraj, S.; Patel, K.; Sankpal, U.T.; Mathew, S.; Basha, R. Impact of the Microbiome on the Immune System. Crit. Rev. Immunol. 2019, 39, 313–328. [Google Scholar] [CrossRef]
- Nedorost, S.; Hammond, M. Art of prevention: Allergic sensitization through damaged skin: Atopic, occupational, and stasis dermatitis. Int. J. Womens Dermatol. 2020, 6, 381–383. [Google Scholar] [CrossRef]
- Mestas, J.; Hughes, C.C. Of mice and not men: Differences between mouse and human immunology. J. Immunol. 2004, 172, 2731–2738. [Google Scholar] [CrossRef]
- Rayamajhi, S.; Sulthana, S.; Ferrel, C.; Shrestha, T.B.; Aryal, S. Extracellular vesicles production and proteomic cargo varies with incubation time and temperature. Exp. Cell Res. 2023, 422, 113454. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).