NF-κB in the Radiation Response of A549 Non-Small Cell Lung Cancer Cells to X-rays and Carbon Ions under Hypoxia
Abstract
1. Introduction
2. Results
2.1. Expression of NF-κB Target Genes in A549 Cells after Exposure to X-rays or Carbon Ions under Normoxia and Hypoxia
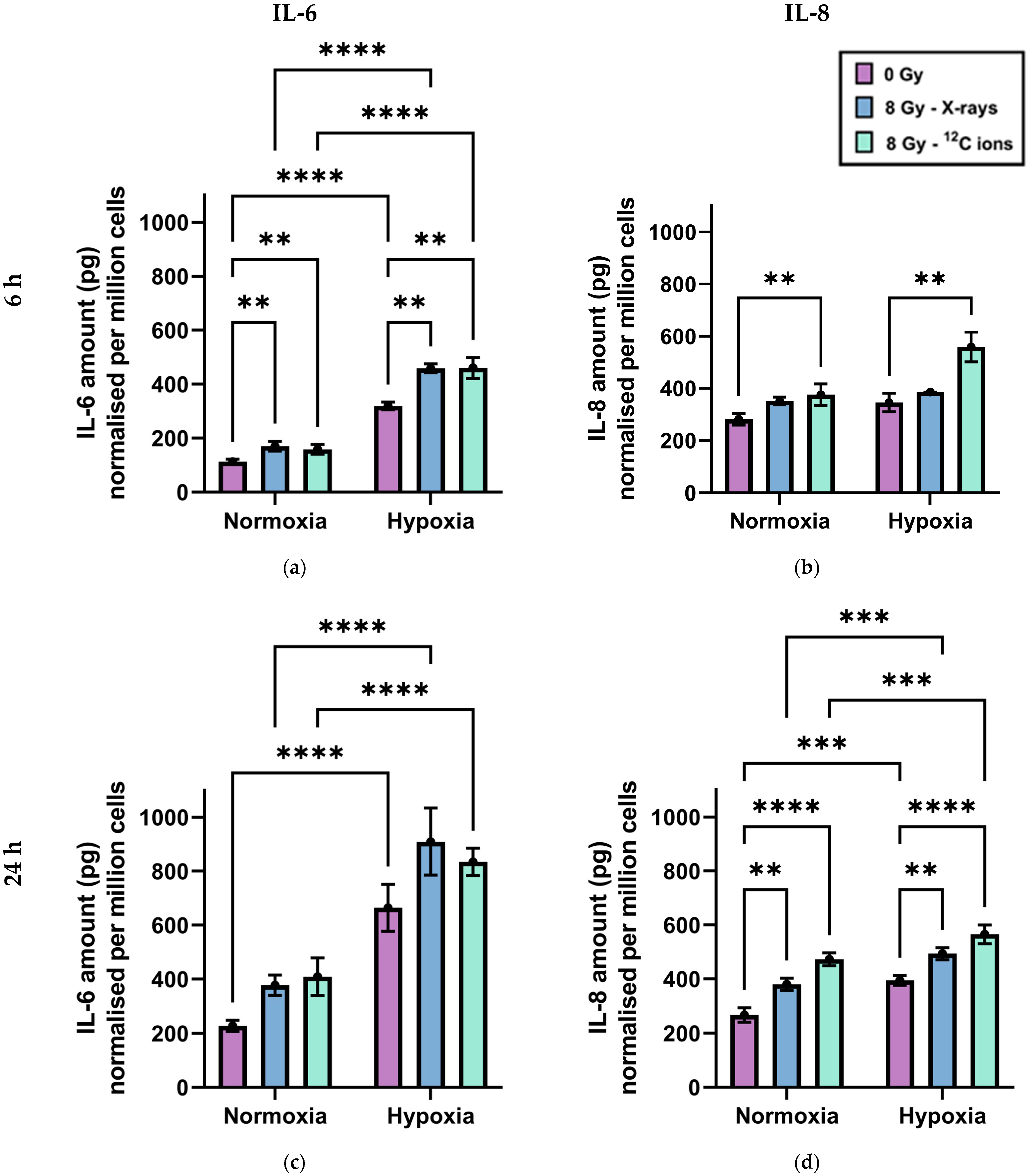
2.2. Secretion of the Cytokines IL-6 and IL-8 by A549 Cells after Exposure to X-rays or Carbon Ions under Normoxia and Hypoxia
3. Discussion
3.1. Irradiation Upregulates NF-κB Target Genes Influencing Apoptosis, Cell Cycle, and Cell Migration in A549 Cells
3.2. High-LET Radiation Enhances NF-κB Target Genes Expression in A549 Cells Compared to Low-LET Radiation
3.3. Chronic Hypoxia Upregulates NF-κB Target Genes Influencing Cell Survival and Cell Migration in A549 Cells
3.4. NF-κB Target Genes’ Activation Signature Following Irradiation May Promote Cancer Progression under Hypoxia, Especially after X-ray Exposure
3.5. Chronic Hypoxia Results in an Inflammatory Response in A549 Cells Distinct from Normoxia, Irrespective of Irradiation Status
4. Materials and Methods
4.1. Cell Line and Cultivation
4.2. Irradiation
4.3. Gene Expression Analysis
4.4. Quantification of Cytokines
4.5. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Siegel, R.; Naishadham, D.; Jemal, A. Cancer Statistics, 2013. CA Cancer J. Clin. 2013, 63, 11–30. [Google Scholar] [CrossRef]
- Kang, K.H.; Okoye, C.C.; Patel, R.B.; Siva, S.; Biswas, T.; Ellis, R.J.; Yao, M.; Machtay, M.; Lo, S.S. Complications from Stereotactic Body Radiotherapy for Lung Cancer. Cancers 2015, 7, 981–1004. [Google Scholar] [CrossRef]
- Zegers, C.M.L.; van Elmpt, W.; Szardenings, K.; Kolb, H.; Waxman, A.; Subramaniam, R.M.; Moon, D.H.; Brunetti, J.C.; Srinivas, S.M.; Lambin, P.; et al. Repeatability of Hypoxia PET Imaging Using [18F]HX4 in Lung and Head and Neck Cancer Patients: A Prospective Multicenter Trial. Eur. J. Nucl. Med. Mol. Imaging 2015, 42, 1840–1849. [Google Scholar] [CrossRef]
- van Elmpt, W.; Zegers, K.; Reymen, B.; Even, A.J.G.; Dingemans, A.-M.C.; Öllers, M.; Wildberger, J.E.; Mottaghy, F.M.; Das, M.; Troost, E.G.C.; et al. Multiparametric Imaging of Patient and Tumour Heterogeneity in Non-Small-Cell Lung Cancer: Quantification of Tumour Hypoxia, Metabolism and Perfusion. Eur. J. Nucl. Med. Mol. Imaging 2016, 43, 240–248. [Google Scholar] [CrossRef]
- Postema, E.J.; McEwan, A.J.B.; Riauka, T.A.; Kumar, P.; Richmond, D.A.; Abrams, D.N.; Wiebe, L.I. Initial Results of Hypoxia Imaging Using 1-α-D-(5-Deoxy-5-[18F]-Fluoroarabinofuranosyl)-2-Nitroimidazole (18F-FAZA). Eur. J. Nucl. Med. Mol. Imaging 2009, 36, 1565–1573. [Google Scholar] [CrossRef]
- Trinkaus, M.E.; Blum, R.; Rischin, D.; Callahan, J.; Bressel, M.; Segard, T.; Roselt, P.; Eu, P.; Binns, D.; MacManus, M.P.; et al. Imaging of Hypoxia with 18F-FAZA PET in Patients with Locally Advanced Non-Small Cell Lung Cancer Treated with Definitive Chemoradiotherapy. J. Med. Imaging Radiat. Oncol. 2013, 57, 475–481. [Google Scholar] [CrossRef]
- Li, L.; Hu, M.; Zhu, H.; Zhao, W.; Yang, G.; Yu, J. Comparison of 18F-Fluoroerythronitroimidazole and 18F-Fluorodeoxyglucose Positron Emission Tomography and Prognostic Value in Locally Advanced Non–Small-Cell Lung Cancer. Clin. Lung Cancer 2010, 11, 335–340. [Google Scholar] [CrossRef]
- Ren, W.; Mi, D.; Yang, K.; Cao, N.; Tian, J.; Li, Z.; Ma, B. The Expression of Hypoxia-Inducible Factor-1α and Its Clinical Significance in Lung Cancer: A Systematic Review and Meta-Analysis. Swiss Med. Wkly. 2013, 143, w13855. [Google Scholar] [CrossRef]
- Li, C.; Lu, H.-J.; Na, F.-F.; Deng, L.; Xue, J.-X.; Wang, J.-W.; Wang, Y.-Q.; Li, Q.-L.; Lu, Y. Prognostic Role of Hypoxic Inducible Factor Expression in Non-Small Cell Lung Cancer: A Meta-Analysis. Asian Pac. J. Cancer Prev. 2013, 14, 3607–3612. [Google Scholar] [CrossRef]
- Yang, S.-L.; Ren, Q.-G.; Wen, L.; Hu, J.-L. Clinicopathological and Prognostic Significance of Hypoxia-Inducible Factor-1 Alpha in Lung Cancer: A Systematic Review with Meta-Analysis. J. Huazhong Univ. Sci. Technol. Med. Sci. 2016, 36, 321–327. [Google Scholar] [CrossRef]
- Salem, A.; Asselin, M.-C.; Reymen, B.; Jackson, A.; Lambin, P.; West, C.M.L.; O’Connor, J.P.B.; Faivre-Finn, C. Targeting Hypoxia to Improve Non–Small Cell Lung Cancer Outcome. JNCI J. Natl. Cancer Inst. 2018, 110, 14–30. [Google Scholar] [CrossRef] [PubMed]
- Vyfhuis, M.A.L.; Onyeuku, N.; Diwanji, T.; Mossahebi, S.; Amin, N.P.; Badiyan, S.N.; Mohindra, P.; Simone, C.B. Advances in Proton Therapy in Lung Cancer. Ther. Adv. Respir. Dis. 2018, 12, 1753466618783878. [Google Scholar] [CrossRef]
- Aoki, S.; Onishi, H.; Karube, M.; Yamamoto, N.; Yamashita, H.; Shioyama, Y.; Matsumoto, Y.; Matsuo, Y.; Miyakawa, A.; Matsushita, H.; et al. Comparative Analysis of Photon Stereotactic Radiotherapy and Carbon-Ion Radiotherapy for Elderly Patients with Stage I Non-Small-Cell Lung Cancer: A Multicenter Retrospective Study. Cancers 2023, 15, 3633. [Google Scholar] [CrossRef]
- Wozny, A.-S.; Rodriguez-Lafrasse, C. The ‘Stealth-Bomber’ Paradigm for Deciphering the Tumour Response to Carbon-Ion Irradiation. Br. J. Cancer 2023, 128, 1429–1438. [Google Scholar] [CrossRef]
- Pouget, J.-P.; Mather, S.J. General Aspects of the Cellular Response to Low- and High-LET Radiation. Eur. J. Nucl. Med. 2001, 28, 541–561. [Google Scholar] [CrossRef]
- Kabakov, A.E.; Yakimova, A.O. Hypoxia-Induced Cancer Cell Responses Driving Radioresistance of Hypoxic Tumors: Approaches to Targeting and Radiosensitizing. Cancers 2021, 13, 1102. [Google Scholar] [CrossRef]
- Klein, C.; Dokic, I.; Mairani, A.; Mein, S.; Brons, S.; Häring, P.; Haberer, T.; Jäkel, O.; Zimmermann, A.; Zenke, F.; et al. Overcoming Hypoxia-Induced Tumor Radioresistance in Non-Small Cell Lung Cancer by Targeting DNA-Dependent Protein Kinase in Combination with Carbon Ion Irradiation. Radiat. Oncol. 2017, 12, 208. [Google Scholar] [CrossRef]
- Zaarour, R.F.; Ribeiro, M.; Azzarone, B.; Kapoor, S.; Chouaib, S. Tumor Microenvironment-Induced Tumor Cell Plasticity: Relationship with Hypoxic Stress and Impact on Tumor Resistance. Front. Oncol. 2023, 13, 1222575. [Google Scholar] [CrossRef] [PubMed]
- Hellweg, C.E.; Spitta, L.F.; Koch, K.; Chishti, A.A.; Henschenmacher, B.; Diegeler, S.; Konda, B.; Feles, S.; Schmitz, C.; Berger, T.; et al. The Role of the Nuclear Factor κB Pathway in the Cellular Response to Low and High Linear Energy Transfer Radiation. Int. J. Mol. Sci. 2018, 19, 2220. [Google Scholar] [CrossRef] [PubMed]
- Pordanjani, S.M.; Hosseinimehr, S.J. The Role of NF-kB Inhibitors in Cell Response to Radiation. Curr. Med. Chem. 2016, 23, 3951–3963. [Google Scholar] [CrossRef]
- Singh, V.; Gupta, D.; Arora, R. NF-kB as a Key Player in Regulation of Cellular Radiation Responses and Identification of Radiation Countermeasures. Discoveries 2015, 3, e35. [Google Scholar] [CrossRef] [PubMed]
- Koong, A.C.; Chen, E.Y.; Giaccia, A.J. Hypoxia Causes the Activation of Nuclear Factor κB through the Phosphorylation of IκBα on Tyrosine Residues. Cancer Res. 1994, 54, 1425–1430. [Google Scholar] [PubMed]
- Patel, H.; Zaghloul, N.; Lin, K.; Liu, S.F.; Miller, E.J.; Ahmed, M. Hypoxia-Induced Activation of Specific Members of the NF-kB Family and Its Relevance to Pulmonary Vascular Remodeling. Int. J. Biochem. Cell Biol. 2017, 92, 141–147. [Google Scholar] [CrossRef] [PubMed]
- Oliver, K.M.; Garvey, J.F.; Ng, C.T.; Veale, D.J.; Fearon, U.; Cummins, E.P.; Taylor, C.T. Hypoxia Activates NF-κB–Dependent Gene Expression Through the Canonical Signaling Pathway. Antioxid. Redox Signal. 2009, 11, 2057–2064. [Google Scholar] [CrossRef]
- Wang, Y.; Li, N.; Wang, Y.; Zheng, G.; An, J.; Liu, C.; Wang, Y.; Liu, Q. NF-κB/P65 Competes With Peroxisome Proliferator-Activated Receptor Gamma for Transient Receptor Potential Channel 6 in Hypoxia-Induced Human Pulmonary Arterial Smooth Muscle Cells. Front. Cell Dev. Biol. 2021, 9, 656625. [Google Scholar] [CrossRef] [PubMed]
- Rastogi, S.; Aldosary, S.; Saeedan, A.S.; Ansari, M.N.; Singh, M.; Kaithwas, G. NF-κB Mediated Regulation of Tumor Cell Proliferation in Hypoxic Microenvironment. Front. Pharmacol. 2023, 14, 1108915. [Google Scholar] [CrossRef]
- Hellweg, C.E. The Nuclear Factor κB Pathway: A Link to the Immune System in the Radiation Response. Cancer Lett. 2015, 368, 275–289. [Google Scholar] [CrossRef]
- Oeckinghaus, A.; Ghosh, S. The NF-κB Family of Transcription Factors and Its Regulation. Cold Spring Harb. Perspect. Biol. 2009, 1, a000034. [Google Scholar] [CrossRef] [PubMed]
- Park, M.H.; Hong, J.T. Roles of NF-κB in Cancer and Inflammatory Diseases and Their Therapeutic Approaches. Cells 2016, 5, 15. [Google Scholar] [CrossRef]
- Yu, H.; Lin, L.; Zhang, Z.; Zhang, H.; Hu, H. Targeting NF-κB Pathway for the Therapy of Diseases: Mechanism and Clinical Study. Sig. Transduct. Target. Ther. 2020, 5, 209. [Google Scholar] [CrossRef]
- Ahmed, K.M.; Li, J.J. NF-κB-Mediated Adaptive Resistance to Ionizing Radiation. Free Radic. Biol. Med. 2008, 44, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Hellweg, C.E.; Baumstark-Khan, C.; Schmitz, C.; Lau, P.; Meier, M.M.; Testard, I.; Berger, T.; Reitz, G. Activation of the Nuclear Factor κB Pathway by Heavy Ion Beams of Different Linear Energy Transfer. Int. J. Radiat. Biol. 2011, 87, 954–963. [Google Scholar] [CrossRef] [PubMed]
- Chishti, A.A.; Baumstark-Khan, C.; Koch, K.; Kolanus, W.; Feles, S.; Konda, B.; Azhar, A.; Spitta, L.F.; Henschenmacher, B.; Diegeler, S.; et al. Linear Energy Transfer Modulates Radiation-Induced NF-Kappa B Activation and Expression of Its Downstream Target Genes. Radiat. Res. 2018, 189, 354–370. [Google Scholar] [CrossRef] [PubMed]
- Xia, Y.; Shen, S.; Verma, I.M. NF-κB, an Active Player in Human Cancers. Cancer Immunol. Res. 2014, 2, 823–830. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Ma, C.; Zhang, Z.; Zhang, H.; Hu, H. NF-κB Signaling in Inflammation and Cancer. MedComm 2021, 2, 618–653. [Google Scholar] [CrossRef] [PubMed]
- Hinz, T.K.; Kalkur, R.; Rabinovitch, J.; Hinkle, W.; Heasley, L.E. TP53 Null Mutations Identify Lung Cancer Cell Lines with Highest Sensitivity to the Nontaxane Microtubule Inhibitor Eribulin. Mol. Pharmacol. 2021, 100, 144–154. [Google Scholar] [CrossRef] [PubMed]
- Dhakar, R.; Dakal, T.C.; Sharma, A. Genetic Determinants of Lung Cancer: Understanding the Oncogenic Potential of Somatic Missense Mutations. Genomics 2022, 114, 110401. [Google Scholar] [CrossRef] [PubMed]
- Uras, I.Z.; Moll, H.P.; Casanova, E. Targeting KRAS Mutant Non-Small-Cell Lung Cancer: Past, Present and Future. Int. J. Mol. Sci. 2020, 21, 4325. [Google Scholar] [CrossRef] [PubMed]
- Dearden, S.; Stevens, J.; Wu, Y.-L.; Blowers, D. Mutation Incidence and Coincidence in Non Small-Cell Lung Cancer: Meta-Analyses by Ethnicity and Histology (mutMap). Ann. Oncol. 2013, 24, 2371–2376. [Google Scholar] [CrossRef]
- Choi, K.-C.; Lee, Y.-H.; Jung, M.G.; Kwon, S.H.; Kim, M.-J.; Jun, W.J.; Lee, J.; Lee, J.M.; Yoon, H.-G. Gallic Acid Suppresses Lipopolysaccharide-Induced Nuclear Factor-κB Signaling by Preventing RelA Acetylation in A549 Lung Cancer Cells. Mol. Cancer Res. 2009, 7, 2011–2021. [Google Scholar] [CrossRef]
- Garofalo, R.; Sabry, M.; Jamaluddin, M.; Yu, R.K.; Casola, A.; Ogra, P.L.; Brasier, A.R. Transcriptional Activation of the Interleukin-8 Gene by Respiratory Syncytial Virus Infection in Alveolar Epithelial Cells: Nuclear Translocation of the RelA Transcription Factor as a Mechanism Producing Airway Mucosal Inflammation. J. Virol. 1996, 70, 8773–8781. [Google Scholar] [CrossRef] [PubMed]
- Jamaluddin, M.; Tian, B.; Boldogh, I.; Garofalo, R.P.; Brasier, A.R. Respiratory Syncytial Virus Infection Induces a Reactive Oxygen Species-MSK1-Phospho-Ser-276 RelA Pathway Required for Cytokine Expression. J. Virol. 2009, 83, 10605–10615. [Google Scholar] [CrossRef] [PubMed]
- Carpenter, L.R.; Moy, J.N.; Roebuck, K.A. Respiratory Syncytial Virus and TNFalpha Induction of Chemokine Gene Expression Involves Differential Activation of Rel A and NF-kappaB1. BMC Infect. Dis. 2002, 2, 5. [Google Scholar] [CrossRef] [PubMed]
- Yamada, A.; Arakaki, R.; Saito, M.; Kudo, Y.; Ishimaru, N. Dual Role of Fas/FasL-Mediated Signal in Peripheral Immune Tolerance. Front. Immunol. 2017, 8, 217624. [Google Scholar] [CrossRef]
- Peter, M.E.; Hadji, A.; Murmann, A.E.; Brockway, S.; Putzbach, W.; Pattanayak, A.; Ceppi, P. The Role of CD95 and CD95 Ligand in Cancer. Cell Death Differ. 2015, 22, 549–559. [Google Scholar] [CrossRef] [PubMed]
- Kreis, N.-N.; Louwen, F.; Yuan, J. The Multifaceted P21 (Cip1/Waf1/CDKN1A) in Cell Differentiation, Migration and Cancer Therapy. Cancers 2019, 11, 1220. [Google Scholar] [CrossRef]
- Ling, J.; Zhang, L.; Chang, A.; Huang, Y.; Ren, J.; Zhao, H.; Zhuo, X. Overexpression of KITLG Predicts Unfavorable Clinical Outcomes and Promotes Lymph Node Metastasis via the JAK/STAT Pathway in Nasopharyngeal Carcinoma. Lab. Investig. 2022, 102, 1257–1267. [Google Scholar] [CrossRef]
- Yang, Z.; Liu, S.; Wang, Y.; Chen, Y.; Zhang, P.; Liu, Y.; Zhang, H.; Zhang, P.; Tao, Z.; Xiong, K. High Expression of KITLG Is a New Hallmark Activating the MAPK Pathway in Type A and AB Thymoma. Thorac. Cancer 2020, 11, 1944–1954. [Google Scholar] [CrossRef] [PubMed]
- Bruikman, C.S.; Zhang, H.; Kemper, A.M.; van Gils, J.M. Netrin Family: Role for Protein Isoforms in Cancer. J. Nucleic Acids 2019, 2019, 3947123. [Google Scholar] [CrossRef]
- Vilalta, M.; Rafat, M.; Graves, E.E. Effects of Radiation on Metastasis and Tumor Cell Migration. Cell. Mol. Life Sci. 2016, 73, 2999–3007. [Google Scholar] [CrossRef]
- Xiao, L.; Mao, Y.; Tong, Z.; Zhao, Y.; Hong, H.; Wang, F. Radiation Exposure Triggers the Malignancy of Non-Small Cell Lung Cancer Cells through the Activation of Visfatin/Snail Signaling. Oncol. Rep. 2021, 45, 1153–1161. [Google Scholar] [CrossRef] [PubMed]
- Aravindan, N.; Aravindan, S.; Manickam, K.; Natarajan, M. High Energy Particle Radiation-Associated Oncogenic Transformation in Normal Mice: Insight into the Connection between Activation of Oncotargets and Oncogene Addiction. Sci. Rep. 2016, 6, 37623. [Google Scholar] [CrossRef] [PubMed]
- Mahony, C.B.; Fish, R.J.; Pasche, C.; Bertrand, J.Y. Tfec Controls the Hematopoietic Stem Cell Vascular Niche during Zebrafish Embryogenesis. Blood 2016, 128, 1336–1345. [Google Scholar] [CrossRef] [PubMed]
- Petratou, K.; Spencer, S.A.; Kelsh, R.N.; Lister, J.A. The MITF Paralog Tfec Is Required in Neural Crest Development for Fate Specification of the Iridophore Lineage from a Multipotent Pigment Cell Progenitor. PLoS ONE 2021, 16, e0244794. [Google Scholar] [CrossRef] [PubMed]
- Zoncu, R.; Perera, R.M. Emerging Roles of the MiT/TFE Factors in Cancer. Trends Cancer 2023, 9, 817–827. [Google Scholar] [CrossRef] [PubMed]
- Liao, K.-L.; Bai, X.-F.; Friedman, A. IL-27 in Combination with Anti-PD-1 Can Be Anti-Cancer or pro-Cancer. J. Theor. Biol. 2024, 579, 111704. [Google Scholar] [CrossRef] [PubMed]
- Gelfo, V.; Romaniello, D.; Mazzeschi, M.; Sgarzi, M.; Grilli, G.; Morselli, A.; Manzan, B.; Rihawi, K.; Lauriola, M. Roles of IL-1 in Cancer: From Tumor Progression to Resistance to Targeted Therapies. Int. J. Mol. Sci. 2020, 21, 6009. [Google Scholar] [CrossRef]
- Hodge, D.R.; Hurt, E.M.; Farrar, W.L. The Role of IL-6 and STAT3 in Inflammation and Cancer. Eur. J. Cancer 2005, 41, 2502–2512. [Google Scholar] [CrossRef] [PubMed]
- Kumari, N.; Dwarakanath, B.S.; Das, A.; Bhatt, A.N. Role of Interleukin-6 in Cancer Progression and Therapeutic Resistance. Tumor Biol. 2016, 37, 11553–11572. [Google Scholar] [CrossRef]
- Ke, W.; Zhang, L.; Dai, Y. The Role of IL-6 in Immunotherapy of Non-Small Cell Lung Cancer (NSCLC) with Immune-Related Adverse Events (irAEs). Thorac. Cancer 2020, 11, 835–839. [Google Scholar] [CrossRef]
- Luppi, F.; Longo, A.M.; de Boer, W.I.; Rabe, K.F.; Hiemstra, P.S. Interleukin-8 Stimulates Cell Proliferation in Non-Small Cell Lung Cancer through Epidermal Growth Factor Receptor Transactivation. Lung Cancer 2007, 56, 25–33. [Google Scholar] [CrossRef]
- Waugh, D.J.J.; Wilson, C. The Interleukin-8 Pathway in Cancer. Clin. Cancer Res. 2008, 14, 6735–6741. [Google Scholar] [CrossRef]
- England, H.; Summersgill, H.R.; Edye, M.E.; Rothwell, N.J.; Brough, D. Release of Interleukin-1α or Interleukin-1β Depends on Mechanism of Cell Death*. J. Biol. Chem. 2014, 289, 15942–15950. [Google Scholar] [CrossRef] [PubMed]
- Martin, S.J. Cell Death and Inflammation: The Case for IL-1 Family Cytokines as the Canonical DAMPs of the Immune System. FEBS J. 2016, 283, 2599–2615. [Google Scholar] [CrossRef] [PubMed]
- Wooff, Y.; Man, S.M.; Aggio-Bruce, R.; Natoli, R.; Fernando, N. IL-1 Family Members Mediate Cell Death, Inflammation and Angiogenesis in Retinal Degenerative Diseases. Front. Immunol. 2019, 10, 462812. [Google Scholar] [CrossRef]
- Vanden Berghe, T.; Kalai, M.; Denecker, G.; Meeus, A.; Saelens, X.; Vandenabeele, P. Necrosis Is Associated with IL-6 Production but Apoptosis Is Not. Cell Signal. 2006, 18, 328–335. [Google Scholar] [CrossRef]
- Sano, E.; Kazaana, A.; Tadakuma, H.; Takei, T.; Yoshimura, S.; Hanashima, Y.; Ozawa, Y.; Yoshino, A.; Suzuki, Y.; Ueda, T. Interleukin-6 Sensitizes TNF-α and TRAIL/Apo2L Dependent Cell Death through Upregulation of Death Receptors in Human Cancer Cells. Biochim. Biophys. Acta (BBA)-Mol. Cell Res. 2021, 1868, 119037. [Google Scholar] [CrossRef]
- Regis, G.; Icardi, L.; Conti, L.; Chiarle, R.; Piva, R.; Giovarelli, M.; Poli, V.; Novelli, F. IL-6, but Not IFN-γ, Triggers Apoptosis and Inhibits in Vivo Growth of Human Malignant T Cells on STAT3 Silencing. Leukemia 2009, 23, 2102–2108. [Google Scholar] [CrossRef]
- Yang, M.; Han, Y.-M.; Han, Q.; Rong, X.-Z.; Liu, X.-F.; Ln, X.-Y. KCTD11 Inhibits Progression of Lung Cancer by Binding to β-Catenin to Regulate the Activity of the Wnt and Hippo Pathways. J. Cell. Mol. Med. 2021, 25, 9411–9426. [Google Scholar] [CrossRef] [PubMed]
- Mochizuki, S.; Okada, Y. ADAMs in Cancer Cell Proliferation and Progression. Cancer Sci. 2007, 98, 621–628. [Google Scholar] [CrossRef]
- Wang, Y. MiR-145 Changes Sensitivity of Non-Small Cell Lung Cancer to Gefitinib through Targeting ADAM19. Eur. Rev. Med. Pharmacol. Sci. 2019, 23, 5831–5839. [Google Scholar] [CrossRef]
- Augoff, K.; Hryniewicz-Jankowska, A.; Tabola, R.; Stach, K. MMP9: A Tough Target for Targeted Therapy for Cancer. Cancers 2022, 14, 1847. [Google Scholar] [CrossRef]
- Mondal, S.; Adhikari, N.; Banerjee, S.; Amin, S.A.; Jha, T. Matrix Metalloproteinase-9 (MMP-9) and Its Inhibitors in Cancer: A Minireview. Eur. J. Med. Chem. 2020, 194, 112260. [Google Scholar] [CrossRef]
- Yu, Y.; Wang, W.; Lu, W.; Chen, W.; Shang, A. Inhibin β-A (INHBA) Induces Epithelial-Mesenchymal Transition and Accelerates the Motility of Breast Cancer Cells by Activating the TGF-β Signaling Pathway. Bioengineered 2021, 12, 4681–4696. [Google Scholar] [CrossRef]
- Zhang, Y.; Yan, S.; Li, Y.; Zhang, J.; Luo, Y.; Li, P.; Yang, Y.; Li, Y.; Huang, Y.; Wang, E. Inhibin βA Is an Independent Prognostic Factor That Promotes Invasion via Hippo Signaling in Non-Small Cell Lung Cancer. Mol. Med. Rep. 2021, 24, 789. [Google Scholar] [CrossRef]
- Bao, J.; Närhi, K.; Teodòsio, A.; Hemmes, A.; Linnavirta, N.M.; Mäyränpää, M.I.; Salmenkivi, K.; Le Quesne, J.; Verschuren, E.W. SOX9 Has Distinct Roles in the Formation and Progression of Different Non-Small Cell Lung Cancer Histotypes. J. Pathol. 2021, 255, 16–29. [Google Scholar] [CrossRef]
- Huang, J.-Q.; Wei, F.-K.; Xu, X.-L.; Ye, S.-X.; Song, J.-W.; Ding, P.-K.; Zhu, J.; Li, H.-F.; Luo, X.-P.; Gong, H.; et al. SOX9 Drives the Epithelial-Mesenchymal Transition in Non-Small-Cell Lung Cancer through the Wnt/β-Catenin Pathway. J. Transl. Med. 2019, 17, 143. [Google Scholar] [CrossRef]
- Piechaczyk, M.; Farràs, R. Regulation and Function of JunB in Cell Proliferation. Biochem. Soc. Trans. 2008, 36, 864–867. [Google Scholar] [CrossRef]
- Pérez-Benavente, B.; Fathinajafabadi, A.; de la Fuente, L.; Gandía, C.; Martínez-Férriz, A.; Pardo-Sánchez, J.M.; Milián, L.; Conesa, A.; Romero, O.A.; Carretero, J.; et al. New Roles for AP-1/JUNB in Cell Cycle Control and Tumorigenic Cell Invasion via Regulation of Cyclin E1 and TGF-Β2. Genome Biol. 2022, 23, 252. [Google Scholar] [CrossRef]
- Ren, F.; Cai, X.; Yao, Y.; Fang, G. JunB: A Paradigm for Jun Family in Immune Response and Cancer. Front. Cell. Infect. Microbiol. 2023, 13, 1222265. [Google Scholar] [CrossRef]
- Jin, L.; Quan, J.; Pan, X.; He, T.; Hu, J.; Li, Y.; Gui, Y.; Yang, S.; Mao, X.; Chen, Y.; et al. Identification of lncRNA EGOT as a Tumor Suppressor in Renal Cell Carcinoma. Mol. Med. Rep. 2017, 16, 7072–7079. [Google Scholar] [CrossRef]
- Qiu, S.; Chen, G.; Peng, J.; Liu, J.; Chen, J.; Wang, J.; Li, L.; Yang, K. LncRNA EGOT Decreases Breast Cancer Cell Viability and Migration via Inactivation of the Hedgehog Pathway. FEBS Open Bio 2020, 10, 817–826. [Google Scholar] [CrossRef] [PubMed]
- Hu, J.; Zhang, P.-J.; Zhang, D.; Chen, Z.-H.; Cao, X.-C.; Yu, Y.; Ge, J. An Autophagy-Associated lncRNAs Model for Predicting the Survival in Non-Small Cell Lung Cancer Patients. Front. Genet. 2022, 13, 919857. [Google Scholar] [CrossRef] [PubMed]
- Cha, J.-H.; Chan, L.-C.; Li, C.-W.; Hsu, J.L.; Hung, M.-C. Mechanisms Controlling PD-L1 Expression in Cancer. Mol. Cell 2019, 76, 359–370. [Google Scholar] [CrossRef] [PubMed]
- Fan, P.; Zhang, N.; Candi, E.; Agostini, M.; Piacentini, M.; Shi, Y.; Huang, Y.; Melino, G. Alleviating Hypoxia to Improve Cancer Immunotherapy. Oncogene 2023, 42, 3591–3604. [Google Scholar] [CrossRef]
- Abu Hejleh, T.; Furqan, M.; Ballas, Z.; Clamon, G. The Clinical Significance of Soluble PD-1 and PD-L1 in Lung Cancer. Crit. Rev. Oncol./Hematol. 2019, 143, 148–152. [Google Scholar] [CrossRef]
- Ghiringhelli, F.; Bibeau, F.; Greillier, L.; Fumet, J.-D.; Ilie, A.; Monville, F.; Laugé, C.; Catteau, A.; Boquet, I.; Majdi, A.; et al. Immunoscore Immune Checkpoint Using Spatial Quantitative Analysis of CD8 and PD-L1 Markers Is Predictive of the Efficacy of Anti-PD1/PD-L1 Immunotherapy in Non-Small Cell Lung Cancer. eBioMedicine 2023, 92, 104633. [Google Scholar] [CrossRef]
- Zhang, Z.; Liu, X.; Chen, D.; Yu, J. Radiotherapy Combined with Immunotherapy: The Dawn of Cancer Treatment. Sig. Transduct. Target. Ther. 2022, 7, 258. [Google Scholar] [CrossRef] [PubMed]
- Shang, S.; Liu, J.; Verma, V.; Wu, M.; Welsh, J.; Yu, J.; Chen, D. Combined Treatment of Non-Small Cell Lung Cancer Using Radiotherapy and Immunotherapy: Challenges and Updates. Cancer Commun. 2021, 41, 1086–1099. [Google Scholar] [CrossRef]
- Qu, F.; Zhou, Y.; Wu, S. Progress of Immune Checkpoint Inhibitors Therapy for Non-Small Cell Lung Cancer with Liver Metastases. Br. J. Cancer 2024, 130, 165–175. [Google Scholar] [CrossRef]
- Wu, X.; Chen, Y.; Guo, W.; Li, T.; Dong, H.; Wang, W.; Xie, M.; Ma, G.; Pei, D. HMGB1 Regulates SNAI1 during NSCLC Metastasis, Both Directly, through Transcriptional Activation, and Indirectly, in a RSF1-IT2-dependent Manner. Mol. Oncol. 2020, 14, 1348. [Google Scholar] [CrossRef]
- Sun, Y.; Ma, L. A Milestone in Epithelial-Mesenchymal Transition. Nat. Cell Biol. 2024, 26, 29–30. [Google Scholar] [CrossRef]
- da Silva-Junior, I.A.; Dalmaso, B.; Herbster, S.; Lepique, A.P.; Jancar, S. Platelet-Activating Factor Receptor Ligands Protect Tumor Cells from Radiation-Induced Cell Death. Front. Oncol. 2018, 8, 10. [Google Scholar] [CrossRef]
- Tian, T.; Leng, Y.; Tang, B.; Dong, X.; Ren, Q.; Liang, J.; Liu, T.; Liu, Y.; Feng, W.; Liu, S.; et al. The Oncogenic Role and Regulatory Mechanism of PGK1 in Human Non-Small Cell Lung Cancer. Biol. Direct 2024, 19, 1. [Google Scholar] [CrossRef]
- Zhao, Q.; Sun, Z.; Pan, Y.; Jing, Q.; Li, W.; Wang, C. Role of ALOX5 in Non-Small Cell Lung Cancer: A Potential Therapeutic Target Associated with Immune Cell Infiltration. Zhong Nan Da Xue Xue Bao Yi Xue Ban 2023, 48, 311–322. [Google Scholar] [CrossRef]
- Pan, R.; Zhang, X.; Chen, Z.; Liu, Y. Arachidonate Lipoxygenases 5 Is a Novel Prognostic Biomarker and Correlates with High Tumor Immune Infiltration in Low-Grade Glioma. Front. Genet. 2023, 14, 1027690. [Google Scholar] [CrossRef]
- Nisar, H.; Labonté, F.M.; Roggan, M.D.; Schmitz, C.; Chevalier, F.; Konda, B.; Diegeler, S.; Baumstark-Khan, C.; Hellweg, C.E. Hypoxia Modulates Radiosensitivity and Response to Different Radiation Qualities in A549 Non-Small Cell Lung Cancer (NSCLC) Cells. Int. J. Mol. Sci. 2024, 25, 1010. [Google Scholar] [CrossRef]
- D’Ignazio, L.; Rocha, S. Hypoxia Induced NF-κB. Cells 2016, 5, 10. [Google Scholar] [CrossRef]
- Tong, J.; Hu, C.; Wu, Y.; Liu, Q.; Sun, D. Radiation-Induced NF-κB Activation Is Involved in Cochlear Damage in Mice via Promotion of a Local Inflammatory Response. J. Radiat. Res. 2023, 64, 63–72. [Google Scholar] [CrossRef]
- Wang, J.; Zhang, W.; Xiong, W.; Lu, W.; Zheng, H.; Zhou, X.; Yuan, J. PM2.5 Stimulated the Release of Cytokines from BEAS-2B Cells through Activation of IKK/NF-κB Pathway. Hum. Exp. Toxicol. 2019, 38, 311–320. [Google Scholar] [CrossRef]
- Favaro, F.; Luciano-Mateo, F.; Moreno-Caceres, J.; Hernández-Madrigal, M.; Both, D.; Montironi, C.; Püschel, F.; Nadal, E.; Eldering, E.; Muñoz-Pinedo, C. TRAIL Receptors Promote Constitutive and Inducible IL-8 Secretion in Non-Small Cell Lung Carcinoma. Cell Death Dis. 2022, 13, 1046. [Google Scholar] [CrossRef]
- Yang, J.-L.; Lin, W.-L.; Tai, S.-B.; Ciou, Y.-S.; Chung, C.-L.; Chen, J.-J.; Liu, P.-F.; Lin, M.-W.; Chen, C.-L. Suppression of TGFβ-Induced Interleukin-6 Secretion by Sinulariolide from Soft Corals through Attenuation of the P38-NF-kB Pathway in Carcinoma Cells. Int. J. Mol. Sci. 2023, 24, 11656. [Google Scholar] [CrossRef] [PubMed]
- Angeles, A.K.; Janke, F.; Daum, A.-K.; Reck, M.; Schneider, M.A.; Thomas, M.; Christopoulos, P.; Sültmann, H. Integrated Circulating Tumour DNA and Cytokine Analysis for Therapy Monitoring of ALK-Rearranged Lung Adenocarcinoma. Br. J. Cancer 2023, 129, 112–121. [Google Scholar] [CrossRef]
- Dib, B.; Lin, H.; Maidana, D.E.; Tian, B.; Miller, J.B.; Bouzika, P.; Miller, J.W.; Vavvas, D.G. Mitochondrial DNA Has a Pro-Inflammatory Role in AMD. Biochim. Biophys. Acta (BBA)-Mol. Cell Res. 2015, 1853, 2897–2906. [Google Scholar] [CrossRef]
- Sakai, C.; Ueda, K.; Goda, K.; Fujita, R.; Maeda, J.; Nakayama, S.; Sotomaru, Y.; Tashiro, S.; Yoshizumi, M.; Ishida, T.; et al. A Possible Role for Proinflammatory Activation via cGAS-STING Pathway in Atherosclerosis Induced by Accumulation of DNA Double-Strand Breaks. Sci. Rep. 2023, 13, 16470. [Google Scholar] [CrossRef]
- Hong, C.; Schubert, M.; Tijhuis, A.E.; Requesens, M.; Roorda, M.; van den Brink, A.; Ruiz, L.A.; Bakker, P.L.; van der Sluis, T.; Pieters, W.; et al. cGAS-STING Drives the IL-6-Dependent Survival of Chromosomally Instable Cancers. Nature 2022, 607, 366–373. [Google Scholar] [CrossRef]
- Al-Asmari, S.S.; Rajapakse, A.; Ullah, T.R.; Pépin, G.; Croft, L.V.; Gantier, M.P. Pharmacological Targeting of STING-Dependent IL-6 Production in Cancer Cells. Front. Cell Dev. Biol. 2022, 9, 709618. [Google Scholar] [CrossRef]
- Yum, S.; Li, M.; Fang, Y.; Chen, Z.J. TBK1 Recruitment to STING Activates Both IRF3 and NF-κB That Mediate Immune Defense against Tumors and Viral Infections. Proc. Natl. Acad. Sci. USA 2021, 118, e2100225118. [Google Scholar] [CrossRef]
- Zhang, L.; Wei, X.; Wang, Z.; Liu, P.; Hou, Y.; Xu, Y.; Su, H.; Koci, M.D.; Yin, H.; Zhang, C. NF-κB Activation Enhances STING Signaling by Altering Microtubule-Mediated STING Trafficking. Cell Rep. 2023, 42, 112185. [Google Scholar] [CrossRef]
- Abe, T.; Barber, G.N. Cytosolic-DNA-Mediated, STING-Dependent Proinflammatory Gene Induction Necessitates Canonical NF-κB Activation through TBK1. J. Virol. 2014, 88, 5328–5341. [Google Scholar] [CrossRef]
- De Simone, V.; Franzè, E.; Ronchetti, G.; Colantoni, A.; Fantini, M.C.; Di Fusco, D.; Sica, G.S.; Sileri, P.; MacDonald, T.T.; Pallone, F.; et al. Th17-Type Cytokines, IL-6 and TNF-α Synergistically Activate STAT3 and NF-kB to Promote Colorectal Cancer Cell Growth. Oncogene 2015, 34, 3493–3503. [Google Scholar] [CrossRef] [PubMed]
- Taylor, C.T.; Colgan, S.P. Regulation of Immunity and Inflammation by Hypoxia in Immunological Niches. Nat. Rev. Immunol. 2017, 17, 774–785. [Google Scholar] [CrossRef] [PubMed]
- Pham, K.; Parikh, K.; Heinrich, E.C. Hypoxia and Inflammation: Insights From High-Altitude Physiology. Front. Physiol. 2021, 12, 676782. [Google Scholar] [CrossRef]
- Chen, Y.; Gaber, T. Hypoxia/HIF Modulates Immune Responses. Biomedicines 2021, 9, 260. [Google Scholar] [CrossRef] [PubMed]
- Hekim, N.; Cetin, Z.; Nikitaki, Z.; Cort, A.; Saygili, E.I. Radiation Triggering Immune Response and Inflammation. Cancer Lett. 2015, 368, 156–163. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, D.; Coates, P.J.; Lorimore, S.A.; Wright, E.G. Responses to Ionizing Radiation Mediated by Inflammatory Mechanisms. J. Pathol. 2014, 232, 289–299. [Google Scholar] [CrossRef] [PubMed]
- Krysztofiak, A.; Szymonowicz, K.; Hlouschek, J.; Xiang, K.; Waterkamp, C.; Larafa, S.; Goetting, I.; Vega-Rubin-de-Celis, S.; Theiss, C.; Matschke, V.; et al. Metabolism of Cancer Cells Commonly Responds to Irradiation by a Transient Early Mitochondrial Shutdown. iScience 2021, 24, 103366. [Google Scholar] [CrossRef] [PubMed]
- Love, M.I.; Huber, W.; Anders, S. Moderated Estimation of Fold Change and Dispersion for RNA-Seq Data with DESeq2. Genome Biol. 2014, 15, 550. [Google Scholar] [CrossRef]
- Subramanian, A.; Tamayo, P.; Mootha, V.K.; Mukherjee, S.; Ebert, B.L.; Gillette, M.A.; Paulovich, A.; Pomeroy, S.L.; Golub, T.R.; Lander, E.S.; et al. Gene Set Enrichment Analysis: A Knowledge-Based Approach for Interpreting Genome-Wide Expression Profiles. Proc. Natl. Acad. Sci. USA 2005, 102, 15545–15550. [Google Scholar] [CrossRef]
- Rouillard, A.D.; Gundersen, G.W.; Fernandez, N.F.; Wang, Z.; Monteiro, C.D.; McDermott, M.G.; Ma’ayan, A. The Harmonizome: A Collection of Processed Datasets Gathered to Serve and Mine Knowledge about Genes and Proteins. Database 2016, 2016, baw100. [Google Scholar] [CrossRef]

| Regulated DEGs (Gene Name Abbreviation) | Gene Expression Based on p-Adjusted log2 Fold Changes | ||||||
|---|---|---|---|---|---|---|---|
| Irradiated vs. Unirradiated | Hypoxic vs. Normoxic | ||||||
| X-rays | 12C Ions | Controls (H0 vs. N0) | X-rays (H8 vs. N8) | 12C Ions (H8 vs. N8) | |||
| Normoxia (N8 a vs. N0) | Hypoxia (H8 vs. H0) | Normoxia (N8 vs. N0) | Hypoxia (H8 vs. H0) | ||||
| FAS | 2.06 | 2.75 | 2.31 | 2.85 | −0.71 | −0.02 | −0.17 |
| NTN1 | 1.36 | 1.93 | 2.19 | 2.29 | −0.03 | 0.54 | 0.07 |
| CDKN1A | 2.63 | 2.62 | 2.61 | 2.38 | −0.19 | −0.21 | −0.42 |
| KITLG | 1.01 | 1.38 | 1.36 | 1.46 | −1.10 | −0.73 | −1.00 |
| PLK3 | 1.28 | 1.14 | 1.13 | 1.16 | −0.26 | −0.40 | −0.23 |
| IL1A | 1.37 | 1.54 | 0.43 | 1.26 | 2.63 | 2.81 | 3.46 |
| EBI3 | 0.60 | 0.81 | 1.04 | 1.45 | −0.29 | −0.08 | 0.12 |
| TFEC | 2.49 | N/A b | 2.95 | 2.87 | N/A | N/A | N/A |
| REV3L | 0.78 | 1.08 | 0.79 | 0.89 | −0.43 | −0.13 | −0.33 |
| PTAFR | 0.83 | 1.15 | 0.72 | 0.97 | −0.55 | −0.23 | −0.29 |
| GAD1 | 0.70 | 0.76 | 0.69 | 1.32 | −0.63 | −0.57 | 0.00 |
| BAX | 0.58 | 0.83 | 0.74 | 1.01 | −0.27 | −0.03 | 0.00 |
| MX1 | 0.22 | 0.18 | 0.12 | 1.16 | −0.31 | −0.35 | 0.73 |
| ALOX5 | 1.16 | 1.35 | 1.43 | 2.49 | 0.48 | 0.68 | 1.54 |
| GADD45A | 1.11 | 0.95 | 0.77 | 0.55 | 0.18 | 0.02 | −0.04 |
| CCR7 | 0.76 | 0.49 | 1.02 | 0.62 | 0.00 | −0.27 | −0.40 |
| EMX1 | 1.33 | 0.48 | 3.10 | 2.55 | −0.71 | −1.56 | N/A |
| KCTD11 | 0.26 | −0.02 | 0.16 | 0.34 | 1.96 | 1.67 | 2.14 |
| ADAM19 | −0.19 | −0.04 | −0.15 | −0.02 | 1.46 | 1.60 | 1.58 |
| INHBA | 0.15 | 0.28 | 0.05 | 0.19 | 1.41 | 1.55 | 1.55 |
| MMP9 | 0.26 | 0.81 | −0.45 | −0.75 | 1.87 | 2.43 | 1.58 |
| PDGFB | −0.17 | 0.15 | −0.21 | −0.08 | 1.63 | 1.95 | 1.75 |
| F3 | −0.05 | −0.02 | −0.62 | 0.03 | 2.00 | 2.02 | 2.65 |
| CXCL8 | −0.15 | 0.03 | 0.01 | 0.10 | 1.31 | 1.49 | 1.41 |
| SERPINE1 | 0.37 | 0.57 | 0.09 | 0.36 | 1.64 | 1.84 | 1.91 |
| IL6 | 0.37 | 0.32 | 0.65 | 0.70 | 1.23 | 1.18 | 1.28 |
| CARD11 | 0.00 | 0.14 | −0.17 | 0.20 | 1.05 | 1.18 | 1.42 |
| BCL2A1 | 0.54 | 0.58 | 0.19 | −0.17 | 1.54 | 1.59 | 1.18 |
| JUNB | −0.18 | −0.03 | −0.10 | 0.09 | 1.21 | 1.36 | 1.40 |
| EGOT | 0.73 | 1.03 | −1.68 | 0.65 | 1.94 | 2.24 | N/A |
| IL1B | 0.56 | 1.33 | 1.94 | 0.19 | 1.80 | 2.58 | 0.05 |
| SOX9 | −0.57 | −0.11 | 0.12 | −0.11 | 1.01 | 1.47 | 0.79 |
| CCL28 | 0.83 | −0.19 | −0.34 | 0.26 | 1.49 | 0.47 | 2.09 |
| CD274 | −0.26 | −0.06 | −0.26 | −0.11 | 0.91 | 1.11 | 1.05 |
| TRAF1 | 0.16 | 0.51 | 0.19 | −0.21 | 0.98 | 1.32 | 0.58 |
| SNAI1 | −0.68 | 0.25 | −0.52 | −0.21 | 0.32 | 1.24 | 0.63 |
| CCL20 | −1.45 | −0.02 | 0.63 | −0.10 | 0.62 | 2.05 | −0.11 |
| LIPG | −0.28 | 0.66 | −0.09 | 0.21 | 0.53 | 1.47 | 0.83 |
| ENO2 | 0.06 | −0.20 | −0.29 | −0.02 | 0.82 | 0.56 | 1.08 |
| CCL2 | 0.16 | 0.27 | −0.02 | 0.25 | 0.89 | 1.00 | 1.17 |
| PGK1 | 0.24 | 0.03 | −0.49 | 0.08 | 0.92 | 0.70 | 1.49 |
| AGT | 0.39 | −0.38 | −1.30 | 1.56 | 0.02 | −0.75 | 2.88 |
| ICAM1 | 0.19 | 0.45 | −0.23 | 0.33 | 0.52 | 0.78 | 1.09 |
| GADD45B | 0.07 | 0.01 | −0.03 | 0.16 | 0.99 | 0.93 | 1.19 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nisar, H.; Sanchidrián González, P.M.; Labonté, F.M.; Schmitz, C.; Roggan, M.D.; Kronenberg, J.; Konda, B.; Chevalier, F.; Hellweg, C.E. NF-κB in the Radiation Response of A549 Non-Small Cell Lung Cancer Cells to X-rays and Carbon Ions under Hypoxia. Int. J. Mol. Sci. 2024, 25, 4495. https://doi.org/10.3390/ijms25084495
Nisar H, Sanchidrián González PM, Labonté FM, Schmitz C, Roggan MD, Kronenberg J, Konda B, Chevalier F, Hellweg CE. NF-κB in the Radiation Response of A549 Non-Small Cell Lung Cancer Cells to X-rays and Carbon Ions under Hypoxia. International Journal of Molecular Sciences. 2024; 25(8):4495. https://doi.org/10.3390/ijms25084495
Chicago/Turabian StyleNisar, Hasan, Paulina Mercedes Sanchidrián González, Frederik M. Labonté, Claudia Schmitz, Marie Denise Roggan, Jessica Kronenberg, Bikash Konda, François Chevalier, and Christine E. Hellweg. 2024. "NF-κB in the Radiation Response of A549 Non-Small Cell Lung Cancer Cells to X-rays and Carbon Ions under Hypoxia" International Journal of Molecular Sciences 25, no. 8: 4495. https://doi.org/10.3390/ijms25084495
APA StyleNisar, H., Sanchidrián González, P. M., Labonté, F. M., Schmitz, C., Roggan, M. D., Kronenberg, J., Konda, B., Chevalier, F., & Hellweg, C. E. (2024). NF-κB in the Radiation Response of A549 Non-Small Cell Lung Cancer Cells to X-rays and Carbon Ions under Hypoxia. International Journal of Molecular Sciences, 25(8), 4495. https://doi.org/10.3390/ijms25084495








